Microstructures and Mechanical Properties of Nanocrystalline AZ31 Magnesium Alloy Powders with Submicron TiB2 Additions Prepared by Mechanical Milling
Abstract
1. Introduction
2. Experimental Procedures
3. Results and Discussion
3.1. Morphology Evolution of Powders
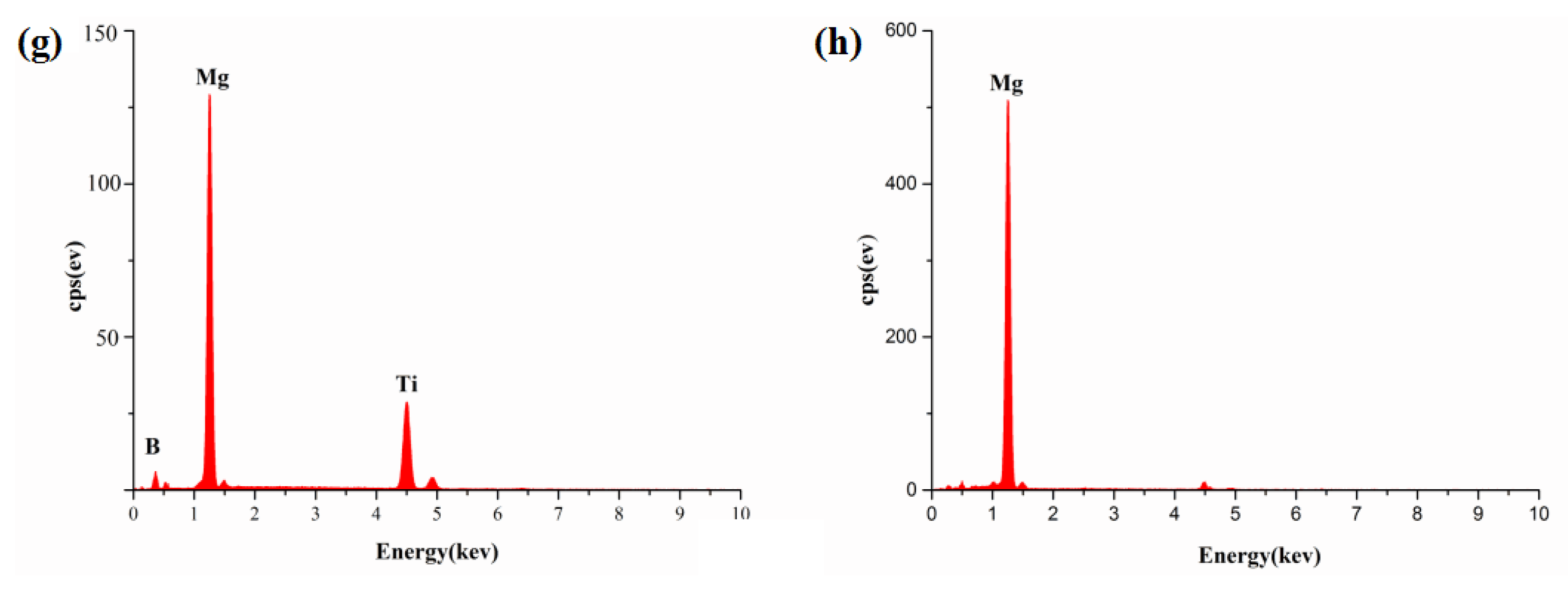
3.2. Evolution of TiB2 Particles
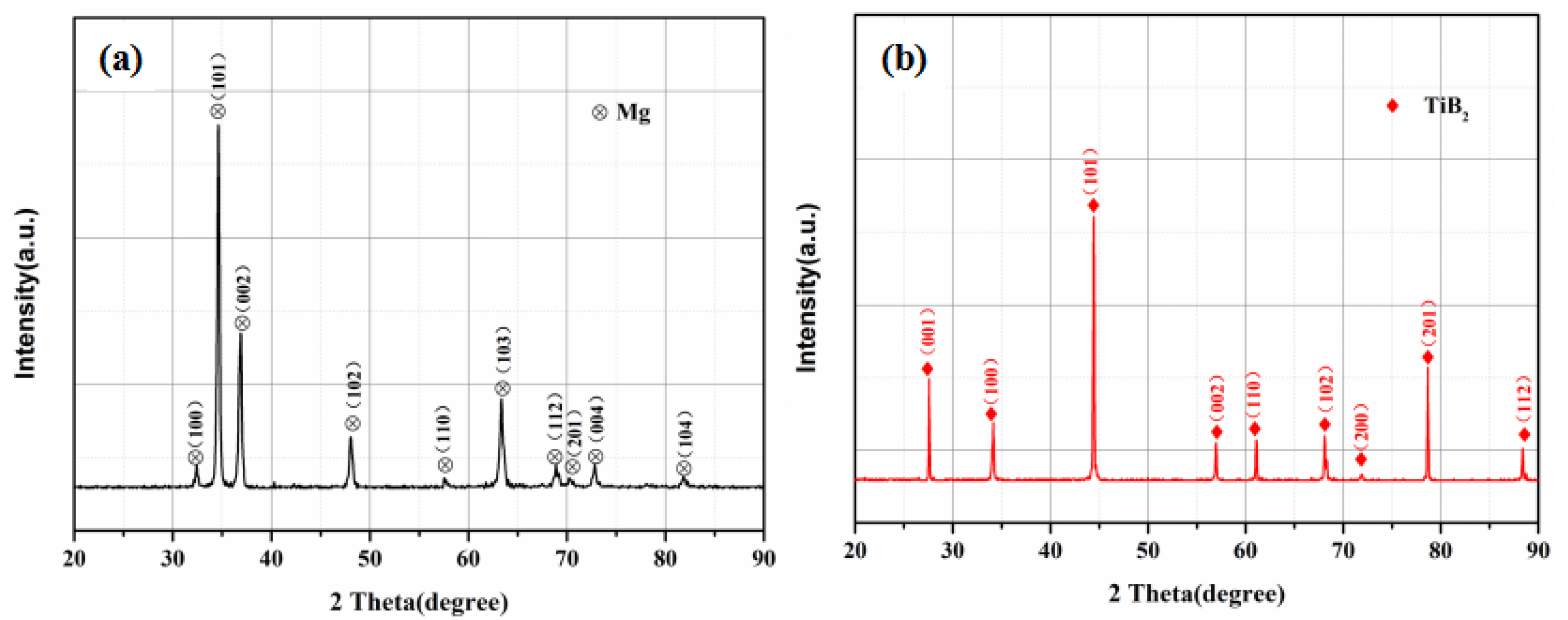
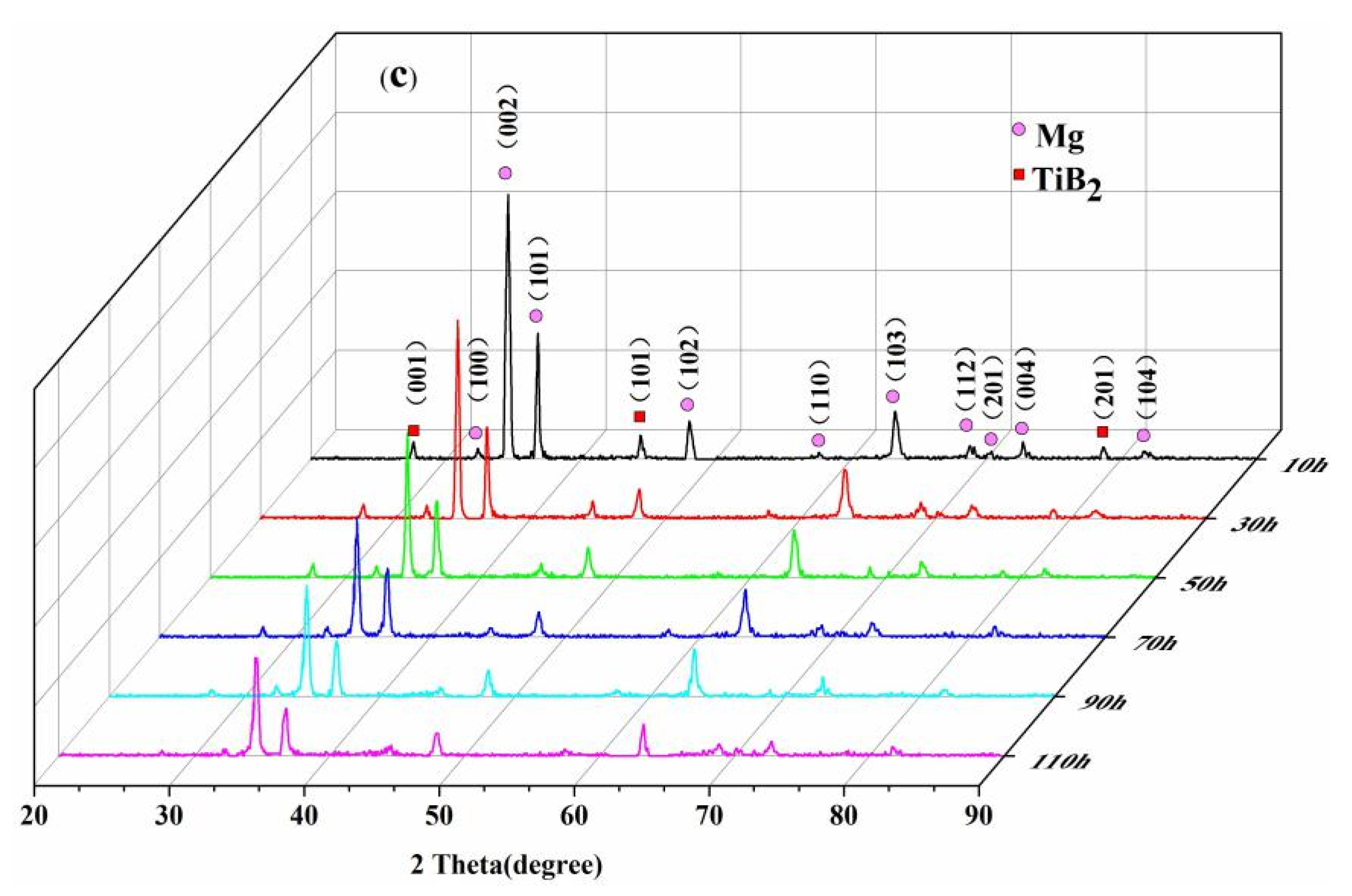
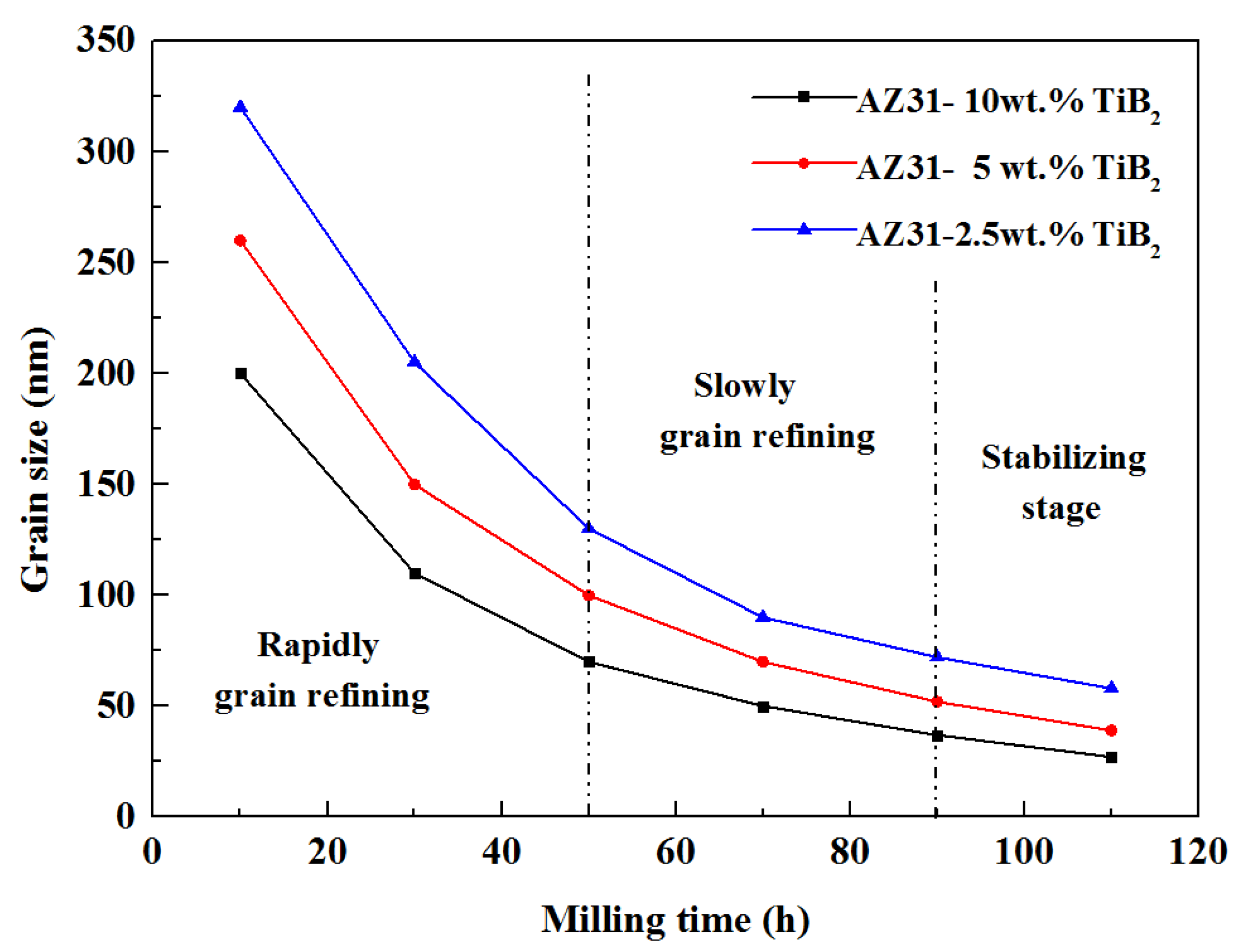
3.3. XRD Analysis
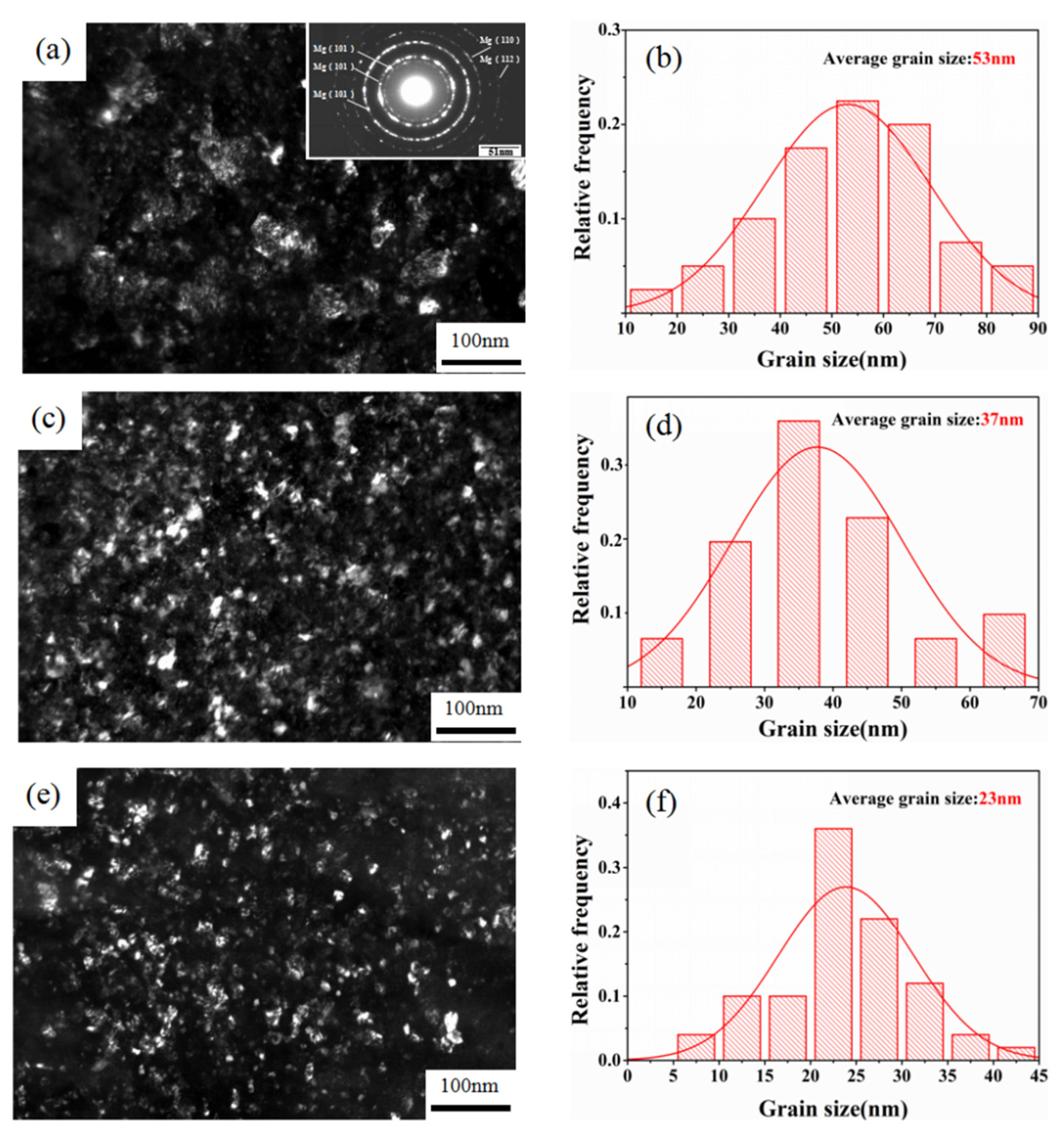
3.4. TEM Analysis
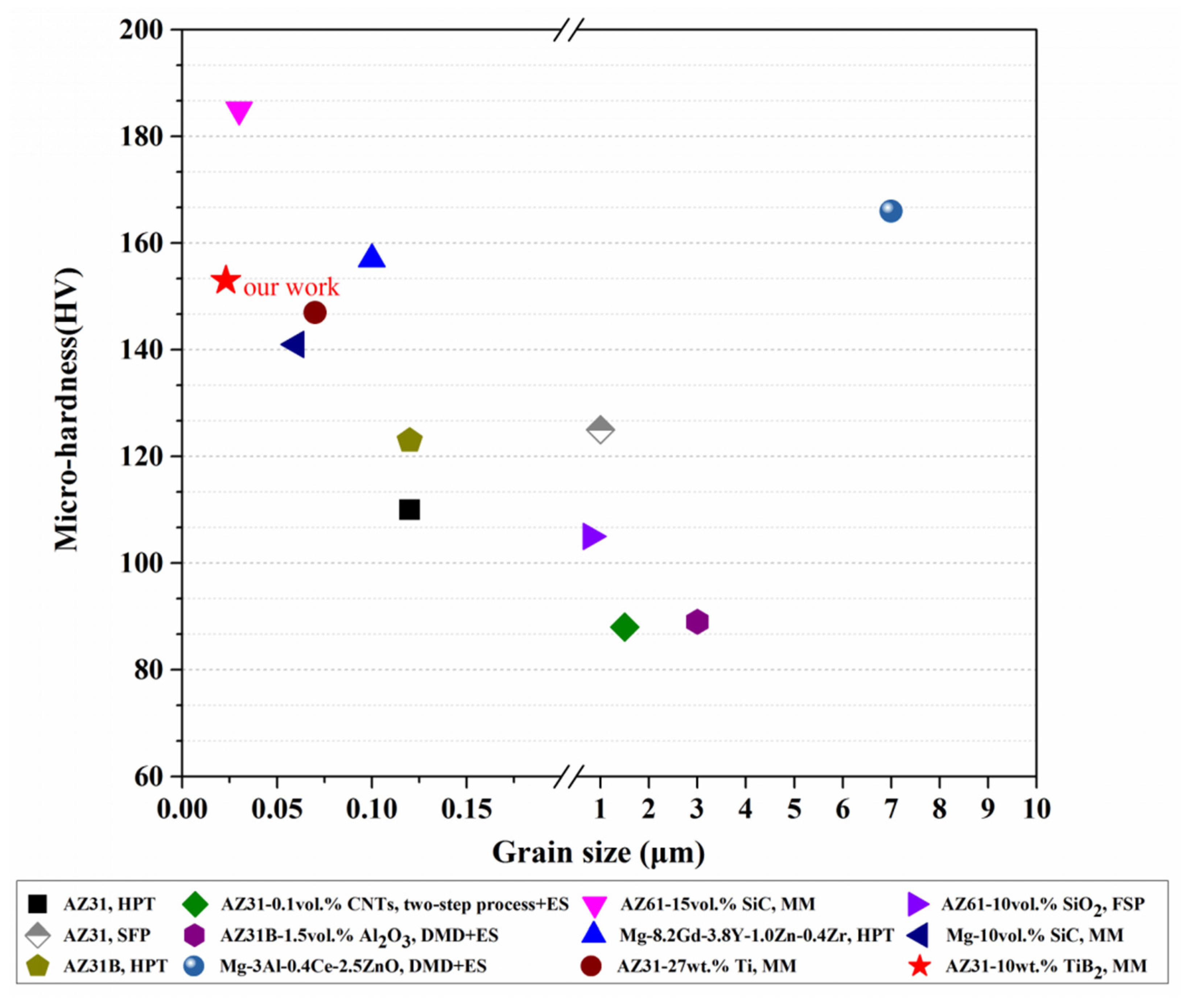
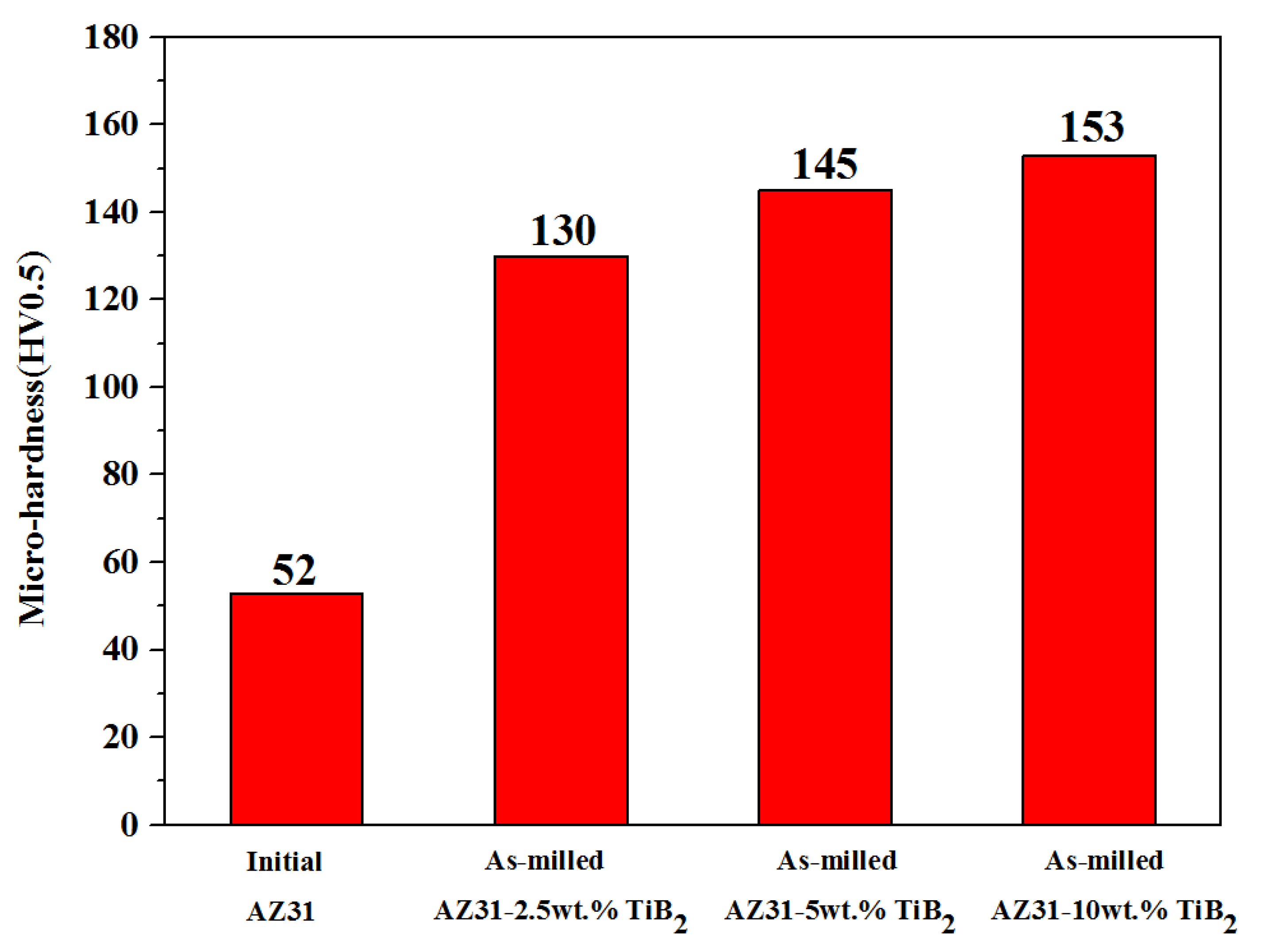
3.5. Hardness Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Suryanarayana, C.; Al-Aqeeli, N. Mechanically alloyed nanocomposites. Prog. Mater. Sci. 2013, 58, 383–502. [Google Scholar] [CrossRef]
- Feng, J.; Sun, H.; Li, X.; Zhang, J.; Fang, W.; Fang, W. Microstructures and mechanical properties of the ultrafine-grained Mg–3Al–Zn alloys fabricated by powder metallurgy. Adv. Powder Technol. 2016, 27, 550–556. [Google Scholar] [CrossRef]
- Pourbahari, B.; Emamy, M.; Mirzadeh, H. Synergistic effect of Al and Gd on enhancement of mechanical properties of magnesium alloys. Prog. Nat. Sci. Mater. Int. 2017, 27, 228–235. [Google Scholar] [CrossRef]
- Asgharzadeh, H.; Yoon, E.Y.; Chae, H.J.; Kim, T.S.; Lee, J.W.; Kim, H.S. Microstructure and mechanical properties of a Mg–Zn–Y alloy produced by a powder metallurgy route. J. Alloys Compd. 2014, 586, S95–S100. [Google Scholar] [CrossRef]
