Twist-Bend Nematogenic Supramolecular Dimers and Trimers Formed by Hydrogen Bonding
Abstract
1. Introduction
2. Materials and Methods
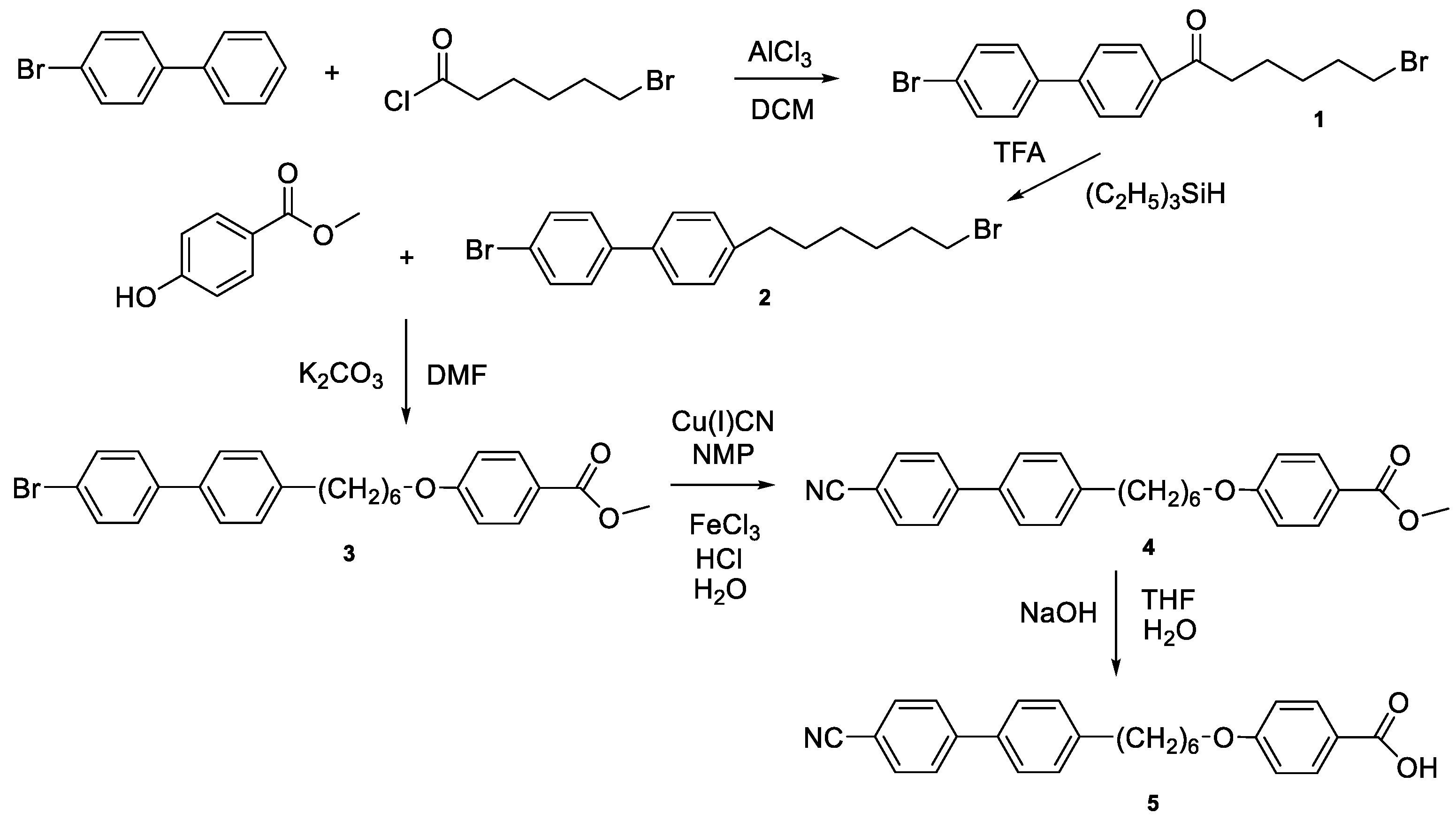
2.1. Synthesis 4-[6-(4’-Cyanobiphenyl-4-yl)Hexyloxy]Benzoic Acid (CB6OBA)
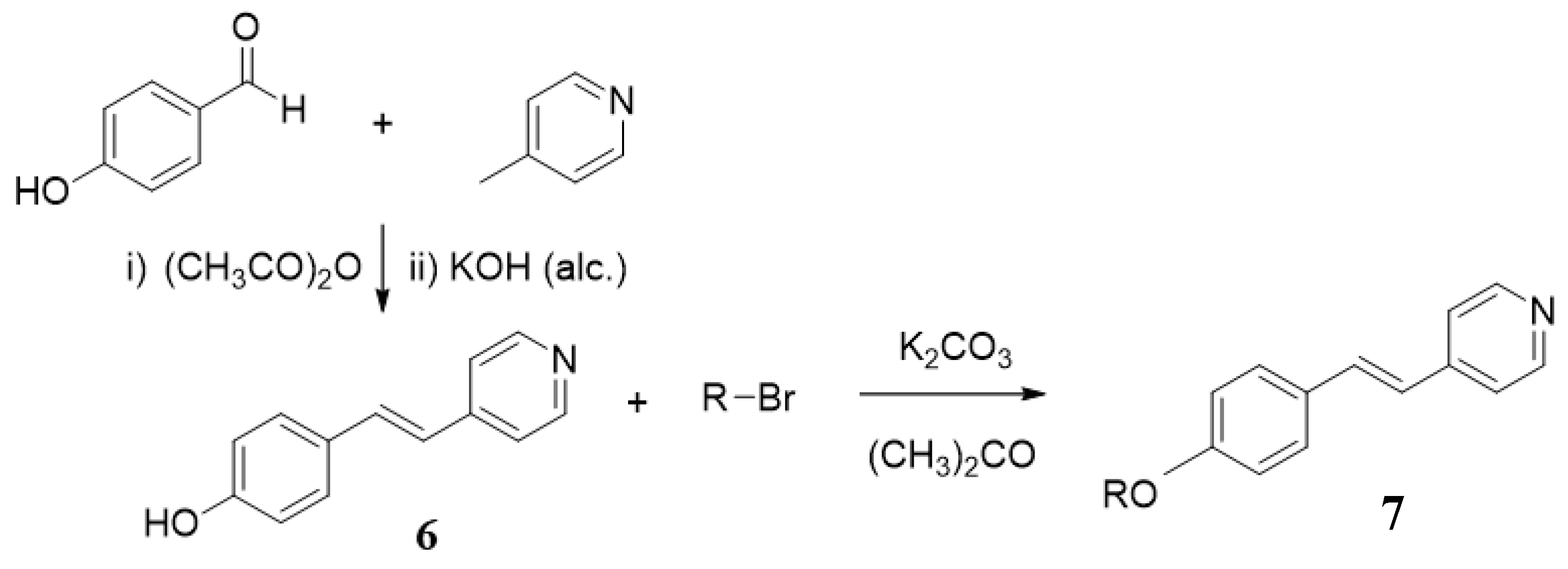
2.2. Synthesis 4-[(E)-2-(4-Alkoxyphenyl)Ethenyl]Pyridines, nOS
2.3. Binary Mixtures
2.4. Characterisation
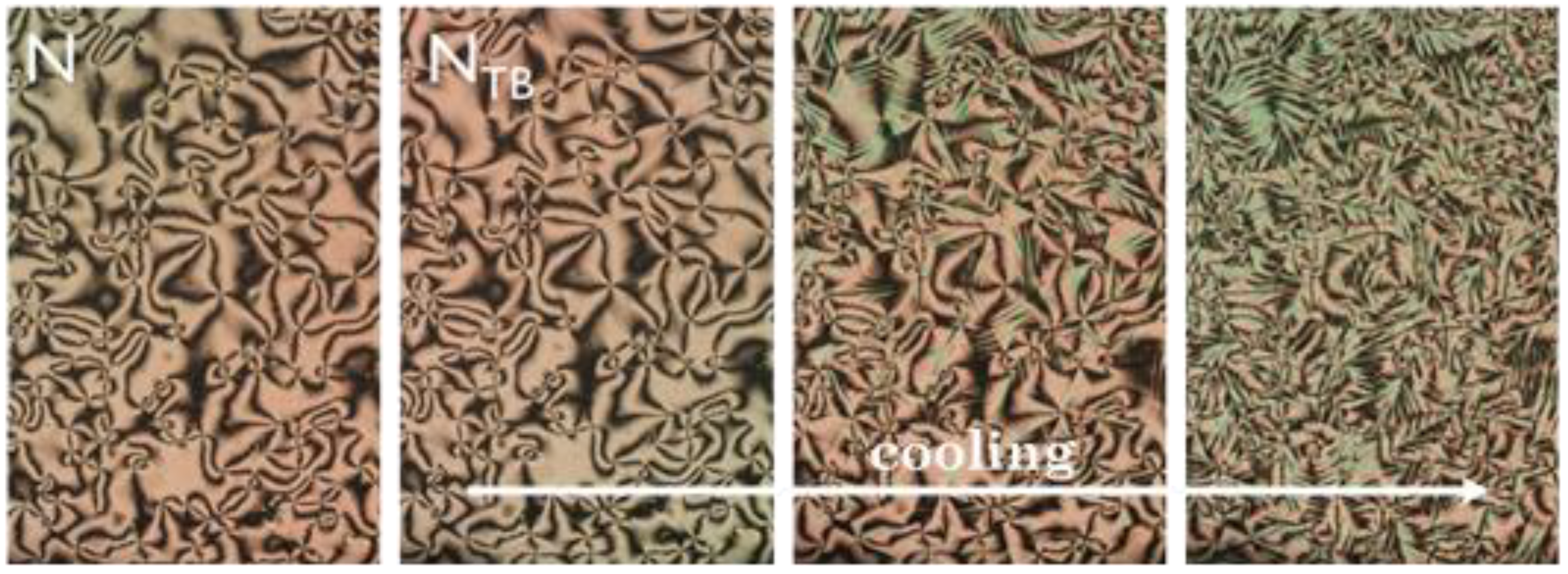
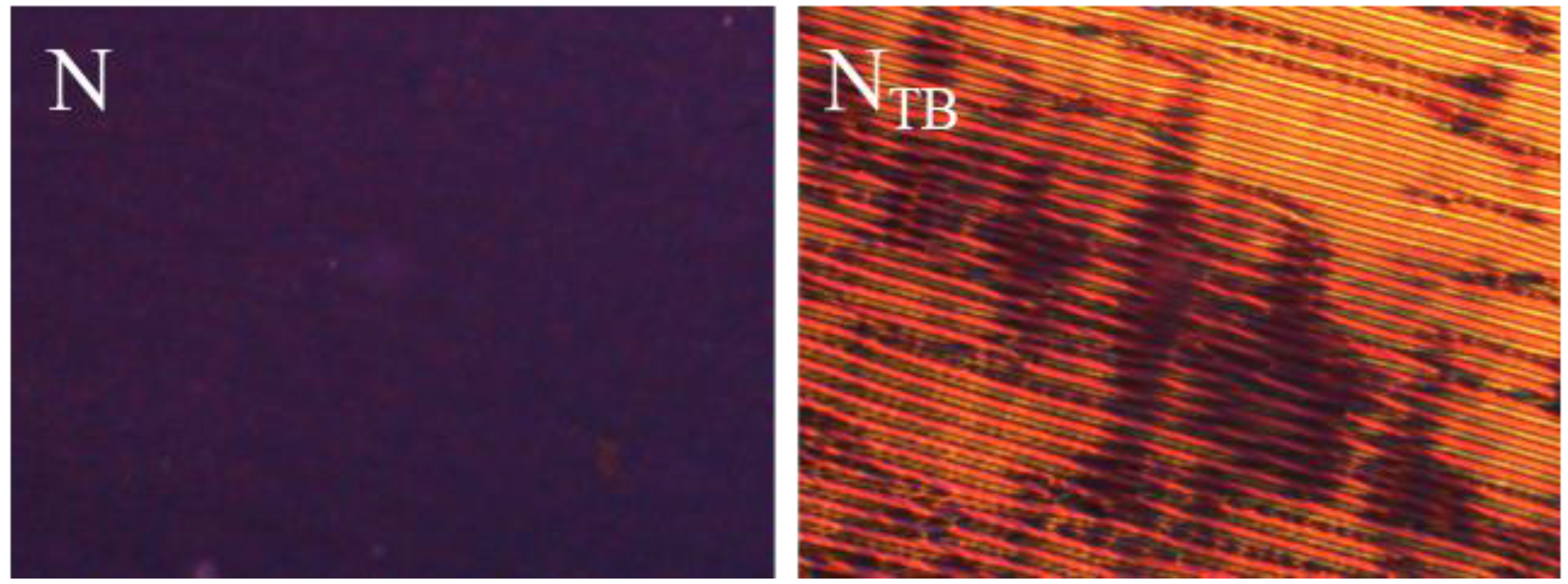
2.4.1. Polarised Optical Microscopy (POM)
2.4.2. Differential Scanning Calorimetry (DSC)
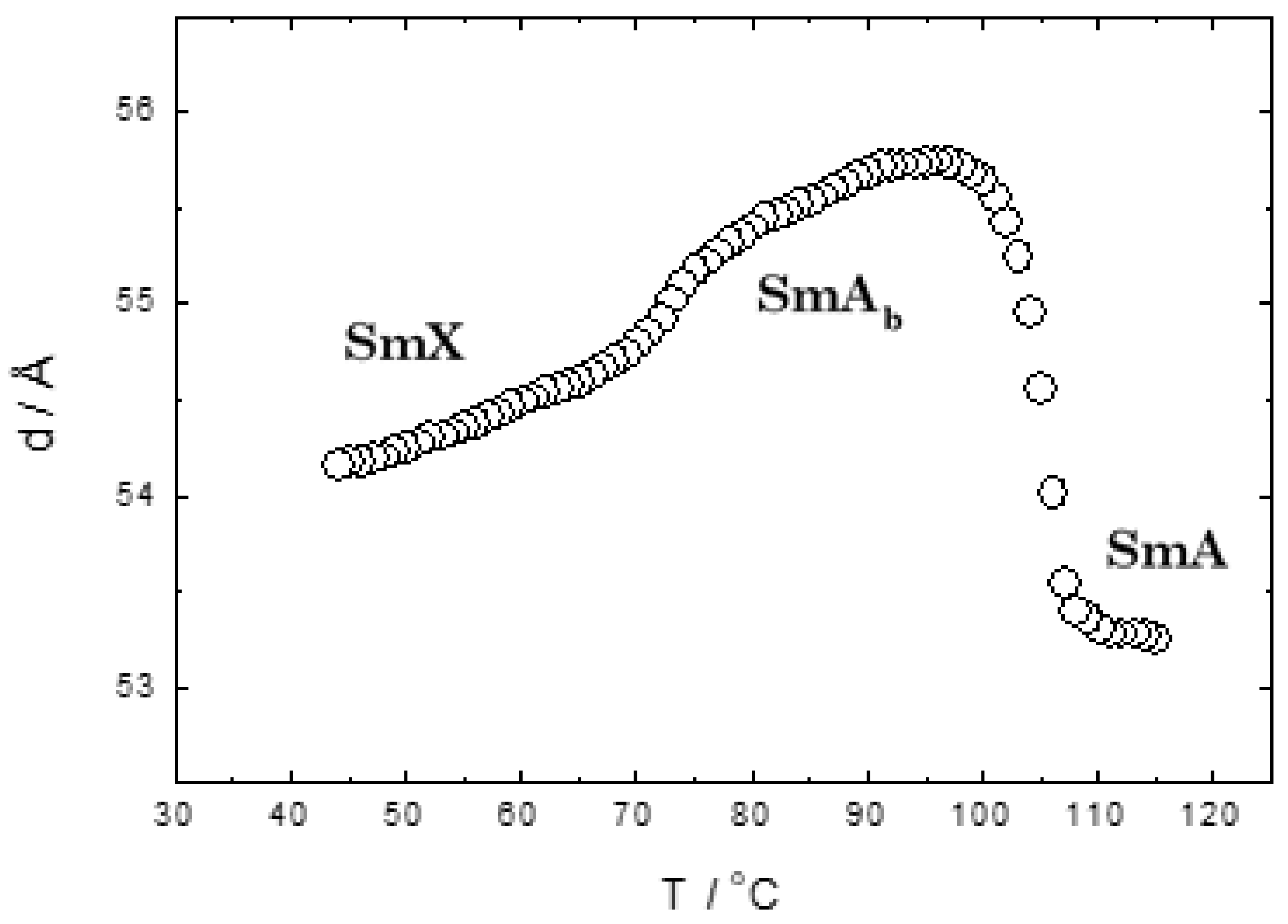
2.4.3. X-Ray Diffraction (XRD)
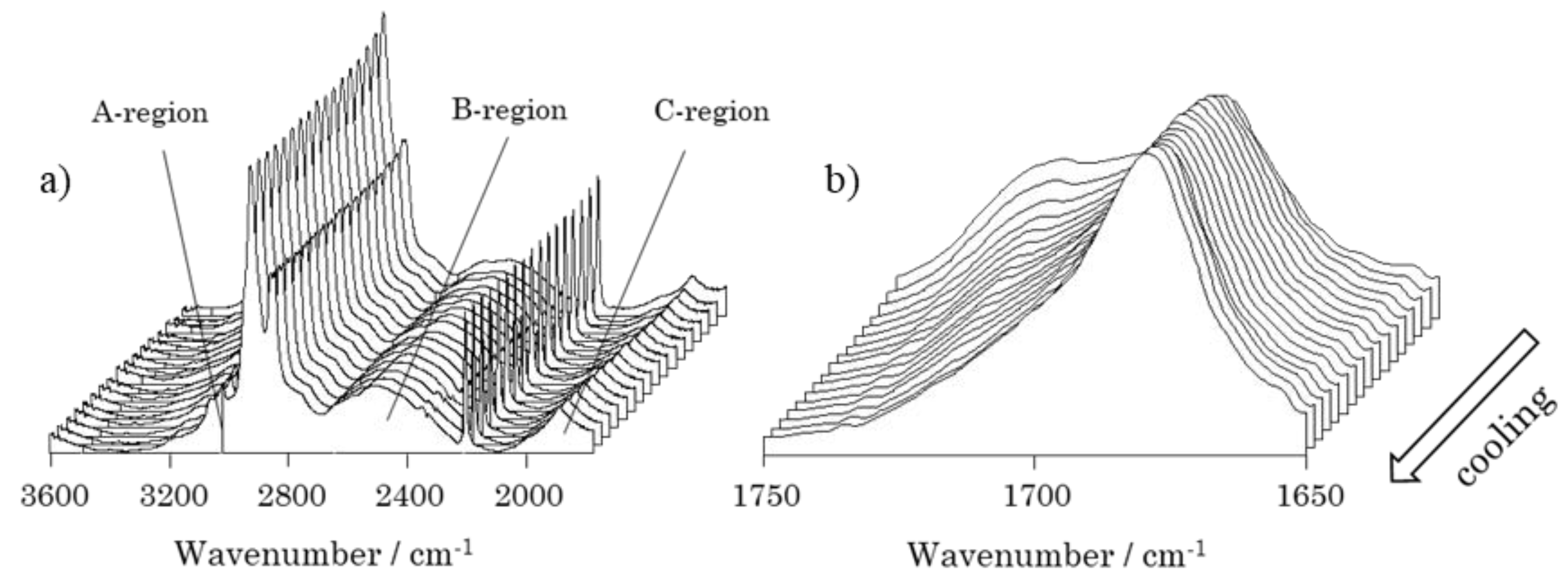
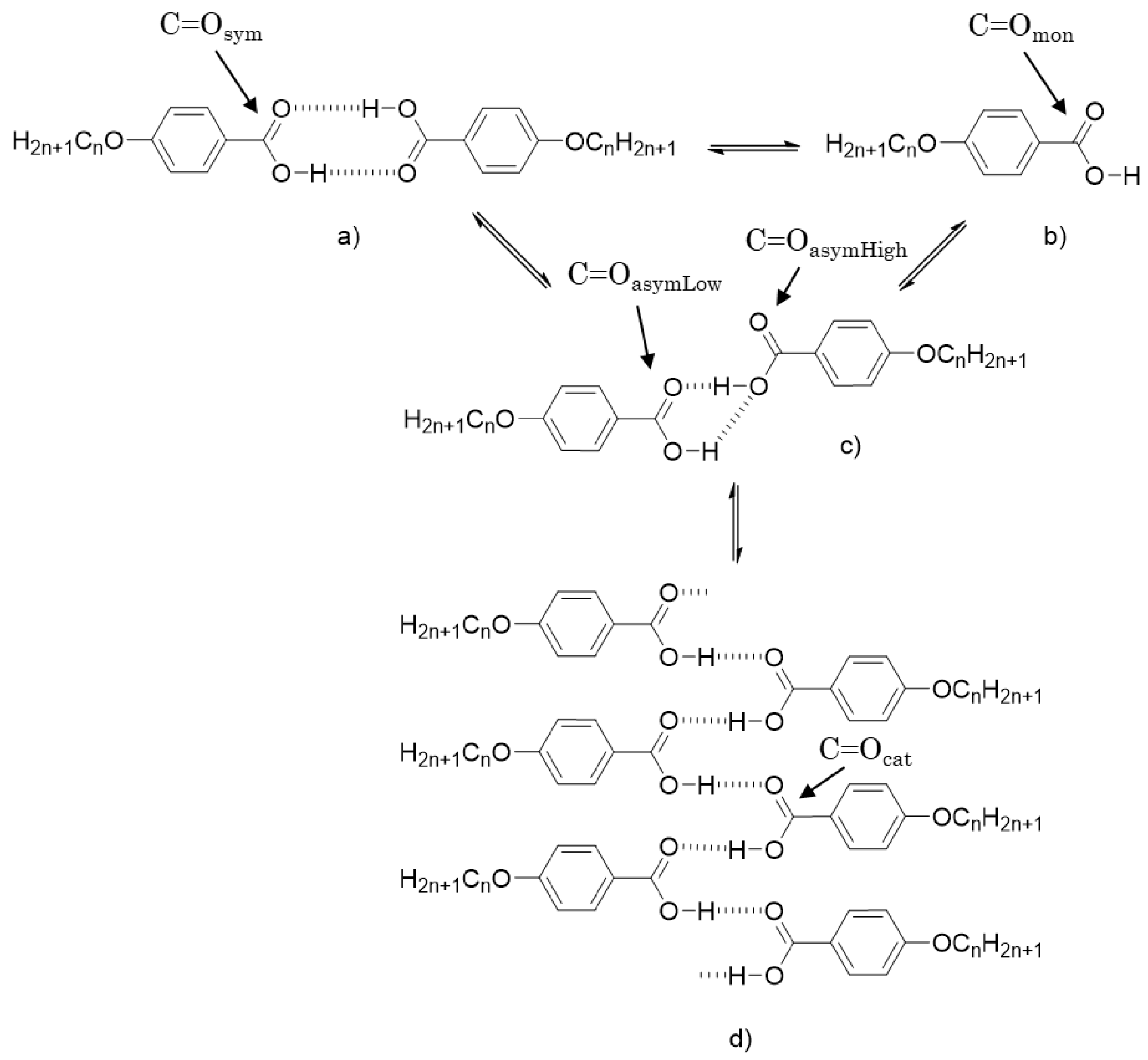
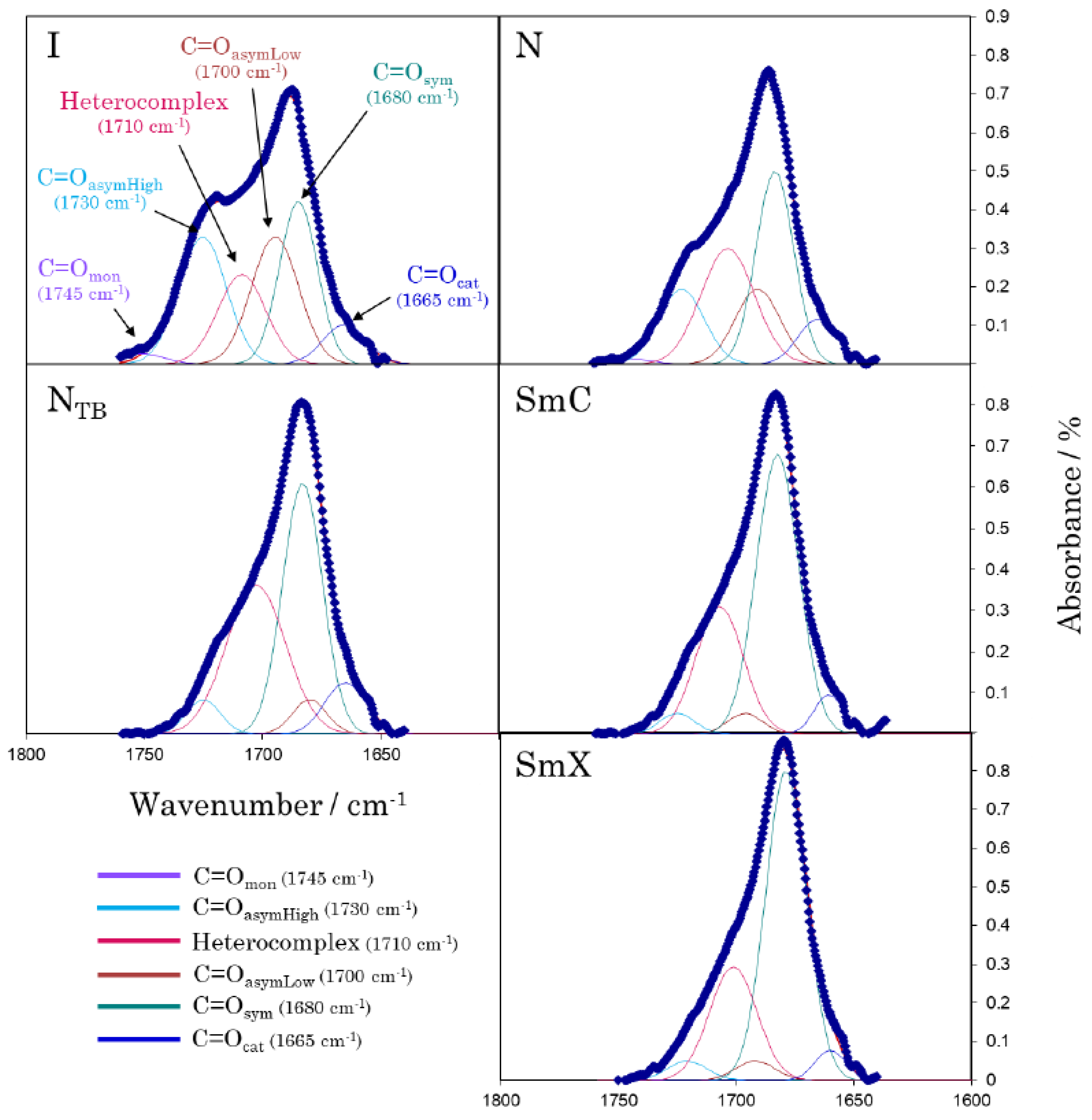
2.4.4. Temperature-dependent Fourier Transform Infrared Spectroscopy
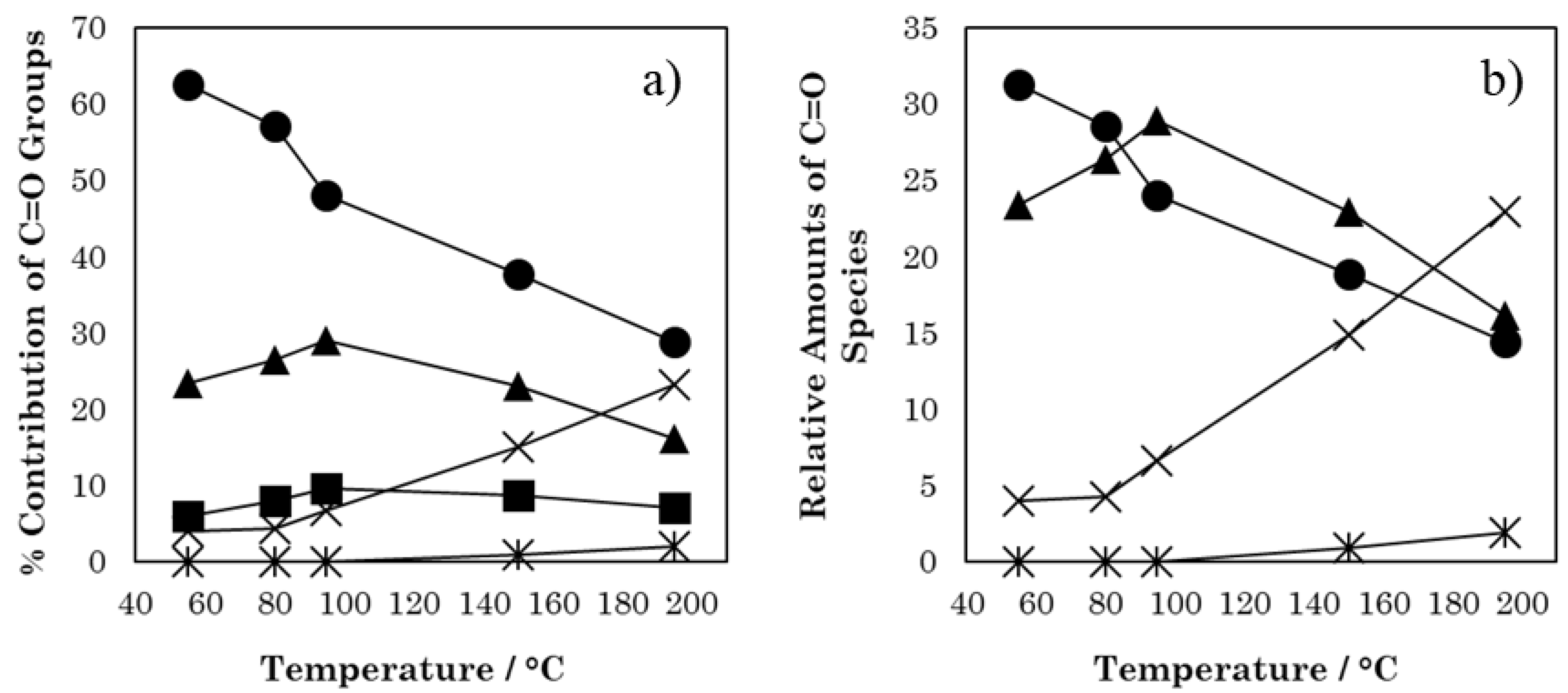
3. Results
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cestari, M.; Diez-Berart, S.; Dunmur, D.A.; Ferrarini, A.; de la Fuente, M.R.; Jackson, D.J.; Lopez, D.O.; Luckhurst, G.R.; Perez-Jubindo, M.A.; Richardson, R.M.; et al. Phase behavior and properties of the liquid-crystal dimer 1′′,7′′-bis(4-cyanobiphenyl-4′-yl) heptane: A twist-bend nematic liquid crystal. Phys. Rev. E Stat. Nonlinear Soft Matter Phys. 2011, 84, 031704. [Google Scholar] [CrossRef]
- Meyer, R.B. Structural problems in liquid crystal physics. In Les Houches Summer School in Theoretical Physics; Molecular Fluids: Gordon and Bearch, New York, NY, USA, 1976; pp. 316–320. [Google Scholar]
