The Development of New Perovskite-Type Oxygen Transport Membranes Using Machine Learning
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Reference Data
2.2. Bond-Valence Modeling
2.3. Data Analysis and Python Programming
2.4. Machine Learning
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Auto-WEKA | Automatic Model Selection and Hyperparameter Optimization in WEKA |
| BVM | Bond Valence Model |
| CLI | Command Line Interface |
| DFT | Density Functional Theory |
| FV | Free Volume |
| lazy.IBK | K-nearest Neighbours (WEKA Classifier) |
| MD | Molecular Dynamics |
| ML | Machine Learning |
| Oxygen Partial Pressure | |
| SG | Space Group |
| SVM | Support Vector Machine (WEKA Classifier) |
| STO | |
| SVO | |
| STVO | |
| STFO | |
| SBTFO | |
| SBTVO | |
| SBTVFO | |
| WEKA | Waikato Environment for Knowledge Analysis |
Appendix A
| SrTiO3 | -> | T = 0 to 2313 K |
| SrVO3 | -> | T = 0 to 1956 K |
| SrTi(1-y)V(y)O3 | -> | T = 973 to 1173 K |
| SrTi(1-z)Fe(z)O3 | -> | T = 973 to 1173 K |
| Sr(0.5)Ba(0.5)Ti(0.5)Fe(0.5)O3 | -> | T = 1073 to 1223 K |
| Sr(1-x)Ba(x)Ti(1-y)V(y)O3 | -> | T = 973 to 1173 K |
| Sr(1-x)Ba(x)Ti(1-y-z)V(y)Fe(z)O3 | -> | T = 973 to 1173 K |
| SrTiO3 | -> | T = 950 to 1173 K |
| SrVO3 | -> | T = 973 to 1173 K |
| SrTi(1-y)V(y)O3 | -> | T = 1173 K |
| SrTi(1-z)Fe(z)O3 | -> | T = 973 to 1223 K |
| Sr(0.5)Ba(0.5)Ti(0.5)Fe(0.5)O3 | -> | T = 1073 to 1223 K |
| Sr(1-x)Ba(x)Ti(1-y)V(y)O3 | -> | T = no data available |
| Sr(1-x)Ba(x)Ti(1-y-z)V(y)Fe(z)O3 | -> | T = no data available |
| SrTiO3 | -> | pO2 = to 1.0 bar |
| SrVO3 | -> | pO2 = to bar |
| SrTi(1-y)V(y)O3 | -> | pO2 = to bar |
| SrTi(1-z)Fe(z)O3 | -> | pO2 = 0.213 bar |
| Sr(0.5)Ba(0.5)Ti(0.5)Fe(0.5)O3 | -> | pO2 = 0.213 bar |
| Sr(1-x)Ba(x)Ti(1-y)V(y)O3 | -> | pO2 = to bar |
| Sr(1-x)Ba(x)Ti(1-y-z)V(y)Fe(z)O3 | -> | pO2 = bar |
| Input x-value equal 0.0 (no Ba2+ on the A-site) or > 0.0 and <= 0.5: |
| Input y-value equal 0.0 (SrTiO3) or 1.0 (SrVO3) or > 0.0 and <= 0.5 (SrTi(1-y)V(y)O3): |
| Input z-value equal 0.0 (no Fe2+/3+ on the B-site) or > 0.0 and <= 0.5 or <= 0.8 |
| (only SrTi(1-z)Fe(z)O3): |
| Input temperature T (K): |
| Input oxygen partial pressure pO2 (bar): |
Appendix B
| Phase | = | SrTiO3 | |
| User input | x | = | 0.000000 |
| User input | y | = | 0.000000 |
| User input | z | = | 0.000000 |
| User input | T | = | 973.000000 K |
| User input | pO2 | = | bar |
| Cubic crystal structure | SG | = | |
| Cell constant | a | = | 3.918795 Å |
| Volume of the unit cell | V | = | 60.180762 Å |
| Atomic number density | N | = | 0.083083 atoms/Å |
| Tolerance factor | t | = | 1.014169 |
| O-O distance (1. order) | = | 2.771007 Å | |
| O-O distance (2. order) | = | 3.918795 Å | |
