Abstract
Since cancer is a serious threat to public health worldwide, the development of novel methods and materials for treating cancer rapidly and thoroughly is of great significance. This review summarizes the mechanism and application of photocatalytic materials used to kill cancer cells. The photosensitivity and toxicological properties of several common photcatalysts used in anti-cancer treatment are discussed in detail. The ideal photocatalyst must possess the following characteristics: a highly stable production of active oxygen species and high selectivity to cancer cells without causing any damage to healthy tissues. This work concluded the existing photocatalytic materials used to treat cancer, as well as the current challenges in the application of cancer therapy. We aim to provide a basis for the development of new photocatalytic anti-cancer materials with high stability and selectivity while maintaining high photodynamic reaction performance.
1. Introduction
Nowadays, cancer has become a serious threat to public health worldwide [1]. In 2018, there were 18.1 million cancer patients and an estimated 9.6 million individuals died from cancer [2,3,4]. With the continuous emergence of targeted therapy and immunotherapy, modern oncology has made breakthrough progress. Although the relative survival rate of cancer patients has been improved and the mortality rate of cancer has decreased, most clinical cancer therapies, often accompanied by severe and sometimes irreversible side effects, are still difficult to completely cure cancer. Therefore, there is still an urgent need to explore new methods for rapid, thorough, targeted, and safe cancer treatment.
Phototherapy methods with intrinsic non-invasiveness, minimal side effects and high spatial selectivity upon a localized irradiation at the tumor sites can precisely target tumors and minimize damage to normal cells [5]. Owing to these advantages, Phototherapy methods have been widely used in preclinical and clinical treatment of cancer. Photodynamic therapy (PDT) and photothermal therapy (PTT) are two typical phototherapy methods [6]. PTT is a treatment that converts light energy into heat energy to kill cancer cells using nanomaterials with high light-to-heat conversion efficiency under the irradiation of an external light source [7]. As a minimally invasive, precise and controllable treatment approach, PTT has made great achievements in recent years. However, photothermal agents (PTA), the cornerstone and key component of photothermal therapy, still have deficiencies and room for improvement in terms of metabolism, stability, photothermal conversion efficiency, and synthesis strategies. PDT is a well-established and clinically approved treatment approach. In the PDT process, the photosensitizer molecules absorb energy from an appropriate wavelength and then transfer the energy to the surrounding oxygen molecules to generate reactive oxygen species (ROS), which have significant cytotoxicity, leading to cancer cell killing and tumor ablation [8,9]. Since ROS are chemically reactive free radicals or non-radical molecules derived from oxygen molecules, PDT is an oxygen-dependent process, which requires the participation of oxidative intermediates in the tumor hypoxic microenvironment [10]. PDT therapy is usually an inefficient procedure and requires repeated treatments.
Due to the enhanced permeability and retention effect, nanomaterials tend to passively accumulate in tumors and often act as nanocarriers of chemotherapeutic drugs [11]. Semiconductor TiO2 is a common photocatalyst material, which is generally used to degrade organic substrates [12] and deactivate microorganisms [13,14,15] and viruses [16]. It was reported that TiO2 nanoparticles of various sizes and morphologies exhibited cytotoxicity toward tumors [17,18,19]. The catalytic action of the photocatalysts driven by photon energy can oxidize or reduce substrate molecules. Photocatalysts have been widely used in life-related antibacterial and antiviral fields [20,21]. Recently, researchers have conducted vast and in-depth explorations on the potential value of photocatalysts in the field of cancer treatment. Zhu et al. [22] prepared a self-assembled supramolecular photocatalyst of tetracarboxyphenyl porphyrin (SA-TCPP) and proved that it can be excited by the light with a wavelength of 420–750 nm. Porphyrin-based molecular drugs are widely used in PDT due to their excellent biocompatibility and release of singlet oxygen, some of which have already achieved clinical applications. Li et al. [23] thought that photocatalytic materials can generate ROS for tumor DNA damage when they were irradiated by targeted wavelength light, achieving high efficiency at low radiation doses with limited damage to normal tissues. In this work, we discussed the photosensitivity and toxicological properties of several common photocatalysts used in anti-cancer treatments in detail. The current limitations and challenges of photocatalytic physiotherapy were summarized and feasible solutions were proposed. We aim to provide a basis for the development of novel photocatalysts for cancer therapy with higher stability, selectivity, and high photodynamic reaction performance.
