Entrapped Transient Chloroform Solvates of Bilastine
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Bilastine Crystal Forms
2.1.1. Form I
2.1.2. Form II
2.1.3. Form III
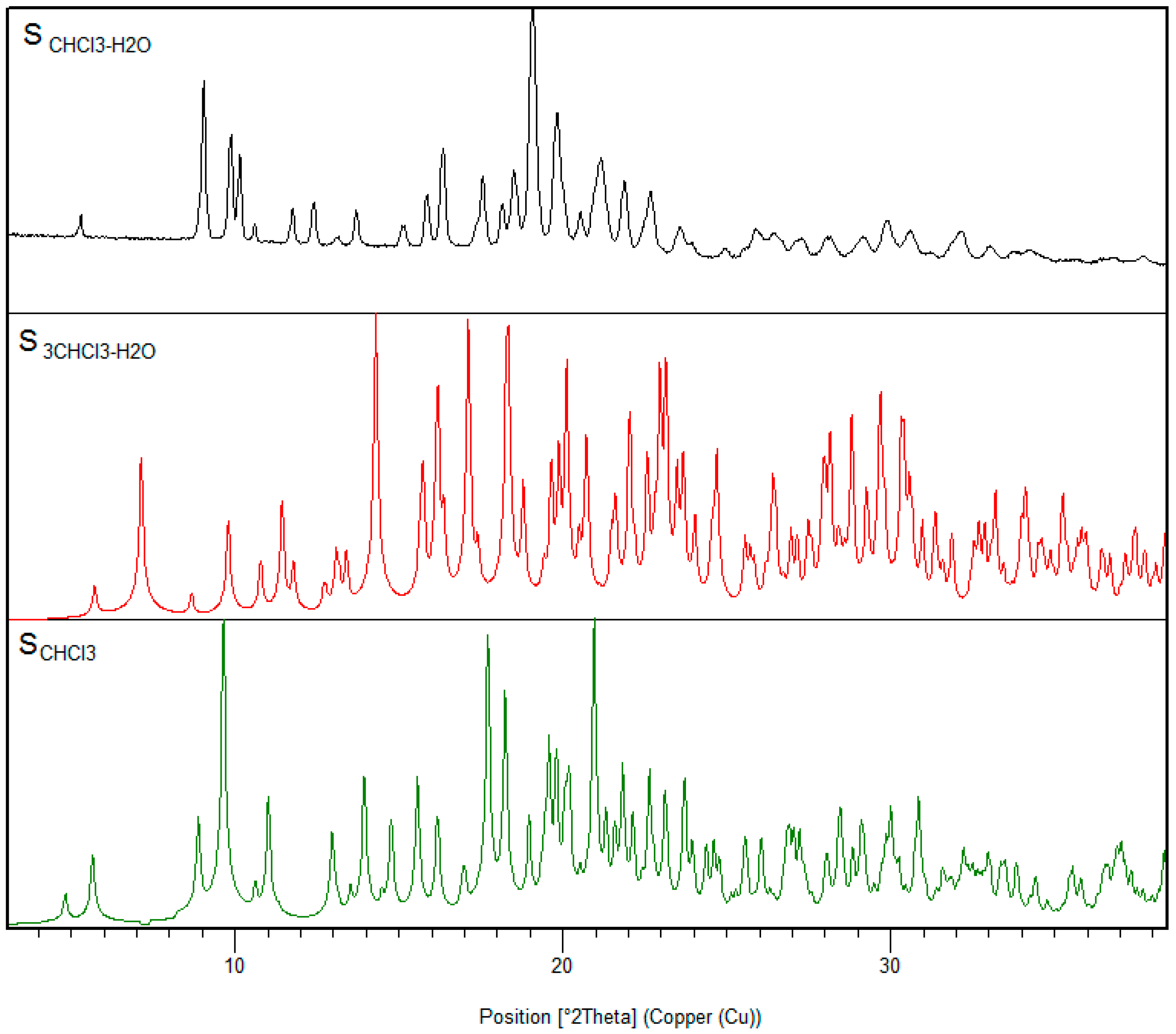
2.1.4. S3CHCl3-H2O
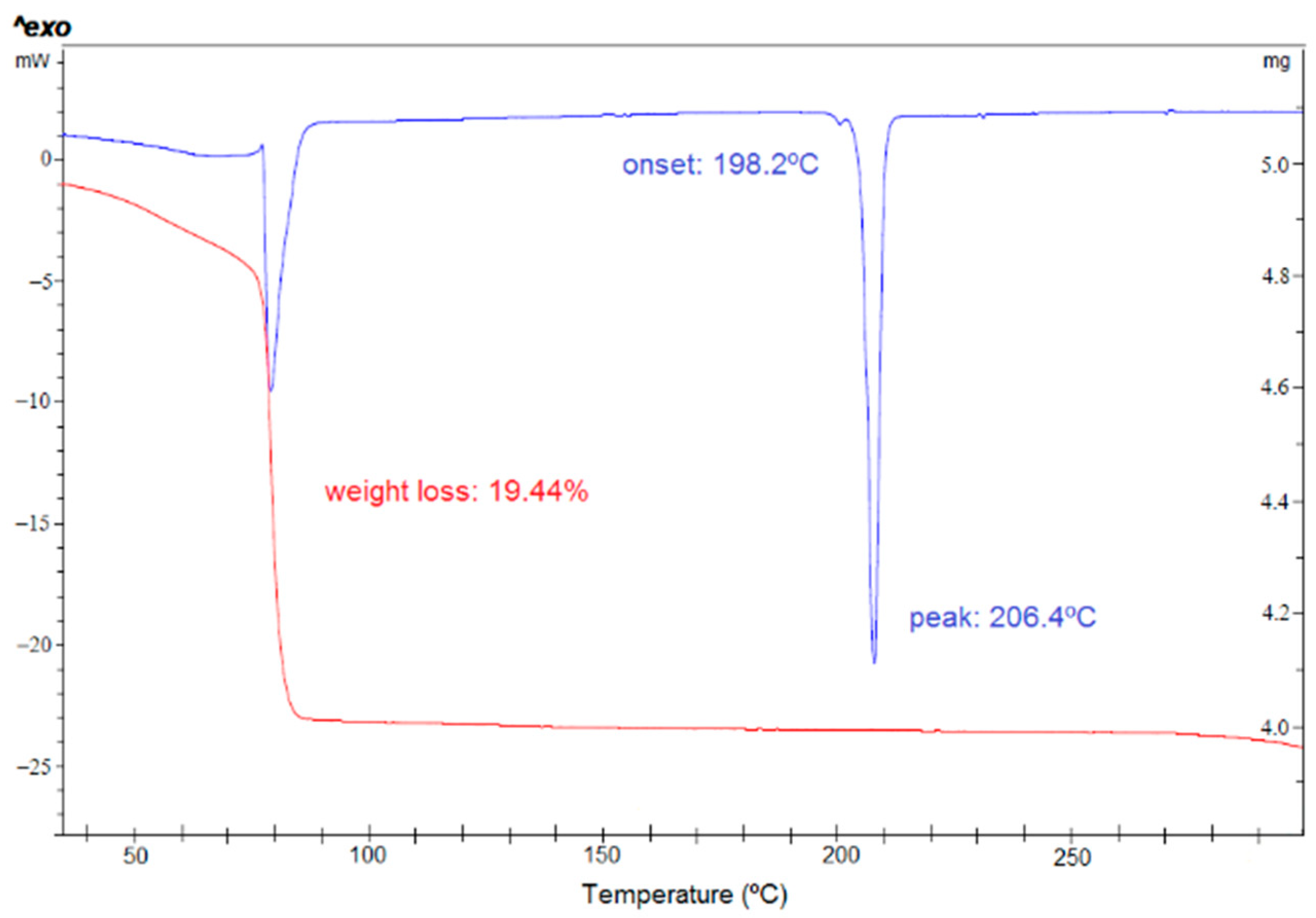
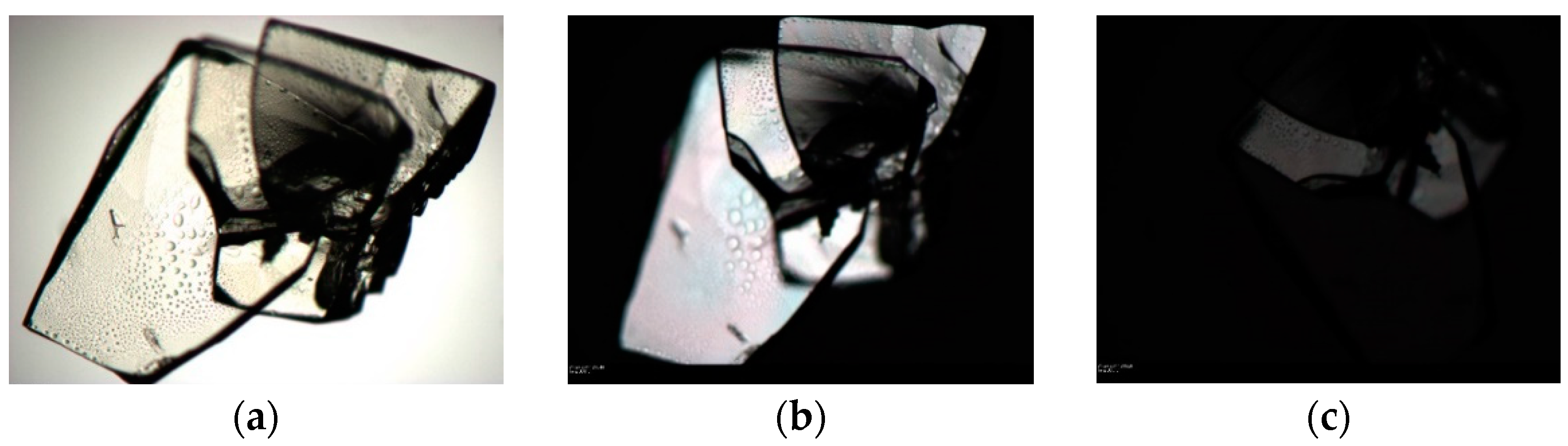
2.1.5. SCHCl3-H2O
2.1.6. SCHCl3
2.2. Methods
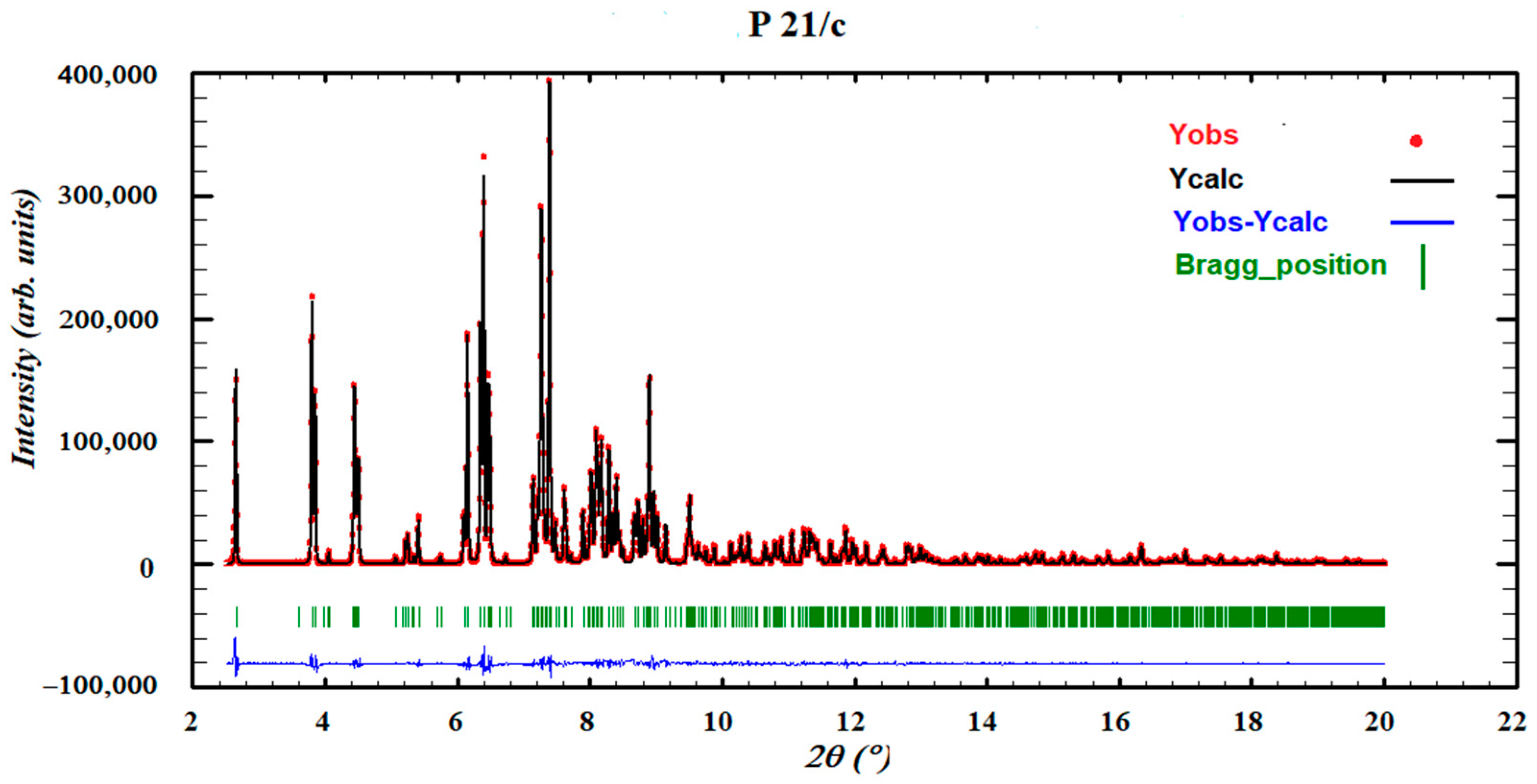
2.2.1. Powder X-ray Diffraction (PXRD)
2.2.2. Single Crystal X-ray Diffraction (SCXRD)
2.2.3. Differential Scanning Calorimetry (DSC)
2.2.4. Thermogravimetric Analysis (TGA)
3. Results and Discussion
3.1. Anhydrous Forms
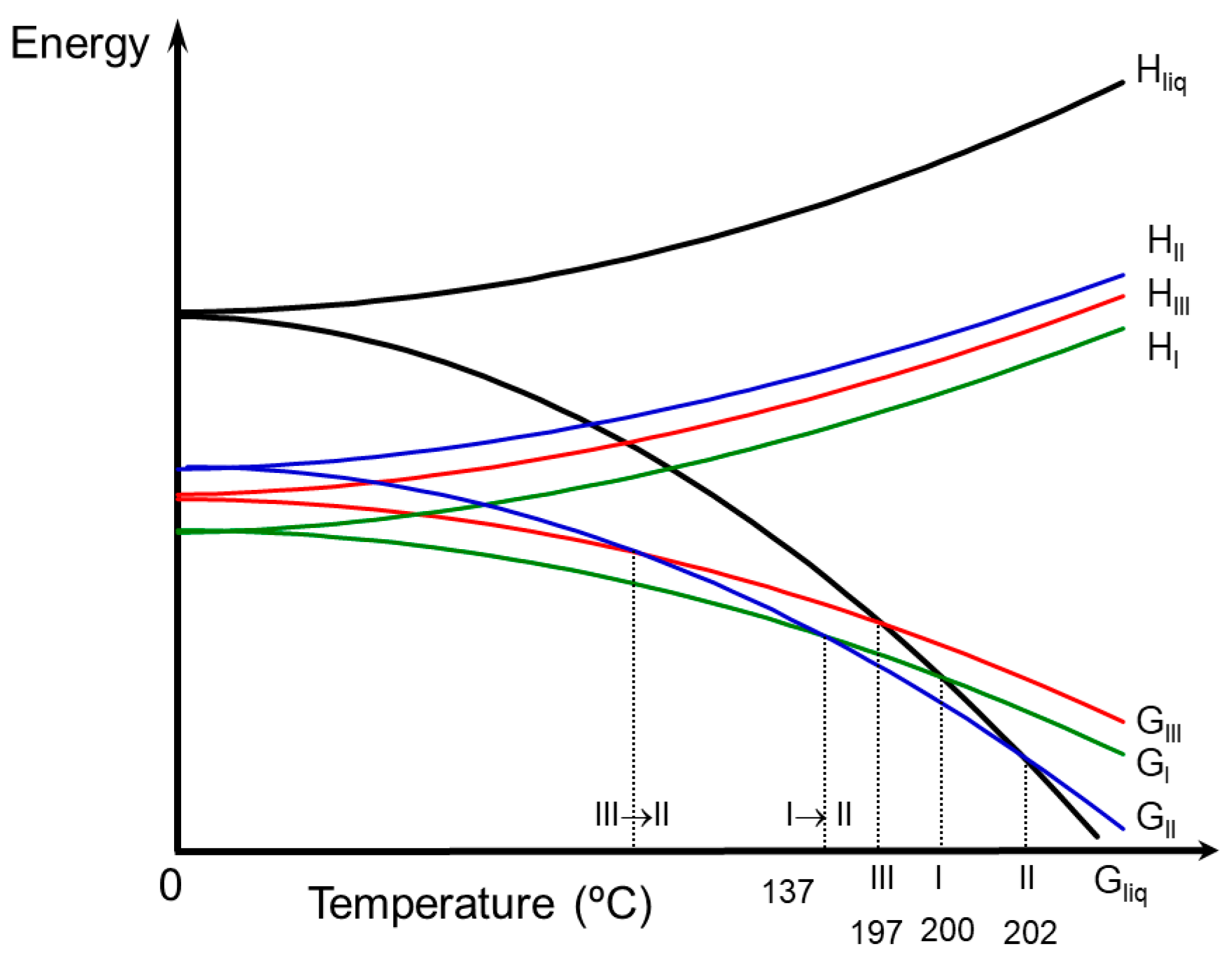
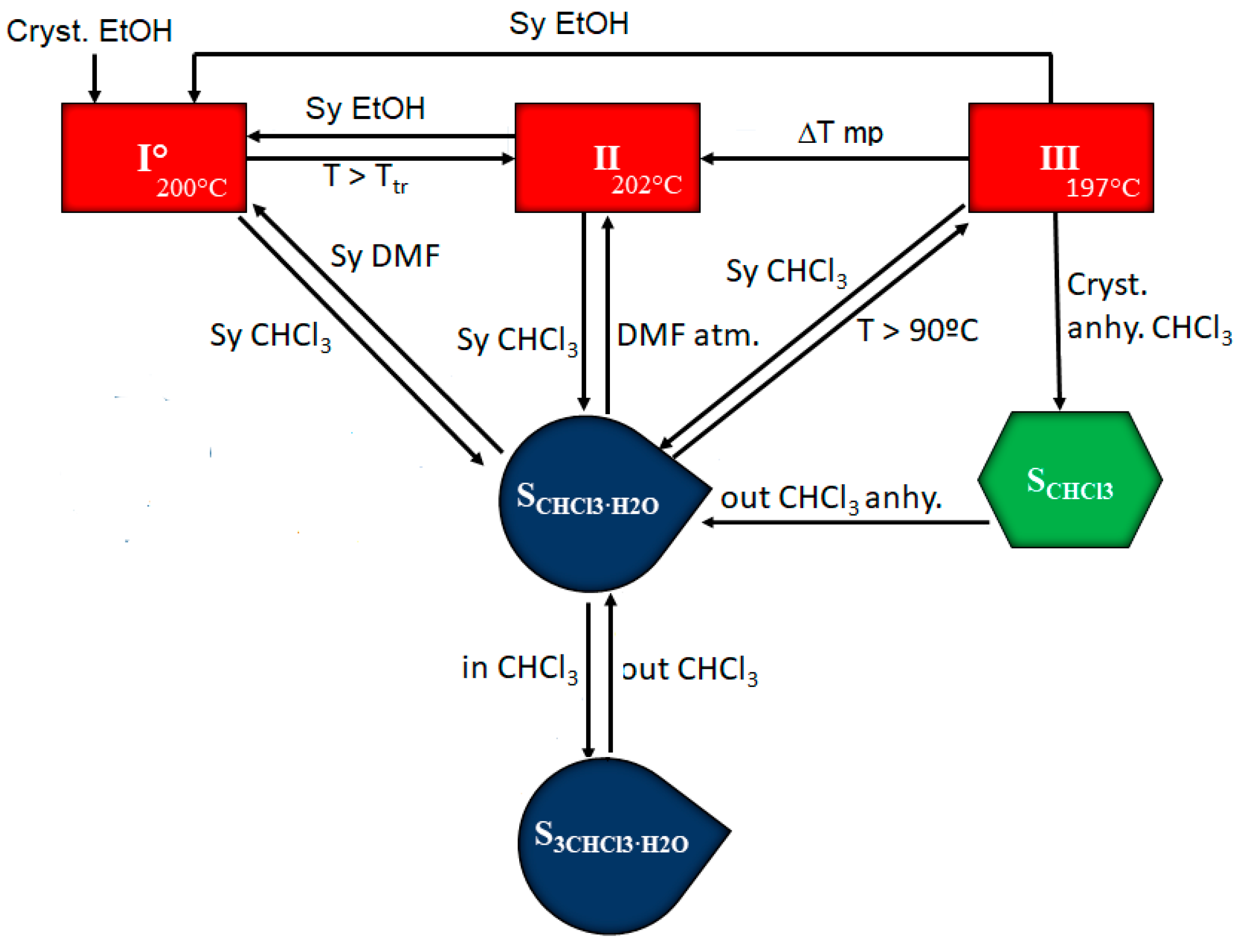
3.2. Relative Thermodynamic Stability among the Anhydrous Solid Forms
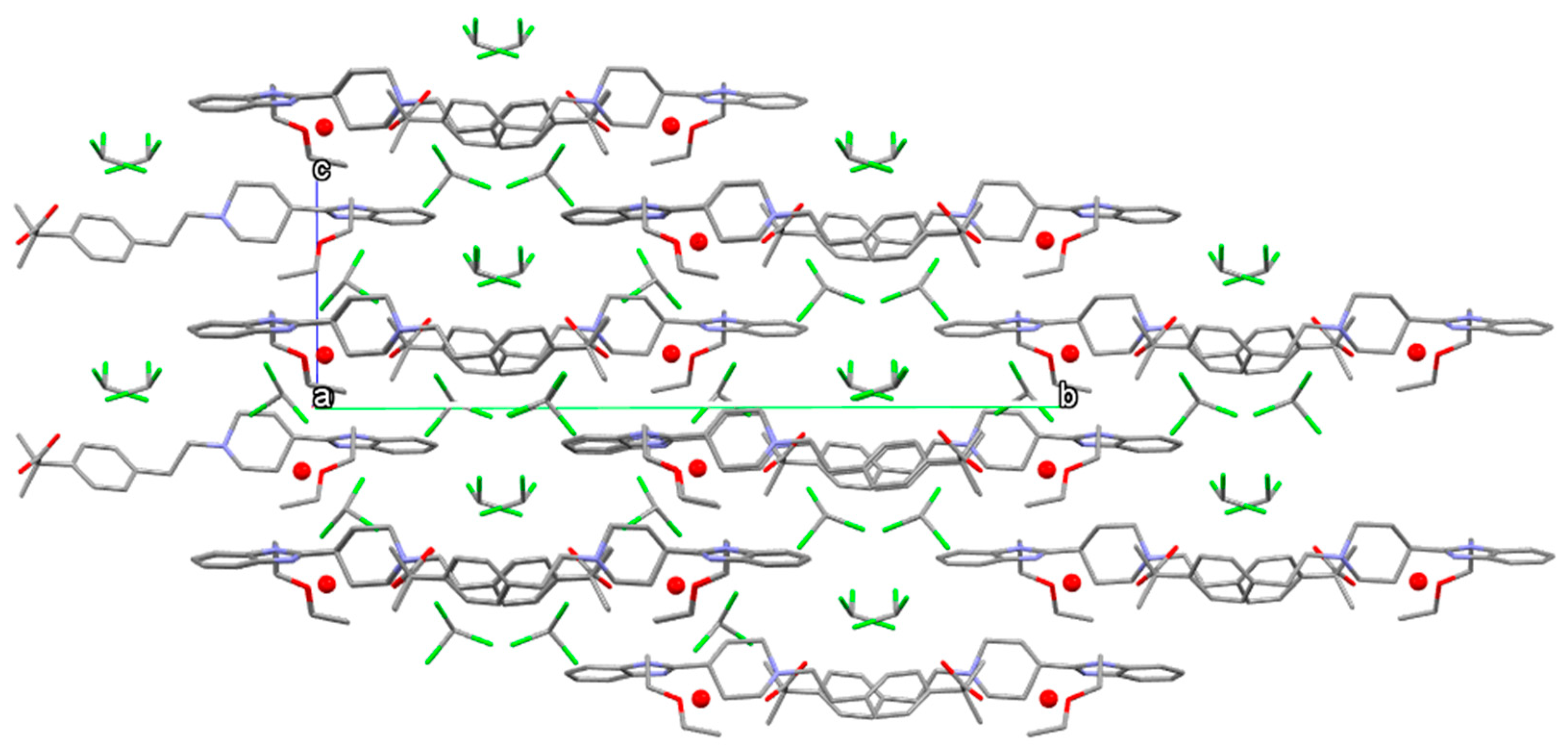
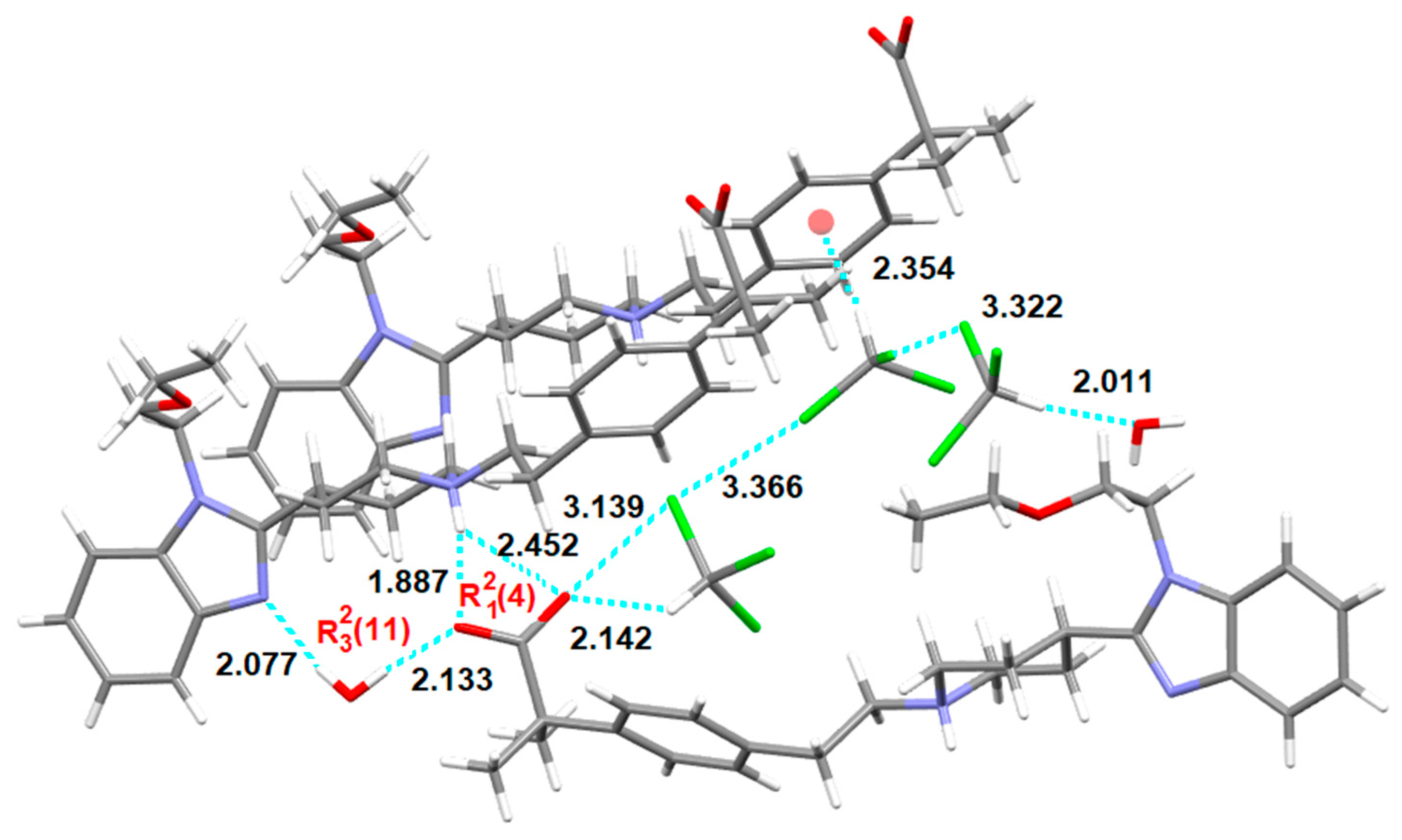
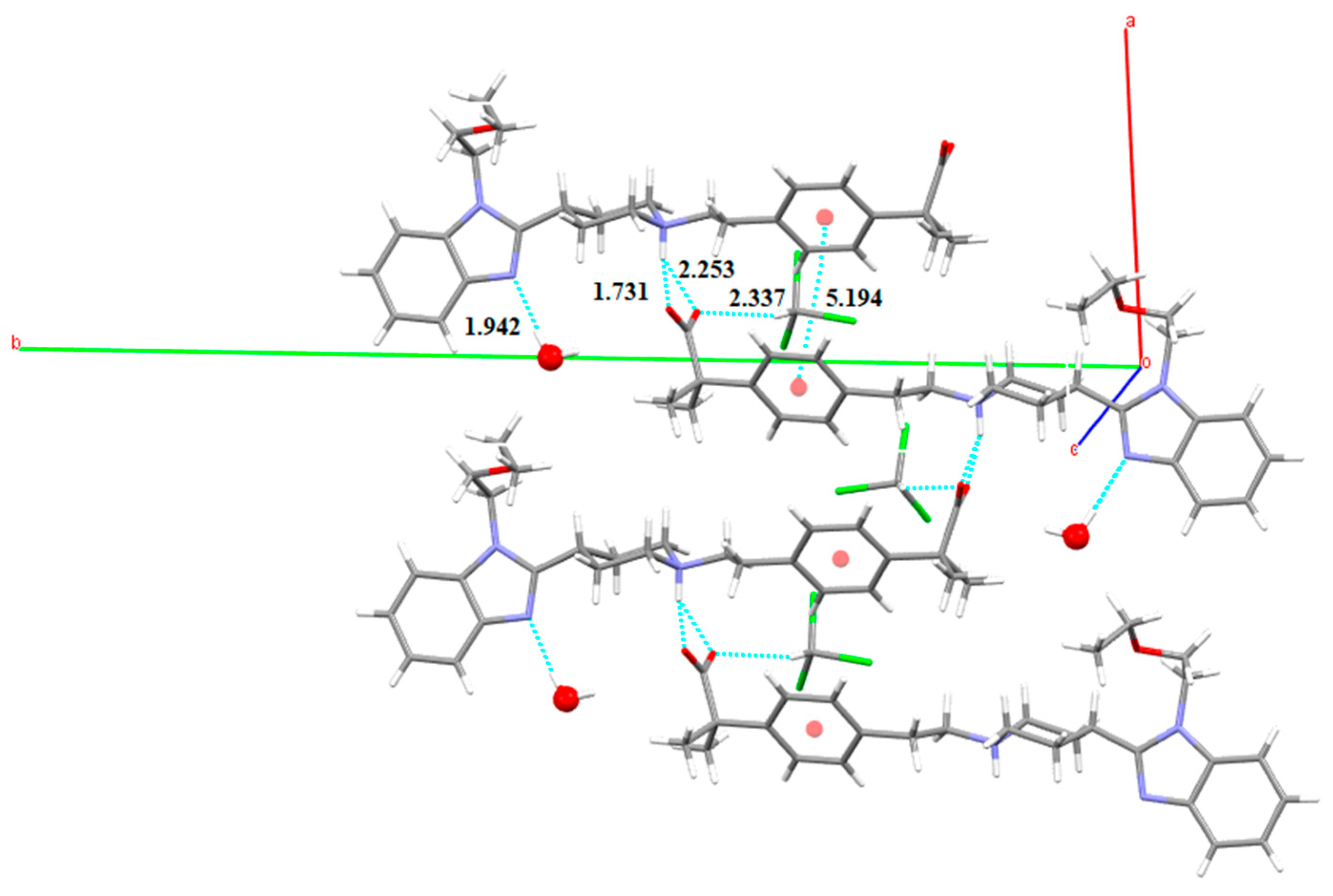
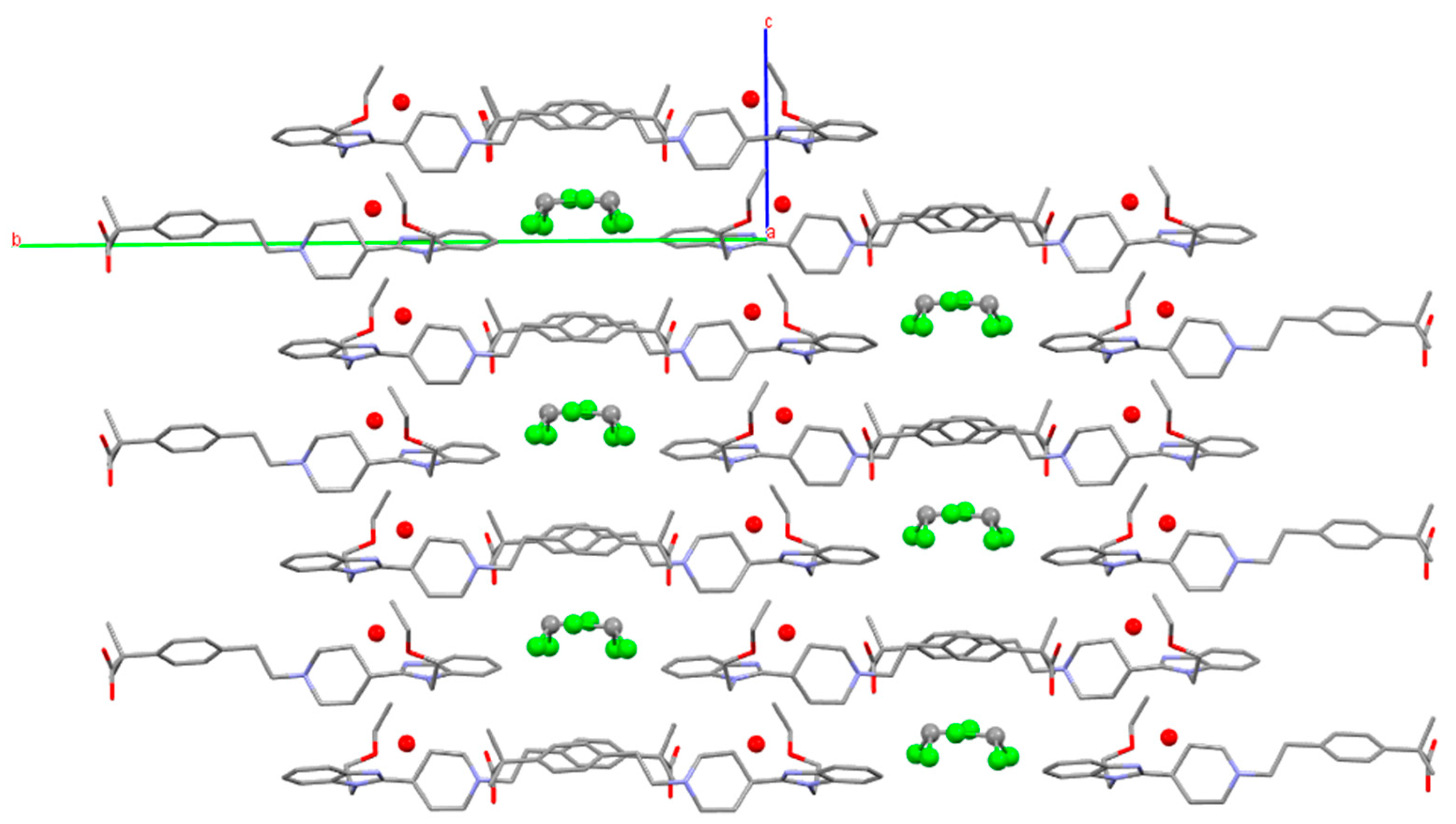
3.3. Chloroform Solvates
3.4. Stability and Interrelation Pathways among the Solvates and Anhydrous Forms
