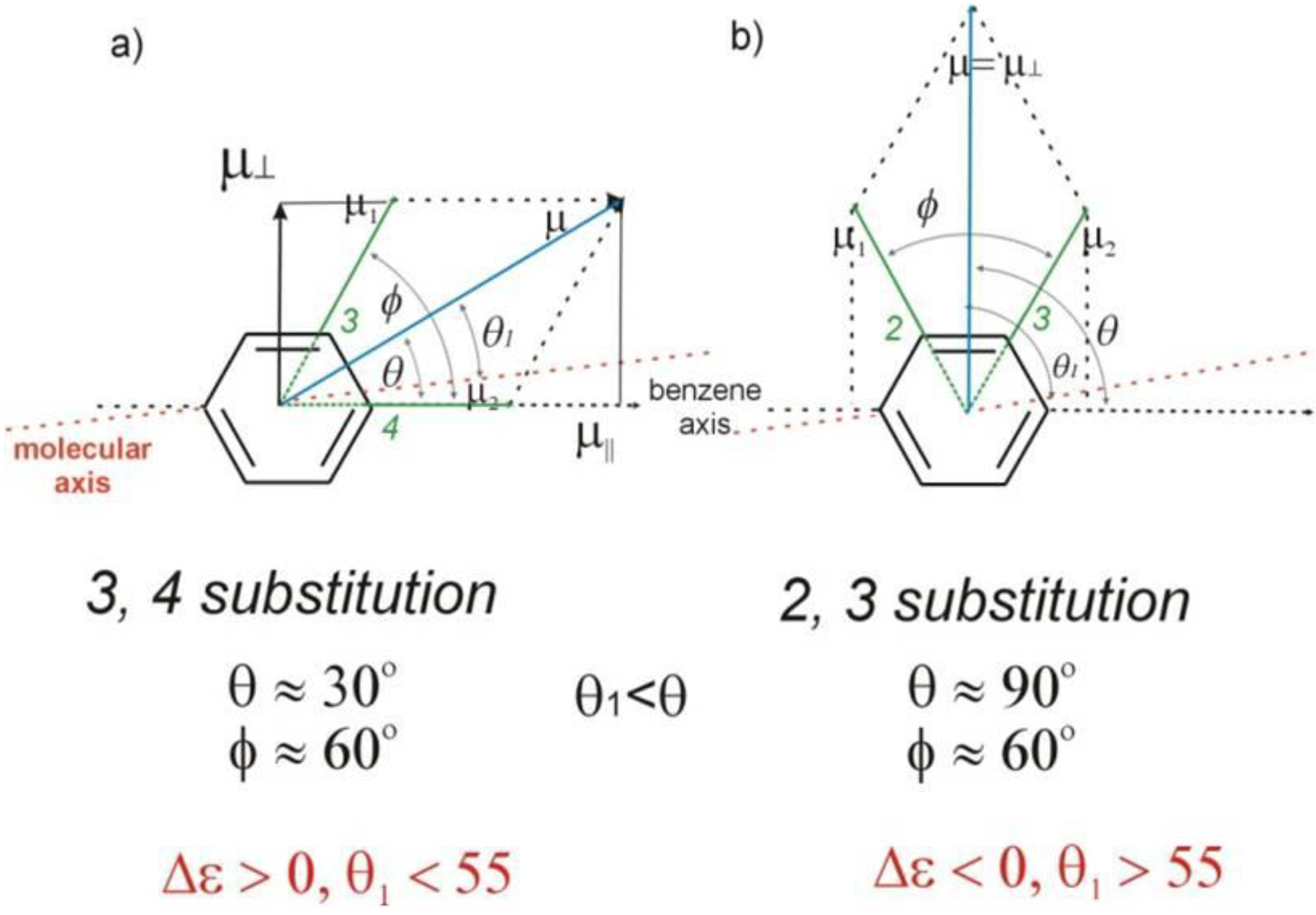
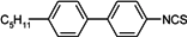
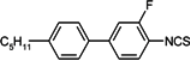
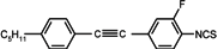
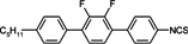
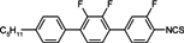
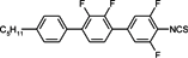
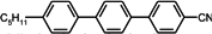
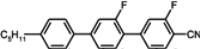
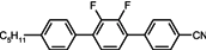
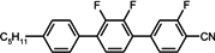
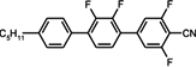
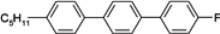
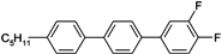
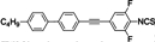
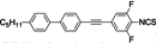
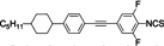
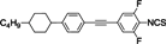
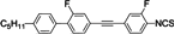
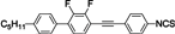
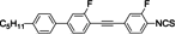
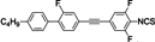
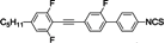
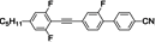
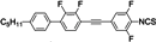
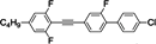
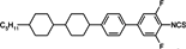
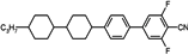
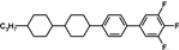
