Crystal Structures of Two 1,4-Diamino-1,2,4-triazolium Salts
Abstract
:1. Introduction
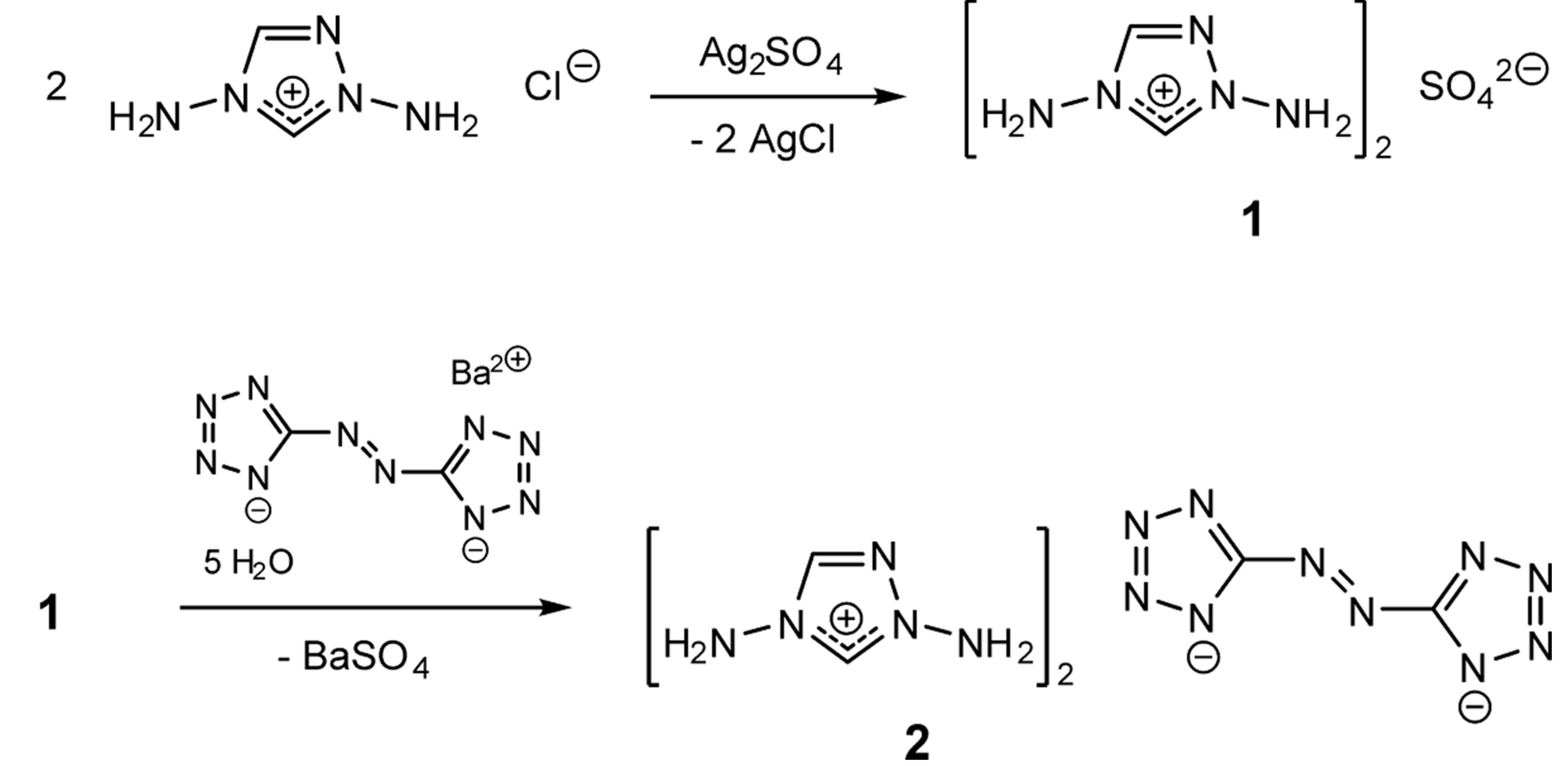
2. Results and Discussion
| Compound | 1 | 2 |
|---|---|---|
| CCDC No. | 1441522 | 1441523 |
| Empirical formula | 2(C2H6N5)·O4S | 2(C2H6N5)·C2N10 |
| Formula weight | 296.30 | 364.36 |
| Crystal system | Monoclinic | Monoclinic |
| Space group | Pc | P21/c |
| a/Å | 11.0114(3) | 3.5735(3) |
| b/Å | 8.4671(2) | 14.8270(12) |
| c/Å | 12.8156(5) | 13.4559(11) |
| β/° | 92.356(3) | 95.664(3) |
| Volume/Å3 | 1193.85(6) | 709.47(10) |
| Z | 4 | 2 |
| Dx/g·cm−3 | 1.648 | 1.706 |
| μ/mm−1 | 0.31 | 0.13 |
| F(000) | 616 | 376 |
| T/K | 173 | 183 |
| Crystal size/mm3 | 0.54 × 0.20 × 0.16 | 0.18 × 0.04 × 0.03 |
| θmax/° | 25.4 | 25.0 |
| Index ranges | –13 ≤ h ≤ 11, –10 ≤ k ≤ 10, –14 ≤ l ≤ 15 | –4 ≤ h≤ 3, –17 ≤ k ≤ 17, –15 ≤ l ≤ 15 |
| Reflections collected | 7080 | 9515 |
| Independent reflections (Rint) | 3571 (0.028) | 1250 (0.060) |
| Observed reflections [I > 2σ(I)] | 3428 | 1001 |
| Absorption correction | multi-scan | multi-scan |
| Restraints/parameters | 18/392 | 4/135 |
| Goodness-of-fit on F2 | 1.09 | 1.06 |
