The Effects of Benzene on the Structure and Properties of Triethylamine Hydrochloride/Chloroaluminate
Abstract
:1. Introduction
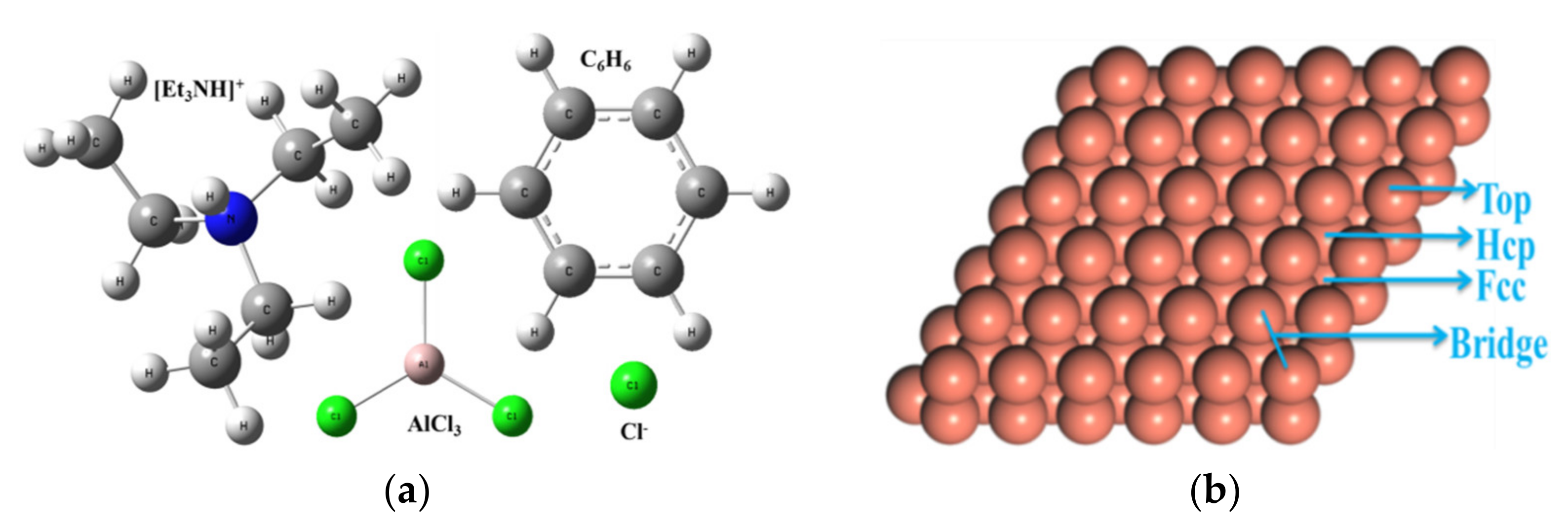
2. Materials and Methods
3. Results and Discussion
3.1. Effect of Additives on Microstructure and Physicochemical Properties of the System
3.1.1. Density
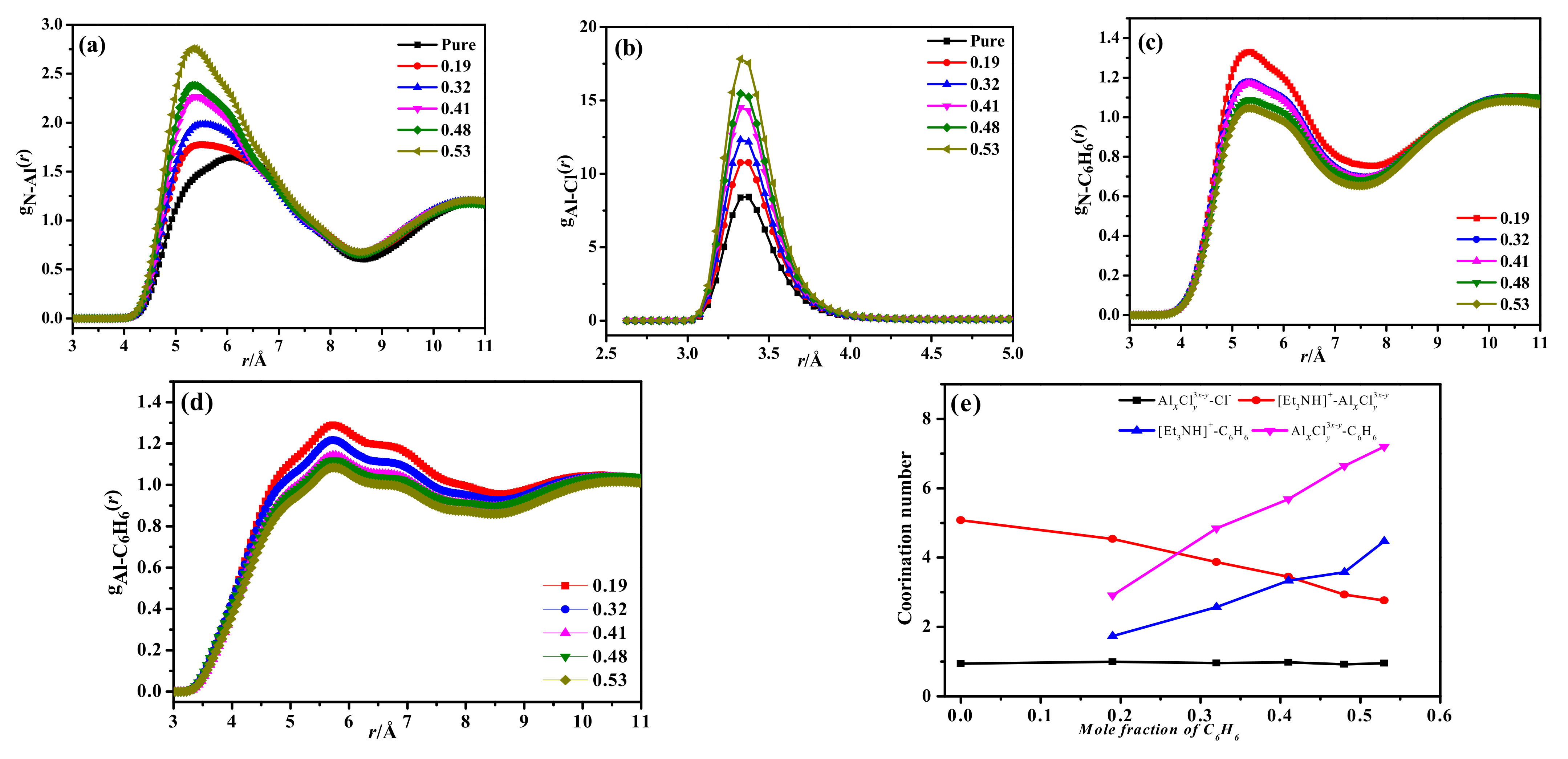
3.1.2. Radial Distribution Function and Coordination Numbers
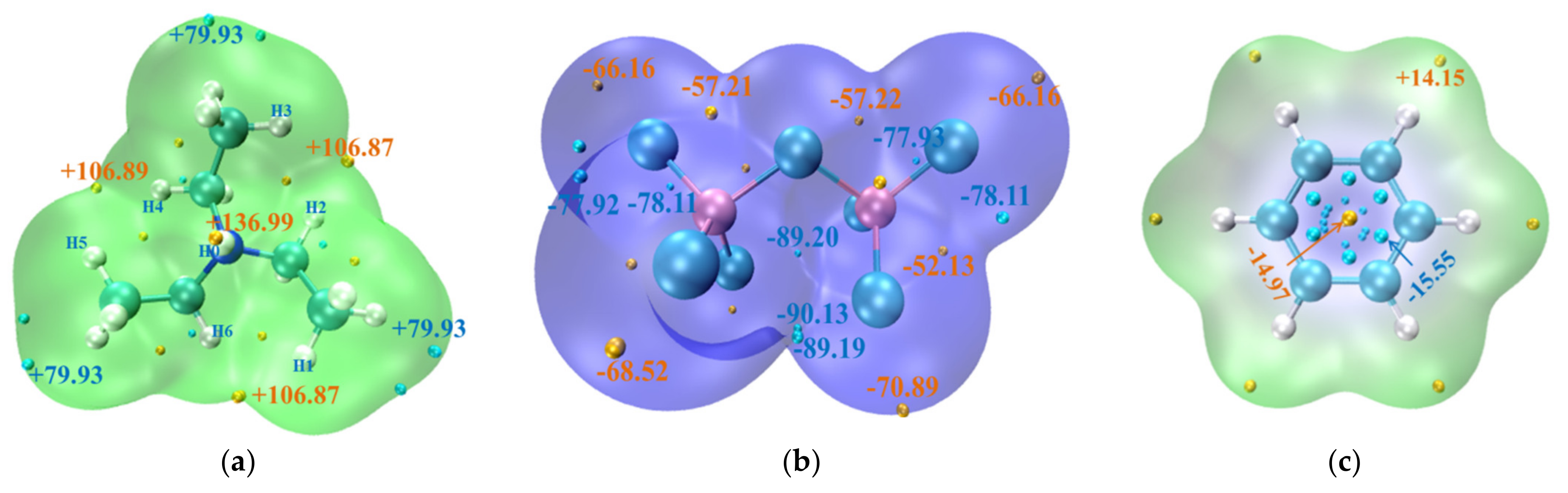
3.1.3. Spatial Distribution Function
3.1.4. Self-Diffusion Coefficient
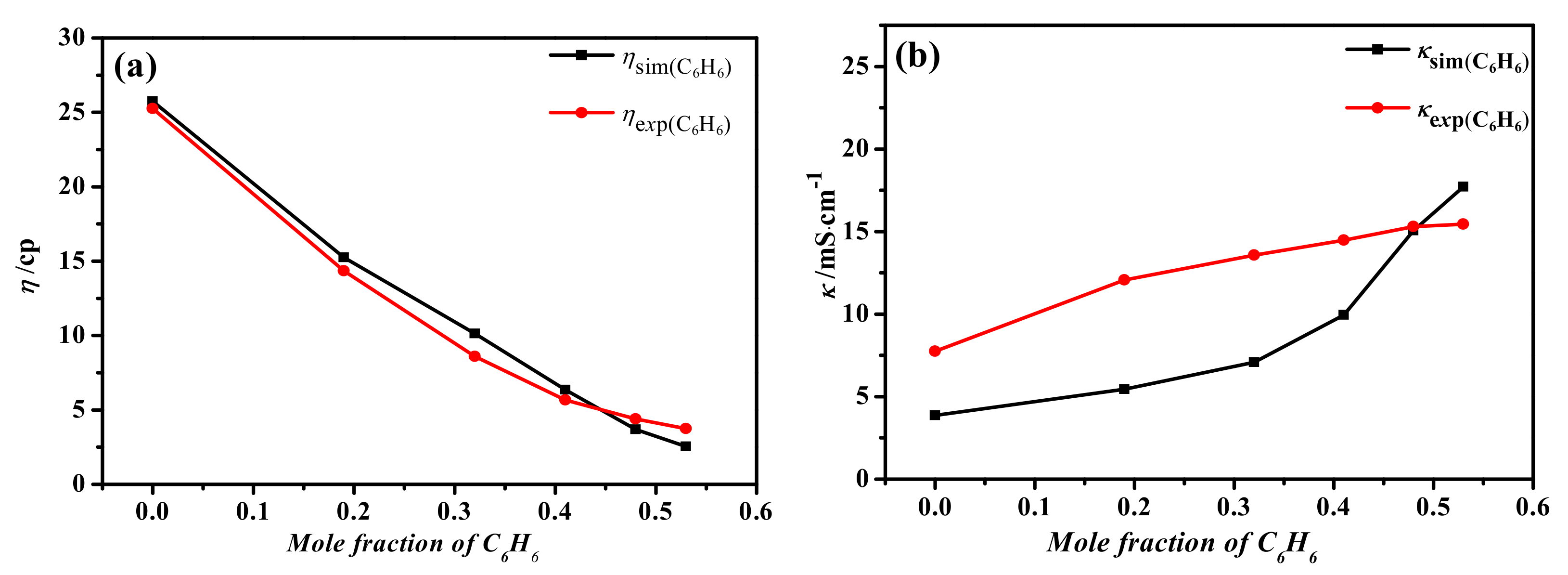
3.1.5. Viscosity and Conductivity
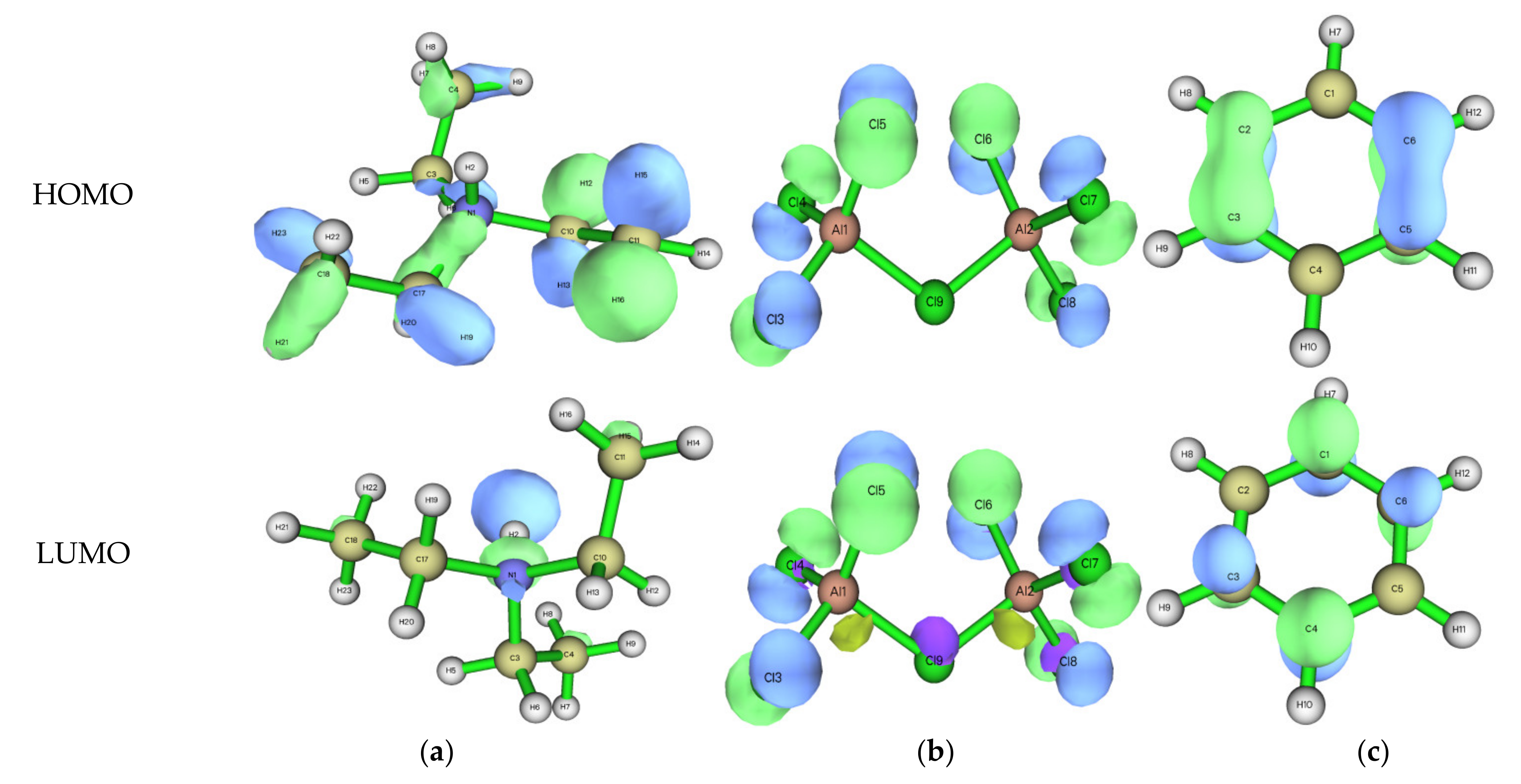
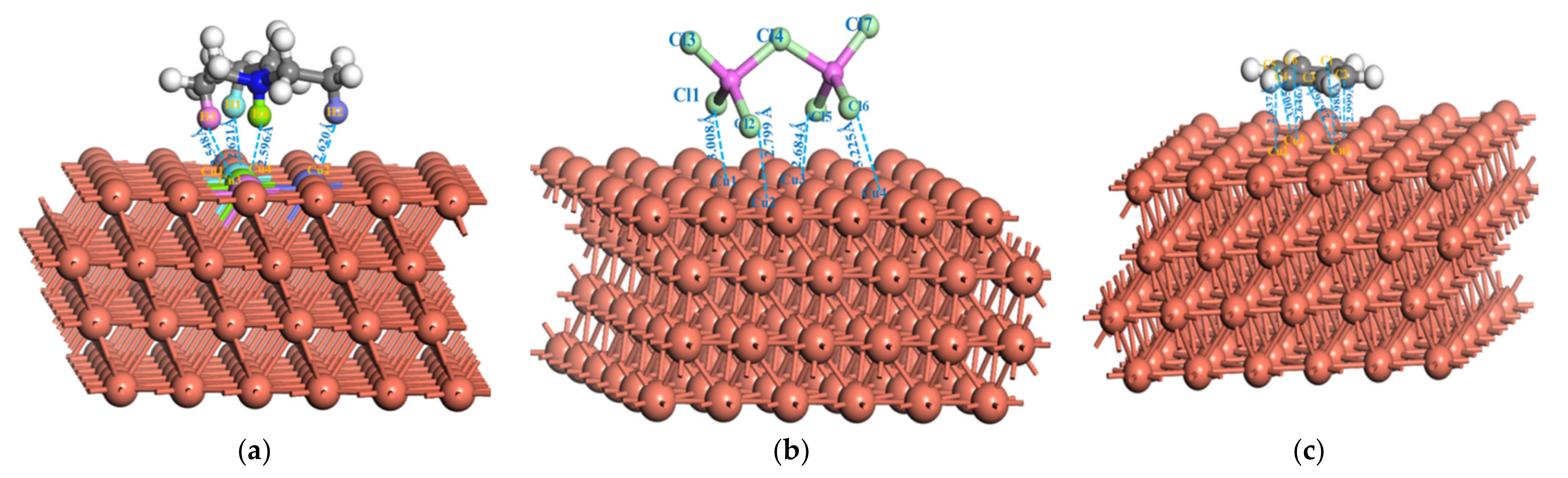
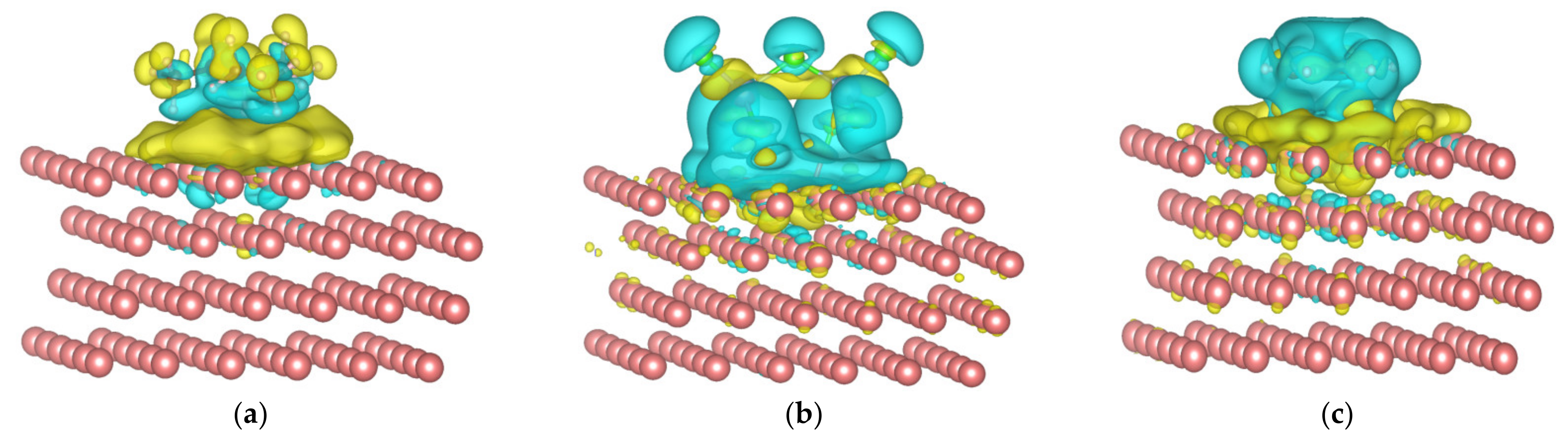
3.2. Structure, Frontier Orbitals and Adsorption of Cations, Anions and Additives on Surface
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhang, S.J.; Lu, X.M. Ionic Liquids, from Basic Research to Industrial Application; Science Press: Beijing, China, 2006. [Google Scholar]
- Deng, Y.Q. Ionic Liquids-Properties, Preparation and Application; China Petrochemical Press: Beijing, China, 2006. [Google Scholar]
- Abbott, A.P.; McKenzie, K.J. Application of ionic liquids to the electrodeposition of metals. Phys. Chem. Chem. Phys. 2006, 8, 4265–4279. [Google Scholar] [CrossRef] [PubMed]
- Tian, G.C.; Li, J.; Hua, Y.X. Application of ionic liquids in metallurgy of nonferrous metals. Chin. J. Process. Eng. 2009, 9, 200. [Google Scholar]
- Tian, G.C.; Li, J.; Hua, Y.X. Application of ionic liquids in hydrometallurgy of nonferrous metals. Trans. Nonferrous Met. Soc. 2010, 20, 513–520. [Google Scholar] [CrossRef]
- Zhang, M.; Kamavarum, V.; Reddy, R.G. New electrolytes for aluminum production: Ionic liquids. JOM 2003, 55, 54–57. [Google Scholar] [CrossRef]
- Liu, F.; Deng, Y.; Han, X. Electrodeposition of metals and alloys from ionic liquids. J. Alloy. Compd. 2016, 654, 163–170. [Google Scholar] [CrossRef]