| Ti-O distance | = | 1.959398 Å | |
| Sr-O distance | = | 2.771007 Å | |
| Ti-Ti distance | = | 3.918795 Å | |
| Sr-Sr distance | = | 3.918795 Å | |
| Ti-Sr distance | = | 3.393776 Å |
| Critical radius | r(c) | = | 0.895343 Å |
| Free volume | FV | = | 15.827530 Å |
| O2- diffusion saddle point | ODSP | = | 0.439857 |
| Total conductivity | Sigma(t) | = | S/cm |
| Electronic conductivity | Sigma(e-) | = | S/cm |
| Oxygen conductivity | Sigma(O2-) | = | S/cm |
References
- Deibert, W.; Ivanova, M.E.; Baumann, S.; Guillon, O.; Meulenberg, W.A. Ion-conducting ceramic membrane reactors for high-temperature applications. J. Membr. Sci. 2017, 543, 79–97. [Google Scholar] [CrossRef]
- Hashim, S.M.; Mohamed, A.R.; Bhatia, S. Current status of ceramic-based membranes for oxygen separation from air. Adv. Colloid Interface Sci. 2010, 160, 88–100. [Google Scholar] [CrossRef] [PubMed]
- Hashim, S.M.; Mohamed, A.R.; Bhatia, S. Oxygen separation from air using ceramic-based membrane technology for sustainable fuel production and power generation. Renew. Sustain. Energy Rev. 2011, 15, 1284–1293. [Google Scholar] [CrossRef]
- Hwang, J.; Rao, R.R.; Giordano, L.; Katayama, Y.; Yu, Y.; Shao-Horn, Y. Perovskites in catalysis and electrocatalysis. Science 2017, 358, 751–756. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sunarso, J.; Baumann, S.; Serra, J.M.; Meulenberg, W.A.; Liu, S.; Lin, Y.S.; da Costa, J.C.D. Mixed ionic-electronic conducting (MIEC) ceramic-based membranes for oxygen separation. J. Membr. Sci. 2008, 320, 13–41. [Google Scholar] [CrossRef]
- Sunarso, J.; Hashim, S.S.; Zhu, N.; Zhou, W. Perovskite oxides applications in high temperature oxygen separation, solid oxide fuel cell and membrane reactor: A review. Prog. Energy Combust. Sci. 2017, 61, 57–77. [Google Scholar] [CrossRef]
- Ubic, R.; Tolman, K.; Chan, K.; Lundy, N.; Letourneau, S.; Kriven, W.M. Effective size of vacancies in aliovalently doped SrTiO3 perovskites. J. Alloys Compd. 2013, 575, 239–245. [Google Scholar] [CrossRef]
- Ubic, R.; Tolman, K.; Talley, K.; Joshi, B.; Schmidt, J.; Faulkner, E.; Owens, J.; Papac, M.; Garland, A.; Rumrill, C.; et al. Lattice-constant prediction and effect of vacancies in aliovalently doped perovskites. J. Alloys Compd. 2015, 644, 982–995. [Google Scholar] [CrossRef] [Green Version]
- Schulze-Küppers, F.; ten Donkelaar, S.F.P.; Baumann, S.; Prigorodov, P.; Sohn, Y.J.; Bouwmeester, H.J.M.; Meulenberg, W.A.; Guillon, O. Structural and functional properties of SrTi1−xFexO3−δ (0 ≤ x ≤ 1) for the use as oxygen transport membrane. Sep. Purif. Technol. 2015, 147, 414–421. [Google Scholar] [CrossRef]
