Carbothermal Reduction Nitridation of Fly Ash, Diatomite and Raw Illite: Formation of Nitride Powders with Different Morphology and Photoluminescence Properties
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
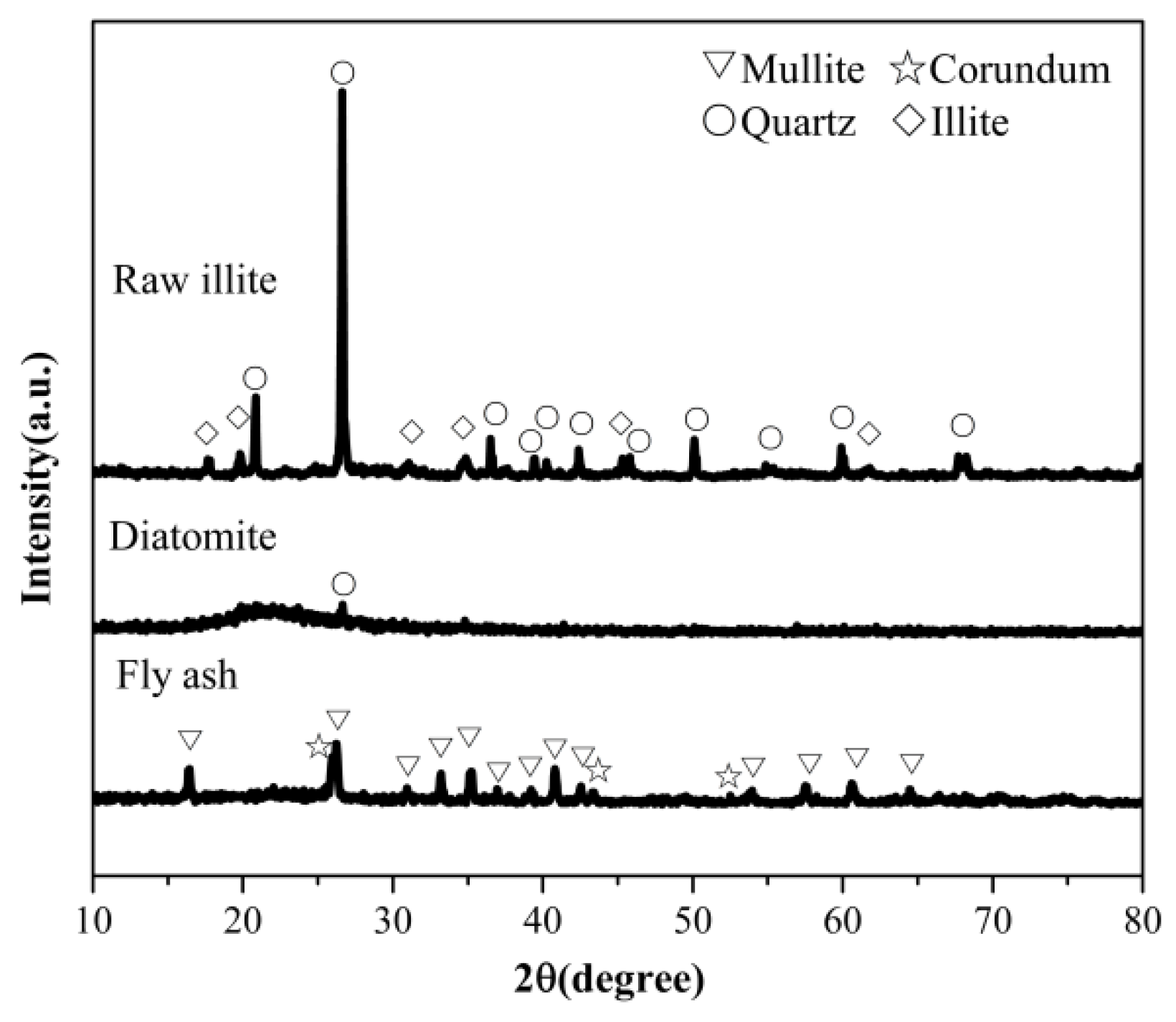
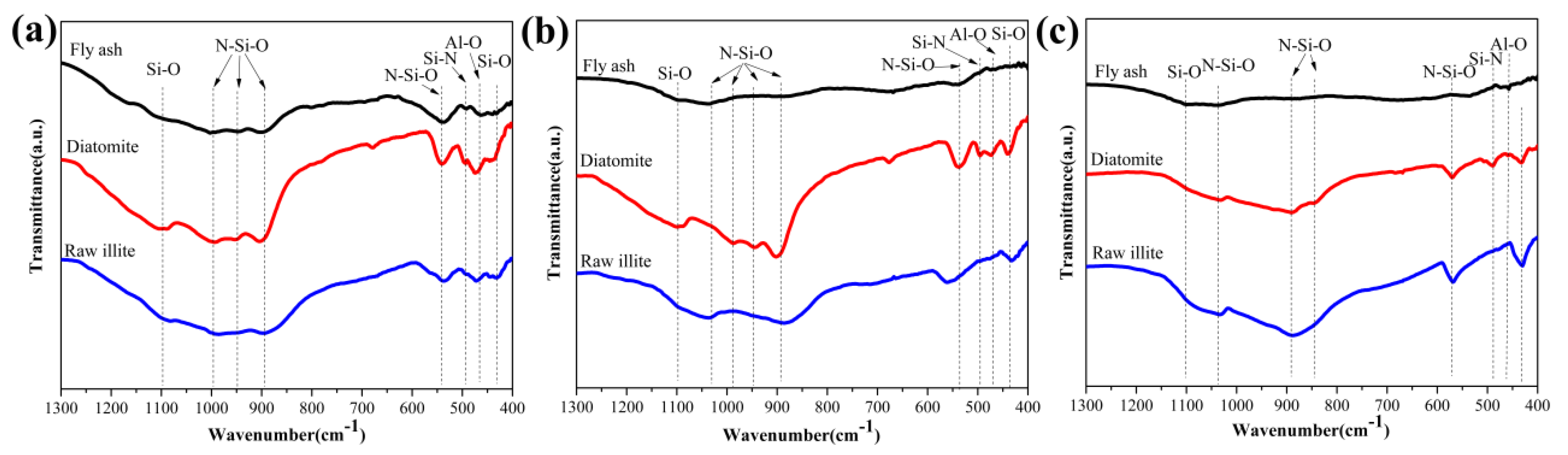
3.1. Analysis of Experimental Raw Materials
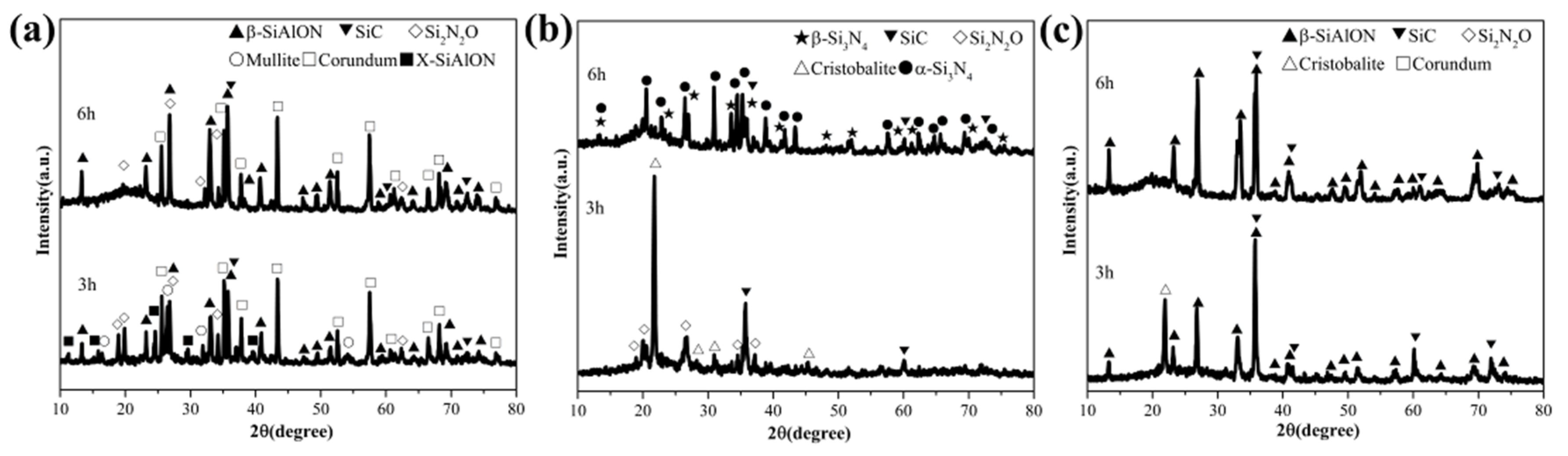
3.2. The Effect of the Si Sources on the Phase Composition of Products
3.3. The Effect of the Holding Time on the Phase Composition of Products
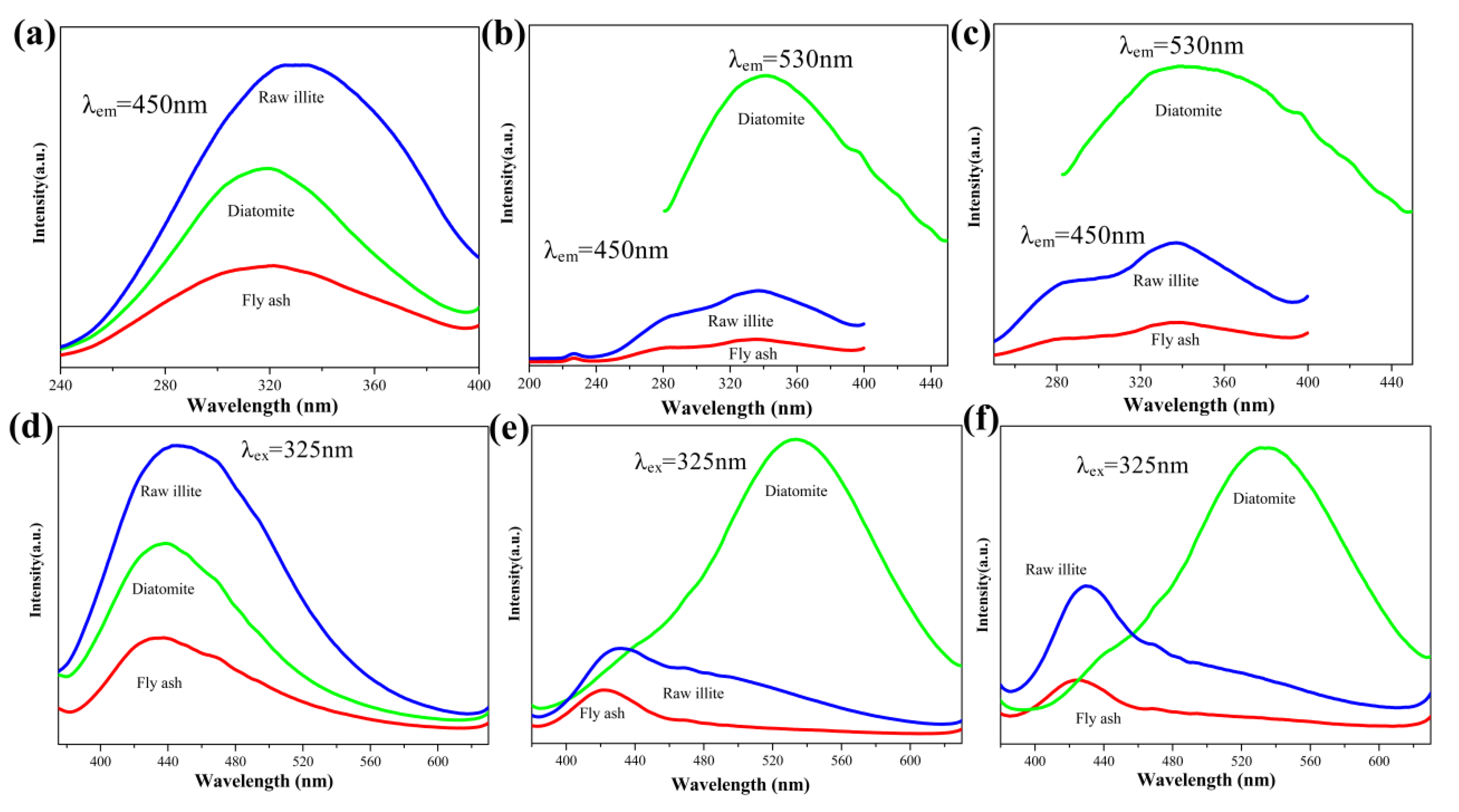
3.4. The Effect of the Si Sources on the Photoluminescence Properties of Products
3.5. Product Morphology
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tanaka, I.; Nasu, S.; Adachi, H.; Miyamoto, Y.; Niihara, K. Electronic structure behind the mechanical properties of β-sialons. Acta Metall. Mater. 1992, 40, 1995–2001. [Google Scholar] [CrossRef]
- Izhevskiy, V.A.; Genova, L.A.; Bressiani, J.C.; Aldinger, F. Progress in SiAlON ceramics. J. Eur. Ceram. Soc. 2000, 20, 2275–2295. [Google Scholar] [CrossRef]
- Xie, R.J.; Hirosaki, N.; Mitomo, M.; Suehiro, T.; Xu, X.; Tanaka, H. Photoluminescence of rare-earth-doped Ca-α-SiAlON phosphors: Composition and concentration dependence. J. Am. Ceram. Soc. 2005, 88, 2883–2888. [Google Scholar] [CrossRef]
- Xianqing, P.; Machida, K.; Horikawa, T.; Hanzawa, H. Synthesis and luminescent properties of low oxygen contained Eu2+-doped Ca-α-SiAlON phosphor from calcium cyanamide reduction. J. Rare Earth 2008, 26, 198–202. [Google Scholar]
