1. Introduction
A continuing increase in the electrical energy demand, which has been observed in recent years, altogether with rapid industrialization, has led to advanced research of more efficient energy storage solutions. On the other hand, excessive consumption of natural resources (oil, coal, gas) is not in accordance with European Union climate policy. The development of highly advanced energy storage technologies may reduce fossil fuel consumption and the greenhouse effect. Efficient storage of electrical energy would also prevent power outages, observed all over the world [
1,
2]. The most important modern energy storage devices are lithium-ion batteries and supercapacitors. There are some constructional differences between both groups; however, in recent years, the technological progress has blurred these differences. Generally, the main feature of supercapacitors, contrary to lithium-ion batteries, is the possibility of fast charging/discharging, and hence the quick delivery of huge energy within a short time. Consequently, the main advantages are a high power density, a long cycle life, relatively simple construction from easily accessible materials, eco-friendliness (no disposal issues), and high coulombic efficiency. Electrochemical supercapacitors are promising energy storage materials that, due to a long cycle life and high power density, may be applied in many areas of everyday living, i.e., back-up memories, traffic warning signals, security and alarm systems, smoke detectors, engine starting and acceleration, and load lifting [
3]. The perspective electrode materials for supercapacitors should meet numerous requirements, like high electrical conductivity, chemical inertness, gas or liquid adsorption permission, a relatively high surface area, and state of material, whether it is powder, fiber, or monolith. Regarding these criteria, active carbons seem to be the most adequate materials. Within applied electrolytes, aqueous, organic, or ionic liquids can be found. Each group has their pros and cons. Aqueous electrolytes, either alkali hydroxides or sulfates (most commonly used), are relatively cheap and easily available, but the drawback is a voltage window limited to 1.2 V. Organic electrolytes show a broader stability range (2–2.5 V) but suffer from flammability, volatility, and toxicity. Ionic liquids may give a voltage window as high as 4 V, but their preparation is complex and expensive, and still insufficient for industrial applications.
The capacitance of supercapacitors is proportional to the surface area of the electrode material. The higher the specific surface area, the higher the charge contact interface; therefore, chemically and electrochemically inert carbons are considered the best choice for matrices [
4]. Further increases in capacitance may be achieved through intercalation of an active species like conducting polymers, metal nanoparticles, and metal oxides. Such composites containing, e.g., metal oxides on carbons are generally classified as conversion electrodes, with the capacitance arising from typical charge separation and pseudocapacitance caused by Faradaic reactions [
5]. Redox reactions between electrolytes and metal oxides can significantly increase the specific capacitance. Within many proposals of metal oxide–carbon composites, transition metal oxides are privileged. Transition metals may offer several possible oxidation states. Consequently, during charge/discharge cycles, such a material is able to store many more electrolyte ions. Active carbon is mainly applied in supercapacitors; however, other porous carbons with a high specific surface area are often examined as potential competitors. One such example is expanded graphite with high electrical conductivity up to 10
8 S/cm, a material with high corrosion resistance that can be obtained at a low cost [
6]. Data concerning the application of expanded graphite with the intercalated metal species are presented in
Table 1.