- Unger, L.-S.; Niedrig, C.; Wagner, S.F.; Menesklou, W.; Baumann, S.; Meulenberg, W.A.; Ivers-Tiffée, E. Yttrium doping of Ba0.5Sr0.5Co0.8Fe0.2OO3−δ part I: Influence on oxygen permeation, electrical properties, reductive stability, and lattice parameters. J. Eur. Ceram. Soc. 2018, 38, 2378–2387. [Google Scholar] [CrossRef]
- Unger, L.-S.; Ruhl, R.; Meffert, M.; Niedrig, C.; Menesklou, W.; Wagner, S.F.; Gerthsen, D.; Bouwmeester, H.J.M.; Ivers-Tiffée, E. Yttrium doping of Ba0.5Sr0.5Co0.8Fe0.2OO3−δ part II: Influence on oxygen transport and phase stability. J. Eur. Ceram. Soc. 2018, 38, 2388–2395. [Google Scholar] [CrossRef]
- Meulenberg, W.A.; Schulze-Küppers, F.; Deibert, W.; Van Gestel, T.; Baumann, S. Ceramic Membranes: Materials—Components—Potential Applications. ChemBioEng Rev. 2019, 6, 198–208. [Google Scholar] [CrossRef]
- Zhu, X.; Yang, W. Perovskite-type MIEC membranes. In Mixed Conducting Ceramic Membranes; Springer: Berlin, Germany, 2017; pp. 179–226. [Google Scholar]
- De Souza, R.A. Oxygen Diffusion in SrTiO3 and Related Perovskite Oxides. Adv. Funct. Mater. 2015, 25, 6326–6342. [Google Scholar] [CrossRef]
- Khan, T.T.; Mahmud, I.; Ur, S.-C. Synthesis and Thermoelectric Properties of the B-Site Substituted SrTiO3 with Vanadium. Korean J. Mater. Res. 2017, 27, 416–421. [Google Scholar] [CrossRef]
- Liu, Y.; Baumann, S.; Schulze-Küppers, F.; Mueller, D.N.; Guillon, O. Co and Fe co-doping influence on functional properties of SrTiO3 for use as oxygen transport membranes. J. Eur. Ceram. Soc. 2018, 38, 5058–5066. [Google Scholar] [CrossRef]
- Mantry, S.P.; Yadav, A.; Fahad, M.; Sarun, P.M. Effect of vanadium substitution on the dielectric and electrical conduction properties of SrTiO3 ceramics. Mater. Res. Express 2018, 5, 036303. [Google Scholar] [CrossRef]
- Teraoka, Y.; Nobunaga, T.; Yamazoe, N. Effect of Cation Substitution on the Oxygen Semipermeability of Perovskite-type Oxides. Chem. Lett. 1988, 3, 503–506. [Google Scholar] [CrossRef] [Green Version]
- Shannon, R.D.; Prewitt, C.T. Effective Ionic Radii in Oxides and Fluorides. Acta Cryst. 1969, B25, 925–946. [Google Scholar] [CrossRef]
- Sammels, A.F.; Cook, R.F.; White, J.H.; Osborne, J.J.; MacDuff, R.C. Rational selection of advanced solid electrolytes for intermediate temperature fuel cells. Solid State Ion. 1992, 52, 111–123. [Google Scholar] [CrossRef]
- Kawada, T.; Watanabe, T.; Kaimai, A.; Kawamura, K.; Nigara, Y.; Mizusaki, J. High temperature transport properties in SrTiO3 under an oxygen potential gradient. Solid State Ion. 1998, 108, 391–402. [Google Scholar] [CrossRef]
- Hayashi, H.; Inaba, H.; Matsuyama, M.; Lan, N.G.; Dokiya, M.; Tagawa, H. Structural consideration on the ionic conductivity of perovskite-type oxides. Solid State Ion. 1999, 122, 1–15. [Google Scholar] [CrossRef]
