High-Pressure Gas Adsorption on Covalent Organic Framework CTF-1
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Instruments
2.3. CTF-1 Synthesis
2.4. Adsorption Studies
3. Results and Discussion
3.1. Composition of CTF-1
3.2. Characterization of CTF-1
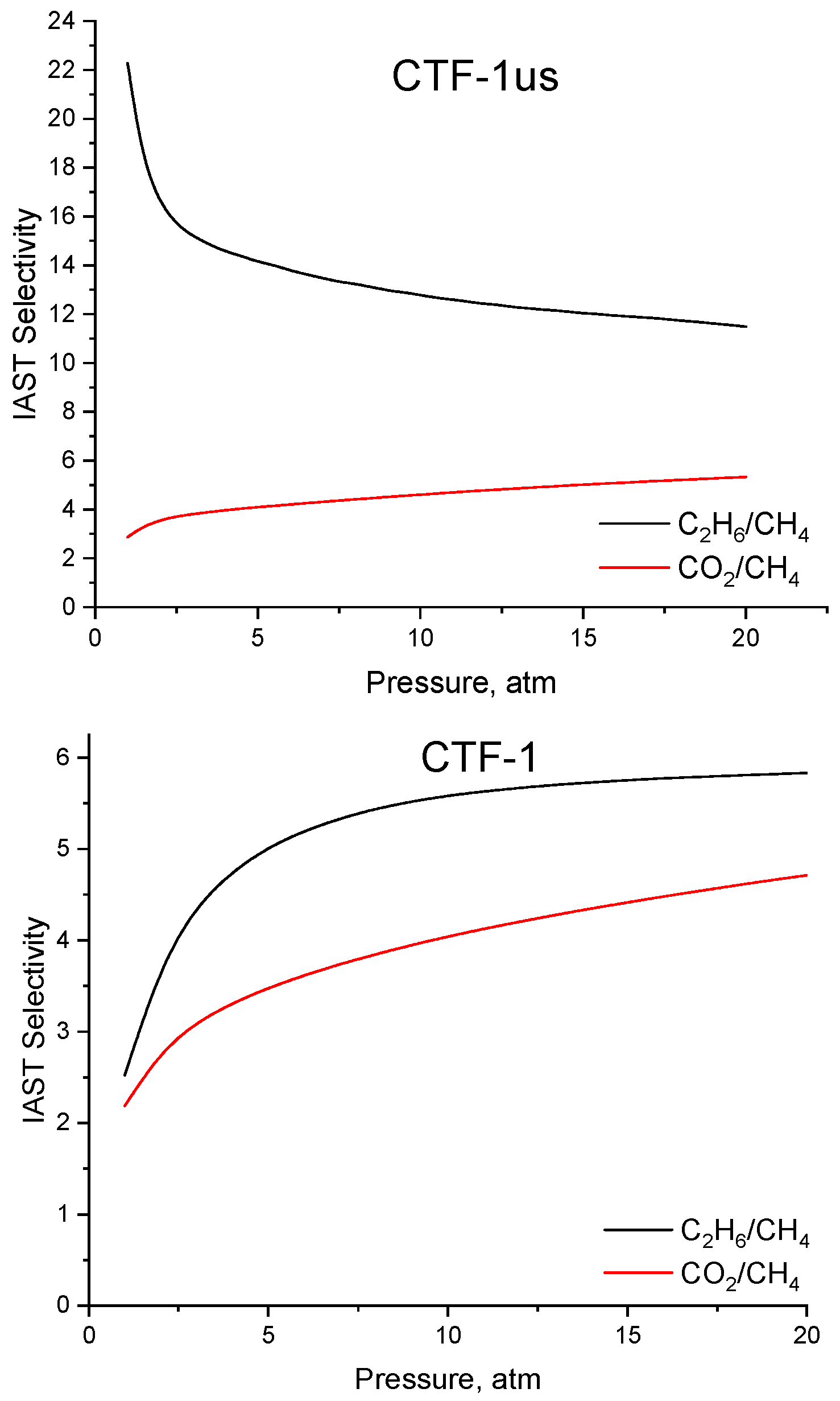
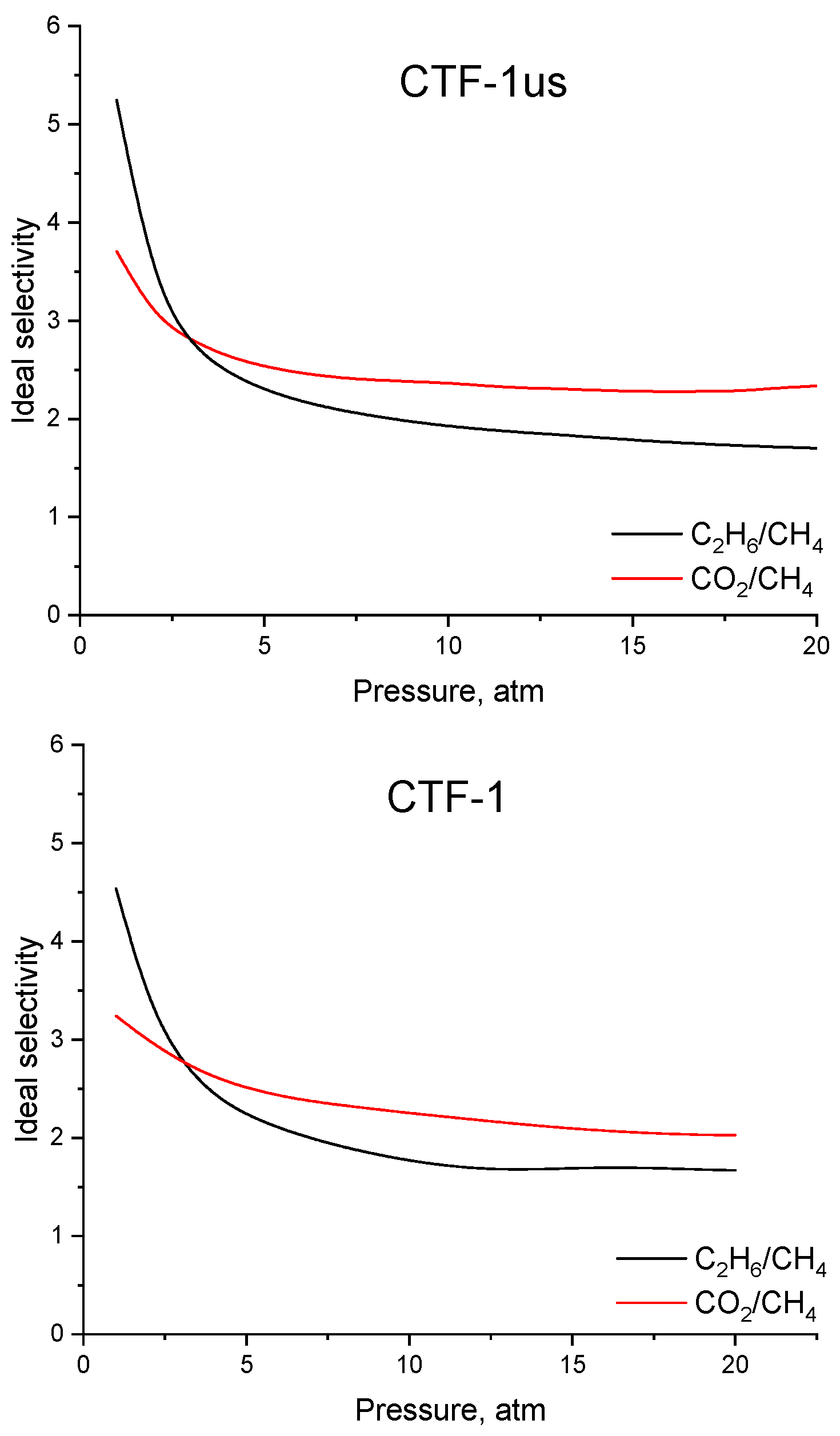
3.3. Adsorption on CTF-1
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Freund, R.; Zaremba, O.; Arnauts, G.; Ameloot, R.; Skorupskii, G.; Dincă, M.; Bavykina, A.; Gascon, J.; Ejsmont, A.; Goscianska, J.; et al. The Current Status of MOF and COF Applications. Angew. Chem. Int. Ed. 2021, 60, 23975–24001. [Google Scholar] [CrossRef] [PubMed]
- Chung, Y.G.; Haldoupis, E.; Bucior, B.J.; Haranczyk, M.; Lee, S.; Zhang, H.; Vogiatzis, K.D.; Milisavljevic, M.; Ling, S.; Camp, J.S.; et al. Advances, Updates, and Analytics for the Computation-Ready, Experimental Metal–Organic Framework Database: CoRE MOF 2019. J. Chem. Eng. Data 2019, 64, 5985–5998. [Google Scholar] [CrossRef]
- Ongari, D.; Yakutovich, A.V.; Talirz, L.; Smit, B. Building a Consistent and Reproducible Database for Adsorption Evaluation in Covalent–Organic Frameworks. ACS Cent. Sci. 2019, 5, 1663–1675. [Google Scholar] [CrossRef]
- Kuhn, P.; Antonietti, M.; Thomas, A. Porous, Covalent Triazine-Based Frameworks Prepared by Ionothermal Synthesis. Angew. Chem. Int. Ed. 2008, 47, 3450–3453. [Google Scholar] [CrossRef]
- Liao, L.; Li, M.; Yin, Y.; Chen, J.; Zhong, Q.; Du, R.; Liu, S.; He, Y.; Fu, W.; Zeng, F. Advances in the Synthesis of Covalent Triazine Frameworks. ACS Omega 2023, 8, 4527–4542. [Google Scholar] [CrossRef]
- Zakharov, V.N.; Kudryavtsev, I.K.; Dunaev, S.F.; Paseshnichenko, K.A.; Aslanov, L.A. Triazine 2D Nanosheets as a New Class of Nanomaterials: Crystallinity, Properties and Applications. Colloids Interfaces 2022, 6, 20. [Google Scholar] [CrossRef]
- Yu, S.; Mahmood, J.; Noh, H.; Seo, J.; Jung, S.; Shin, S.; Im, Y.; Jeon, I.; Baek, J. Direct Synthesis of a Covalent Triazine-Based Framework from Aromatic Amides. Angew. Chem. Int. Ed. 2018, 57, 8438–8442. [Google Scholar] [CrossRef] [PubMed]