2. Photocatalysts for Cancer Therapy
Anti-cancer activity of photocatalysts belongs to photodynamic therapy (PDT), which is a new technique for cancer treatment. Typically, the PDT process involves three main components: light, oxygen, and light-responsive materials (photosensitizers). Hybrid semiconductor photocatalysts absorb energy from light and then transfer it to molecular oxygen to produce cytotoxic reactive oxygen species (ROS) [24]. Compared with conventional PDT, the advantages of nano photocatalysts are the results of the synergistic combination of inorganic materials with unique physical properties and the targeting performance of biomolecules, as well as the multi-functional drugs molecules loaded in an ideal therapeutic system [25]. In addition, nano photocatalysts can overcome biological barriers, including the blood-brain barrier.
Mechanism of PDT
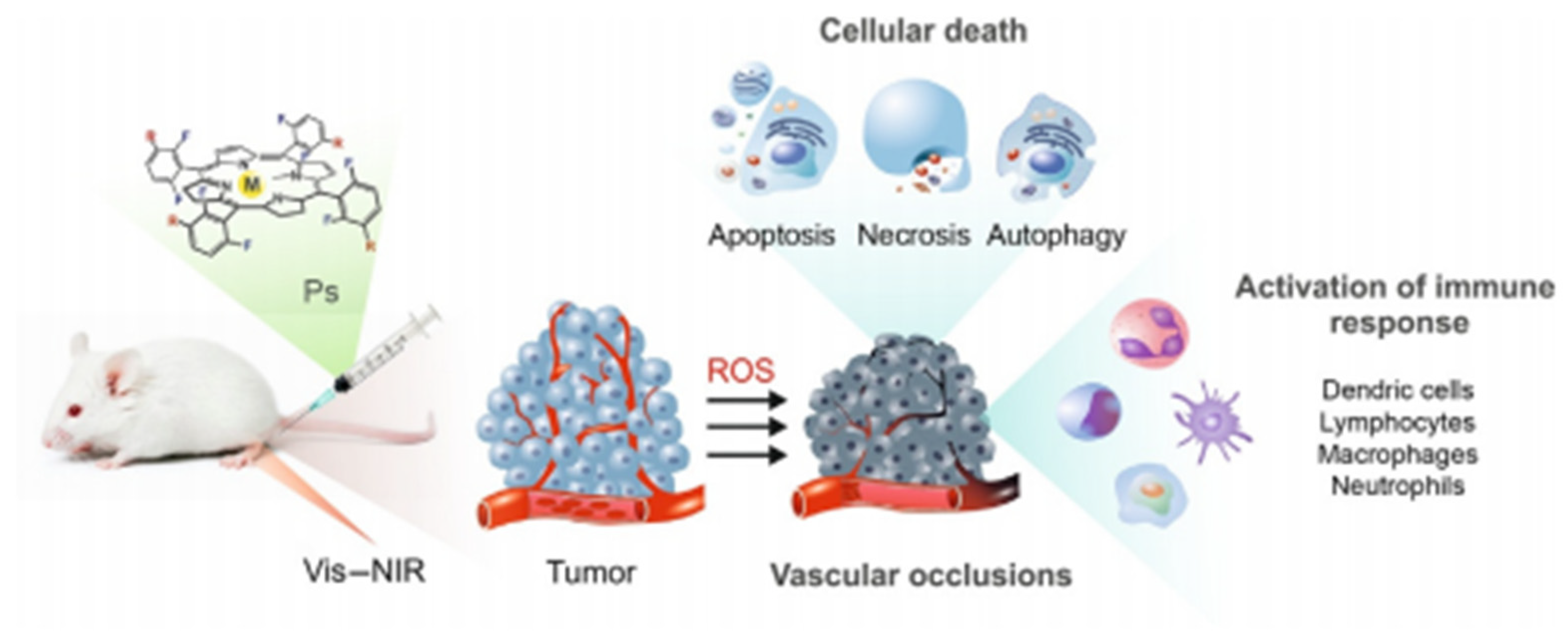
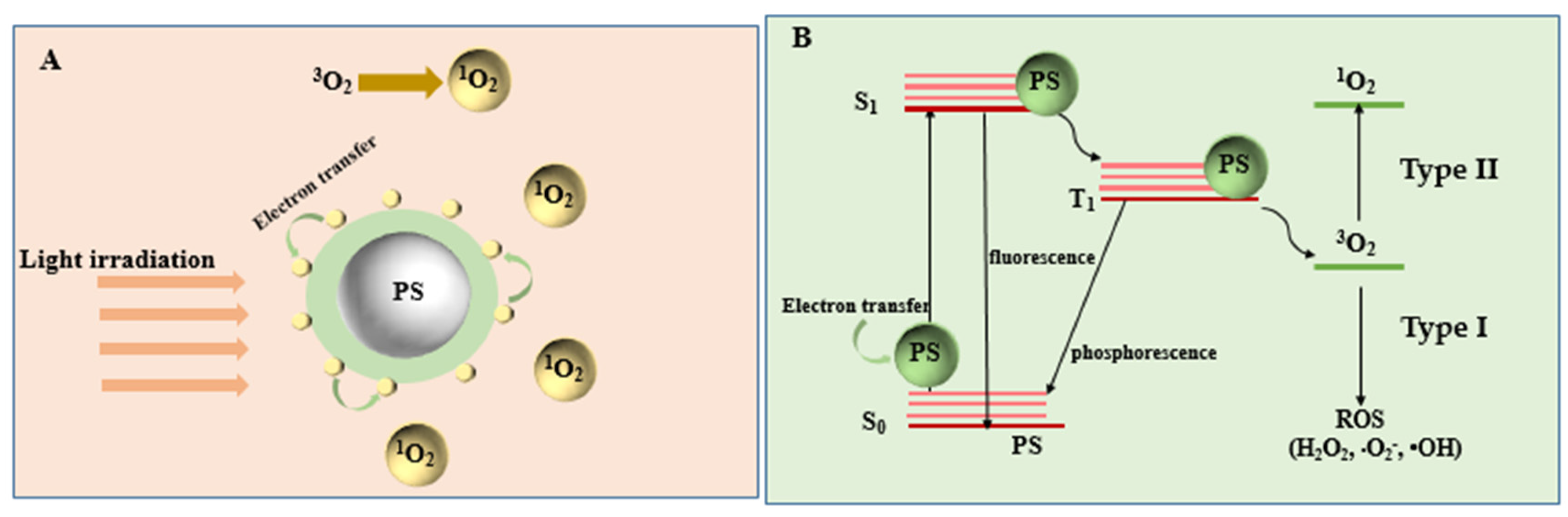
PDT is a new method for the treatment of tumor diseases, which uses photosensitive drugs and laser activation [26,27]. The general process of PDT is to irradiate the tumor site with a specific wavelength and selectively activate the photosensitive drugs accumulated in the tumor tissue and trigger a photochemical reaction to destroy the tumor cells (Figure 1). Photosensitizers can trigger a photodynamic reaction to produce a large amount of ROS in the presence of oxygen. ROS possess remarkable cytotoxic properties, which can cause changes in the morphology and functions of cells or biomolecules, leading to cancer cell killing and tumor ablation [28]. PDT treatment depends on three vital components: photosensitizers, light, and tissue oxygen [29]. According to the type of ROS, PDT can be divided into type I and type II [30]. Type I: the photosensitizer undergoes an electron transfer reaction in the excited state, obtaining an electron to form a radical anion, which can react with oxygen to form a superoxide radical anion (•O2−). Finally, the disproportionation or single-electron reduction of •O2− will generate hydroxyl radicals that cause extensive oxidative damage. Type II: photosensitizers in the excited singlet state can form a relatively stable excited triplet state through the process of intersystem crossing, transferring energy to molecular oxygen, and producing single oxygen (1O2). The detailed mechanism of PDT has been concluded in Figure 2. Singlet oxygen interacts with adjacent cancer tissues to induce apoptosis, necrosis, and autophagy-related cell death. Singlet oxygen participates in many biological processes, such as the membrane destruction process, metabolic hydroxylation, oxidative DNA damage, carcinogenesis, and so on. All of the above processes have genotoxic, virus-killing and cytotoxic effects.
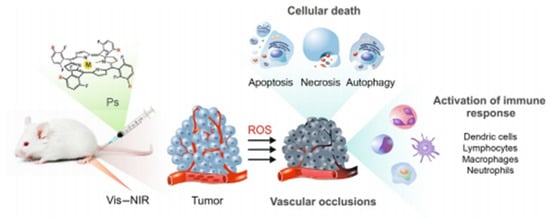
Figure 1.
The processes of PDT to destroy the tumor cells (PS means photosensitizer). (Reproduced from [30] with permission from [Elsevier], copyright [2017]) [31].
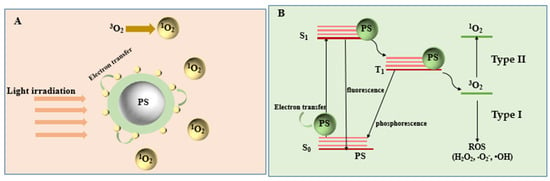
Figure 2.
Diagram of the PDT mechanism at the moment when energy is transferred from photosensitizers (PS) to activate the photosensitizers. (A) The generation of 1O2 through the electron transfer process. (B) The process of energy transfer and the PDT mechanism.
3. Common Photocatalysts for Cancer Therapy
The photodynamic therapy, by using photocatalysts as photosensitizers to treat tumors, has theoretical value and practical significance. Recently, nano-photocatalysts with anti-tumor activity have attracted increasing attention from medical researchers. Nowadays, medical researchers have some experience in the theoretical basis of nano-photocatalysts with anti-tumor activity. Nano photocatalysts are likely to become a new anti-tumor drug in the 21st century. Common photocatalysts for cancer therapy have been concluded in Table 1.

Table 1.
Common photocatalysts for cancer therapy.