- Suehiro, T.; Onuma, H.; Hirosaki, N.; Xie, R.J.; Sato, T.; Miyamoto, A. Powder synthesis of Y-α-SiAlON and its potential as a phosphor host. J. Phys. Chem. C 2010, 114, 1337–1342. [Google Scholar] [CrossRef]
- Ryu, J.H.; Park, Y.G.; Won, H.S.; Suzuki, H.; Kim, S.H.; Yoon, C. Luminescent properties of β-SiAlON: Eu2+ green phosphors synthesized by gas pressured sintering. J. Ceram. Soc. Jpn. 2008, 116, 389–394. [Google Scholar] [CrossRef][Green Version]
- Ryu, J.H.; Won, H.S.; Park, Y.G.; Kim, S.H.; Song, W.Y.; Suzuki, H.; Yoon, C.B.; Kim, D.H.; Park, W.J.; Yoon, C. Photoluminescence of Ce3+-activated β-SiAlON blue phosphor for UV-LED. Electrochem. Solid-State Lett. 2010, 13, H30–H32. [Google Scholar] [CrossRef]
- Wang, Z.; Ye, W.; Chu, I.H.; Ong, S.P. Elucidating structure-composition-property relationships of the β-SiAlON: Eu2+ phosphor. Chem. Mater. 2016, 28, 8622–8630. [Google Scholar] [CrossRef]
- Zhu, X.W.; Masubuchi, Y.; Motohashi, T.; Kikkawa, S. The z value dependence of photoluminescence in Eu2+-doped β-SiAlON (Si6-zAlzOzN8-z) with 1 ≤ z ≤ 4. J. Alloy. Compd. 2010, 489, 157–161. [Google Scholar] [CrossRef]
- Ryu, J.H.; Park, Y.G.; Won, H.S.; Kim, S.H.; Suzuki, H.; Yoon, C. Luminescence properties of Eu2+-doped β-Si6-zAlzOzN8-z microcrystals fabricated by gas pressured reaction. J. Cryst. Growth 2009, 311, 878–882. [Google Scholar] [CrossRef]
- Li, Y.Q.; Hirosaki, N.; Xie, R.J.; Takeda, T.; Mitomo, M. Crystal and electronic structures, luminescence properties of Eu2+-doped Si6-zAlzOzN8-z and My Si6-zAlz-yOz+yN8-z-y (M = 2Li, Mg, Ca, Sr, Ba). J. Solid State Chem. 2008, 181, 3200–3210. [Google Scholar] [CrossRef]
