Abstract
A search of the Cambridge Structural Database (CSD) was carried out for phosphine-water and arsine-water complexes in which water is either the proton donor in hydrogen-bonded complexes, or the electron-pair donor in pnicogen-bonded complexes. The range of experimental P-O distances in the phosphine complexes is consistent with the results of ab initio MP2/aug’-cc-pVTZ calculations carried out on complexes H2XP:OH2, for X = NC, F, Cl, CN, OH, CCH, H, and CH3. Only hydrogen-bonded complexes are found on the H2(CH3)P:HOH and H3P:HOH potential surfaces, while only pnicogen-bonded complexes exist on H2(NC)P:OH2, H2FP:OH2, H2(CN)P:OH2, and H2(OH)P:OH2 surfaces. Both hydrogen-bonded and pnicogen-bonded complexes are found on the H2ClP:OH2 and H2(CCH)P:OH2 surfaces, with the pnicogen-bonded complexes more stable than the corresponding hydrogen-bonded complexes. The more electronegative substituents prefer to form pnicogen-bonded complexes, while the more electropositive substituents form hydrogen-bonded complexes. The H2XP:OH2 complexes are characterized in terms of their structures, binding energies, charge-transfer energies, and spin-spin coupling constants 2hJ(O-P), 1hJ(H-P), and 1J(O-H) across hydrogen bonds, and 1pJ(P-O) across pnicogen bonds.
1. Introduction
Chloroform, dichloromethane, and water have been observed as solvent molecules in X-ray structures of crystals [1,2,3,4,5,6]. Such structures have long been used as a tool for identifying and confirming the presence of weak intermolecular interactions. The most prevalent intermolecular interaction in the Cambridge Structural Database (CSD) is the X-H…Y hydrogen bond, which has been at the forefront of intermolecular interactions since Pimentel’s book “The Hydrogen Bond” [7]. The hydrogen bond is defined as an attractive interaction between a hydrogen atom from a molecule or a molecular fragment X–H in which X is more electronegative than H, and an atom or a group of atoms in the same or a different molecule, in which there is evidence of bond formation [8,9]. Of particular interest are the hydrogen bonds in the X-ray structures of organic hydrates. Hydrogen bonds involving water molecules interacting with different chemical groups have been identified and classified [3,4,5,6].
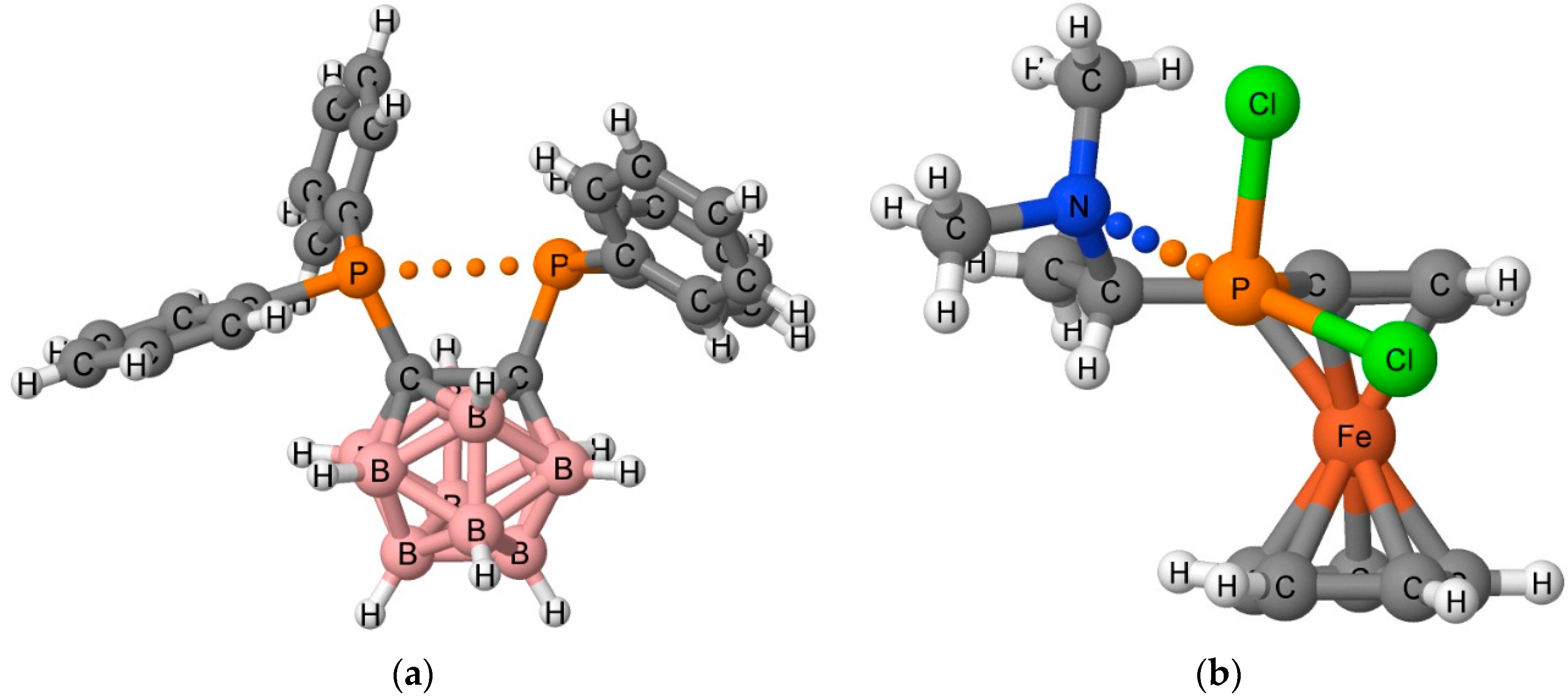
A relatively new intermolecular interaction, the pnicogen bond, was initially detected in the crystal structures of 1,2-dicarba-closo-dodecaboranes [10,11] and aminoalkyl-ferrocenylphosphanes [12]. The structures of two of these complexes are illustrated in Figure 1. A large number of intermolecular and intramolecular pnicogen interactions have also been observed in the solid phase [13,14,15]. Pnicogen bonds were first described theoretically for model complexes [16,17], and subsequent studies confirmed the stabilizing nature of pnicogen interactions [18,19,20]. The pnicogen bond is a Lewis acid-Lewis base interaction in which the Lewis acid is a group 15 element (N, P, As, or Sb) acting as an electron-pair acceptor.
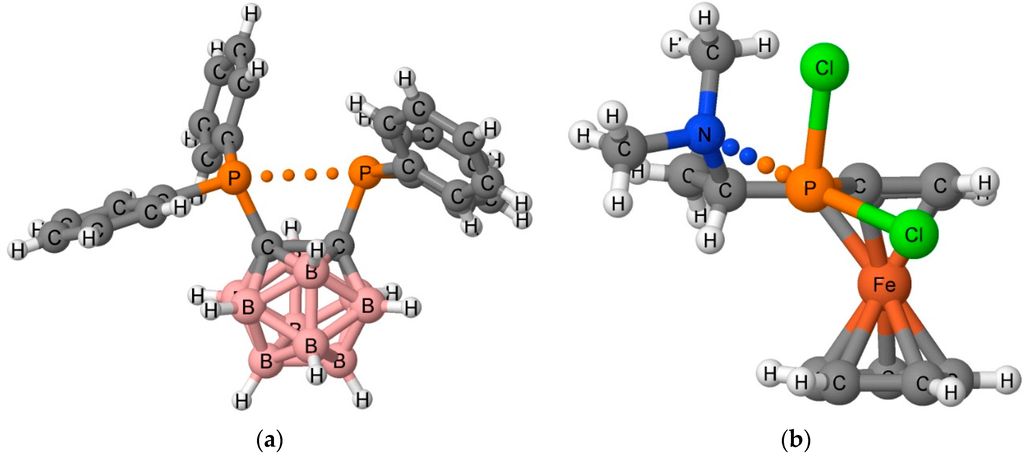
Figure 1.
X-ray structure of Cambridge Structural Database (CSD) Refcodes (a) XEBBEM01 and (b) QEZDOP. The pnicogen bond interaction is indicated with dots.
Figure 1.
X-ray structure of Cambridge Structural Database (CSD) Refcodes (a) XEBBEM01 and (b) QEZDOP. The pnicogen bond interaction is indicated with dots.
In the present article, we present the results of our search of the CSD for phosphine-water and arsine-water complexes in which water is either the proton donor in hydrogen-bonded complexes, or the electron-pair donor in pnicogen-bonded complexes. We also report the results of ab initio calculations on a series of complexes H2XP:OH2, for X = NC, F, Cl, CN, OH, CCH, H, and CH3, stabilized by either hydrogen bonds or pnicogen bonds. We present and discuss the structures, binding energies, and charge-transfer energies of these complexes, as well as equation-of-motion coupled cluster singles and doubles (EOM-CCSD) spin–spin coupling constants across hydrogen bonds and pnicogen bonds.