- Gold, T. The Origin of Natural Gas and Petroleum, and the Prognosis for Future Supplies. Annu. Rev. Energy 1985, 10, 53–77. [Google Scholar] [CrossRef]
- Faramawy, S.; Zaki, T.; Sakr, A.A.-E. Natural Gas Origin, Composition, and Processing: A Review. J. Nat. Gas. Sci. Eng. 2016, 34, 34–54. [Google Scholar] [CrossRef]
- Zhang, R.; Wang, Z.; Wei, X.; Peng, X.; Chen, W.; Deng, C.; Liu, B.; Sun, C.; Chen, G. Modelling and Optimization of Ethane Recovery Process from Natural Gas via ZIF-8/Water-Glycol Slurry with Low Energy Consumption. Energy 2023, 263, 125645. [Google Scholar] [CrossRef]
- Siddiqi, K.S.; Thomas, W.J. The Adsorption of Methane-Ethane Mixtures on Activated Carbon. Carbone 1982, 20, 473–479. [Google Scholar] [CrossRef]
- Triebe, R.W.; Tezel, F.H.; Khulbe, K.C. Adsorption of Methane, Ethane and Ethylene on Molecular Sieve Zeolites. Gas. Sep. Purif. 1996, 10, 81–84. [Google Scholar] [CrossRef]
- Lin, R.-B.; Xiang, S.; Zhou, W.; Chen, B. Microporous Metal-Organic Framework Materials for Gas Separation. Chem 2020, 6, 337–363. [Google Scholar] [CrossRef]
- Deyko, G.S.; Glukhov, L.M.; Isaeva, V.I.; Vergun, V.V.; Chernyshev, V.V.; Kapustin, G.I.; Kustov, L.M. Adsorption of Methane and Ethane on HKUST-1 Metal–Organic Framework and Mesoporous Silica Composites. Mendeleev Commun. 2023, 33, 817–820. [Google Scholar] [CrossRef]
- Koyama, R.; Tsunoda, F.; Ichinose, I.; Kanoh, H. Adsorption Properties of Methane, Ethane, and Hexane on Mesoporous Organic Polymers Prepared by the Flash Freezing Method. Langmuir 2020, 36, 2184–2190. [Google Scholar] [CrossRef]
- Wang, H.; Liu, Y.; Liu, Y.; Wang, Z.; Li, C.; Sun, S.; Lyu, Q.; Hu, S. Two-Dimensional Imine Covalent Organic Frameworks for Methane and Ethane Separation: A GCMC Simulation Study. Microporous Mesoporous Mater. 2021, 326, 111386. [Google Scholar] [CrossRef]
- Katsoulidis, A.P.; Kanatzidis, M.G. Mesoporous Hydrophobic Polymeric Organic Frameworks with Bound Surfactants. Selective Adsorption of C2H6 versus CH4. Chem. Mater. 2012, 24, 471–479. [Google Scholar] [CrossRef]