- Hofstetter, J.; Rüedi, S.; Baumgartner, I.; Kilian, H.; Mingler, B.; Povoden-Karadeniz, E.; Pogatscher, S.; Uggowitzer, P.J.; Löffler, J.F. Processing and microstructure–property relations of high-strength low-alloy (HSLA) Mg–Zn–Ca alloys. Acta Mater. 2015, 98, 423–432. [Google Scholar] [CrossRef]
- Yu, H.; Sun, Y.; Hu, L.; Zhou, H.; Wan, Z. Microstructural evolution of AZ61-10at.%Ti composite powders during mechanical milling. Mater. Des. 2016, 104, 265–275. [Google Scholar] [CrossRef]
- Chaubey, A.K.; Scudino, S.; Khoshkhoo, M.S.; Prashanth, K.G.; Mukhopadhyay, N.K.; Mishra, B.K.; Eckert, J. High-strength ultrafine grain Mg–7.4%Al alloy synthesized by consolidation of mechanically alloyed powders. J. Alloys Compd. 2014, 610, 456–461. [Google Scholar] [CrossRef]
- Shen, M.J.; Ying, W.F.; Wang, X.J.; Zhang, M.F.; Wu, K. Development of High Performance Magnesium Matrix Nanocomposites Using Nano-SiC Particulates as Reinforcement. J. Mater. Eng. Perform. 2015, 24, 3798–3807. [Google Scholar] [CrossRef]
- Yuan, W.; Panigrahi, S.K.; Su, J.Q.; Mishra, R.S. Influence of grain size and texture on Hall–Petch relationship for a magnesium alloy. Scr. Mater. 2011, 65, 994–997. [Google Scholar] [CrossRef]
- Stráská, J.; Janeček, M.; Gubicza, J.; Krajňák, T.; Yoon, E.Y.; Kim, H.S. Evolution of microstructure and hardness in AZ31 alloy processed by high pressure torsion. Mater. Sci. Eng. A 2015, 625, 98–106. [Google Scholar] [CrossRef]
- Pérez-Prado, M.T.; Valle, d.; Ruano, O.A. Grain refinement of Mg–Al–Zn alloys via accumulative roll bonding. Scr. Mater. 2004, 51, 1093–1097. [Google Scholar] [CrossRef]