2. Methods
2.1. Cambridge Structural Database Search
The Cambridge Structural Database [21] version 5.36 with updates from November 2014, February 2015, and May 2015 was searched for complexes that contain P(III) and As(III) with water molecules. Included structures have a distance of 2.0 to 4.0 Å between the pnicogen atom and the oxygen atom of water.
2.2. Ab Initio Calculations
The structures of the isolated monomers and the binary complexes H2XP:OH2 were optimized at second-order Møller-Plesset perturbation theory (MP2) [22,23,24,25] with the aug'-cc-pVTZ basis set [26]. This basis set is derived from the Dunning aug-cc-pVTZ basis set [27,28] by removing diffuse functions from H atoms. Frequencies were computed to establish that the optimized structures correspond to equilibrium structures on their potential surfaces. Optimization and frequency calculations were performed using the Gaussian 09 program [29]. The binding energies (ΔE) of all complexes have been calculated as the total energy of the complex minus the sum of the total energies of the corresponding isolated monomers.
The electron densities of complexes have been analyzed using the Atoms in Molecules (AIM) methodology [30,31,32,33] employing the AIMAll [34] program. The topological analysis of the electron density produces the molecular graph of each complex. This graph identifies the location of electron density features of interest, including the electron density (ρ) maxima associated with the various nuclei, and saddle points which correspond to bond critical points (BCPs). The zero gradient line which connects a BCP with two nuclei is the bond path.
The Natural Bond Orbital (NBO) [35] method has been used to analyze the stabilizing charge-transfer interactions using the NBO6 program [36]. Since MP2 orbitals are nonexistent, the charge-transfer interactions have been computed using the B3LYP functional [37,38] with the aug’-cc-pVTZ basis set at the MP2/aug’-cc-pVTZ complex geometries, so that at least some electron correlation effects could be included.
Spin-spin coupling constants were evaluated using the EOM-CCSD method in the CI (configuration interaction)-like approximation [39,40], with all electrons correlated. For these calculations, the Ahlrichs [41] qzp basis set was placed on 13C, 15N, 17O, and 19F, and the qz2p basis set on 31P, 35Cl, and hydrogen-bonded 1H atoms. The Dunning cc-pVDZ basis set was placed on all other 1H atoms. All terms, namely, the paramagnetic spin-orbit (PSO), diamagnetic spin orbit (DSO), Fermi contact (FC), and spin dipole (SD), have been evaluated. The EOM-CCSD calculations were performed using ACES II [42] on the IBM Cluster 1350 (Glenn) at the Ohio Supercomputer Center.
3. Results and Discussion
3.1. CSD Search
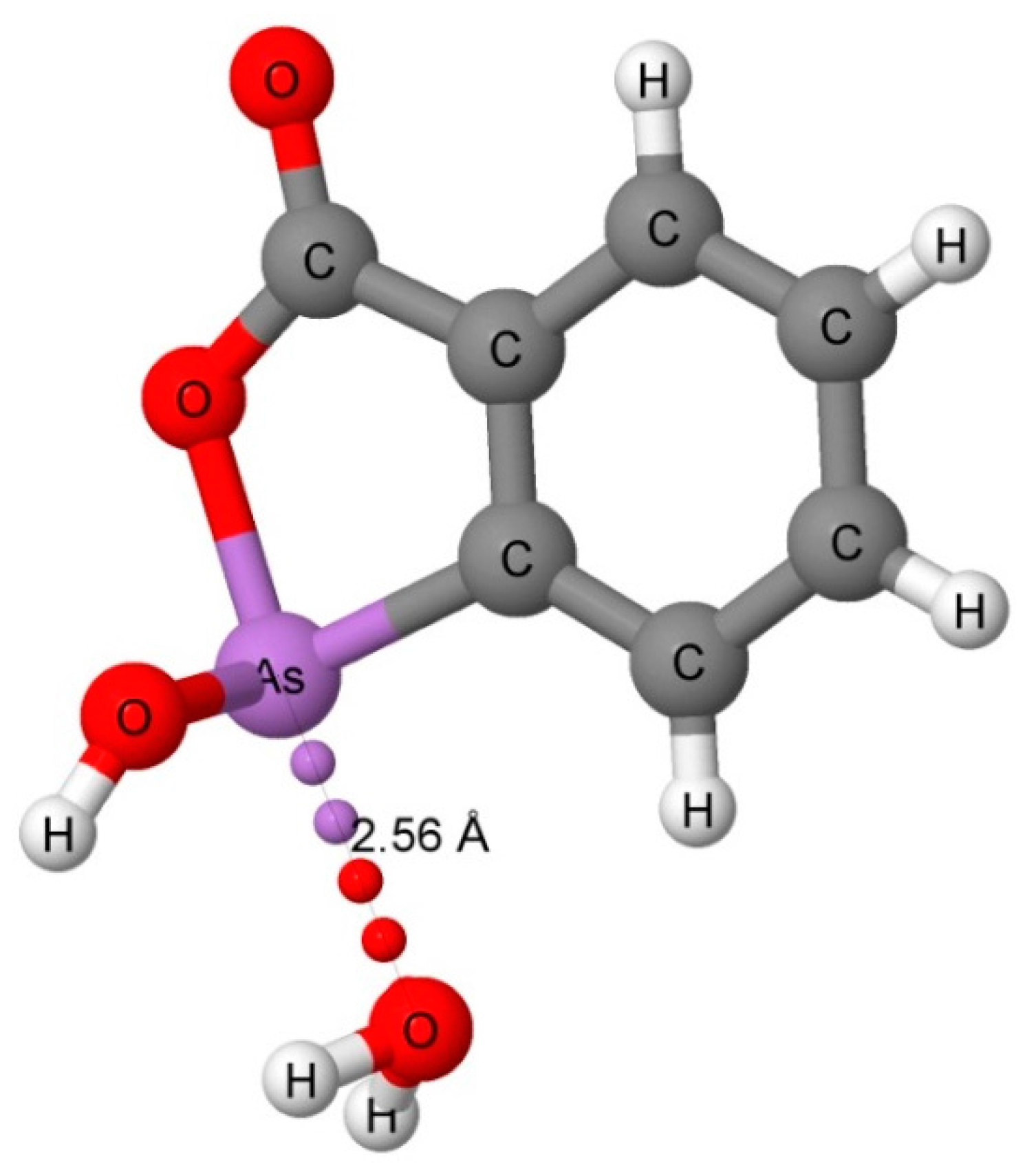
The CSD search found only three water-phosphine complexes and seven water-arsine complexes that have a distance of 2.0 to 4.0 Å between the pnicogen atom and the oxygen atom of water. Two of the water–phosphine complexes are hydrogen bonded (CSD Refcodes: AGAHIB and BEZTOR) with P···H distances of 2.48 and 2.72 Å, and P···O distances of 3.315 and 3.465 Å, respectively. The third structure (NOPYEX) corresponds to a pnicogen-bonded complex between a triphenylphosphine derivative and water with a longer P···O distance of 3.76 Å. Four of the water-arsine complexes (TELFAR, NOPYAT, FUTDUU, and IBAKIH) are pnicogen-bonded, and three (NIVWAQ, FUTDUU, and HAVKEW) are hydrogen-bonded. The pnicogen-bonded complex TELFAR which is illustrated in Figure 2 has a short As-O pnicogen bond distance of 2.56 Å. The O-As-O angle is 171°, which allows for the interaction of the O of water with the σ-hole of As. The short distance for this bond suggests that it has significant covalent character. The As···O distances in the other pnicogen-bonded complexes range between 3.61 and 3.78 Å. The hydrogen-bonded complexes have As···H distances between 2.81 and 3.22 Å, and As···O distances between 3.70 and 3.96 Å.
