Application of Reactive Oxygen Species-Based Nanomaterials in Dentistry: A Review
Abstract
1. Introduction
2. Reaction Oxide Species
2.1. ROS in Dentistry
2.2. Source of ROS Generation in Dentistry
2.2.1. Nonthermal Plasma (NTP) for ROS Generation
2.2.2. Laser for ROS Generation
2.2.3. ROS Generating from Bleaching Agents
2.2.4. Photodynamic Therapy and Light Sources for ROS Generation
2.2.5. Other Sources for ROS Generation
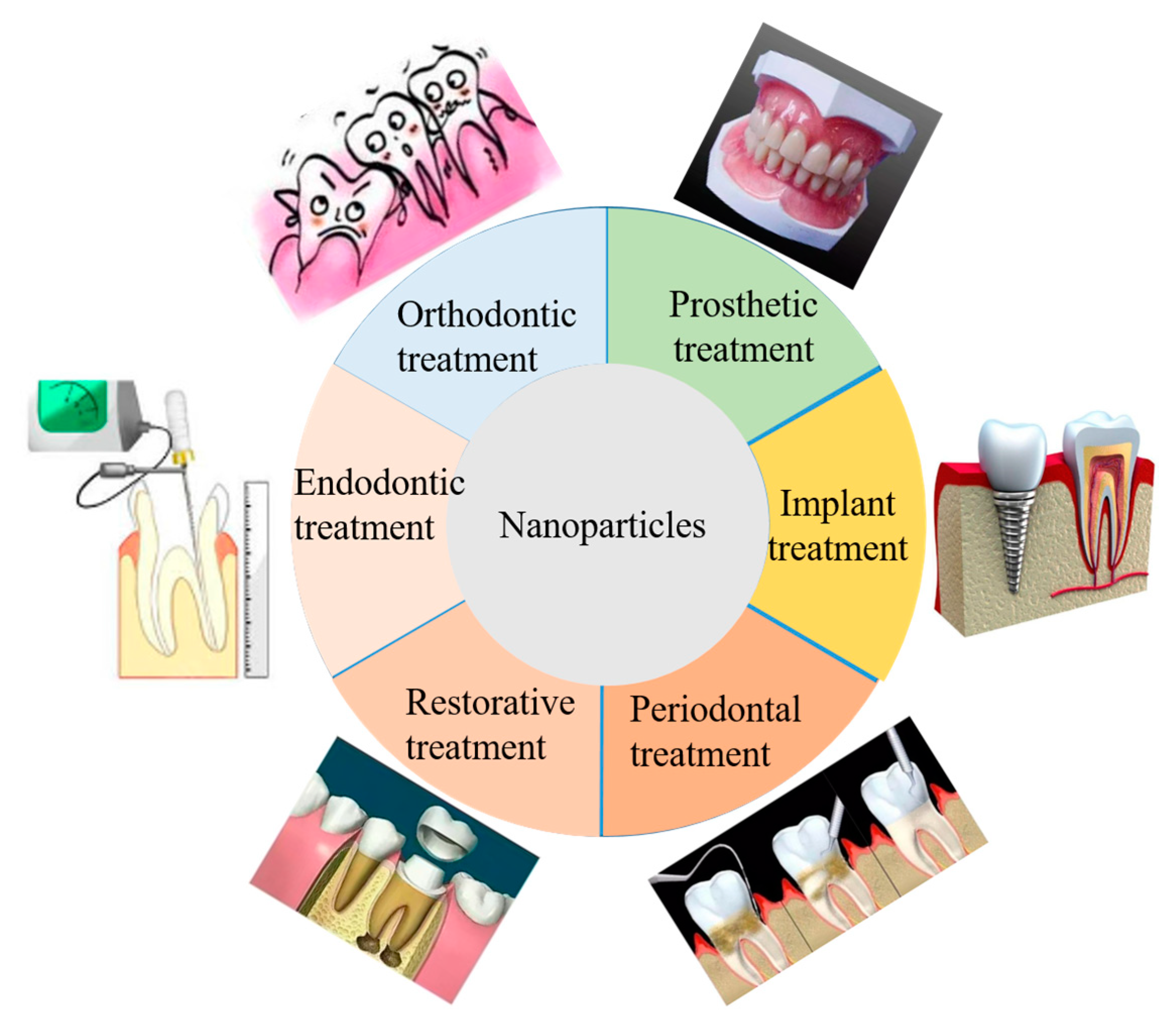
3. ROS-Based Nanomaterials Used in Dentistry
3.1. Metal Nanoparticles in Dental Treatment
3.1.1. ROS-Based Silver Nanoparticles
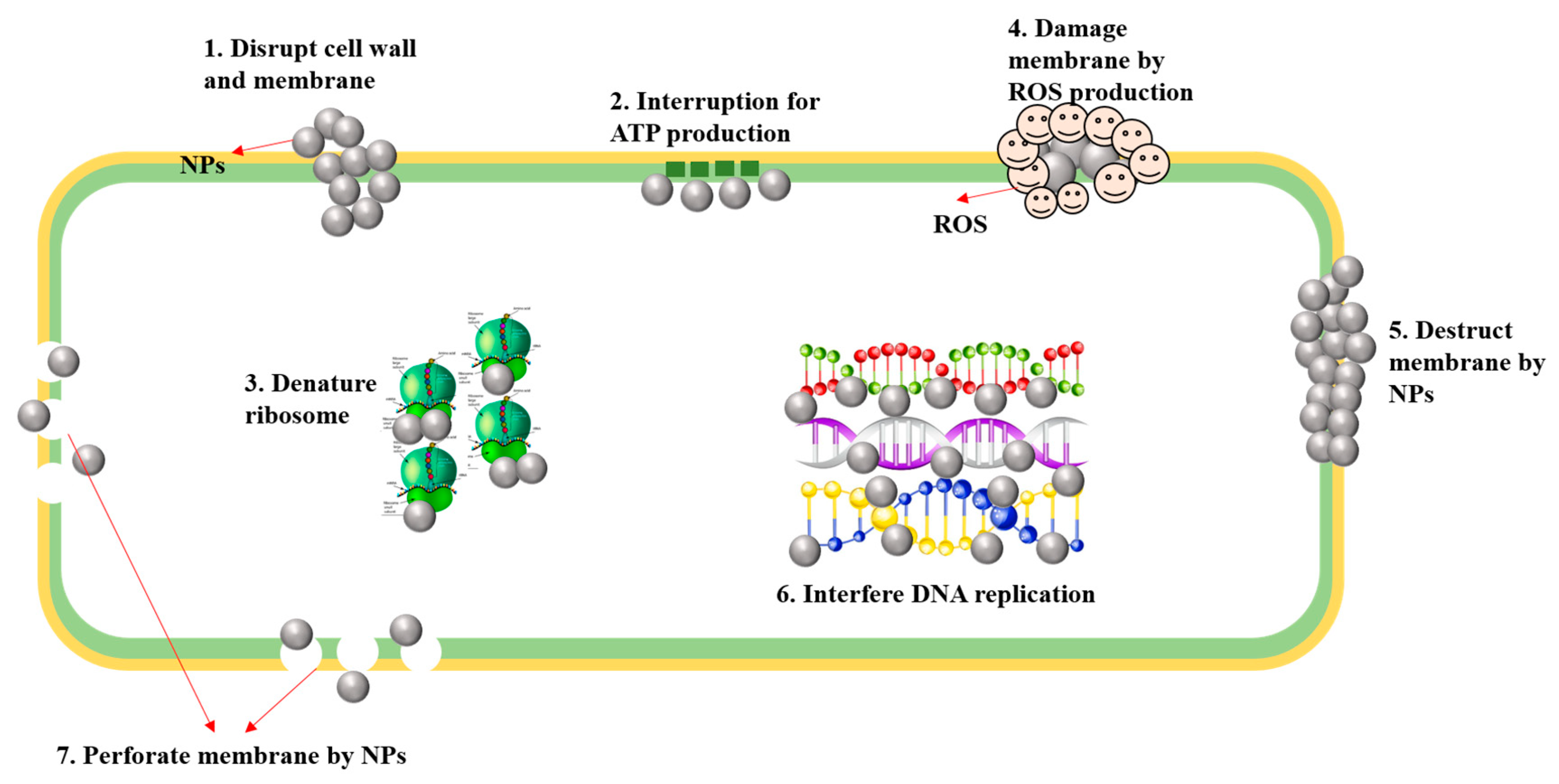
3.1.2. The Antibacterial Mechanism of ROS-Based Silver Nanoparticles in Dentistry
3.1.3. ROS-Based Au Nanoparticles
3.2. ROS-Based Metal Oxide Nanoparticles in Dentistry
3.2.1. ROS-Based Titanium Oxide Nanoparticles
3.2.2. ROS-Based ZnO Nanoparticles
3.3. Other Nanoparticles Used in Dentistry
4. Clinical Applications of ROS-Nanoparticles in Dentistry
5. Safety Evaluation of Nanoparticles in Dental Application
6. Conclusions and Future Prospect
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Marsh, P.D.; Martin, M.V.; Lewis, M.A.; Williams, D. Oral Microbiology e-Book; Elsevier: Amsterdam, The Netherlands, 2009. [Google Scholar]
- Filoche, S.; Wong, L.; Sissons, C.H. Oral biofilms: Emerging concepts in microbial ecology. J. Dent. Res. 2010, 89, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Melo, M.A.; Guedes, S.F.; Xu, H.H.; Rodrigues, L.K. Nanotechnology-based restorative materials for dental caries management. Trends Biotechnol. 2013, 31, 459–467. [Google Scholar] [CrossRef]
- Cury, J.A.; Tenuta, L.M.A. Enamel remineralization: Controlling the caries disease or treating early caries lesions? Braz. Oral Res. 2009, 23, 23–30. [Google Scholar] [CrossRef]
- Fontana, M.; Young, D.A.; Wolff, M.S.; Pitts, N.B.; Longbottom, C. Defining dental caries for 2010 and beyond. Dent. Clin. 2010, 54, 423–440. [Google Scholar] [CrossRef]
- Engel, E.; Michiardi, A.; Navarro, M.; Lacroix, D.; Planell, J.A. Nanotechnology in regenerative medicine: The materials side. Trends Biotechnol. 2008, 26, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Solga, A.; Cerman, Z.; Striffler, B.F.; Spaeth, M.; Barthlott, W. The dream of staying clean: Lotus and biomimetic surfaces. Bioinspiration Biomim. 2007, 2, S126. [Google Scholar] [CrossRef]
- Watson, P.S.; Pontefract, H.A.; Devine, D.A.; Shore, R.C.; Nattress, B.R.; Kirkham, J.; Robinson, C. Penetration of fluoride into natural plaque biofilms. J. Dent. Res. 2005, 84, 451–455. [Google Scholar] [CrossRef]
- Hannig, M.; Kriener, L.; Hoth-Hannig, W.; Becker-Willinger, C.; Schmidt, H. Influence of nanocomposite surface coating on biofilm formation in situ. J. Nanosci. Nanotechnol. 2007, 7, 4642–4648. [Google Scholar] [CrossRef]