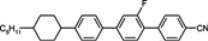
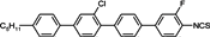
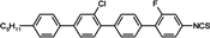
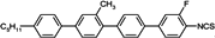
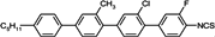
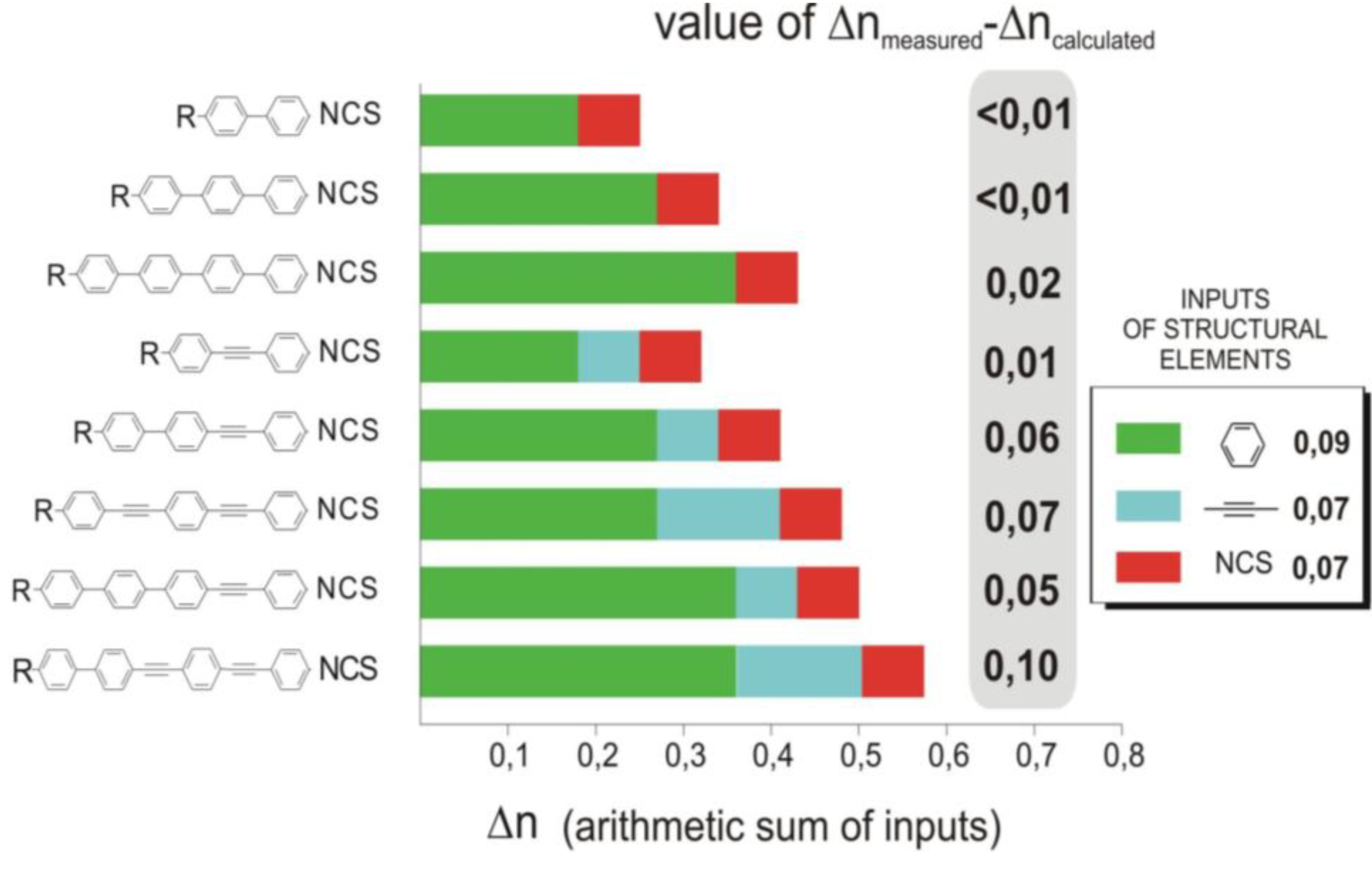
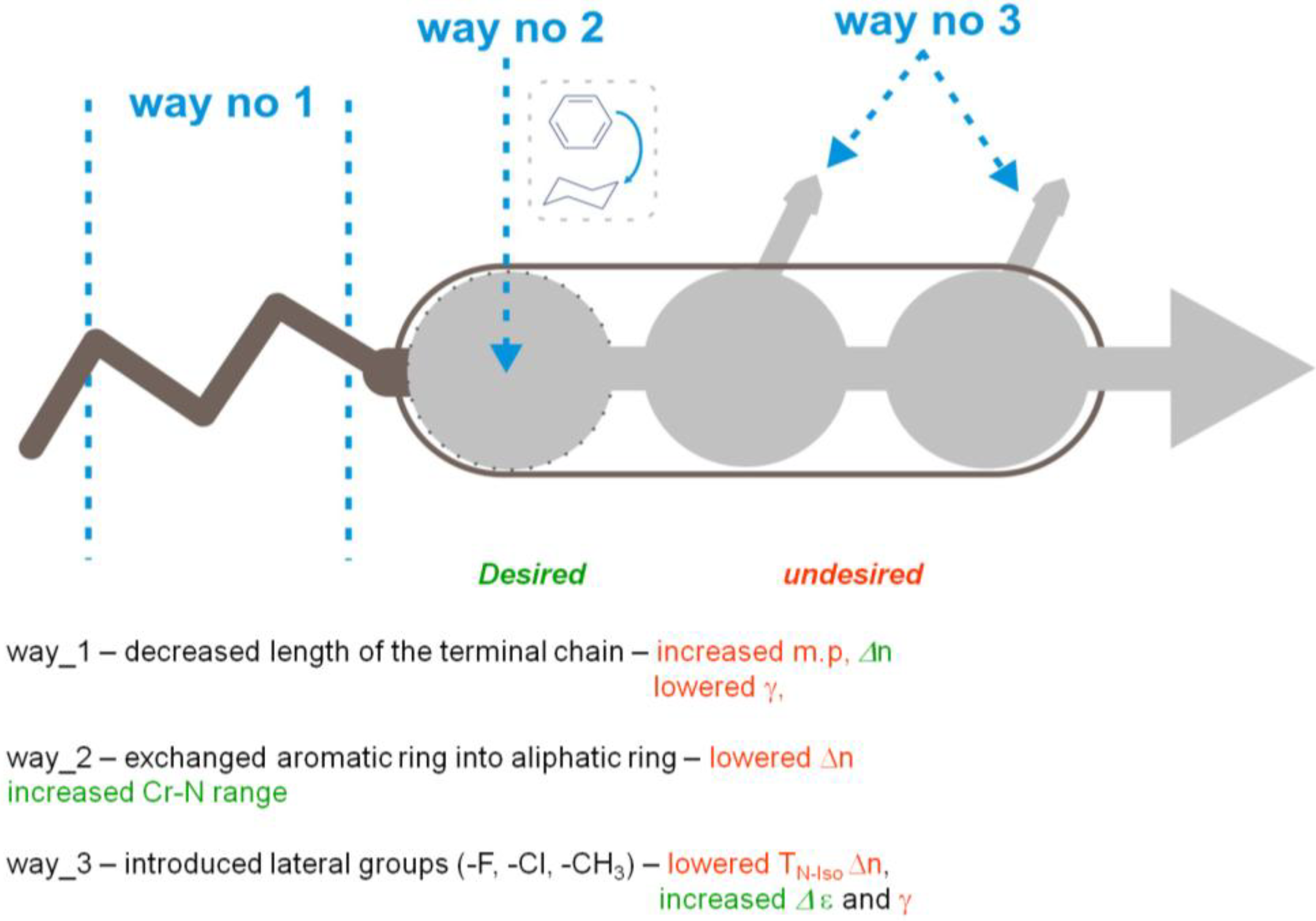
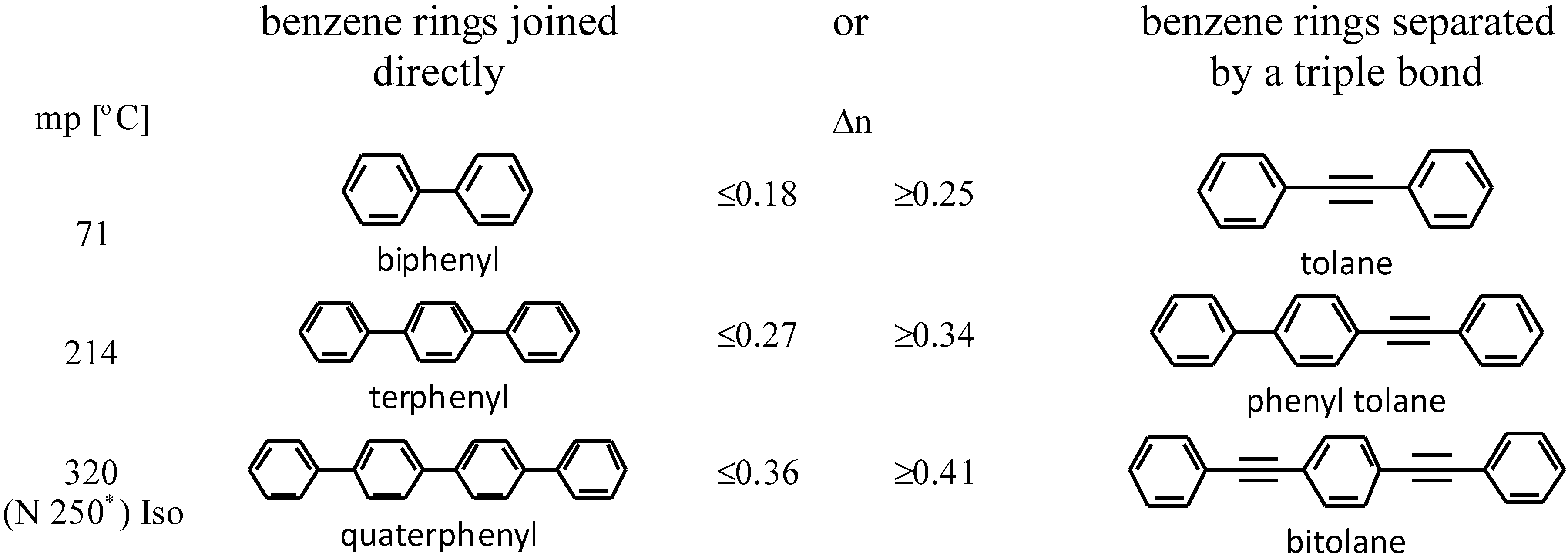
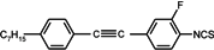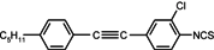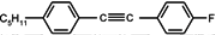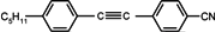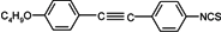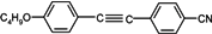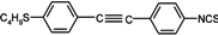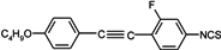The mesogenic and physical properties of two, three and four-ring aromatic compounds with the rings connected directly or via carbon-carbon triple bond will be reviewed to show structural correlations. We are reviewing the compounds with positive dielectric anisotropy (terminated with NCS or CN groups or F atom) and pay special attention to the isothiocyanato compounds as being one of the most important for formulation of large birefringence LC mixtures. These compounds have also been the object of our studies for many years. The compounds with negative dielectric anisotropy will be considered in the separate elaborations. The properties of many of them were also the subject of recent work presented by Hird [
71]. Homologous members with five carbon atoms in terminal chain (C
5H
11) or C
4H
9O including an oxygen atom were taken under consideration for the comparison of the properties of different liquid crystal families. They are thought to be the most representative ones. The compounds with index
n = 5 in the chain are marked only by a number (
1 …
101) while other homologous members are marked by adding the letter a, b, c, d, e and f for
n = 1, 2, 3, 4, 6 and 7, respectively; for example, compound
10 with the ethyl chain is marked
10b. The compounds with chains (H
2n+1C
n– and –C
mH
2m+1 or H
2n+1C
nO– and –OC
mH
2m+1) are marked as
10.
n.
m (10.1.2 for the methyl and ethyl group or the methoxy and ethoxy group, respectively).
3.1. Two-Ring Compounds
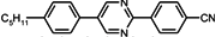
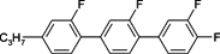
The characteristic examples of two-ring compounds with biphenyl core and its heterocyclic analogues are given in
Table 1.
