Abstract
Graphene derivatives such as reduced graphene oxide and graphene-based composites are regarded as highly promising materials for optoelectronics and photodetection applications. Recently, considerable interest has arisen in using facile and environmentally friendly methods of graphene production. Despite significant progress in experimental studies of graphene and graphene-based composites, some fundamental questions about their structures, and the interaction between components in these systems, remain open. In the present work, several atomistic models of oxidized graphene fragments have been proposed based on XPS compositional analysis and DFT calculations, representing reduced graphene oxide produced by laser irradiation. The composition of the oxygen-containing functional groups, their equilibrium configuration and influence on the electronic and optical properties of graphene sheets were determined. The nature of the low-lying excited states, as well as the photoactive regions, has also been studied for the proposed models. The calculated absorption spectra of the graphene sheets were compared with experimental UV-Vis spectrum of rGO produced by laser ablation.
1. Introduction
From its discovery in 2004 to the present, graphene continues to attract great attention in various fields of scientific research and applications due to its unique electronic, thermal, mechanical and optical properties [1]. Graphene is an attractive material for optoelectronic and photodetection applications [2,3] because it offers a broad spectral bandwidth and fast response times. However, pure graphene exhibits zero bandgap and weak light absorption; in addition, it has low solubility in water and tends to aggregate due to interlayer π-π interactions, which limits its optoelectronic and some other potential applications. For example, the oxygen functional groups attached to the surfaces and edges of graphene sheet can significantly modify its electronic, mechanical and optical properties and allows graphene to be processed easily in dispersions, leading to the plethora of applications, including solar cells [4,5,6,7], sensors [8], supercapacitors [9], and many more. For this reason, most of the new promising composite materials are based on graphene derivatives such as graphene oxide (GO), reduced graphene oxide (rGO) [10,11] and zero-dimensional nanostructures composed of one or few layers [12]. The above materials have tunable properties and can form stable aqueous suspensions.
Recently GO considered only a simple and inexpensive material for the preparation of single and multilayer graphene by reduction, but the results of the latest research demonstrated a large number of unique optical and electronic properties of GO and rGO that means they should be regarded as independent nanomaterials with a wide range of applications [13,14,15]. Reduced graphene oxide is regarded as a highly promising material for optoelectronic applications. One of the widely discussed applications for rGO, due to the growing interest in flexible and stretchable electronic devices, is thin transparent protective and conductive films, which are mostly used in optoelectronic and photovoltaic devices as electrodes. Films with a thickness of several atomic layers could be easily deposited on any hydrophilic surface by simple drop-casting or spin coating methods [16]. Graphene derivatives are less expensive and more environmentally friendly in comparison with traditional ITO and can successfully replace it in most applications [10,17]. Recently, silver- and gold-decorated rGO hybrid sheets were successfully fabricated and demonstrated low resistance at more than 83% transparency [17]. Ag–rGO films achieved a sheet conductivity two orders of magnitude higher than that of rGO films [17]. Flexible field-effect light-emitting diodes based on rGO were constructed by employing a graphene field-effect transistor [18]. To date, rGO has been successfully used in the fabrication of organic, perovskite, silicon and dye-sensitized solar cells and photodetectors [4,5,6,7,19]. Dye functionalized graphenes, such as rGO nanohybrids with porphyrins, were shown to possess great potential in the field of photocatalytic hydrogen production and optical sensing [20,21].
The common methods to obtain rGO include chemical and thermal reduction of GO. Recently, laser irradiation has been proposed as a promising tool for the reduction of GO [22,23,24,25,26]. In addition to the spatial control and energy efficiency, the laser reduction process is much cleaner and less toxic than any chemical method, as it does not require chemical reactions in acids, or long-term washing procedures [23,25]. Pulsed laser ablation in liquid has recently attracted increased attention for the synthesis of graphene and GO quantum dots [27,28]. In our previous work, we reported on graphenic oxidized films deposited on metal and dielectric substrates by pulsed laser ablation in an inert gas atmosphere [29]. We have found a significant reduction of oxygen-containing groups in comparison with the target material. The methods of graphene oxidation/reduction and fragmentation significantly affect its physical properties, including the mechanisms of conduction, light absorption and emission. Taking into account the diversity of the possible graphene derivatives, the application of theoretical methods that could correctly predict the spectroscopic properties of those materials can serve as a guidance for fabrication of such materials with desired properties. Generally, rGO is an amorphous system, and there is no direct experimental information that can provide a clear understanding of its local microscopic structure. At the same time, an understanding of the atomic structure can help reveal the mechanisms of interaction of rGO with other molecules, surfaces and nanostructures, and allow correct interpretation of spectroscopic data. To date there have been a number of papers on modelling graphene and graphene derivatives. There are two approaches in modelling graphene oxide. The first is based on periodic density functional theory calculations and assumes some degree of repeating order in oxygen atoms’ distribution on the graphene surface [30,31,32]. The other approach consider graphene as a finite sheet decorated with oxygen-containing groups [21,33,34]. The finite models, in spite of the computational complexity, become more preferable as they are physically more reasonable. They allow reproduction of the edge functionalization of graphene and correctly describe the adsorption and interaction of graphene with molecules and different surfaces. Finite models have also been shown to reproduce the optical properties of graphene-based nanohybrid systems with good accuracy [21,34,35]. Among the various finite structural models proposed recently [36,37,38] the Lerf–Klinowski model (LK) [39] was explicitly or implicitly assumed in the majority of successful functionalization studies [38]. While the LK model is quite successful in describing the properties of graphene produced by “classical” chemical methods, it does not account for the structural features of reduced graphene oxide produced by physical methods, such as hydrothermal and laser reduction [25,40]. The successful validation of structural models relies on the combination of theoretical and experimental approaches. In this work, we present atomistic models of oxidized graphene sheets, produced by laser ablation, based on XPS compositional analysis and DFT calculations of electronic and optical properties.
