Crystallization of Calcium Carbonate in Alginate and Xanthan Hydrogels
Abstract
:1. Introduction
2. Results and Discussion
2.1. CaCO3 Precipitation in Xanthan Gels
2.2. CaCO3 Precipitation in Alginate Gels
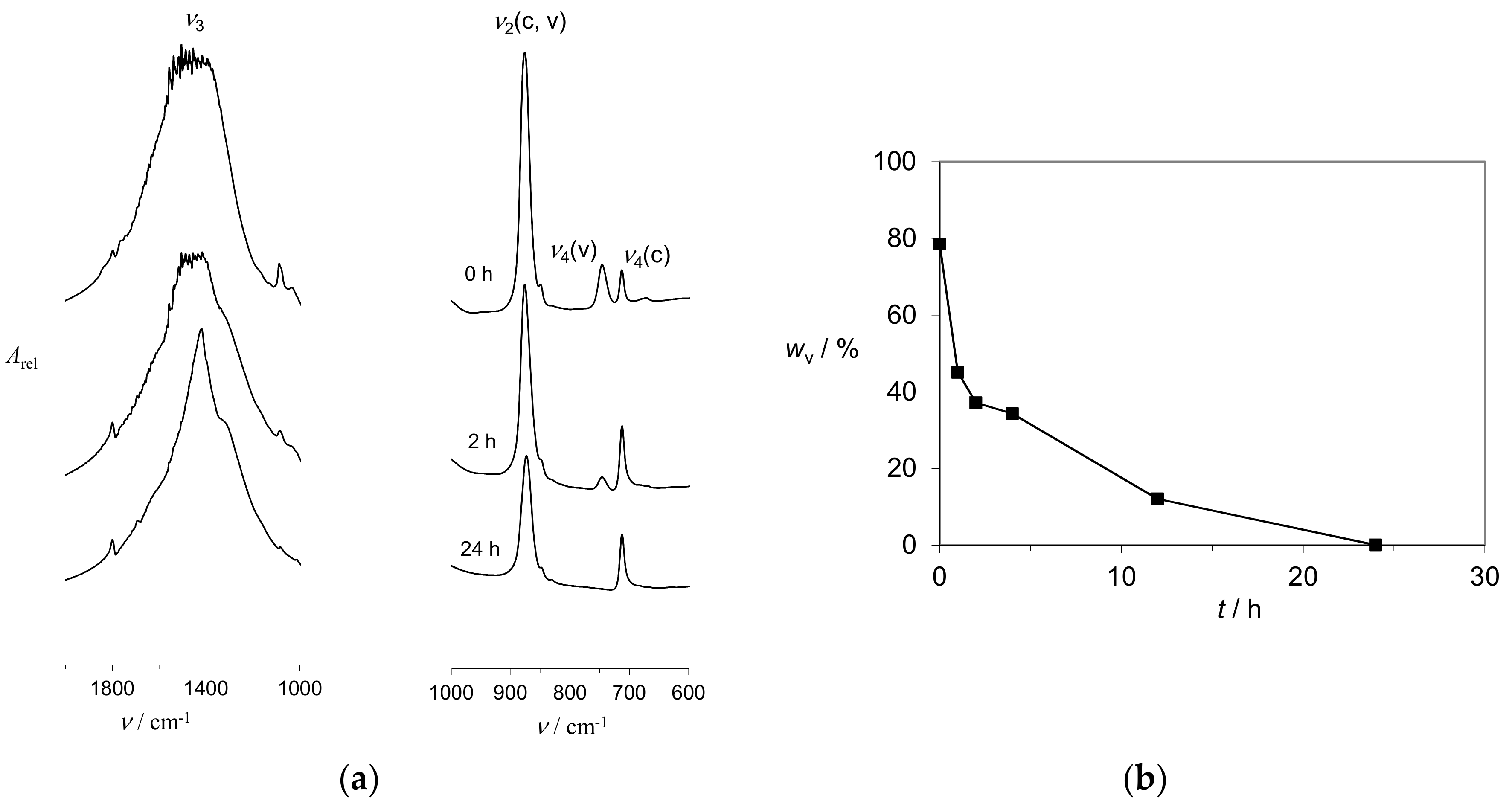
2.3. Kinetics of CaCO3 Phase Transition in the Alginate Gels
3. Materials and Methods
3.1. CO3-Gel, pH = 10.5
3.2. C-Gel, pH = 9.0
3.3. Ca-Gel, pH = 10.5
3.4. Model System
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Brecevic, L.; Kralj, D. On calcium carbonates: From fundamental research to application. Croat. Chem. Acta 2007, 80, 467–484. [Google Scholar]
- Sommerdijk, N.A.J.M.; de With, G. Biomimetic CaCO3 mineralization using designer molecules and interfaces. Chem. Rev. 2008, 108, 4499–4550. [Google Scholar] [CrossRef] [PubMed]
- Gebauer, D.; Völkel, A.; Cölfen, H. Stable prenucleation calcium carbonate clusters. Science 2008, 322, 1819–1822. [Google Scholar] [CrossRef] [PubMed]
- Meldrum, F.C.; Cölfen, H. Controlling mineral morphologies and structures in biological and synthetic systems. Chem. Rev. 2008, 108, 4332–4432. [Google Scholar] [CrossRef] [PubMed]
- Demichelis, R.; Raiteri, P.; Gale, J.D. Structure of hydrated calcium carbonates: A first-principles study. J. Cryst. Growth 2014, 401, 33–37. [Google Scholar] [CrossRef]
- Carlson, W.D. The polymorphs of CaCO3 and the aragonite-calcite transformation. Rev. Mineral. Geochem. 1983, 11, 191–225. [Google Scholar]
- Asenath-Smith, E.; Li, H.; Keene, E.C.; Seh, Z.V.; Estroff, L.A. Crystal growth of calcium carbonate in hydrogels as a model of biomineralization. Adv. Funct. Mater. 2012, 22, 2891–2914. [Google Scholar] [CrossRef]
- Flory, P.J. Introductory lecture. Faraday Discuss. Chem. Soc. 1974, 57, 7–18. [Google Scholar] [CrossRef]
- Estroff, L.A.; Hamilton, A.D. Water gelation by small organic molecules. Chem. Rev. 2004, 104, 1201–1218. [Google Scholar] [CrossRef] [PubMed]
- Nindiyasari, F.; Fernández-Díaz, L.; Griesshaber, E.; Astilleros, J.M.; Sánchez-Pastor, N.; Schmahl, W.W. Influence of gelatin hydrogel porosity on the crystallization of CaCO3. Cryst. Growth Des. 2014, 14, 1531–1542. [Google Scholar] [CrossRef]
- Max, A.; Lauffer, M.A. Theory of diffusion in gels. Biophys. J. 1961, 1, 205–213. [Google Scholar]
- Kosanovic, C.; Falini, G.; Kralj, D. Mineralization of calcium carbonates in gelling media. Cryst. Growth Des. 2011, 11, 269–277. [Google Scholar] [CrossRef]