Although they do not yet possess very large Δ
n, they are very important as necessary components of nematic mixtures. We have also been provided with more information about the properties of these compounds than what is available for multi-ring compounds. Among laterally unsubstituted alkylbiphenyl derivatives, only compound
3 terminated with the cyano group is the enantiotropic low-melting nematogen. Also, alkoxy analogue
4d and pyrimidine analogue
7 are nematic compounds, but their melting points are much higher and the nematic phase is monotropic only (observed during cooling below melting). The presence of a heteroatom inside the chain (oxygen) or nitrogen inside the ring increases the clearing point, but simultaneously, strongly increases viscosity (compound
4d has
γ = 370 mPa·s [
72], over three times higher than what compound
3 has). The birefringence increase is also noticeable but it is rather small. In the homologous series
3 and
4a, smectic A
d phase appears for longer chain (
n ≥ 8) [
73,
74].
For unsubstituted biphenyl isothiocyanate (compound
1) as well pyrimidine and tiophene analogues (compounds
6 and
9d), only smectic phases are observed in all ranges of mesophase (a higher ordered smectic E phase in compound
1, a smectic A
1 phase in compound
6 and a smectic B phase in compound
9d). Only smectic phase E is observed for homologous series
1 for alkyl
n = 2–10 [
75]. Also, fluoro terminated compound
5 is a smectic B. Virtual N–I transition temperatures (hypothetical
N–I transition if they are nematics) of compounds
1,
5 and
9d are 31 °C, −28.5 °C and 26 °C, respectively, estimated from the properties of nematic solution.
The compound
1 has the highest birefringence, even a little higher than compounds
9d with an easily polarized thiophene ring. Seed
et al. [
48,
72] gives the values 0.33 and 0.32 for compounds
1 and
9d, respectively, at 25 °C, while for reduced temperatures,
T/TN–I = 0.7815, Δ
n = 0.41 and 0.38. The compound
1 has a higher order parameter because its molecules are more linear than for
9d, wherein the thiophenyl ring is not coaxial with the benzene ring. Their value is higher than that obtained by extrapolation from the mixture with compound
8e. They used cyano mixture E7. It is known that in mixtures consisted of cyano and isothiocyanato compounds,
ε‖ and Δ
ε increase because the dimerization process is reduced [
76]; therefore, the E7 mixture is not appropriate for estimation of optical properties of isothiocyanato compounds.
Table 1.
Properties of biphenyl derivatives and their analogues at temperatures 22–25 °C, where η and γ represents flow and rotationalviscosity, respectively.
Substitution of compound
1 with one fluorine atom in position 3 (the closest to the isothiocyanato group) transforms this compound into nematic compound
2 with a lower clearing point (19.8 °C) than for cyano compound
3 (37.2 °C), simultaneously destabilizing smectic E phase. Substitution in the position 2 gives the compound with virtual
N–
I transition at temperature −15.4 °C [
85]. The compound
2 exhibits much higher birefringence and lower viscosity than cyano compound
3, so it acts as a better component for manufacturing a moderately birefringence mixture than cyano compound
3. Also, the transformation of the left benzene ring in the isothiocyanato biphenyl molecule into a cyclohexane unit totally depresses the smectic phase. The compound
8 is a monotropic nematic with a high enough melting point and clearing point, but it also has other polymorphic crystal forms that arise during crystallization from its nematic phase, which melts only at 30 °C. On the contrary, the next homologue of this series (compound
8e,
n = 6) has a very low melting point—only 12.5 °C—one of the lowest among polar compounds. Its clearing point is higher than cyano compound
3e. In the family of compound
8, a smectic phase does not appear in analogue
n = 12 [
69]. This compound also has very low shear and rotational viscosities. The compound
8e has high
k33 and
k11 values [
80,
81] but its ratio
k33/
k11 is lower than observed for cyano compound 5 and other cyano compounds. Especially, a low
k33/
k11 is observed for isothiocyanato pyrimidine 6,
k33/
k11 ≈ 0.5, see data in
Table 2.
Table 2.
Comparison of elastic constants [10
−12 N] for cyano- and isothiocyanato compounds at temperature
TN–I–
T = 10 [
80].
Table 2.
Comparison of elastic constants [10−12 N] for cyano- and isothiocyanato compounds at temperature TN–I–T = 10 [80].
| Compound No. | k11 | k22 | k33 | k33/k11 |
|---|
| 3 | 6.7 | 3.6 | 9.1 | 1.23 |
| 4d | – | – | – | 1.24 |
| 6 | 5.04 | 3.19 | 4.61 | 0.7 |
| 8e | 8.57 | 3.70 | 9.51 | 1.11 |
| 7 | 7.0 | 3.9 | 8.0 | 1.16 |
The values of elastic constants in isothiocyanates as well as in other compounds may be shifted to the higher level (more than two times) by introducing a double bond to the chain [
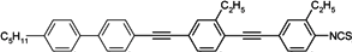
80,
81]. The value of ratio
k33/
k11 strongly depends on the distance of a double bond from the rigid core of a molecule. For example, among the family of compound
8, with a double bond in the alkyl chain, the ratio
k33/
k11 changes from 1.10 to 2.00 upon a distance of –CH=CH– group from the rigid core increases [
80]. In the case of cyano compounds, the observed difference is much larger; also, the position of the double bond in the terminal chain has much influence on the viscosity [
80,
81].
The compounds with an alkylsulphanyl and an alkylselanyl chain were prepared at Hull University [
48,
72,
86] in order to increase polarizability and birefringence. The replacement of the a butoxy chain in compound
9d into butyl sulphanyl (H
9C
4S–) lead to a small increase in both
ne and
no and Δ
n. The increase of n
o is explained by the increase in the molecular breadth, while the increase of
ne is explained by the presence of a much more polarizable sulfur atom which is conjugated to the aromatic core. The increase of the order parameter and birefringence for compounds with a butylselanyl (H
9C
4Se–) chain instead of a butyl sulphanyl one was also observed [
48].