2. Materials and Methods
The widely accepted Lerf–Klinowski model [39] and other finite structural models [36,37] describe GO as carbon planes heavily decorated with epoxy and hydroxyl groups on the surfaces and carbonyl and carboxyl groups at the edges of the nanosheets. These oxygen groups are mostly removed from the surface in rGO by the use of chemical reducing agents such as hydrazine and its derivatives, sodium borohydride or hydroiodic acid, or physical reducing agents such as heat and optical radiation. In the case of laser irradiation of GO or oxidized carbon material, several processes can occur including photochemical and photothermal removal of oxygen functional groups, which is accompanied by laser ablation, and structural reorganization of the newly formed carbon lattice into the planar sp2-conjugated graphenic structure [26]. Fragmentation of carbon materials in solvents by laser irradiation can be explained within the model of Coulomb explosion [41]. As a result of this process in the laser plume, carbon precursors are ablated to the quantum size, and solvent can decompose into small molecules. The decomposed molecules can functionalize ablated graphenic fragments, which can significantly modify the final optoelectronic properties of the graphenic material. In contrast to chemically reduced graphene oxide, not only carboxyl and hydroxyl groups, but also ketones and quinones can remain on the edges of graphene sheets after laser ablation. Because of this, the Lerf–Klinowski model may not accurately describe the structure of graphene sheets produced by laser irradiation.
In this work, we propose several models of oxidized graphene, based on XPS compositional analysis of carbon films deposited on a silver substrate by laser ablation. The details of sample preparation and characterization are presented in supplementary information and in [29]. In our simulations, we considered rGO as a two-dimensional finite system, which contains randomly distributed graphene sheets of different sizes; recently this approach has been successfully applied to study GO/rGO-based nanohybrid systems [21,34,35]. XPS spectra allowed us to determine the type and amount of functional groups attached to carbon atoms, as well as the average carbon-to-oxygen ratio C/O in the graphene sheet. The core-level 1s carbon and oxygen spectra (Figure S2 in the Supplementary Materials) has shown that the ablated nanocarbon contains oxygen, singly bonded to aliphatic carbon (hydroxyl and/or epoxy groups), oxygen doubly bonded to aromatic and aliphatic carbon (carboxyl, carbonyl and quinone groups), and oxygen singly bonded to aromatic carbon (phenol groups) (Table S1 in the Supplementary Materials). The C/O ratio was estimated at ca. 6.6 which indicates that a large amount of the oxygen-containing groups were removed upon ablation. The partial restoration of the sp2 honeycomb lattice was confirmed by Raman spectroscopy (Figure S3 in the Supplementary Materials). In this study we neglected the occurrence of point defects within the graphene sheets. Thus, in our models, graphene sheets consisted of a polyaromatic core with honeycomb lattice and oxygen functional groups at the edges, as illustrated in Figure 1.
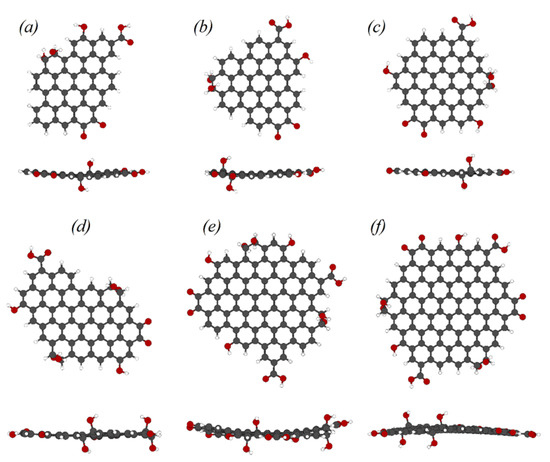
Figure 1.
Atomistic models of oxidized graphene sheets: (a)—C47H18O7, (b)—C49H18O7, (c)—C55H18O8, (d)—C61H22O10, (e)—C84H26O13, (f)—C98H24O14.
Our approach was based on the search for structures with minimum total energy and a singlet ground-state electronic configuration. The characteristic dimensions of the sheets ranged from 1.6 nm to 2.3 nm, and the carbon-to-oxygen ratio within sheets ranged from 6.1 to 7. As a polyaromatic core, we considered stable structures with low and high symmetry (including D3h and D6h), containing both zigzag and armchair edges. For the final oxidized structures with the lowest ground state energy, the complete structural optimization was carried out using density functional theory within the plane-wave pseudopotential method [42].