3.5. Crystal Structures Analysis
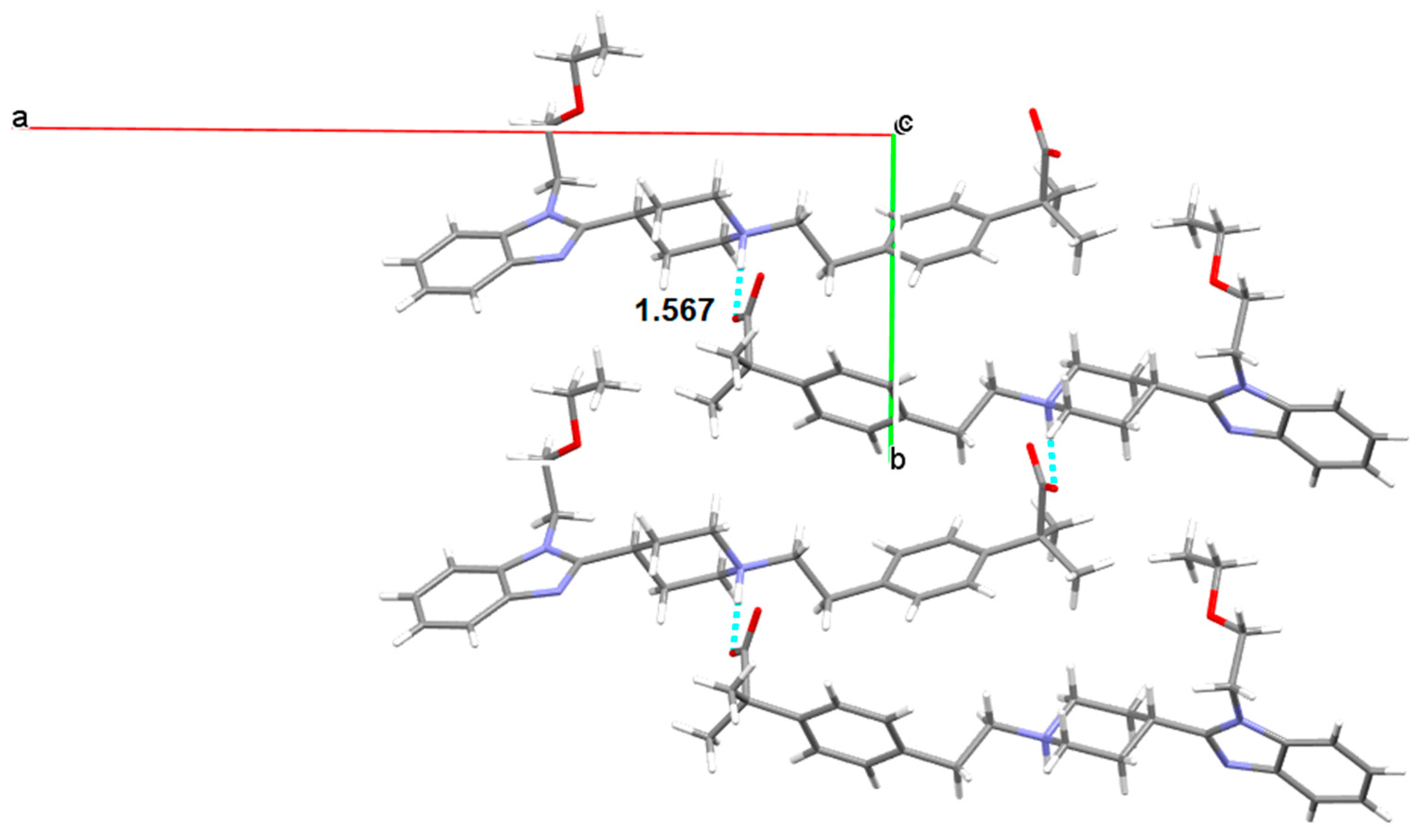
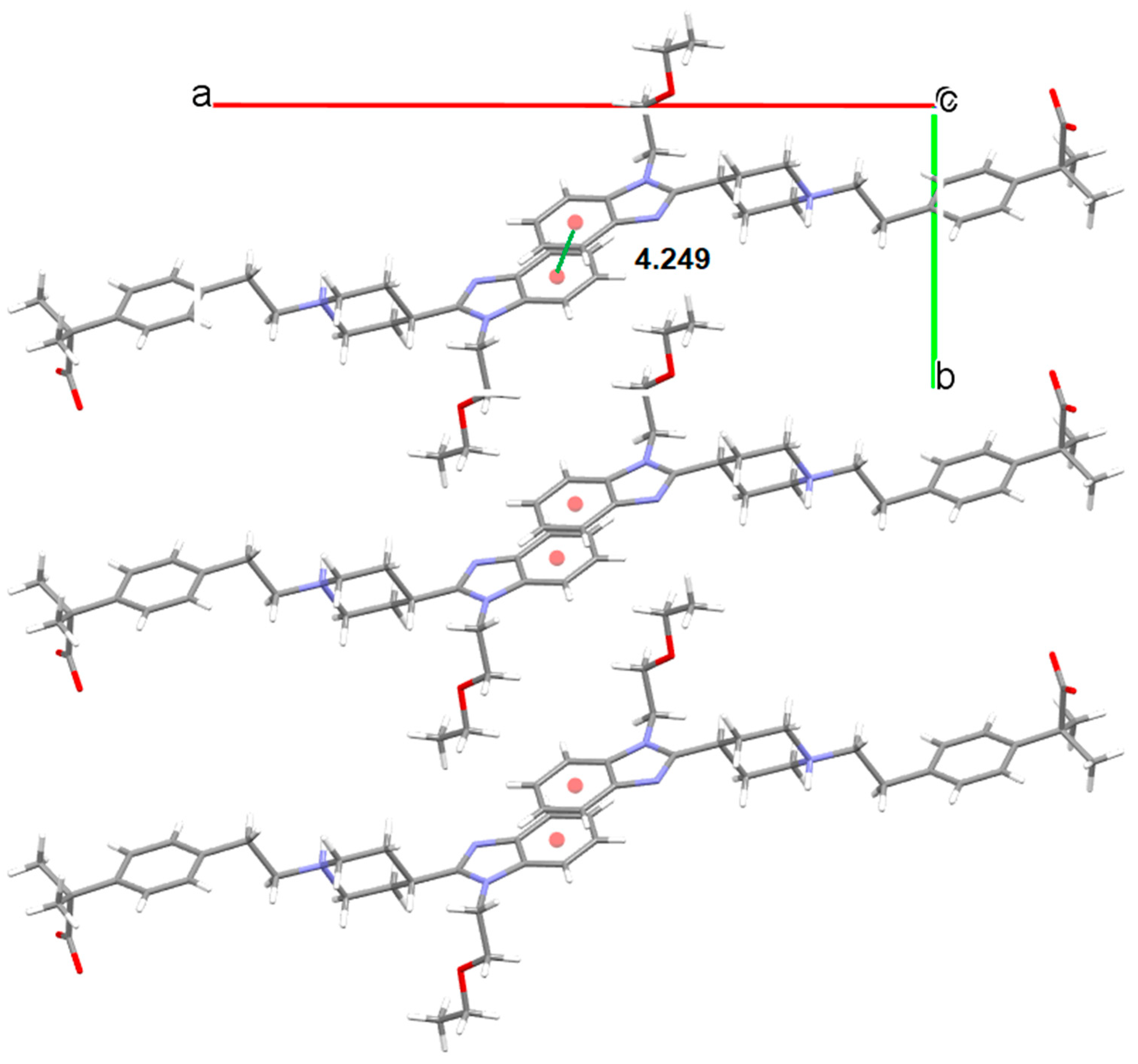
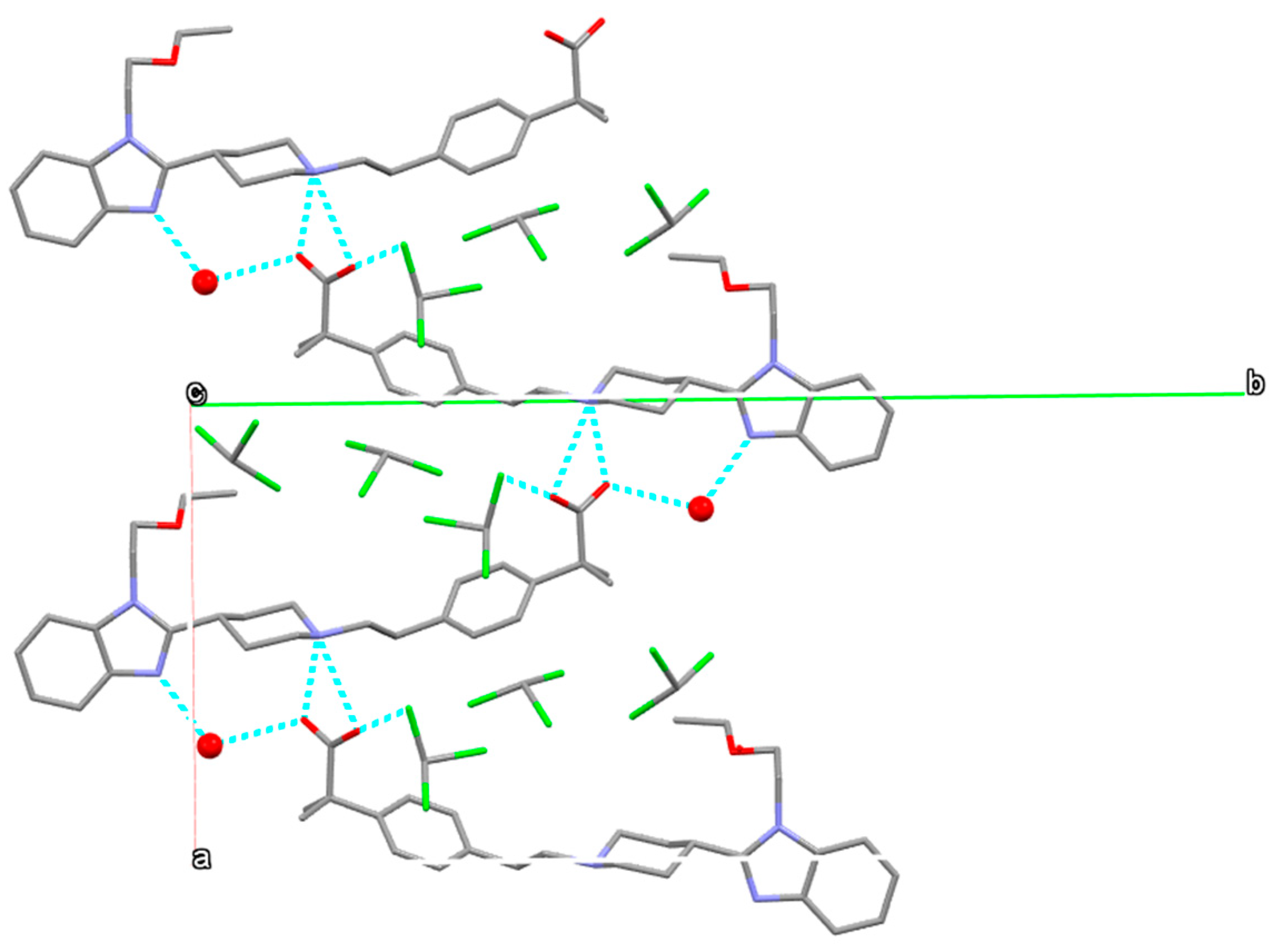
3.5.1. Bilastine Anhydrates
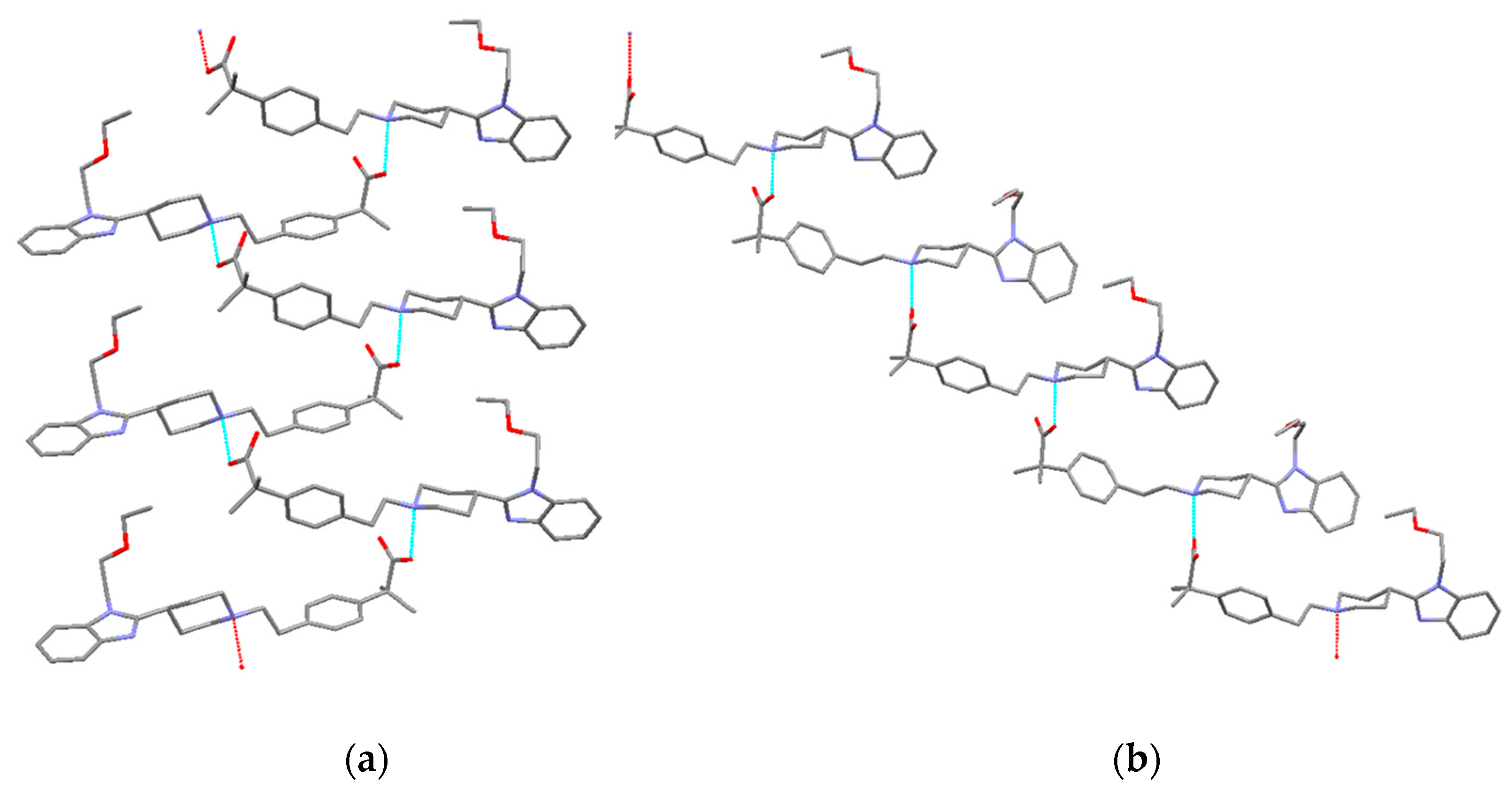
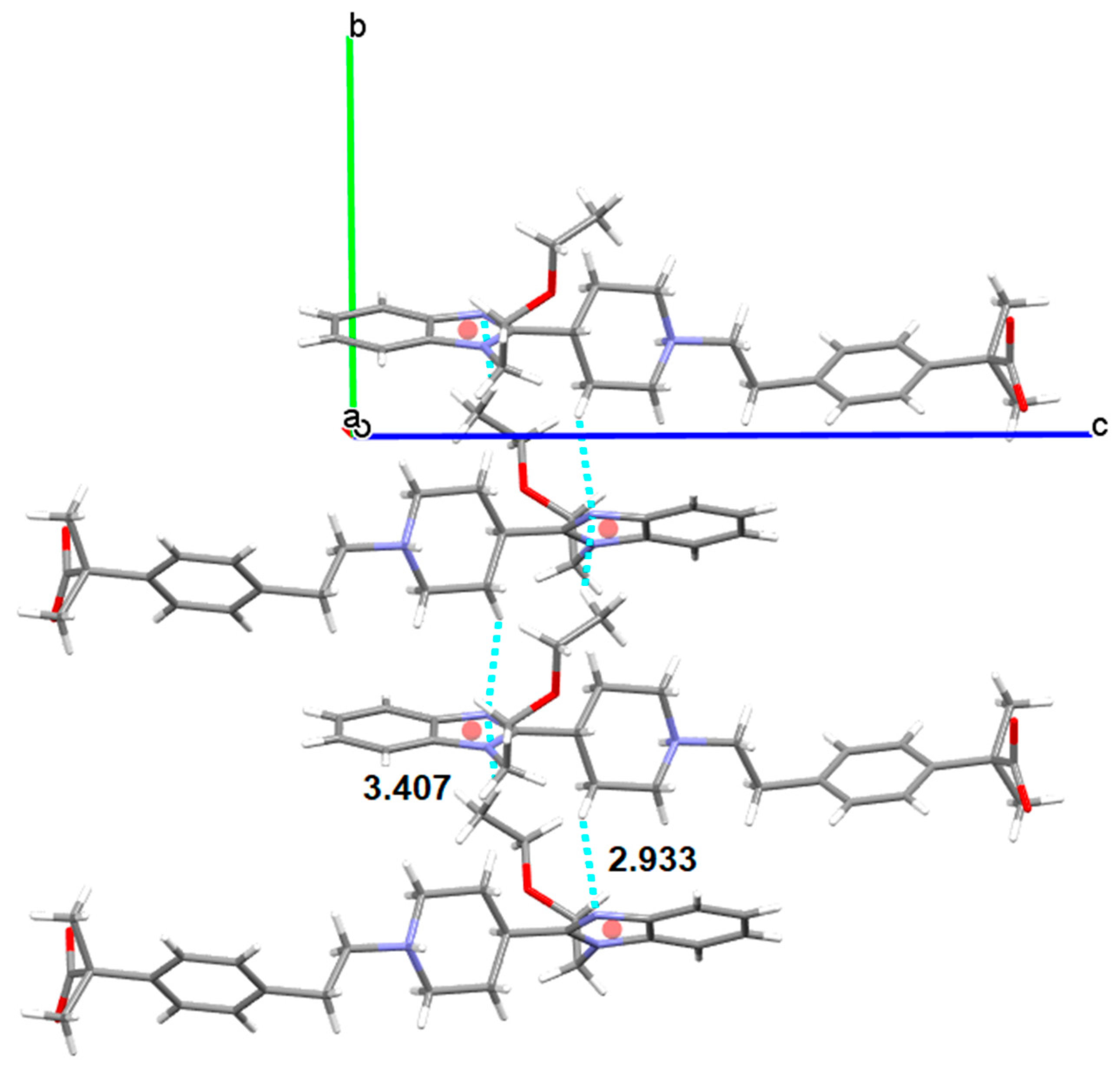
3.5.2. Bilastine Chloroform Solvates
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Singhal, D.; Curatolo, W. Drug polymorphism and dosage form design: A practical perspective. Adv. Drug Deliv. Rev. 2004, 56, 335–347. [Google Scholar] [CrossRef]
- Bladgen, N.; Davey, R.J. Polymorph Selection: Challenges for the Future? Cryst. Growth Des. 2003, 3, 873–885. [Google Scholar]
- Zalte, A.G.; Darekar, A.B.; Gondkar, S.B.; Saudagar, R.B. Cocrystals: An Emerging Approach to Modify Physicochemical Properties of Drugs. Am. J. PharmTech Res. 2014, 4, 2056–2072. [Google Scholar]
- Rodriguez-Spong, B.; Price, C.P.; Jayasankar, A.; Matzger, A.J.; Rodriguez-Hornedo, N. General principles of pharmaceutical solid polymorphism: A supramolecular perspective. Adv. Drug Delivery Rev. 2004, 56, 241–274. [Google Scholar] [CrossRef]