- Van Krevel, J.W.H.; van Rutten, J.W.T.; Mandal, H.; Hintzen, H.T.; Metselaar, R. Luminescence properties of terbium-, cerium-, or europium-doped α-Sialon materials. J. Solid State Chem. 2002, 165, 19–24. [Google Scholar] [CrossRef]
- Nekouee, K.A.; Khosroshahi, R.A. Preparation and characterization of β-SiAlON/TiN nanocomposites sintered by spark plasma sintering and pressureless sintering. Mater. Des. 2016, 112, 419–428. [Google Scholar] [CrossRef]
- Jiang, P.; Wu, X.F.; Xue, W.D.; Chen, J.H.; Wang, W.; Li, Y. In-situ synthesis and reaction mechanism of β-SiAlON in the Al-Si3N4-Al2O3 composite material. Ceram. Int. 2017, 43, 1335–1340. [Google Scholar] [CrossRef]
- Yi, X.M.; Niu, J.; Nakamura, T.; Akiyama, T. Reaction mechanism for combustion synthesis of β-SiAlON by using Si, Al, and SiO2 as raw materials. J. Alloys Compd. 2013, 561, 1–4. [Google Scholar] [CrossRef]
- He, E.Q.; Yue, J.S.; Fan, L.; Wang, C.; Wang, H.J. Synthesis of single phase β-SiAlON ceramics by reaction-bonded sintering using Si and Al2O3 as raw materials. Scr. Mater. 2011, 65, 155–158. [Google Scholar] [CrossRef]
- Jun, K.; Lee, K.; Kim, G.; Kim, Y.J. A new route for the synthesis of β-sialon: Eu2+ phosphors using pyrophyllite powders. Ceram. Int. 2013, 39, S349–S353. [Google Scholar] [CrossRef]
- O’Leary, B.G.; MacKenzie, K.J.D. Inorganic polymers (geopolymers) as precursors for carbothermal reduction and nitridation (CRN) synthesis of SiAlON ceramics. J. Eur. Ceram. Soc. 2015, 35, 2755–2764. [Google Scholar] [CrossRef]
- Bahramian, A.R.; Kokabi, M. Carbonitriding synthesis of β-SiAlON nanopowder from kaolinite–polyacrylamide precursor. Appl. Clay Sci. 2011, 52, 407–413. [Google Scholar] [CrossRef]