- Hajipour, M.J.; Fromm, K.M.; Ashkarran, A.A.; de Aberasturi, D.J.; de Larramendi, I.R.; Rojo, T.; Serpooshan, V.; Parak, W.J.; Mahmoudi, M. Antibacterial properties of nanoparticles. Trends Biotechnol. 2012, 30, 499–511. [Google Scholar] [CrossRef]
- Li, X.; Cui, R.; Liu, W.; Sun, L.; Yu, B.; Fan, Y.; Feng, Q.; Cui, F.; Watari, F. The use of nanoscaled fibers or tubes to improve biocompatibility and bioactivity of biomedical materials. J. Nanomater. 2013, 2013, 16. [Google Scholar] [CrossRef]
- Li, X.M.; Lee, S.C.; Zhang, S.M.; Akasaka, T. Biocompatibility and Toxicity of Nanobiomaterials 2014. J. Nanomater. 2015, 2015, 259264. [Google Scholar] [CrossRef]
- Kalyanaraman, B.; Darley-Usmar, V.; Davies, K.J.; Dennery, P.A.; Forman, H.J.; Grisham, M.B.; Mann, G.E.; Moore, K.; Roberts, L.J., II; Ischiropoulos, H. Measuring reactive oxygen and nitrogen species with fluorescent probes: Challenges and limitations. Free Radic. Biol. Med. 2012, 52, 1–6. [Google Scholar] [CrossRef]
- Atashi, F.; Modarressi, A.; Pepper, M.S. The role of reactive oxygen species in mesenchymal stem cell adipogenic and osteogenic differentiation: A review. Stem. Cells Dev. 2015, 24, 1150–1163. [Google Scholar] [CrossRef]
- Park, H.J.; Park, S.; Roh, J.; Kim, S.; Choi, K.; Yi, J.; Kim, Y.; Yoon, J. Biofilm-inactivating activity of silver nanoparticles: A comparison with silver ions. J. Ind. Eng. Chem. 2013, 19, 614–619. [Google Scholar] [CrossRef]
- Ashkarran, A.A.; Hamidinezhad, H.; Haddadi, H.; Mahmoudi, M. Double-doped TiO2 nanoparticles as an efficient visible-light-active photocatalyst and antibacterial agent under solar simulated light. Appl. Surf. Sci. 2014, 301, 338–345. [Google Scholar] [CrossRef]
- Baumgardner, K.R.; Sulfaro, M.A. The anti–inflammatory effects of human recombinant copper–zinc superoxide dismutase on pulp inflammation. J. Endod. 2001, 27, 190–195. [Google Scholar] [CrossRef]
- Krifka, S.; Spagnuolo, G.; Schmalz, G.; Schweikl, H. A review of adaptive mechanisms in cell responses towards oxidative stress caused by dental resin monomers. Biomaterials 2013, 34, 4555–4563. [Google Scholar] [CrossRef]
- Nasir, N.M.; Lee, B.K.; Yap, S.S.; Thong, K.L.; Yap, S.L. Cold plasma inactivation of chronic wound bacteria. Arch. Biochem. Biophys. 2016, 605, 76–85. [Google Scholar] [CrossRef]
- Yang, H.L.; Liu, S.; Hu, T. Application of low-temperature plasma in dental clinical sterilization. Foreign Med. Sci. Stomatol. 2013, 40, 483–485. [Google Scholar]
- Laroussi, M. Low temperature plasma-based sterilization: Overview and state-of-the-art. Plasma Process. Polym. 2005, 2, 391–400. [Google Scholar] [CrossRef]
- Laroussi, M.; Tendero, C.; Lu, X.; Alla, S.; Hynes, W.L. Inactivation of bacteria by the plasma pencil. Plasma Process. Polym. 2006, 3, 470–473. [Google Scholar] [CrossRef]