- Grodowska, K.; Parezewski, A. Organic solvents in the pharmaceutical industry. Acta Pol. Pharm. Drug Res. 2010, 67, 3–12. [Google Scholar]
- Zvoníček, V.; Skořepová, E.; Dušek, M.; Babor, M.; Žvátora, P.; Šoóš, M. First crystal structures of pharmaceutical ibrutinib: Systematic solvate screening and characterization. Cryst. Growth Des. 2017, 17, 3116–3127. [Google Scholar] [CrossRef]
- Wang, Y.; Chi, Y.; Zhang, W.; Yang, Q.; Yang, S.; Su, C.; Lin, Z.; Gu, J.; Hu, C. Structural diversity of diosgenin hydrates: Effect of initial concentration, water volume fraction, and solvent on crystallization. Cryst. Growth Des. 2016, 16, 1492–1501. [Google Scholar] [CrossRef]
- Berzins, A.; Trimdale, A.; Kons, A.; Zvaniņa, D. On the formation and desolvation mechanism of organic molecule solvates: A structural study of methyl cholate solvates. Cryst. Growth Des. 2017, 17, 5712–5724. [Google Scholar] [CrossRef]
- Tieger, E.; Kiss, V.; Pokol, G.; Finta, Z.; Rohlíček, J.; Skořepová, E.; Dušek, M. Rationalization of the formation and stability of bosutinib solvated forms. CrystEngComm 2016, 18, 9260–9274. [Google Scholar] [CrossRef]
- Council of Europe. Residual Solvents. In European Pharmacopoeia; Council of Europe: Strasbourg, France, 2013; p. 5967. [Google Scholar]
- Morissette, S.L.; Almarsson, O.; Peterson, M.L.; Remenar, J.F.; Read, M.J.; Lemmo, A.V.; Ellis, S.; Cima, M.J.; Gardner, C.R. High-throughput crystallization: Polymorphs, salts, co-crystals and solvates of pharmaceutical solids. Adv. Drug Deliv. Rev. 2004, 56, 275–300. [Google Scholar] [CrossRef] [PubMed]
- Wolthers, O.D. Bilastine: A new nonsedating oral H1 antihistamine for treatment of allergic rhinoconjunctivitis and urticaria. BioMed Res. Int. 2013, 2013, 1–6. [Google Scholar] [CrossRef]
- Corcóstegui, R.; Labeaga, L.; Innerárity, A.; Berisa, A.; Orjales, A. Preclinical pharmacology of bilastine, a new selective histamine H 1 receptor antagonist. Drugs R D 2005, 6, 371–384. [Google Scholar] [CrossRef] [PubMed]