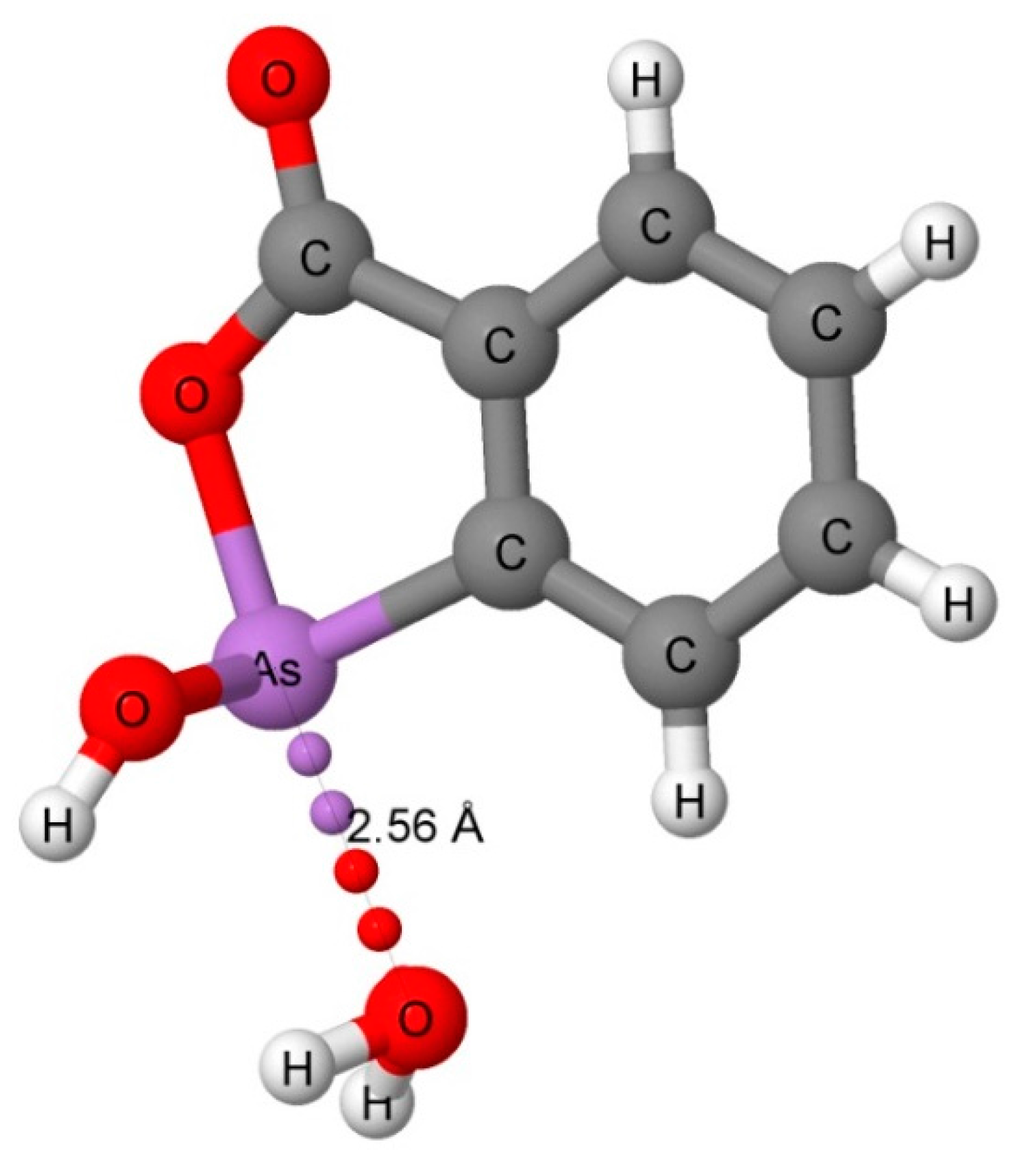
Figure 2.
The X-ray structure of CSD Refcode: TELFAR showing the As···O pnicogen bond interaction.
Figure 2.
The X-ray structure of CSD Refcode: TELFAR showing the As···O pnicogen bond interaction.
3.2. Computational Results
We have attempted to optimize eight complexes H2XP:HOH with O-H…P hydrogen bonds, and eight complexes with P…O pnicogen bonds, with X = NC, F, Cl, CN, OH, CCH, H, and CH3. However, only four hydrogen-bonded (HB) and six pnicogen-bonded (ZB) equilibrium complexes have been found on the potential surfaces. The structures, total energies, and molecular graphs of these complexes are reported in Table S1 of the Supporting Information, and their binding energies are given in Table 1. ZB complexes have binding energies which vary from –11.3 to –21.1 kJ·mol−1, while HB complexes have binding energies between –9.4 and –15.4 kJ·mol−1. The absolute values of the binding energies of the ZB complexes decrease in the order
while those of the HB complexes decrease in the reverse order
NC > F > Cl > CN > OH > CCH
CH3 > H > CCH > Cl.
It is apparent from Table 1 that the more electronegative substituents prefer to form pnicogen-bonded complexes, while the more electropositive substituents form hydrogen-bonded complexes. This trend follows the general trend of the Molecular Electrostatic Potential (MEP) values around the phosphorous atom [43]. H2ClP and H(CCH)P form both hydrogen-bonded and pnicogen-bonded complexes with H2O, with the latter more stable by 7.9 and 0.5 kJ∙mol−1, respectively.

Table 1.
Binding energies of equilibrium pnicogen-bonded (ZB) and hydrogen-bonded (HB) complexes of H2XP with H2O.
| H2XP, X = | Binding Energies (ΔE, kJ·mol−1) | |
|---|---|---|
| ZB | HB | |
| NC | –21.1 | |
| F | –19.5 | |
| Cl | –17.3 | –9.4 |
| CN | –16.9 | |
| OH | –12.8 | |
| CCH | –11.3 | –10.8 |
| H | –11.1 | |
| CH3 | –15.4 | |
3.2.1. Hydrogen-Bonded Complexes
Only four equilibrium hydrogen-bonded complexes have been found on the H2XP:HOH surfaces; namely, H2ClP:HOH, H2(CCH)P:HOH, H3P:HOH, and H2(CH3)P:HOH. The binding energies of these are given in Table 1, and selected structural parameters are reported in Table 2. The shortest intermolecular O···P and H···P distances are 3.360 and 2.536 Å, respectively, in H2(CH3)P:HOH. The remaining three complexes have O···P distances between 3.55 and 3.59 Å, and H···P distances between 2.62 and 2.66 Å. These distances are consistent with the experimental distances from the CSD. As evident from Table 2, the hydrogen bonds in H2(CCH)P:HOH and H3P:HOH are close to linear, with H-O-P angles of 7°, while these bonds in H2ClP:HOH and H2(CH3)P:HOH are nonlinear with values of 19 and 27°, respectively. Moreover, the H2O molecule is positioned similarly in H2ClP:HOH and H2(CCH)P:HOH, but has a different orientation in H3P:HOH and H2(CH3)P:HOH, as illustrated in Figure 3. In these two complexes, but particularly H2(CH3)P:HOH, there appears to be an attractive interaction between the water oxygen and the hydrogens of the substituent. All of these differences lead to a lack of correlation between the binding energies of these complexes and the O···P distances. However, the electrostatic minimum associated with the lone pair of the phosphorous atom [43] and the H···P distances do correlate. Although not included in Table 1 and Table 2, there is an equilibrium hydrogen-bonded complex formed between H2(OH)P and H2O with a binding energy of –28.5 kJ·mol−1. However, it is stabilized by an O-H…O hydrogen bond with the substituent O-H as the proton donor to H2O.

Table 2.
O···P, H···P, and O-H distances, and H-O-P angles for hydrogen-bonded complexes H2XP:HOH with Cs symmetry.
| H2XP, X = | Distance (R, Å) | Angles (<, °) | ||
|---|---|---|---|---|
| R(O···P) | R(H···P) | R(O-H) a | <H-O-P | |
| Cl | 3.553 | 2.656 | 0.965 | 19 |
| CCH | 3.585 | 2.629 | 0.966 | 7 |
| H | 3.575 | 2.620 | 0.966 | 7 |
| CH3 | 3.360 | 2.536 | 0.967 | 27 |
(a) The O-H distance in isolated H2O is 0.961 Å.
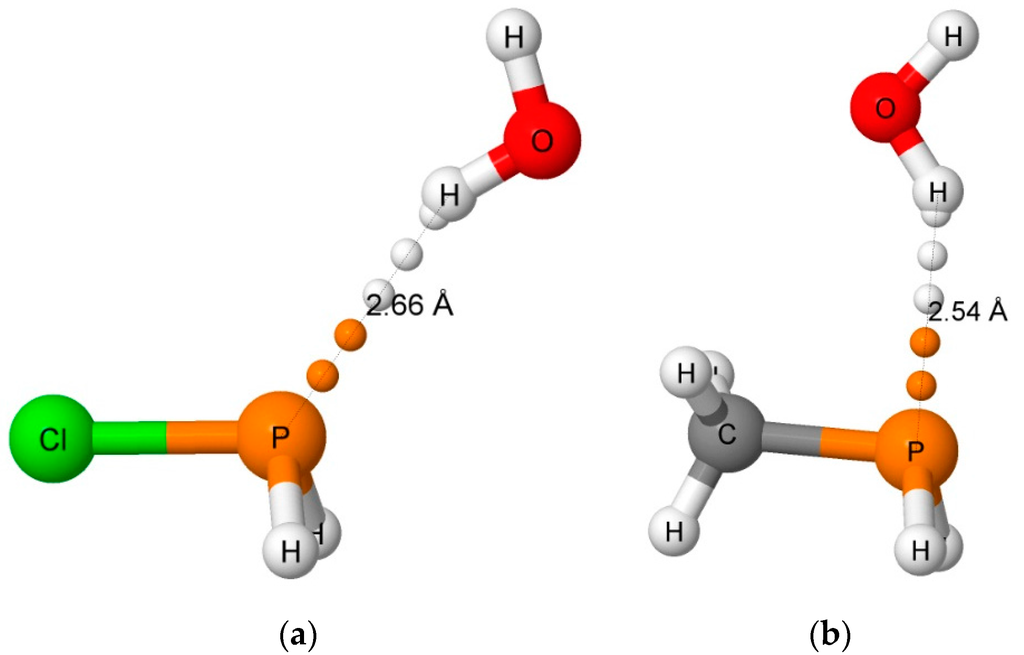
Figure 3.