- Dozov, I. On the spontaneous symmetry breaking in the mesophases of achiral banana-shaped molecules. Europhys. Lett. 2001, 56, 247–253. [Google Scholar] [CrossRef]
- Borshch, V.; Kim, Y.-K.; Xiang, J.; Gao, M.; Jákli, A.; Panov, V.P.; Vij, J.K.; Imrie, C.T.; Tamba, M.G.; Mehl, G.H.; et al. Nematic twist-bend phase with nanoscale modulation of molecular orientation. Nat. Commun. 2013, 4, 2635. [Google Scholar] [CrossRef]
- Walker, R.; Pociecha, D.; Storey, J.; Gorecka, E.; Imrie, C.T. The Chiral Twist-Bend Nematic Phase (N*TB). Chem. A Eur. J. 2019, 25, 13329–13335. [Google Scholar] [CrossRef]
- Walker, R.; Pociecha, D.; Strachan, G.; Storey, J.M.D.; Gorecka, E.; Imrie, C.T. Molecular curvature, specific intermolecular interactions and the twist-bend nematic phase: The synthesis and characterisation of the 1-(4-cyanobiphenyl-4′-yl)-6-(4-alkylanilinebenzylidene-4′-oxy)hexanes (CB6O.m). Soft Matter 2019, 15, 3188–3197. [Google Scholar] [CrossRef]
- Salamończyk, M.; Vaupotič, N.; Pociecha, D.; Walker, R.; Storey, J.M.D.; Imrie, C.T.; Wang, C.; Zhu, C.; Gorecka, E. Multi-level chirality in liquid crystals formed by achiral molecules. Nat. Commun. 2019, 10, 1922. [Google Scholar] [CrossRef]
- Abberley, J.P.; Killah, R.; Walker, R.; John, M.D.; Storey, C.T.; Imrie, M.S.; Zhu, C.; Gorecka, E.; Damian, P. Heliconical smectic phases formed by achiral molecules. Nat. Commun. 2018, 9, 228. [Google Scholar] [CrossRef]
