The Structural Characteristics and Chemical Composition of Serpentine Jade Weathering Rinds: Implications for the Formation Process
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Microstructure Characteristics
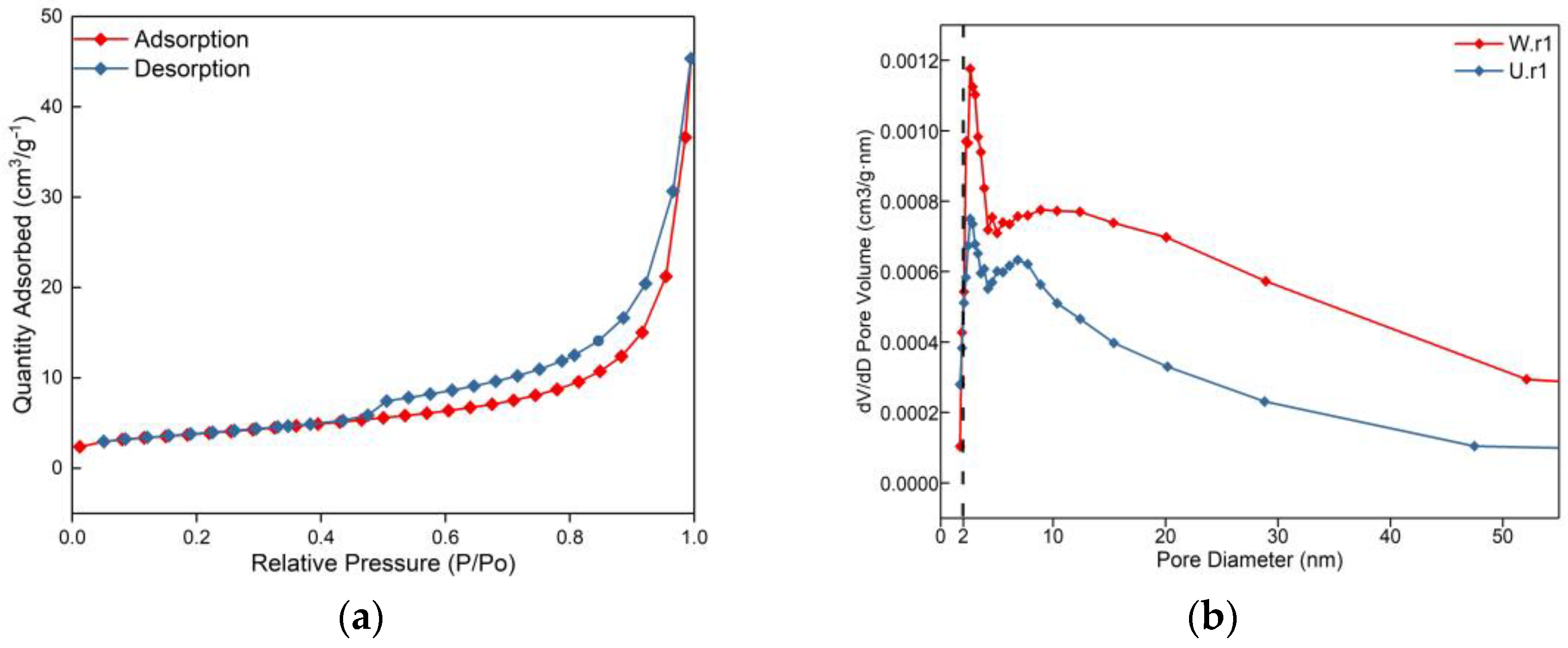
3.2. Nitrogen Adsorption by the BET Method
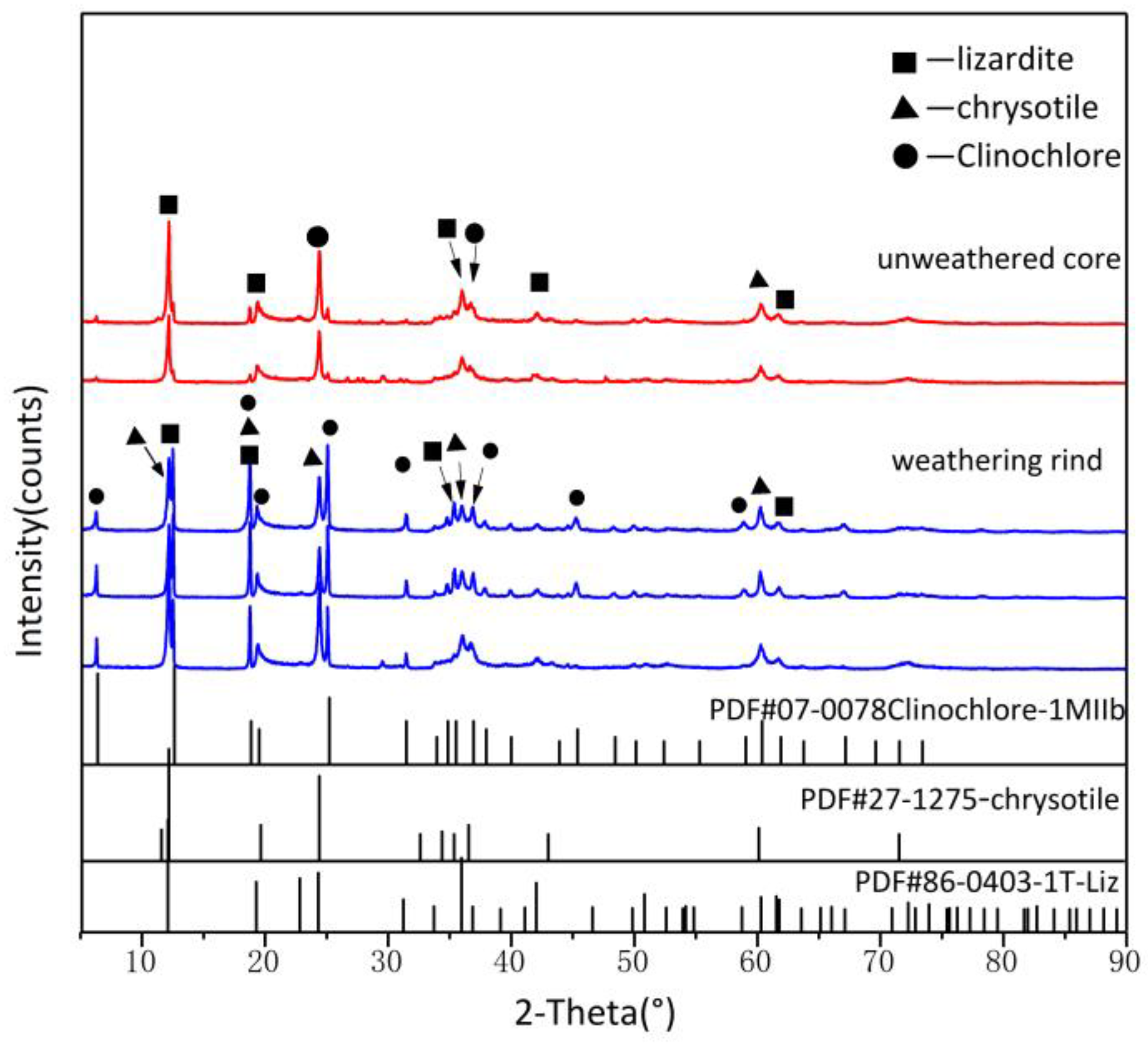
3.3. X-ray Diffraction
3.4. μ-XRF Mapping
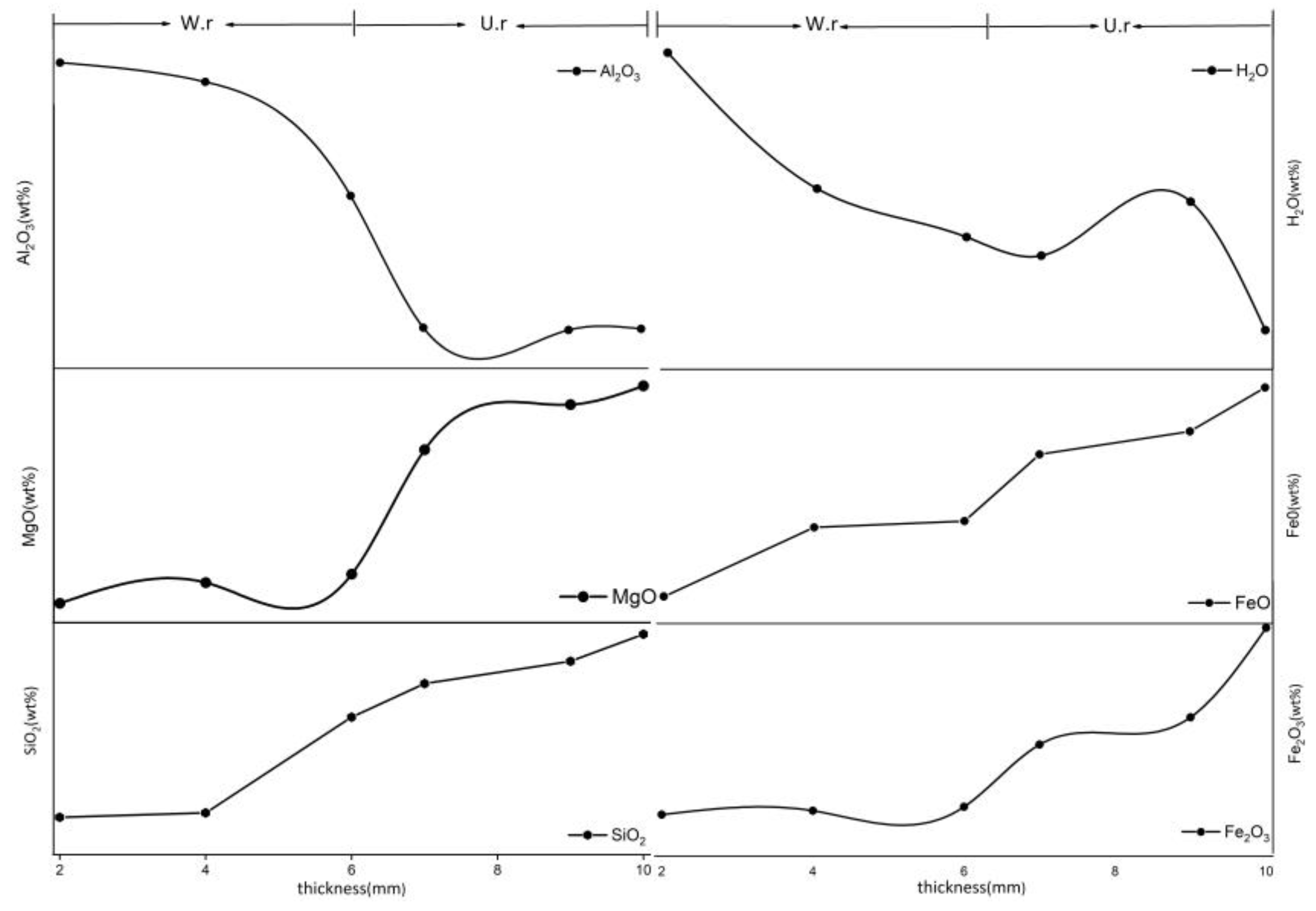
3.5. EPMA