Refractory metals like molybdenum, tungsten, and rhenium seem to be very attractive prospective components of supercapacitor electrodes, which is mainly attributed to oxidation states from +2 to +6 and numerous polymeric-like structures [
15,
16]. These groups, including oxides and oxo-hydroxides of tungsten, molybdenum, and rhenium, were marginally examined. Combinations of active carbon with ammonium perrhenate allowed us to produce an electrode material with an enhanced specific capacitance around 60 F/g [
17]. Similar results were obtained by reacting multiwalled carbon nanotubes with potassium permanganate and ammonium perrhenate [
18]. ReS
2 was also examined as a potential pseudocapacitive material, presenting high coulombic efficiency and a dramatic increase during the initial hundreds of cycles [
19]. A material with a high specific capacitance was produced by a combined ternary hybrid of MoS
2–ReS
2/rGO [
20]. Rhenium sulfide-based structures may result in a specific capacitance in the range from 85 F/g up to 190 F/g depending on the synthesis method (either sonochemical or hydrothermal) [
21,
22]. Rhenium nanoparticles decorated on biomass-derived active carbon were tested in applications of electrochemical sensing and supercapacitance, resulting in a high energy density and good long-term stability [
23]. Owing to the huge irreversible capacitance of lithium rhenium oxide, it was proposed as a good candidate for positive activated carbon electrodes of a lithium-ion capacitor to be used for pre-lithiating the graphite negative electrode [
24]. The biggest drawbacks of rhenium compound application are the limited availability of rhenium and its high market price in comparison to other metals of interest like nickel, cobalt, lithium, manganese, zinc, and iron. However, despite these facts, the interest in rhenium application can still be found in the literature and new ideas of its potential application are under review. In this report, the possibility to produce rhenium-doped carbon electrodes was examined using popular rhenium compounds synthesized within the progress of RenMet project activities. The center of Hydroelectrometallurgy at LUKASIEWICZ—IMN, Poland, is a highly qualified R&D group specialized in rhenium chemistry, including the recovery of rhenium from various materials and the production of a vast portfolio of rhenium compounds.
2. Materials and Methods
2.1. Carbon Matrix
Thermally expanded graphite was produced through thermal treatment of graphite oxide. It was chosen as a carbon matrix for the intercalated active species. Although structural defects generated in thermal shock significantly limit the Brunauer–Emmett–Teller (BET) specific surface area, expanded graphite has a much bigger pore size in comparison to other modified carbon precursors, which is an important factor regarding the intercalation of large perrhenate molecules [
3]. Synthetic graphite was oxidized by the Hummers method mixing 10 g synthetic graphite with 230 cm
3 95–97% sulfuric acid (Acros Organics, Geel, Belgium) in a round-bottomed flask equipped with a reflux and thermometer, and ice-cooled at a temperature close to 273 K [
25]. Then, 5 g sodium nitrate and 30 g potassium permanganate were added. The produced graphite oxide slurry was diluted with deionized water, washed several times by decantation, vacuum-filtrated using two filter papers, and dried at 110 °C. The produced graphite oxide powder was placed in a tubular furnace purged with inert gas (N
2). The temperature was increased above 700 °C for 10 min. It allowed for the desorption of the oxygen-containing groups and the expansion of the material to give a fluffy, light carbon powder (EXPANDED).
2.2. Metal Salt
Cobalt(II) perrhenate was chosen as a potential candidate for the energy storage material. It was synthesized based on the following equation:
A total of 50 cm3 perrhenic acid at a concentration of 200 g Re/dm3 was mixed with a cobalt(II) nitrate(V) solution in 10% stochiometric excess. The solution was vigorously mixed and evaporated at 90 °C. The solid product was dried at 120 °C for 3 days to produce a constant mass violet product.
2.3. Electrode Preparation
Experiments were carried out using a two-electrode symmetric system with electrolytic nickel current collectors and with electrode materials pasted on it. Each electrode was composed of 0.08 g of expanded graphite mechanically mixed in an agate mortar with 0.02 g of cobalt(II) perrhenate and 0.02 g of polytetrafluoroethylene. When pasted on the current collectors, a membrane (micro glass fiber paper, MGC, Ahlstrom Munksjo) soaked with 6 M KOH was used as a separator. A high-precision balance (Mettler Toledo AB 204 S, Columbus, OH, USA) was used to weigh the mass of electrode materials.
2.4. Electrochemical Characterization
The whole assembly was pressed in a poly(methyl methacrylate) casing. One current collector was mounted with the working electrode while the latter had both reference and counter electrodes. An Autolab PGSTAT 302 N workstation (Metrohm, Herisau, Switzerland)was used to perform cyclic voltammetry (to evaluate the cycle life of the electrode) in a potential window of 0–1 V at a scan rate of 500 mV/s, with galvanostatic charge/discharge (GC) at a current density in the range of 0.1–2 A/g (to evaluate the specific capacitance), and the electrochemical impedance spectroscopy (EIS) in the frequency range of 100 kHz–100 mHz, with the amplitude of sinusoidal voltage signal equal to 10 mV (to determine the resistance of the electrode processes and the charge transfer characteristics).