The Kohn–Sham orbitals have been expanded in plane waves up to a kinetic energy cutoff of 60 Ry. The norm-conserving Hartwigsen–Goedeker–Hutter pseudopotentials were used. The oxidized sheets were separated from their periodic images in the supercell by at least 12 Å of vacuum. The atomic positions in the supercell were fully optimized with the force threshold of 2·10−3 Ry/Å.
The optical properties of the systems were studied using the linear-response time-dependent density functional theory. All computations were performed with the B3LYP hybrid functional. We compared the performance of different exchange-correlation functionals, including hybrid PBE0, BHLYP, and range-separated ωB97X functional. Semilocal GGA-functionals are known to underestimate excitation energies of simple polyaromatic molecules. However, it was found that hybrid functionals with different amounts of Hartree–Fock exchange give a rather large spread in the values of the excitation energies. The best agreement of calculated excitation energies with experimental absorption spectra of simple polyaromatic molecules was found using the B3LYP level of theory (Table S2 in the Supplementary Materials).
For each atomistic model, the first 25 singlet excited states corresponding to vertical electronic transitions were calculated. The excitation energies were determined by calculation the eigenvalues and eigenvectors of a Liouvillian superoperator using the iterative procedure analogous to Davidson diagonalization, as implemented in the turboTDDFT2.0 code [43]. Absorption spectra were calculated using the linear-response recursive Lanczos approach [44]. In this method the frequency-dependent dipole susceptibility is written as a matrix element of the resolvent of the system’s Liouvillian. The imaginary part of the susceptibility has been plotted representing the absorption spectra.
3. Results and Discussion
Total energy calculations showed, that oxygen-containing functional groups in graphene sheets tend to be located on edge carbon atoms that do not belong to aromatic sextets (so-called K-regions). It has also been found that carbonyl groups are most stable when they form dimers on the K-regions, similar to the 1,2-benzoquinone structure. Calculations have shown that sp3-bonded hydroxyl groups also tend to be located in the 1,2-configuration on the edge carbon atoms in K-regions, but are oriented on opposite sides of the rGO sheet. The rearrangement of phenolic and carboxyl groups on the edges did not significantly change the total energy of the systems under study.
The electronic structure and absorption spectra of the polyaromatic cores were calculated for a better understanding of the role of oxygen functional groups. The electronic structure and optical properties of polyaromatic molecules largely depend on their symmetry. Structures C48H18, C54H18 and C96H24 with D3h and D6h symmetry respectively exhibit degenerate HOMO/HOMO-1 and LUMO/LUMO + 1 orbitals leading to wider energy gaps and transitions at shorter wavelengths (Figure S4 in the Supplementary Materials). The highest energy gap of 3.11 eV was observed for the C48H18 molecule with D3h symmetry (Table S3 in the Supplementary Materials).
Oxidation leads to symmetry reduction and splitting of degenerate levels. The ground state Kohn–Sham (KS) energy levels diagram of the oxidized graphene sheets is shown in Figure 2. All structures demonstrate singlet ground state electronic configuration and an energy gap in the range from 1.77 eV to 2.29 eV. Analysis of charge density distribution for the chosen molecular orbitals has shown that the highest occupied molecular orbitals of the systems are formed predominantly by π-electrons in an aromatic core, while the lowest unoccupied MOs are localized at the K-regions with carbonyl groups and originate from p-electrons of carbon and oxygen (Figure S5 in the Supplementary Materials).
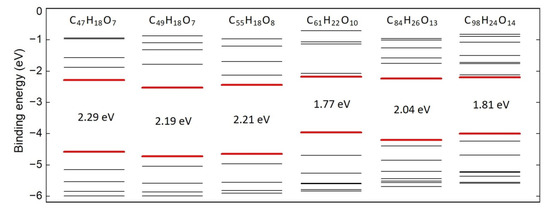
Figure 2.
Ground state KS energy levels of rGO fragments.
Typically, large polycyclic aromatic molecules reveal three characteristic optical bands α, p, and β with increasing intensity in the absorption edge [45,46]. The α-band with the highest wavelength is usually very weak and symmetry-forbidden in high symmetry groups. The p-band originates from HOMO → LUMO transition, it can be observed in the low-symmetry structures C46H18, C60H20, and C82H24 as the first intense absorption peak (the S1 peak in Figure 3g,j,k). The β-band has the lowest wavelength of the three and corresponds to a transition from HOMO to the second lowest unoccupied orbital (LUMO + 1) [47]. In the high-symmetry polyaromatic cores C48H18, C54H18, C96H24 p-bands are symmetry-forbidden, and very intense β-bands represent the absorption edge (S3–S4 bands in Figure 3h,i,l). These structures exhibit narrow absorption spectra with only few transitions.
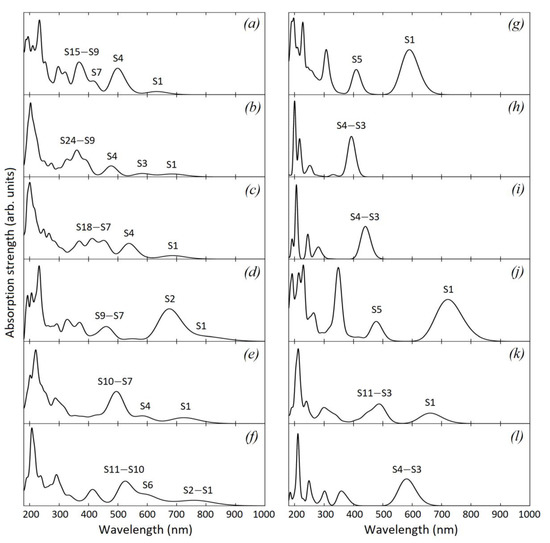
Figure 3.