The hydrogen-bonded complexes H2ClP:HOH (a) and H2(CH3)P:HOH (b). The orientation of the H2O molecule in H2(CCH)P:HOH is similar to that in H2ClP:HOH, while the orientation of H2O in H3P:HOH is similar to that in H2(CH3)P:HOH.
Figure 3.
The hydrogen-bonded complexes H2ClP:HOH (a) and H2(CH3)P:HOH (b). The orientation of the H2O molecule in H2(CCH)P:HOH is similar to that in H2ClP:HOH, while the orientation of H2O in H3P:HOH is similar to that in H2(CH3)P:HOH.
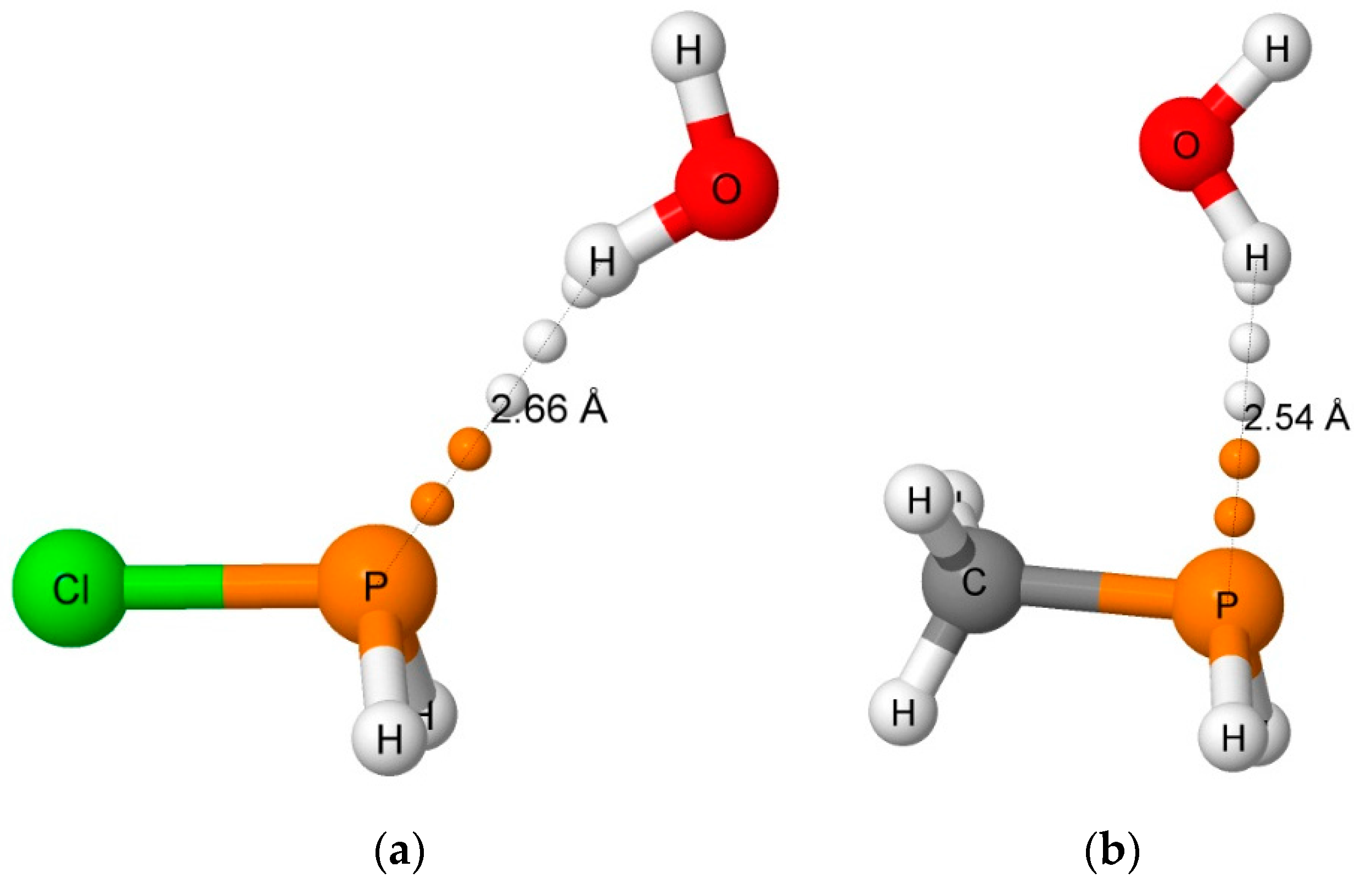
Table 3 presents the stabilizing NBO Plp→σ*H-O charge-transfer energies for the four hydrogen-bonded complexes. These energies range from 9 kJ·mol−1 for H2ClP:HOH to between 14 and 15 kJ·mol−1 for the remaining complexes. The charge-transfer energies do not correlate with either the O···P or the H···P distances.

Table 3.
Charge-transfer energies (Plp→σ*H-O) and coupling constants 2hJ(O-P), 1hJ(H-P), and 1J(O-H) for hydrogen-bonded complexes H2XP:HOH.
| H2XP, X = | Charge-Transfer Energies (kJ·mol−1) | Coupling Constants (Hz) | ||
|---|---|---|---|---|
| Plp→σ*H-O a | 2hJ(O-P) | 1hJ(H-P) | 1J(O-H) b | |
| Cl | 9.0 | –18.2 | –12.9 | –78.0 |
| CCH | 13.8 | –14.4 | –13.0 | –77.9 |
| H | 14.0 | –14.0 | –13.5 | –78.1 |
| CH3 | 15.0 | –24.1 | –15.0 | –78.4 |
a The Olp→σ*H-O charge-transfer energy in the complex of H2(OH)P with H2O that has the substituent O-H as the proton donor is 41.0 kJ·mol−1; b 1J(O-H) in isolated H2O is –77.0 Hz.
