Processability and Optimization of Laser Parameters for Densification of Hypereutectic Al–Fe Binary Alloy Manufactured by Laser Powder Bed Fusion
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
3. Results
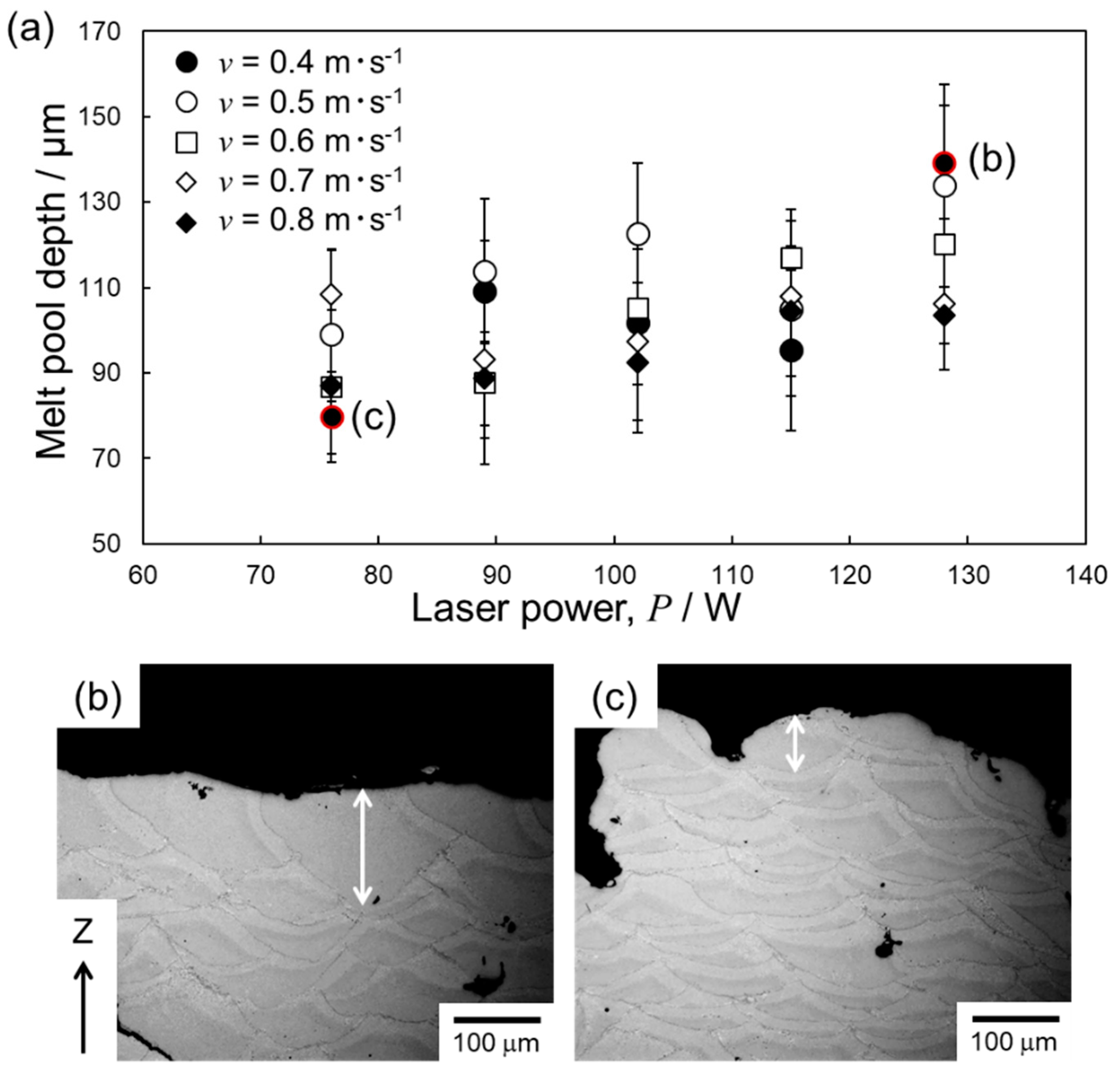
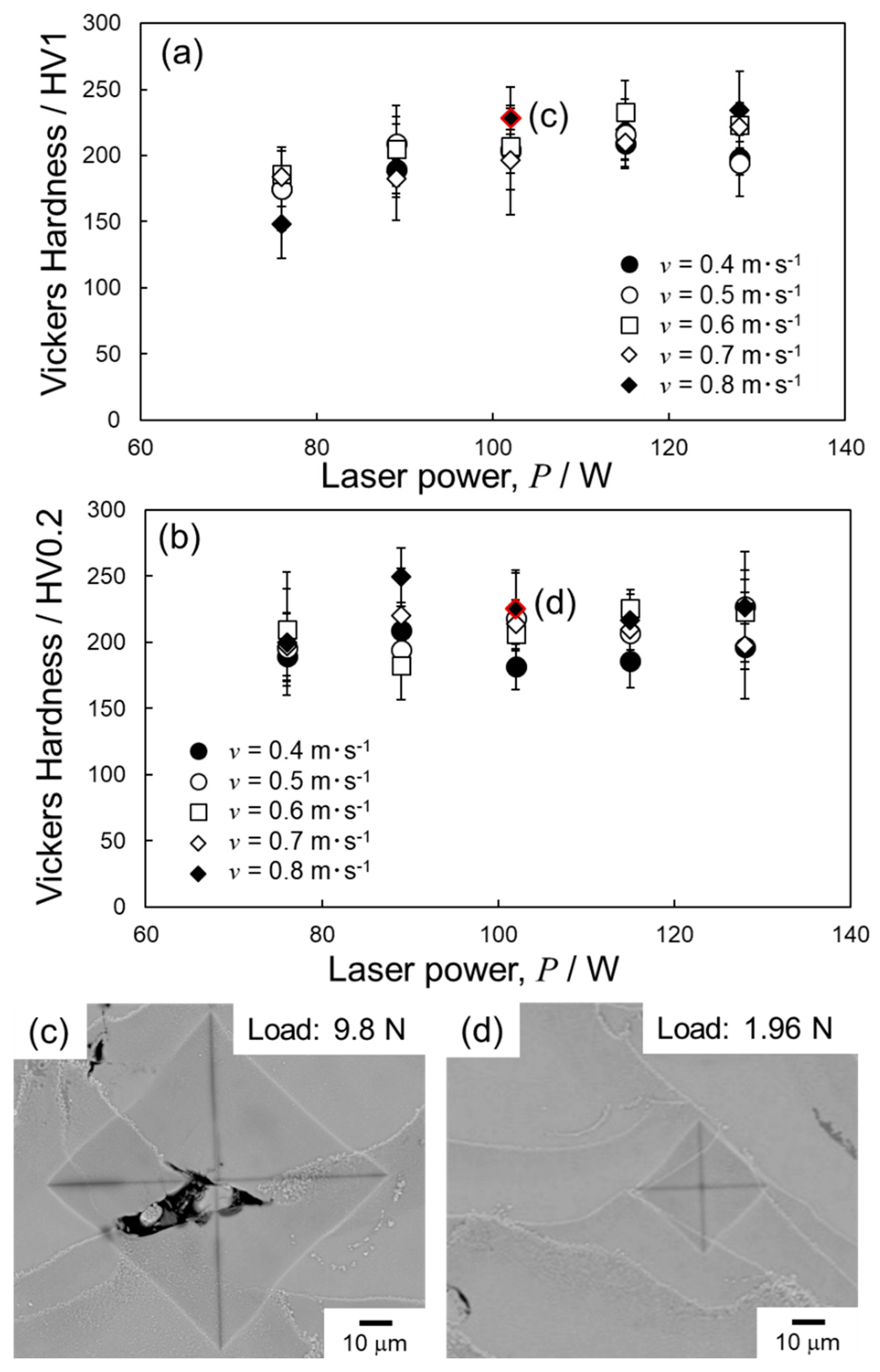