Calculated absorption spectra of oxidized graphene sheets and corresponding polyaromatic cores: (a)—C47H18O7; (b)—C49H18O7; (c)—C55H18O8; (d)—C61H22O10; (e)—C84H26O13; (f)—C98H24O14; (g)—C46H18; (h)—C48H18; (i)—C54H18; (j)—C60H20; (k)—C82H24; (l)—C96H24.
Oxygen functional groups modify the absorption spectra of unoxidized structures. Oxidized graphene sheets have the lowest C1 symmetry, which leads to more allowed transitions and broader, less structured absorption spectra. For all oxidized structures, we can observe the occurrence of a long-wavelength absorption tail formed by the low-energy excited states (Figure 3a). We numbered the most characteristic absorption bands in the visible range in accordance with those excited states that make the dominant contribution at corresponding frequencies. Thus, for example, in the model C47H18O7, the absorption edge is formed due to S0 → S1 transition (Figure 3a), and the second intense absorption peak (denoted S4) at 500 nm is due to S0 → S4 (it originates from the S1 transition of the polyaromatic core C46H18 (Figure 3g). The data on excitation energies, wavelengths, oscillator strengths and transition coefficients for the considered models are presented in Table 1.

Table 1.
Compound, the most intense singlet–singlet transition, photon energy, wavelength, sum of diagonal elements of the imaginary part of dynamic polarizability and principal transition components in terms of ground-state KS orbitals (H = HOMO; L = LUMO).
In order to better understand the individual absorption bands and photo-active regions we calculated the real-space electron charge-density response to a perturbing electric field n’(r,ω) [48]:
where -unperturbed (ground-state) KS orbitals, Nv-number of occupied KS states, -first-order response orbitals at frequency ω. The factor 2 accounts for the spin degeneracy of molecular orbitals in non-magnetic systems.
Response charge density of the most prominent transitions in the oxidized graphene sheets is shown in Figure 4. So, for example, in the case of the C47H18O7 model, it can be seen that the weak S1 absorption band appears due to an electron-density response associated with electron-withdrawing carbonyl groups and neighboring carbon atoms. In contrast, the intense S4 band in this model, which has a maximum at 500 nm, is formed due to the response of the electron density of carbon atoms in the polyaromatic core. This density is delocalized throughout the graphene sheet and is not related to the activity of oxygen groups. Therefore, the S4 band originates from the S0 → S1 transition in the C46H18 core. Transitions in the short-wave visible and near-UV regions are of mixed origin. Thus, for example, the absorption peak at 378 nm in the C47H18O7 structure is due to the response of the electron density localized both on the carbon atoms of the polyaromatic core and on the hydroxyl, carboxyl, and carbonyl groups.
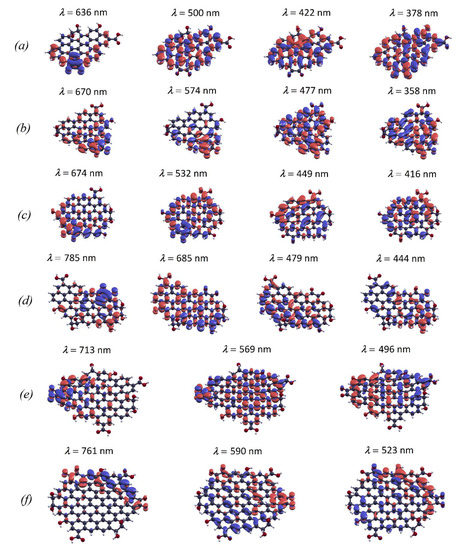
Figure 4.
Response charge density to a perturbing electric field of selected wavelength for the oxidized graphene models: (a)—C47H18O7; (b)—C49H18O7; (c)—C55H18O8; (d)—C61H22O10; (e)—C84H26O13; (f)—C98H24O14.