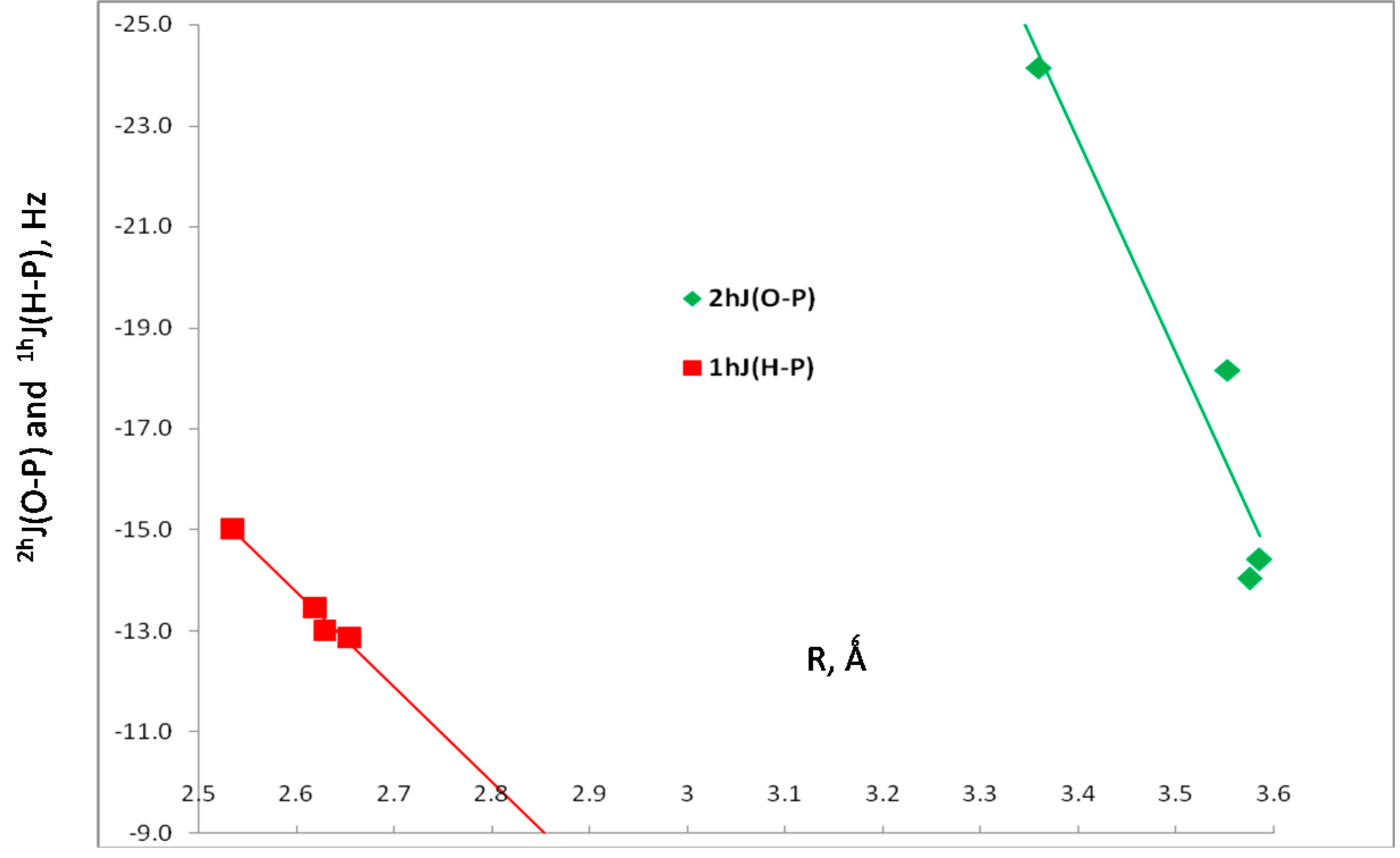
The two-bond coupling constant 2hJ(O-P) and the one-bond coupling constant 1hJ(H-P) across the hydrogen bonds, and the one-bond coupling constant 1J(O-H) for the hydrogen-bonded O-H group are also reported in Table 3. The components of these coupling constants are reported in Table S2 of the Supporting Information. Values of 2hJ(O-P) vary from –14 to –24 Hz, while values of 1hJ(H-P) lie between –13 and –15 Hz. The dependence of these two coupling constants on the corresponding O···P and H···P distances is shown graphically in Figure 4. Since there are only 4 points in each set and at least two of them have similar values of the coupling constant and the corresponding distance, only linear trendlines were used to illustrate the distance dependence. The correlation coefficients of these trendlines are 0.915 for 2hJ(O-P) and 0.973 for 1hJ(H-P). The third coupling constant, 1J(O-H) has a value of –77.0 Hz in the isolated H2O molecule, and increases in absolute value only slightly upon complex formation to between –77.9 and –78.4 Hz. The O-H distance of 0.961 Å in the monomer also increases only slightly upon complexation, with values between 0.965 and 0.967 Å. The one-bond coupling constant 1J(O-H) for the non-hydrogen-bonded O-H decreases to about –62 Hz.
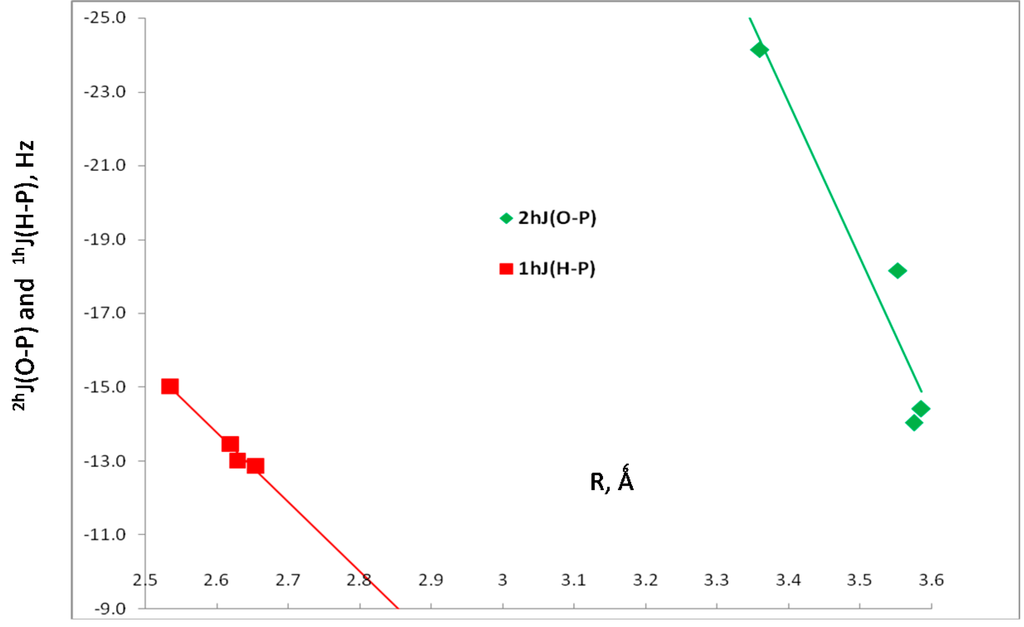
Figure 4.
2hJ(O-P) versus the O···P distance, and 1hJ(H-P) versus the H···P distance for HB complexes H2XP:HOH.
Figure 4.
2hJ(O-P) versus the O···P distance, and 1hJ(H-P) versus the H···P distance for HB complexes H2XP:HOH.
3.2.2. Pnicogen-Bonded Complexes
Three of the six equilibrium pnicogen-bonded H2XP:OH2 complexes have Cs symmetry with the H2O molecule in the symmetry plane, and three have C1 symmetry. For reasons of computational efficiency, particularly for the coupling constant calculations, we have re-optimized the C1 structures under the constraint of Cs symmetry with an in-plane H2O molecule. The binding energies of C1 and Cs structures are compared in Table 4. The Cs structures of H2FP:OH2, H2ClP:OH2, and H2(OH)P:OH2 with H2O in the Cs symmetry plane are only 0.1 to 0.4 kJ·mol−1 less stable than the C1 equilibrium structures. Moreover, the ordering of complexes according to decreasing binding energy is the same for the equilibrium structures and those with Cs symmetry, and the structures of C1 and corresponding Cs complexes are very similar. To ensure that there are no other pnicogen-bonded complexes with P···O pnicogen bonds, we also optimized a set of these complexes with Cs symmetry in which the H2O molecule does not lie in the symmetry plane. All of these complexes have one imaginary frequency, and smaller absolute values of the binding energies than the complexes with in-plane H2O molecules, as evident from Table 4. The two types of Cs complexes are illustrated in Figure 5.
The structures, total energies, and molecular graphs of the three pnicogen-bonded ZB complexes H2XP:OH2 with Cs symmetry, one imaginary frequency, and in-plane H2O molecules are reported in Table S3 of the Supporting Information. This table also provides these data for the six less-stable ZB complexes with Cs symmetry, one imaginary frequency, and out-of-plane H2O molecules. Table 5 presents selected data for the more stable Cs complexes with in-plane H2O molecules. The P···O distances in these complexes range from 2.755 Å in H2FP:OH2 to 3.036 Å in H2(CCH)P:OH2. Their binding energies do not correlate well with the P···O distances, as indicated by correlation coefficients of 0.7 for linear, quadratic, and exponential trendlines. The O-P-A angles, with A the atom of X directly bonded to P, are also reported in Table 5. These angles vary between 161 and 168°, indicating that the O-P-A arrangement approaches linearity. These values are consistent with the values of the P-P-A and N-P-A angles in the pnicogen-bonded complexes (H2XP)2 [44] and H2XP:NXH2 [45].

Table 4.
Binding energies of pnicogen-bonded complexes with C1 and Cs symmetries
| H2XP, X = | Equilibrium Symmetry | ΔE (kJ·mol−1) for Equilibrium Structures | ΔE (kJ·mol−1) for Cs Structures with H2O in-Plane | ΔE (kJ·mol−1) for Cs Structures with H2O out-of-Plane |
|---|---|---|---|---|
| NC | Cs | –21.1 | –21.1 | –19.9b |
| F | C1 | –19.5 | –19.2a | –18.5b |
| Cl | C1 | –17.3 | –17.2a | –16.3b |
| CN | Cs | –16.9 | –16.9 | –15.7b |
| OH | C1 | –12.8 | –12.4a | –12.1b |
| CCH | Cs | –11.3 | –11.3 | –10.1b |
a These Cs structures with water in-plane have one imaginary frequency; b These complexes with out-of-plane H2O molecules have one imaginary frequency.