3.1. Supermolecular Photocatalyst
It is reported that organic supramolecular photocatalysts have huge potential in rapid, complete, targeted, and safe treatment for tumors, which is of great significance to human health [55,56,57]. Wang’s team [32] found that supermolecular photocatalyst of J-aggregated perylenetetracarboxylic diimide has high growth inhibition efficiency for Hela cells, low cytotoxicity, and high photostability, and thereby can be used as a photocatalytic anti-breast cancer treatment. After injecting MDA-MB-231 into the mammary gland cells of mice to generate xenograft tumors for 12 h, the tumor site was irradiated with light of 600 nm wavelength (220 mW/cm²) for 10 min. The results showed that J-aggregate perylenetetracarboxylic diimide had a high 1O2 quantum yield under red light irradiation, verified it had potential application value in photocatalytic anti-cancer therapy. Based on the combination of biological light-absorbing molecules and self-assembling peptides, Zou et al. [33] constructed highly stable supramolecular assembled peptide−porphyrin photothermal nanodots. The assembled peptide-porphyrin photothermal nanodots showed high antitumor therapy properties, negligible toxicity, and effective MCF-7 tumor ablation. Zhang has established a new method for the treatment of solid tumors using a self-assembled tetra-carboxyphenyl porphyrin supermolecular photocatalyst by irradiating with light of 600–700 nm wavelength [34]. Solid tumors can be eliminated by self-assembled tetra-carboxyphenyl porphyrin supermolecular photocatalyst within 10 min. This kind of supramolecular photocatalytic therapy has a significant therapeutic effect on cancer cells of Hela cells, MCF-7, HepG-2, and so on.
Photocatalytic cancer treatment uses conjugated organic supramolecular photocatalysts to generate a strong oxidative photo-generated hole under strong red light irradiation (>650 nm), which has overcome the dependence of traditional phototherapy on oxygen in hypoxic tumor tissues. Photocatalytic cancer treatment can completely oxidize and kill tumor cells or solid tumor tissues without the risk of metastasis and recurrence, and is an efficient treatment of cancer cells. In addition, organic supramolecular photocatalytic anti-cancer materials have an excellent biocompatibility, with no side effects to normal cells or tissues. Supramolecular is a safe cancer therapy since it can be degraded by biological metabolism.
3.2. Metal and Metal Oxide Photocatalysts for Cancer Therapy
Metal and its oxide nanoparticles showed an excellent performance in pharmacological activity, especially in anti-tumor therapy.
3.2.1. IrIII Photocatalyst for Cancer Therapy
Ir3 is a water-soluble coumarin-functionalized IrIII complex with excellent photophysical and anti-cancer properties [58]. Ir3 exhibits excellent photocatalytic oxidation of cellular coenzymes, and reduced nicotinamide adenine dinucleotide phosphate (NADPH) and amino acids through a single electron transfer mechanism. Light-induced intracellular redox imbalance and changes in the mitochondrial membrane potential lead to necrosis and apoptosis of cancer cells [48]. Sadler and colleagues reported that nicotinamide adenine dinucleotide (NADH) photooxidized can be reduced to NAD+ in the cell under the catalysis of acyclic metallized IrIII complex, which can cause cancer cells even under hypoxic conditions [59]. However, this concept of photocatalytic cancer treatment is limited to in vitro. Compared with ordinary photosensitizers, stable iridium photocatalysts have an abnormally high excited state reduction potential [58]. The iridium photocatalyst, when irradiated by specific wavelength light, has high NADH oxidation performance in biological media [60]. The treatment of hypoxic tumors is the most difficult problem for photodynamic therapy. Huang prepared a highly oxidizing Ir(III) photocatalyst [Ir(ttpy)(pq)Cl]PF6 (“ttpy” means 4′-(p-tolyl)-2,2′:6′, 2″-Terpyridine and “pq” means 3-phenylisoquinoline), which has a good phototoxic performance on normoxic or hypoxic cancer cells [59]. This Ir(III) photocatalyst can induce the depletion of NADH, the imbalance of intracellular redox and the death of immunogenic apoptotic cancer cells under light irradiation. This photocatalytic redox imbalance strategy provides a new method for effective hypoxic cancer phototherapy. Therefore, the iridium photocatalyst can target the mitochondria in cancer cells. It exhibited good photocytotoxicity regardless of whether it was under aerobic or hypoxic conditions, but had low toxicity to normal cells.
3.2.2. Cerium Oxide for Cancer Therapy
Recent research reported that photocatalytic nano cerium oxide can scavenge hydroxyl radicals [61], and showed intrinsic oxidase activity [62]. It was reported that cerium oxide had cytotoxic and anti-invasive properties to a variety of cancer cells [42,63,64]. Cerium oxide has been widely used in anti-cancer treatment. The redox-active polymer-coated cerium oxide nanoparticles can affect the formation of myofibroblasts, exhibiting cytotoxicity, and invading tumor cells. In addition, it has non-toxic effects on normal stromal cells. Lin45 reported that the oxygen free radicals produced by cerium oxide nanoparticles can generate significant oxidative stress to kill lung cancer cells. In Melissa’s work [40], photocatalytic nano cerium oxide can induce ROS production and cell death, selectively adsorbing to human pancreatic tumor cells, protecting normal tissues when exposed to an acidic environment. Pešic [65] discussed the toxicity of nano cerium oxide with a size of 4nm to eight different types of cancer cells. The results showed that nano cerium oxide had low cytotoxicity to normal cells, as well as the keratinocyte HaCaT, lung fetal fibroblast cell MRC-5, while it had a good inhibitory effect on melanoma 518A2 and colorectal adenocarcinoma HT-29 by increasing the production of ROS. Cerium oxide may provide a new strategy for cancer treatment by enhancing anti-tumor activity and reducing the destructive side effects caused by classic chemotherapy drugs.
3.2.3. Titanium Dioxide for Cancer Therapy
The nano TiO2 photocatalyst has a significant killing effect on tumor cells, which can generate ROS and induce cell apoptosis by increasing the permeability of the cell membrane through the oxidation of lipids in the cell membrane, leading to an influx of calcium ions [66]. Hidaka et al. proposed that nano-TiO2 can catalyze and oxidize DNA or RNA molecules under light irradiation [67]; Dunford et al. found that TiO2 can catalyze DNA damage in vitro under ultraviolet light irradiation, and he believed that the hydroxyl radicals produced by TiO2 were the main factor leading to DNA breakage [68]; Kubota et al. confirmed that nano-TiO2 under light irradiation can cause cell membrane damage and kill cells [47]. The mechanism of nano TiO2 photocatalytic oxidation to kill cancer cells is not yet clear, and it may be the result of the synergistic effect of multiple mechanisms.
The anti-cancer research on nano TiO2 has attracted more and more attention, and satisfactory results have been achieved. Huang et al. has found that ultrafine TiO2 had a significant killing effect on U937 leukemia cells after photocatalytic oxidation [69]. The agarose gel electrophoresis of DNA showed that light-excited TiO2 could damage the DNA in cells and cause cell death, which have provided a new idea for killing cancer. Li et al. evaluated the expression levels of endoplasmic reticulum stress sensors (PERK and ATF6) and Bax under light induction by the Western blotting method, and studied the killing effect of TiO2 on HepG2 human liver cancer cells [70]. Under light induction, nano TiO2 not only has an anti-tumor effect, but also shows a high tumor-inhibition rate at an appropriate concentration. The anti-tumor process can well conform to the first-order kinetic equation and related laws. Botelho et al. conducted a blank experiment of ultraviolet light and TiO2 on gastric cells in vitro, along with a control experiment of nano-TiO2 photocatalytic inhibition of cancer cells [71]. The results showed that nano TiO2 had a significant killing effect on gastric cancer cells. The anti-cancer mechanism of nano TiO2 is to use a photosensitizer to cause a series of chemical reactions to kill cancer cells. TiO2 is stable and has a strong killing performance on cancer cells. The cancer cells can be killed within 30–50 min by using nano TiO2 photocatalyst.