- Zhong, X.W.; Xiong, T.; Lu, J.; Shi, Z.N. Advances of electro-deposition and aluminum refining of aluminum and aluminum alloyin ionic liquid electrolytes system. Nonferrous Met. Sci. Eng. 2014, 5, 44. [Google Scholar]
- Liu, Y.X.; Li, J. Modern Aluminum Electrolysis; Metallurgical Industry Press: Beijing, China, 2008. [Google Scholar]
- Carlin, R.T.; Crawford, W.; Bersch, M. Nucleation and morphology studies of aluminum deposited from an ambient-temperature chloroaluminate molten salt. J. Electrochem. Soc. 1992, 139, 2720–2727. [Google Scholar] [CrossRef]
- Suneesh, P.V.; Babu, T.S.; Ramachandran, T. Electrodeposition of aluminium and aluminium-copper alloys from a room temperature ionic liquid electrolyte containing aluminium chloride and triethylamine hydrochloride. Int. J. Miner. Metall. Mater. 2013, 20, 909–916. [Google Scholar] [CrossRef]
- Xu, H.; Bai, T.; Chen, H. Low-cost AlCl3/Et3NHCl electrolyte for high-performance aluminum-ion battery. Energy Storage Mater. 2019, 17, 38–45. [Google Scholar] [CrossRef]
- Tian, G.C. Ionic liquids as green electrolytes for Aluminum and Aluminum-alloy production. Mater. Res. Found. 2019, 54, 249. [Google Scholar]
- Ueda, M.; Hariyama, S.; Ohtsuka, T. Al electroplating on the AZ121 Mg alloy in an EMIC-AlCl3 ionic liquid containing ethylene glycol. J. Solid State Electrochem. 2012, 16, 3423. [Google Scholar] [CrossRef]
- Liu, L.; Lu, X.; Cai, Y.; Zheng, Y.; Zhang, S. Influence of additives on the speciation, morphology, and nanocrystallinity of aluminum electrodeposition. Aust. J. Chem. 2012, 65, 1523. [Google Scholar] [CrossRef]
- Zhang, Q.; Wang, Q.; Zhang, S.; Lu, X. Effect of nicotinamide on electrodeposition of Al from aluminum chloride (AlCl3)-1-butyl-3-methylimidazolium chloride ([BMIM]Cl) ionic liquids. J. Solid State Electrochem. 2014, 18, 257. [Google Scholar] [CrossRef]