2.5. Characterization
Elemental analysis was performed using atomic absorption spectroscopy (FAAS—iCE 3000 Series AA spectrometer, Waltham, MA, USA, Thermo Scientific, Miami, OK, USA), while quantitative analysis was performed using X-ray diffractometry (Rigaku MiniFlex 600 equipped with a Cu tube as a source of Kα radiation with a wavelength 1.5406 Å). The specific surface area was calculated using the Brunauer–Emmett–Teller method. Pore size distribution and the mean values were calculated based on the adsorption part of the hysteresis loop using the Barrett–Joyner–Halenda (BJH) method. Morphology was analyzed by using a scanning electron microscope (SEM) (NOVA NanoSEM 200 (FEI Company, Hillsboro, OR, USA)). Thermogravimetry (TG) and (DSC) differential scanning calorimetry were performed in a nitrogen atmosphere with a temperature increment of 10 °C/min up to 1000 °C in corundum crucibles, using NETZSCH STA 449F3 (NETZSCH-Gerätebau GmbH, Selb, Germany).
3. Results
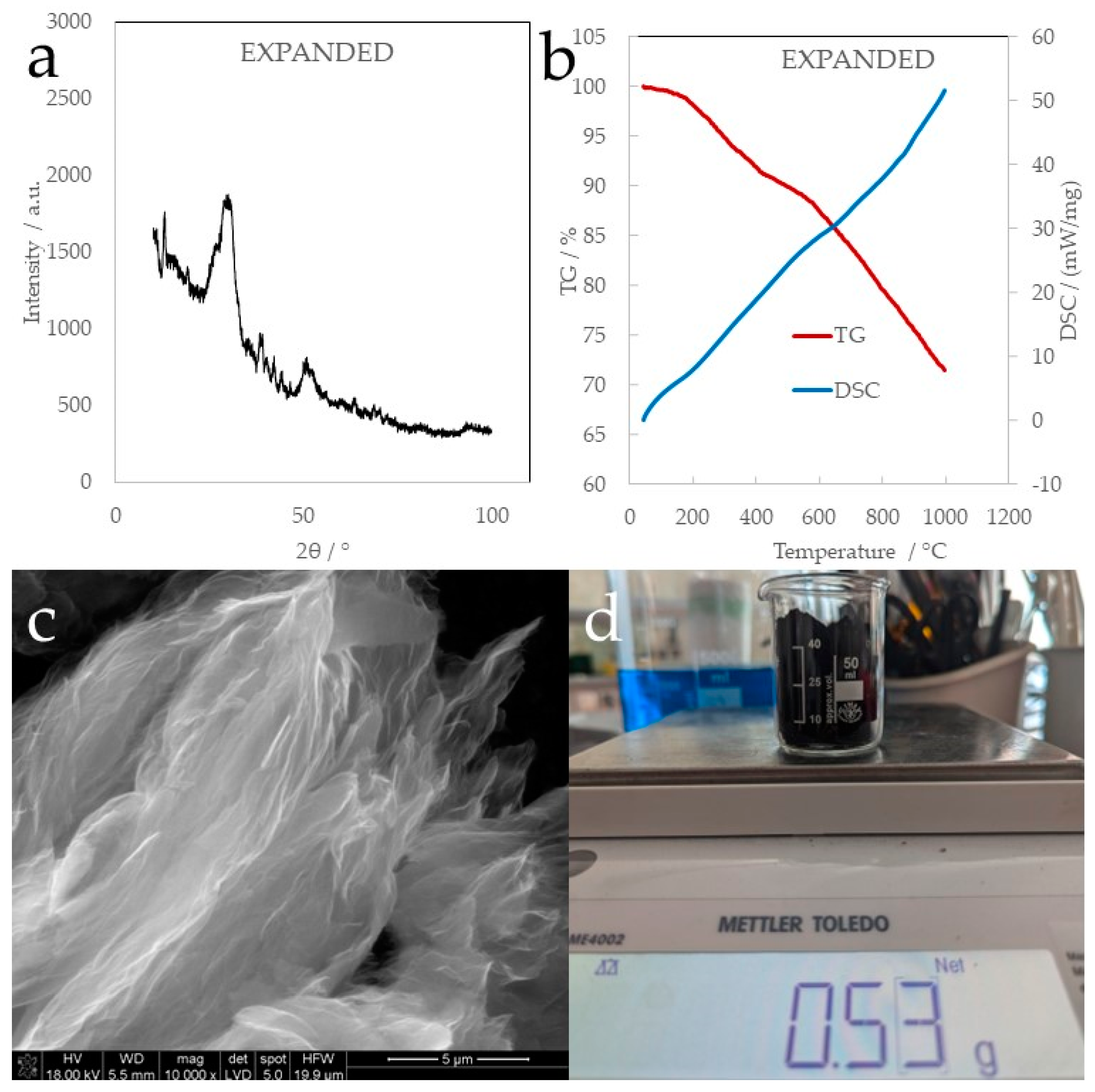
The expanded graphite that was produced by thermal shock in an inert gas flow has a strongly disordered structure with small packets of multiple graphene layers. This is a consequence of the extremely high pressure exerted on graphene layers by the desorbing gases. Consequently, there is no characteristic strong graphitic signal at 26°, but rather a small intensity and wide peak that can be attributed to the highly corrugated amorphous structure of EXPANDED (