4. Discussion
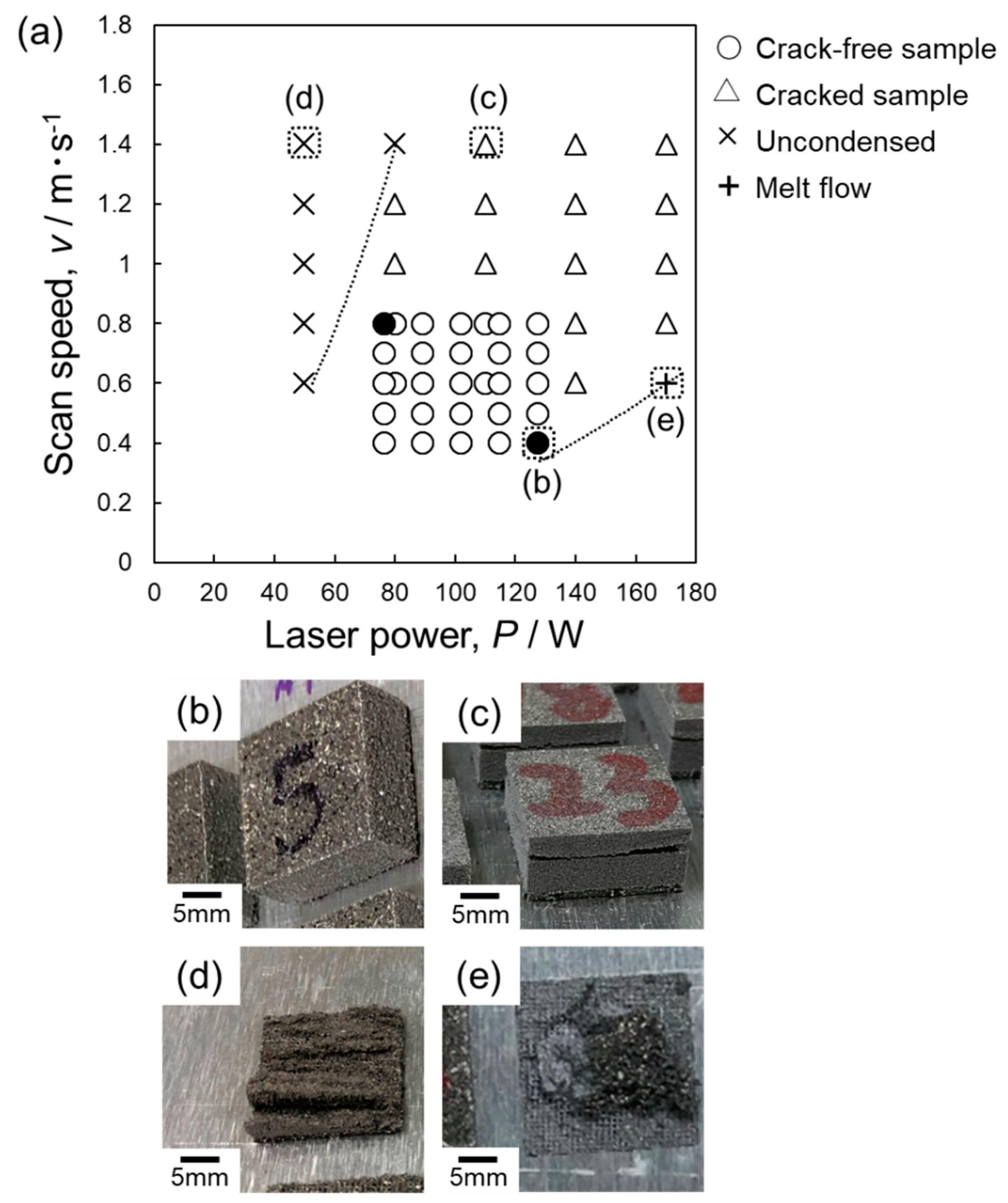
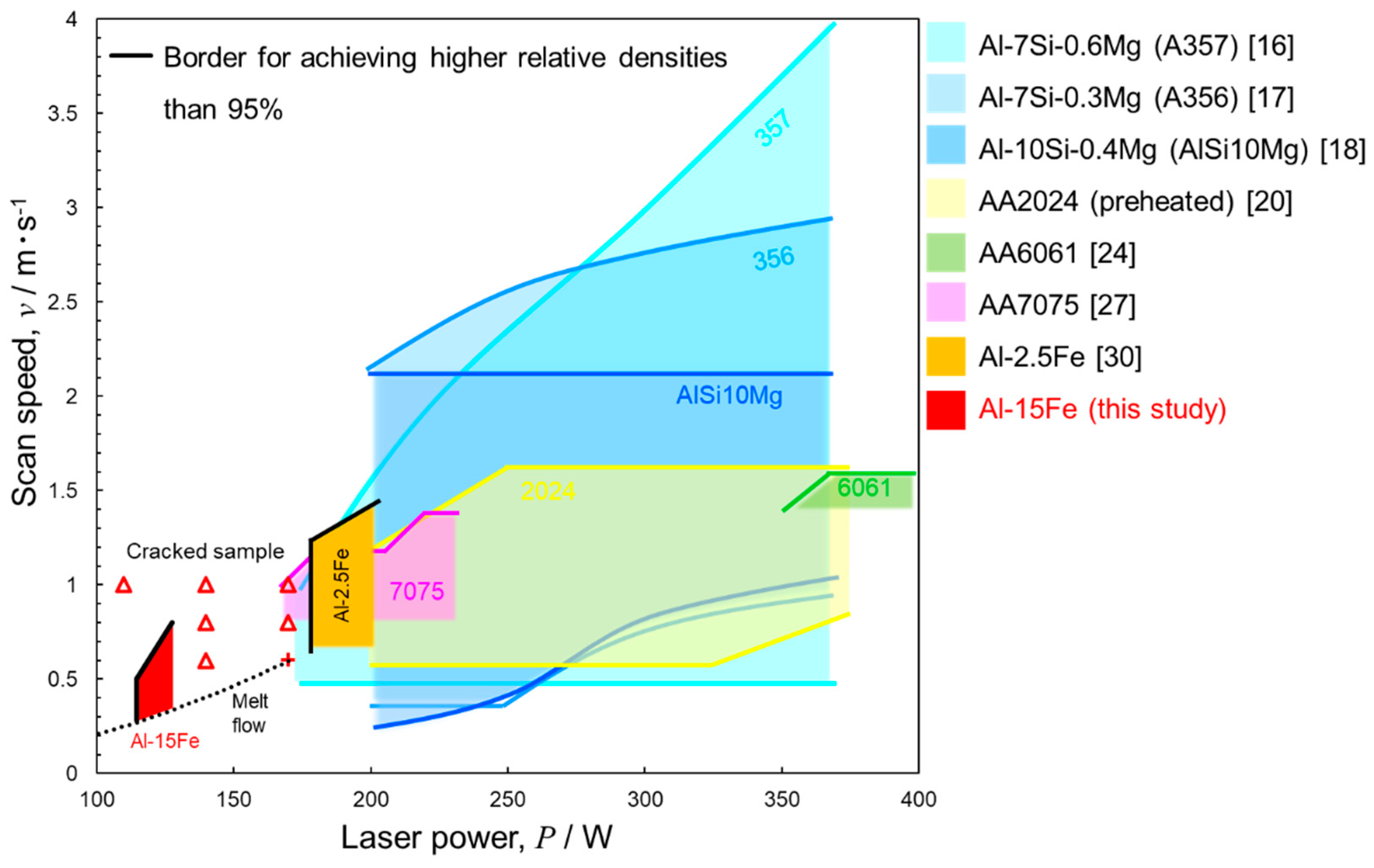
4.1. Process Window of Laser Parameters for Manufacturing Samples
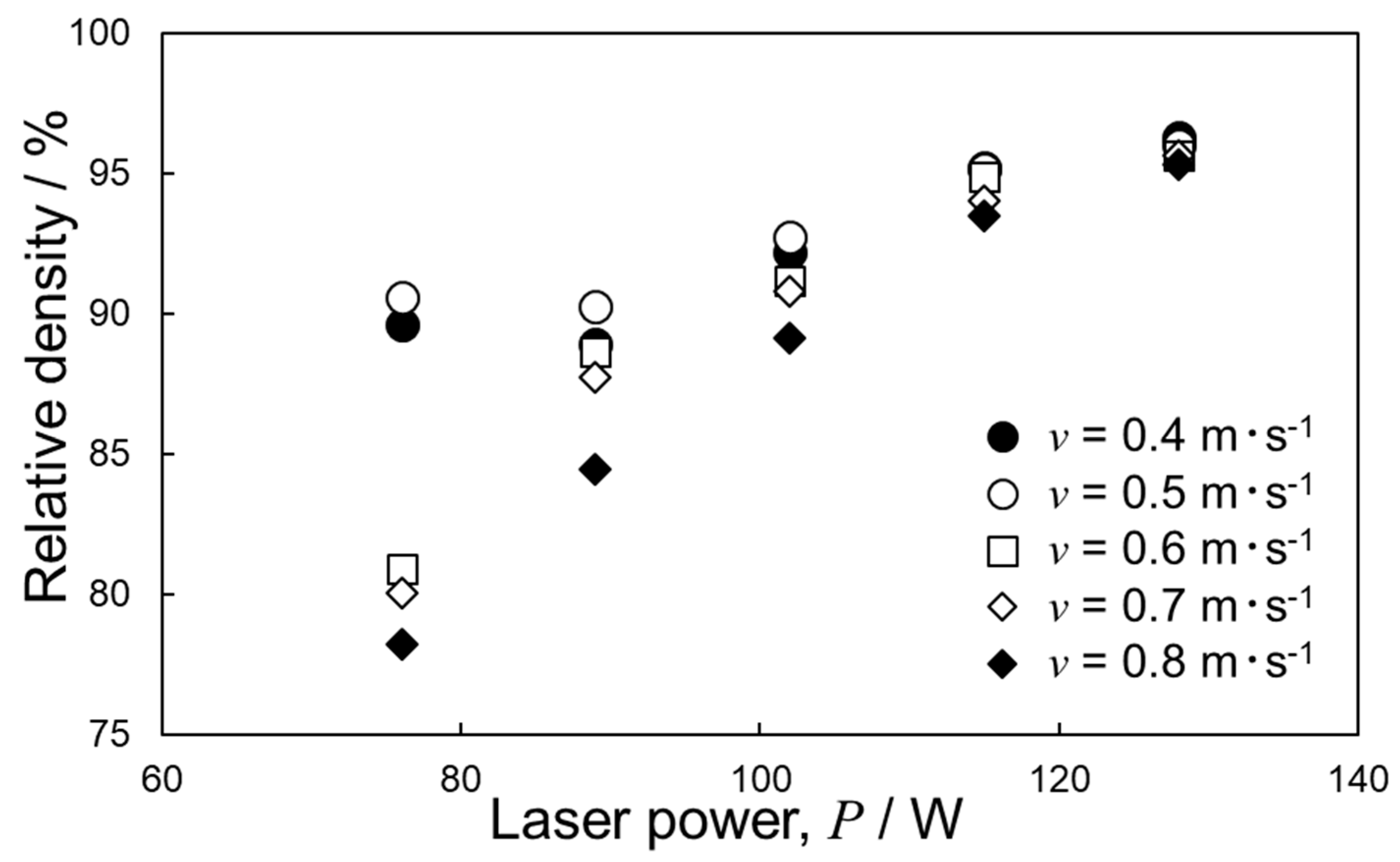
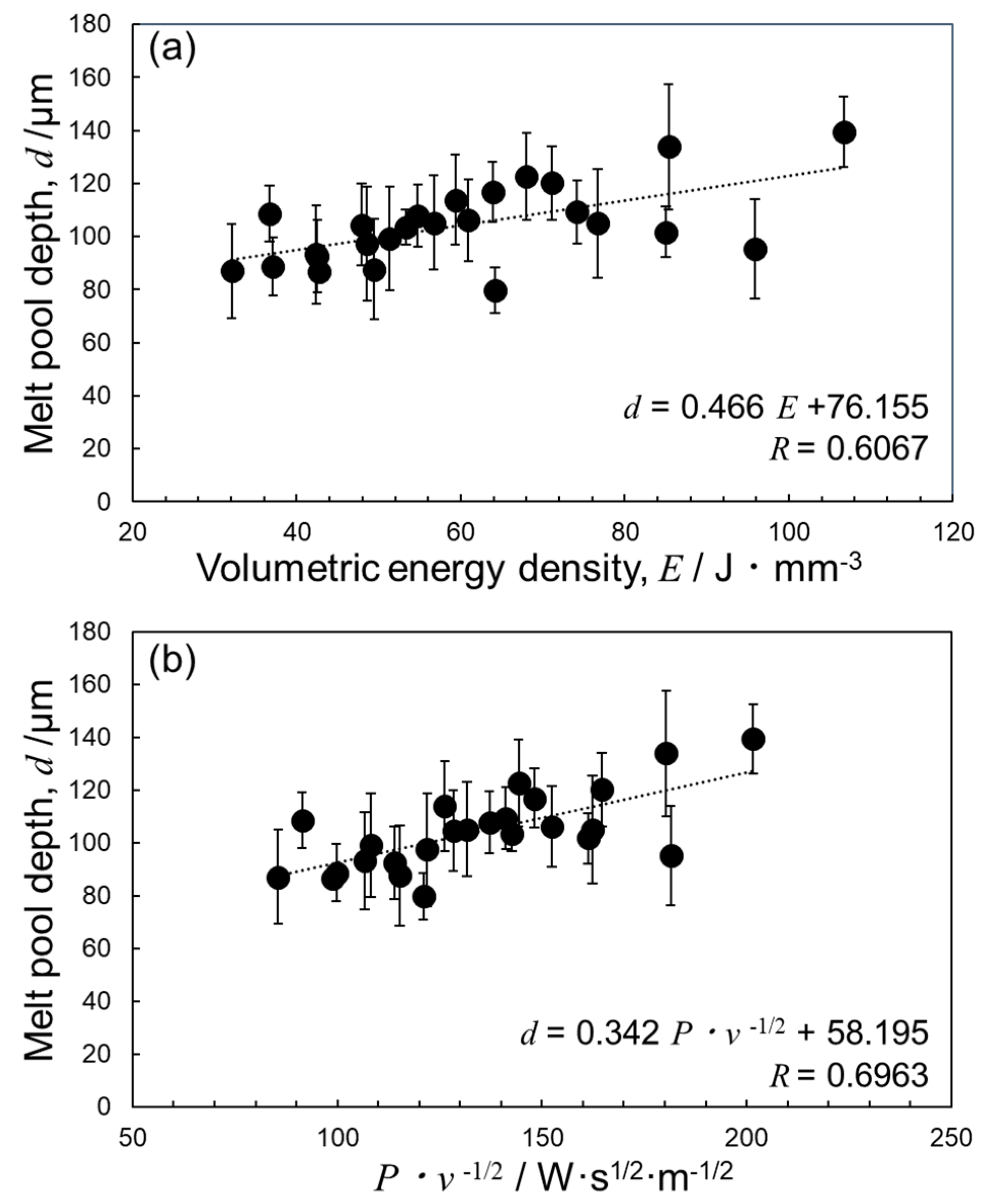