- Gzyl, M.; Rosochowski, A.; Boczkal, S.; Olejnik, L. The role of microstructure and texture in controlling mechanical properties of AZ31B magnesium alloy processed by I-ECAP. Mater. Sci. Eng. A 2015, 638, 20–29. [Google Scholar] [CrossRef]
- Chang, C.I.; Du, X.H.; Huang, J.C. Producing nanograined microstructure in Mg–Al–Zn alloy by two-step friction stir processing. Scr. Mater. 2008, 59, 356–359. [Google Scholar] [CrossRef]
- Fang, W.-B.; Fang, W.; Sun, H.-F. Preparation of bulk ultrafine-grained Mg-3Al-Zn alloys by consolidation of ball milling nanocrystalline powders. Trans. Nonferrous Metals Soc. China 2011, 21, s247–s251. [Google Scholar] [CrossRef]
- Suryanarayana, C. Mechanical alloying and milling. Prog. Mater. Sci. 2011, 46, 1–184. [Google Scholar] [CrossRef]
- Matin, A.; Saniee, F.F.; Abedi, H.R. Microstructure and mechanical properties of Mg/SiC and AZ80/SiC nano-composites fabricated through stir casting method. Mater. Sci. Eng. A 2015, 625, 81–88. [Google Scholar] [CrossRef]
- Aydin, F.; Sun, Y.; Ahlatci, H.; Turen, Y. Investigation of Microstructure, Mechanical and Wear Behaviour of B4C Particulate Reinforced Magnesium Matrix Composites by Powder Metallurgy. Trans. Indian Inst. Metals 2018, 71, 873–882. [Google Scholar] [CrossRef]
- Su, H.; Gao, W.; Feng, Z.; Lu, Z. Processing, microstructure and tensile properties of nano-sized Al2O3 particle reinforced aluminum matrix composites. Mater. Des. 2012, 36, 590–596. [Google Scholar] [CrossRef]