- Sung, S.J.; Huh, J.B.; Yun, M.J.; Chang, B.M.W.; Jeong, C.M.; Jeon, Y.C. Sterilization effect of atmospheric pressure non-thermal air plasma on dental instruments. J. Adv. Prosthodont. 2013, 5, 2–8. [Google Scholar] [CrossRef]
- Kumar, S.C.; Sarada, P.; Reddy, S.C.; Reddy, S.M.; Dsv, N. Plasma torch toothbrush a new insight in fear free dentistry. J. Clin. Diagn. Res. JCDR 2014, 8, 7–10. [Google Scholar]
- Chen, M.; Zhang, Y.; Driver, M.S.; Caruso, A.N.; Yu, Q.; Wang, Y. Surface modification of several dental substrates by non-thermal, atmospheric plasma brush. Dent. Mater. 2013, 29, 871–880. [Google Scholar] [CrossRef] [PubMed]
- Kwon, J.H.; Park, H.C.; Zhu, T.; Yang, H.C. Inhibition of odontogenic differentiation of human dental pulp cells by dental resin monomers. Biomater. Res. 2015, 19, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Geurtsen, W.; Spahl, W.; Leyhausen, G. Residual monomer/additive release and variability in cytotoxicity of light-curing glass-ionomer cements and compomers. J. Dent. Res. 1998, 77, 2012–2019. [Google Scholar] [CrossRef] [PubMed]
- Saita, M.; Kobatashi, K.; Yoshino, F.; Hase, H.; Nonami, T.; Kimoto, K.; Masaichi, C.I. ESR investigation of ROS generated by H2O2 bleaching with TiO2 coated Hap. Dent. Mater. J. 2012, 31, 458–464. [Google Scholar] [CrossRef]
- Lee, D.H.; Lim, B.S.; Lee, Y.K.; Yang, H.C. Effects of hydrogen peroxide (H2O2) on alkaline phosphatase activity and matrix mineralization of odontoblast and osteoblast cell lines. Cell Biol. Toxicol. 2006, 22, 39–46. [Google Scholar] [CrossRef]
- Andersen, R.; Loebel, N.; Hammond, D.; Wilson, M. Treatment of periodontal disease by photodisinfection compared to scaling and root planning. J. Clin. Dent. 2007, 18, 34. [Google Scholar]
- Wilson, M. Lethal photosensitisation of oral bacteria and its potential application in the photodynamic therapy of oral infections. Photochem. Photobiol. Sci. 2004, 3, 412–418. [Google Scholar] [CrossRef]
- Jha, N.; Ryu, J.J.; Choi, E.H.; Kaushik, N.K. Generation and role of reactive oxygen and nitrogen species induced by plasma, lasers, chemical agents, and other systems in dentistry. Oxidative Med. Cell. Longev. 2017, 2017, 7542540. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, C.; Berganza, C.; Zhang, J. Cold Atmospheric Plasma: Methods of production and application in dentistry and oncology. Med. Gas Res. 2013, 3, 2045–9912. [Google Scholar] [CrossRef]
- Gherardi, M.; Tonini, R.; Colombo, V. Plasma in dentistry: Brief history and current status. Trends Biotechnol. 2018, 36, 583–585. [Google Scholar] [CrossRef]
- Rosa, D.S.A.; Aranha, A.C.C.; de Paula Eduardo, C.; Aoki, A. Esthetic treatment of gingival melanin hyperpigmentation with Er: YAG laser: Short-term clinical observations and patient follow-up. J. Periodontol. 2007, 78, 2018–2025. [Google Scholar] [CrossRef]