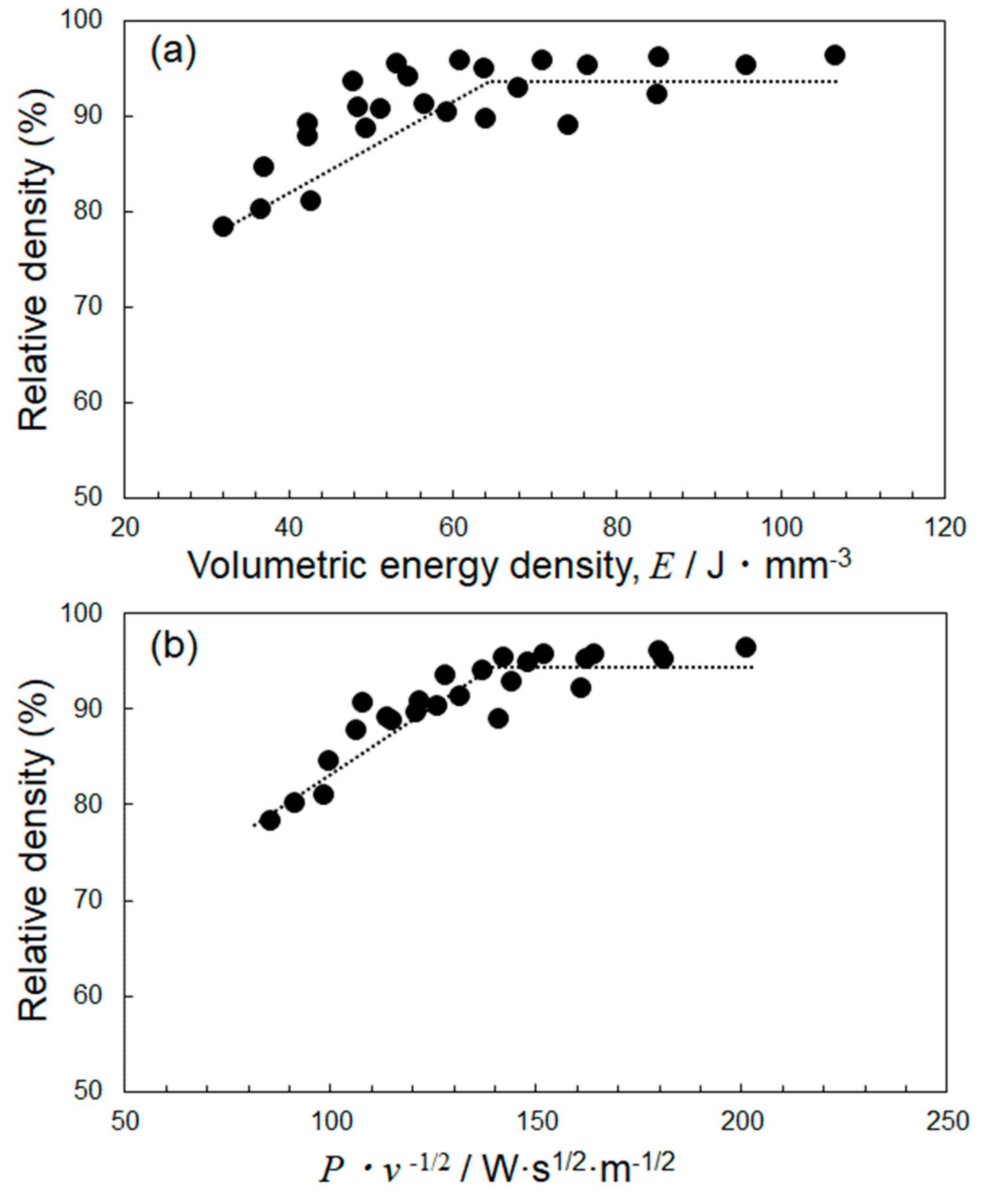
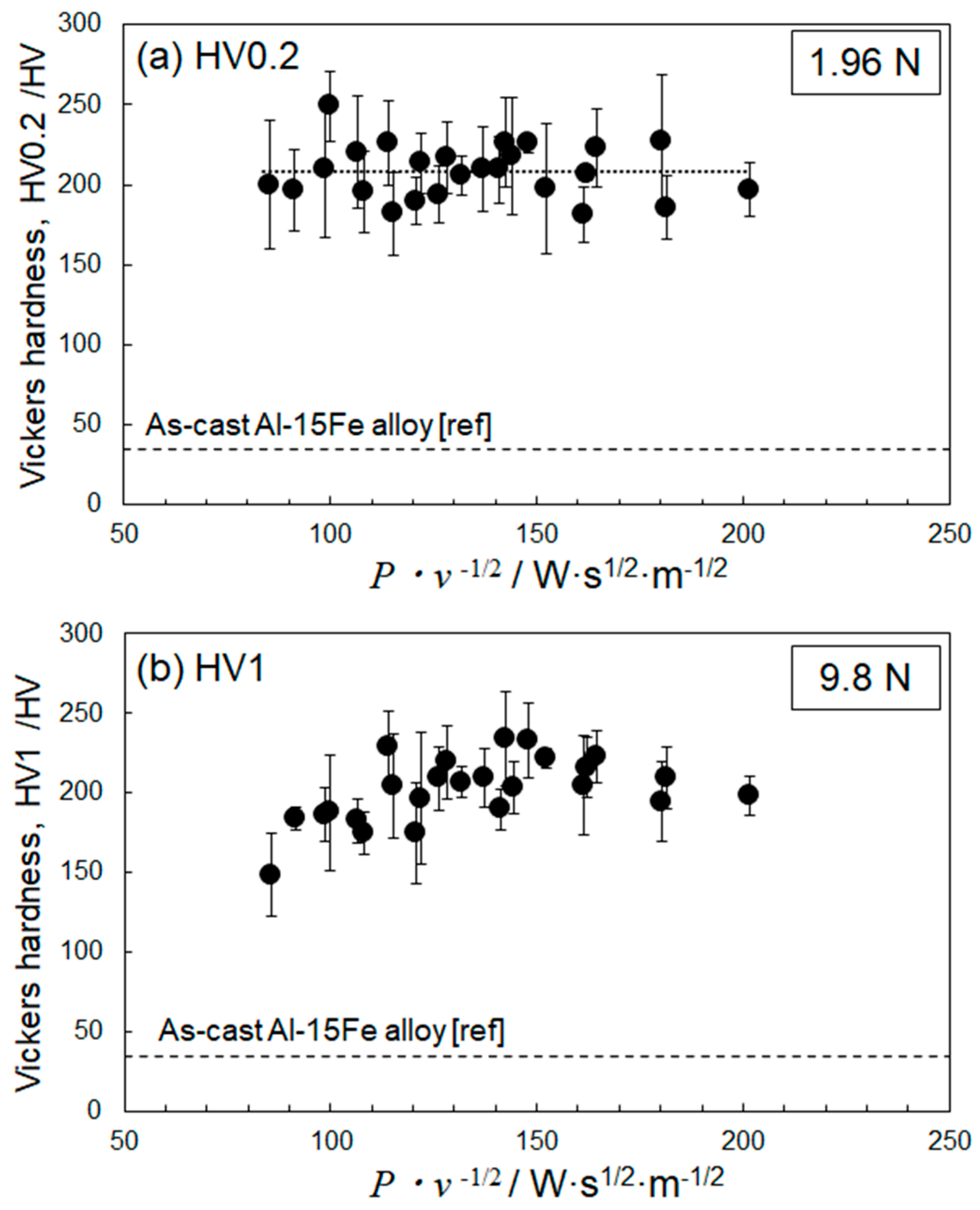
4.2. Optimization of Laser Parameters for Densification
5. Conclusions
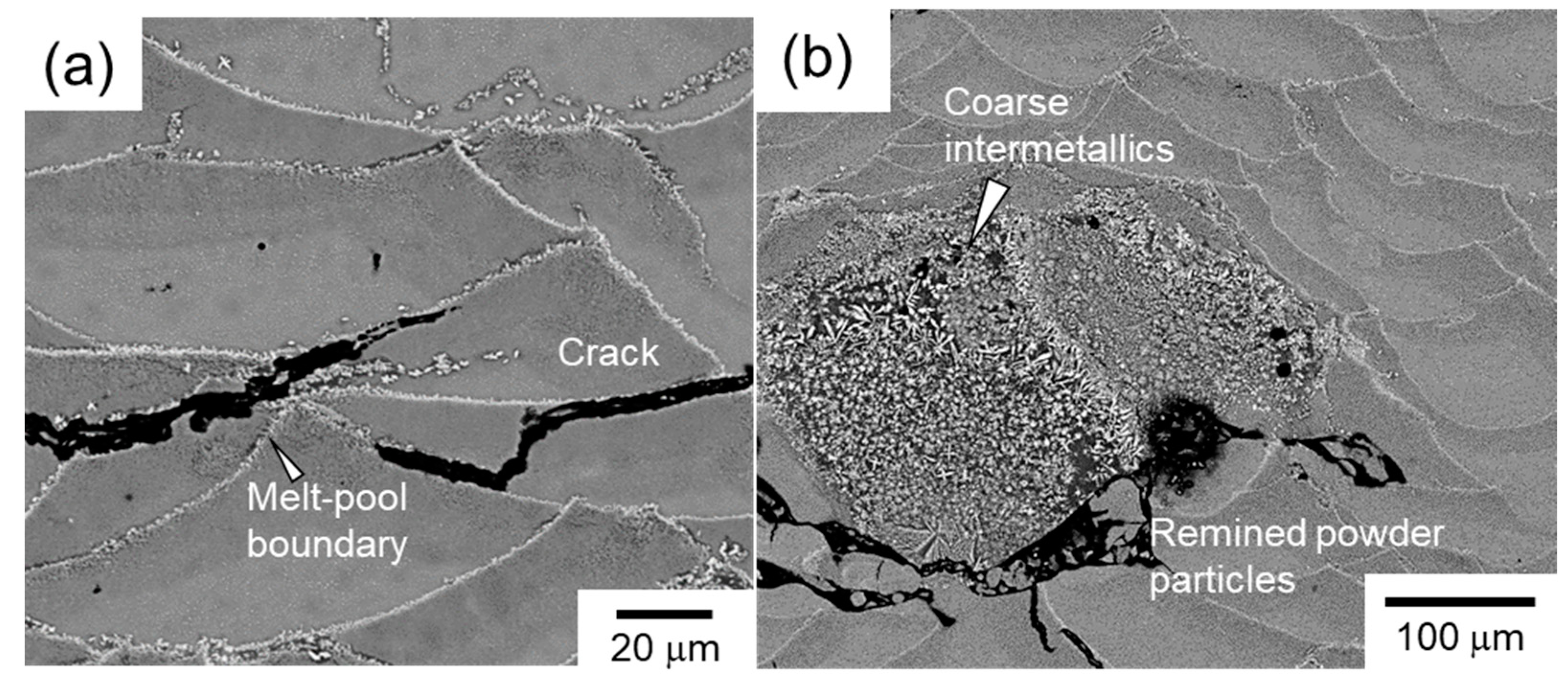
- A laser process window of P = 77–128 W and v = 0.4–0.8 m·s−1 was identified that can ensure the fabrication of macroscopically crack-free Al–15%Fe alloy samples. This process window is much smaller than those of other Al alloys. When a higher scan speed (v ≥ 1.0 m·s−1) or a higher laser power (P ≥ 140 W) is used, the samples tend to include a number of macroscopic cracks propagating parallel to the powder bed plane. These cracks appear to preferentially propagate along the melt pool boundaries decorated with the brittle θ-Al13Fe4 phase (primary solidified phase), resulting in a low L-PBF processability for manufacturing Al–15%Fe alloy samples.