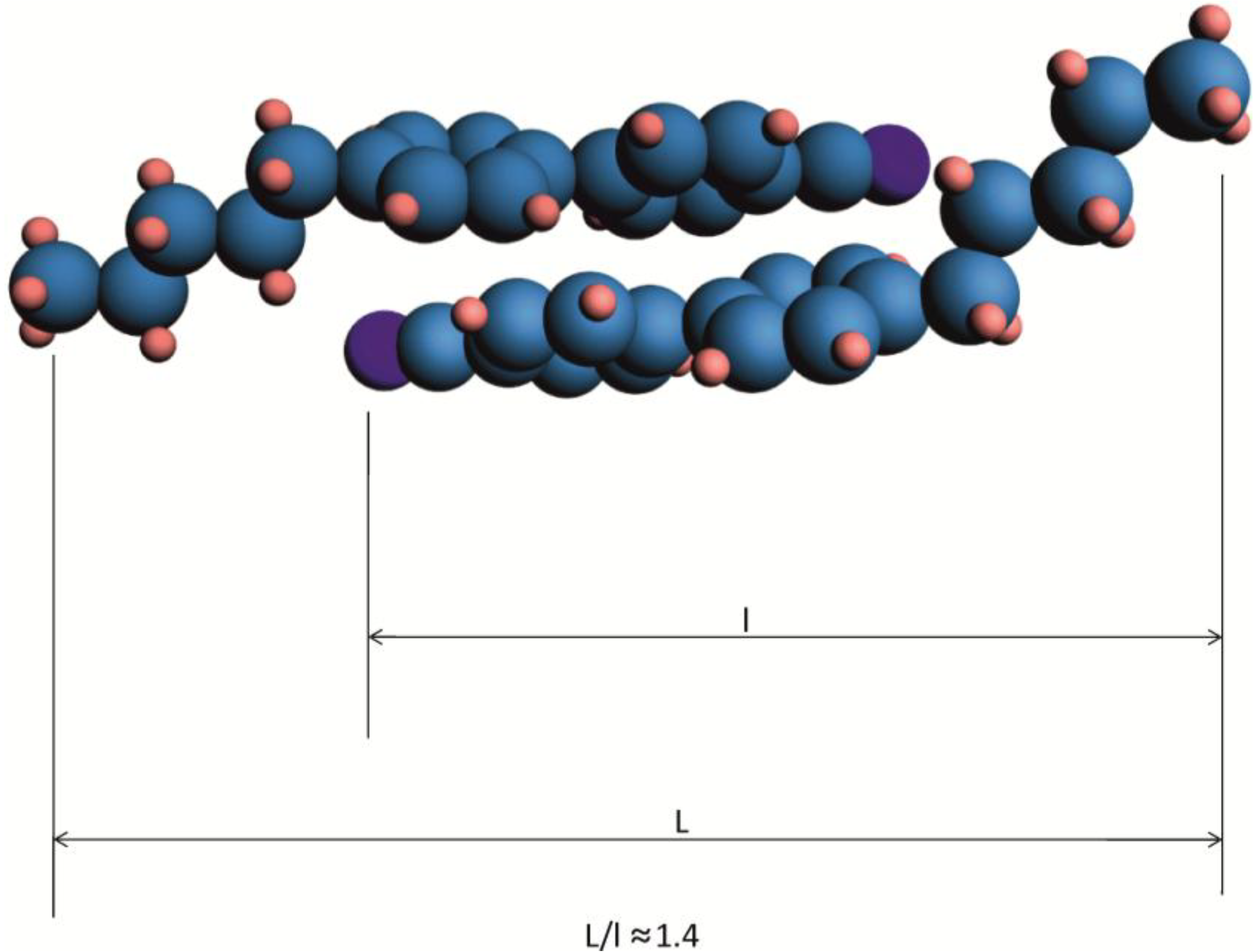
A low viscosity of fluorine was substituted in compound
2 and cyclohexane derivative 8 results from its monomeric character. In the nematic phase of compound
8e, X-ray diffraction pattern shows a small concentration of bodies with apparent length L/l of 1.2l [
70], while for CN compounds, high concentration of dimer with L/l ≈ 1.4 is observed [
87]. Substitution of compound
3 in the position close to the cyano group results in a strong increase in electric permittivity [
69], which tells us that dimerization disappears (L is the length of aggregate and l is the single molecule length).
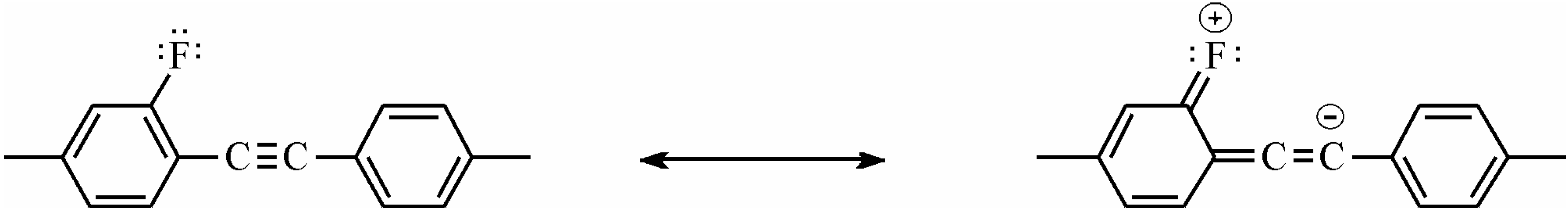
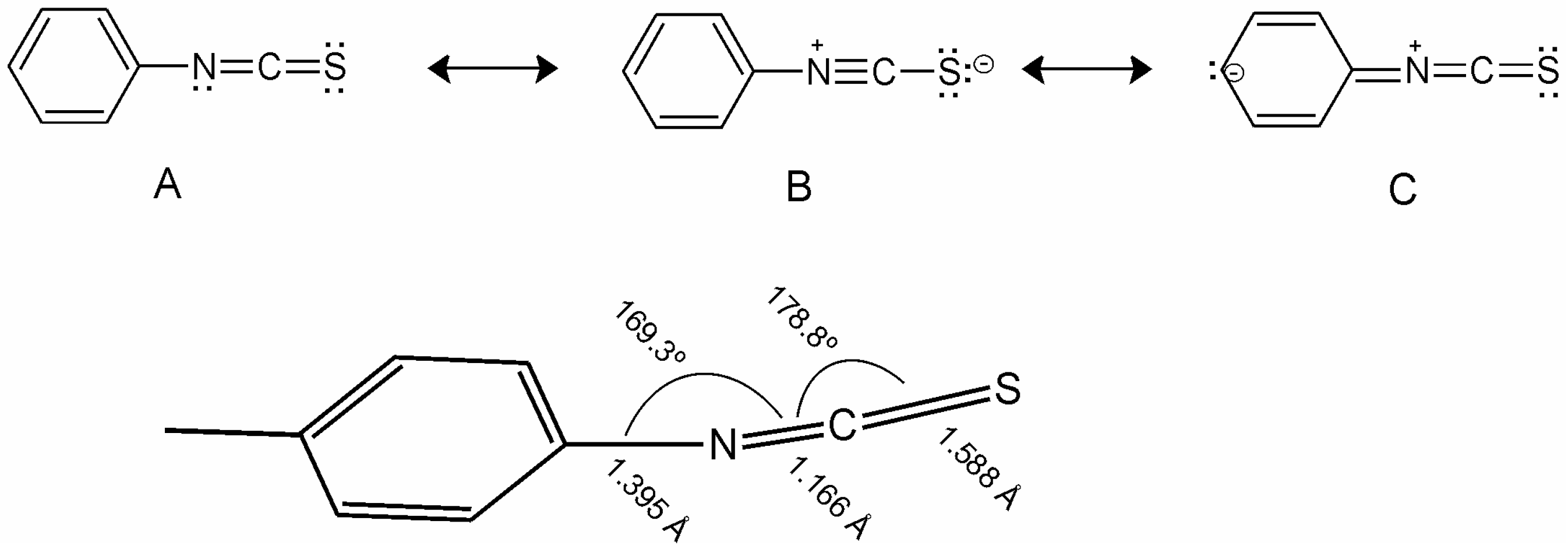
NCS group is coplanar with benzene ring plane and lies almost coaxially to it, see
Figure 10. From the monocrystal reflection investigations, the tilt values were obtained between the axis C–N–C bond 169.3 °C and N–C–S bond 179.9 °C, respectively [
88,
89]. The length of the bond between benzene ring and N atom is 1.395 Å, which is very close for a C–C standard aromatic bond. Resonance structures lead to coplanarity of the NCS group with benzene rings and average the C–N bond to the characteristic length for an aromatic bond (C–C, 1.389 Å), while the length of N–C bond is very similar to the triple bond length (C≡C, 0.121 Å).
Figure 10.
Resonance structures and geometry of NCS group bonded directly to the benzene ring.
Figure 10.
Resonance structures and geometry of NCS group bonded directly to the benzene ring.
For the phenyl NCS unit, the following resonance structures (
Figure 10) are considered [
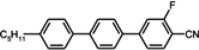
90], which are responsible for NCS conjugation with the benzene ring and are lying in the benzene plane. Laterally unsubstituted, two-ring alkyltolanes exhibit similar mesogenic properties to that observed in biphenyl derivatives, but the melting and clearing points are higher, see
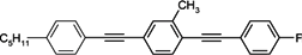
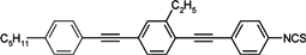
Table 3.
Table 3.
Phase transition temperatures [°C], melting enthalpies [kJ/mol] and birefringenceof alkyltolanes.
In isothiocyanato alkyltolanes (compound
10), only smectic E is observed, but its chain contains an even number of carbon atoms (
n = 4 and