- Ma, H.; Ren, H.; Meng, S.; Yan, Z.; Zhao, H.; Sun, F.; Zhu, G. A 3D Microporous Covalent Organic Framework with Exceedingly High C3H8/CH4 and C2 Hydrocarbon/CH4 Selectivity. Chem. Commun. 2013, 49, 9773. [Google Scholar] [CrossRef]
- Deyko, G.S.; Glukhov, L.M.; Isaeva, V.I.; Chernyshev, V.V.; Vergun, V.V.; Archipov, D.A.; Kapustin, G.I.; Tkachenko, O.P.; Nissenbaum, V.D.; Kustov, L.M. Modifying HKUST-1 Crystals for Selective Ethane Adsorption Using Ionic Liquids as Synthesis Media. Crystals 2022, 12, 279. [Google Scholar] [CrossRef]
- Zhang, S.; Taylor, M.K.; Jiang, L.; Ren, H.; Zhu, G. Light Hydrocarbon Separations Using Porous Organic Framework Materials. Chem.—A Eur. J. 2020, 26, 3205–3221. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, J.; Yang, Q.; Zhong, C. Two-Dimensional Covalent Triazine Framework Membrane for Helium Separation and Hydrogen Purification. ACS Appl. Mater. Interfaces 2016, 8, 8694–8701. [Google Scholar] [CrossRef] [PubMed]
- Deeg, K.S.; Damasceno Borges, D.; Ongari, D.; Rampal, N.; Talirz, L.; Yakutovich, A.V.; Huck, J.M.; Smit, B. In Silico Discovery of Covalent Organic Frameworks for Carbon Capture. ACS Appl. Mater. Interfaces 2020, 12, 21559–21568. [Google Scholar] [CrossRef] [PubMed]
- Lan, J.; Cao, D.; Wang, W.; Smit, B. Doping of Alkali, Alkaline-Earth, and Transition Metals in Covalent-Organic Frameworks for Enhancing CO2 Capture by First-Principles Calculations and Molecular Simulations. ACS Nano 2010, 4, 4225–4237. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Cao, D. Effect of Li Doping on Diffusion and Separation of Hydrogen and Methane in Covalent Organic Frameworks. J. Phys. Chem. C 2012, 116, 12591–12598. [Google Scholar] [CrossRef]
- Zakharov, V.; Kardasheva, Y.; Chernyshev, V.; Terenina, M.; Kalmykov, K.; Ovsyannikov, D.; Savilov, S.; Filippova, S.; Karakhanov, E.; Dunaev, S.; et al. Hydroformylation of Olefins over a Novel Active Rhodium Catalyst Supported on a Melamine–Cyanuric Acid Co-Crystal. Mol. Catal. 2023, 550, 113598. [Google Scholar] [CrossRef]