- Sobouti, F.; Rakhshan, V.; Chiniforush, N.; Khatami, M. Effects of laser-assisted cosmetic smile lift gingivectomy on postoperative bleeding and pain in fixed orthodontic patients: A controlled clinical trial. Prog. Orthod. 2014, 15, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Farivar, S.; Malekshahabi, T.; Shiari, R. Biological effects of low level laser therapy. J. Lasers Med Sci. 2014, 5, 58. [Google Scholar] [PubMed]
- Matsui, S.; Takahashi, C.; Tsujimoto, Y.; Matsushima, K. Stimulatory effects of low-concentration reactive oxygen species on calcification ability of human dental pulp cells. J. Endodontia 2009, 35, 67–72. [Google Scholar] [CrossRef] [PubMed]
- Eimar, H.; Siciliano, R.; Abdallah, M.N.; Abi Nader, S.; Amin, W.M.; Martinez, P.P.; Celemin, A.; Cerruti, M.; Tamimi, F. Hydrogen peroxide whitens teeth by oxidizing the organic structure. J. Dent. 2012, 40, e25–e33. [Google Scholar] [CrossRef]
- Diogo, P.; Gonçalves, T.; Palma, P.; Santos, J.M. Photodynamic antimicrobial chemotherapy for root canal system asepsis: A narrative literature review. Int. J. Dent. 2015, 2015, 269205. [Google Scholar] [CrossRef]
- Yoshida, A.; Iwata, S.; Iizuka, J.; Takahashi, S.S.; Wada-Takahashi, S.; Miyamoto, C.; Maehata, Y.; Ogura, Y.; Lee, M.; Yo, F. Blue light from dental resin curing unit causes light-induced vasoconstriction in isolated rat aorta. Oral Health Dent. Manag. 2014, 13, 1147–1151. [Google Scholar]
- Nasrin, A.; Hassan, M.; Gomes, V.G. Two-photon active nucleus-targeting carbon dots: Enhanced ROS generation and photodynamic therapy for oral cancer. Nanoscale 2020, 12, 20598–20603. [Google Scholar] [CrossRef]
- Desouky, O.; Ding, N.; Zhou, G. Targeted and non-targeted effects of ionizing radiation. J. Radiat. Res. Appl. Sci. 2015, 8, 247–254. [Google Scholar] [CrossRef]
- Bush, M.A.; Hermanson, A.S.; Yetto, R.J.; Wieczkowski, G. The use of ultraviolet LED illumination for composite resin removal: An in vitro study. Gen. Dent. 2010, 58, 214–218. [Google Scholar]
- Yin, I.X.; Zhang, J.; Zhao, I.S.; Mei, M.L.; Li, Q.; Chu, C.H. The Antibacterial Mechanism of Silver Nanoparticles and Its Application in Dentistry. Int. J. Nanomed. 2020, 15, 2555. [Google Scholar] [CrossRef]
- Jandt, K.D.; Watts, D.C. Nanotechnology in dentistry: Present and future perspectives on dental nanomaterials. Dent. Mater. 2020, 36, 1365–1378. [Google Scholar] [CrossRef]
- Radad, K.; Al-Shraim, M.; Moldzio, R.; Rausch, W.D. Recent advances in benefits and hazards of engineered nanoparticles. Environ. Toxicol. Pharm. 2012, 34, 661–672. [Google Scholar] [CrossRef]
- Bapat, R.A.; Chaubal, T.V.; Joshi, C.P.; Bapat, P.R.; Choudhury, H.; Pandey, M.; Gorain, B.; Kesharwani, P. An overview of application of silver nanoparticles for biomaterials in dentistry. Mater. Sci. Eng. C 2018, 91, 881–898. [Google Scholar] [CrossRef]