n = 6) and at a lower temperature, smectic K phase is observed [
97]. The temperature of virtual
N–
I transition is 44.0 °C. Fluorine analogue (compound
15) is not mesogenic, while cyano terminated compound
16 with a monotropic nematic with a clearing point at 70.5 °C. Also, the introduction here of one fluorine atom into the benzene ring (compound
11) completely destroys the smectic E phase. A monotropic nematic phase appears for pentyl member (compound
11) while heptyl member (compound
11f) is already enantiotropic nematic. This is the general relation for tolane compounds, also observed among three ring compounds. Substitution of benzene ring in tolane core by a chlorine atom (compound
13) or by two fluorine atoms (compound
14) strongly depresses the clearing point. Two fluorine atoms decrease the clearing point with less than one chlorine atom. A three-component eutectic mixture, consisting of
11c,
11 and
11f, had viscosity (
η = 19 mPa·s and
γ = 88 mPa·s, Δ
n = 0.34 at 589 nm and 20 °C, melting point (−2 °C) and clearing point 40.1 °C) [
92]. Higher clearing point can be observed for isomeric compound
12, wherein fluorine is close to the triple bond. Alkoxy tolanes have higher clearing points than alkyl tolanes, see
Table 4.
Table 4.
Phase transitiontemperatures [°C], melting enthalpies [kJ/mol] and birefringence of alkoxytolanes and alkylsulphanyltolanes.
Unsubstituted 4′-butoxy-4-isothiocyanatotolane (compound
17d) has a high melting point and clearing point; similar behavior is observed for the cyano analogues
18d. However, the monotropic smectic E phase and enantiotropic nematic phase is observed for
17d, which appears in a narrow temperature range. Analogous alkylsulphanyl derivative
19d possess only smectic B phase, but its melting and clearing temperatures are lower than these observed in alkoxy derivative.
N–I transition is lower by about 50 Kelvins. Cyano derivative
20d is a monotropic nematic compound with low clearing point. The exchange of alkoxy chain into analogue alkylsulphanyl chain involves a significant decrease in clearing points [
48].
Seed
et al. [
48] found for compound
19d and
20d Δ
n values 0.49 and 0.29, respectively (0.5 and 0.34 at
T/
TN–I = 0.7815). It seems that the first value is too large, probably because of the host used, which was an E7 mixture.