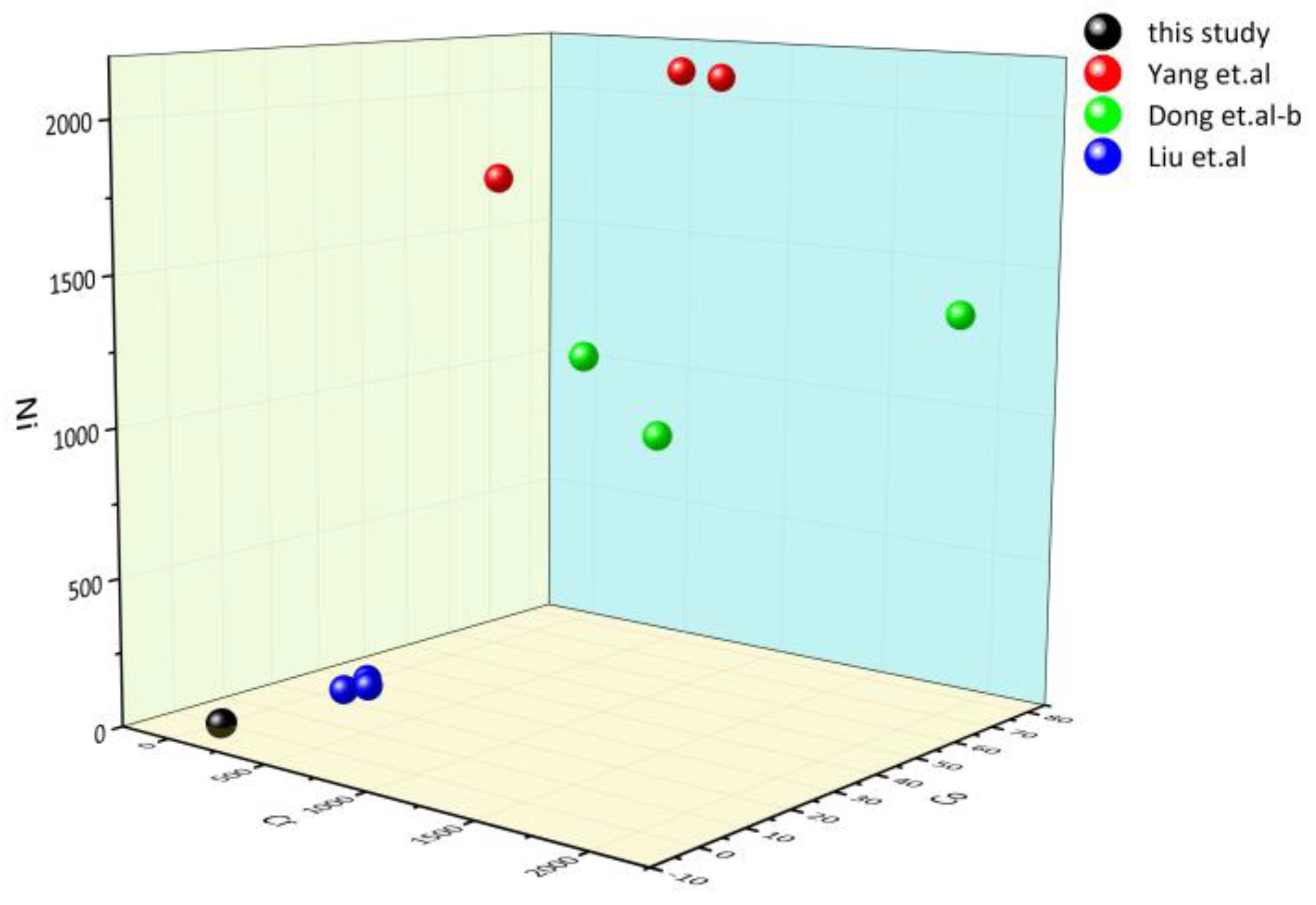
3.6. LA-ICP-MS
4. Discussion
4.1. The Structure Characteristics of Weathering Rinds and Unweathered Cores
4.2. The Formation Mechanism of Weathering Rinds
4.3. The Genesis Analysis of Serpentine Jade in Ji’an
5. Conclusions
- (1)
- The density of the weathering rind of Jilin Ji’an serpentine jade is 2.36–2.42 g/cm3, which is lower than the standard value of serpentine. The specific surface area is 13.3987 m2/g and the pore volume is 0.0314 cm3/g, which is much larger than the unweathered jade. Both are characterized by mesoporous (2–10 nm) distribution, accounting for about 80%. The adsorption isotherm of the weathering rind belongs to the IV isotherm type, H3 hysteresis type, and the structural characteristics show that serpentine crystals appear partially dissolved in the weathering rind, with increased porosity and a loose structure.