- Ohly, C.; Hoffmann-Eifert, S.; Szot, K.; Waser, R. Electrical conductivity and segregation effects of doped SrTiO3 thin films. J. Eur. Ceram. Soc. 2001, 21, 1673–1676. [Google Scholar] [CrossRef]
- Schmidbauer, M.; Kwasniewski, A.; Schwarzkopf, J. High-precision absolute lattice parameter determination of SrTiO3, DyScO3 and NdGaO3 single crystals. Acta Cryst. 2012, B68, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Yaremchenko, A.A.; Brinkmann, B.; Janssen, R.; Frade, J.R. Electrical conductivity, thermal expansion and stability of Y- and Al-substituted SrVO3 as prospective SOFC anode material. Solid State Ion. 2013, 247–248, 86–93. [Google Scholar] [CrossRef]
- De Souza, R.A.; Ramadan, A.H.H. Ionic conduction in the SrTiO3|YSZ|SrTiO3 heterostructure. Phys. Chem. Chem. Phys. 2013, 15, 4505–4509. [Google Scholar] [CrossRef] [PubMed]
- Macias, J.; Yaremchenko, A.A.; Frade, J.R. Redox transition in strontium vanadates: Electrical conductivity and dimensional changes. J. Alloys Compd. 2014, 601, 186–194. [Google Scholar] [CrossRef]
- Metlenko, V.; Ramadan, A.H.H.; Gunkel, F.; Du, H.; Schraknepper, H.; Hoffmann-Eifert, S.; Dittmann, R.; Waser, R.; De Souza, R.A. Do dislocations act as atomic autobahns for oxygen in the perovskite oxide SrTiO3? Nanoscale 2014, 6, 12864–12876. [Google Scholar] [CrossRef]
- Schie, M.; Waser, R.; De Souza, R.A. A Simulation Study of Oxygen-Vacancy Behavior in Strontium Titanate: Beyond Nearest-Neighbor Interactions. J. Phys. Chem. C 2014, 118, 15185–15192. [Google Scholar] [CrossRef]
- Schulz, M.; Orland, T.; Mehlmann, A.; Rothschild, A.; Fritze, H. Oxygen transport in epitaxial SrTiO3/SrTi1-xFexO3 multilayer stacks. J. Sens. Sens. Syst. 2017, 6, 107–119. [Google Scholar] [CrossRef] [Green Version]
- Liu, Z.T.Y.; Podraza, N.J.; Khare, S.V.; Sarin, P. Transparency enhancement for SrVO3 by SrTiO3 mixing: A first-principles study. Comput. Mater. Sci. 2018, 144, 139–146. [Google Scholar] [CrossRef]
- Macias, J.; Yaremchenko, A.A.; Rodriguez-Castellon, E.; Starykevich, M.; Frade, J.R. Compromising Between Phase Stability and Electrical Performance: SrVO3-SrTiO3 Solid Solutions as Solid Oxide Fuel Cell Anode Components. ChemSusChem 2019, 12, 240–251. [Google Scholar] [CrossRef] [PubMed]
- Kolotygin, V.A.; Viskup, A.P.; Pivak, E.V.; Kharton, V.V. The Mixed Electronic and Ionic Conductivity of Perovskite-Like Ba1−xSrxFe1−yTiyO3−δ and BaTi0.5Fe0.5−zCezO3−δ Solid Solutions. Russ. J. Electrochem. 2020, 56, 110–117. [Google Scholar] [CrossRef]
- Brese, N.E.; O’Keeffe, M. Bond-Valence Parameters For Solids. Acta Cryst. 1991, B47, 192–197. [Google Scholar] [CrossRef]
- Brown, I.D.; Poppelmeier, K.R. Bond Valences; Springer: Berlin, Germany, 2014. [Google Scholar]