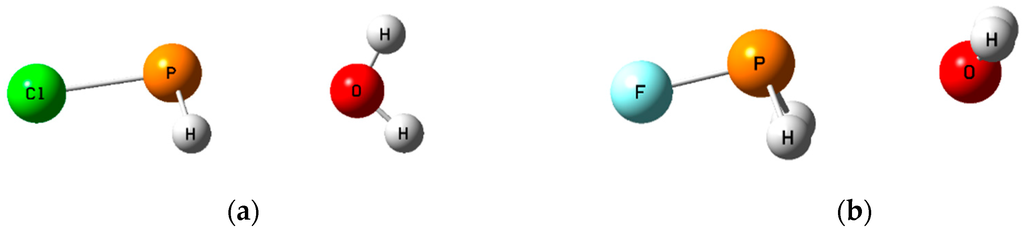
Figure 5.
The pnicogen-bonded complexes H2ClP:OH2 (a) with the H2O molecule in the Cs symmetry plane; and H2FP:OH2 (b) with Cs symmetry and an out-of-plane H2O molecule.
Figure 5.
The pnicogen-bonded complexes H2ClP:OH2 (a) with the H2O molecule in the Cs symmetry plane; and H2FP:OH2 (b) with Cs symmetry and an out-of-plane H2O molecule.
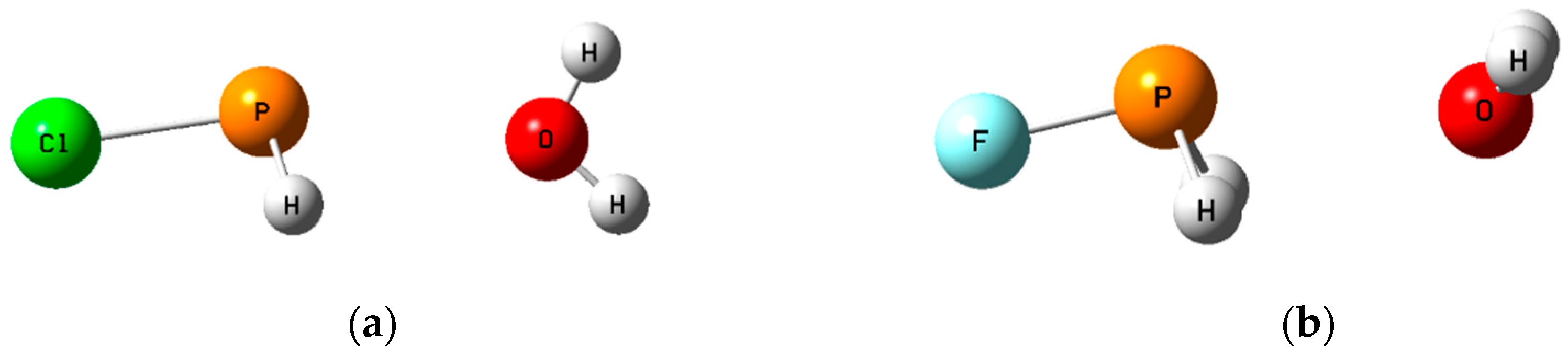
Table 5 also presents the NBO Olp→σ*P-A charge-transfer energies in these pnicogen-bonded complexes. Charge-transfer energies vary from 8 kJ·mol−1 in H2(CCH)P:OH2 to 20 kJ·mol−1 in H2(NC)P:OH2. They exhibit an exponential dependence on the P···O distance, with a correlation coefficient of 0.949.

Table 5.
P···O distances, O-P-A angles, charge-transfer energies, and 1pJ(P-O) coupling constants for pnicogen-bonded complexes H2XP:OH2 with Cs symmetry and in-plane H2O molecules.
| H2XP, X = | Distance (R, Å) | Angles (<, °) | Charge-Pransfer Energies (kJ·mol−1) | Coupling Constants (Hz) |
|---|---|---|---|---|
| R(P···O) | <O-P-Aa | Olp→σ*P-A | 1pJ(P-O) | |
| NCb | 2.800 | 165 | 20.3 | –62.5 |
| F | 2.755 | 167 | 19.5 | –69.8 |
| Cl | 2.835 | 166 | 17.4 | –61.2 |
| CNb | 2.944 | 161 | 12.3 | –41.9 |
| OH | 2.919 | 166 | 11.6 | –46.7 |
| CCH | 3.036 | 167 | 8.0 | –36.7 |
a A is the atom of X directly bonded to P. b The atom written first is directly bonded to P.
Table 5 also reports the spin–spin coupling constants 1pJ(P-O) across the pnicogen bonds. The components of these coupling constants are reported in Table S4 of the Supporting Information. 1pJ(P-O) values are dominated by the Fermi-contact terms, and vary from –37 Hz in H2(CCH)P:OH2 to –70 Hz in H2FP:OH2. Figure 6 illustrates the second-order dependence of 1pJ(P-O) on the P···O distance, with a correlation coefficient of 0.979.
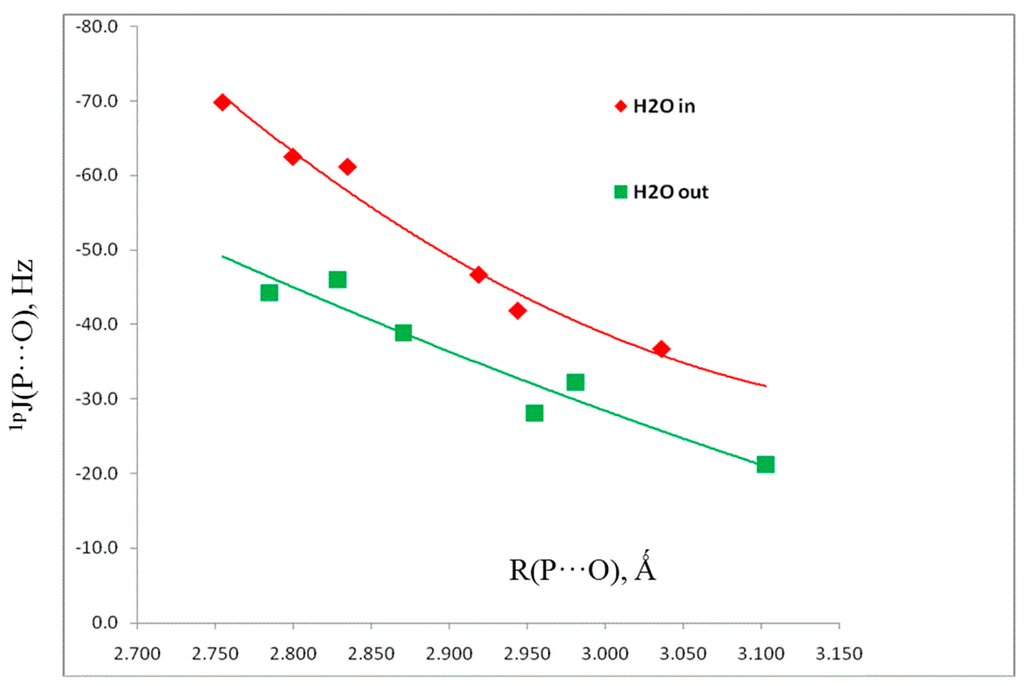
Figure 6.
1pJ(P-O) versus the P···O distance for pnicogen-bonded complexes H2XP:OH2 with Cs symmetry and in-plane and out-of-plane H2O molecules.
Figure 6.
1pJ(P-O) versus the P···O distance for pnicogen-bonded complexes H2XP:OH2 with Cs symmetry and in-plane and out-of-plane H2O molecules.