- Imrie, C.T.; Henderson, P.A. Liquid crystal dimers and higher oligomers: Between monomers and polymers. Chem. Soc. Rev. 2007, 36, 2096–2124. [Google Scholar] [CrossRef]
- Imrie, C.T.; Henderson, P.; Yeap, G.-Y. Liquid crystal oligomers: Going beyond dimers. Liq. Cryst. 2009, 36, 755–777. [Google Scholar] [CrossRef]
- Paterson, D.A.; Abberley, J.P.; Harrison, W.T.A.; Storey, J.; Imrie, C.T. Cyanobiphenyl-based liquid crystal dimers and the twist-bend nematic phase. Liq. Cryst. 2017, 44, 127–146. [Google Scholar] [CrossRef]
- Mandle, R.J.; Davis, E.; Voll, C.-C.A.; Archbold, C.; Goodby, J.; Cowling, S. The relationship between molecular structure and the incidence of the NTB phase. Liq. Cryst. 2015, 42, 688–703. [Google Scholar]
- Lu, Z.; Henderson, P.A.; Paterson, B.J.A.; Imrie, C.T. Liquid crystal dimers and the twist-bend nematic phase. The preparation and characterisation of the α,ω-bis(4-cyanobiphenyl-4′-yl) alkanedioates. Liq. Cryst. 2014, 41, 471–483. [Google Scholar] [CrossRef]
- Cruickshank, E.; Salamończyk, M.; Pociecha, D.; Strachan, G.; Storey, J.M.D.; Wang, C.; Feng, J.; Zhu, C.; Gorecka, E.; Imrie, C.T. Sulfur-linked cyanobiphenyl-based liquid crystal dimers and the twist-bend nematic phase. Liq. Cryst. 2019, 46, 1595–1609. [Google Scholar] [CrossRef]