Figure 1a). Thermogravimetric analysis and differential scanning calorimetry were used to characterize the thermal stability of the examined materials, while DSC allowed us to observe the thermal effects accompanying the dehydration, degassing, and thermal decomposition of samples. The material showed a 28 wt.% mass loss, which can be attributed to the desorption of the residual oxygen interlayer functionalities and the decomposition of smaller intralamellar heteroatomic packets (
Figure 1b). The SEM image (
Figure 1c) shows the highly corrugated structure of expanded graphite satisfying the high deformation of lamellar graphite oxide. Additionally, the specific surface area (BET) of EXPANDED was as high as 278 m
2/g, with a pore volume of 0.59 cm
3/g and pore size of 12.22 nm. A corrugated structure with big voids and interlamellar distances, in fact, can be an advantage for the accumulation of big anions like perrhenate.
Qualitative analysis of synthesized perrhenate revealed that the cobalt species was in fact a mixture of cobalt bis(rhenate(VII))—Co(ReO
4)
2 (01-089-6940)—commonly known as cobalt(II) rhenate(VII) or cobalt(II) perrhenate, and cobalt rhenium oxide hydrate—Co(H
2O)
4(ReO
4)
2 (01-088-1324). The diffraction pattern is presented in
Figure 2.
The synthesized material was crystalline with a major fraction of cobalt bis(rhenate(VII)); however, two signals around 16.6° and 25.5° were attributed to a hydrated form of cobalt salt.
Quantitative analysis of the obtained cobalt rhenium species showed that the contents of cobalt and rhenium were 10.4 wt.% and 66.8 wt.%, respectively, which was in line with the stoichiometry results: 10.5 wt.% and 66.5 wt.%, respectively. The contents of the contaminants were <10 ppm Na, <5 ppm Mg, <10 ppm Al, <10 ppm K, <10 ppm Ca, <10 ppm Fe, <5 ppm Cu, <5 ppm Ni, <5 ppm Co, and <10 ppm Zn.