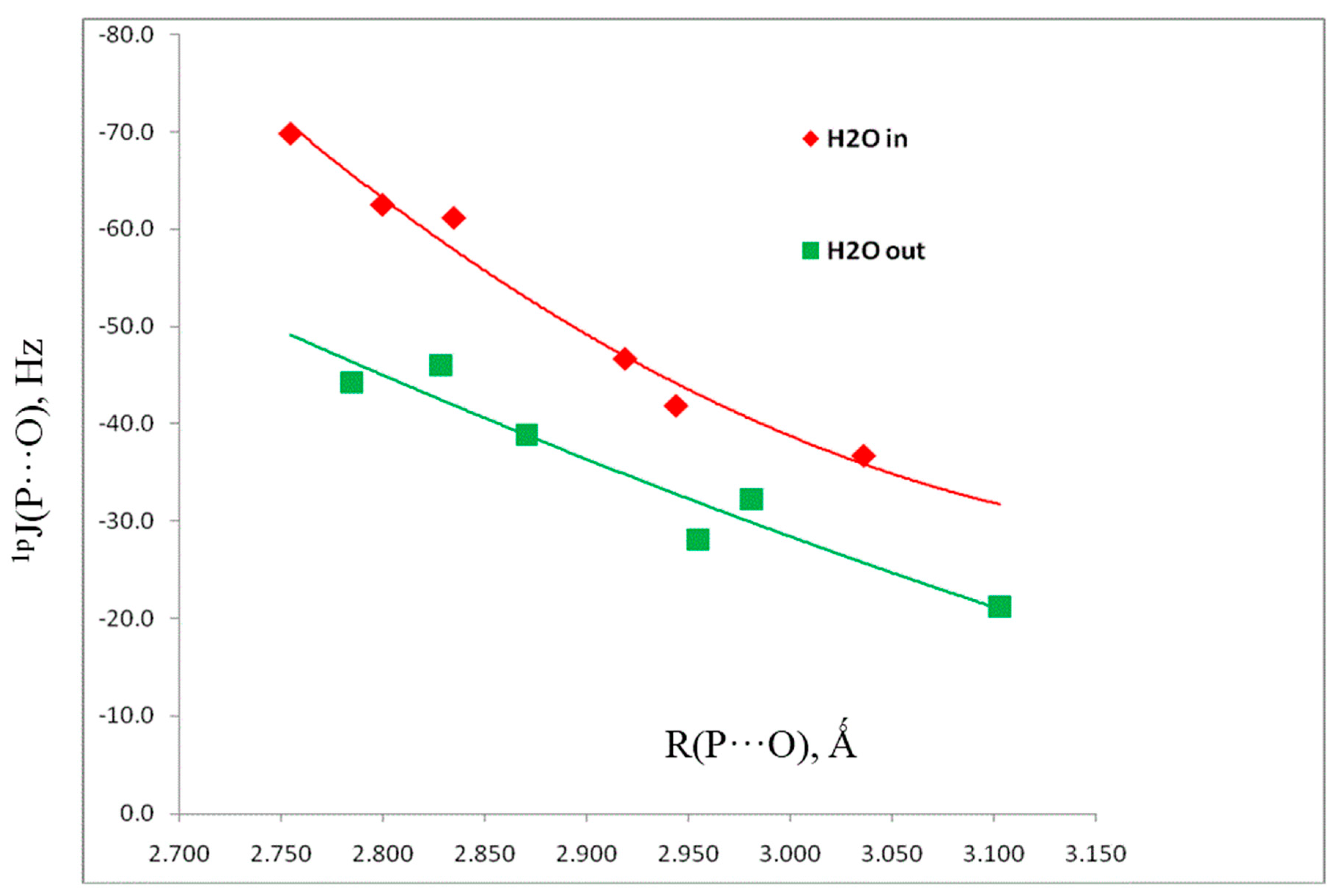
A second reason for optimizing the set of complexes with Cs symmetry and out-of-plane H2O molecules is to examine the coupling constants of these structures, the components of which are reported in Table S5. Since 1pJ(P-O) values are also dominated by the Fermi-contact terms, it is expected that the s electron densities at O and at P interacting with O will be very different in the ground state and the excited states which couple to the ground state for these two orientations of H2O molecules. It is apparent from Figure 6 that such is the case, since at the same P···O distances, the points for structures with out-of-plane H2O molecules lie below those for in-plane H2O molecules.
Finally, a plot of 2hJ(O-P) for hydrogen-bonded complexes and 1pJ(P-O) for pnicogen-bonded complexes with Cs symmetry and in-plane H2O molecules versus the P···O distance is reported as Figure S1 of the Supporting Information. At the shorter P···O distances, the absolute values of 2hJ(O-P) are greater than the values of 1pJ(P-O) at longer distances, but a single second-order trendline with a correlation coefficient of 0.981 describes the distance dependence of both coupling constants.
4. Conclusions
Crystal structures have long been used as a tool for identifying and confirming the presence of weak intermolecular interactions, including hydrogen bonds and pnicogen bonds. A search of the CSD for complexes of water with phosphine and arsine identified two water–phosphine complexes stabilized by hydrogen bonds and one stabilized by a pnicogen bond, as well as three water–arsine complexes with hydrogen bonds and four with pnicogen bonds. The range of P···O distances in the phosphine complexes is consistent with the results of ab initio MP2/aug’-cc-pVTZ calculations carried out on complexes H2XP:OH2, for X = NC, F, Cl, CN, OH, CCH, H, and CH3. Only hydrogen-bonded complexes are found on the H2(CH3)P:OH2 and H3P:OH2 potential surfaces, while only pnicogen-bonded complexes exist on the H2(NC)P:OH2, H2FP:OH2, H2(CN)P:OH2, and H2(OH)P:OH2 surfaces. Both hydrogen-bonded and pnicogen-bonded complexes are found on the H2ClP:OH2 and H2(CCH)P:OH2 surfaces, with the pnicogen-bonded complexes more stable than the corresponding hydrogen-bonded complexes. It is apparent that the more electronegative substituents prefer to form pnicogen-bonded complexes, while the more electropositive substituents form hydrogen-bonded complexes. The binding energies of pnicogen-bonded complexes range from –11 to –21 kJ·mol−1, while the pnicogen-bonded complexes have binding energies from –9 to –15 kJ·mol−1. The hydrogen-bonded complexes are stabilized by charge transfer from the lone pair on P to the antibonding σ* H-O orbital, while pnicogen-bonded complexes are stabilized by charge transfer from the lone pair on O to the antibonding σ* P-A orbital, with A the atom of X directly bonded to P.
Spin-spin coupling constants 2h(O-P) and 1hJ(H-P) correlate with the O···P and H···P distances, respectively, while 1J(O-H) for the hydrogen-bonded O-H group increases in absolute value only slightly upon complex formation. 1pJ(P-O) coupling constants were computed for two sets of pnicogen-bonded complexes, one with Cs symmetry and the H2O molecule in the symmetry plane, and the other also with Cs symmetry but with an out-of-plane H2O molecule. 1pJ(P-O) for both sets are quadratically dependent on the P···O distance. The different orientations of the H2O molecule in these two sets alter the s electron densities at O and P, and lead to greater absolute values of 1pJ(P-O) for complexes with in-plane H2O molecules compared to those with out-of-plane H2O molecules.
Supplementary Materials
Supplementary materials can be found at http://www.mdpi.com/2073-4352/6/2/19/s1.
Acknowledgments
This work was carried out with financial support from the Ministerio de Economía y Competitividad (Project No. CTQ2012-35513-C02-02) and Comunidad Autónoma de Madrid (Project FOTOCARBON, ref. S2013/MIT-2841). Thanks are also given to the Ohio Supercomputer Center and CTI (CSIC) for their continued computational support.