- Brown, I.D. The Chemical Bond in Inorganic Chemistry: The Bond Valence Model; Oxford University Press: Oxford, UK, 2016. [Google Scholar]
- Gagné, O.C.; Hawthorne, F.C. Visualizing Lithium-Ion Migration Pathways in Battery Materials. Acta Cryst. 2015, B71, 562–578. [Google Scholar]
- Filsø, M.O.; Turner, M.J.; Gibbs, G.V.; Adams, S.; Spackmann, M.A.; Iversen, B.B. Visualizing Lithium-Ion Migration Pathways in Battery Materials. Chem. Eur. J. 2013, 19, 15535–15544. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Chu, G.; He, M.; Zhang, S.; Xiao, R.; Li, H.; Chen, L. Screening possible solid electrolytes by calculating the conduction pathways using Bond Valence method. Sci. China Phys. Mech. Astron. 2014, 57, 1526–1535. [Google Scholar] [CrossRef]
- Inoue, R.; Fuji, K.; Shiraiwa, M.; Niwa, E.; Yashima, M. A new structure family of oxide-ion conductors Ca0.8Y2.4Sn0.8O6 discovered by a combined technique of the bond-valence method and experiments. Dalton Trans. 2018, 47, 7515–7521. [Google Scholar] [CrossRef]
- Sale, M.; Avdeev, M. 3DBVSMAPPER: A program for automatically generating bond-valence sum landscapes. J. Appl. Cryst. 2012, 45, 1054–1056. [Google Scholar] [CrossRef]
- Yamada, I.; Takamatsu, A.; Ikeno, H. Complementary evaluation of structure stability of perovskite oxides using bond-valence and density-functional-theory calculations. Sci. Technol. Adv. Mater. 2018, 19, 101–107. [Google Scholar] [CrossRef] [Green Version]
- Lufaso, M.W.; Woodward, P.M. Prediction of the Crystal Structures of Perovskites Using the Software Program SPuDS. Acta Cryst. 2001, B57, 725–738. [Google Scholar] [CrossRef] [Green Version]
- Lufaso, M.W.; Barness, P.W.; Woodward, P.M. Structure prediction of ordered and disordered multiple octahedral cation perovskites using SPuDS. Acta Cryst. 2006, B62, 397–410. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Witten, I.H.; Frank, E.; Hall, M.A.; Pal, C.J. Data Mining, Practical Machine Learning Tools and Techniques, 4th ed.; Elsevier: Cambridge, MA, USA, 2017. [Google Scholar]
- Witten, I.H.; Frank, E.; Hall, M.A.; Pal, C.J. The WEKA Workbench, 4th ed.; Morgan Kaufmann: Burlington, NJ, USA, 2016. [Google Scholar]
- Kotthoff, L.; Thornton, C.; Hoos, H.H.; Hutter, F.; Leyton-Brown, K. Auto-WEKA 2.0: Automatic model selection and hyperparameter optimization in WEKA. J. Mach. Learn. Res. 2017, 18, 1–5. [Google Scholar]
- Aha, D.; Kibler, D. Instance-based learning algorithms. Mach. Learn. 1991, 6, 37–66. [Google Scholar] [CrossRef] [Green Version]
- Chang, C.-C.; Lin, C.-J. LIBSVM—A Library for Support Vector Machines. 2001. Available online: http://www.csie.ntu.edu.tw/cjlin/libsvm/ (accessed on 1 April 2022).