- The deposited energy density model (parametrized by P·v−1/2) appeared useful for identifying the optimum L-PBF process conditions towards densification of Al–15%Fe alloy samples, in comparison with the volumetric energy density (parametrized by P·v−1). However, the correlation with P·v−1/2 for the Al–15%Fe alloy was modest compared to the cases of Al–2.5%Fe alloy and maraging steel with high thermal conductivities. The possible reason was that the L-PBF-built Al–15%Fe alloy samples contained inhomogeneous microstructures having a number of coarsened θ-Al13Fe4 phases with low thermal conductivity.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wang, S.; Cheng, S.; Yu, H.; Rao, Z.; Liu, Z. Experimental investigation of Al–Cu composed tube–fin heat exchangers for air conditioner. Exp. Therm. Fluid Sci. 2013, 51, 264–270. [Google Scholar] [CrossRef]
- Choi, S.H.; Sung, S.Y.; Choi, H.J.; Sohn, Y.H.; Han, B.S.; Lee, K.A. High temperature tensile deformation behavior of new heat resistant aluminum alloy. Procedia Eng. 2011, 10, 159–164. [Google Scholar] [CrossRef]
- Tiwary, C.S.; Kashyap, S.; Chattopadhyay, K. Development of alloys with high strength at elevated temperatures by tuning the bimodal microstructure in the Al–Cu–Ni eutectic system. Scr. Mater. 2014, 93, 20–23. [Google Scholar] [CrossRef]
- Tiwary, C.S.; Kashyap, S.; Kim, D.H.; Chattopadhyay, K. Al based ultra-fine eutectic with high room temperature plasticity and elevated temperature strength. Mater. Sci. Eng. A 2015, 639, 359–369. [Google Scholar] [CrossRef]
- Erdeniz, D.; De Luca, A.; Seidman, D.N.; Dunand, D.C. Effects of Nb and Ta additions on the strength and coarsening resistance of precipitation-strengthened Al-Zr-Sc-Er-Si alloys. Mater. Charact. 2018, 141, 260–266. [Google Scholar] [CrossRef]
- Michi, R.A.; Toinin, J.; Farkoosh, A.R.; Seidman, D.N.; Dunand, D.C. Effects of Zn and Cr additions on precipitation and creep behavior of a dilute Al–Zr–Er–Si alloy. Acta Mater. 2019, 181, 249–261. [Google Scholar] [CrossRef]
- Black, P.J. The structure of FeAl3. I. Acta Cryst. 1955, 8, 43–48. [Google Scholar] [CrossRef]
- Tsukahara, T.; Takata, N.; Kobayashi, S.; Takeyama, M. Mechanical properties of Fe2Al5 and FeAl3 intermetallic phases at ambient temperature. Tetsu-To-Hagané 2016, 102, 29–35. [Google Scholar] [CrossRef]
- Khorasani, A.; Gibson, I.; Veetil, J.K.; Ghasemi, A.H. A review of technological improvements in laser-based powder bed fusion of metal printers. Int. J. Adv. Manuf. Technol. 2020, 108, 191–209. [Google Scholar] [CrossRef]
- Thijs, L.; Kempen, K.; Kruth, J.P.; Van Humbeeck, J. Fine-structured aluminium products with controllable texture by selective laser melting of pre-alloyed AlSi10Mg powder. Acta Mater. 2013, 61, 1809–1819. [Google Scholar] [CrossRef]
- Takata, N.; Kodaira, H.; Sekizawa, K.; Suzuki, A.; Kobashi, M. Change in microstructure of selectively laser melted AlSi10Mg alloy with heat treatments. Mater. Sci. Eng. A 2017, 704, 218–228. [Google Scholar] [CrossRef]
- Takata, N.; Liu, M.; Kodaira, H.; Suzuki, A.; Kobashi, M. Anomalous strengthening by supersaturated solid solutions of selectively laser melted Al–Si-based alloys. Addit. Manuf. 2020, 33, 101152. [Google Scholar] [CrossRef]
- Qi, X.; Takata, N.; Suzuki, A.; Kobashi, M.; Kato, M. Managing both high strength and thermal conductivity of a laser powder bed fused Al–2.5 Fe binary alloy: Effect of annealing on microstructure. Mater. Sci. Eng. A 2020, 805, 140591. [Google Scholar] [CrossRef]
- Wang, W.; Takata, N.; Suzuki, A.; Kobashi, M.; Kato, M. Formation of multiple intermetallic phases in a hypereutectic Al-Fe binary alloy additively manufactured by laser powder bed fusion. Intermetallics 2020, 125, 106892. [Google Scholar] [CrossRef]
- Wallace, G.; Jackson, A.P.; Midson, S.P. Novel method for casting high quality aluminum turbocharger impellers. Int. J. Mater. Manuf. 2010, 3, 405–412. [Google Scholar] [CrossRef]
- Rao, H.; Giet, S.; Yang, K.; Wu, X.; Davies, C.H. The influence of processing parameters on aluminium alloy A357 manufactured by Selective Laser Melting. Mater. Des. 2016, 109, 334–346. [Google Scholar] [CrossRef]
- Kimura, T.; Nakamoto, T. Microstructures and mechanical properties of A356 (AlSi7Mg0.3) aluminum alloy fabricated by selective laser melting. Mater. Des. 2016, 89, 1294–1301. [Google Scholar] [CrossRef]
- Kimura, T.; Nakamoto, T. Microstructures and mechanical properties of Al-10%Si-0.4%Mg fabricated by selective laser melting. J. Jpn. Soc. Powder Metall. 2014, 61, 531–537. [Google Scholar] [CrossRef][Green Version]
- Koutny, D.; Palousek, D.; Pantelejev, L.; Hoeller, C.; Pichler, R.; Tesicky, L.; Kaiser, J. Influence of scanning strategies on processing of aluminum alloy EN AW 2618 using selective laser melting. Materials 2018, 11, 298. [Google Scholar] [CrossRef]