- Tatlı, Z.; Demir, A.; Yılmaz, R.; Çalışkan, F.; Kurt, A.O. Effects of processing parameters on the production of β-SiAlON powder from kaolinite. J. Eur. Ceram. Soc. 2007, 27, 743–747. [Google Scholar] [CrossRef]
- Kudyba-Jansen, A.A.; Hintzen, H.T.; Metselaar, R. Ca-α/β-sialon ceramics synthesised from fly ash-preparation, characterization and properties. Mater. Res. Bull. 2001, 36, 1215–1230. [Google Scholar] [CrossRef]
- Zhao, H.; Wang, P.Y.; Yu, J.L.; Zhang, J. A mechanistic study on the synthesis of β-Sialon whiskers from coal fly ash. Mater. Res. Bull. 2015, 65, 47–52. [Google Scholar] [CrossRef]
- Ma, B.Y.; Li, Y.; Yan, C.; Ding, Y.S. Effects of synthesis temperature and raw materials composition on preparation of β-Sialon based composites from fly ash. Trans. Nonferr. Met. Soc. China 2012, 22, 129–133. [Google Scholar] [CrossRef]
- Gilbert, J.E.; Mosset, A. Preparation of β-SiAlON from fly ashes. Mater. Res. Bull. 1998, 33, 117–123. [Google Scholar] [CrossRef]
- Dou, K.Z.; Jiang, Y.S.; Xue, B.; Wei, C.D.; Li, F.F. The carbon environment effects on phase composition and photoluminescence properties of β-SiAlON multiphase materials prepared from fly ash acid slag. Ceram. Int. 2019, 45, 7850–7856. [Google Scholar] [CrossRef]
- Seidel, A.; Sluszny, A.; Shelef, G.; Zimmels, Y. Self inhibition of aluminum leaching from coal fly ash by sulfuric acid. Chem. Eng. J. 1999, 72, 195–207. [Google Scholar] [CrossRef]
- Matjie, R.H.; Bunt, J.R.; van Heerden, J.H.P. Extraction of alumina from coal fly ash generated from a selected low rank bituminous South African coal. Miner. Eng. 2005, 18, 299–310. [Google Scholar] [CrossRef]