- Sankaranarayanan, S.; Habibi, M.K.; Jayalakshmi, S.; Ai, K.J.; Almajid, A.; Gupta, M. Nano-AlN particle reinforced Mg composites: Microstructural and mechanical properties. Mater. Sci. Technol. 2015, 31, 1122–1131. [Google Scholar] [CrossRef]
- Rahmany-Gorji, R.; Alizadeh, A.; Jafari, H. Microstructure and mechanical properties of stir cast ZX51/Al2O3p magnesium matrix composites. Mater. Sci. Eng. A 2016, 674, 413–418. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, H.-Y.; Xiu, K.; Wang, H.-Y.; Jiang, Q.-C. Fabrication of TiB2 particulate reinforced magnesium matrix composites by two-step processing method. Mater. Lett. 2006, 60, 1533–1537. [Google Scholar] [CrossRef]
- Xiao, P.; Gao, Y.; Xu, F.; Yang, S.; Li, B.; Li, Y.; Huang, Z.; Zheng, Q. An investigation on grain refinement mechanism of TiB2 particulate reinforced AZ91 composites and its effect on mechanical properties. J. Alloys Compd. 2019, 780, 237–244. [Google Scholar] [CrossRef]
- Aydin, F.; Sun, Y. Investigation of wear behaviour and microstructure of hot-pressed TiB2 particulate-reinforced magnesium matrix composites. Can. Metall. Q. 2018, 57, 455–469. [Google Scholar] [CrossRef]
- Lei, R.S.; Wang, M.P.; Li, Z.; Wei, H.G.; Yang, W.C.; Jia, Y.L.; Gong, S. Structure evolution and solid solubility extension of copper–niobium powders during mechanical alloying. Mater. Sci. Eng. A 2011, 528, 4475–4481. [Google Scholar] [CrossRef]
- Zhou, H.; Hu, L.; Sun, Y.; Zhang, H.; Duan, C.; Yu, H. Synthesis of nanocrystalline AZ31 magnesium alloy with titanium addition by mechanical milling. Mater. Charact. 2016, 113, 108–116. [Google Scholar] [CrossRef]
