Case Study of High-Throughput Drug Screening and Remote Data Collection for SARS-CoV-2 Main Protease by Using Serial Femtosecond X-ray Crystallography
Abstract
:1. Introduction
2. Materials and Methods
2.1. Gene Construct Design, Protein Expression, and Purification
2.2. Crystallization of Protein with Drug by Using Soaking Method
2.3. Transport of Microcrystals
2.4. Injection of Microcrystals
2.5. Data Collection at LCLS
2.6. Data Processing
2.7. Structure Determination
3. Results
3.1. Serial Crystallography Based Faster High-Throughput Drug Screening at an XFEL
3.2. Determining Mpro Structures with Mitigated Radiation Damage at Near Physiological Temperature
3.3. Interpretation of Experimental Findings
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Morse, S.S. Factors in the emergence of infectious diseases. Plagues Politics 1995, 1, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Fauci, A.S.; Folkers, G.K. The world must build on three decades of scientific advances to enable a new generation to live free of HIV/AIDs. Health Aff. 2012, 31, 1529–1536. [Google Scholar] [CrossRef] [Green Version]
- Steere, A.C.; Coburn, J.; Glickstein, L. The emergence of Lyme disease. J. Clin. Investig. 2004, 113, 1093–1101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morens, D.M.; Taubenberger, J.K.; Fauci, A.S. The persistent legacy of the 1918 influenza virus. N. Engl. J. Med. 2009, 361, 225–229. [Google Scholar] [CrossRef] [Green Version]
- Andersen, K.G.; Rambaut, A.; Lipkin, W.I.; Holmes, E.C.; Garry, R.F. The proximal origin of SARS-CoV-2. Nat. Med. 2020, 26, 450–452. [Google Scholar] [CrossRef] [Green Version]
- Jones, K.E.; Patel, N.G.; Levy, M.A.; Storeygard, A.; Balk, D.; Gittleman, J.L.; Daszak, P. Global trends in emerging infectious diseases. Nature 2008, 451, 990–993. [Google Scholar] [CrossRef]
- Qiao, J.; Li, Y.-S.; Zeng, R.; Liu, F.-L.; Luo, R.-H.; Huang, C.; Wang, Y.-F.; Zhang, J.; Quan, B.; Shen, C.; et al. SARS-CoV-2 mpro inhibitors with antiviral activity in a transgenic mouse model. Science 2021, 371, 1374–1378. [Google Scholar] [CrossRef] [PubMed]
- Dai, W.; Zhang, B.; Jiang, X.-M.; Su, H.; Li, J.; Zhao, Y.; Xie, X.; Jin, Z.; Peng, J.; Liu, F.; et al. Structure-based design of antiviral drug candidates targeting the SARS-CoV-2 main protease. Science 2020, 368, 1331–1335. [Google Scholar] [CrossRef] [Green Version]
- Li, Q.; Kang, C.B. Progress in developing inhibitors of SARS-CoV-2 3c-like protease. Microorganisms 2020, 8, 1250. [Google Scholar] [CrossRef] [PubMed]
- Mengist, H.M.; Dilnessa, T.; Jin, T. Structural basis of potential inhibitors targeting SARS-CoV-2 main protease. Front. Chem. 2021, 9, 622898. [Google Scholar] [CrossRef] [PubMed]
- Sacco, M.D.; Ma, C.; Lagarias, P.; Gao, A.; Townsend, J.A.; Meng, X.; Dube, P.; Zhang, X.; Hu, Y.; Kitamura, N.; et al. Structure and inhibition of the SARS-CoV-2 main protease reveal strategy for developing dual inhibitors against Mpro and cathepsin L. Sci. Adv. 2020, 6, eabe0751. [Google Scholar] [CrossRef] [PubMed]