- Šepelj, M.; Lesac, A.; Baumeister, U.; Diele, S.; Nguyen, H.L.; Bruce, D. Intercalated liquid-crystalline phases formed by symmetric dimers with an α,ω-diiminoalkylene spacer. J. Mater. Chem. 2007, 17, 1154–1165. [Google Scholar] [CrossRef]
- Dawood, A.A.; Grossel, M.C.; Luckhurst, G.; Richardson, R.M.; Timimi, B.A.; Wells, N.J.; Yousif, Y.Z. On the twist-bend nematic phase formed directly from the isotropic phase. Liq. Cryst. 2016, 43, 2–12. [Google Scholar] [CrossRef]
- Arakawa, Y.; Komatsu, K.; Tsuji, H. Twist-bend nematic liquid crystals based on thioether linkage. New J. Chem. 2019, 43, 6786–6793. [Google Scholar] [CrossRef]
- Henderson, P.A.; Imrie, C.T. Methylene-linked liquid crystal dimers and the twist-bend nematic phase. Liq. Cryst. 2011, 38, 1407–1414. [Google Scholar] [CrossRef]
- Paterson, D.; Walker, R.; Abberley, J.P.; Forestier, J.; Harrison, W.T.A.; Storey, J.M.D.; Pociecha, D.; Gorecka, E.; Imrie, C.T. Azobenzene-based liquid crystal dimers and the twist-bend nematic phase. Liq. Cryst. 2017, 44, 2060–2078. [Google Scholar] [CrossRef]
- Abberley, J.P.; Storey, J.M.D.; Imrie, C.T. Structure-property relationships in azobenzene-based twist-bend nematogens. Liq. Cryst. 2019, 42, 2102–2114. [Google Scholar] [CrossRef]
- Abberley, J.P.; Jansze, S.; Walker, R.; Paterson, D.; Henderson, P.; Marcelis, A.T.M.; Storey, J.M.D.; Imrie, C.T. Structure–property relationships in twist-bend nematogens: The influence of terminal groups. Liq. Cryst. 2017, 44, 68–83. [Google Scholar] [CrossRef]