- Wang, Q.; Chen, B.; Zhang, Q.; Lu, X.; Zhang, S. Aluminum deposition from lewis acidic 1-butyl-3-methylimidazolium chloroaluminate ionic liquid ([BMIM]Cl/AlCl3) modified with methyl nicotinate. ChemElectroChem 2015, 2, 1794. [Google Scholar] [CrossRef]
- Wang, Q.; Zhang, Q.; Chen, B.; Lu, X.; Zhang, S. Electrodeposition of bright Al coatings from 1-butyl-3-methylimidazolium chloroaluminate ionic liquids with specific additives. J. Electrochem. Soc. 2015, 162, D320. [Google Scholar] [CrossRef]
- Sheng, P.F.; Chen, B.; Shao, H.B.; Wang, J.M.; Zhang, J.Q.; Cao, C.N. Electrodeposition and corrosion behavior of nanocrystalline aluminum from a chloroaluminate ionic liquid. Mater. Corros. 2015, 66, 1338. [Google Scholar] [CrossRef]
- Lang, H.; Zhang, J.; Kang, Y.; Chen, S.; Zhang, S. Effects of lithium bis (oxalato) borate on electrochemical stability of [EMIM][Al2Cl7] ionic liquid for aluminum electrolysis. Ionics 2017, 23, 959. [Google Scholar] [CrossRef]
- Jiang, T.; Brym, M.J.C.; Dubé, G. Electrodeposition of aluminium from ionic liquids: Part II-studies on the electrodeposition of aluminum from aluminum chloride (AlCl3)-trimethylphenylammonium chloride (TMPAC) ionic liquids. Surf. Coat. Technol. 2006, 201, 10. [Google Scholar] [CrossRef]
- Jiang, T.; Brym, M.J.C.; Dubé, G. Electrodeposition of aluminium from ionic liquids: Part I—Electrodeposition and surface morphology of aluminium from aluminium chloride (AlCl3)-l-ethyl-3-methylimidazolium chloride ([EMIm]Cl) ionic liquids. Surface and Coatings Technology. Surf. Coat. Technol. 2006, 201, 1–9. [Google Scholar] [CrossRef]
- Hou, Y.Y.; Li, R.Q.; Liang, J.; Su, P.B.; Ju, P.F. Electropolishing of Al and Al alloys in AlCl3/trimethylamine hydrochloride ionic liquid. Surf. Coat. Technol. 2018, 335, 7. [Google Scholar] [CrossRef]
- Liu, K.R.; Liu, Q.; Han, Q.; Tu, G.F. Electrodeposition of Al on AZ31 magnesium alloy in TMPAC-AlCl3 ionic liquids. Trans. Nonferrous Met. Soc. 2011, 21, 2104–2110. [Google Scholar] [CrossRef]