- Yasser, E.-M. WLSVM. 2005. Available online: http://www.cs.iastate.edu/yasser/wlsvm/ (accessed on 1 April 2022).
- Liu, Y.; Zhao, T.; Ju, W.; Shi, S. Materials discovery and design using machine learning. J. Mater. 2017, 3, 159–177. [Google Scholar] [CrossRef]
- Wang, A.Y.-T.; Murdock, R.J.; Kauwe, S.K.; Oliynyk, A.O.; Gurlo, A.; Brgoch, J.; Persson, K.A.; Sparks, T.D. Machine Learning for Materials Scientists: An introductory Guide toward Best Practices. Chem. Mater. 2020, 32, 4954–4965. [Google Scholar] [CrossRef]
- Butler, K.T.; Davies, D.W.; Cartwright, H.; Isayev, O.; Walsh, A. Machine learning for molecular and materials science. Nature 2018, 559, 547–555. [Google Scholar] [CrossRef]
- Schleder, G.R.; Padilha, A.C.M.; Acosta, C.M.; Costa, M.; Fazzio, A. From DFT to machine learning: Recent approaches to materials science—A review. J. Phys. Mater. 2019, 2, 032001. [Google Scholar] [CrossRef]
- Schmidt, J.; Marques, M.R.G.; Botti, S.; Marques, M.A.L. Recent advances and applications of machine learning in solid-state materials science. NPJ Comput. Mater. 2019, 5, 83. [Google Scholar] [CrossRef]
- Chen, C.; Zuo, Y.; Ye, W.; Li, X.; Deng, Z.; Ong, S.P. A Critical Review of Machine Learning of Energy Materials. Adv. Energy Mater. 2020, 10, 1903242. [Google Scholar] [CrossRef]
- Xu, L.; Wencong, L.; Chunrong, P.; Qiang, S.; Jin, G. Two semi-empirical approaches for the prediction of oxide ionic conductivities in ABO3 perovskites. Comput. Mater. Sci. 2009, 46, 860–868. [Google Scholar] [CrossRef]
- Dai, D.; Liu, Q.; Hu, R.; Wei, X.; Ding, G.; Xu, B.; Xu, T.; Zhang, J.; Xu, Y.; Zhang, H. Method construction of structure-property relationships from data by machine learning assisted mining for materials design applications. Mater. Des. 2020, 196, 109194. [Google Scholar] [CrossRef]
- Mistry, A.; Franco, A.A.; Cooper, S.J.; Roberts, S.A.; Viswanathan, V. How Machine Learning Will Revolutionize Electrochemical Sciences. ACS Energy Lett. 2021, 6, 1422–1431. [Google Scholar] [CrossRef]
- Zhang, L.; He, B.; Zhao, Q.; Zou, Z.; Chi, S.; Mi, P.; Ye, A.; Li, Y.; Wang, D.; Avdeev, M.; et al. A Database of Ionic Transport Characteristics for Over 29,000 Inorganic Compounds. Adv. Funct. Mater. 2020, 2003087. [Google Scholar] [CrossRef]
- Pilania, G.; Balachandran, P.V.; Kim, C.; Lookman, T. Finding New Perovskite Halides via Machine Learning. Front. Mater. 2016, 3, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Balachandran, P.V.; Lin, L.; Anchell, J.S.; Bridges, C.A.; Ganesh, P. Defect Genome of Cubic Perovskites for Fuel Cell Applications. J. Phys. Chem. C 2017, 121, 26637–26647. [Google Scholar] [CrossRef]