3.2.4. Cuprous Oxide for Cancer Therapy
Cuprous oxide nanoparticles can selectively induce tumor cell apoptosis both in vitro and in vivo. Wang [72] used cuprous oxide particles to treat B16-F10 mouse subcutaneous melanoma cells and metastatic lung tumors by injected intratumorally and systemically, respectively. The results showed that cuprous oxide nanoparticles can significantly reduce the growth of melanoma, inhibiting the metastasis of B16-F10 cells, and improving the survival rate of tumor-bearing mice. Importantly, the results showed that cuprous oxide nanoparticles can be quickly removed from the organs, and these particles showed weak systemic toxicity. In Yang’s research [73], he believed that cuprous oxide was a promising nanomaterial to treat patients with advanced renal cell carcinoma by regulating the copper chaperones ATOX1 and CCS in RCC cells to disrupt copper transport, promoting the accumulation of intracellular calcium and inducing endoplasmic reticulum stress in vitro and in vivo, thereby destroying cancer cells.
3.3. Z-Scheme Structure Photocatalyst
Drug-free therapy generally means that a therapeutic methodology with inexhaustible therapeutic capability is able to maximize the benefits of treatment without any drug usage. To achieve the goal of drug-free therapy, researchers have conducted a lot of research in past years. Tang et al. developed a calcification method to kill cancer cells by injecting folic acid and Ca2+ into tumors; however, these two therapeutic agents were easily consumed during cancer treatment, which limited their application [74]. Shi et al. proposed a concept that nanocatalytic drugs can be used to improve the curative effect of cancer treatment and he believed that nanocatalysis can realize drug-free treatment in principle, which is beneficial to avoid the toxic side effects of drugs [75,76,77,78]. However, some toxic or easily depleted drugs as auxiliary drugs have been loaded in current catalytic nanomedicines. Thus, the development of nanocatalytic nanomedicine for drug-free treatment is still a big challenge. Z-scheme structure photocatalyst provides a new feasible solution for drug-free therapy. Recently, He et al. developed a Z-scheme structured SnS1.68-WO2.41 nano-photocatalyst, which realized in-situ NIR photocatalytic hydrogen production in tumors to inhibit cancer cell growth, and at the same time destroyed tumors by using the generated holes to in-situ oxidize over-expressed GSH in tumors, destroying the tumor microenvironment through the “hydrogen-cavity combined therapy”, and achieving a highly effective anti-cancer effect [52]. Pan prepared a Z-scheme heterojunction functionalized pyrite nanosheets with FeS2 core and Fe2O3 shell and the results found that this catalyst can destroy the tumor microenvironment by consuming glutathione and producing O2, meanwhile it can produce •OH through Fenton reaction [79]. Sang prepared a Z-scheme Ni3S2/Cu1.8S@HA for hypoxic tumor therapy. This catalyst relieved the hypoxia in the tumor microenvironment through the release of photocatalytic O2 in the cell and realized the enhancement of PDT. In addition, the nanocomposite material also exhibited new biodegradability, which can be metabolized and eliminated by feces and urine within 2 weeks [53].
3.4. Piezocatalysis for Cancer Therapy
Due to the advantages of non-invasiveness, low energy attenuation, and strong tissue penetration, ultrasound therapy is playing an increasingly important role in the diagnosis and treatment of clinical diseases. Recently, researchers have proposed a method to treat tumors that adopted a piezoelectric catalyst to catalyze the production of reactive oxygen species (ROS) using ultrasound as a microscopic pressure source. Under ultrasonic vibration, electrons and holes are separated by piezoelectricity, forming a strong built-in electric field, catalyzing the production of toxic hydroxyl (•OH) and superoxide radicals (•O2) and other reactive oxygen species, eradicating tumors in situ. Compared with typical sonoluminescence activated sonodynamic therapy, this approach has many advantages including stable sensitizers and dynamical control of redox reaction outcomes [80]. Kang constructed a natural sphalerite nanosheet heterojunction by using piezoelectric photocatalytic therapy to separate the charges in the interface under the irradiation of ultrasound and laser, which limited the recombination of charges (electron–hole pairs) and led to efficient catalytic performance, exhibiting high-performance superoxide radical (•O2−) and hydroxyl radical (•OH) generation and glutathione (GSH) depletion, causing ROS burst for cancer therapy [81].
4. Up-Conversion Nanoparticles for Photocatalytic Anti-Cancer Therapy
Due to the limited penetration, the ultraviolet light that excites the photocatalyst and the visible light that excites the organic photosensitizer are not suitable for the application of PDT in deep tissues and organs. The penetration ability of ultraviolet light and visible light into the skin is usually a few millimeters; therefore, they can’t be used for the treatment and removal of deep tumors. Since X-ray or NIR light can penetrate the human body, they are the best alternative to ultraviolet and visible light. However, most photocatalysts and organic photosensitizers are insensitive to X-ray and NIR light. To solve these issues, up-conversion nanoparticles (UCN) have been used in PDT and showed outstanding prospects in PDT application [82]. After being excited by X-rays or NIR light, UCN can emit a large number of low-energy photons. Rimoldi et al. synthesized a composite nanostructure for self-luminous PDT by combined ZnO nanoparticles with cerium fluoride (CeF3) and this composite nanostructure can generate ROS after excited by high-penetrating X-rays [83]. Compared with X-rays, NIR light is safer for the human body. Lanthanides are also sensitive to near-infrared light and emit ultraviolet fluorescence, which can activate semiconductors to produce reactive oxygen species, thereby killing cancer cells. Wang et al. [82] used Yb3+ and Tm3+ co-doped with lanthanide metal ions of NaYF4 upconversion nanoparticles to transfer NIR photons to high-energy photons to activate ZnO nanoparticles, thereby generating a large amount of ROS. However, how to improve the conversion efficiency by adjusting the ratio of ingredients is still a direction worthy of in-depth discussion by researchers.
Although UCNs excited by near-infrared light can produce abundant ROS to kill cancer cells, the upconversion spectrum of neutrons is strictly determined by the energy level of lanthanide ions. Thus, it is very difficult to match various absorption band of photosensitizers by adjusting the wavelength of upconversion light. Recently, Gu’s team [82] developed a completely different method that used the nonlinear optical interaction between incident NIR laser radiation and tumor-targeted molecular components to excite ZnO through second-harmonic generation, thereby triggering PDT via a single photon absorption. Since the excitation efficiency of single-photon absorption is higher than that of traditional two-photon absorption, ZnO nanoparticles can produce higher PDT efficiency. This new concept of PDT will be a more convenient and efficient treatment for in-depth tumors.