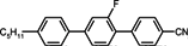
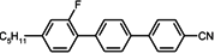
The placement of a lateral substituent such as fluorine, chlorine or methyl group totally removes the smectic phases in alkoxytolanes [
93,
94], but simultaneously, the clearing points decrease as follows: Cl < CH
3 < F, comparing
TN–I temperatures of compounds:
23d,
24d,
21d. It is a common relation found also in multi-ring compounds. The tolanes with alkoxy terminal chain generally have higher clearing points, higher viscosity and very often, higher melting points than alkyl terminated derivatives. Due to their high clearing points, they may be used as the main components of medium birefringence mixtures (Δ
n ~ 0.3) without or only with a small number of three-ring compounds. Another general rule validated for tolanes is the following: compounds with fluorine next to the triple bond have higher clearing points than those with the fluorine atom positioned further from the triple bond (compare compounds
22d and
21d or
25d). The last compound with fluorine close to the butoxy group has a lower
TN–I of 18 Kelvin, but chlorine in this position influences the
TN–I in the opposite way, with compound
26d having a higher
TN–I than compound
23d. Dielectric anisotropy of tolanes with fluorine in the neighborhood of the triple bond is lower than those with fluor pending in the same directions, but at a long distance from the triple bond [
100]. Refractive indices of differently substituted alkyl and alkoxy isothiocyanato tolanes are compared at temperature
TN–I–
T = 10 °C in
Table 5.
Table 5.
Birefringence and refractive indices of alkyl and alkoxy isothiocyantotolanes at reduced temperature TN–I = −10°C.
Table 5.
Birefringence and refractive indices of alkyl and alkoxy isothiocyantotolanes at reduced temperature TN–I = −10°C.
| No. | Δn | no | ne |
|---|
| 11 | 0.2928 | 1.5674 | 1.8602 |
| 13 | 0.2828 | 1.5964 | 1.8792 |
| 21d | 0.3121 | 1.5574 | 1.8695 |
| 22d | 0.2963 | 1.5615 | 1.8682 |
| 23d | 0.2618 | 1.5769 | 1.8392 |
| 24d | 0.2508 | 1.5734 | 1.8242 |
| 25d | 0.2890 | 1.5720 | 1.8582 |
| 26d | 0.2768 | 1.5974 | 1.8742 |
Chloro-substituted compounds 23d and 26d as well as methyl substituted one 24d have lower birefringence than analogous fluorosubstituted compounds 21d and 25d. For the first two compounds, a decrease of Δn results from a higher ordinary index no (chlorocompounds). The compound 24d has no a little higher and ne little lower.
Bulky substituents make the molecules broader and they become more polarizable in the direction perpendicular to the long axis. The same is observed for sulphanyl derivatives 18d and 19d.
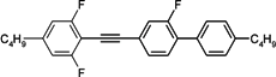
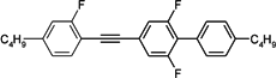
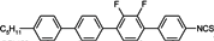
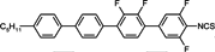
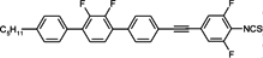
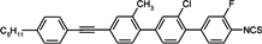
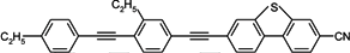
3.3. Four-Ring Compounds: Quaterphenyls, Phenylethynyl Terphenyls
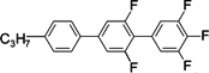
Di-, tri- and tetra-fluorosubstituted isothiocyanato quaterphenyls have smectic A phase in a very broad temperature range. Melting points are also high for them, see
Table 14. Clearing temperatures are very high (close to 400 °C) for di-substituted compound
97.
TN–I,
Tm and
TSmA–N temperatures systematically decrease with an increase in the number of fluorine atoms in the lateral position of molecules, but they are still above an acceptable level even for tetra substituted compounds. Also, quaterphenyls
100 and
101 terminated with fluorine atom and having additionally three or four fluorine atoms in the lateral positions are smectics A with
TSmA-N transition at 195 °C and 166 °C, respectively. Similarly structured compound
102, with CF
2O bridge in the core on the right side, has a distinctly lower melting point and stability in its smectic A phase. Unfortunately, the CF
2O bridge breaks conjugation among the aromatic rings and decreases Δ
n values to the level 0.27. Other property changes are very desirable [
85]. Rotational viscosity
γ is lower than that observed for other such large molecules and dielectric anisotropy Δ
ε increases [
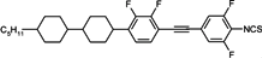
85]. Shortly, they are very useful components for formulation of the medium birefringence mixtures with high photochemical stability. Total depression of the smectic phase is obtained through the exchange of biphenyl unit for bicyclohexyl unit, see isothiocyanato compounds
105c and
105 [
101]. With this move, low melting compounds with an extremely broad range of nematic only phase are obtained with high Δ
n ~ 0.35–0.4, which is involved by way of the high order parameter of these compounds. Another interesting feature of such compounds is also its strong dependence on permittivity constant
ε‖ upon frequency, which makes them excellent components for dual frequency mixtures [
120]. Fluoro terminated compound
106c melts at higher temperatures but still at an acceptable level, while compounds
104c and
103c with cyano terminal group melt at unacceptably high temperatures. Such features are observed not only for compound
104c, but also for slightly different analogue substitution [
121].
Only nematic phase is present in quaterphenyl derivatives
107–
112 laterally substituted by more bulky moiety such as chlorine atom or methyl group [
117].
Table 14.
Phase transition temperatures [°C], melting enthalpies [kJ/mol] and birefringence of four-ring compounds.
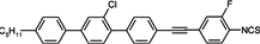
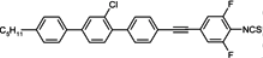
Compounds with one chlorine atom (
107,
108) or one methyl group (
109) in the lateral central position still have high clearing points, while the simultaneous presence of both these groups reduces melting point, and also rapidly, the
TN–I point. According to the authors’ knowledge, compound
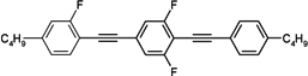
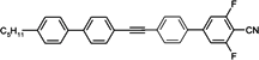
108 is the lowest melting alkyl isothiocyanato quaterphenyl obtained up to now. We also found that, more interesting than quaterphenyls, are their four-ring analogues with the triple bond positioned inside the core, see compounds
113–
118, see
Table 15.