- Xie, M.; Olderoy, M.O.; Andreassen, J.P.; Selbach, S.M.; Strand, B.L.; Sikorski, P. Alginate-controlled formation of nanoscale calcium carbonate and hydroxyapatite mineral phase within hydrogel networks. Acta Biomater. 2010, 6, 3665–3675. [Google Scholar] [CrossRef] [PubMed]
- Munro, N.H.; McGrath, K.M. Biomimetic approach to forming chitin/aragonite composites. Chem. Commun. (Camb.) 2012, 48, 4716–4718. [Google Scholar] [CrossRef] [PubMed]
- Volodkin, V.; Petrov, A.I.; Prevot, M.; Sukhorukov, G.B. Matrix polyelectrolyte microcapsules: New system for macromolecule encapsulation. Langmuir 2004, 20, 3398–3406. [Google Scholar] [CrossRef] [PubMed]
- Trushina, D.B.; Bukreeva, T.V.; Kovalchuk, M.V.; Antipina, M.N. CaCO3 vaterite microparticles for biomedical and personal care applications. Mater. Sci. Eng. C 2014, 45, 644–658. [Google Scholar] [CrossRef] [PubMed]
- Volodkin, D.; Klitzing, R.; Möhwald, H. Pure protein microspheres by calcium carbonate templating. Angew. Chem. Int. Ed. 2010, 122, 9444–9447. [Google Scholar] [CrossRef]
- Lengert, E.; Saveleva, M.; Abalymov, A.; Atkin, V.; Wuytens, P.C.; Kamyshinsky, R.; Vasiliev, A.L.; Gorin, D.A.; Sukhorukov, G.B.; Skirtach, A.G.; et al. Silver alginate hydrogel micro- and nanocontainers for theranostics: Synthesis, encapsulation, remote release, and detection. ACS Appl. Mater. Interfaces 2017, 9, 21949–21958. [Google Scholar] [CrossRef] [PubMed]
- Parakhonskiy, B.V.; Yashchenok, A.M.; Konrad, M.; Skirtach, A.G. Colloidal micro- and nano-particles as templates for polyelectrolyte multilayer capsules. Adv. Colloid Interface Sci. 2014, 207, 253–264. [Google Scholar] [CrossRef] [PubMed]
- Yashchenok, A.; Parakhonskiy, B.; Donatan, S.; Kohler, D.; Skirtachad, A.; Mohwald, H. Polyelectrolyte multilayer microcapsules templated on spherical, elliptical and square calcium carbonate particles. J. Mater. Chem. B 2013, 1, 1223–1228. [Google Scholar] [CrossRef]
- Yi, Q.; Sukhorukov, G.B. Externally triggered dual function of complex microcapsules. ACS Nano 2013, 7, 8693–8705. [Google Scholar] [CrossRef] [PubMed]
- Donatan, S.; Yashchenok, A.; Khan, N.; Parakhonskiy, B.; Cocquyt, M.; Pinchasik, B.; Khalenkow, D.; Möhwald, H.; Konrad, M.; Skirtach, M. Loading capacity versus enzyme activity in anisotropic and spherical calcium carbonate microparticles. ACS Appl. Mater. Interfaces 2016, 8, 14284–14292. [Google Scholar] [CrossRef] [PubMed]
- Svenskaya, Y.I.; Fattah, H.; Zakharevich, A.M.; Gorin, D.A.; Sukhorukov, G.B.; Parakhonskiy, B.V. Ultrasonically assisted fabrication of vaterite submicron-sized carriers. Adv. Powder Technol. 2016, 27, 618–624. [Google Scholar] [CrossRef]