- Ullrich, S.; Nitsche, C. The SARS-CoV-2 main protease as drug target. Bioorg. Med. Chem. Lett. 2020, 30, 127377. [Google Scholar] [CrossRef] [PubMed]
- Mahase, E. COVID-19: Pfizer’s paxlovid is 89% effective in patients at risk of serious illness, company reports. BMJ 2021, 375, n2697. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Lin, D.; Kusov, Y.; Nian, Y.; Ma, Q.; Wang, J.; von Brunn, A.; Leyssen, P.; Lanko, K.; Neyts, J.; et al. α-Ketoamides as broad-spectrum inhibitors of coronavirus and enterovirus replication: Structure-based design, synthesis, and activity. J. Med. Chem. 2020, 63, 4562–4578. [Google Scholar] [CrossRef] [PubMed]
- Pillaiyar, T.; Manickam, M.; Namasivayam, V.; Hayashi, Y.; Jung, S.-H. An overview of severe acute respiratory syndrome–coronavirus (SARS-CoV) 3CL protease inhibitors: Peptidomimetics and small molecule chemotherapy. J. Med. Chem. 2016, 59, 6595–6628. [Google Scholar] [CrossRef] [PubMed]
- Huang, N.; Deng, H.; Liu, B.; Wang, D.; Zhao, Z. Features and futures of X-ray free-electron lasers. Innovation 2021, 2, 100097. [Google Scholar] [CrossRef]
- Durdagi, S.; Dag, Ç.; Dogan, B.; Sierra, R.; Snell, E.; DeMirci, H. Near-physiological-temperature serial crystallography reveals conformations of SARS-CoV-2 main protease active site for improved drug repurposing. Structure 2021, 29, 1–15. [Google Scholar] [CrossRef]
- Blay, V.; Tolani, B.; Ho, S.P.; Arkin, M.R. High-throughput screening: Today’s biochemical and cell-based approaches. Drug Discov. Today 2020, 25, 1807–1821. [Google Scholar] [CrossRef]
- Aceti, D.J.; Ahmed, H.; Westler, W.M.; Wu, C.; Dashti, H.; Tonelli, M.; Eghbalnia, H.; Amarasinghe, G.K.; Markley, J.L. Fragment screening targeting Ebola virus nucleoprotein C-terminal domain identifies lead candidates. Antivir. Res. 2020, 180, 104822. [Google Scholar] [CrossRef] [PubMed]
- Aretz, J.; Baukmann, H.; Shanina, E.; Hanske, J.; Wawrzinek, R.; Zapol’skii, V.A.; Seeberger, P.H.; Kaufmann, D.E.; Rademacher, C. Identification of multiple druggable secondary sites by fragment screening against DC-SIGN. Angew. Chem. Int. Ed. 2017, 56, 7292–7296. [Google Scholar] [CrossRef]
- Schuller, M.; Correy, G.J.; Gahbauer, S.; Fearon, D.; Wu, T.; Díaz, R.E.; Young, I.D.; Carvalho Martins, L.; Smith, D.H.; Schulze-Gahmen, U.; et al. Fragment binding to the nsp3 macrodomain of SARS-COV-2 identified through crystallographic screening and computational docking. Sci. Adv. 2021, 7, eabf8711. [Google Scholar] [CrossRef]
- Gupta, S.; Singh, A.K.; Kushwaha, P.P.; Prajapati, K.S.; Shuaib, M.; Senapati, S.; Kumar, S. Identification of potential natural inhibitors of SARS-CoV2 main protease by molecular docking and Simulation Studies. J. Biomol. Struct. Dyn. 2020, 39, 4334–4345. [Google Scholar] [CrossRef] [PubMed]
- Reiner, Ž.; Hatamipour, M.; Banach, M.; Pirro, M.; Al-Rasadi, K.; Jamialahmadi, T.; Radenkovic, D.; Montecucco, F.; Sahebkar, A. Statins and the COVID-19 main protease: In silico evidence on direct interaction. Arch. Med. Sci. 2020, 16, 490–496. [Google Scholar] [CrossRef]