| R1/wR2 [I > 2σ(I)] | 0.026/0.077 | 0.037/0.092 |
| R1/wR2 (all data) | 0.029/0.079 | 0.052/0.098 |
| Δρmax/Δρmin/e.Å−3 | 0.23/–0.25 | 0.54/–0.22 |
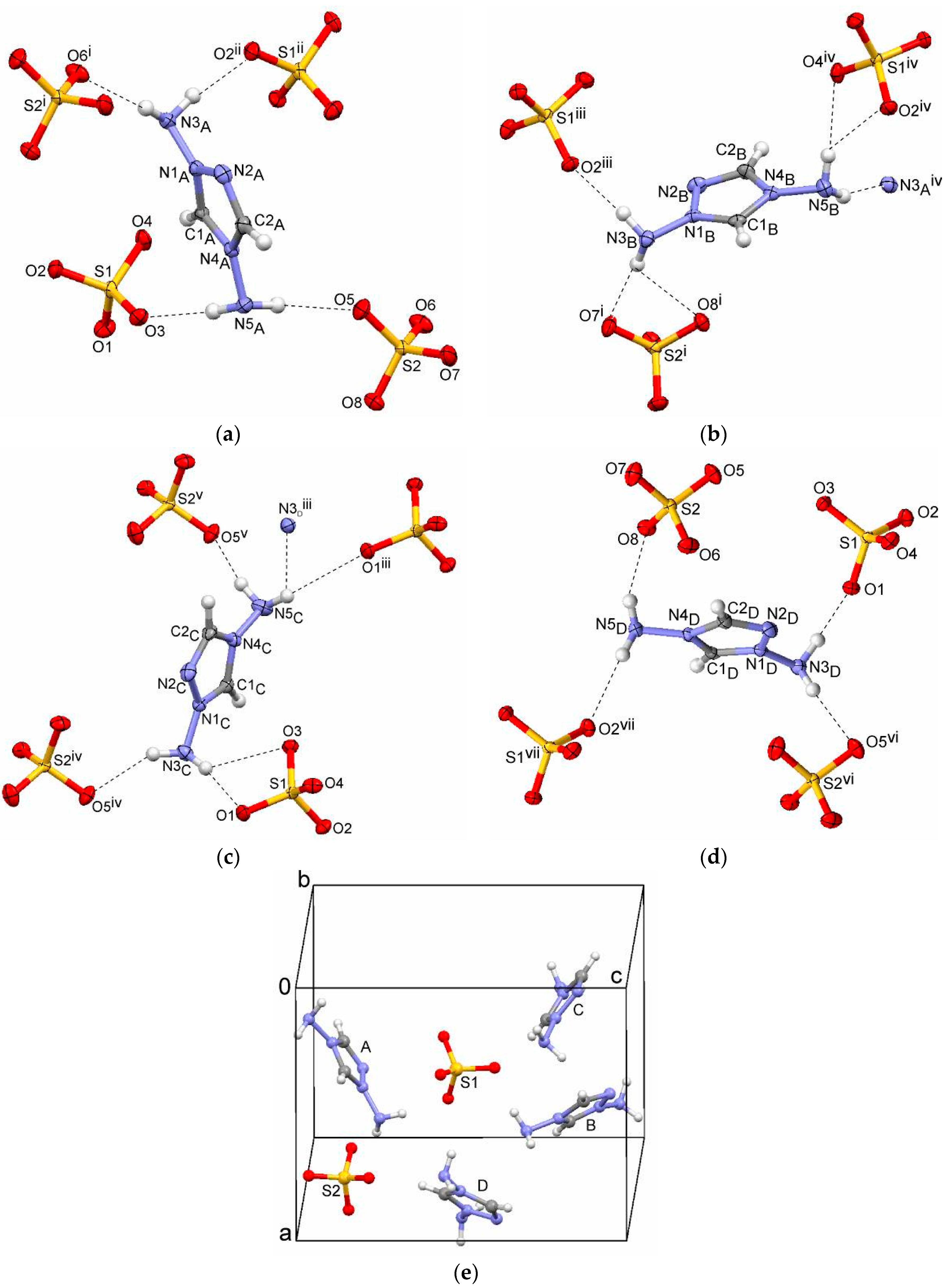
2.1. Bis(1,4-diamino-1,2,4-triazolium) Sulfate (1)
| Compound | Interaction | H···A | D···A | D–H···A | Symmetry Operation A |
|---|---|---|---|---|---|
| 1 | N5A–H51A···O5 | 2.04(2) | 2.913(3) | 160(3) | x, y, z |
| N5A–H52A···O3 | 2.04(2) | 2.946(3) | 173(3) | x, y, z | |
| N3C–H31C···O1 | 2.11(3) | 3.008(3) | 169(3) | x, y, z | |
| N3C–H31C···O3 | 2.48(3) | 3.115(3) | 128(3) | x, y, z | |
| N3C–H32C···O5 | 2.19(3) | 2.962(4) | 144(3) | x, −y, −1/2 + z | |
| N3B–H31B···O2 | 2.15(2) | 2.932(3) | 145(3) | x, 1 + y, z | |
| N3B–H32B···O7 | 2.07(2) | 2.951(3) | 172(3) | 1 + x, y, z | |
| N5D–H51D···O8 | 2.06(3) | 2.871(3) | 151(3) | x, y, z | |
| N5D–H52D···O2 | 2.16(2) | 2.996(3) | 155(3) | −1 + x, y, z | |
| N3D–H31D···O1 | 2.00(2) | 2.889(3) | 163(3) | x, y, z | |
| N3D–H32D···O5 | 2.16(2) | 3.042(4) | 170(3) | x, −1 + y, z | |
| N5B–H51B···O4 | 2.14(2) | 2.989(4) | 162(3) | x, −y, −1/2 + z | |
| N5B–H52B···N3A | 2.26(2) | 3.108(4) | 159(3) | x, −y, −1/2 + z | |