- Fogagnolo, J.B.; Velasco, F.; Robert, M.H.; Torralba, J.M. Effect of mechanical alloying on the morphology, microstructure and properties of aluminium matrix composite powders. Mater. Sci. Eng. A 2003, 342, 131–143. [Google Scholar] [CrossRef]
- Bemanifar, S.; Rajabi, M.; Hosseinipour, S.J. Microstructural Characterization of Mg-SiC Nanocomposite Powders Fabricated by High Energy Mechanical Milling. Silicon 2017, 9, 823–827. [Google Scholar] [CrossRef]
- Hesabi, Z.R.; Simchi, A.; Reihani, S.M.S. Structural evolution during mechanical milling of nanometric and micrometric Al2O3 reinforced Al matrix composites. Mater. Sci. Eng. A 2006, 428, 159–168. [Google Scholar] [CrossRef]
- Slipenyuk, A.; Kuprin, V.; Milman, Y.; Goncharuk, V.; Eckert, J. Properties of P/M processed particle reinforced metal matrix composites specified by reinforcement concentration and matrix-to-reinforcement particle size ratio. Acta Mater. 2006, 54, 157–166. [Google Scholar] [CrossRef]
- Zhang, S.L.; Zhao, Y.T.; Chen, G.; Cheng, X.N.; She, C.J.; Wang, X.Y.; Wu, D.N. Effects of in situ TiB2 particle on microstructures and mechanical properties of AZ91 alloy. J. Alloys Compd. 2010, 494, 94–97. [Google Scholar] [CrossRef]
- Yu, H.; Sun, Y.; Hu, L.; Wan, Z.; Zhou, H. The effect of Ti addition on microstructure evolution of AZ61 Mg alloy during mechanical milling. J. Alloys Compd. 2017, 704, 537–544. [Google Scholar] [CrossRef]
- Mazilkin, A.A.; Straumal, B.B.; Kilmametov, A.R.; Boll, T.; Baretzky, B.; Kogtenkova, O.A.; Korneva, A.; Zieba, P. Competition for impurity atoms between defects and solid solution during high pressure torsion. Scr. Mater. 2019, 173, 46–50. [Google Scholar] [CrossRef]