- Martínez-Gutiérrez, F.; Guajardo-Pacheco, J.M.; Noriega-Trevino, M.E.; Thi, E.P.; Reiner, N.; Orrantia, E.; Av-Gay, Y.; Ruiz, F.; Bach, H. Antimicrobial activity, cytotoxicity and inflammatory response of novel plastics embedded with silver nanoparticles. Future Microbiol. 2013, 8, 403–411. [Google Scholar] [CrossRef]
- Durner, J.; Stojanovic, M.; Urcan, E.; Hickel, R.; Reichl, F.X. Influence of silver nano-particles on monomer elution from light-cured composites. Dent. Mater. 2011, 27, 631–636. [Google Scholar] [CrossRef]
- Durán, N.; Nakazato, G.; Seabra, A.B. Antimicrobial activity of biogenic silver nanoparticles, and silver chloride nanoparticles: An overview and comments. Appl. Microbiol. Biotechnol. 2016, 100, 6555–6570. [Google Scholar] [CrossRef]
- Regiel-Futyra, A.; Kus-Liśkiewicz, M.; Sebastian, V.; Irusta, S.; Arruebo, M.; Stochel, G.; Kyzioł, A. Development of noncytotoxic chitosan–gold nanocomposites as efficient antibacterial materials. ACS Appl. Mater. Interfaces 2015, 7, 1087–1099. [Google Scholar] [CrossRef]
- Braz, A.K.S.; de Araujo, R.E.; Ohulchanskyy, T.Y.; Shukla, S.; Bergey, E.J.; Gomes, A.S.; Prasad, P.N. In situ gold nanoparticles formation: Contrast agent for dental optical coherence tomography. J. Biomed. Opt. 2012, 17, 066003. [Google Scholar] [CrossRef]
- Mcintyre, R.A. Common nano-materials and their use in real world applications. Sci. Prog. 2012, 95, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Monteiro, N.R.; Basting, R.T.; Amaral, F.L.B.D.; França, F.M.G.; Turssi, C.P.; Gomes, O.P.; Lisboa Filho, P.N.; Kantovitz, K.R.; Basting, R.T. Titanium dioxide nanotubes incorporated into bleaching agents: Physicochemical characterization and enamel color change. J. Appl. Oral Sci. 2020, 28. [Google Scholar]
- Komatsu, O.; Nishida, H.; Sekino, T.; Yamamoto, K. Application of titanium dioxide nanotubes to tooth whitening. Nano Biomed. 2014, 6, 63–72. [Google Scholar]
- Argueta-Figueroa, L.; Torres-Gómez, N.; Scougall-Vilchis, R.J.; García-Contreras, R. Biocompatibility and antibacterial activity of nitrogen-doped titanium dioxide nanoparticles for use in dental resin formulations. Int. J. Nanomed. 2016, 11, 6459. [Google Scholar]
- Kurzmann, C.; Verheyen, J.; Coto, M.; Kumar, R.V.; Divitini, G.; Shokoohi-Tabrizi, H.A.; Verheyen, P.; De Moor, R.J.G.; Moritz, A.; Agis, H. In vitro evaluation of experimental light activated gels for tooth bleaching. Photochem. Photobiol. Sci. 2019, 18, 1009–1019. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Petersen, E.J.; Watson, S.S.; Sims, C.M.; Kassman, A.; Frukhtbeyn, S.; Skrtic, D.; Ok, M.T.; Jacobs, D.S.; Reipa, V.; et al. Biophysical characterization of functionalized titania nanoparticles and their application in dental adhesives. Acta Biomater. 2017, 53, 585–597. [Google Scholar] [CrossRef] [PubMed]