Electrochemical analysis of the synthesized material showed the gradual improvement in the reversibility. In the first three cycles, relatively narrow curves with oxidation and reduction peaks were observed (
Figure 3). Then, it started to resemble a box shape, typical for supercapacitors. The final cycle is represented by a more or less rectangular-like curve, not very typical for metal oxides or other metal salts used in supercapacitors. The electrochemical stability of this material after 1000 cycles was as high as 80%.
EIS analysis showed a low resistance combined with electrode processes, with a mere 1 Ω of real impedance at the maximum (
Figure 4), which is a very good result as the other tested perrhenates resulted in values of 200–1000 Ω. These electrochemical tests proved that cobalt(II) perrhenate can be a perspective material for energy storage devices.
Mechanical intercalation of cobalt(II) perrhenate led to a synergistic effect on the specific surface area, as it increased by 22% to 339 m
2/g, while the parameter of pore volume was 0.61 cm
3/g and the pore size was 13.71 nm. Expanded graphite and co-modified expanded graphite were electrochemically analyzed to show their characteristics and advantages for Co(ReO
4)
2 application. Cyclic voltammetry was used to specify the charge/discharge behavior, emphasizing the loss in the specific capacitance between the first and last work cycle. Supercapacitors should be quickly charged and discharged, with a box-like shape of their CV curves. The curves obtained for the examined carbon matrix and composite are presented in
Figure 5.
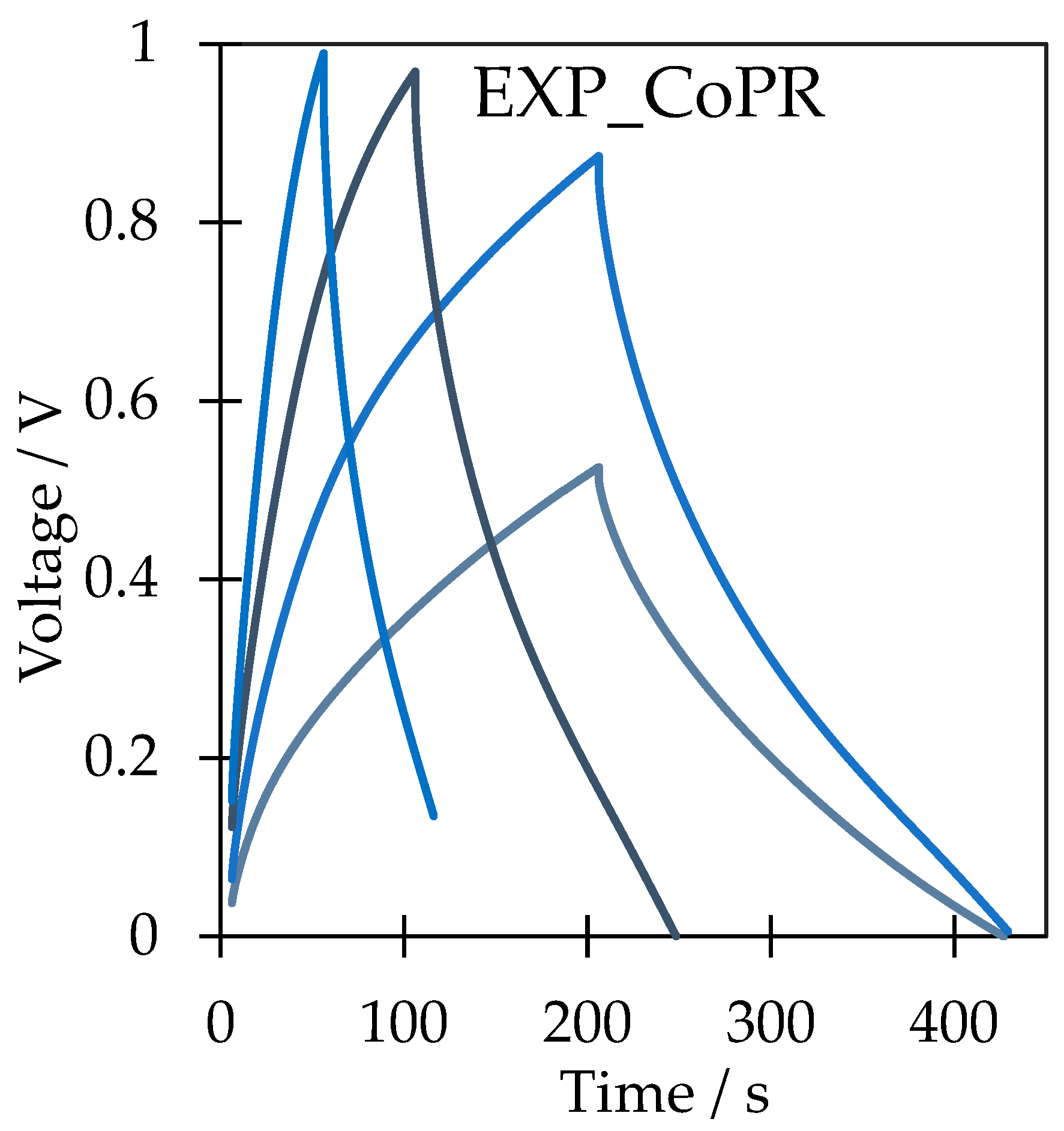