The most significant changes in absorption spectra were found for the structures C49H18O7 and C55H18O8 (Figure 3b,c). While the C48H18 structure exhibits absorption only in the ultraviolet region, the optical spectrum of the structure C49H18O7 covers the entire visible range. The C49H18O7 and C55H18O8 models have a similar absorption spectrum. In the long-wave range, according to Figure 3 and Table 1, the absorption spectrum of these systems is formed due to the transitions between two highest occupied and two lowest unoccupied molecular orbitals. In both structures the S1 absorption edge corresponds to the transition from the ground state to the first singlet excited state. It can be seen from the analysis of transition components that the S0 → S1 excitation is entirely determined by the HOMO → LUMO transition in these models. From Figure 4b,c one can see that the absorption edge is related to the optical activity of the carbonyl groups and neighboring carbon atoms, similar to the C47H18O7 model. In the C49H18O7 model, the S3 absorption band at 574 nm is also related to this photo-active region because it is formed mainly by the transition HOMO-1 → LUMO to the lowest unoccupied orbital. According to Figure 4b,c the S4 absorption band in the models C49H18O7 and C55H18O8 is largely due to the optical response of carboxylic and carbonyl functional groups. In the case of the C61H22O10 model the low-symmetry polyaromatic core produces an intense S2 absorption band associated with the low-energy π → π* transitions (HOMO → LUMO + 1). The spectrum also features the weak absorption edge at 785 nm due to the response of carbonyl groups as in the previous models (Figure 4d). The models C84H26O13 and C98H24O14 with the largest number of carbon atoms in the core demonstrate absorption spectra similar in structure despite differences in symmetry and C/O ratio. The long-wavelength absorption tail as in the previous models is formed by the HOMO → LUMO and HOMO → LUMO + 1 transitions (Figure 3e,f) and is related to the response of the electron-withdrawing oxygen functional groups and response of polyaromatic core (Figure 4e,f). The most intense absorption bands in these models are observed in the wavelength range of 500–600 nm. In the system C84H26O13 the absorption maximum in the visible region is at 496 nm and is related to the electronic transitions in the sp2-carbon network. In the C98H24O14 model, the absorption maximum is at 523 nm, and is due to the response of the zigzag edges as well as phenolic and carbonyl functional groups.
In order to understand which models better reproduce the optical properties of rGO we made a comparison of the calculated absorption spectra with the experimental UV-Vis spectrum of rGO suspensions, produced by laser ablation of high purity carbon tape (Figure 5). It should be noted that rGO samples measured in the experiment consist of multiple graphene sheets with different size, symmetry and arrangement of functional groups, thus the experimental absorption spectrum yields an averaged picture, and a single finite model is unable to reproduce this. The experimental spectrum features the absorption maximum at 240 nm which corresponds to a π-π* plasmon peak, and a relatively continuous absorption tail up to 1000 nm. There are also no distinguishable features corresponding to n-π* transitions.
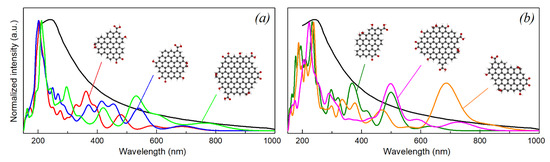
Figure 5.
Comparison of experimental UV-Vis spectrum and the calculated absorption spectra of rGO fragments with high-symmetry (a) and low-symmetry polyaromatic core (b).
We divided the calculated spectra into two parts. In Figure 5a,b we have compared the experimental spectrum with calculated spectra representing models with a high-symmetry and low-symmetry polyaromatic core, respectively. From Figure 5b it can be seen that the calculated spectra of the models with a low-symmetry polyaromatic core accurately reproduce the position of the UV absorption maximum, but feature intense absorption peaks in the visible region, which are not observed in the experiment. In contrast, the spectra of the high-symmetry models are shown to better reproduce the experimental absorption tail throughout the visible range, though the absorption maximum was found to be blue-shifted in these models. The best agreement with experiment in the visible range was found for the combination of models C47H18O7, C49H18O7 and C55H18O8. The calculated coefficient of determination for the weighted sum of these models in the wavelength range from 380 to 800 nm was 0.91.
4. Conclusions
In the present work, we proposed several atomistic models of rGO fragments based on XPS composition analysis of graphenic carbon produced by pulsed laser ablation and studied their optical properties using time-dependent DFT. It was shown that carbonyl and sp3-bonded hydroxyl functional groups in graphene sheets tend to locate on edge carbon atoms that do not belong to aromatic sextets; in addition, carbonyl groups are most stable when they form dimers.
Optical absorption of rGO, produced by laser reduction in the long-wavelength region, may be due to the optical activity of carbonyl groups, and low-energy π → π* transitions in the polyaromatic core of graphene sheets. The symmetry of the polyaromatic core affects the absorption intensity in the visible region, and the position of the absorption maximum in the UV region. A low-symmetry polyaromatic core in oxidized graphene sheet produces intense absorption bands in the visible range, which originate from intense p-bands. These bands were found to be blue-shifted and had a lower intensity when compared to the original p-bands. The calculated absorption spectra of the low-symmetry models were shown to accurately reproduce the experimental position of the π → π* plasmon peak in the UV region, while the high-symmetry models were better at reproducing the absorbance in the visible region.
Understanding the atomic structure of graphene derivatives and its correlation with electronic and optical properties can help in the interpretation of new spectroscopic data and can serve as a guidance for the fabrication of new graphene-based hybrid materials and interfaces for optoelectronic applications.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/cryst13040600/s1, Figure S1: Survey XPS spectrum and carbon 1s deconvoluted core-level spectrum of target material; Figure S2: Survey XPS spectrum of rGO deposited on silver substrate (top) and 1s deconvoluted core-level spectra of carbon and oxygen (bottom) Table S1: XPS composition analysis, fitting parameters and C/O ratio of the as-prepared sample; Table S2: Calculated and experimental positions of p- and β-bands in the absorption spectra of simple nanographenes. Calculations performed using plane-wave pseudopotential method and B3LYP XC functional. Figure S3: Raman spectrum of the as-prepared sample; Figure S4: Ground state KS energy levels of polyaromatic cores; Table S3: Composition, C/O ratio in graphene sheet, positions of HOMO and LUMO, and energy gap of the rGO sheets and corresponding aromatic cores, calculated using plane-wave pseudopotential approach and B3LYP functional; Figure S5: Charge density distribution in real space for the frontier molecular orbitals: HOMO, LUMO, HOMO-1, LUMO + 1. (a)-C47H18O7, (b)-C49H18O7, (c)-C55H18O8, (d)-C61H22O10, (e)-C84H26O13, (f)-C98H24O14.