- Dhote, A.M.; Patil, V.R.; Lokwani, D.K.; Amnerkar, N.D.; Ugale, V.G.; Charbe, N.B.; Bhongade, B.A.; Khadse, S.C. Strategic analyses to identify key structural features of antiviral/antimalarial compounds for their binding interactions with 3CLpro, PLpro and RdRp of SARS-CoV-2: In silico molecular docking and dynamic simulation studies. J. Biomol. Struct. Dyn. 2021, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Choudhary, S.; Silakari, O. Scaffold morphing of arbidol (umifenovir) in search of multi-targeting therapy halting the interaction of SARS-CoV-2 with ACE2 and other proteases involved in COVID-19. Virus Res. 2020, 289, 198146. [Google Scholar] [CrossRef]
- Ekins, S.; Mottin, M.; Ramos, P.R.P.S.; Sousa, B.K.P.; Neves, B.J.; Foil, D.H.; Zorn, K.M.; Braga, R.C.; Coffee, M.; Southan, C.; et al. Déjà Vu: Stimulating open drug discovery for SARS-CoV-2. Drug Discov. Today 2020, 25, 928–941. [Google Scholar] [CrossRef]
- Copertino, D.C.; Duarte, R.R.; Powell, T.R.; Mulder Rougvie, M.; Nixon, D.F. Montelukast drug activity and potential against severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). J. Med. Virol. 2020, 93, 187–189. [Google Scholar] [CrossRef]
- Durdagi, S.; Avsar, T.; Orhan, M.D.; Serhatli, M.; Balcioglu, B.K.; Ozturk, H.U.; Kayabolen, A.; Cetin, Y.; Aydinlik, S.; Bagci-Onder, T.; et al. The neutralization effect of montelukast on SARS-CoV-2 is shown by multiscale in silico simulations and combined in vitro studies. Mol. Ther. 2021, in press. [Google Scholar] [CrossRef]
- Sierra, R.G.; Laksmono, H.; Kern, J.; Tran, R.; Hattne, J.; Alonso-Mori, R.; Lassalle-Kaiser, B.; Glöckner, C.; Hellmich, J.; Schafer, D.W.; et al. Nanoflow electrospinning serial femtosecond crystallography. Acta Crystallogr. Sect. D Struct. Biol. 2012, 68, 1584–1587. [Google Scholar] [CrossRef]
- Sierra, R.G.; Gati, C.; Laksmono, H.; Dao, E.H.; Gul, S.; Fuller, F.; Kern, J.; Chatterjee, R.; Ibrahim, M.; Brewster, A.S.; et al. Concentric-flow electrokinetic injector enables serial crystallography of ribosome and photosystem II. Nat. Methods 2015, 13, 59–62. [Google Scholar] [CrossRef] [PubMed]
- Mariani, V.; Morgan, A.; Yoon, C.H.; Lane, T.J.; White, T.A.; O’Grady, C.; Kuhn, M.; Aplin, S.; Koglin, J.; Barty, A.; et al. OnDA: Online data analysis and feedback for serial X-ray imaging. J. Appl. Cryst. 2016, 49, 1073–1080. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Damiani, D.; Dubrovin, M.; Gaponenko, I.; Kroeger, W.; Lane, T.J.; Mitra, A.; O’Grady, C.P.; Salnikov, A.; Sanchez-Gonzalez, A.; Schneider, D.; et al. Linac coherent light source data analysis using psana. J. Appl. Cryst. 2016, 49, 672–679. [Google Scholar] [CrossRef]
- Thayer, J.; Damiani, D.; Ford, C.; Dubrovin, M.; Gaponenko, I.; O’Grady, C.P.; Kroeger, W.; Pines, J.; Lane, T.J.; Salnikov, A.; et al. Data systems for the linac coherent light source. Adv. Struct. Chem. Imag. 2017, 3, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Driel, T.B.; Nelson, S.; Armenta, R.; Blaj, G.; Boo, S.; Boutet, S.; Doering, D.; Dragone, A.; Hart, P.; Haller, G.; et al. The epix10k 2-megapixel hard X-ray detector at LCLS. J. Synchrotron Radiat. 2020, 27, 608–615. [Google Scholar] [CrossRef]