- Sharma, V.; Shukla, R.K.; Saxena, N.; Parmar, D.; Das, M.; Dhawan, A. DNA damaging potential of zinc oxide nanoparticles in human epidermal cells. Toxicol. Lett. 2009, 185, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Gu, H.; Fan, D.; Gao, J.; Zou, W.; Peng, Z.; Zhao, Z.; Ling, J.; LeGeros, R.Z. Effect of ZnCl2 on plaque growth and biofilm vitality. Arch. Oral Biol. 2012, 57, 369–375. [Google Scholar] [CrossRef]
- Chronopoulou, L.; Nocca, G.; Castagnola, M.; Paludetti, G.; Ortaggi, G.; Sciubba, F.; Bevilacqua, M.; Lupi, A.; Gambarini, G.; Palocci, C. Chitosan based nanoparticles functionalized with peptidomimetic derivatives for oral drug delivery. New Biotechnol. 2016, 33, 23–31. [Google Scholar] [CrossRef]
- Khan, S.T.; Ahamed, M.; Al-Khedhairy, A.; Musarrat, J. Biocidal effect of copper and zinc oxide nanoparticles on human oral microbiome and biofilm formation. Mater. Lett. 2013, 97, 67–70. [Google Scholar] [CrossRef]
- Misawa, M.; Takahashi, J. Generation of reactive oxygen species induced by gold nanoparticles under X-ray and UV Irradiations. Nanomed. Nanotechnol. Biol. Med. 2011, 7, 604–614. [Google Scholar] [CrossRef] [PubMed]
- Priyadarsini, S.; Mukherjee, S.; Mishra, M. Nanoparticles used in dentistry: A review. J. Oral Biol. Craniofacial Res. 2018, 8, 58–67. [Google Scholar] [CrossRef]
- Dwivedi, S.; Wahab, R.; Khan, F.; Mishra, Y.K.; Musarrat, J.; Al-Khedhairy, A.A. Reactive oxygen species mediated bacterial biofilm inhibition via zinc oxide nanoparticles and their statistical determination. PLoS ONE 2014, 9, e111289. [Google Scholar] [CrossRef] [PubMed]
- Guerreiro-Tanomaru, J.M.; Trindade-Junior, A.; Cesar Costa, B.; da Silva, G.F.; Drullis Cifali, L.; Basso Bernardi, M.I.; Tanomaru-Filho, M. Effect of zirconium oxide and zinc oxide nanoparticles on physicochemical properties and antibiofilm activity of a calcium silicate-based material. Sci. World J. 2014, 2014, 975213. [Google Scholar] [CrossRef]
- Tsai, Y.Y.; Oca-Cossio, J.; Lin, S.M.; Woan, K.; Yu, P.C.; Sigmund, W. Reactive oxygen species scavenging properties of ZrO2–CeO2 solid solution nanoparticles. Future Med. 2008, 637–645. [Google Scholar]
- Toodehzaeim, M.H.; Zandi, H.; Meshkani, H.; Firouzabadi, A.H. The effect of CuO nanoparticles on antimicrobial effects and shear bond strength of orthodontic adhesives. J. Dent. 2018, 19, 1. [Google Scholar]
- Bitar, A.; Ahmad, N.M.; Fessi, H.; Elaissari, A. Silica-based nanoparticles for biomedical applications. Drug Discov. Today 2012, 17, 1147–1154. [Google Scholar] [CrossRef]
- Timpe, N.; Fullriede, H.; Borchers, L.; Stiesch, M.; Behrens, P.; Menzel, H. Nanoporous silica nanoparticles with spherical and anisotropic shape as fillers in dental composite materials. BioNanoMaterials 2014, 15, 89–99. [Google Scholar] [CrossRef]
- Akasaka, T.; Nakata, K.; Uo, M.; Watari, F. Modification of the dentin surface by using carbon nanotubes. Bio-Med Mater. Eng. 2009, 19, 179–185. [Google Scholar] [CrossRef]