5. Challenges and Prospects of Photocatalysts for Cancer Cell Treatment
Photocatalytic therapy targeting the tumor microenvironment triggers a photocatalytic reaction by absorbing photon energy, destroying the redox balance in the tumor area, thereby killing tumor cells. Photocatalytic therapy is expected to become one of the treatment methods that can completely treat cancer in the future. However, photocatalytic therapy is still facing problems and challenges. The penetration depth of ultraviolet-visible light used in traditional photocatalytic reactions in human tissues is only a few millimeters, and it is difficult to reach deep tissues. Thus, traditional photocatalysts excited by ultraviolet-visible light are only suitable for the treatment of superficial tumors. Near-infrared light has a deeper penetration power, but the stimulated photodynamic therapy usually requires the consumption of oxygen, which will aggravate the hypoxia of tumor tissues, thereby limiting the efficacy of photodynamic therapy. X-ray with strong penetrating power is an ideal light source for the treatment of deep-seated tumors. However, radiotherapy can easily cause damage to the healthy tissues around the tumors, leading to greater side effects. Some new physical mechanisms including two-photon excitation and up-conversion process, as well as X-ray catalysis based on high atomic number materials or scintillators, provide feasible solutions to the above-mentioned problems. At present, further research is still needed for many photocatalytic materials used in tumor treatment before they are put into general clinical use. Most of the developed catalytic nanomedicines must be loaded with toxic drugs or easily exhaustible therapeutic adjuvants. The development of nanocatalytic nanomedicine for drug-free treatment is the focus of future research. Several major issues regarding photocatalytic therapy that need to be solved in the future are summarized as follows:
- How to control the ultraviolet-visible light to trigger the photocatalytic reaction for cancer therapy, breaking the limitation of penetration depth.
- Whether it can break through the mechanism of conventional reactive oxygen species to avoid further deterioration of the hypoxic environment in the tumor area.
- Whether the abundant reactive oxygen species can be produced in reducing environment in vivo to attack the DNA molecules in tumor cells.
- How to control the toxicity of photocatalysts and drug consumption caused by metabolism or immunity.
6. Conclusions
The development of nano-photocatalysts with anti-tumor efficiency has high theoretical value and practical significance. At present, the photocatalyst materials that have been applied in tumor therapy include supramolecular photocatalysts, metal and metal oxide photocatalysts, piezocatalysis, and up-conversion nanoparticles. They all showed high ROS production which can destroy cancer cells. The tumor cells can be killed by disrupting the redox balance in the tumor area. Photocatalytic therapy has great potential in the thorough treatment of cancer. However, the photocatalyst corresponding to ultraviolet-visible light faces a huge challenge in the treatment of in-depth cancer cells or tissues due to the limitation of the penetration power. Since most of the developed catalytic nanomedicines must be loaded with toxic drugs or therapeutic adjuvants that are easily depleted, the development of photocatalysts with drug-free treatment of cancer is the focus of future research.
Author Contributions
H.Y. wrote the paper. Z.C. guided the writing of the paper and reviewed the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by National Natural Science Foundation of China, grant number 82073932.
Data Availability Statement
The data used to support the findings of this study are available from the corresponding author upon request.
Conflicts of Interest
The authors declare no conflict of interest in this paper.
References
- Ali, E.S.; Sharker, S.M.; Islam, M.T.; Khan, I.N.; Shaw, S.; Rahman, M.A.; Uddin, S.J.; Shill, M.C.; Rehman, S.; Das, N. Targeting Cancer Cells with Nanotherapeutics and Nanodiagnostics: Current Status and Future Perspectives; Seminars in Cancer Biology; Elsevier: Amsterdam, The Netherlands, 2021; pp. 52–68. [Google Scholar]
- Boehm, J.S.; Garnett, M.J.; Adams, D.J.; Francies, H.E.; Golub, T.R.; Hahn, W.C.; Iorio, F.; McFarland, J.M.; Parts, L.; Vazquez, F. Cancer Research Needs a Better Map; Nature Publishing Group: Cambridge, MA, USA, 2021. [Google Scholar]
- Hahn, W.C.; Bader, J.S.; Braun, T.P.; Califano, A.; Clemons, P.A.; Druker, B.J.; Ewald, A.J.; Fu, H.; Jagu, S.; Kemp, C.J. An expanded universe of cancer targets. Cell 2021, 184, 1142–1155. [Google Scholar] [CrossRef] [PubMed]
- Ferlay, J.; Colombet, M.; Soerjomataram, I.; Parkin, D.M.; Piñeros, M.; Znaor, A.; Bray, F. Cancer statistics for the year 2020: An overview. Int. J. Cancer 2021, 149, 778–789. [Google Scholar] [CrossRef] [PubMed]
- Bao, Z.; Li, K.; Hou, P.; Xiao, R.; Yuan, Y.; Sun, Z. Nanoscale metal–organic framework composites for phototherapy and synergistic therapy of cancer. Mater. Chem. Front. 2021, 5, 1632–1654. [Google Scholar] [CrossRef]
- Bown, S. Phototherapy of tumors. World J. Surg. 1983, 7, 700–709. [Google Scholar] [CrossRef]
- Huang, X.; Jain, P.K.; El-Sayed, I.H.; El-Sayed, M.A. Plasmonic photothermal therapy (PPTT) using gold nanoparticles. Lasers Med. Sci. 2008, 23, 217–228. [Google Scholar] [CrossRef] [PubMed]
- Hou, Z.; Deng, K.; Wang, M.; Liu, Y.; Chang, M.; Huang, S.; Li, C.; Wei, Y.; Cheng, Z.; Han, G. Hydrogenated titanium oxide decorated upconversion nanoparticles: Facile laser modified synthesis and 808 nm near-infrared light triggered phototherapy. Chem. Mater. 2019, 31, 774–784. [Google Scholar] [CrossRef]