- Ivanisenko, Y.; Sauvage, X.; Mazilkin, A.A.; Kilmametov, A.R.; Beach, J.A.; Straumal, B.B. Bulk Nanocrystalline Ferrite Stabilized through Grain Boundary Carbon Segregation. Adv. Eng. Mater. 2018, 20, 1800443. [Google Scholar] [CrossRef]
- Straumal, B.B.; Mazilkin, A.A.; Protasova, S.G.; Myatiev, A.A.; Straumal, P.B.; Baretzky, B. Increase of Co solubility with decreasing grain size in ZnO. Acta Mater. 2008, 56, 6246–6256. [Google Scholar] [CrossRef]
- Su, S.; Zhou, J.; Tang, S.; Yu, H.; Su, Q.; Zhang, S. Synthesis of Nanocrystalline AZ91 Magnesium Alloy Dispersed with 15 vol.% Submicron SiC Particles by Mechanical Milling. Materials 2019, 12, 901. [Google Scholar] [CrossRef]
- Schuh, C.A.; Nieh, T.G.; Iwasaki, H. The effect of solid solution W additions on the mechanical properties of nanocrystalline Ni. Acta Mater. 2003, 51, 431–443. [Google Scholar] [CrossRef]
- Goh, C.S.; Wei, J.; Lee, L.C.; Gupta, M. Properties and deformation behaviour of Mg–Y2O3 nanocomposites. Acta Mater. 2007, 55, 5115–5121. [Google Scholar] [CrossRef]
- Silva, C.L.P.; Soares, R.B.; Pereira, P.H.R.; Figueiredo, R.B.; Lins, V.F.C.; Langdon, T.G. The Effect of High-Pressure Torsion on Microstructure, Hardness and Corrosion Behavior for Pure Magnesium and Different Magnesium Alloys. Adv. Eng. Mater. 2019, 21, 1801081. [Google Scholar] [CrossRef]
- Saravanan, M.; Sivaiah, B.; Srivastava, A.K.; Dhar, A. Ultrafine grain structure features in spray-formed AZ31 magnesium alloy. Mater. Des. 2014, 60, 21–25. [Google Scholar] [CrossRef]