- (2)
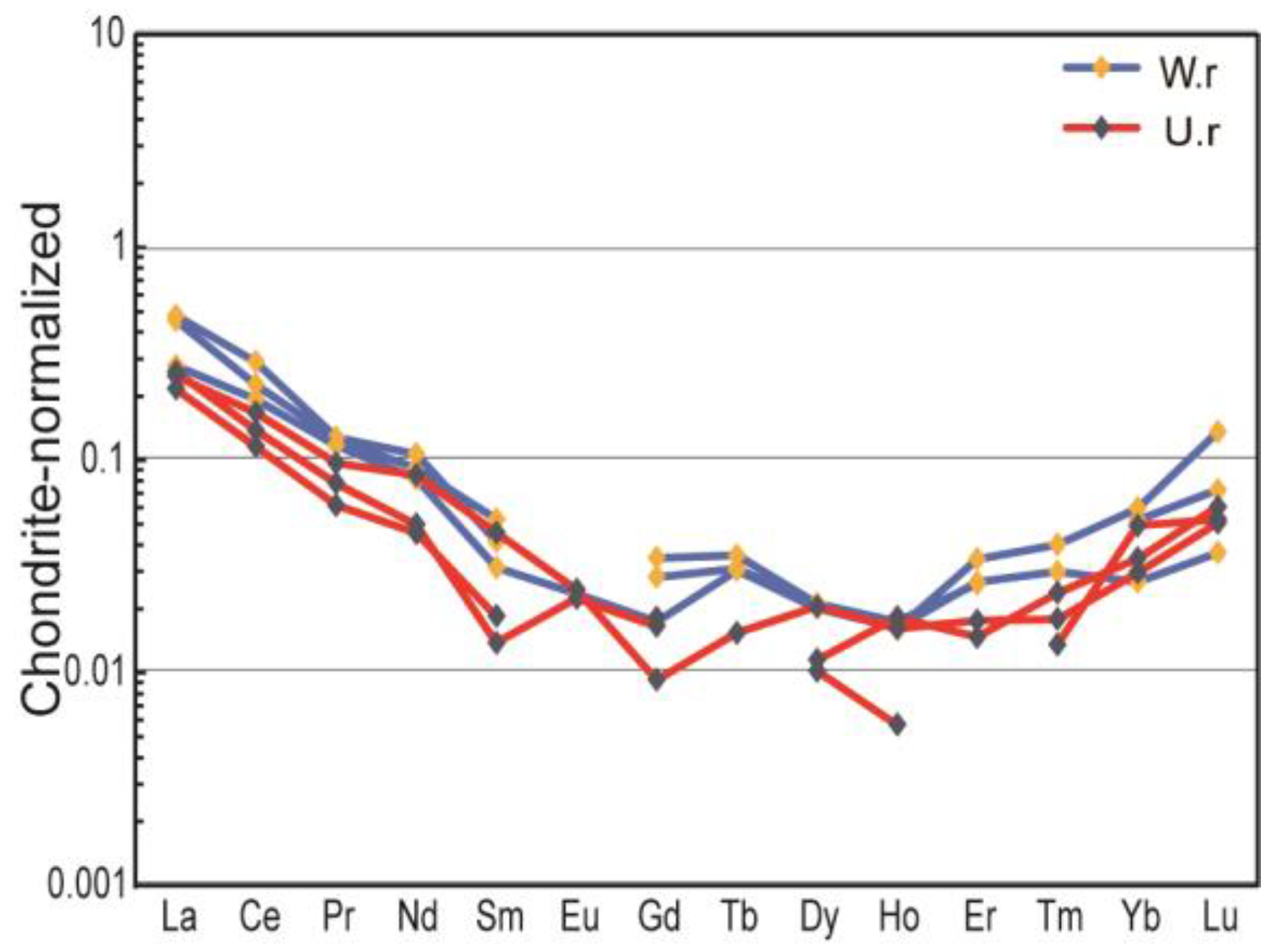
- The major trace element test shows that Ji’an serpentinite belongs to the Mg-bearing carbonate rock genesis, which is marine sedimentation with a relatively stable depositional environment. Overall, the serpentine jade is enriched in LILE such as K, Rb, Th, and U, and deficient in HFSE such as Nb, Sr, and Ti. ΣREE is 9.55–12.99 ppm, and Eu and Ce elements are slightly negative anomalies after chondrite-normalized.