- Li, W.; Jacobs, R.; Morgan, D. Predicting the thermodynamic stability of perovskite oxides using machine learning models. Comput. Mater. Sci. 2018, 150, 454–463. [Google Scholar] [CrossRef] [Green Version]
- Yamada, I.; Takamatsu, A.; Asai, K.; Shirakawa, T.; Ohzuku, H.; Seno, A.; Uchimura, T.; Fujii, H.; Kawaguchi, S.; Wada, K.; et al. Systematic Study of Descriptors for Oxygen Evolution Reaction Catalysis in Perovskite Oxides. J. Phys. Chem. C 2018, 122, 27885–27892. [Google Scholar] [CrossRef] [Green Version]
- Xu, Q.; Li, Z.; Liu, M.; Yin, W.-J. Rationalizing Perovskite Data for Machine Learning and Materials Design. J. Phys. Chem. Lett. 2018, 9, 6948–6954. [Google Scholar] [CrossRef]
- Li, J.; Pradhan, B.; Gaur, S.; Thomas, J. Predictions and Strategies Learned from Machine Learning to Develop High-Performing Perovskite Solar Cells. Adv. Energy Mater. 2019, 9, 1901891. [Google Scholar] [CrossRef]
- Sun, S.; Hartono, N.T.P.; Ren, Z.D.; Oviedo, F.; Buscemi, A.M.; Layurova, M.; Chen, D.X.; Ogunfunmi, T.; Thapa, J.; Ramasamy, S.; et al. Accelerated Development of Perovskite-Inspired Materials via High-Throughput Synthesis and Machine-Learning Diagnosis. Joule 2019, 3, 1437–1451. [Google Scholar] [CrossRef]
- Vieten, J.; Bulfin, B.; Huck, P.; Horton, M.; Guban, D.; Zhu, L.; Lu, Y.; Persson, K.A.; Roeb, M.; Sattler, C. Materials design of perovskite solid solutions for thermochemical applications. Energy Environ. Sci. 2019, 12, 1369–1384. [Google Scholar] [CrossRef]
- Li, Z.; Najeeb, M.A.; Alves, L.; Sherman, A.Z.; Shekar, V.; Parilla, P.C.; Pendleton, I.M.; Wang, W.; Nega, P.W.; Zeller, M.; et al. Robot-Accelerated Perovskite Investigation and Discovery. Chem. Mater. 2020, 32, 5650–5663. [Google Scholar] [CrossRef]
- Srivastava, M.; Howard, J.M.; Gong, T.; Dias, M.R.S.; Leite, M.S. Machine Learning Road map for Perovskite Photovoltaics. J. Phys. Chem. Lett. 2021, 12, 7866–7877. [Google Scholar] [CrossRef] [PubMed]
- Tao, Q.; Xu, P.; Li, M.; Lu, W. Machine Learning for perovskite materials design and discovery. NPJ Comput. Mater. 2021, 7, 23. [Google Scholar] [CrossRef]
- Park, H.; Ali, A.; Mall, R.; Bensmail, H.; Sanvito, S.; El-Mellouhi, F. Data-driven enhancement of cubic phase stability in mixed-cation perovskites. Mach. Learn. Sci. Technol. 2021, 2, 025030. [Google Scholar] [CrossRef]
- Deng, Q.; Lin, B. Exploring structure-composition relationships of cubic perovskite oxides via extreme feature engineering and automated machine learning. Mater. Today Commun. 2021, 28, 102590. [Google Scholar] [CrossRef]
- Chenebuah, E.T.; Nganbe, M.; Tchagang, A.B. Comparative analysis of machine learning approaches on the prediction of the electronic properties of perovskites: A case study of ABX3 and A2BB′X6. Mater. Today Commun. 2021, 27, 102462. [Google Scholar] [CrossRef]