- Tan, Q.; Liu, Y.; Fan, Z.; Zhang, J.; Yin, Y.; Zhang, M.X. Effect of processing parameters on the densification of an additively manufactured 2024 Al alloy. J. Mater. Sci. Technol. 2020, 58, 34–45. [Google Scholar] [CrossRef]
- Ahuja, B.; Karg, M.; Nagulin, K.Y.; Schmidt, M. Fabrication and characterization of high strength Al-Cu alloys processed using laser beam melting in metal powder bed. Phys. Procedia 2014, 56, 135–146. [Google Scholar] [CrossRef]
- Xu, R.; Li, R.; Yuan, T.; Niu, P.; Wang, M.; Lin, Z. Microstructure, metallurgical defects and hardness of Al–Cu–Mg–Li–Zr alloy additively manufactured by selective laser melting. J. Alloys Compd. 2020, 835, 155372. [Google Scholar] [CrossRef]
- Knoop, D.; Lutz, A.; Mais, B.; von Hehl, A. A tailored AlSiMg alloy for laser powder bed fusion. Metals 2020, 10, 514. [Google Scholar] [CrossRef]
- Uddin, S.Z.; Murr, L.E.; Terrazas, C.A.; Morton, P.; Roberson, D.A.; Wicker, R.B. Processing and characterization of crack-free aluminum 6061 using high-temperature heating in laser powder bed fusion additive manufacturing. Addit. Manuf. 2018, 22, 405–415. [Google Scholar] [CrossRef]
- Rutherford, B.A.; Avery, D.Z.; Phillips, B.J.; Rao, H.M.; Doherty, K.J.; Allison, P.G.; Brewer, L.N.; Jordon, J.B. Effect of Thermomechanical Processing on Fatigue Behavior in Solid-State Additive Manufacturing of Al-Mg-Si Alloy. Metals 2020, 10, 947. [Google Scholar] [CrossRef]
- Stopyra, W.; Gruber, K.; Smolina, I.; Kurzynowski, T.; Kuźnicka, B. Laser powder bed fusion of AA7075 alloy: Influence of process parameters on porosity and hot cracking. Addit. Manuf. 2020, 35, 101270. [Google Scholar]
- Otani, Y.; Kusaki, Y.; Itagaki, K.; Sasaki, S. Microstructure and mechanical properties of A7075 alloy with additional Si objects fabricated by selective laser melting. Mater. Trans. 2019, 60, 2143–2150. [Google Scholar] [CrossRef]
- Zhou, S.Y.; Su, Y.; Wang, H.; Enz, J.; Ebel, T.; Yan, M. Selective laser melting additive manufacturing of 7xxx series Al-Zn-Mg-Cu alloy: Cracking elimination by co-incorporation of Si and TiB2. Addit. Manuf. 2020, 36, 101458. [Google Scholar] [CrossRef]
- Qi, T.; Zhu, H.; Zhang, H.; Yin, J.; Ke, L.; Zeng, X. Selective laser melting of Al7050 powder: Melting mode transition and comparison of the characteristics between the keyhole and conduction mode. Mater. Des. 2017, 135, 257–266. [Google Scholar] [CrossRef]
- Qi, X.; Takata, N.; Suzuki, A.; Kobashi, M.; Kato, M. Laser powder bed fusion of a near-eutectic Al–Fe binary alloy: Processing and microstructure. Addit. Manuf. 2020, 35, 101308. [Google Scholar]
- Sun, S.B.; Zheng, L.J.; Liu, J.H.; Zhang, H. Selective laser melting of an Al–Fe–V–Si alloy: Microstructural evolution and thermal stability. J. Mater. Sci. Technol. 2017, 33, 389–396. [Google Scholar] [CrossRef]
- Yamasaki, S.; Okuhira, T.; Mitsuhara, M.; Nakashima, H.; Kusui, J.; Adachi, M. Effect of Fe addition on heat-resistant aluminum alloys produced by selective laser melting. Metals 2019, 9, 468. [Google Scholar] [CrossRef]
- Takata, N.; Nishida, R.; Suzuki, A.; Kobashi, M.; Kato, M. Crystallographic features of microstructure in maraging steel fabricated by selective laser melting. Metals 2018, 8, 440. [Google Scholar] [CrossRef]
- Suzuki, A.; Nishida, R.; Takata, N.; Kobashi, M.; Kato, M. Design of laser parameters for selectively laser melted maraging steel based on deposited energy density. Addit. Manuf. 2019, 28, 160–168. [Google Scholar] [CrossRef]
- Takata, N.; Okano, T.; Suzuki, A.; Kobashi, M. Microstructure of intermetallic-reinforced Al-based alloy composites fabricated using eutectic reactions in Al–Mg–Zn ternary system. Intermetallics 2018, 95, 48–58. [Google Scholar] [CrossRef]
- Takata, N.; Kodaira, H.; Suzuki, A.; Kobashi, M. Size dependence of microstructure of AlSi10Mg alloy fabricated by selective laser melting. Mater. Charact. 2018, 143, 18–26. [Google Scholar] [CrossRef]
- Sames, W.J.; List, F.A.; Pannala, S.; Dehoff, R.R.; Babu, S.S. The metallurgy and processing science of metal additive manufacturing. Int. Mater. Rev. 2016, 61, 315–360. [Google Scholar] [CrossRef]
- Liu, M.; Takata, N.; Suzuki, A.; Kobashi, M. Development of gradient microstructure in the lattice structure of AlSi10Mg alloy fabricated by selective laser melting. J. Mater. Sci. Technol. 2020, 36, 106–117. [Google Scholar] [CrossRef]
- Walford, L.K. The structure of the intermetallic phase FeAl6. Acta Cryst. 1965, 18, 287–291. [Google Scholar] [CrossRef]
- Ding, X.; Koizumi, Y.; Wei, D.; Chiba, A. Effect of process parameters on melt pool geometry and microstructure development for electron beam melting of IN718: A systematic single bead analysis study. Addit. Manuf. 2019, 26, 215–226. [Google Scholar] [CrossRef]
- Zhao, Y.; Koizumi, Y.; Aoyagi, K.; Wei, D.; Yamanaka, K.; Chiba, A. Molten pool behavior and effect of fluid flow on solidification conditions in selective electron beam melting (SEBM) of a biomedical Co-Cr-Mo alloy. Addit. Manuf. 2019, 26, 202–214. [Google Scholar] [CrossRef]