- Douglas, T.E.L. Biomimetic Mineralization of Hydrogels in Biomineralization and Biomaterials, Fundamentals and Applications; Aparicio, C., Ginebra, M., Eds.; Woodhead Publishing: Cambridge, UK; 2015; pp. 291–313. [Google Scholar]
- Gkioni, K.; Leeuwenburgh, S.C.; Douglas, T.E.; Mikos, A.G.; Jansen, J.A. Mineralization of hydrogels for bone regeneration. Tissue Eng. B 2010, 16, 577–585. [Google Scholar] [CrossRef] [PubMed]
- Douglas, T.E.L.; Sobczyk, K.; Łapa, A.; Włodarczyk, K.; Brackman, G.; Vidiasheva, I.; Reczyńska, K.; Pietryga, K.; Schaubroeck, D.; Bliznuk, V.; et al. Ca:Mg:Zn:CO3 and Ca:Mg:CO3—tri- and bi-elemental carbonate microparticles for novel injectable self-gelling hydrogel–microparticle composites for tissue regeneration. Biomed. Mater. 2017, 12, 025015. [Google Scholar] [CrossRef] [PubMed]
- Jang, J.; Seol, Y.J.; Kim, H.J.; Kundu, J.; Kim, S.W.; Cho, D.W. Effects of alginate hydrogel cross-linking density on mechanical and biological behaviors for tissue engineering. J. Mech. Behav. Biomed. Mater. 2014, 37, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Kuo, C.K.; Ma, P.X. Maintaining dimensions and mechanical properties of ionically crosslinked alginate hydrogel scaffolds in vitro. J. Biomed. Mater. Res. A 2008, 84, 899–907. [Google Scholar] [CrossRef] [PubMed]
- Corni, I.; Harvey, T.J.; Wharton, J.A.; Stokes, K.R.; Walsh, F.C.; Wood, R.J.K. A review of experimental techniques to produce a nacre-like structure. Bioinspir. Biomim. 2012, 7, 031001. [Google Scholar] [CrossRef] [PubMed]
- Bidarra, S.J.; Barrias, C.C.; Fonseca, K.B.; Barbosa, M.A.; Soares, R.A.; Granja, P.L. Injectable in situ crosslinkable RGD-modified alginate matrix for endothelial cells delivery. Biomaterials 2011, 32, 7897–7904. [Google Scholar] [CrossRef] [PubMed]
- Ogomi, D.; Serizawa, T.; Akashi, M. Bioinspired organic–inorganic composite materialsprepared by an alternate soaking process as a tissue reconstitution matrix. J. Biomed. Mater. Res. A 2003, 67, 1360–1366. [Google Scholar] [CrossRef] [PubMed]
- Suzawa, Y.; Funaki, T.; Watanabe, J.; Iwai, S.; Yura, Y.; Nakano, T.; Umakoshi, Y.; Akashi, M. Regenerative behavior of biomineral/agarose composite gels as bone grafting materials in rat cranial defects. J. Biomed. Mater. Res. A 2010, 93, 965–975. [Google Scholar] [CrossRef] [PubMed]