- Jiang, T.; Xue, X.X.; Duan, P.N.; Liu, X.; Zhang, S.H.; Liu, R. Carbothermal reduction–nitridation of titania-bearing blast furnace slag. Ceram. Int. 2008, 34, 1643–1651. [Google Scholar] [CrossRef]
- Tang, Y.; Yin, H.F.; Yuan, H.D.; Shuai, H.; Xin, Y.L. Carbothermal reduction nitridation of slag, glass and minerals: Formation process of SiAlON powders with different morphology. Ceram. Int. 2016, 42, 7499–7505. [Google Scholar] [CrossRef]
- Hotta, M.; Tatami, J.; Zhang, C.; Komeya, K.; Meguro, T.; Terner, M.R.; Cheng, Y.B. Ca-α SiAlON hollow spheres prepared by carbothermal reduction–nitridation from different SiO2 powders. Ceram. Int. 2010, 36, 1553–1559. [Google Scholar] [CrossRef]
- Panda, P.K.; Mariappan, L.; Kannan, T.S. Carbothermal reduction of kaolinite under nitrogen atmosphere. Ceram. Int. 2000, 26, 455–461. [Google Scholar] [CrossRef]
- Antsiferov, V.N.; Gilyov, V.G.; Karmanov, V.I. IR-spectra and phases structure of sialons. Vib. Spectrosc. 2002, 30, 169–173. [Google Scholar] [CrossRef]
- Wang, F.; Jin, G.; Guo, X. Formation Mechanism of Si3N4 Nanowires via Carbothermal Reduction of Carbonaceous Silica Xerogels. J. Phys. Chem. B 2006, 110, 14546–14549. [Google Scholar] [CrossRef] [PubMed]
- Yariv, S.; Lapides, I.; Nasser, A.; Lahav, N.; Brodsky, I.; Michaelian, K.H. Infrared study of the intercalation of potassium halides in kaolinite. Clays Clay Miner. 2000, 48, 10–18. [Google Scholar] [CrossRef]
- Yu, F.; Yang, J.; Delsing, A.; Hintzen, B. Preparation, characterization and luminescence properties of porous Si3N4 ceramics with Eu2O3 as sintering additive. J. Lumin. 2010, 130, 2298–2304. [Google Scholar] [CrossRef]