- Paterson, D.; Crawford, C.A.; Pociecha, D.; Walker, R.; Storey, J.M.; Gorecka, E.; Imrie, C.T. The role of a terminal chain in promoting the twist-bend nematic phase: The synthesis and characterisation of the 1-(4-cyanobiphenyl-4′-yl)-6-(4-alkyloxyanilinebenzylidene-4′-oxy) hexanes. Liq. Cryst. 2018, 45, 2341–2351. [Google Scholar] [CrossRef]
- Zhang, Z.; Panov, V.P.; Nagaraj, M.; Goodby, J.; Luckhurst, G.R.; Gleeson, H.; Mandle, R.J.; Jones, J.C. Raman scattering studies of order parameters in liquid crystalline dimers exhibiting the nematic and twist-bend nematic phases. J. Mater. Chem. C 2015, 3, 10007–10016. [Google Scholar] [CrossRef]
- Sreenilayam, S.; Panov, V.P.; Vij, J.K.; Shanker, G. The NTB phase in an achiral asymmetrical bent-core liquid crystal terminated with symmetric alkyl chains. Liq. Cryst. 2017, 44, 244–253. [Google Scholar] [CrossRef]
- Tamba, M.G.; Baumeister, U.; Pelzl, G.; Weissflog, W. Banana-calamitic dimers: Further variations of the bent-core mesogenic unit. Ferroelectrics 2014, 468, 52–76. [Google Scholar] [CrossRef]
- Mandle, R.J.; Goodby, J. A Liquid Crystalline Oligomer Exhibiting Nematic and Twist-Bend Nematic Mesophases. ChemPhysChem 2016, 17, 967–970. [Google Scholar] [CrossRef]
- Simpson, F.P.; Mandle, R.J.; Moore, J.N.; Goodby, J. Investigating the Cusp between the nano- and macro-sciences in supermolecular liquid-crystalline twist-bend nematogens. J. Mater. Chem. C 2017, 5, 5102–5110. [Google Scholar] [CrossRef]