- Orjales, A.; Rubio, V.; Bordell, M. Benzimidazole Derivatives with Antihistaminic Activity. EP 0818454B1, 3 June 1997. [Google Scholar]
- Orjales, A.; Bordell, M.; Canal, G.; Blanco, H. Polymorph of Acid 4-[2-[4-[1-(2-Ethoxyethyl)-1h-Benzimidazole-2-Il]-1-Piperidinyl]Ethyl]-$G(A), $G(A)-Dimethyl-Benzeneacetic. WO 2003/089425, 30 October 2003. [Google Scholar]
- Fauth, F.; Peral, I.; Popescu, C.; Knapp, M. The new Material Science Powder Diffraction beamline at ALBA Synchrotron. Powder Diffr. 2013, 28, S360–S370. [Google Scholar] [CrossRef]
- Boultif, A.; Louër, D.J. Powder pattern indexing with the dichotomy method. J. Appl. Crystallogr. 2004, 37, 724–731. [Google Scholar] [CrossRef]
- Vallcorba, O.; Rius, J.; Frontera, C.; Peral, I.; Miravitlles, C. DAJUST: A suite of computer programs for pattern matching, space-group determination and intensity extraction from powder diffraction data. J. Appl. Crystallogr. 2012, 45, 844–848. [Google Scholar] [CrossRef]