- Barty, A.; Kirian, R.A.; Maia, F.R.; Hantke, M.; Yoon, C.H.; White, T.A.; Chapman, H. Cheetah: Software for high-throughput reduction and analysis of serial femtosecond X-ray diffraction data. J. Appl. Cryst. 2014, 47, 1118–1131. [Google Scholar] [CrossRef] [Green Version]
- White, T.A.; Kirian, R.A.; Martin, A.V.; Aquila, A.; Nass, K.; Barty, A.; Chapman, H.N. CrystFEL: A software suite for snapshot serial crystallography. J. Appl. Cryst. 2012, 45, 335–341. [Google Scholar] [CrossRef] [Green Version]
- White, T.A.; Mariani, V.; Brehm, W.; Yefanov, O.; Barty, A.; Beyerlein, K.R.; Chervinskii, F.; Galli, L.; Gati, C.; Nakane, T.; et al. Recent developments in CrystFEL. J. Appl. Cryst. 2016, 49, 680–689. [Google Scholar] [CrossRef] [Green Version]
- Winn, M.D. Overview of the CCP4 suite and current developments. Acta Crystallogr. Sect. D Struct. Biol. 2011. [Google Scholar] [CrossRef] [Green Version]
- McCoy, A.J.; Grosse-Kunstleve, R.W.; Adams, P.D.; Winn, M.D.; Storoni, L.C.; Read, R.J. Phaser crystallographic software. J. Appl. Cryst. 2007, 40, 658–678. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adams, P.D.; Afonine, P.V.; Bunkóczi, G.; Chen, V.B.; Davis, I.W.; Echols, N.; Headd, J.J.; Hung, L.W.; Kapral, G.J.; Grosse-Kunstleve, R.W.; et al. PHENIX: A comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. Sect. D Biol. Crystallogr. 2010, 66, 213–221. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Emsley, P.; Cowtan, K. Coot: Model-building tools for molecular graphics. Acta Crystallogr. Sect. D Biol. Crystallogr. 2004, 60, 2126–2132. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- DeLano, W. Pymol. In The PyMOL Molecular Graphics System, Version; Schrödinger, LLC: New York, NY, USA, 2020; Volume 2. [Google Scholar]
- Wienen-Schmidt, B.; Oebbeke, M.; Ngo, K.; Heine, A.; Klebe, G. Two methods, one goal: Structural differences between cocrystallization and crystal soaking to discover ligand binding poses. ChemMedChem 2020, 16, 292–300. [Google Scholar] [CrossRef]
- Mansoor, S.; Saadat, S.; Amin, A.; Ali, I.; Ghaffar, M.T.; Amin, U.; Mukhtar, M. A case for montelukast in COVID-19: “The use of computational docking to estimate the effects of montelukast on potential viral main protease catalytic site”. Res. Sq. 2020. [Google Scholar] [CrossRef]
- Watashi, K. Identifying and repurposing antiviral drugs against severe acute respiratory syndrome coronavirus 2 with in silico and in vitro approaches. Biochem. Biophys. Res. Commun. 2021, 538, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Malla, T.N.; Pandey, S.; Poudyal, I.; Feliz, D.; Noda, M.; Phillips, G.; Stojkovic, E.; Schmidt, M. Ebselen Reacts with SARS Coronavirus-2 Main Protease Crystals. bioRxiv 2020. [Google Scholar] [CrossRef]