- (3)
- Ji’an serpentine jade weathering rind is the weathering band with further alteration of serpentine, accompanied by the dissolution of serpentine grains and the reduction of the Fe3+/Fe2+ ratio in the process. It can be explained as follows: weathering increases the porosity of the exterior of serpentine jade, and long-term exposure to water erosion near rivers causes H2O and external impurities to enter pores. This causes the decrease in some major elements (Si, Mg, and Fe) of serpentine and the concentration of impurity elements (K, Na, Ca, Al, and Cl), the dissolution of serpentine grains, the generation of clay mineral (chlorite), structural loosening, the change in color, and finally the formation of a brownish-yellow or khaki weathering rind.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Qin, Y. Geochrnoligicl Constraints on the Tectonic Evolution of the Liao-Ji Paleoproterozoic Rift Zone. Ph.D. Thesis, Jilin University, Changchun, China, 2013. [Google Scholar]
- Wang, Y.; He, M.; Yan, W.; Yang, M.; Liu, X. Jianite: Massive Dunite Solely Made of Virtually Pure Forsterite from Ji’an County, Jilin Province, Northeast China. Minerals 2020, 10, 220. [Google Scholar] [CrossRef]
- Chen, Q.; Wang, Y.Y.; Gan, F.X. The Chemical Compositions and Origin Characters of Serpentine Jade from Different Deposits. Appl. Mech. Mater. 2014, 624, 119–123. [Google Scholar] [CrossRef]
- Wang, Y.Y.; Gan, F.X.; Zhao, H.X. Inclusions of Black-Green Serpentine Jade Determined by Raman Spectroscopy. Vib. Spectrosc. 2013, 66, 19–23. [Google Scholar] [CrossRef]
- Zhang, B.S.; Wu, X.T.; Sun, Y.F.; Ritchey, M.; Fan, A.C.; Zhang, Y.Y.; Yu, G.; Song, Y.B. Complex Raw Materials and the Supply System: Mineralogical and Geochemical Study of the Jade Artefacts of the Longshan Culture (2400–2000 Bce) from Sujiacun Site in Coastal Shandong, China. Archaeometry 2021, 63, 1–18. [Google Scholar] [CrossRef]
- Kloprogge, J.T.; Frost, R.L.; Rintoul, L. Single Crystal Raman Microscopic Study of the Asbestos Mineral Chrysotile. Phys. Chem. Chem. Phys. 1999, 1, 2559–2564. [Google Scholar] [CrossRef]
- Wu, S.; He, M.; Yang, M.; Zhang, B.; Wang, F.; Li, Q. Near-Infrared Spectroscopy Study of Serpentine Minerals and Assignment of the OH Group. Crystals 2021, 11, 1130. [Google Scholar] [CrossRef]
- Yariv, S. Infrared Evidence for the Occurrence of SiO Groups with Double-Bond Character in Antigorite, Sepiolite and Palygorskite. Clay Miner. 1986, 21, 925–935. [Google Scholar] [CrossRef]
- Li, J. Research on Serpentine Jade from Ji’an City, Jilin Province. Master’s Thesis, Jilin University, Changchun, China, April 2012. [Google Scholar]
- Baatar, A.; Kim, W.S. Mineralogical Characteristics of Serpentine Jade and Thulite Gemminerals from Booyo Area, South Korea. In Proceedings of the European Geosciences Union General Assembly 2009, Vienna, Austria, 19–24 April 2009; p. 573. [Google Scholar]
- Qin, H.; Liu, R. Mineral Composition and Genetic Analysis of Ji’an Serpentine Jade, Tonghua, Jinlin Province. Acta Petrol. Mineral. 2016, 35, 344–348. [Google Scholar]
- Colman, S.M.; Pierce, K.L. Weathering Rinds on Andesitic and Basaltic Stones as a Quaternary Age Indicator, Western United States. Geol. Surv. Prof. Pap. 1981. [Google Scholar] [CrossRef]
- Dixon, J.C.; Thorn, C.E.; Darmody, R.G.; Campbell, S.W. Weathering Rinds and Rock Coatings from an Arctic Alpine Environment, Northern Scandinavia. Geol. Soc. Am. Bull. 2002, 114, 226–238. [Google Scholar] [CrossRef]