- Su, C.J.; Hsieh, Y.T.; Chen, C.C.; Sun, I.W. Electrodeposition of aluminum wires from the lewis acidic AlCl3/trimethylamine hydrochloride ionic liquid without using a template. Electrochem. Commun. 2013, 34, 170–173. [Google Scholar] [CrossRef]
- Bakkar, A.; Neubert, V. Electrodeposition and corrosion characterisation of microand nano-crystalline aluminium from AlCl3/1-ethyl-3-methylimidazolium chloride ionic liquid. Electrochim. Acta 2013, 103, 211–216. [Google Scholar] [CrossRef]
- Gao, L.X.; Wang, L.N.; Qi, T. Electrodeposition of aluminium from AlCl3/Et3NHCl ionic liquids. Acta Phys.-Chim. Sin. 2008, 24, 939–944. [Google Scholar] [CrossRef]
- Liao, Q.; Pitner, W.R.; Stewart, G.; Hussey, C.L.; Stafford, G.R. Electrodeposition of aluminum from the aluminum chloride-1-methyl-3-ethylimidazolium chloride room temperature molten salt + benzene. J. Electrochem. Soc. 1997, 144, 936–943. [Google Scholar] [CrossRef]
- Uehara, K.; Yamazaki, K.; Gunji, T.; Kaneko, S.; Tanabe, T.; Ohsakab, T.; Matsumoto, F. Evaluation of key factors for preparing high brightness surfaces of aluminum films electrodeposited from AlCl3-1-ethyl-3-methylimidazolium chloride-organic additive baths. Electrochim. Acta 2016, 215, 556–565. [Google Scholar] [CrossRef]
- Xia, S.; Zhang, X.M.; Huang, K. Ionic liquid electrolytes for aluminum secondary battery: Influence of organic solvents. J. Electroanal. Chem. 2015, 757, 167–175. [Google Scholar] [CrossRef]
- Smith, B.; Akimov, A.V. Modeling nonadiabatic dynamics in condensed matter materials: Some recent advances and applications. J. Phys. Cond. Matter 2019, 32, 073001. [Google Scholar] [CrossRef]
- Tian, G.C.; Wang, D.; Wang, P.F. Molecular Dynamics Simulations of Structure and Properties of 1-Ethyl -3-methylimidazolium Tetrafluoroborate Ionic Liquid and Ethanol Mixtures. J. Kunming Univ. Sci. Technol. 2013, 37, 21–27. [Google Scholar]
- Li, Z.T.; Tian, G.C. Simulation study of the effect of benzene on the structure and properties of 1-ethyl-3-methylimidazolium chloroaluminate. Comput. Appl. Chem. 2015, 32, 1044. [Google Scholar]