- Tuchband, M.; Paterson, D.; Salamończyk, M.; Norman, V.A.; Scarbrough, A.N.; Forsyth, E.; Garcia, E.; Wang, C.; Storey, J.M.D.; Walba, D.M.; et al. Distinct differences in the nanoscale behaviors of the twist–bend liquid crystal phase of a flexible linear trimer and homologous dimer. Proc. Natl. Acad. Sci. USA 2019, 166, 10698–10704. [Google Scholar] [CrossRef]
- Mandle, R.J.; Goodby, J. A novel nematic-like mesophase induced in dimers, trimers and tetramers doped with a high helical twisting power additive. Soft Matter 2018, 14, 8846–8852. [Google Scholar] [CrossRef]
- Mandle, R.J.; Goodby, J.W. A Nanohelicoidal Nematic Liquid Crystal Formed by a Non-Linear Duplexed Hexamer. Angew. Chem. Int. Ed. 2018, 57, 7096–7100. [Google Scholar] [CrossRef]
- Stevenson, W.D.; An, J.; Zeng, X.; Xue, M.; Zou, H.-X.; Liu, Y.; Ungar, G. Twist-bend nematic phase in biphenylethane-based copolyethers. Soft Matter 2018, 14, 3003–3011. [Google Scholar] [CrossRef]
- Greco, C.; Luckhurst, G.R.; Ferrarini, A. Enantiotopic discrimination and director organization in the twist-bend nematic phase. Phys. Chem. Chem. Phys. 2013, 15, 14961–14965. [Google Scholar] [CrossRef] [PubMed]
- Jansze, S.M.; Martínez-Felipe, A.; Storey, J.M.D.; Marcelis, A.T.M.; Imrie, C.T. A twist-bend nematic phase driven by hydrogen bonding. Angew. Chem. Int. Ed. 2015, 54, 643–646. [Google Scholar] [CrossRef]