- Sychev, V.; Vasserman, A.; Zagoruchenko, V.; Kozlov, A.; Spiridonov, G.; Tsymarny, V. Thermodynamic Properties of Methane; Izdatel’stvo Standartov: Moscow, Russia, 1979. [Google Scholar]
- Sychev, V.; Wasserman, A.; Zagoruchenko, V.; Kozlov, A.; Spiridonov, G.; Tsymarnyi, V. Thermodynamic Properties of Ethane; Izdatel’stvo Standartov: Moscow, Russia, 1982; ISBN 9780891166115. [Google Scholar]
- Span, R.; Wagner, W. A New Equation of State for Carbon Dioxide Covering the Fluid Region from the Triple-Point Temperature to 1100 K at Pressures up to 800 MPa. J. Phys. Chem. Ref. Data 1996, 25, 1509–1596. [Google Scholar] [CrossRef]
- Myers, A.L.; Prausnitz, J.M. Thermodynamics of Mixed-Gas Adsorption. AIChE J. 1965, 11, 121–127. [Google Scholar] [CrossRef]
- Fraux, G.; Boutin, A.; Fuchs, A.H.; Coudert, F.-X. On the Use of the IAST Method for Gas Separation Studies in Porous Materials with Gate-Opening Behavior. Adsorption 2018, 24, 233–241. [Google Scholar] [CrossRef]
- Pourebrahimi, S.; Pirooz, M.; Kazemeini, M.; Vafajoo, L. Synthesis, Characterization, and Gas (SO2, CO2, NO2, CH4, CO, NO, and N2) Adsorption Properties of the CTF-1 Covalent Triazine Framework-Based Porous Polymer: Experimental and DFT Studies. J. Porous Mater. 2024, 31, 643–657. [Google Scholar] [CrossRef]
- Pawley, G.S. Unit-Cell Refinement from Powder Diffraction Scans. J. Appl. Crystallogr. 1981, 14, 357–361. [Google Scholar] [CrossRef]
- Zlokazov, V.B.; Chernyshev, V.V. MRIA—A Program for a Full Profile Analysis of Powder Neutron-Diffraction Time-of-Flight (Direct and Fourier) Spectra. Mater. Sci. Forum 1991, 79–82, 283–288. [Google Scholar] [CrossRef]
- Li, Z.; Han, Y.; Guo, Y.; Xu, S.; Chen, F.; Ye, L.; Luo, Z.; Liu, X.; Zhou, H.; Zhao, T. Evolution of the Formation of a Covalent Triazine-Based Framework Catalyzed by p-Toluenesulfonic Acid Monohydrate. RSC Adv. 2017, 7, 45818–45823. [Google Scholar] [CrossRef]
- Yan, J.; Zhang, B.; Wang, Z. Highly Selective Separation of CO2, CH4, and C2–C4 Hydrocarbons in Ultramicroporous Semicycloaliphatic Polyimides. ACS Appl. Mater. Interfaces 2018, 10, 26618–26627. [Google Scholar] [CrossRef] [PubMed]