- Izgorodina, E.I.; Seeger, Z.L.; Scarborough, D.L.A.; Tan, S.Y.S. Quantum chemical methods for the prediction of energetic, physical, and spectroscopic properties of ionic liquids. Chem. Rev. 2017, 117, 6696–6754. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Fu, F.; Peng, K. Understanding of structures, dynamics, and hydrogen bonds of imidazolium-based ionic liquid mixture from molecular dynamics simulation. Chem. Phys. 2019, 525, 110391. [Google Scholar] [CrossRef]
- Varela, L.M.; Méndez-Morales, T.; Carrete, J.; Gonzáleza, B.G.V.; Álvarez, B.D.; Gallego, L.J.; Cabeza, O.; Russinad, O. Solvation of molecular cosolvents and inorganic salts in ionic liquids: A review of molecular dynamics simulations. J. Mol. Liq. 2015, 210, 178–188. [Google Scholar] [CrossRef]
- Frisch, M.J.E.A.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.; et al. Gaussian 09 Revision A, 3rd ed.; Gaussian, Inc.: Wallingford, UK, 2009. [Google Scholar]
- Lyubartsev, A.P.; Laaksonen, A.M. DynaMix–a scalable portable parallel MD simulation package for arbitrary molecular mixtures. Comput. Phys. Commun. 2000, 128, 565–589. [Google Scholar] [CrossRef]
- Wang, J.; Wolf, R.M.; Caldwell, J.W. Development and testing of a general amber force field. J. Comp. Chem. 2004, 25, 1157–1174. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Lee, S.H. Molecular dynamics simulation studies of benzene, toluene, and p-xylene in a canonical ensemble. Bull. Korean Chem. Soc. 2002, 23, 441–446. [Google Scholar]
- De Andrade, J.; Böes, E.S.; Stassen, H. Computational study of room temperature molten salts composed by 1-alkyl-3-methylimidazolium cations force-field proposal and validation. J. Phys. Chem. B 2002, 106, 13344–13351. [Google Scholar] [CrossRef]
- Wang, J.; Cieplak, P.; Kollman, P.A. How well does a restrained electrostatic potential (RESP) model perform in calculating conformational energies of organic and biological molecules. J. Comput. Chem. 2000, 21, 1049–1074. [Google Scholar] [CrossRef]