- Vallcorba, O.; Rius, J.; Frontera, C.; Miravitlles, C. TALP: A multisolution direct-space strategy for solving molecular crystals from powder diffraction data based on restrained least squares. J. Appl. Crystallogr. 2012, 45, 1270–1277. [Google Scholar] [CrossRef]
- Sheldrick, G.M. A Short History of SHELX. Acta Crystallogr. 2008, A64, 112–122. [Google Scholar] [CrossRef] [PubMed]
- Hübschle, C.B.; Sheldrick, G.M.; Dittrich, B.J. ShelXle: A Qt graphical user interface for SHELXL. J. Appl. Crystallogr. 2011, 44, 1281–1284. [Google Scholar] [CrossRef]
- Burger, A.; Ramberger, R. On the polymorphism of pharmaceuticals and other molecular crystals. I. Mikrochim. Acta 1979, 259–271. [Google Scholar] [CrossRef]
- Yu, L.J. Inferring thermodynamic stability relationship of polymorphs from melting data. J. Pharm. Sci. 1995, 84, 966–974. [Google Scholar] [CrossRef]
- Haleblian, J.; Crone, W.M. Pharmaceutical applications of polymorphism. J. Pharm. Sci. 1969, 58, 911–929. [Google Scholar] [CrossRef]