- Furukawa, H.; Yaghi, O.M. Storage of Hydrogen, Methane, and Carbon Dioxide in Highly Porous Covalent Organic Frameworks for Clean Energy Applications. J. Am. Chem. Soc. 2009, 131, 8875–8883. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Yao, K.X.; Teng, B.; Zhang, T.; Han, Y. A Perfluorinated Covalent Triazine-Based Framework for Highly Selective and Water–Tolerant CO2 Capture. Energy Environ. Sci. 2013, 6, 3684. [Google Scholar] [CrossRef]
- Buyukcakir, O.; Je, S.H.; Talapaneni, S.N.; Kim, D.; Coskun, A. Charged Covalent Triazine Frameworks for CO2 Capture and Conversion. ACS Appl. Mater. Interfaces 2017, 9, 7209–7216. [Google Scholar] [CrossRef]
- Dong, J.; Wang, Y.; Liu, G.; Cheng, Y.; Zhao, D. Isoreticular Covalent Organic Frameworks for Hydrocarbon Uptake and Separation: The Important Role of Monomer Planarity. CrystEngComm 2017, 19, 4899–4904. [Google Scholar] [CrossRef]
- Meng, S.; Ma, H.; Jiang, L.; Ren, H.; Zhu, G. A Facile Approach to Prepare Porphyrinic Porous Aromatic Frameworks for Small Hydrocarbon Separation. J. Mater. Chem. A 2014, 2, 14536–14541. [Google Scholar] [CrossRef]
- Das, M.C.; Xu, H.; Wang, Z.; Srinivas, G.; Zhou, W.; Yue, Y.-F.; Nesterov, V.N.; Qian, G.; Chen, B. A Zn4O-Containing Doubly Interpenetrated Porous Metal–Organic Framework for Photocatalytic Decomposition of Methyl Orange. Chem. Commun. 2011, 47, 11715. [Google Scholar] [CrossRef]
- He, Y.; Zhou, W.; Krishna, R.; Chen, B. Microporous Metal–Organic Frameworks for Storage and Separation of Small Hydrocarbons. Chem. Commun. 2012, 48, 11813. [Google Scholar] [CrossRef]



| Elemental Composition of CTF-1, C8N2H4 | C, % | N, % | H, % | Ratio of C/N | 100- ΣCHN, % |
|---|---|---|---|---|---|
| Theoretical values | 74.99 | 21.86 | 3.15 | 3.43 | - |
| CTF-1us | 71.88 | 20.68 | 3.18 | 3.47 | 4.26 |
| CTF-1 | 69.10 | 16.58 | 3.57 | 4.17 | 10.75 |
| Sample Name | CTF-1 | CTF-1us |
|---|---|---|
| a = b, Å | 13.79(2) | 13.91(2) |
| c, Å | 3.377(14) | 3.399(13) |