Author Contributions
Conceptualization, I.V.E. and A.A.L.; Data curation, A.A.L. and O.M.H.; Formal analysis, A.A.L.; Investigation, I.V.E., I.V.B. and N.V.P.; Methodology, I.V.E. and A.A.L.; Project administration, A.A.L.; Resources, I.V.E.; Software, I.V.B., D.L.R. and T.P.Z.; Supervision, I.V.E.; Validation, N.V.P. and I.V.B.; Visualization, O.M.H.; Writing-original draft, I.V.E.; Writing-review & editing, A.A.L. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
Authors would like to acknowledge I.N. Efremenko and E.A. Portnova for the assistance in organizing high performance computations at the Don Engineering Centre of DSTU.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Geim, A.K. Graphene: Status and prospects. Science 2009, 324, 1530–1534. [Google Scholar] [CrossRef]
- Bonaccorso, F.; Sun, Z.; Hasan, T.; Ferrari, A.C. Graphene photonics and optoelectronics. Nat. Photon. 2010, 4, 611–622. [Google Scholar] [CrossRef]
- Mueller, T.; Xia, F.; Avouris, P. Graphene photodetectors for high-speed optical communications. Nat. Photon. 2010, 4, 297–301. [Google Scholar] [CrossRef]
- Mahakul, P.C.; Subramaniam, B.V.R.S.; Mahanandia, P. Mesoscopic investigation of the effect of MWCNT/rGO network on the performance of P3HT:PC60BM solar cells. Mater. Chem. Phys. 2019, 226, 113–117. [Google Scholar] [CrossRef]
- Liu, R.; Qiu, R.; Zou, T.; Liu, C.; Chen, J.; Dai, Q.; Zhang, S.; Zhou, H. SnO2-rGO nanocomposite as an efficient electron transport layer for stable perovskite solar cells on AZO substrate. Nanotechnology 2018, 30, 075202. [Google Scholar] [CrossRef]
- Jiang., X.; Wang, Z.; Han, W.; Liu, Q.; Lu, S.; Wen, Y.; Hou, J.; Huang, F.; Peng, S.; He, D.; et al. High performance silicon–organic hybrid solar cells via improving conductivity of PEDOT:PSS with reduced graphene oxide. Appl. Surf. Sci. 2017, 407, 398–404. [Google Scholar] [CrossRef]
- Gopi, C.V.V.M.; Singh, S.; Reddy, A.E.; Kim, H.J. CNT@rGO@MoCuSe composite as an efficient counter electrode for quantum dot-sensitized solar cells. ACS Appl. Mater. Interfaces 2018, 10, 10036–10042. [Google Scholar] [CrossRef]
- Liu, J.; Xu, M.; Wang, B.; Zhou, Z.; Wang, L. Fluorescence sensor for detecting protamines based on competitive interactions of polyacrylic acid modified with sodium 4-amino-1-naphthalenesulfonate with protamines and aminated graphene oxide. RSC Adv. 2017, 7, 1432–1438. [Google Scholar] [CrossRef]
- Li, Z.; Gadipelli, S.; Yang, Y.; Guo, Z. Design of 3D Graphene-Oxide Spheres and Their Derived Hierarchical Porous Structures for High Performance Supercapacitors. Small 2017, 13, 1702474. [Google Scholar] [CrossRef]
- Dideikin, A.T.; Vul’, A.Y. Graphene oxide and derivatives: The place in graphene family. Front. Phys. 2019, 6, 149. [Google Scholar] [CrossRef]
- Tarcan, R.; Todor-Boer, O.; Petrovai, I.; Leordean, C.; Astilean, S.; Botiz, I. Reduced graphene oxide today. J. Mater. Chem. C 2020, 8, 1198–1224. [Google Scholar] [CrossRef]
- Bacon, M.; Bradley, S.J.; Nann, T. Graphene quantum dots. Part. Part. Syst. Charact. 2014, 31, 415–428. [Google Scholar] [CrossRef]
- Novoselov, K.S.; Fal′ko, V.I.; Colombo, L.; Gellert, P.R.; Schwab, M.G.; Kim, K. A roadmap for graphene. Nature 2012, 490, 192–200. [Google Scholar] [CrossRef]
- Kakavelakis, G.; Maksudov, T.; Konios, D.; Paradisanos, I.; Kioseoglou, G.; Stratakis, E.; Kymakis, E. Efficient and highly air stable planar inverted perovskite solar cells with reduced graphene oxide doped PCBM electron transporting layer. Adv. Energy Mater. 2017, 7, 1602120. [Google Scholar] [CrossRef]
- Naderi, H.R.; Sobhani-Nasab, A.; Rahimi-Nasrabadi, M.; Ganjali, M.R. Decoration of nitrogen-doped reduced graphene oxide with cobalt tungstate nanoparticles for use in high-performance supercapacitors. Appl. Surf. Sci. 2017, 423, 1025–1034. [Google Scholar] [CrossRef]