- Zencirci, N.; Griesser, U.J.; Gelbrich, T.; Kahlenberg, V.; Jetti, R.K.R.; Apperley, D.C.; Harris, R.K. New solvates of an old drug compound (phenobarbital): Structure and stability. J. Phys. Chem. B 2014, 118, 3267–3280. [Google Scholar] [CrossRef] [PubMed]
- Ward, M.R.; Oswald, D.H. Hidden solvates and transient forms of trimesic acid. Crystals 2020, 10, 1098. [Google Scholar] [CrossRef]
- Braun, D.E.; Schneeberger, A.; Griesser, U.J. Understanding the role of water in 1, 10-phenanthroline monohydrate. CrystEngComm 2017, 19, 6133–6145. [Google Scholar] [CrossRef] [PubMed]
- Petkune, S.; Bobrovs, R.; Actins, A. Organic solvents vapor pressure and relative humidity effects on the phase transition rate of α and β forms of tegafur. Pharm. Dev. Technol. 2012, 17, 625–631. [Google Scholar] [CrossRef] [PubMed]
- Hunter, C.A.; Sanders, J.K.M. The nature of. pi.-. pi. interactions. J. Am. Chem. Soc. 1990, 112, 5525–5534. [Google Scholar] [CrossRef]
- Takieddin, K.; Zhimyak, Y.Z.; Fábián, L. Prediction of hydrate and solvate formation using statistical models. Cryst. Growth Des. 2016, 16, 70–81. [Google Scholar] [CrossRef]