- Kresse, G.; Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 1996, 6, 15–50. [Google Scholar] [CrossRef]
- Kresse, G.; Hafner, J. Ab initio molecular dynamics for liquid metals. Phys. Rev. B 1993, 47, 558–561. [Google Scholar] [CrossRef]
- Perdew, J.P.; Burke, K.; Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 1996, 77, 3865–3868. [Google Scholar] [CrossRef] [Green Version]
- Grimme, S.; Ehrlich, S.; Goerigk, L. Effect of the damping function in dispersion corrected density functional theory. J. Comput. Chem. 2011, 32, 1456–1503. [Google Scholar] [CrossRef] [PubMed]
- Padama, A.A.; Kishi, H.; Arevalo, R.L.; Moreno, J.L.V.; Kasai, H.; Taniguchi, M.; Uenishi, M.; Tanaka, H.; Nishihata, Y. NO dissociation on Cu(111) and Cu2O(111)surfaces: A density functional theory based study. J. Phys. Condens. Matter 2012, 24, 175005. [Google Scholar] [CrossRef]
- Lide, D.R. CRC Handbook of Chemistry and Physics, 84th ed.; CRC Press: Los Angeles, CA, USA, 2003–2004. [Google Scholar]
- Silbaugh, T.L.; Giorgi, J.B.; Xu, Y.; Tillekaratne, A.; Zaera, F.; Campbell, C.T. Adsorption energy of tert-Butyl on Pt (111) by dissociation of tert-Butyl Iodide: Calorimetry and DFT. J. Phys. Chem. C 2014, 118, 427–438. [Google Scholar] [CrossRef]
- Methfessel, M.; Paxton, A.T. High-precision sampling for Brillouin-zone integration in metals. Phys. Rev. B Condens. Matter 1989, 40, 3616–3621. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bader, R.F.W. Atoms in Molecules: A Quantum Theory; Oxford University Press: Oxford, UK, 1990. [Google Scholar]
- Momma, K.; Izumi, F. VESTA 3 for three-dimensional visualization of crystal, volumetric and morphology data. J. Appl. Crystallogr. 2011, 44, 1272–1276. [Google Scholar] [CrossRef]
- Lu, T.; Chen, F. Multiwfn: A multifunctional wavefunction analyzer. J. Comput. Chem. 2012, 33, 580–592. [Google Scholar] [CrossRef] [PubMed]