| N5C–H52C···O5 | 2.11(3) | 2.922(4) | 152(3) | x, 1 − y, −1/2 + z | |
| N3A–H31A···O6 | 2.03(2) | 2.874(3) | 157(3) | 1 + x, y, z | |
| N3A–H32A···O2 | 1.98(2) | 2.856(3) | 166(3) | x, −y, 1/2 + z | |
| N3B–H32B···O8 | 2.54(3) | 3.123(3) | 124(2) | 1 + x, y, z | |
| N5C–H51C···O1 | 2.56(3) | 3.353(3) | 150(3) | x, 1 + y, z | |
| N5C–H51C···N3D | 2.68(3) | 3.301(4) | 129(2) | x, 1 + y, z | |
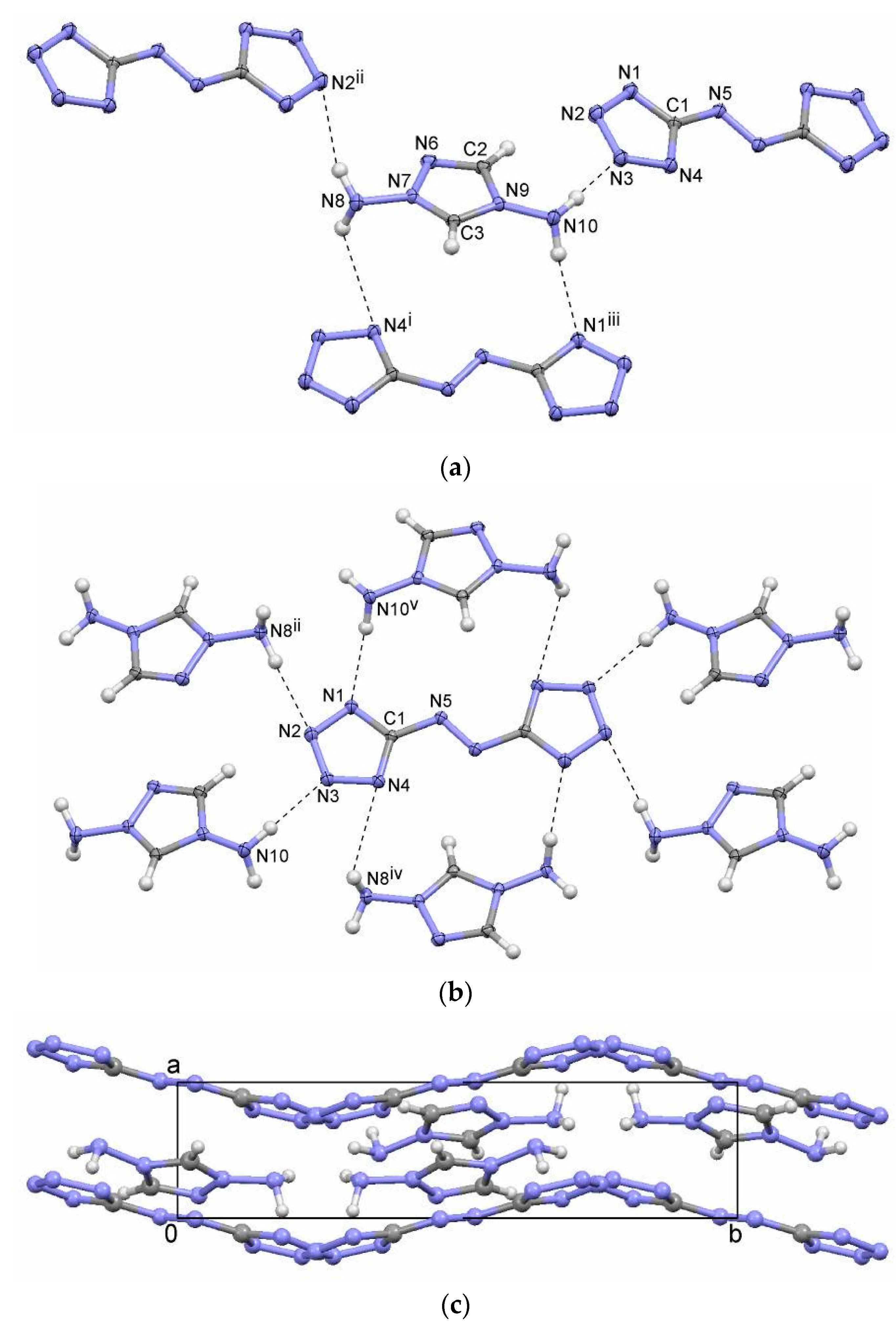
| 2 | N10–HA···N3 | 2.12(2) | 3.018(2) | 169(2) | x, y, z |
| N10–HB···N1 | 2.16(2) | 3.048(2) | 167(2) | x, 3/2 – y, –1/2 + z | |
| N8–HA···N2 | 2.38(2) | 3.152(2) | 146(2) | 1 – x, 1 – y, 1 – z | |
| N8–HB···N4 | 2.64(3) | 3.221(2) | 125(2) | –x, –1/2 + y, 1/2 – z |
2.2. Bis(1,4-diamino-1,2,4-triazolium) 5,5′-Azotetrazolate (2)
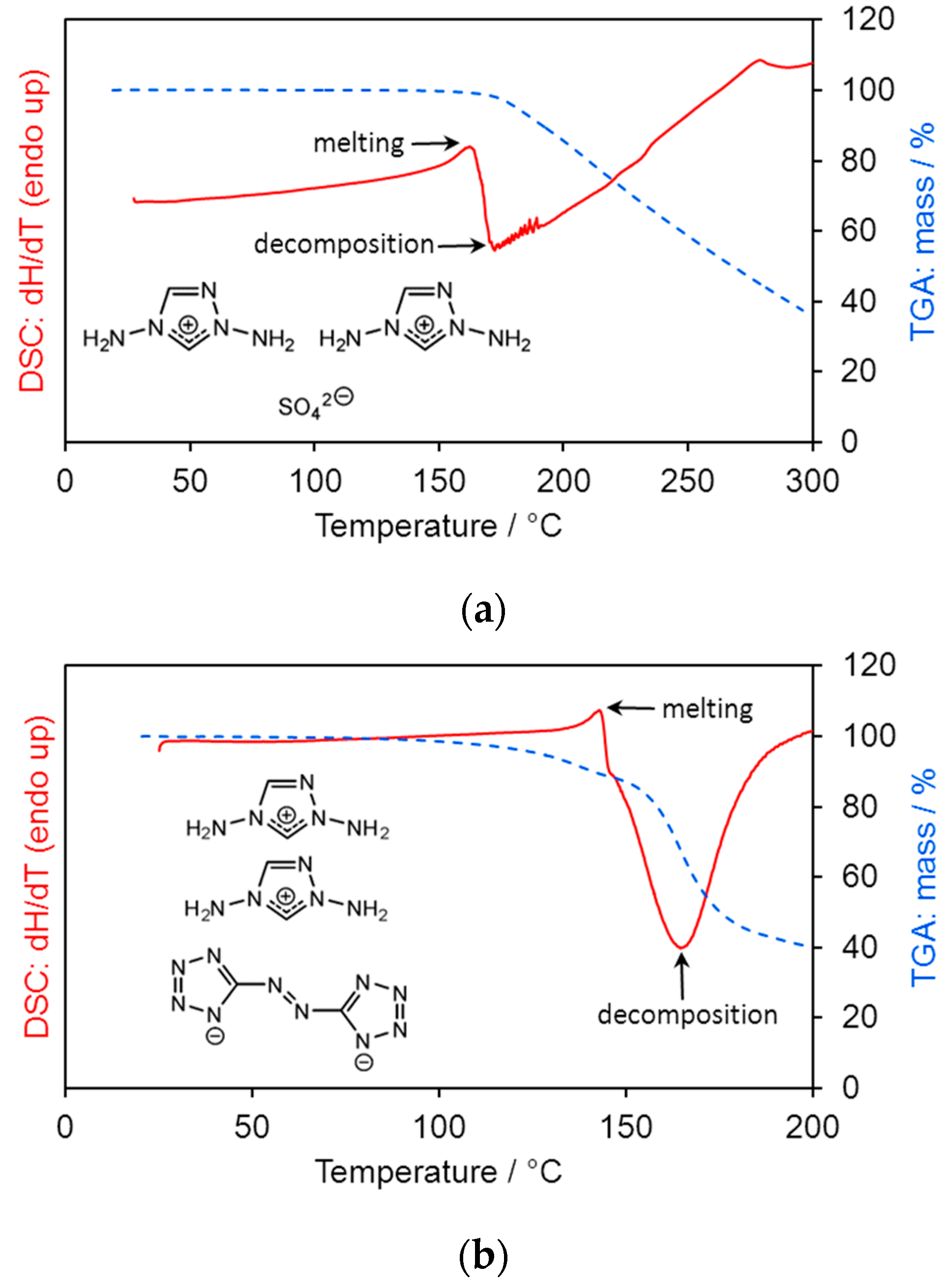
2.3. Differential Scanning Calorimetry (DSC) and Thermogravimetric Analysis (TGA)
3. Experimental Section
3.1. Bis(1,4-diamino-1,2,4-triazolium) Sulfate (1)
3.2. Bis(1,4-diamino-1,2,4-triazolium) 5,5′-Azotetrazolate (2)