- Grapes, R.H.; Yun, S.-T. Geochemistry of a New Zealand Nephrite Weathering Rind. N. Z. J. Geol. Geophys. 2010, 53, 413–426. [Google Scholar] [CrossRef]
- Oguchi, C.T. Formation of Weathering Rinds on Andesite. Earth Surf. Process. Landf. 2001, 26, 847–858. [Google Scholar] [CrossRef]
- Zhang, Z. Study on the Minerals Composition and Structural Characteristics of Weathering Crust of Primary Nephrite in Russia. Master’s Thesis, China University of Geosciences (Beijing), Beijing, China, May 2019. [Google Scholar]
- Colman, S.M. Rock-Weathering Rates as Functions of Time. Quat. Res. 1981, 15, 250–264. [Google Scholar] [CrossRef]
- Oguchi, C.T. A Porosity-Related Diffusion Model of Weathering-Rind Development. CATENA 2004, 58, 65–75. [Google Scholar] [CrossRef]
- Oguchi, C.T.; Matsukura, Y. Effect of Porosity on the Increase in Weathering-Rind Thicknesses of Andesite Gravel. Eng. Geol. 2000, 55, 77–89. [Google Scholar] [CrossRef]
- Tang, C.; Shi, B.; Wang, B. Factors Affecting Analysis of Soil Microstructure Using SEM. Chin. J. Geotech. Eng. 2008, 30, 560–565. [Google Scholar]
- Xiong, C.; Tang, H.; Liu, B.; Zhang, J. Using SEM Photos to the Pore Structural Parameters of Soil Samples. Earth Sci.-J. Univ. Geosci. 2007, 32, 415–419. [Google Scholar]
- Xu, R.; Deng, Y.; Xu, B.; Lai, J.; Zhan, X.; Xu, L.; Lu, J. Quantitative Analysis of Soft Clay Three-dimensional Porosity Based on SEM Image Information. J. Earth Sci. Environ. 2015, 37, 104–110. [Google Scholar]
- Liu, L. Study on Origin, Factors and Grading of the Color of Turquoise from China. Master’s Thesis, China University of Geosciences, Wuhan, China, May 2018. [Google Scholar]
- Chen, S.; Zhu, Y.; Wang, H.; Liu, H.; Wei, W.; Fang, J. Structure Characteristics and Accumulation Significance of Nanopores in Longmaxi Shale Gas Reservoir in the Southern Sichuan Basin. J. China Coal Soc. 2012, 37, 438–444. [Google Scholar] [CrossRef]
- Li, Z.; Shen, X.; Qi, Z.; Hu, R. Comparations Between Mercury Intrusion and Gas Adsorption for Pore Structure Characteristics of Shale. J. Eng. Geol. 2017, 25, 1405–1413. [Google Scholar] [CrossRef]
- Liu, P.; Guo, H.; Shen, R.; Li, H.; Ren, H.; Zhang, C. Study of the Structure of Shale Based on Gas Adsorption Method and Mercury Intrusion method. Mech. Eng. 2018, 40, 514–519. [Google Scholar]
- Yang, F.; Ning, Z.; Hu, C.; Wang, B.; Peng, K.; Liu, H. Characterization of Microscopic Pore Structures in Shale Reservoirs. Acta Pet. Sin. 2013, 34, 301–311. [Google Scholar] [CrossRef]
- Colman, S. Chemical Weathering of Basalts and Andesites; Evidence from Weathering Rinds; U.S. Government Publishing Office: Washington, DC, USA, 1982. [Google Scholar]
- Etienne, S. The Role of Biological Weathering in Periglacial Areas: A Study of Weathering Rinds in South Iceland. Geomorphology 2002, 47, 75–86. [Google Scholar] [CrossRef]
- Etienne, S.; Dupont, J. Fungal Weathering of Basaltic Rocks in a Cold Oceanic Environment (Iceland): Comparison between Experimental and Field Observations. Earth Surf. Process. Landf. 2002, 27, 737–748. [Google Scholar] [CrossRef]
- Butler, D.R. Rock Coatings.: R. Dorn. Elsevier, Amsterdam, 1998. Developments in Earth Surface Processes 6, Hardbound, Xii and 429 Pages. Price US$207.00, ISBN 0-444-82919-9. Geomorphology 2000, 33, 123–124. [Google Scholar] [CrossRef]
- Adsorption of Gases in Multimolecular Layers. J. Am. Chem. Soc. 1938, 60, 309–319. [CrossRef]