- Ni, C.; Zhou, J.; Kong, N.; Bian, T.; Zhang, Y.; Huang, X.; Xiao, Y.; Yang, W.; Yan, F. Gold nanoparticles modulate the crosstalk between macrophages and periodontal ligament cells for periodontitis treatment. Biomaterials 2019, 206, 115–132. [Google Scholar] [CrossRef]
- Sun, X.; Wang, L.; Lynch, C.D.; Sun, X.; Li, X.; Qi, M.; Ma, C.; Li, C.; Dong, B.; Zhou, Y.; et al. Nanoparticles having amphiphilic silane containing Chlorin e6 with strong anti-biofilm activity against periodontitis-related pathogens. J. Dent. 2019, 81, 70–84. [Google Scholar] [CrossRef] [PubMed]
- Bapat, R.A.; Joshi, C.P.; Bapat, P.; Chaubal, T.V.; Pandurangappa, R.; Jnanendrappa, N.; Gorain, B.; Khurana, S.; Kesharwani, P. The use of nanoparticles as biomaterials in dentistry. Drug Discov. Today 2019, 24, 85–98. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.; Chen, A.; Zhang, Y.; Wang, J.; Shao, L.; Wei, L. Application of dental nanomaterials: Potential toxicity to the central nervous system. Int. J. Nanomed. 2015, 10, 3547. [Google Scholar]
| ROS Source in Dentistry | Application in Dentistry | Ref. |
|---|---|---|
| Nonthermal Plasma | Wound healing, sterilization in gingival crevices, biofilm removal, dental implants, and cleaning dentin during periodontal regeneration. | [23,24,25] |
| Laser | Tissue repair, gingivectomy | [26,27] |
| Bleaching Agents | Destroy root pathogens, sterilization | [28,29] |
| Photodynamic Therapy and Light Sources | Cause cellular damage, break biofilm, act as adhensive materials | [30,31] |
| Ionizing Radiation and UV rays | Break DNA molecule of bacteria, radiotherapy in dentistry, elimination cancer cells | [32] |
| Nanoparticles | Dental Application | Mechanisms/ROS Generation | Superiorities | Inferiorities | Ref. |
|---|---|---|---|---|---|
| Silver | Antibacterial agents, dental restorative materials, dental implants, root canal irrigation, orthodontic treatment. | Producing ROS, Releasing free silver ions leads to interruption of ATP molecules, preventing DNA replication or silver ions (Ag+) directly damage cell membranes. | Reduce bacterial colonization and improve oral health. biocompatibility, low toxicity and long-term antibacterial activity. | AgNPs are toxic, may cause silver-poisoning, lacking of research data in vivo to prove its safety. | [48] |
| Gold | Gingival inflammation, dental caries treatment, tissue engineering, dental implants, cancer diagnosis. | ROS production. | Small size, large specific surface area, with minimal adverse to mammals. | Toxic. Particles distribution in various organs. Larger gold nanoparticles scattered throughout the liver and spleen, harmful effects on various systems. | [64] |
| TiO2 | Preparing dental adhesives, dental restoration. | ROS production. | Compared with traditional dental adhesives, dental adhesives with acrylic functionalized nano-titanium dioxide have a stronger binding effect on human teeth. | The cytotoxicity and genotoxicity cannot be warranted. | [59,65] |