- Yamaguchi, H.; Eda, G.; Mattevi, C.; Kim, H.K.; Chhowalla, M. Highly uniform 300 mm wafer-Scale deposition of single and multilayered chemically derived graphene thin films. ACS Nano 2010, 4, 524–528. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Tjong, S.C. Silver-decorated reduced graphene oxides as novel building blocks for transparent conductive films. RSC Adv. 2017, 7, 2058–2065. [Google Scholar] [CrossRef]
- Wang, X.; Tian, H.; Mohammad, M.A.; Li, C.; Wu, C.; Yang, Y.; Ren, T.L. A spectrally tunable all-graphene-based flexible field-effect light-emitting device. Nat. Commun. 2015, 6, 7767. [Google Scholar] [CrossRef]
- Yu, X.X.; Yin, H.; Li, H.-X.; Zhao, H.; Li, C.; Zhu, M.-Q. A novel high-performance self-powered UV-vis-NIR photodetector based on a CdS nanorod array/reduced graphene oxide film heterojunction and its piezo-phototronic regulation. J. Mater. Chem. C 2018, 6, 630–636. [Google Scholar] [CrossRef]
- Xu, Y.; Zhao, L.; Bai, H.; Hong, W.; Li, C.; Shi, G. Chemically converted graphene induced molecular flattening of 5,10,15,20-Tetrakis(1-methyl-4-pyridinio)porphyrin and its application for optical detection of cadmium(II) ions. J. Am. Chem. Soc. 2009, 131, 13490–13497. [Google Scholar] [CrossRef]
- Siklitskaya, A.; Gacka, E.; Larowska, D.; Mazurkiewicz-Pawlicka, M.; Malolepszy, A.; Stobiński, L.; Marciniak, B.; Lewandowska-Andrałojć, A.; Kubas, A. Lerf–Klinowski-type models of graphene oxide and reduced graphene oxide are robust in analyzing non-covalent functionalization with porphyrins. Sci. Rep. 2021, 11, 7977. [Google Scholar] [CrossRef] [PubMed]
- Kymakis, E.; Petridis, C.; Anthopoulos, T.D.; Stratakis., E. Laser-assisted reduction of graphene oxide for flexible, large-area optoelectronics. IEEE J. Sel. Top. Quant. Electr. 2014, 20, 106–115. [Google Scholar] [CrossRef]
- Bhattacharjya, D.; Kim, C.-H.; Kim, J.-H.; You, I.-K.; In, J.B.; Lee, S.-M. Fast and controllable reduction of graphene oxide by low-cost CO2 laser for supercapacitor application. Appl. Surf. Sci. 2018, 462, 353–361. [Google Scholar] [CrossRef]
- Evlashin, S.; Dyakonov, P.; Khmelnitsky, R.; Dagesyan, S.; Klokov, A.; Sharkov, A.; Timashev, P.; Minaeva, S.; Maslakov, K.; Svyakhovskiy, S.; et al. Controllable Laser Reduction of Graphene Oxide Films for Photoelectronic Applications. ACS Appl. Mater. Interfaces 2016, 8, 28880–28887. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, R.D.; Khalelov, A.; Postnikov, P.S.; Lipovka, A.; Dorozhko, E.; Amin, I.; Murastov, G.V.; Chen, J.-J.; Sheng, W.; Trusova, M.E.; et al. Beyond graphene oxide: Laser engineering functionalized graphene for flexible electronics. Mater. Horiz. 2020, 7, 1030–1041. [Google Scholar] [CrossRef]
- Zhao, Y.; Han, Q.; Cheng, Z.; Jiang, L.; Qu, L. Integrated graphene systems by laser irradiation for advanced devices. Nano Today 2017, 12, 14–30. [Google Scholar] [CrossRef]
- Donate-Buendia, C.; Torres-Mendieta, R.; Pyatenko, A.; Falomir, E.; Fernández-Alonso, M.; Mínguez-Vega, G. Fabrication by laser irradiation in a continuous flow jet of carbon quantum dots for fluorescence imaging. ACS Omega 2018, 3, 2735–2742. [Google Scholar] [CrossRef]
- Kang, S.; Ryu, J.H.; Lee, B.; Jung, K.H.; Shim, K.B.; Han, H.; Kim., K.M. Laser wavelength modulated pulsed laser ablation for selective and efficient production of graphene quantum dots. RSC Adv. 2019, 9, 13658–13663. [Google Scholar] [CrossRef]
- Ershov, I.V.; Lavrentyev, A.A.; Prutsakova, N.V.; Holodova, O.M.; Mardasova, I.V.; Zhdanova, T.P.; Kozakov, A.T. Characterization of graphenic carbon produced by pulsed laser ablation of sacrificial carbon tapes. Appl. Sci. 2021, 11, 11972. [Google Scholar] [CrossRef]
- Boukhvalov, D.W.; Katsnelson, M.I. Modeling of Graphite Oxide. J. Am. Chem. Soc. 2008, 130, 10697–10701. [Google Scholar] [CrossRef]