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Link, H.; Klötzer, W.; Karpitschka, E.M.; Montavon, M.; Müssner, R.; Singewald, N. 1-Amino-and 1,3-Diaminoimidazolium Salts. Angew. Chem. Int. Ed. Engl. 1990, 29, 556–557. [Google Scholar] [CrossRef]
- Laus, G.; Kahlenberg, V.; Többens, D.M.; Jetti, R.K.R.; Griesser, U.J.; Schütz, J.; Kristeva, E.; Wurst, K.; Schottenberger, H. Lattice architecture and hydrogen-bonding networks of N-aminoazolium and N,N′-diaminoazolium chlorides. Cryst. Growth Des. 2006, 6, 404–410. [Google Scholar] [CrossRef]
- Thiele, J. Ueber Azo- und Hydrazoverbindungen des Tetrazols. Ann. Chem. 1898, 303, 57–75. (In German) [Google Scholar] [CrossRef]
- Laus, G.; Kahlenberg, V.; Wurst, K.; Schottenberger, H.; Fischer, N.; Stierstorfer, J.; Klapötke, T.M. Synthesis and crystal structures of new 5,5′-azotetrazolates. Crystals 2012, 2, 127–136. [Google Scholar] [CrossRef]
- Lampl, M.; Laus, G.; Braun, D.E.; Kahlenberg, V.; Wurst, K.; Fuhrmann, G.; Schottenberger, H.; Huppertz, H. New crystal structures in the realm of 5,5′-azotetrazolates. Z. Naturforsch. 2015, 70, 125–134. [Google Scholar] [CrossRef]
- Klapötke, T.M.; Sabaté, C.M. New energetic compounds based on the nitrogen-rich 5,5′-azotetrazolate anion ([C2N10]2−). New J. Chem. 2009, 33, 1605–1617. [Google Scholar] [CrossRef]