- Munro, N.H.; McGrath, K.M. Biomimetic mineralisation of polymeric scaffolds using a combined soaking approach: Adaptation with various mineral salts. Dalton Trans. 2011, 40, 9269–9275. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Estroff, L.A. Hydrogels coupled with self-assembled monolayers: An in vitro matrix to study calcite biomineralization. J. Am. Chem. Soc. 2007, 129, 5480–5483. [Google Scholar] [CrossRef] [PubMed]
- Helbig, U. Growth of calcium carbonate in polyacrylamide hydrogel: Investigation of the influence of polymer content. J. Cryst. Growth 2008, 310, 2863–2870. [Google Scholar] [CrossRef]
- Sancho-Tomas, M.; Fermani, S.; Goeffredo, S.; Dubinsky, Z.; Garcia-Ruiz, J.M.; Gomez-Morales, J.; Falini, G. Exploring coral biomineralization in gelling environment by means of a counter diffusion system. CrystEngComm 2014, 16, 1257–1267. [Google Scholar] [CrossRef]
- Sancho-Tomas, M.; Fermani, S.; Duran-Olivencia, M.D.; Otalora, F.; Gomez-Morales, J.; Falini, G.; Garcia-Ruiz, J.M. Influence of carged polypeptides on nucleation and growth of CaCO3 evaluated by counterdiffusion experiments. Cryst. Growth Des. 2013, 13, 3884–3891. [Google Scholar] [CrossRef]
- Iijima, M.; Shinozaki, M.; Hatakeyama, T.; Takahashi, M.; Hatakeyama, H. AFM studies on gelation mechanism of xanthan gum hydrogels. Carbohydr. Polym. 2007, 68, 701–707. [Google Scholar] [CrossRef]
- Takahashi, M.; Hatakeyama, T.; Hatakeyama, H. Phenomenological theory describing the behaviour of non-freezing water in structure formation process of polysaccharide aqueous solutions. Carbohydr. Polym. 2000, 41, 91–95. [Google Scholar] [CrossRef]
- Smidsrød, O. Molecular basis for some physical properties of alginates in the gel state. Faraday Discuss. Chem. Soc. 1974, 57, 263–274. [Google Scholar] [CrossRef]
- Huynh, U.T.D.; Lerbret, A.; Neiers, F.; Chambin, O.; Assifaoui, A. Binding of divalent cations to polygalacturonate: A mechanism driven by the hydration water. J. Phys. Chem. B 2016, 120, 1021–1032. [Google Scholar] [CrossRef] [PubMed]
- Leea, K.Y.; Mooneya, D.J. Alginate: Properties and biomedical applications. Prog. Polym. Sci. 2012, 37, 106–126. [Google Scholar] [CrossRef] [PubMed]
- Braissant, O.; Cailleau, G.; Dupraz, C.; Verrecchia, E.P. Bacterially induced mineralization of calcium carbonate in terrestrial environments: The role of exopolysaccarides and amino acids. J. Sediment. Res. 2003, 73, 485–490. [Google Scholar] [CrossRef]
- Kralj, D.; Brecević, L.; Kontrec, J. Vaterite growth and dissolution in aqueous solutions. III. Kinetics of transformation. J. Cryst. Growth 1997, 177, 248–257. [Google Scholar] [CrossRef]