| ZnO | Deformation and damage of cells, surface coating material with antibacterial activity. | ROS production, inhibition on bacterial growth and biofilm formation. | Cell wall of bacteria can be destroyed at low concentration of ZnO-NPs. | Its mechanism of inhibition on bacterial growth and biofilm formation is still obscure. | [66] |
| Zirconia | Reducing bacterial adhesion to the tooth surface, providing protection against dental caries, effective polishing agent. | Yield ROS. | Similar mechanical properties and color like a tooth, have low cytotoxicity, sensible biocompatibility, and high fracture resistance. | Cytotoxic, inducing oxidative stress in cells, leading to cell death. | [67,68] |
| CuO | CuO NPs can improve the antibacterial performance and binding strength as orthodontic adhesives. | ROS production. | Low-priced and more stable, have strong antimicrobial effects on inhibition of bacterial colonization and plaque development. | Exhibit toxic effects. | [69] |
| Silica | Tooth filler, tooth whitening, prevention for tooth cavity, antibacterial agent. | By polishing prevents dental caries, which acts as a primary defense mechanism against the cariogenic bacteria. | Good biocompatibility, low toxicity, low density, significant adsorption capacity and low cost. | Silica nanoparticles, like crystal particles, can induce silicosis and lung cancer. SiNPs are cytotoxic. In addition, it can also induce oxidative stress and mediate cell apoptosis. SiNPs also have genotoxic effects (DNA damage, gene regulation involving apoptosis and autophagy) and immunotoxic effects. | [70,71] |
| Carbon nanotube | Teeth filling, coating layer on the teeth surface. | Carbon nanotubes can adhere to the surface of dentin and cementum by interacting with exposed collagen fibers. | Large surface area, easy to adhere to the surface of teeth, dentin and cementum. | May induce inflammation and fibrosis under certain conditions. | [72] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, F.; Hong, T.; Xie, J.; Zhan, X.; Wang, Y. Application of Reactive Oxygen Species-Based Nanomaterials in Dentistry: A Review. Crystals 2021, 11, 266. https://doi.org/10.3390/cryst11030266
Liu F, Hong T, Xie J, Zhan X, Wang Y. Application of Reactive Oxygen Species-Based Nanomaterials in Dentistry: A Review. Crystals. 2021; 11(3):266. https://doi.org/10.3390/cryst11030266
Chicago/Turabian StyleLiu, Fenglin, Tingting Hong, Jingxian Xie, Xuehui Zhan, and Yuehong Wang. 2021. "Application of Reactive Oxygen Species-Based Nanomaterials in Dentistry: A Review" Crystals 11, no. 3: 266. https://doi.org/10.3390/cryst11030266
APA StyleLiu, F., Hong, T., Xie, J., Zhan, X., & Wang, Y. (2021). Application of Reactive Oxygen Species-Based Nanomaterials in Dentistry: A Review. Crystals, 11(3), 266. https://doi.org/10.3390/cryst11030266