- Barrett, E.P.; Joyner, L.G.; Halenda, P.P. The Determination of Pore Volume and Area Distributions in Porous Substances. I. Computations from Nitrogen Isotherms. J. Am. Chem. Soc. 1951, 73, 373–380. [Google Scholar] [CrossRef]
- Sing, K.S.W. Reporting Physisorption Data for Gas/Solid Systems with Special Reference to the Determination of Surface Area and Porosity (Recommendations 1984). Pure Appl. Chem. 1985, 57, 603–619. [Google Scholar] [CrossRef]
- Brunauer, S.; Deming, L.S.; Deming, W.E.; Teller, E. On a Theory of the van Der Waals Adsorption of Gases. J. Am. Chem. Soc. 1940, 62, 1723–1732. [Google Scholar] [CrossRef]
- Sun, W.; McDonough, W. Chemical and Isotopic Systematics of Oceanic Basalts: Implications for Mantle Composition and Processes. Geol. Soc. Lond. Spec. Publ. 1989, 42, 313–345. [Google Scholar] [CrossRef]
- Zhang, Y.; Jiang, Z.; Li, S.; Wang, Y.; Yu, L. The Process of Oceanic Peridotite Serpentinization: From Seafloor Hydration to Subduction Dehydration. Acta Petrol. Sin. 2022, 38, 1063–1080. [Google Scholar] [CrossRef]
- Tang, Y.; Jia, J.; Xie, X. Environment Significance of Clay Minerals. Earth Sci. Front. 2002, 30, 337–344. [Google Scholar]
- Biscaye, P.E. Mineralogy and Sedimentation of Recent Deep-Sea Clay in the Atlantic Ocean and Adjacent Seas and Oceans. Geol. Soc. Am. Bull. 1965, 76, 803–832. [Google Scholar] [CrossRef]
- Huang, J. Study on Spectroscopy and Color-causing Mechanism of Anlu Jade. Volcanol. Miner. Resour. 1992, 13, 71–78. [Google Scholar]
- Wang, Y.; Gan, F. Coloration Mechanism and Chromaticity of Xiuyan Jade of China. Spectrosc. Spectr. Anal. 2012, 32, 2305–2310. [Google Scholar]
- Walter, L.M.; Huston, T.J.; Stueber, A.M. Br-Cl-Na systematics in Illinois basin fluids: Constraints on fluid origin and evolution. Geology 1990, 18, 315–318. [Google Scholar] [CrossRef]
- Yang, J.; Zhang, Y.; Qiu, Z.; Jia, D.; Zheng, X. Geochemistry and Petrogenesis of Greenstone Belt Type Serpentine Jade from Taishan, Shandongd. Geotecton. Metallog. 2021, 45, 1044–1059. [Google Scholar] [CrossRef]
- Leeman, W. Petrogenesis of McKinney (Snake River) Olivine Tholeiite in Light of Rare-Earth Element and Cr/Ni Distributions. Geol. Soc. Am. Bull. 1976, 87, 1582–1586. [Google Scholar] [CrossRef]
- Liu, Y. Metallogenic Regularity and Prospecing Indicator of the Jade Deposit in the Xiuyan Area, Liaoning Province. Master’s Thesis, Jilin University, Changchun, China, November 2013. [Google Scholar]
- Dong, J.; Wang, Y.; Gan, F.; Li, Q. Trace Element Analysis by PIYE and ICP-AES of Raw Material and Ancient Serpentine Artifacts from China. Guang Pu Xue Yu Guang Pu Fen Xi Guang Pu 2016, 36, 3780–3788. [Google Scholar]
- Zhuo, S.; Liu, Y.; Wang, J.; Jia, H.; Fu, Z.; Chi, H.; Mei, S.; Guo, P. The Petrological and Geochemical Characteristics of the Serpentinized Marble in the Wusutu Region, Inner Mongolia, China. Bull. Mineral. Ptrol. Geochem. 2016, 35, 239–246+1. [Google Scholar] [CrossRef]
- Taylor, S.R.; McLennan, S.M. The Continental Crust: Its Composition and Evolution; Blackwell Scientific Publications: Oxford, UK, 1985; 312p. [Google Scholar]
- Jewell, P.W.; Stallard, R.F. Geochemistry and Paleoceanographic Setting of Central Nevada Bedded Barites. J. Geol. 1991, 99, 151–170. [Google Scholar] [CrossRef]



| Samples | W.r-1 | W.r-2 | W.r-3 | U.r-1 | U.r-2 | U.r-3 | Serpentine |
|---|---|---|---|---|---|---|---|
| relative density (g/cm3) | 2.417 | 2.354 | 2.388 | 2.455 | 2.491 | 2.502 | 2.57(+0.23, −0.13) |
| Lattice Parameters | Weathering Rinds | Unweathered Cores | Lizardite |
|---|---|---|---|
| a0/nm | 0.529 | 0.531 | 0.531 |