- King, W.E.; Barth, H.D.; Castillo, V.M.; Gallegos, G.F.; Gibbs, J.W.; Hahn, D.E.; Kamath, C.; Rubenchik, A.M. Observation of keyhole-mode laser melting in laser powder-bed fusion additive manufacturing. J. Mater. Process. Technol. 2014, 214, 2915–2925. [Google Scholar] [CrossRef]
- Aboulkhair, N.T.; Simonelli, M.; Parry, L.; Ashcroft, I.; Tuck, C.; Hague, R. 3D printing of aluminium alloys: Additive manufacturing of aluminium alloys using selective laser melting. Prog. Mater. Sci. 2019, 106, 100578. [Google Scholar] [CrossRef]
- Wang, P.; Gammer, C.; Brenne, F.; Prashanth, K.G.; Mendes, R.G.; Rümmeli, M.H.; Gemming, T.; Eckert, J.; Scudino, S. Microstructure and mechanical properties of a heat-treatable Al-3.5 Cu-1.5 Mg-1Si alloy produced by selective laser melting. Mater. Sci. Eng. 2018, 711, 562–570. [Google Scholar] [CrossRef]
- Kou, S. A criterion for cracking during solidification. Acta Mater. 2015, 88, 366–374. [Google Scholar] [CrossRef]
- Roy, G.G.; Elmer, J.W.; DebRoy, T. Mathematical modeling of heat transfer, fluid flow, and solidification during linear welding with a pulsed laser beam. J. Appl. Phys. 2006, 100, 034903. [Google Scholar] [CrossRef]
- Eskin, D.G.; Katgerman, L. Thermal contraction during solidification of aluminium alloys. Mater. Sci. Forum 2006, 519, 1681–1686. [Google Scholar] [CrossRef]
- Lee, Y.S.; Kirka, M.M.; Dinwiddie, R.B.; Raghavan, N.; Turner, J.; Dehoff, R.R.; Babu, S.S. Role of scan strategies on thermal gradient and solidification rate in electron beam powder bed fusion. Addit. Manuf. 2018, 22, 516–527. [Google Scholar] [CrossRef]
- Hovig, E.W.; Holm, H.D.; Sørby, K. Effect of processing parameters on the relative density of AlSi10Mg processed by laser powder bed fusion. In Advanced Manufacturing and Automation VIII; Springer: Singapore, 2018; pp. 268–276. [Google Scholar]
- Bertoli, U.S.; Wolfer, A.J.; Matthews, M.J.; Delplanque, J.P.; Schoenung, J.M. On the limitations of volumetric energy density as a design parameter for selective laser melting. Mater. Des. 2017, 113, 331–340. [Google Scholar] [CrossRef]
- Tian, J.; Huang, Z.; Qi, W.; Li, Y.; Liu, J.; Hu, G. Dependence of microstructure, relative density and hardness of 18Ni-300 maraging steel fabricated by selective laser melting on the energy density. In Advances in Materials Processing; Springer: Singapore, 2018; pp. 229–241. [Google Scholar]
- Hann, D.B.; Iammi, J.; Folkes, J. A simple methodology for predicting laser-weld properties from material and laser parameters. J. Phys. D Appl. Phys. 2011, 44, 445401. [Google Scholar] [CrossRef]
- Popčević, P.; Smontara, A.; Ivkov, J.; Wencka, M.; Komelj, M.; Jeglič, P.; Vrtnik, S.; Bobnar, M.; Jagličić, Z.; Bauer, B.; et al. Anisotropic physical properties of the Al13Fe4 complex intermetallic and its ternary derivative Al13(Fe, Ni)4. Phys. Rev. B 2010, 81, 184203. [Google Scholar] [CrossRef]
- Martin, J.H.; Yahata, B.D.; Hundley, J.M.; Mayer, J.A.; Schaedler, T.A.; Pollock, T.M. 3D printing of high-strength aluminium alloys. Nature 2017, 549, 365–369. [Google Scholar] [CrossRef]
- Tan, Q.; Yin, Y.; Fan, Z.; Zhang, J.; Liu, Y.; Zhan, M.X. Uncovering the roles of LaB6-nanoparticle inoculant in the AlSi10Mg alloy fabricated via selective laser melting. Mater. Sci. Eng. A 2021, 800, 140365. [Google Scholar] [CrossRef]



| Element | Si | Fe | Cu | Mn | Mg | Zr | Zn | O | Al |
|---|---|---|---|---|---|---|---|---|---|
| Content (mass%) | 0.09 | 15.2 | 0.01 | 0.02 | 0.03 | 0.01 | 0.04 | 0.29 | Bal. |
| Parameter | Value |
|---|---|
| Laser power (P) | 50, 77, 80, 89, 102, 110, 115, 128, 140, 170 W |
| Scan speed (v) | 0.4, 0.5, 0.6, 0.7, 0.8, 1.0, 1.2, 1.4 m·s−1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, W.; Takata, N.; Suzuki, A.; Kobashi, M.; Kato, M. Processability and Optimization of Laser Parameters for Densification of Hypereutectic Al–Fe Binary Alloy Manufactured by Laser Powder Bed Fusion. Crystals 2021, 11, 320. https://doi.org/10.3390/cryst11030320
Wang W, Takata N, Suzuki A, Kobashi M, Kato M. Processability and Optimization of Laser Parameters for Densification of Hypereutectic Al–Fe Binary Alloy Manufactured by Laser Powder Bed Fusion. Crystals. 2021; 11(3):320. https://doi.org/10.3390/cryst11030320
Chicago/Turabian StyleWang, Wenyuan, Naoki Takata, Asuka Suzuki, Makoto Kobashi, and Masaki Kato. 2021. "Processability and Optimization of Laser Parameters for Densification of Hypereutectic Al–Fe Binary Alloy Manufactured by Laser Powder Bed Fusion" Crystals 11, no. 3: 320. https://doi.org/10.3390/cryst11030320
APA StyleWang, W., Takata, N., Suzuki, A., Kobashi, M., & Kato, M. (2021). Processability and Optimization of Laser Parameters for Densification of Hypereutectic Al–Fe Binary Alloy Manufactured by Laser Powder Bed Fusion. Crystals, 11(3), 320. https://doi.org/10.3390/cryst11030320