- Sharma, A.; Kushvaha, V. Predictive modeling of fracture behavior in silica-filled polymer composite subjected to impact with varying loading rates using artificial neural network. Eng. Fract. Mech. 2020, 239, 107328. [Google Scholar] [CrossRef]
- Sharma, A.; Mukhopadhyay, T.; Rangappa, S.M.; Siengchin, S.; Kushvaha, V. Advances in Computational Intelligence of Polymer Composite Materials: Machine Learning Assisted Modeling, Analysis and Design. Arch. Comput. Methods Eng. 2022; in press. [Google Scholar] [CrossRef]
- Alam, M.A.; Ya, H.H.; Azeem, M.; Yusuf, M.; Soomro, I.A.; Masood, F.; Shozib, I.A.; Sapuan, S.M.; Akhter, J. Artificial Neural Network Modeling to Predict the Effect of Milling Time and TiC Content on the Crystallite Size and Lattice Strain of Al7075-TiC Composites Fabricated by Powder Metallurgy. Crystals 2022, 12, 372. [Google Scholar] [CrossRef]
- Gómez-Peralta, J.I.; García-Peña, N.G.; Bokhimi, X. Crystal-Site-Based Artificial Neural Networks for Material Classification. Crystals 2021, 11, 1039. [Google Scholar] [CrossRef]


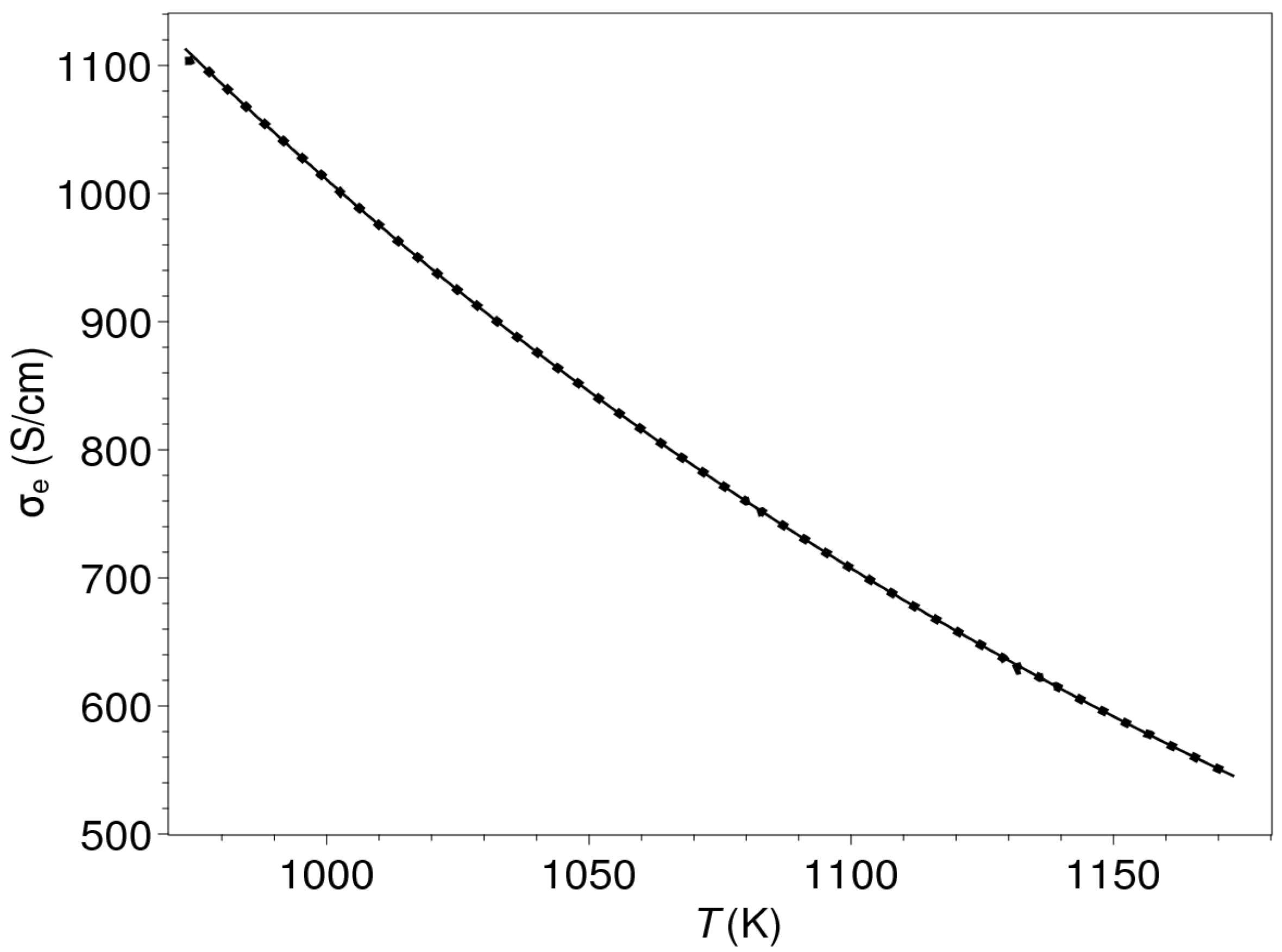

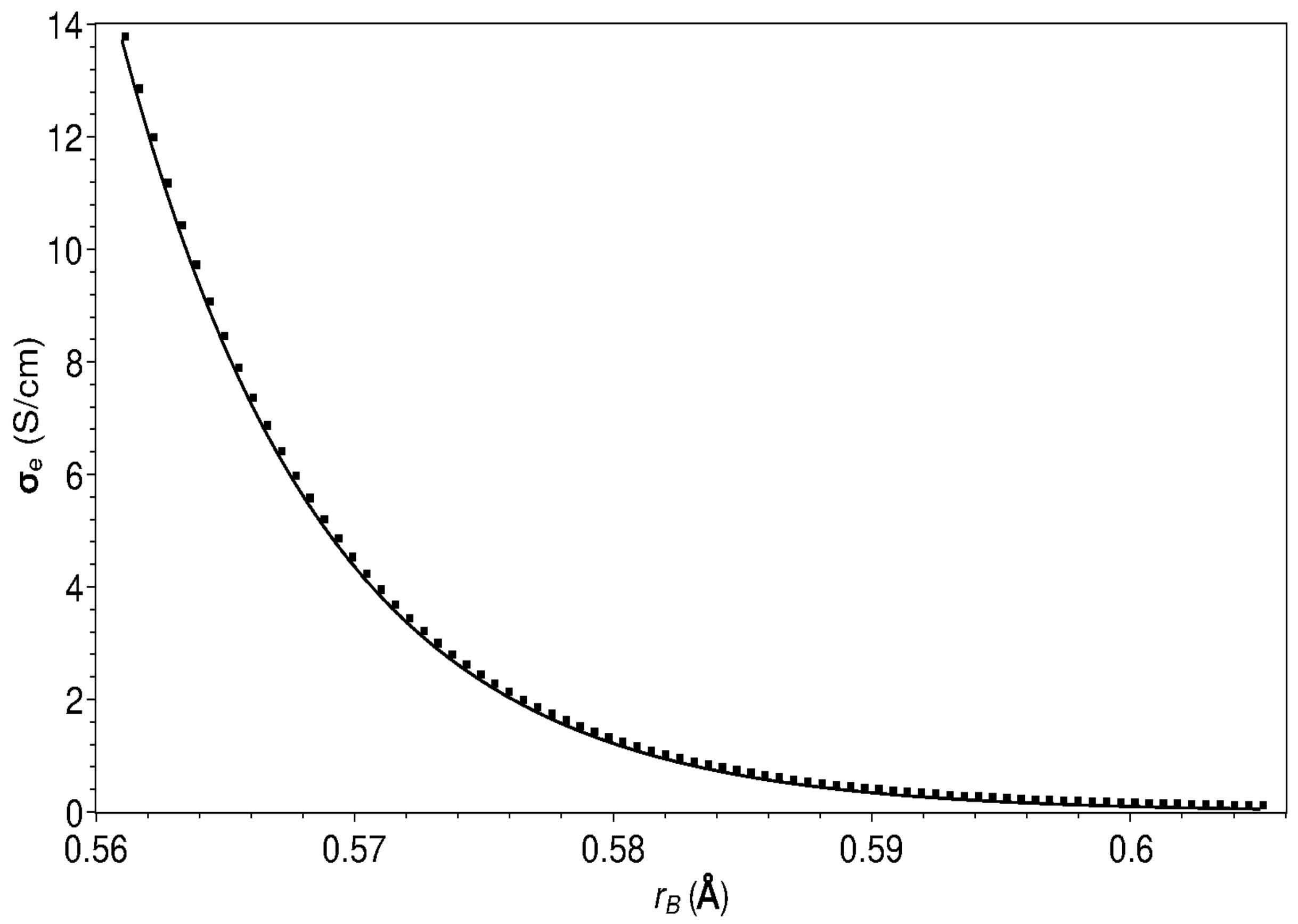

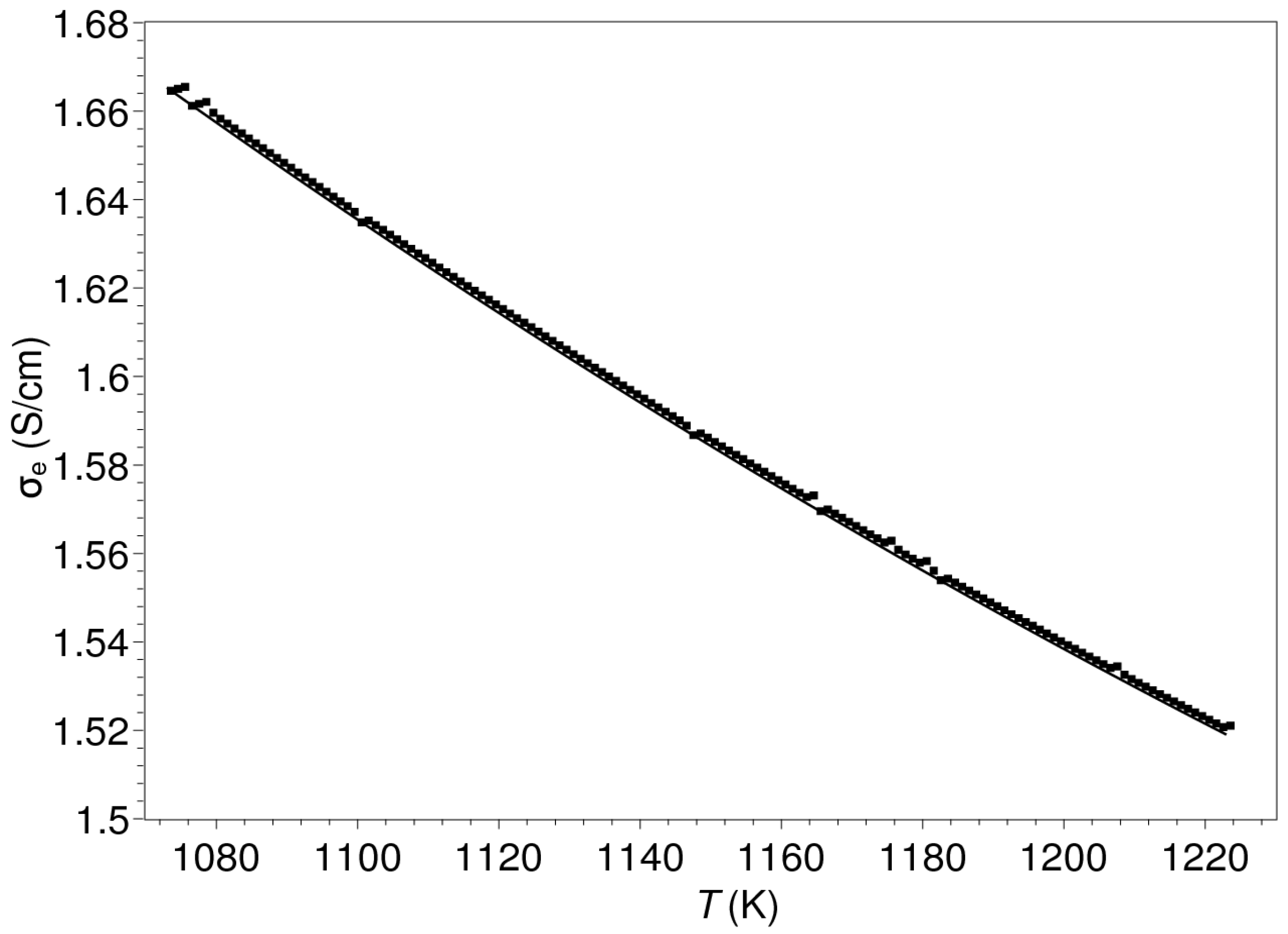
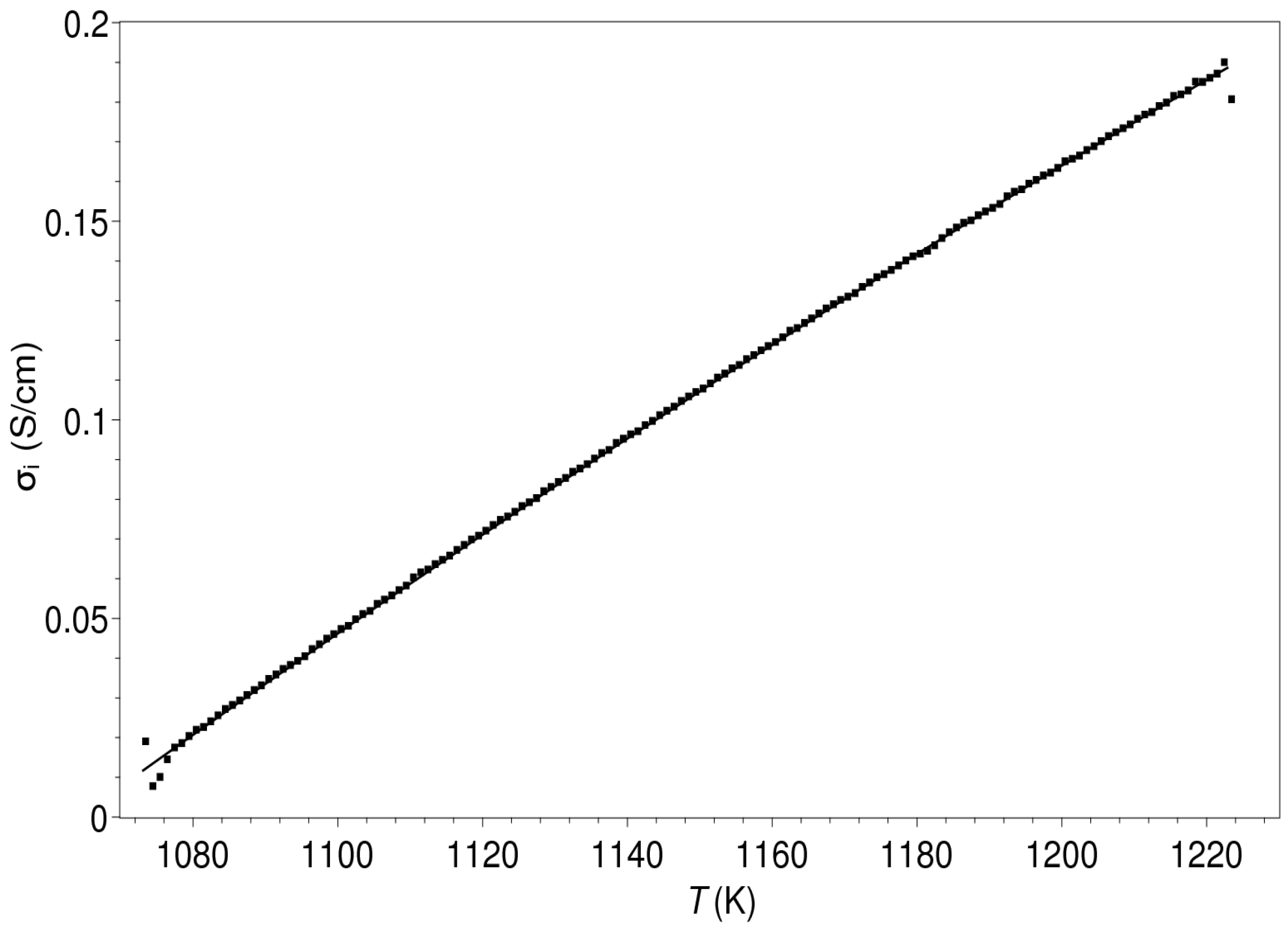

| Composition | ||
|---|---|---|
| SrTiO | YES | YES |
| SrVO | YES | NO |
| YES | NO | |
| YES | YES | |
| YES | YES | |
| NO | NO | |
| NO | NO |
| Composition | (bar) | FV (Å) | ||
|---|---|---|---|---|
| 16.011 | ||||
| 13.377 | 545.09 | |||
| 15.320 | 20.01 | |||
| 0.213 | 13.360 | 1.67 | 0.05 | |
| 0.213 | 17.904 | 1.56 | 0.13 | |
| 16.729 | 1.60 | |||
| 16.827 | 1.59 | |||
| 16.654 | 1.60 | |||
| 16.585 | 1.60 | |||
| 16.757 | 1.59 | |||
| 17.149 | 1.59 | |||
| 16.678 | 1.60 | |||
| 16.860 | 1.59 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schlenz, H.; Baumann, S.; Meulenberg, W.A.; Guillon, O. The Development of New Perovskite-Type Oxygen Transport Membranes Using Machine Learning. Crystals 2022, 12, 947. https://doi.org/10.3390/cryst12070947
Schlenz H, Baumann S, Meulenberg WA, Guillon O. The Development of New Perovskite-Type Oxygen Transport Membranes Using Machine Learning. Crystals. 2022; 12(7):947. https://doi.org/10.3390/cryst12070947
Chicago/Turabian StyleSchlenz, Hartmut, Stefan Baumann, Wilhelm Albert Meulenberg, and Olivier Guillon. 2022. "The Development of New Perovskite-Type Oxygen Transport Membranes Using Machine Learning" Crystals 12, no. 7: 947. https://doi.org/10.3390/cryst12070947
APA StyleSchlenz, H., Baumann, S., Meulenberg, W. A., & Guillon, O. (2022). The Development of New Perovskite-Type Oxygen Transport Membranes Using Machine Learning. Crystals, 12(7), 947. https://doi.org/10.3390/cryst12070947