| b0/nm | 0.924 | 0.926 | 0.920 |
| c0/nm | 0.731 | 0.731 | 0.731 |
| β (°) | 89.586 | 89.908 | 90 |
| Date | W.r-1 | W.r-2 | W.r-3 | U.r-1 | U.r-2 |
|---|---|---|---|---|---|
| SiO2 | 33.03 | 32.71 | 39.97 | 44.03 | 42.4 |
| TiO2 | 0.04 | 0 | 0 | 0.07 | 0 |
| MgO2 | 36.92 | 35.93 | 37.33 | 45.51 | 43.33 |
| K2O | 0.02 | 0.02 | 0.02 | 0.06 | 0.02 |
| Cr2O3 | 0.02 | 0.07 | 0 | 0.02 | 0 |
| Al2O3 | 17.10 | 18.42 | 9.31 | 0.13 | 0.29 |
| CaO | 0.06 | 0.01 | 0.03 | 0.06 | 0.01 |
| MnO | 0 | 0 | 0.05 | 0.06 | 0.03 |
| FeO * | 0.89 | 0.86 | 0.53 | 1.32 | 1.53 |
| Fe2O3 * | 0 | 0 | 0 | 0.18 | 0.48 |
| NiO | 0.05 | 0.04 | 0.02 | 0 | 0 |
| Na2O | 0.04 | 0.05 | 0.08 | 0.02 | 0.01 |
| # H20 | 1.79 | 1.71 | 2.6 | 1.48 | 1.9 |
| Total | 88.21 | 88.29 | 87.40 | 88.52 | 88.1 |
| Date | W.r-1 | W.r-2 | W.r-3 | U.r-1 | U.r-2 | U.r-3 |
|---|---|---|---|---|---|---|
| Li | 0.069 | 0.076 | 0.097 | 0.010 | 0.010 | 0.024 |
| B | 19.9 | 15.2 | 12.1 | 24.07 | 14.23 | 15 |
| Ti | 27.9 | 59 | 3.43 | 2.56 | 1.38 | 1.95 |
| K | 50 | 60.5 | 60 | 0.72 | 1.019 | 560 |
| Na | 1280 | 1560 | 1660 | 4.33 | 3.85 | 320 |
| Ca | 720 | 940 | 900 | 6.4 | 5.2 | 320 |
| V | 0.41 | 0.7 | 0.06 | 0.026 | 0.044 | 0.043 |
| Cr | 0.49 | 2.47 | 0.14 | 0.128 | 0.121 | 0.125 |
| Co | 0.148 | 0.331 | 0.056 | 0.168 | 0.23 | 0.065 |
| Ni | 0.31 | 0.24 | 0.153 | 0.253 | 0.268 | 0.221 |
| Sr | 0.626 | 0.91 | 0.718 | 0.057 | 0.032 | 0.275 |
| Zr | 0.56 | 0.67 | 0.67 | 0.058 | 0.066 | 0.27 |
| Cl | 43 | 71 | 44 | 23.3 | 21.8 | 27.8 |
| S | 62 | 66 | 41.6 | 27.7 | 26.9 | 30.7 |
| La | 0.141 | 0.148 | 0.086 | 0.081 | 0.067 | 0.077 |
| Ce | 0.183 | 0.234 | 0..156 | 0.112 | 0.094 | 0.135 |
| Pr | 0.015 | 0.016 | 0.014 | 0.01 | 0.008 | 0.012 |
| Nd | 0.055 | 0.064 | 0.049 | 0.03 | 0.027 | 0.051 |
| Sm | 0.01 | 0.008 | 0.006 | 0.003 | 0.004 | 0.009 |
| Eu | 0.002 | 0.002 | 0.001 | 0.002 | 0.002 | 0.002 |
| Gd | 0.007 | 0.009 | 0.005 | 0.004 | 0.005 | 0.002 |
| Tb | 0.001 | 0.002 | 0.001 | b.d.l | b.d.l | 0.001 |
| Dy | 0.007 | 0.001 | 0.007 | 0.004 | 0.003 | 0.007 |
| Ho | 0.001 | 0.006 | 0.001 | 0.001 | 0.001 | 0.001 |
| Er | 0.007 | b.d.l | 0.006 | 0.003 | b.d.l | 0.004 |
| Tm | 0.001 | 0.011 | 0.001 | 0.001 | 0.001 | 0.001 |
| Yb | 0.012 | 0.002 | 0.006 | 0.007 | 0.01 | 0.006 |
| Lu | 0.005 | 0.049 | 0.001 | 0.002 | 0.002 | 0.002 |
| Y | 0.045 | 0.558 | 0.042 | 0.029 | 0.021 | 0.034 |
| Th | 0.038 | 0.044 | 0.053 | 0.004 | 0.005 | 0.019 |
| U | 0.014 | 0.018 | 0.015 | 0.005 | 0.005 | 0.014 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xue, S.; He, M.; Yang, M.; Wu, S. The Structural Characteristics and Chemical Composition of Serpentine Jade Weathering Rinds: Implications for the Formation Process. Crystals 2023, 13, 239. https://doi.org/10.3390/cryst13020239
Xue S, He M, Yang M, Wu S. The Structural Characteristics and Chemical Composition of Serpentine Jade Weathering Rinds: Implications for the Formation Process. Crystals. 2023; 13(2):239. https://doi.org/10.3390/cryst13020239
Chicago/Turabian StyleXue, Shanna, Mingyue He, Mei Yang, and Shaokun Wu. 2023. "The Structural Characteristics and Chemical Composition of Serpentine Jade Weathering Rinds: Implications for the Formation Process" Crystals 13, no. 2: 239. https://doi.org/10.3390/cryst13020239
APA StyleXue, S., He, M., Yang, M., & Wu, S. (2023). The Structural Characteristics and Chemical Composition of Serpentine Jade Weathering Rinds: Implications for the Formation Process. Crystals, 13(2), 239. https://doi.org/10.3390/cryst13020239