- Ye, C.; Xiao, J.-C.; Twamley, B.; Shreeve, J.M. Energetic salts of azotetrazolate, iminobis(5-tetrazolate) and 5,5′-bis(tetrazolate). Chem. Commun. 2005, 21, 2750–2752. [Google Scholar] [CrossRef] [PubMed]
- Laus, G.; Wurst, K.; Schottenberger, H. Crystal structure of bis(1,3-diaminoimidazolium) 5,5′-azotetrazolate, (C3H7N4)2 (C2N10), C8H14N18. Z. Kristallogr. New Cryst. Struct. 2012, 227, 293–294. [Google Scholar] [CrossRef]
- Laus, G.; Kahlenberg, V.; Schottenberger, H. Crystal structure of 1,3-diaminoimidazolium hexafluorophosphate, [C3H7N4]PF6. Z. Kristallogr. New Cryst. Struct. 2010, 225, 463–464. [Google Scholar] [CrossRef]
- Bentivoglio, G.; Schwärzler, A.; Wurst, K.; Kahlenberg, V.; Nauer, G.; Bonn, G.; Schottenberger, H.; Laus, G. Hydrogen Bonding in the Crystal Structures of New Imidazolium Triflimide Protic Ionic Liquids. J. Chem. Cryst. 2009, 39, 662–668. [Google Scholar] [CrossRef]
- Bernstein, J.; Davis, R.E.; Shimoni, L.; Chang, N.-L. Patterns in Hydrogen Bonding: Functionality and Graph Set Analysis in Crystals. Angew. Chem. Int. Ed. Engl. 1995, 34, 1555–1573. [Google Scholar] [CrossRef]