- Ma, C.; Hu, Y.; Townsend, J.A.; Lagarias, P.I.; Marty, M.T.; Kolocouris, A.; Wang, J. Ebselen, disulfiram, carmofur, PX-12, tideglusib, and shikonin are nonspecific promiscuous SARS-CoV-2 main protease inhibitors. ACS Pharmacol. Transl. Sci. 2020, 3, 1265–1277. [Google Scholar] [CrossRef]
- Amporndanai, K.; Meng, X.; Shang, W.; Jin, Z.; Rogers, M.; Zhao, Y.; Rao, Z.; Liu, Z.-J.; Yang, H.; Zhang, L.; et al. Inhibition Mechanism of SARS-CoV-2 Main Protease by Ebselen and Its Derivatives. Nat. Commun. 2021, 12, 3061. [Google Scholar] [CrossRef]
- Nogara, P.A.; Omage, F.B.; Bolzan, G.R.; Delgado, C.P.; Aschner, M.; Orian, L.; Teixeira Rocha, J.B. In silico studies on the interaction between Mpro and PLpro from SARS-CoV-2 and Ebselen, its metabolites and derivatives. Mol. Inform. 2021, 40, 2100028. [Google Scholar] [CrossRef]
- Jin, Z.; Du, X.; Xu, Y.; Deng, Y.; Liu, M.; Zhao, Y.; Zhang, B.; Li, X.; Zhang, L.; Peng, C.; et al. Structure of Mpro from SARS-CoV-2 and discovery of Its inhibitors. Nature 2020, 582, 289–293. [Google Scholar] [CrossRef] [Green Version]
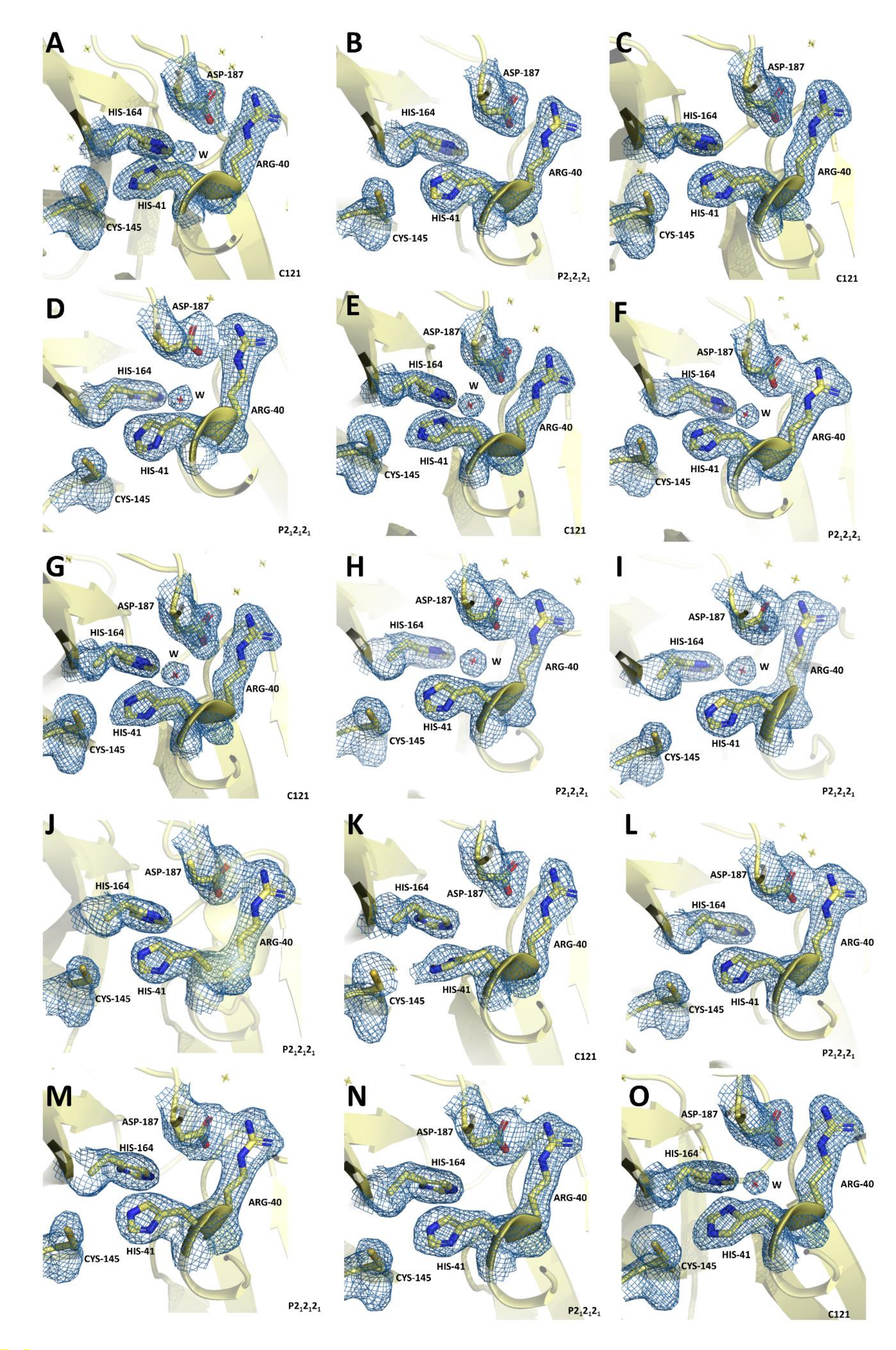
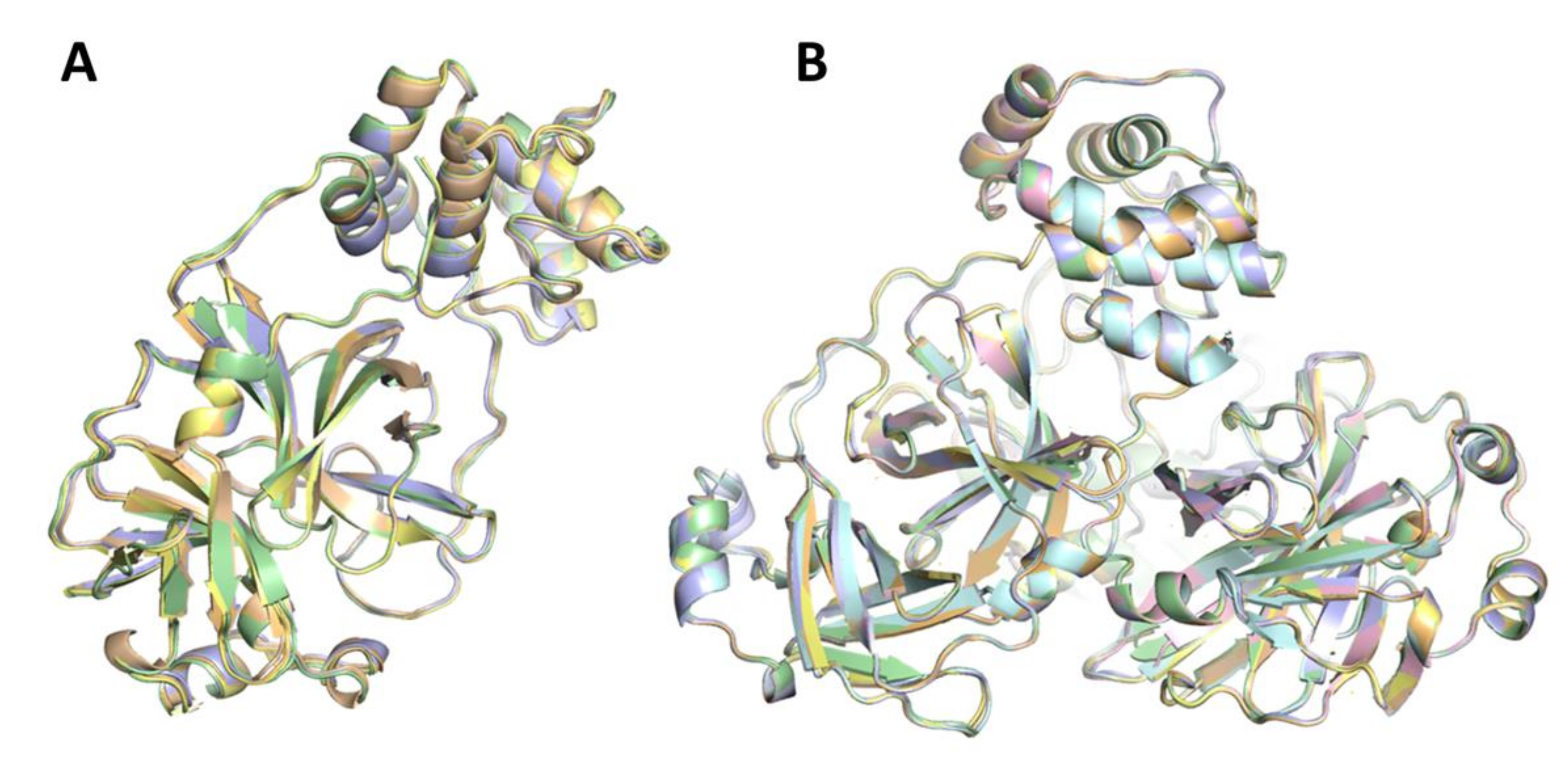
| Sample ID | PDB ID | Total Run Time | Effective Run Time | Runs | Run Number | Total Number Frames Collected | Total Number Frames with More Than 30 Bragg Reflections and a Signal to Noise Ratio > 4.5 | Hit Rate (%) | Total Number of Crystal Lattices Indexed to the Appropriate Unit Cell | Total Number of Crystal Lattices Merged into the Final SFX Dataset |
|---|---|---|---|---|---|---|---|---|---|---|
| Mpro3 | 7VJY | 5 h 20 min | 4 h 20 min | 20–80 | 60 | 1,465,292 | 279,428 | 19.07 | 73,605 | 68,314 |
| Mpro4 | 7VK3 | 3 h 28 min | 2 h 59 min | 82–115 | 33 | 1,121,297 | 182,774 | 16.3 | 33,384 | 67,918 |
| Mpro5 | 7CWB | 2 h 59 min | 2 h 52 min | 118–144 | 26 | 1,163,413 | 208,839 | 17.95 | 168,655 | 160,510 |
| Mpro6 | 7VK4 | 2 h 18 min | 2 h 18 min | 235–257 | 22 | 790,492 | 229,422 | 29.02 | 315,158 | 313,250 |
| Mpro7 | 7VK8 | 1 h 25 min | 45 min | 176–188 | 12 | 327,156 | 2078 | 0.63 | 1177 | 947 |
| Mpro8 | N/A | N/A | N/A | N/A | N/A | N/A | 13 | N/A | N/A | N/A |
| Mpro9 | 7VK1 | 3 h 04 min | 3 h 04 min | 145–174 | 29 | 1,148,580 | 51,112 | 4.45 | 33,384 | 30,732 |
| Mpro10 | 7VK0 | 2 h 11 min | 2 h 11 min | 264–291 | 27 | 858,742 | 932,35 | 10.86 | 116,315 | 115,577 |
| Mpro11 | 7VJZ | 2 h 07 min | 1 h 57 min | 189–213 | 24 | 669,237 | 132,447 | 19.79 | 70,272 | 65,579 |
| Mpro12 | 7VK5 | 1 h 30 min | 1 h 30 min | 292–310 | 18 | 609,036 | 80,383 | 13.19 | 120,147 | 119,430 |
| Mpro13 | 7VK2 | 2 h 47 min | 2 h 47 min | 216–234 | 18 | 873,323 | 180,625 | 20.68 | 88,551 | 81,563 |
| Mpro14 | 7VJW | 1 h 12 min | 1 h 12 min | 312–234 | 7 | 192,352 | 15,899 | 8.26 | 22,470 | 21,278 |
| Mpro15 | N/A | N/A | N/A | N/A | N/A | N/A | 0 | N/A | N/A | N/A |
| Mpro16 | 7CWC | 1 h 53 min | 1 h 53 min | 320–340 | 20 | 686,808 | 214,355 | 31.21 | 15,7976 | 156,512 |
| Mpro17 | 7VK7 | 1 h 28 min | 1 h 28 min | 341–357 | 16 | 439,830 | 86,490 | 19.66 | 174,120 | 173,796 |
| Mpro18 | 7VJX | 1 h 22 min | 1 h 22 min | 358–372 | 15 | 466,014 | 111,815 | 23.99 | 170,214 | 169,695 |