- Yang, X.; Xu, G. The influence of xanthan on the crystallization of calcium carbonate. J. Cryst. Growth 2011, 314, 231–238. [Google Scholar] [CrossRef]
- Li, H.; Estroff, L.A. Calcite growth in hydrogels: Assessing the mechanism of polymer-network incorporation into single crystals. Adv. Mater. 2009, 21, 470–473. [Google Scholar] [CrossRef]
- Butler, M.F.; Glaser, N.; Weaver, A.C.; Kirkland, M.; Heppenstall-Butler, M. Calcium carbonate crystallization in the presence of biopolymers. Cryst. Growth Des. 2006, 6, 781–794. [Google Scholar] [CrossRef]
- Diaz-Dosque, M.; Aranda, P.; Darder, M.; Retuert, J.; Yazdani-Pedrame, M.; Arias, J.L.; Ruiz-Hitzky, E. Use of biopolymers as oriented supports for the stabilization of different polymorphs of biomineralized calcium carbonate with complex shape. J. Cryst. Growth 2008, 310, 5331–5340. [Google Scholar] [CrossRef]
- Flemming, A.A.; Kralj, D. Determination of the composition of calcite-vaterite mixtures by infrared spectrophotometry. Appl. Spectrosc. 1991, 45, 1748–1751. [Google Scholar]
- Fleming, A.A.; Brecevic, L. Infrared spectra of amorphous and crystalline calcium carbonate. Acta Chem. Scand. 1991, 45, 1018–1024. [Google Scholar]
- Kontrec, J.; Ukrainczyk, M.; Babic-Ivancic, V.; Kralj, D. Synthesis of calcium carbonate by semicontinuous carbonation method in the presence of dextrans. Croat. Chem. Acta 2011, 84, 25–32. [Google Scholar] [CrossRef]
- Kralj, D.; Kontrec, J.; Brecevic, L.; Falini, G.; Nothing-Laslo, V. Effect of inorganic anions on the Morphology and structure of magnesium calcite. Chem. Eur. J. 2004, 10, 1647–1656. [Google Scholar] [CrossRef] [PubMed]
- Raz, S.; Hamilton, P.S.; Wilt, F.H.; Weiner, S.; Addadi, L. The transient phase of amorphous calcium carbonate in sea urchin larval spicules: The involvement of proteins and magnesium ions in its formation and stabilization. Adv. Funct. Mater. 2003, 13, 480–486. [Google Scholar] [CrossRef]
- Beniash, E.; Aizenberg, J.; Addadi, L.; Weiner, S. Amorphous calcium carbonate transforms into calcite during sea urchin larval spicule growth. Proc. R. Soc. Lond. B 1997, 264, 461–465. [Google Scholar] [CrossRef]
- International Centre for Diffraction Data. Powder Diffraction File, Inorganic Volume; Table Nos. 88-1087, 86-0174, 83-0578 for Calcilte; 33-0268 for Vaterite; International Centre for Diffraction Data: Swarthmore, PA, USA, 1988. [Google Scholar]
- Plummer, L.N.; Busenberg, E. The solubilities of calcite, aragonite and vaterite in CO2-H2O solutions between 0 and 90 °C, and an evaluation of the aqueous model for the system CaCO3-CO2-H2O. Geochim. Cosmochim. Acta 1982, 46, 1011–1040. [Google Scholar] [CrossRef]