- Walker, R.; Pociecha, D.; Abberley, J.P.; Martinez-Felipe, A.; Paterson, D.; Forsyth, E.; Lawrence, G.B.; Henderson, P.; Storey, J.M.D.; Gorecka, E.; et al. Spontaneous chirality through mixing achiral components: A twist-bend nematic phase driven by hydrogen-bonding between unlike components. Chem. Commun. 2018, 54, 3383–3386. [Google Scholar] [CrossRef] [PubMed]
- Walker, R.; Pociecha, D.; Crawford, C.; Storey, J.; Gorecka, E.; Imire, C. Hydrogen bonding and the design of twist-bend nematogens. J. Mol. Liq. 2020. [Google Scholar] [CrossRef]
- Paterson, D.A.; Gao, M.; Kim, Y.-K.; Jamali, A.; Finley, K.L.; Robles-Hernández, B.; Diez-Berart, S.; Salud, J.; de la Fuente, M.R.; Timimi, B.A.; et al. Understanding the twist-bend nematic phase: The characterisation of 1-(4-cyanobiphenyl-4′-yloxy)-6-(4-cyanobiphenyl-4′-yl)hexane (CB6OCB) and comparison with CB7CB. Soft Matter 2016, 12, 6827–6840. [Google Scholar] [CrossRef]
- Kato, T.; Frechet, J. New Approach to Mesophase Stabilization through Hydrogen-Bonding Molecular Interactions in Binary Mixtures. J. Am. Chem. Soc. 1989, 111, 8533–8534. [Google Scholar] [CrossRef]
- Al-Janabi, A.; Mandle, R.J.; Goodby, J. Isomeric trimesogens exhibiting modulated nematic mesophases. RSC Adv. 2017, 7, 47235–47242. [Google Scholar] [CrossRef]
- Mandle, R.J.; Stevens, M.P.; Goodby, J.W. Developments in liquid-crystalline dimers and oligomers. Liq. Cryst. 2017, 44, 2046–2059. [Google Scholar] [CrossRef]