Cyclic voltammetry showed that in both cases, the shape of the curves is not ideal, with uneven charge and discharge behavior. Reversibility of the charge/discharge processes was poor in bare expanded graphite, resulting from the residual oxygen-containing groups reacting with the electrolyte and the worse wettability of the interlayer spaces. The low stability is also attributed to a shift in the oxidation–reduction curves toward a higher voltage. On the other hand, the intercalation of cobalt(II) perrhenate allowed us to limit capacitance deterioration in cyclic mode and improved the wettability; so, the diffusion of the electrolyte within the electrode was enhanced and the reversibility was better. The specific capacitance was calculated from the CV curves using Equation (1):
where
V1 and
V2 are the initial and final potential,
i is the current intensity,
V is the voltage,
v is the scan rate, m is the mass of the electrode, and Δ
V is the potential window.
Capacitance retention was calculated for both materials with respect to the first and last CV cycle. The intercalation of cobalt rhenium species allowed us to diminish capacitance loss from 11% for EXPANDED to 7% for EXP_CoPR after 1000 cycles.
Galvanostatic charge/discharge is the most reliable technique to evaluate the specific capacitance of an electrode material. Capacitance was calculated from Equation (2):
where
i is current intensity of discharge,
t is the discharge time, m is the mass of the electrode, and Δ
V is the potential window. The specific capacitance of bare expanded graphite was as high as 52 F/g at a current density of 0.2 A/g, while after modification with cobalt rhenium species, this increased to 78 F/g at 0.3 A/g. The results show that simple mechanical intercalation of cobalt(II) perrhenate allowed us to enhance the specific capacitance by 50%. Additionally, the path of the charge and discharge curves was more symmetric after active species addition, and the iR drop well observed in EXPANDED curves did not appear in EXP_CoPR curves (
Figure 6).