| Mpro19 | 7VK6 | 36 min | 36 min | 373–375 | 3 | 326,781 | 78,455 | 24.01 | 111,145 | 107,895 |
| Dataset | Mpro3 | Mpro4 | Mpro5 | Mpro6 | Mpro7 |
|---|---|---|---|---|---|
| PDB ID | 7VJY | 7VK3 | 7CWB | 7VK4 | 7VK8 |
| Data collection | |||||
| Beamline | LCLS (MFX) | LCLS (MFX) | LCLS (MFX) | LCLS (MFX) | LCLS (MFX) |
| Space group | C121 | P212121 | C121 | P212121 | C121 |
| Cell dimensions | |||||
| a, b, c (Å) | 114.0, 53.5, 45.0 | 69.3, 104.4, 105.7 | 114.0, 53.5, 45.0 | 68.9, 103.9, 105.2 | 115.3, 55.2, 45.7 |
| α, β, γ (°) | 90.0, 102.0, 90.0 | 90.0, 90.0, 90.0 | 90.0, 102.0, 90.0 | 90.0, 90.0, 90.0 | 90.0, 101.3, 90.0 |
| Resolution (Å) 1 | 48.24–1.73 (1.80–1.73) 1 | 46.8–2.14 (2.22–2.14) 1 | 55.0–1.9 (1.98–1.90) 1 | 46.9–2.14 (2.22–2.14) 1 | 31.92–2.34 (2.43–2.34) 1 |
| Rsplit | 1.09 (36.1) | 1.12 (54.4) | 0.63 (0.94) | 0.49 (45.2) | 5.94 (13.5) |
| CC1/2 | 0.998 (0.179) | 0.982 (0.089) | 0.996 (0.598) | 0.998 (0.331) | 0.477 (0.194) |
| I/σI | 0.25 | 0.22 | 0.57 | 0.23 | 0.97 |
| CC* | 0.997 (0.552) | 0.998 (0.405) | 0.999 (0.865) | 1.000 (0.705) | 0.804 (0.410) |
| Completeness (%) | 100.0 (100.0) | 100.0 (26.0) | 100.0 (100.0) | 100.0 (100.0) | 99.2 (98.0) |
| Redundancy | 387 | 1779 | 825 | 5066 | 9.2 |
| Refinement | |||||
| Resolution (Å) | 48.2–1.9 (1.95–1.90) 1 | 46.8–2.22 (2.29–2.22) 1 | 34.0–1.9 (1.95–1.90) 1 | 46.9–2.1 (2.15–2.10) 1 | 31.9–2.4 (2.51–2.40) 1 |
| No. reflections | 21,030 (1359) | 42,380 (3277) | 21,029 (1359) | 44,742 (3028) | 11,028 (1197) |
| Rwork/Rfree | 0.23/0.28 (0.49/0.52) | 0.24/0.28 (0.41/0.43) | 0.22/0.26 (0.43/0.49) | 0.21/0.23 (0.30/0.34) | 0.22/0.25 (0.34/0.41) |
| No. atoms | |||||
| Protein | 2427 | 4710 | 2447 | 4675 | 2435 |
| Ligand/Ion/Water | 60 | 114 | 113 | 7 | 67 |
| B-factors | |||||
| Protein | 38.59 | 53.56 | 42.86 | 64.78 | 31.50 |
| Ligand/Ion/Water | 38.02 | 52.13 | 51.0 | 32.72 | 24.57 |
| Coordinate errors | 0.39 | 0.39 | 0.34 | 0.29 | 0.49 |
| R.m.s deviations | |||||
| Bond lengths (Å) | 0.010 | 0.004 | 0.007 | 0.013 | 0.004 |
| Bond angles (°) | 1.183 | 0.873 | 0.869 | 1.567 | 0.746 |
| Ramachandran plot | |||||
| Favored (%) | 293 (96.4) | 573 (96.3) | 294 (96.7) | 573 (97.3) | 290 (96.4) |
| Allowed (%) | 6 (2.0) | 17 (2.9) | 9 (3.0) | 12 (2.0) | 9 (3.0) |
| Disallowed (%) | 5 (1.6) | 5 (0.8) | 1 (0.3) | 4 (0.7) | 2 (0.7) |
| Dataset | Mpro9 | Mpro10 | Mpro11 | Mpro12 | Mpro13 |
| PDB ID | 7VK1 | 7VK0 | 7VJZ | 7VK5 | 7VK2 |
| Data collection 1 | |||||
| Beamline | LCLS (MFX) | LCLS (MFX) | LCLS (MFX) | LCLS (MFX) | LCLS (MFX) |
| Space group | C121 | P212121 | C121 | P212121 | C121 |
| Cell dimensions | |||||
| a, b, c (Å) | 114.0, 53.5, 45.0 | 69.2, 104.3, 105.7 | 115.7, 55.2, 45.6 | 69.2, 104.3, 105.6 | 115.0, 54.8, 45.4 |
| α, β, γ (°) | 90.0, 102.0, 90.0 | 90.0, 90.0, 90.0 | 90.0, 101.2, 90.0 | 90.0, 90.0, 90.0 | 90.0, 101.4, 90.0 |
| Resolution (Å) 1 | 34.076–1.83 (1.90–1.83) 1 | 46.77–2.14 (2.22–2.14) 1 | 34.74–1.93 (2.11–1.93) 1 | 47.11–2.14 (2.22–2.14) 1 | 34.6–2.04 (2.11–2.04) 1 |