- Chen, Q.; Shu, D.; Hu, C.; Zhao, Z.; Yuan, B. Grain refinement in an as-cast AZ61 magnesium alloy processed by multi-axial forging under the multitemperature processing procedure. Mater. Sci. Eng. A 2012, 541, 98–104. [Google Scholar] [CrossRef]
- Shanthi, M.; Jayaramanavar, P.; Vyas, V.; Seenivasan, D.V.S.; Gupta, M. Effect of niobium particulate addition on the microstructure and mechanical properties of pure magnesium. J. Alloys Compd. 2012, 513, 202–207. [Google Scholar] [CrossRef]
- Han, G.; Wang, Z.; Liu, K.; Li, S.; Du, X.; Du, W. Synthesis of CNT-reinforced AZ31 magnesium alloy composites with uniformly distributed CNTs. Mater. Sci. Eng. A 2015, 628, 350–357. [Google Scholar] [CrossRef]
- Penther, D.; Ghasemi, A.; Riedel, R.; Fleck, C.; Kamrani, S. Effect of SiC nanoparticles on manufacturing process, microstructure and hardness of Mg-SiC nanocomposites produced by mechanical milling and hot extrusion. Mater. Sci. Eng. A 2018, 738, 264–272. [Google Scholar] [CrossRef]
- Lee, C.J.; Huang, J.C.; Hsieh, P.J. Mg based nano-composites fabricated by friction stir processing. Scr. Mater. 2006, 54, 1415–1420. [Google Scholar] [CrossRef]
- Nguyen, Q.B.; Gupta, M. Increasing significantly the failure strain and work of fracture of solidification processed AZ31B using nano-Al2O3 particulates. J. Alloys Compd. 2008, 459, 244–250. [Google Scholar] [CrossRef]
- Seenuvasaperumal, P.; Doi, K.; Basha, D.A.; Singh, A.; Elayaperumal, A.; Tsuchiya, K. Wear behavior of HPT processed UFG AZ31B magnesium alloy. Mater. Lett. 2018, 227, 194–198. [Google Scholar] [CrossRef]