- Barbeira, A.N.; Dickinson, S.P.; Bonazzola, R.; Zheng, J.; Wheeler, H.E.; Torres, J.M.; Torstenson, E.S.; Shah, K.P.; Garcia, T.; Edwards, T.L. Exploring the phenotypic consequences of tissue specific gene expression variation inferred from GWAS summary statistics. Nat. Commun. 2018, 9, 1–20. [Google Scholar] [CrossRef]
- Huang, Z. A review of progress in clinical photodynamic therapy. Technol. Cancer Res. Treat. 2005, 4, 283–293. [Google Scholar] [CrossRef] [Green Version]
- Bae, K.H.; Chung, H.J.; Park, T.G. Nanomaterials for cancer therapy and imaging. Mol. Cells 2011, 31, 295–302. [Google Scholar] [CrossRef] [Green Version]
- Szaciłowski, K.; Macyk, W.; Drzewiecka-Matuszek, A.; Brindell, M.; Stochel, G. Bioinorganic photochemistry: Frontiers and mechanisms. Chem. Rev. 2005, 105, 2647–2694. [Google Scholar] [CrossRef]
- Sunada, K.; Watanabe, T.; Hashimoto, K. Studies on photokilling of bacteria on TiO2 thin film. J. Photochem. Photobiol. A Chem. 2003, 156, 227–233. [Google Scholar] [CrossRef]
- Ziental, D.; Czarczynska-Goslinska, B.; Mlynarczyk, D.T.; Glowacka-Sobotta, A.; Stanisz, B.; Goslinski, T.; Sobotta, L. Titanium dioxide nanoparticles: Prospects and applications in medicine. Nanomaterials 2020, 10, 387. [Google Scholar] [CrossRef] [Green Version]
- de Dicastillo, C.L.; Correa, M.G.; Martínez, F.B.; Streitt, C.; Galotto, M.J. Antimicrobial effect of titanium dioxide nanoparticles. In Antimicrobial Resistance-A One Health Perspective; IntechOpen: London, UK, 2020. [Google Scholar]
- Yamaguchi, K.; Sugiyama, T.; Kato, S.; Kondo, Y.; Ageyama, N.; Kanekiyo, M.; Iwata, M.; Koyanagi, Y.; Yamamoto, N.; Honda, M. A novel CD4–conjugated ultraviolet light-activated photocatalyst inactivates HIV–1 and SIV efficiently. J. Med. Virol. 2008, 80, 1322–1331. [Google Scholar] [CrossRef]
- Seo, J.W.; Chung, H.; Kim, M.Y.; Lee, J.; Choi, I.H.; Cheon, J. Development of water–soluble single–crystalline TiO2 nanoparticles for photocatalytic cancer–cell treatment. Small 2007, 3, 850–853. [Google Scholar] [CrossRef] [PubMed]
- Kalbacova, M.; Macak, J.; Schmidt-Stein, F.; Mierke, C.; Schmuki, P. TiO2 nanotubes: Photocatalyst for cancer cell killing. Phys. Status Solidi (RRL)—Rapid Res. Lett. 2008, 2, 194–196. [Google Scholar] [CrossRef]
- Xu, J.; Sun, Y.; Huang, J.; Chen, C.; Liu, G.; Jiang, Y.; Zhao, Y.; Jiang, Z. Photokilling cancer cells using highly cell-specific antibody–TiO2 bioconjugates and electroporation. Bioelectrochemistry 2007, 71, 217–222. [Google Scholar] [CrossRef]
- Mao, C.; Xiang, Y.; Liu, X.; Cui, Z.; Yang, X.; Yeung, K.W.K.; Pan, H.; Wang, X.; Chu, P.K.; Wu, S. Photo-inspired antibacterial activity and wound healing acceleration by hydrogel embedded with Ag/Ag@ AgCl/ZnO nanostructures. ACS Nano 2017, 11, 9010–9021. [Google Scholar] [CrossRef]
- Qi, K.; Cheng, B.; Yu, J.; Ho, W. Review on the improvement of the photocatalytic and antibacterial activities of ZnO. J. Alloy. Compd. 2017, 727, 792–820. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhu, Y.; Chen, X.; Zhang, H.; Wang, J. A full–spectrum metal–free porphyrin supramolecular photocatalyst for dual functions of highly efficient hydrogen and oxygen evolution. Adv. Mater. 2019, 31, 1806626. [Google Scholar] [CrossRef]
- Li, W.; Wang, C.; Yao, Y.; Wu, C.; Luo, W.; Zou, Z. Photocatalytic Materials: An Apollo’s Arrow to Tumor Cells. Trends Chem. 2020, 2, 1126–1140. [Google Scholar] [CrossRef]
- Rozhkova, E.A.; Ulasov, I.; Lai, B.; Dimitrijevic, N.M.; Lesniak, M.S.; Rajh, T. A high-performance nanobio photocatalyst for targeted brain cancer therapy. Nano Lett. 2009, 9, 3337–3342. [Google Scholar] [CrossRef] [Green Version]
- Ferrari, M. Cancer nanotechnology: Opportunities and challenges. Nat. Rev. Cancer 2005, 5, 161–171. [Google Scholar] [CrossRef]
- Dougherty, T.J.; Gomer, C.J.; Henderson, B.W.; Jori, G.; Kessel, D.; Korbelik, M.; Moan, J.; Peng, Q. Photodynamic therapy. JNCI J. Natl. Cancer Inst. 1998, 90, 889–905. [Google Scholar] [CrossRef] [Green Version]
- Henderson, B.W. Photodynamic Therapy: Basic Principles and Clinical Applications; CRC Press: New York, NY, USA, 2020. [Google Scholar]
- Zhou, Z.; Song, J.; Nie, L.; Chen, X. Reactive oxygen species generating systems meeting challenges of photodynamic cancer therapy. Chem. Soc. Rev. 2016, 45, 6597–6626. [Google Scholar] [CrossRef] [Green Version]
- Agostinis, P.; Berg, K.; Cengel, K.A.; Foster, T.H.; Girotti, A.W.; Gollnick, S.O.; Hahn, S.M.; Hamblin, M.R.; Juzeniene, A.; Kessel, D. Photodynamic therapy of cancer: An update. CA Cancer J. Clin. 2011, 61, 250–281. [Google Scholar] [CrossRef]
- Ni, J.; Wang, Y.; Zhang, H.; Sun, J.Z.; Tang, B.Z. Aggregation-Induced Generation of Reactive Oxygen Species: Mechanism and Photosensitizer Construction. Molecules 2021, 26, 268. [Google Scholar] [CrossRef] [PubMed]
- Dąbrowski, J.M. Reactive oxygen species in photodynamic therapy: Mechanisms of their generation and potentiation. Adv. Inorg. Chem. 2017, 70, 343–394. [Google Scholar]
- Wang, J.; Liu, D.; Zhu, Y.; Zhou, S.; Guan, S. Supramolecular packing dominant photocatalytic oxidation and anticancer performance of PDI. Appl. Catal. B Environ. 2018, 231, 251–261. [Google Scholar] [CrossRef]
- Zou, Q.; Abbas, M.; Zhao, L.; Li, S.; Shen, G.; Yan, X. Biological photothermal nanodots based on self-assembly of peptide–porphyrin conjugates for antitumor therapy. J. Am. Chem. Soc. 2017, 139, 1921–1927. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, L.; Liu, W.; Yan, Z.; Zhu, Y.; Zhou, S.; Guan, S. Photogenerated-hole-induced rapid elimination of solid tumors by the supramolecular porphyrin photocatalyst. Natl. Sci. Rev. 2021, 8, nwaa155. [Google Scholar] [CrossRef]
- Wang, Y.; Du, W.; Zhang, T.; Zhu, Y.; Ni, Y.; Wang, C.; Raya, F.M.S.; Zou, L.; Wang, L.; Liang, G. A Self-Evaluating Photothermal Therapeutic Nanoparticle. ACS Nano 2020, 14, 9585–9593. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.; Lu, Y.; Mu, X.; Chen, Z.; Liu, S.; Zhou, X.; Liu, S.; Li, Z. Intriguing H-Aggregates of Heptamethine Cyanine for Imaging-Guided Photothermal Cancer Therapy. ACS Appl. Mater. Interfaces 2020, 12, 32388–32396. [Google Scholar] [CrossRef] [PubMed]