| hkl | ZK225 | ZK225-UZ |
|---|---|---|
| 1 0 0 | 7.38 | 7.32 |
| 1 1 0 | 12.80 | 12.70 |
| 2 0 0 | 14.79 | 14.66 |
| 0 0 1 | 26.36 | 26.19 |
| Sample | SBET, m2/g | Vtot., cm3/g | Vmicro, cm3/g | Vmeso, cm3/g | Pore Diameter, nm |
|---|---|---|---|---|---|
| CTF-1 | 451 | 0.26 | 0.26 | - | 1.9–2.6 |
| CTF-1us | 550 | 0.30 | 0.30 | - | 1.9–2.6 |
| Sample | Ideal Selectivity | IAST Selectivity at y(CH4) = 0.9 | ||||
|---|---|---|---|---|---|---|
| C2H6:CH4, P = 1 atm | C2H6:CH4, P = 5 atm | C2H6:CH4, P = 20 atm | C2H6:CH4, P = 1 atm | C2H6:CH4, P = 5 atm | C2H6:CH4, P = 20 atm | |
| CTF-1 | 4.53 | 2.23 | 1.67 | 2.52 | 5.02 | 5.84 |
| CTF-us | 5.24 | 2.29 | 1.70 | 22.7 | 14.2 | 11.49 |
| CO2:CH4, P = 1 atm | CO2:CH4, P = 5 atm | CO2:CH4, P = 20 atm | CO2:CH4, P = 1 atm | CO2:CH4, P = 5 atm | CO2:CH4, P = 20 atm | |
| CTF-1 | 3.24 | 2.50 | 2.03 | 2.19 | 3.48 | 4.71 |
| CTF-us | 3.71 | 2.53 | 2.34 | 2.87 | 4.14 | 5.33 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Deyko, G.S.; Zakharov, V.N.; Glukhov, L.M.; Charkin, D.O.; Kultin, D.Y.; Chernyshev, V.V.; Aslanov, L.A.; Kustov, L.M. High-Pressure Gas Adsorption on Covalent Organic Framework CTF-1. Crystals 2024, 14, 1066. https://doi.org/10.3390/cryst14121066
Deyko GS, Zakharov VN, Glukhov LM, Charkin DO, Kultin DY, Chernyshev VV, Aslanov LA, Kustov LM. High-Pressure Gas Adsorption on Covalent Organic Framework CTF-1. Crystals. 2024; 14(12):1066. https://doi.org/10.3390/cryst14121066
Chicago/Turabian StyleDeyko, Gregory S., Valery N. Zakharov, Lev M. Glukhov, Dmitry O. Charkin, Dmitry Yu. Kultin, Vladimir V. Chernyshev, Leonid A. Aslanov, and Leonid M. Kustov. 2024. "High-Pressure Gas Adsorption on Covalent Organic Framework CTF-1" Crystals 14, no. 12: 1066. https://doi.org/10.3390/cryst14121066
APA StyleDeyko, G. S., Zakharov, V. N., Glukhov, L. M., Charkin, D. O., Kultin, D. Y., Chernyshev, V. V., Aslanov, L. A., & Kustov, L. M. (2024). High-Pressure Gas Adsorption on Covalent Organic Framework CTF-1. Crystals, 14(12), 1066. https://doi.org/10.3390/cryst14121066