- Kralj, D.; Brecevic, L.; Nielsen, A.E. Vaterite growth and dissolution in aqueous solutions. I. Kinetics of crystal growth. J. Cryst. Growth 1990, 104, 793–800. [Google Scholar] [CrossRef]



| cxan/wt % | wcalc/wt % | Shape | L/μm # | |
|---|---|---|---|---|
| Ca-gel pH = 9.0 | 0.20 | 99 | c-axis elongated rhombohedra | 15(5) |
| 0.35 | 95 | c-axis elongated rhombohedra | 10(5) | |
| 0.40 | 99 | etched rhombohedra | 10(5) | |
| 2.00 | 21 | etched rhombohedra-spheres | 10(5)–10(5) | |
| Ca-gel pH = 10.5 | 0.20 | 100 | Rhombohedra | 10(5) |
| 0.35 | 99 | Rhombohedra | 10(5) | |
| 0.40 | 61 | etched rhombohedra-spheres | 10(5)–10(5) | |
| 2.00 | 19 | etched rhombohedra-spheres | 10(5)–10(5) | |
| CO3-gel pH = 10.5 | 0.20 | 100 | rhombohedra * | 15(5) |
| 0.35 | 100 | rhombohedra * | 10(5) | |
| 0.40 | 100 | rhombohedra * | 10(5) | |
| 2.00 | 54 | Rhombohedra-spheres | 10(5)–10(5) |
| calg/wt % | wcalc/wt % | Shape | L/μm # | |
|---|---|---|---|---|
| Ca-gel pH = 9.0 | 0.20 | 78 | rhombohedra */spheres | 10(3)–5(1) |
| 0.50 | 100 | rhombohedra | 5(2) | |
| 0.80 | 100 | rhombohedra * | 5(2) | |
| 2.00 | 100 | rhombohedra * | 5(2) | |
| Ca-gel pH = 10.5 | 0.20 | 63 | rhombohedra *-spheres | 15(5)–5(2) |
| 0.50 | 76 | rhombohedra *-spheres | 10(5)–5(3) | |
| 0.80 | 61 | rhombohedra *-spheres | 10(5)–5(4) | |
| 2.00 | 77 | rhombohedra *-spheres | 10(5)–5(3) | |
| CO3-gel pH = 10.5 | 0.20 | 100 | rhombohedra * | 15(5) |
| 0.50 | 100 | rhombohedra | 10(3) | |
| 0.80 | 100 | rhombohedra | 10(5) | |
| 2.00 | 100 | rhombohedra | 10(5) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kosanović, C.; Fermani, S.; Falini, G.; Kralj, D. Crystallization of Calcium Carbonate in Alginate and Xanthan Hydrogels. Crystals 2017, 7, 355. https://doi.org/10.3390/cryst7120355
Kosanović C, Fermani S, Falini G, Kralj D. Crystallization of Calcium Carbonate in Alginate and Xanthan Hydrogels. Crystals. 2017; 7(12):355. https://doi.org/10.3390/cryst7120355
Chicago/Turabian StyleKosanović, Cleo, Simona Fermani, Giuseppe Falini, and Damir Kralj. 2017. "Crystallization of Calcium Carbonate in Alginate and Xanthan Hydrogels" Crystals 7, no. 12: 355. https://doi.org/10.3390/cryst7120355
APA StyleKosanović, C., Fermani, S., Falini, G., & Kralj, D. (2017). Crystallization of Calcium Carbonate in Alginate and Xanthan Hydrogels. Crystals, 7(12), 355. https://doi.org/10.3390/cryst7120355