- Lahaye, R.J.W.E.; Jeong, H.K.; Park, C.Y.; Lee, Y.H. Density functional theory study of graphite oxide for different oxidation levels. Phys. Rev. B 2009, 79, 125435. [Google Scholar] [CrossRef]
- Lundie, M.; Šljivančanin, Z.; Tomić., S. Electronic and optical properties of reduced graphene oxide. J. Mater. Chem. C 2015, 3, 7632–7641. [Google Scholar] [CrossRef]
- Hernández Rosas, J.J.; Ramírez Gutiérrez, R.E.; Escobedo-Morales, A.; Chigo Anota, E. First principles calculations of the electronic and chemical properties of graphene, graphane, and graphene oxide. J. Mol. Model. 2011, 17, 1133–1139. [Google Scholar] [CrossRef] [PubMed]
- Gacka, E.; Burdzinski, G.; Marciniak, B.; Kubas, A. Interaction of light with a non-covalent zinc porphyrin–graphene oxide nanohybrid. Phys. Chem. Chem. Phys. 2020, 22, 13456–13466. [Google Scholar] [CrossRef] [PubMed]
- Gacka, E.; Wojcik, A.; Mazurkiewicz-Pawlicka, M.; Malolepszy, A.; Stobiński, L.; Kubas, A.; Hug, G.L.; Marciniak, B.; Lewandowska-Andralojc. A. Noncovalent Porphyrin–Graphene Oxide Nanohybrids: The pH-Dependent Behavior. J. Phys. Chem. C 2019, 123, 3368–3380. [Google Scholar] [CrossRef]
- Szabo, T.; Berkesi, O.; Forgo, P.; Josepovits, K.; Sanakis, Y.; Petridis, D.; Dekany, I. Evolution of Surface Functional Groups in a Series of Progressively Oxidized Graphite Oxides. Chem. Mater. 2006, 18, 2740–2749. [Google Scholar] [CrossRef]
- Gao, W.; Alemany, L.B.; Ci, L.; Ajayan, P.M. New insights into the structure and reduction of graphite oxide. Nature Chem. 2009, 1, 403–408. [Google Scholar] [CrossRef]
- Dreyer, D.R.; Todd, A.D.; Bielawski., C.W. Harnessing the chemistry of graphene oxide. Chem. Soc. Rev. 2014, 43, 5288–5301. [Google Scholar] [CrossRef]
- Lerf, A.; He, H.; Forster, M.; Klinowski, J. Structure of graphite oxide revisited. J. Phys. Chem. B 1998, 102, 4477–4482. [Google Scholar] [CrossRef]
- Diez, N.; Sliwak, A.; Gryglewicz, S.; Grzyb, B.; Gryglewicz, G. Enhanced reduction of graphene oxide by high-pressure hydrothermal treatment. RSC Adv. 2015, 5, 81831. [Google Scholar] [CrossRef]
- Zhang, D.; Gokce, B.; Barcikowski, S. Laser Synthesis and Processing of Colloids: Fundamentals and Applications. Chem. Rev. 2017, 117, 3990–4103. [Google Scholar] [CrossRef] [PubMed]
- Giannozzi, P.; Andreussi, O.; Brumme, T.; Bunau, O.; Nardelli, M.B.; Calandra, M.; Car, R.; Cavazzoni, C.; Ceresoli, D.; Cococcioni, M.; et al. Advanced capabilities for materials modelling with Quantum ESPRESSO. J. Phys. Condens. Matter. 2017, 29, 465901. [Google Scholar] [CrossRef] [PubMed]
- Ge, X.; Binnie, S.J.; Rocca, D.; Gebauer, R.; Baroni, S. turboTDDFT 2.0—Hybrid functionals and new algorithms within time-dependent density-functional perturbation theory. Comp. Phys. Comm. 2014, 185, 2080–2089. [Google Scholar] [CrossRef]
- Gruning, M.; Marini, A.; Gonze, X. Implementation and testing of Lanczos-based algorithms for Random-Phase Approximation eigenproblems. Comput. Mater. Sci. 2011, 50, 2148–2156. [Google Scholar] [CrossRef]
- Clar, E. Polycyclic Hydrocarbons; Academic Press and Springer-Verlag: London, UK, 1964; Volume 1–2. [Google Scholar]
- Rieger, R.; Mullen, K. Forever young: Polycyclic aromatic hydrocarbons as model cases for structural and optical studies. J. Phys. Org. Chem. 2010, 23, 315–325. [Google Scholar] [CrossRef]
- Fetzer, J.C. Large (C >= 24) Polycyclic Aromatic Hydrocarbons; John Wiley & Sons: New York, NY, USA, 2000. [Google Scholar]
- Malcioglu, O.B.; Gebauer, R.; Rocca, D.; Baroni, S. turboTDDFT—A code for the simulation of molecular spectra using the Liouville–Lanczos approach to time-dependent density-functional perturbation theory. Comp. Phys. Commun. 2011, 182, 1744–1754. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).