- Wahab, R.; Dwivedi, S.; Umar, A.; Singh, S.; Hwang, I.; Shin, H.-S.; Musarrat, J.; Al-Khedhairy, A.A.; Kim, Y.-S. ZnO nanoparticles induce oxidative stress in Cloudman S91 melanoma cancer cells. J. Biomed. Nanotechnol. 2013, 9, 441–449. [Google Scholar] [CrossRef]
- Wahab, R.; Kaushik, N.K.; Kaushik, N.; Choi, E.H.; Umar, A.; Dwivedi, S.; Musarrat, J.; Al-Khedhairy, A.A. ZnO nanoparticles induces cell death in malignant human T98G gliomas, KB and non-malignant HEK cells. J. Biomed. Nanotechnol. 2013, 9, 1181–1189. [Google Scholar] [CrossRef] [Green Version]
- Wahab, R.; Siddiqui, M.A.; Saquib, Q.; Dwivedi, S.; Ahmad, J.; Musarrat, J.; Al-Khedhairy, A.A.; Shin, H.-S. ZnO nanoparticles induced oxidative stress and apoptosis in HepG2 and MCF-7 cancer cells and their antibacterial activity. Colloids Surf. B Biointerfaces 2014, 117, 267–276. [Google Scholar] [CrossRef]
- Wason, M.S.; Colon, J.; Das, S.; Seal, S.; Turkson, J.; Zhao, J.; Baker, C.H. Sensitization of pancreatic cancer cells to radiation by cerium oxide nanoparticle-induced ROS production. Nanomed. Nanotechnol. Biol. Med. 2013, 9, 558–569. [Google Scholar] [CrossRef] [Green Version]
- Sack, M.; Alili, L.; Karaman, E.; Das, S.; Gupta, A.; Seal, S.; Brenneisen, P. Combination of conventional chemotherapeutics with redox-active cerium oxide nanoparticles—A novel aspect in cancer therapy. Mol. Cancer Ther. 2014, 13, 1740–1749. [Google Scholar] [CrossRef] [Green Version]
- Alili, L.; Sack, M.; Karakoti, A.S.; Teuber, S.; Puschmann, K.; Hirst, S.M.; Reilly, C.M.; Zanger, K.; Stahl, W.; Das, S. Combined cytotoxic and anti-invasive properties of redox-active nanoparticles in tumor–stroma interactions. Biomaterials 2011, 32, 2918–2929. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Yang, F.; Zhang, H.; Zi, X.; Pan, X.; Chen, F.; Luo, W.; Li, J.; Zhu, H.; Hu, Y. Cuprous oxide nanoparticles inhibit the growth and metastasis of melanoma by targeting mitochondria. Cell Death Dis. 2013, 4, e783. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yuan, Y.; He, Y.; Bo, R.; Ma, Z.; Wang, Z.; Dong, L.; Lin, T.-Y.; Xue, X.; Li, Y. A facile approach to fabricate self-assembled magnetic nanotheranostics for drug delivery and imaging. Nanoscale 2018, 10, 21634–21639. [Google Scholar] [CrossRef] [PubMed]
- Zhi, D.; Yang, T.; Yang, J.; Fu, S.; Zhang, S. Targeting strategies for superparamagnetic iron oxide nanoparticles in cancer therapy. Acta Biomater. 2020, 102, 13–34. [Google Scholar] [CrossRef]
- Jukapli, N.M.; Bagheri, S. Recent developments on titania nanoparticle as photocatalytic cancer cells treatment. J. Photochem. Photobiol. B Biol. 2016, 163, 421–430. [Google Scholar] [CrossRef]
- Kubota, Y.; Shuin, T.; Kawasaki, C.; Hosaka, M.; Kitamura, H.; Cai, R.; Sakai, H.; Hashimoto, K.; Fujishima, A. Photokilling of T-24 human bladder cancer cells with titanium dioxide. Br. J. Cancer 1994, 70, 1107–1111. [Google Scholar] [CrossRef]
- Huang, C.; Liang, C.; Sadhukhan, T.; Banerjee, S.; Fan, Z.; Li, T.; Zhu, Z.; Zhang, P.; Raghavachari, K.; Huang, H. In–vitro and In–vivo Photocatalytic Cancer Therapy with Biocompatible Iridium (III) Photocatalysts. Angew. Chem. 2021, 133, 9560–9565. [Google Scholar] [CrossRef]
- Tudor, D.; Nenu, I.; Filip, G.A.; Olteanu, D.; Cenariu, M.; Tabaran, F.; Ion, R.M.; Gligor, L.; Baldea, I. Combined regimen of photodynamic therapy mediated by Gallium phthalocyanine chloride and Metformin enhances anti-melanoma efficacy. PLoS ONE 2017, 12, e0173241. [Google Scholar] [CrossRef]
- Maji, S.K.; Kim, D.H. AgInS2-coated upconversion nanoparticle as a photocatalyst for near-infrared light-activated photodynamic therapy of cancer cells. ACS Appl. Bio Mater. 2018, 1, 1628–1638. [Google Scholar] [CrossRef]
- Wheate, N.J.; Collins, J.G. Multi-nuclear platinum complexes as anti-cancer drugs. Coord. Chem. Rev. 2003, 241, 133–145. [Google Scholar] [CrossRef]
- Zhao, B.; Wang, Y.; Yao, X.; Chen, D.; Fan, M.; Jin, Z.; He, Q. Photocatalysis-mediated drug-free sustainable cancer therapy using nanocatalyst. Nat. Commun. 2021, 12, 1–11. [Google Scholar]
- Sang, D.; Wang, K.; Sun, X.; Wang, Y.; Lin, H.; Jia, R.; Qu, F. NIR-Driven Intracellular Photocatalytic O2 Evolution on Z-Scheme Ni3S2/Cu1. 8S@ HA for Hypoxic Tumor Therapy. ACS Appl. Mater. Interfaces 2021, 13, 9604–9619. [Google Scholar] [CrossRef] [PubMed]
- Zhu, P.; Chen, Y.; Shi, J. Piezocatalytic tumor therapy by ultrasound-triggered and BaTiO3-mediated piezoelectricity. Adv. Mater. 2020, 32, 2001976. [Google Scholar] [CrossRef] [PubMed]
- Chao, S.; Shen, Z.; Pei, Y.; Lv, Y.; Chen, X.; Ren, J.; Yang, K.; Pei, Z. Pillar [5] arene-based supramolecular photosensitizer for enhanced hypoxic-tumor therapeutic effectiveness. Chem. Commun. 2021, 57, 7625–7628. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Mao, D.; Wang, Y.; Wang, K.; Yi, X.; Kong, D.; Yang, Z.; Liu, Q.; Ding, D. Biocompatible fluorescent supramolecular nanofibrous hydrogel for long-term cell tracking and tumor imaging applications. Sci. Rep. 2015, 5, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Li, H.; Zhao, J.; Wang, A.; Li, Q.; Cui, W. Supramolecular assembly of protein-based nanoparticles based on tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) for cancer therapy. Colloids Surf. A Physicochem. Eng. Asp. 2020, 590, 124486. [Google Scholar] [CrossRef]