| Rsplit | 1.45 (7.1) | 0.83 (36.6) | 0.94 (12.6) | 0.75 (56.4) | 0.81 (18.1) |
| CC1/2 | 0.980 (0.255) | 0.995 (0.130) | 0.990 (0.387) | 0.996 (0.238) | 0.993 (0.474) |
| I/σI | 1.06 | 0.27 | 0.52 | 0.20 | 0.32 |
| CC* | 0.995 (0.949) | 0.999 (0.480) | 0.997 (0.747) | 0.999 (0.620) | 0.998 (0.802) |
| Completeness (%) | 87.6 (0.56) | 100.0 (100.0) | 100.0 (100.0) | 100.0 (99.0) | 100.0 (100.0) |
| Redundancy | 166 | 2378 | 400 | 310 | 497 |
| Refinement | |||||
| Resolution (Å) | 34.1–1.93 (1.99–1.93) 1 | 46.8–2.1 (2.15–2.10) 1 | 34.7–1.9 (1.95–1.90) 1 | 47.1–2.17 (2.23–2.17) 1 | 32.0–2.0 (2.05–2.00) 1 |
| No. reflections | 17,608 (949) | 45,326 (3045) | 22,355 (1441) | 41,092 (2971) | 18,846 (1314) |
| Rwork/Rfree | 0.21/0.26 (0.47/0.57) | 0.23/0.24 (0.49/0.46) | 0.21/0.25 (0.32/0.36) | 0.23/0.27 (0.44/0.45) | 0.23/0.27 (0.46/0.47) |
| No. atoms | |||||
| Protein | 2439 | 4662 | 2447 | 4710 | 2447 |
| Ligand/Ion/Water | 112 | 104 | 55 | 79 | 40 |
| B-factors | |||||
| Protein | 45.68 | 58.42 | 44.43 | 57.56 | 49.02 |
| Ligand/Ion/Water | 51.48 | 57.40 | 43.69 | 52.70 | 46.00 |
| Coordinate errors | 0.37 | 0.35 | 0.29 | 0.38 | 0.37 |
| R.m.s deviations | |||||
| Bond lengths (Å) | 0.005 | 0.011 | 0.005 | 0.004 | 0.003 |
| Bond angles (°) | 0.746 | 1.535 | 0.744 | 0.907 | 0.635 |
| Ramachandran plot | |||||
| Favored (%) | 293 (96.4) | 574 (62.5) | 299 (98.4) | 571 (96.5) | 296 (97.4) |
| Allowed (%) | 7 (2.3) | 15 (2.5) | 3 (1.0) | 15 (2.6) | 5 (1.6) |
| Disallowed (%) | 4 (1.3) | 6 (1.0) | 2 (0.7) | 6 (1.0) | 3 (1.0) |
| Dataset | Mpro14 | Mpro16 | Mpro17 | Mpro18 | Mpro19 |
| PDB ID | 7VJW | 7CWC | 7VK7 | 7VJX | 7VK6 |
| Data collection 1 | |||||
| Beamline | LCLS (MFX) | LCLS (MFX) | LCLS (MFX) | LCLS (MFX) | LCLS (MFX) |
| Space group | P212121 | P212121 | P212121 | P212121 | P212121 |
| Cell dimensions | |||||
| a, b, c (Å) | 69.1, 104.1, 105.5 | 69.2, 104.3, 105.6 | 69.1, 104.3, 105.5 | 69.2, 104.3, 105.7 | 104.3, 105.5, 69.1 |
| α, β, γ (°) | 90.0, 90.0, 90.0 | 90.0, 90.0, 90.0 | 90.0, 90.0, 90.0 | 90.0, 90.0, 90.0 | 90.0, 90.0, 90.0 |
| Resolution (Å) 1 | 34.55–2.34 (2.43–2.34) 1 | 75.0–2.1 (2.14–2.10) 1 | 41.93–2.24 (2.32–2.24) 1 | 46.77–2.14 (2.22–2.14) 1 | 16.82–2.14 (2.22–2.14) 1 |
| Rsplit | 1.54 (8.3) | 0.59 (3.33) | 0.84 (2.3) | 0.61 (−498.4) | 1.03 (165.7) |
| CC1/2 | 0.979 (0.217) | 0.997 (0.634) | 0.995 (0.793) | 0.997 (0.384) | 0.993 (0.121) |
| I/σI | 1.29 | 0.33 | 5.25 | −0.02 | 0.08 |
| CC* | 0.995 (0.649) | 0.999 (0.678) | 0.999 (0.941) | 0.999 (0.509) | 0.998 (0.465) |
| Completeness (%) | 100.0 (100.0) | 100.0 (100.0) | 100.0 (100.0) | 100.0 (96.0) | 100.0 (100.0) |
| Redundancy | 449 | 3105 (2025) | 4683 | 4516 | 2370 |
| Refinement | |||||
| Resolution (Å) | 34.6–2.2 (2.25–2.20) 1 | 42.0–2.1 (2.15–2.10) 1 | 41.9–2.4 (2.48–2.40) 1 | 46.8–2.2 (2.26–2.20) 1 | 16.8–2.25 (2.39–2.25) 1 |
| No. reflections | 39,287 (2628) | 45,238 (3044) | 30,465 (2599) | 39,396 (2558) | 36,768 (5902) |
| Rwork/Rfree | 0.22/0.28 (0.37/0.39) | 0.22/0.26 (0.37/0.44) | 0.21/0.24 (0.34/0.36) | 0.25/0.28 (0.37/0.37) | 0.23/0.27 (0.37/0.38) |