- Li, R.; Xin, R.; Liu, Q.; Chapuis, A.; Liu, S.; Fu, G.; Zong, L. Effect of grain size, texture and density of precipitates on the hardness and tensile yield stress of Mg-14Gd-0.5Zr alloys. Mater. Des. 2017, 114, 450–458. [Google Scholar] [CrossRef]






© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, H.; Zhang, C.; Han, B.; Qiu, J.; Qin, S.; Gao, K.; Liu, J.; Sun, S.; Zhang, H. Microstructures and Mechanical Properties of Nanocrystalline AZ31 Magnesium Alloy Powders with Submicron TiB2 Additions Prepared by Mechanical Milling. Crystals 2020, 10, 550. https://doi.org/10.3390/cryst10060550
Zhou H, Zhang C, Han B, Qiu J, Qin S, Gao K, Liu J, Sun S, Zhang H. Microstructures and Mechanical Properties of Nanocrystalline AZ31 Magnesium Alloy Powders with Submicron TiB2 Additions Prepared by Mechanical Milling. Crystals. 2020; 10(6):550. https://doi.org/10.3390/cryst10060550
Chicago/Turabian StyleZhou, Haiping, Chengcai Zhang, Baokun Han, Jianfeng Qiu, Shengxue Qin, Kuidong Gao, Jie Liu, Shuai Sun, and Hongbin Zhang. 2020. "Microstructures and Mechanical Properties of Nanocrystalline AZ31 Magnesium Alloy Powders with Submicron TiB2 Additions Prepared by Mechanical Milling" Crystals 10, no. 6: 550. https://doi.org/10.3390/cryst10060550
APA StyleZhou, H., Zhang, C., Han, B., Qiu, J., Qin, S., Gao, K., Liu, J., Sun, S., & Zhang, H. (2020). Microstructures and Mechanical Properties of Nanocrystalline AZ31 Magnesium Alloy Powders with Submicron TiB2 Additions Prepared by Mechanical Milling. Crystals, 10(6), 550. https://doi.org/10.3390/cryst10060550