- Laaksonen, L. A graphics program for the analysis and display of molecular dynamics trajectories. J. Mol. Graph. 1992, 10, 33–34. [Google Scholar] [CrossRef]
- Abbott, A.P.; Qiu, F.; Abood, H.M.; Ali, M.R.; Ryder, K.S. Double layer, diluent and anode effects upon the electrodeposition of aluminum from chloroaluminate based ionic liquids. Phys. Chem. Chem. Phys. 2010, 12, 1862. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Cao, D.; Zhang, L. Molecular dynamics study on nanoparticle diffusion in polymer melts: A test of the Stokes-Einstein law. J. Phys. Chem. C 2008, 112, 6653–6661. [Google Scholar] [CrossRef]
- Morrow, T.I.; Maginn, E.J. Molecular dynamics study of the ionic liquid 1-n-butyl-3-methylimidazolium hexafluorophosphate. J. Phys. Chem. B 2002, 106, 12807–12813. [Google Scholar] [CrossRef]
- Banipal, P.K.; Chahal, A.K.; Singh, V.; Banipal, T.S. Rheological behaviour of some saccharides in aqueous potassium chloride solutions over temperature range (288.15 to 318.15) K. J. Chem. Thermodynam. 2010, 42, 1024–1035. [Google Scholar] [CrossRef]
- Holz, M.; Heil, S.R.; Sacco, A. Temperature-dependent self-diffusion coefficients of water and six selected molecular liquids for calibration in accurate 1H NMR PFG measurements. Phys. Chem. Chem. Phys. 2000, 2, 4740–4742. [Google Scholar] [CrossRef]
- Noda, A.; Hayamizu, K.; Watanabe, M. Pulsed-gradient spin-echo 1H and 19F NMR ionic diffusion coefficient, viscosity, and ionic conductivity of non-chloroaluminate room-temperature ionic liquids. J. Phys. Chem. B 2001, 105, 4603–4610. [Google Scholar] [CrossRef]
- Fang, Y.; Yoshii, K.; Jiang, X.; Sun, X.; Tsuda, T.; Mehio, N.; Dai, S. An AlCl3 based ionic liquid with a neutral substituted pyridine ligand for electrochemical deposition of aluminum. Electrochim. Acta 2015, 160, 82–88. [Google Scholar] [CrossRef]