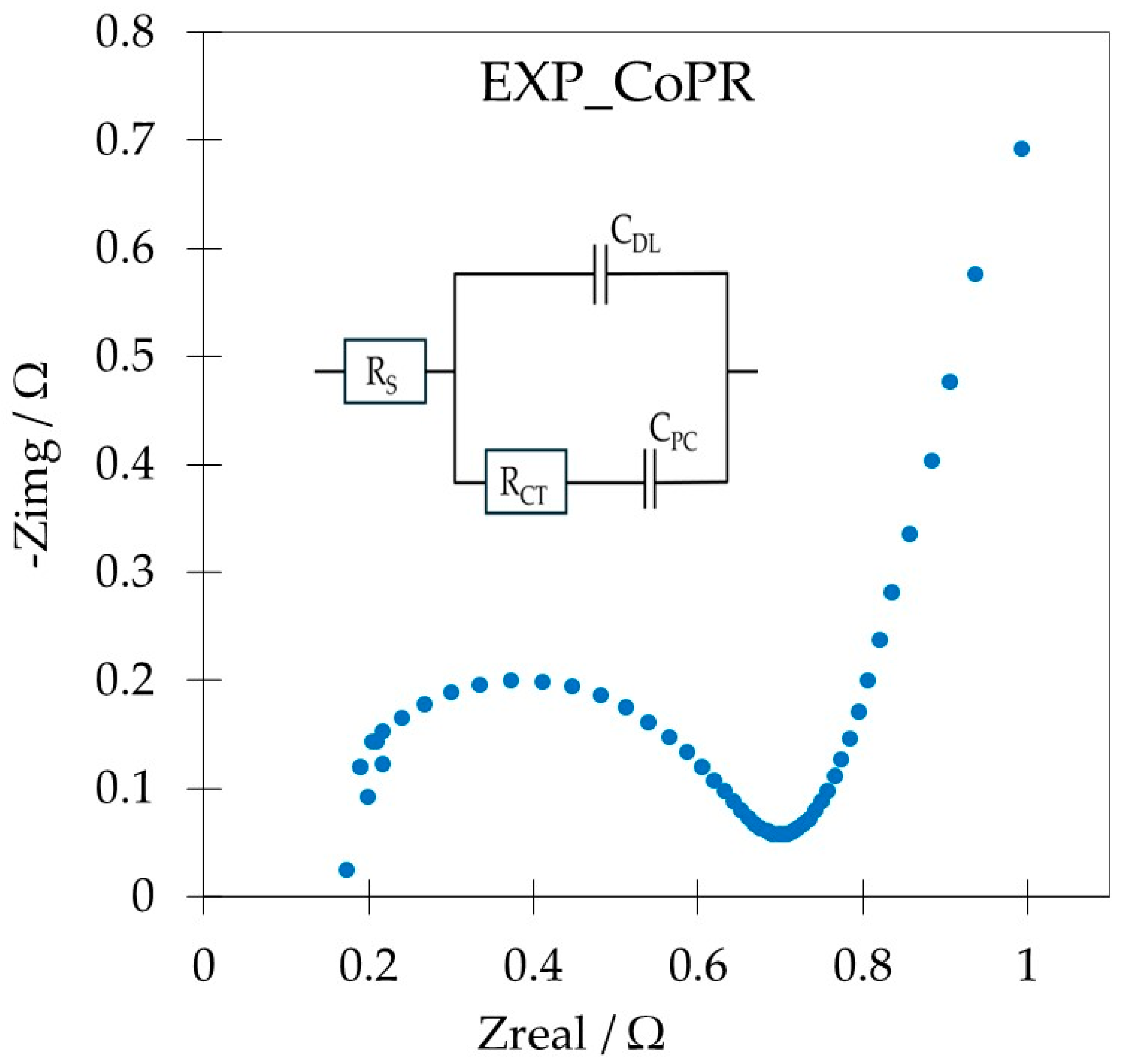
EIS was used to characterize the resistance of processes within the electrode. The relation between the real and imaginary components of impedance, called the Nyquist plot, is a powerful tool to characterize electrodes for energy storage applications. An electrode material that is governed by electrostatic and chemical processes is represented by two components, namely the semicircle in the high-frequency region attributed to the Faradaic charge transfer resistance, and the linear part in the low-frequency region, indicating pure capacitive behavior and representing ion diffusion in the electrode structure (
Figure 7).
This allowed us to estimate the resistance combined with the electrode RS and that combined with the charge transfer RCT. These were 0.26 Ω and 0.17 Ω (RS) and 2.43 Ω and 0.71 Ω (RCT) for EXPANDED and EXP_CoPR, respectively, while the total resistivity component was 2.92 Ω and 0.99 Ω for EXPANDED and EXP_CoPR. Smaller loops and steep curves in the low-frequency region are the consequences of better Warburg diffusion and, consequently, enhanced mass transfer from the electrolyte to the electrode interface in EXP_CoPR.