- Laus, G.; Klötzer, W. Synthesis of 1-amino-1H-1,2,4-triazoles. Synthesis 1989, 4, 269–272. [Google Scholar] [CrossRef]
- Hammerl, A.; Holl, G.; Klapötke, T.M.; Mayer, P.; Nöth, H.; Piotrowski, H.; Warchhold, M. Salts of 5,5′-azotetrazolate. Eur. J. Inorg. Chem. 2002, 4, 834–845. [Google Scholar] [CrossRef]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Laus, G.; Wurst, K.; Kahlenberg, V.; Schottenberger, H. Crystal Structures of Two 1,4-Diamino-1,2,4-triazolium Salts. Crystals 2016, 6, 13. https://doi.org/10.3390/cryst6010013
Laus G, Wurst K, Kahlenberg V, Schottenberger H. Crystal Structures of Two 1,4-Diamino-1,2,4-triazolium Salts. Crystals. 2016; 6(1):13. https://doi.org/10.3390/cryst6010013
Chicago/Turabian StyleLaus, Gerhard, Klaus Wurst, Volker Kahlenberg, and Herwig Schottenberger. 2016. "Crystal Structures of Two 1,4-Diamino-1,2,4-triazolium Salts" Crystals 6, no. 1: 13. https://doi.org/10.3390/cryst6010013
APA StyleLaus, G., Wurst, K., Kahlenberg, V., & Schottenberger, H. (2016). Crystal Structures of Two 1,4-Diamino-1,2,4-triazolium Salts. Crystals, 6(1), 13. https://doi.org/10.3390/cryst6010013