Author Contributions
Ibon Alkorta and Janet E. Del Bene carried out the calculations and analyzed the data. All three authors contributed to the manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Allen, F.H.; Wood, P.A.; Galek, P.T.A. Role of chloroform and dichloromethane solvent molecules in crystal packing: An interaction propensity study. Acta Cryst. 2013, B69, 379–388. [Google Scholar] [CrossRef] [PubMed]
- Van de Streek, J. All series of multiple solvates (including hydrates) from the Cambridge Structural Database. CrystEngComm 2007, 9, 350–352. [Google Scholar] [CrossRef]
- Infantes, L.; Chisholm, J.; Motherwell, S. Extended motifs from water and chemical functional groups in organic molecular crystals. CrystEngComm 2003, 5, 480–486. [Google Scholar] [CrossRef]
- Infantes, L.; Motherwell, S. Water clusters in organic molecular crystals. CrystEngComm 2002, 4, 454–461. [Google Scholar] [CrossRef]
- Gillon, A.L.; Feeder, N.; Davey, R.J.; Storey, R. Hydration in Molecular Crystals-A Cambridge Structural Database Analysis. Cryst. Growth Des. 2003, 3, 663–673. [Google Scholar] [CrossRef]
- Infantes, L.; Fabian, L.; Motherwell, S. Organic crystal hydrates: What are the important factors for formation. CrystEngComm 2007, 9, 65–71. [Google Scholar] [CrossRef]
- Pimentel, G.C.; McClellan, A.L. The Hydrogen Bond; W.H. Freeman: San Francisco, CA, USA, 1960. [Google Scholar]
- Arunan, E.; Desiraju Gautam, R.; Klein Roger, A.; Sadlej, J.; Scheiner, S.; Alkorta, I.; Clary David, C.; Crabtree Robert, H.; Dannenberg Joseph, J.; Hobza, P.; et al. Defining the hydrogen bond: An account (IUPAC Technical Report). Pure Appl. Chem. 2011, 83, 1619–1636. [Google Scholar] [CrossRef]
- Arunan, E.; Desiraju Gautam, R.; Klein, R.-A.; Sadlej, J.; Scheiner, S.; Alkorta, I.; Clary, D.C.; Crabtree, R.H.; Dannenberg, J.J.; Hobza, P.; et al. Definition of the hydrogen bond (IUPAC Recommendations 2011). Pure Appl. Chem. 2011, 83, 1637–1641. [Google Scholar] [CrossRef]
- Sundberg, M.R.; Uggla, R.; Viñas, C.; Teixidor, F.; Paavola, S.; Kivekäs, R. Nature of intramolecular interactions in hypercoordinate C-substituted 1,2-dicarba-closo-dodecaboranes with short P···P distances. Inorg. Chem. Commun. 2007, 10, 713–716. [Google Scholar] [CrossRef]
- Bauer, S.; Tschirschwitz, S.; Lönnecke, P.; Frank, R.; Kirchner, B.; Clarke, M.L.; Hey-Hawkins, E. Enantiomerically pure bis(phosphanyl)carbaborane(12) compounds. Eur. J. Inorg. Chem. 2009, 2009, 2776–2788. [Google Scholar] [CrossRef]
- Tschirschwitz, S.; Lonnecke, P.; Hey-Hawkins, E. Aminoalkylferrocenyldichlorophosphanes: Facile synthesis of versatile chiral starting materials. Dalton Trans. 2007, 1377–1382. [Google Scholar] [CrossRef] [PubMed]
- Politzer, P.; Murray, J.; Janjić, G.; Zarić, S. σ-Hole interactions of covalently-bonded nitrogen, phosphorus and arsenic: A survey of crystal structures. Crystals 2014, 4, 12–31. [Google Scholar] [CrossRef]
- Sánchez-Sanz, G.; Alkorta, I.; Trujillo, C.; Elguero, J. Intramolecular pnicogen interactions in PHF-(CH2)n-PHF (n = 2–6) systems. ChemPhysChem 2013, 14, 1656–1665. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Sanz, G.; Trujillo, C.; Alkorta, I.; Elguero, J. Intramolecular pnicogen interactions in phosphorus and arsenic analogues of proton sponges. Phys. Chem. Chem. Phys. 2014, 16, 15900–15909. [Google Scholar] [CrossRef] [PubMed]
- Scheiner, S. A new noncovalent force: Comparison of P···N interaction with hydrogen and halogen bonds. J. Chem. Phys. 2011, 134, 094315. [Google Scholar] [CrossRef] [PubMed]
- Zahn, S.; Frank, R.; Hey-Hawkins, E.; Kirchner, B. Pnicogen bonds: A new molecular linker? Chem. Eur. J. 2011, 17, 6034–6038. [Google Scholar] [CrossRef] [PubMed]
- Scheiner, S. The pnicogen bond: Its relation to hydrogen, halogen, and other noncovalent bonds. Acc. Chem. Res. 2012, 46, 280–288. [Google Scholar] [CrossRef] [PubMed]
- Scheiner, S. Detailed comparison of the pnicogen bond with chalcogen, halogen, and hydrogen bonds. Int. J. Quantum Chem. 2013, 113, 1609–1620. [Google Scholar] [CrossRef]
- Del Bene, J.E.; Alkorta, I.; Elguero, J. The pnicogen bond in review: Structures, binding energies, bonding properties, and spin-spin coupling constants of complexes stabilized by pnicogen bonds. In Noncovalent Forces; Challenges and Advances in Computational Chemistry and Physics Series; Scheiner, S., Ed.; Springer: Gewerbestrasse, Switzerland, 2015; Volume 19, pp. 191–263. [Google Scholar]
- Allen, F. The Cambridge Structural Database: A quarter of a million crystal structures and rising. Acta Crystallogr. Sect. B 2002, 58, 380–388. [Google Scholar] [CrossRef]
- Pople, J.A.; Binkley, J.S.; Seeger, R. Theoretical models incorporating electron correlation. Int. J. Quantum Chem. Quantum Chem. Symp. 1976, 10, 1–19. [Google Scholar] [CrossRef]
- Krishnan, R.; Pople, J.A. Approximate fourth-order perturbation theory of the electron correlation energy. Int. J. Quantum Chem. 1978, 14, 91–100. [Google Scholar] [CrossRef]
- Bartlett, R.J.; Silver, D.M. Many-body perturbation theory applied to electron pair correlation energies. I. Closed-shell first-row diatomic hydrides. J. Chem. Phys. 1975, 62, 3258–3268. [Google Scholar] [CrossRef]
- Bartlett, R.J.; Purvis, G.D. Many-body perturbation theory, coupled-pair many-electron theory, and the importance of quadruple excitations for the correlation problem. Int. J. Quantum Chem. 1978, 14, 561–581. [Google Scholar] [CrossRef]
- Del Bene, J.E. Proton affinities of ammonia, water, and hydrogen fluoride and their anions: A quest for the basis-set limit using the dunning augmented correlation-consistent basis sets. J. Phys. Chem. 1993, 97, 107–110. [Google Scholar] [CrossRef]
- Dunning, T.H. Gaussian Basis Sets for Use in Correlated Molecular Calculations. I. The Atoms Boron through Neon and Hydrogen. J. Chem. Phys. 1989, 90, 1007–1023. [Google Scholar] [CrossRef]
- Woon, D.E.; Dunning, T.H. Gaussian Basis Sets for Use in Correlated Molecular Calculations. V. Core-Valence Basis Sets for Boron Through Neon. J. Chem. Phys. 1995, 103, 4572–4585. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; et al. Gaussian–09, Revision D.01; Gaussian, Inc.: Wallingford, CT, USA, 2009. [Google Scholar]
- Bader, R.F.W. A quantum theory of molecular structure and its applications. Chem. Rev. 1991, 91, 893–928. [Google Scholar] [CrossRef]
- Bader, R.F.W. Atoms in Molecules, A Quantum Theory; Oxford University Press: Oxford, UK, 1990. [Google Scholar]
- Popelier, P.L.A. Atoms in Molecules. An Introduction; Prentice Hall: Harlow, UK, 2000. [Google Scholar]
- Matta, C.F.; Boyd, R.J. The Quantum Theory of Atoms in Molecules: From Solid State to DNA and Drug Design; Wiley-VCH: Weinheim, Germany, 2007. [Google Scholar]
- Keith, T.A. AIMALL, Version 15.09.27; TK Gristmill Software: Overland Park, KS, USA, 2011. Available online: http://aim.tkgristmill.com (accessed on 31 December 2015).
- Reed, A.E.; Curtiss, L.A.; Weinhold, F. Intermolecular interactions from a Natural Bond Orbital, donor-acceptor viewpoint. Chem. Rev. 1988, 88, 899–926. [Google Scholar] [CrossRef]
- Glendening, E.D.; Badenhoop, J.K.; Reed, A.E.; Carpenter, J.E.; Bohmann, J.A.; Morales, C.M.; Landis, C.R.; Weinhold, F. NBO 6.0; University of Wisconsin: Madison, WI, USA, 2013. [Google Scholar]
- Becke, A.D. Density functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 1993, 98, 5648–5652. [Google Scholar] [CrossRef]
- Lee, C.; Yang, W.; Parr, R.G. Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 1988, 37, 785789. [Google Scholar] [CrossRef]
- Perera, S.A.; Nooijen, M.; Bartlett, R.J. Electron correlation effects on the theoretical calculation of Nuclear Magnetic Resonance spin–spin coupling constants. J. Chem. Phys. 1996, 104, 3290–3305. [Google Scholar] [CrossRef]
- Perera, S.A.; Sekino, H.; Bartlett, R.J. Coupled–cluster calculations of indirect nuclear coupling constants: The importance of non–Fermi-contact contributions. J. Chem. Phys. 1994, 101, 2186–2196. [Google Scholar] [CrossRef]
- Schäfer, A.; Horn, H.; Ahlrichs, R. Fully optimized contracted Gaussian basis sets for atoms Li to Kr. J. Chem. Phys. 1992, 97, 2571–2577. [Google Scholar] [CrossRef]
- Stanton, J.F.; Gauss, J.; Watts, J.D.; Nooijen, M.; Oliphant, N.; Perera, S.A.; Szalay, P.S.; Lauderdale, W.J.; Gwaltney, S.R.; Beck, S.; et al. ACES II; University of Florida: Gainesville, FL, USA, 2014. [Google Scholar]
- Del Bene, J.E.; Alkorta, I.; Elguero, J. Characterizing Complexes with Pnicogen Bonds Involving sp2 Hybridized Phosphorus Atoms: (H2C=PX)2 with X = F, Cl, OH, CN, NC,CCH, H, CH3, and BH2. J. Phys. Chem. A 2013, 117, 6893–6903. [Google Scholar]
- Del Bene, J.E.; Alkorta, I.; Sanchez-Sanz, G.; Elguero, J. 31P-31P Spin-spin coupling constants for Pnicogen Homodimers. Chem. Phys. Lett. 2011, 512, 184–187. [Google Scholar] [CrossRef]
- Del Bene, J.E.; Alkorta, I.; Sanchez-Sanz, G.; Elguero, J. Structures, Energies, Bonding, and NMR Properties of Pnicogen Complexes H2XP:NXH2 (X = H, CH3, NH2, OH, F, Cl). J. Phys. Chem. A 2011, 115, 13724–13731. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