| No. atoms | |||||
| Protein | 4691 | 4710 | 4711 | 4710 | 4695 |
| Ligand/Ion/Water | 63 | 166 | 61 | 97 | 103 |
| B-factors | |||||
| Protein | 56.07 | 65.66 | 77.72 | 62.20 | 60.48 |
| Ligand/Ion/Water | 56.04 | 73.9 | 72.10 | 55.62 | 56.41 |
| Coordinate errors | 0.40 | 0.34 | 0.37 | 0.39 | 0.42 |
| R.m.s deviations | |||||
| Bond lengths (Å) | 0.008 | 0.003 | 0.002 | 0.001 | 0.002 |
| Bond angles (°) | 0.935 | 0.619 | 0.466 | 0.401 | 0.494 |
| Ramachandran plot | |||||
| Favored (%) | 568 (95.5) | 580 (97.5) | 571 (96.0) | 571 (96.0) | 576 (96.8) |
| Allowed (%) | 23 (3.9) | 14 (2.4) | 19 (3.2) | 22 (3.7) | 16 (2.7) |
| Disallowed (%) | 4 (0.7) | 1 (0.1) | 5 (0.8) | 2 (0.3) | 3 (0.5) |
| Mpro03 | Mpro07 | Mpro09 | Mpro11 | |
|---|---|---|---|---|
| Mpro07 | 0.543 | - | - | - |
| Mpro09 | 0.130 | 0.517 | - | - |
| Mpro11 | 0.484 | 0.266 | 0.456 | - |
| Mpro13 | 0.347 | 0.267 | 0.336 | 0.176 |
| Mpro04 | Mpro06 | Mpro10 | Mpro12 | Mpro14 | Mpro17 | Mpro18 | |
|---|---|---|---|---|---|---|---|
| Mpro06 | 0.184 | - | - | - | - | - | - |
| Mpro10 | 0.132 | 0.183 | - | - | - | - | - |
| Mpro12 | 0.134 | 0.165 | 0.142 | - | - | - | - |
| Mpro14 | 0.154 | 0.138 | 0.153 | 0.154 | - | - | - |
| Mpro17 | 0.201 | 0.192 | 0.182 | 0.184 | 0.175 | - | - |
| Mpro18 | 0.160 | 0.182 | 0.144 | 0.156 | 0.145 | 0.161 | - |
| Mpro19 | 0.1180 | 0.187 | 0.172 | 0.168 | 0.158 | 0.157 | 0.137 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guven, O.; Gul, M.; Ayan, E.; Johnson, J.A.; Cakilkaya, B.; Usta, G.; Ertem, F.B.; Tokay, N.; Yuksel, B.; Gocenler, O.; et al. Case Study of High-Throughput Drug Screening and Remote Data Collection for SARS-CoV-2 Main Protease by Using Serial Femtosecond X-ray Crystallography. Crystals 2021, 11, 1579. https://doi.org/10.3390/cryst11121579
Guven O, Gul M, Ayan E, Johnson JA, Cakilkaya B, Usta G, Ertem FB, Tokay N, Yuksel B, Gocenler O, et al. Case Study of High-Throughput Drug Screening and Remote Data Collection for SARS-CoV-2 Main Protease by Using Serial Femtosecond X-ray Crystallography. Crystals. 2021; 11(12):1579. https://doi.org/10.3390/cryst11121579
Chicago/Turabian StyleGuven, Omur, Mehmet Gul, Esra Ayan, J Austin Johnson, Baris Cakilkaya, Gozde Usta, Fatma Betul Ertem, Nurettin Tokay, Busra Yuksel, Oktay Gocenler, and et al. 2021. "Case Study of High-Throughput Drug Screening and Remote Data Collection for SARS-CoV-2 Main Protease by Using Serial Femtosecond X-ray Crystallography" Crystals 11, no. 12: 1579. https://doi.org/10.3390/cryst11121579
APA StyleGuven, O., Gul, M., Ayan, E., Johnson, J. A., Cakilkaya, B., Usta, G., Ertem, F. B., Tokay, N., Yuksel, B., Gocenler, O., Buyukdag, C., Botha, S., Ketawala, G., Su, Z., Hayes, B., Poitevin, F., Batyuk, A., Yoon, C. H., Kupitz, C., ... DeMirci, H. (2021). Case Study of High-Throughput Drug Screening and Remote Data Collection for SARS-CoV-2 Main Protease by Using Serial Femtosecond X-ray Crystallography. Crystals, 11(12), 1579. https://doi.org/10.3390/cryst11121579