- Pulletikurthi, G.; Bödecker, B.; Borodin, A.; Weidenfeller, B.; Endres, F. Electrodeposition of Al from a 1-butylpyrrolidine-AlCl3 ionic liquid. Prog. Nat. Sci. Mater. Int. 2015, 25, 603–611. [Google Scholar] [CrossRef] [Green Version]


| Mole Fraction of C6H6 | [Et3NH]Cl | AlCl3 | C6H6 | Equilibrium Sizes of Simulation Box (nm3) | ρsim/g·cm−3 | ρexp/g·cm−3 [30] |
|---|---|---|---|---|---|---|
| 0 | 128 | 256 | 0 | 4.047 × 4.047 × 4.047 | 1.297 | 1.312 |
| 0.19 | 128 | 256 | 88 | 4.301 × 4.301 × 4.301 | 1.223 | 1.248 |
| 0.32 | 128 | 256 | 177 | 4.506 × 4.506 × 4.506 | 1.190 | 1.201 |
| 0.41 | 128 | 256 | 265 | 4.713 × 4.713 × 4.713 | 1.149 | 1.169 |
| 0.48 | 128 | 256 | 353 | 4.916 × 4.916 × 4.916 | 1.109 | 1.128 |
| 0.53 | 128 | 256 | 441 | 5.082 × 5.082 × 5.082 | 1.091 | 1.095 |
| Adsorption | ECu(111) (eV) | Emol (eV) | ECu(111)−Mol (eV) | Eads (kJ.mol−1) | ∆q(e) |
|---|---|---|---|---|---|
| Cu(111)/[Et3NH]+ | −567.2566 | −117.3809 | −689.5731 | −476.2151 | 0.277 |
| Cu(111)/C6H6 | −567.2566 | −76.3256 | −644.6412 | −102.1741 | −0.045 |
| Cu(111)/Al2Cl7− | −567.2566 | −38.7584 | −604.0223 | 192.2199 | −0.172 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tian, G.; Du, H.; Yuan, Q. The Effects of Benzene on the Structure and Properties of Triethylamine Hydrochloride/Chloroaluminate. Crystals 2021, 11, 1532. https://doi.org/10.3390/cryst11121532
Tian G, Du H, Yuan Q. The Effects of Benzene on the Structure and Properties of Triethylamine Hydrochloride/Chloroaluminate. Crystals. 2021; 11(12):1532. https://doi.org/10.3390/cryst11121532
Chicago/Turabian StyleTian, Guocai, Huanhuan Du, and Qingxiang Yuan. 2021. "The Effects of Benzene on the Structure and Properties of Triethylamine Hydrochloride/Chloroaluminate" Crystals 11, no. 12: 1532. https://doi.org/10.3390/cryst11121532
APA StyleTian, G., Du, H., & Yuan, Q. (2021). The Effects of Benzene on the Structure and Properties of Triethylamine Hydrochloride/Chloroaluminate. Crystals, 11(12), 1532. https://doi.org/10.3390/cryst11121532