Except for cyano compound 118, they show similar low melting temperatures and stronger nematogenic character than isothiocyanato quaterphenyls 110–112, simultaneously their Δn is shifted to a higher range 0.53–0.55 and Δn increment of ~0.05 is observed as a result of the improvement in conjugation.
Table 15.
Phase transition temperatures [°C], melting enthalpies [kJ/mol] and birefringence of four-ring compounds with the triple bond.
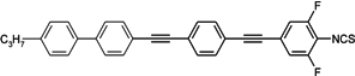
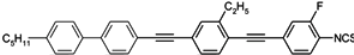
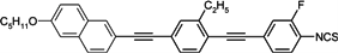
Four-ring diacetylenes substituted only by fluors have high melting points and smectic A phases observed in broad temperature ranges (see compound
119c,
Table 16). Moderately low melting temperatures are observed only for compounds with two ethyl groups, see compounds
120,
121 [
123,
124]. They have large Δ
n ~ 0.64–0.67 values, but also very high melting enthalpies in both cases. Liquid crystal
122b with thiofluorene (dibenzothiophene) moiety is found to be the compound with the highest birefringence reported so far (Δ
n = 0.79), see
Table 16. It is the cyano derivative, so the isothiocyanato analogue should have an even higher Δ
n value. Napthalene derivatives such as compound
123 have only nematic phase in the presence of an ethyl lateral group [
125,
126].
Other heterocyclic derivatives with benzotiazole and dibenzothiophene moiety were prepared by Sekine
et al. [
125,
126], and they obtained compounds reaching Δ
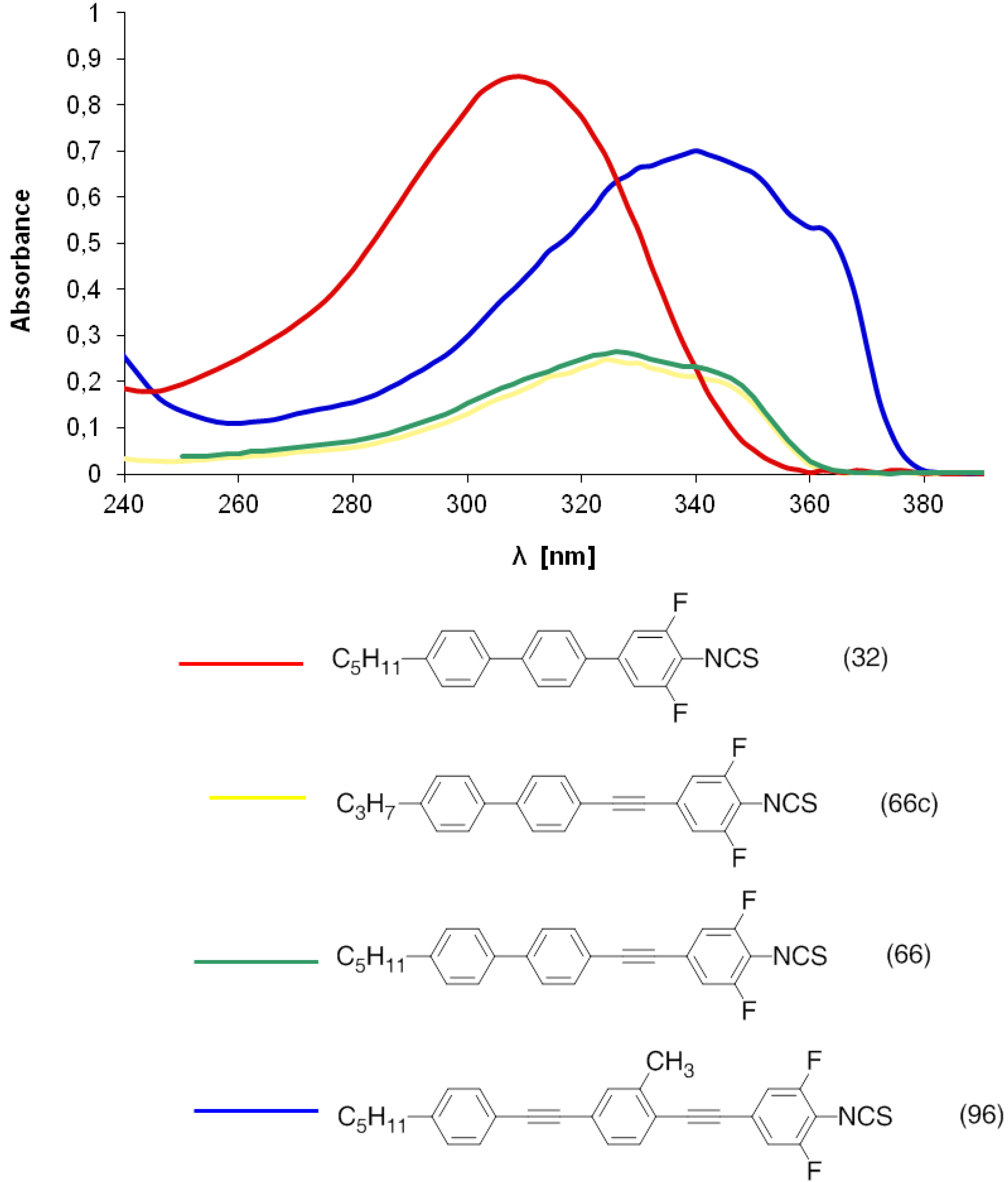
n = 0.6, but which have unfavorably high melting points and additionally, the absorption of light is observed at 300–400 nm.
Table 16.
Phase transition temperatures [°C], melting enthalpies [kJ/mol] and birefringence of four-ring diacetylenes.




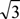 λ/2. The larger Δn is, a smaller cell gap (d) may be used and faster operating devices can be obtained, see Equations (4) and (5).
λ/2. The larger Δn is, a smaller cell gap (d) may be used and faster operating devices can be obtained, see Equations (4) and (5).




 .
.
 .
.