| Composition | Fly Ash | Diatomite | Raw Illite |
|---|---|---|---|
| SiO2 | 40.25 | 87.85 | 70.41 |
| Al2O3 | 49.61 | 2.40 | 16.15 |
| TiO2 | 1.98 | 0.11 | 0.39 |
| CaO | 1.70 | 0.29 | 1.09 |
| MgO | 0.22 | 0.34 | 0.61 |
| Fe2O3 | 1.36 | 1.02 | 1.03 |
| K2O | 0.38 | 0.43 | 5.15 |
| Na2O | 0.07 | 0.17 | 0.03 |
| LOI | 3.27 | 7.37 | 3.50 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dou, K.; Jiang, Y.; Xue, B.; Wei, C.; Li, F. Carbothermal Reduction Nitridation of Fly Ash, Diatomite and Raw Illite: Formation of Nitride Powders with Different Morphology and Photoluminescence Properties. Crystals 2020, 10, 409. https://doi.org/10.3390/cryst10050409
Dou K, Jiang Y, Xue B, Wei C, Li F. Carbothermal Reduction Nitridation of Fly Ash, Diatomite and Raw Illite: Formation of Nitride Powders with Different Morphology and Photoluminescence Properties. Crystals. 2020; 10(5):409. https://doi.org/10.3390/cryst10050409
Chicago/Turabian StyleDou, Kuizhou, Yinshan Jiang, Bing Xue, Cundi Wei, and Fangfei Li. 2020. "Carbothermal Reduction Nitridation of Fly Ash, Diatomite and Raw Illite: Formation of Nitride Powders with Different Morphology and Photoluminescence Properties" Crystals 10, no. 5: 409. https://doi.org/10.3390/cryst10050409
APA StyleDou, K., Jiang, Y., Xue, B., Wei, C., & Li, F. (2020). Carbothermal Reduction Nitridation of Fly Ash, Diatomite and Raw Illite: Formation of Nitride Powders with Different Morphology and Photoluminescence Properties. Crystals, 10(5), 409. https://doi.org/10.3390/cryst10050409