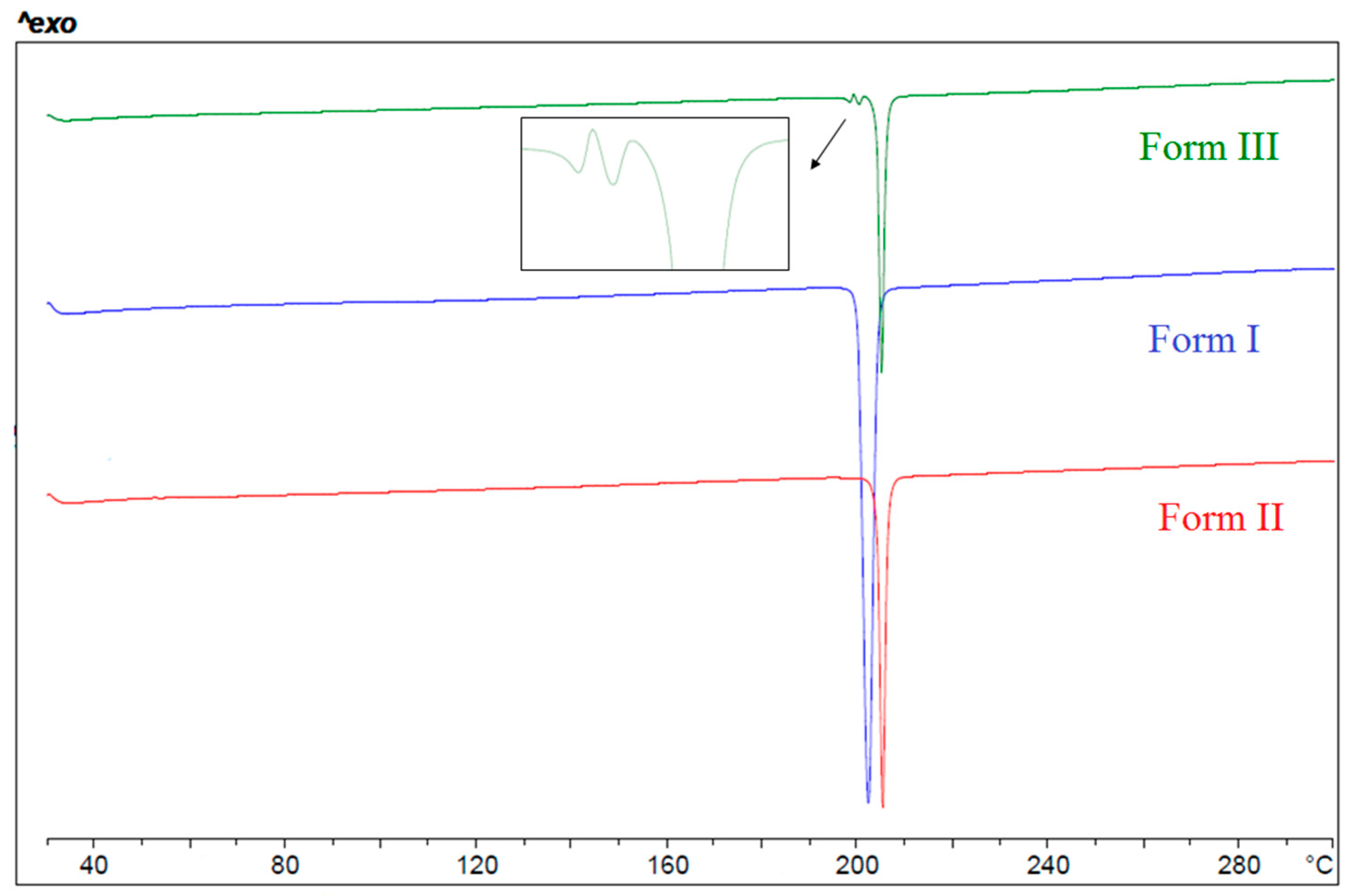









| Tfus (°C) | ΔHfus (J/g) | |
|---|---|---|
| Form I | 200 | 119.9 |
| Form II | 202 | 118.2 |
| Form III | 197 | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Puigjaner, C.; Portell, A.; Blasco, A.; Font-Bardia, M.; Vallcorba, O. Entrapped Transient Chloroform Solvates of Bilastine. Crystals 2021, 11, 342. https://doi.org/10.3390/cryst11040342
Puigjaner C, Portell A, Blasco A, Font-Bardia M, Vallcorba O. Entrapped Transient Chloroform Solvates of Bilastine. Crystals. 2021; 11(4):342. https://doi.org/10.3390/cryst11040342
Chicago/Turabian StylePuigjaner, Cristina, Anna Portell, Arturo Blasco, Mercè Font-Bardia, and Oriol Vallcorba. 2021. "Entrapped Transient Chloroform Solvates of Bilastine" Crystals 11, no. 4: 342. https://doi.org/10.3390/cryst11040342
APA StylePuigjaner, C., Portell, A., Blasco, A., Font-Bardia, M., & Vallcorba, O. (2021). Entrapped Transient Chloroform Solvates of Bilastine. Crystals, 11(4), 342. https://doi.org/10.3390/cryst11040342