- Prier, C.K.; Rankic, D.A.; MacMillan, D.W.C. Visible Light Photoredox Catalysis with Transition Metal Complexes: Applications in Organic Synthesis. Chem. Rev. 2013, 113, 5322–5363. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, H.; Banerjee, S.; Qiu, K.; Zhang, P.; Blacque, O.; Malcomson, T.; Paterson, M.J.; Clarkson, G.J.; Staniforth, M.; Stavros, V.G. Targeted photoredox catalysis in cancer cells. Nat. Chem. 2019, 11, 1041–1048. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Romero-Canelón, I.; Qamar, B.; Hearn, J.M.; Habtemariam, A.; Barry, N.P.; Pizarro, A.M.; Clarkson, G.J.; Sadler, P.J. The potent oxidant anticancer activity of organoiridium catalysts. Angew. Chem. 2014, 126, 4022–4027. [Google Scholar] [CrossRef] [Green Version]
- Xue, Y.; Luan, Q.; Yang, D.; Yao, X.; Zhou, K. Direct evidence for hydroxyl radical scavenging activity of cerium oxide nanoparticles. J. Phys. Chem. C 2011, 115, 4433–4438. [Google Scholar] [CrossRef]
- Asati, A.; Santra, S.; Kaittanis, C.; Nath, S.; Perez, J.M. Oxidase–like activity of polymer–coated cerium oxide nanoparticles. Angew. Chem. 2009, 121, 2344–2348. [Google Scholar] [CrossRef]
- Lin, W.; Huang, Y.-W.; Zhou, X.-D.; Ma, Y. Toxicity of cerium oxide nanoparticles in human lung cancer cells. Int. J. Toxicol. 2006, 25, 451–457. [Google Scholar] [CrossRef]
- Park, E.-J.; Choi, J.; Park, Y.-K.; Park, K. Oxidative stress induced by cerium oxide nanoparticles in cultured BEAS-2B cells. Toxicology 2008, 245, 90–100. [Google Scholar] [CrossRef]
- Pešić, M.; Podolski-Renić, A.; Stojković, S.; Matović, B.; Zmejkoski, D.; Kojić, V.; Bogdanović, G.; Pavićević, A.; Mojović, M.; Savić, A. Anti-cancer effects of cerium oxide nanoparticles and its intracellular redox activity. Chem. Biol. Interact. 2015, 232, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Raja, G.; Cao, S.; Kim, D.-H.; Kim, T.-J. Mechanoregulation of titanium dioxide nanoparticles in cancer therapy. Mater. Sci. Eng. C 2020, 107, 110303. [Google Scholar] [CrossRef] [PubMed]
- Hidaka, H.; Horikoshi, S.; Serpone, N.; Knowland, J. In vitro photochemical damage to DNA, RNA and their bases by an inorganic sunscreen agent on exposure to UVA and UVB radiation. J. Photochem. Photobiol. A Chem. 1997, 111, 205–213. [Google Scholar] [CrossRef]
- Dunford, R.; Salinaro, A.; Cai, L.; Serpone, N.; Horikoshi, S.; Hidaka, H.; Knowland, J. Chemical oxidation and DNA damage catalysed by inorganic sunscreen ingredients. FEBS Lett. 1997, 418, 87–90. [Google Scholar] [CrossRef] [Green Version]
- Huang, N.-P.; Min-hua, X.; Yuan, C.-W.; Rui-rong, Y. The study of the photokilling effect and mechanism of ultrafine TiO2 particles on U937 cells. J. Photochem. Photobiol. A Chem. 1997, 108, 229–233. [Google Scholar] [CrossRef]
- Li, Z.; He, J.; Li, B.; Zhang, J.; He, K.; Duan, X.; Huang, R.; Wu, Z.; Xiang, G. Titanium dioxide nanoparticles induce endoplasmic reticulum stress-mediated apoptotic cell death in liver cancer cells. J. Int. Med Res. 2020, 48, 0300060520903652. [Google Scholar] [CrossRef]
- Botelho, M.C.; Costa, C.; Silva, S.; Costa, S.; Dhawan, A.; Oliveira, P.A.; Teixeira, J.P. Effects of titanium dioxide nanoparticles in human gastric epithelial cells in vitro. Biomed. Pharmacother. 2014, 68, 59–64. [Google Scholar] [CrossRef] [Green Version]
- Yang, Q.; Wang, Y.; Yang, Q.; Gao, Y.; Duan, X.; Fu, Q.; Chu, C.; Pan, X.; Cui, X.; Sun, Y. Cuprous oxide nanoparticles trigger ER stress-induced apoptosis by regulating copper trafficking and overcoming resistance to sunitinib therapy in renal cancer. Biomaterials 2017, 146, 72–85. [Google Scholar] [CrossRef]
- Zhao, R.; Wang, B.; Yang, X.; Xiao, Y.; Wang, X.; Shao, C.; Tang, R. A drug-free tumor therapy strategy: Cancer-cell-targeting calcification. Angew. Chem. Int. Ed. 2016, 55, 5225–5229. [Google Scholar] [CrossRef]
- Huo, M.; Wang, L.; Chen, Y.; Shi, J. Tumor-selective catalytic nanomedicine by nanocatalyst delivery. Nat. Commun. 2017, 8, 1–12. [Google Scholar] [CrossRef]
- Lin, H.; Chen, Y.; Shi, J. Nanoparticle-triggered in situ catalytic chemical reactions for tumour-specific therapy. Chem. Soc. Rev. 2018, 47, 1938–1958. [Google Scholar] [CrossRef]
- Fan, K.; Xi, J.; Fan, L.; Wang, P.; Zhu, C.; Tang, Y.; Xu, X.; Liang, M.; Jiang, B.; Yan, X. In vivo guiding nitrogen-doped carbon nanozyme for tumor catalytic therapy. Nat. Commun. 2018, 9, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Fan, K.; Cao, C.; Pan, Y.; Lu, D.; Yang, D.; Feng, J.; Song, L.; Liang, M.; Yan, X. Magnetoferritin nanoparticles for targeting and visualizing tumour tissues. Nat. Nanotechnol. 2012, 7, 459–464. [Google Scholar] [CrossRef]
- Pan, C.; Ou, M.; Cheng, Q.; Zhou, Y.; Yu, Y.; Li, Z.; Zhang, F.; Xia, D.; Mei, L.; Ji, X. Z-scheme Heterojunction functionalized pyrite Nanosheets for modulating tumor microenvironment and strengthening photo/Chemodynamic therapeutic effects. Adv. Funct. Mater. 2020, 30, 1906466. [Google Scholar] [CrossRef]
- Kang, Y.; Lei, L.; Zhu, C.; Zhang, H.; Mei, L.; Ji, X. Piezo-Photocatalytic Effect Mediating Reactive Oxygen Species Burst for Cancer Catalytic Therapy. Mater. Horiz. 2021, 8, 2273–2285. [Google Scholar] [CrossRef]
- Dou, Q.Q.; Rengaramchandran, A.; Selvan, S.T.; Paulmurugan, R.; Zhang, Y. Core–shell upconversion nanoparticle–semiconductor heterostructures for photodynamic therapy. Sci. Rep. 2015, 5, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Rimoldi, T.; Orsi, D.; Lagonegro, P.; Ghezzi, B.; Galli, C.; Rossi, F.; Salviati, G.; Cristofolini, L. CeF3-ZnO scintillating nanocomposite for self-lighted photodynamic therapy of cancer. J. Mater. Sci. Mater. Med. 2016, 27, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.N.; Zhang, F.; Zhang, C.L.; Guo, Y.C.; Dai, W.; Qian, H.S. Fabrication of Zinc Oxide Composite Microfibers for Near-Infrared-Light-Mediated Photocatalysis. ChemCatChem 2017, 9, 3611–3617. [Google Scholar] [CrossRef]
- Gu, B.; Pliss, A.; Kuzmin, A.N.; Baev, A.; Ohulchanskyy, T.Y.; Damasco, J.A.; Yong, K.-T.; Wen, S.; Prasad, P.N. In-situ second harmonic generation by cancer cell targeting ZnO nanocrystals to effect photodynamic action in subcellular space. Biomaterials 2016, 104, 78–86. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).