- El Sayed, S.; De La Torre, C.; Santos-Figueroa, L.E.; Martínez-Máñez, R.; Sancenón, F.; Orzaez, M.; Costero, A.M.; Parra, M.; Gil, S. 2,4-Dinitrophenyl Ether-Containing Chemodosimeters for the Selective and Sensitive in Vitro and in Vivo Detection of Hydrogen Sulfide. Supramol. Chem. 2015, 27, 244–254. [Google Scholar] [CrossRef]
- Marquardt, D.W.; Mathematics, A. An Algorithm for Least-Squares Estimation of Nonlinear Parameters. J. Soc. Ind. Appl. Math. 1963, 11, 431–441. [Google Scholar] [CrossRef]
- Paterson, D.; Martinez-Felipe, A.; Jansze, S.; Marcelis, A.T.; Storey, J.M.; Imrie, C.T. New insights into the liquid crystal behaviour of hydrogen-bonded mixtures provided by temperature-dependent FTIR spectroscopy. Liq. Cryst. 2015, 42, 928–939. [Google Scholar] [CrossRef]
- Odinokov, S.; Iogansen, A. Torsional γ (OH) vibrations, Fermi resonance [2γ(OH) ⇐ ν(OH)] and isotopic effects in i.r. spectra of H-complexes of carboxylic acids with strong bases. Spectrochim. Acta Part A Mol. Spectrosc. 1972, 28, 2343–2350. [Google Scholar] [CrossRef]
- Lee, J.; Painter, P.; Coleman, M. Hydrogen Bonding in Polymer Blends. 4. Blends Involving Polymers Containing Methacrylic Acid and Vinylpyridinegroups. Macromolecules 1988, 21, 954–960. [Google Scholar] [CrossRef]
- Martinez-Felipe, A.; Cook, A.G.; Abberley, J.P.; Walker, R.; Storey, J.M.D.; Imrie, C.T. An FT-IR spectroscopic study of the role of hydrogen bonding in the formation of liquid crystallinity for mixtures containing bipyridines and 4-pentoxybenzoic acid. RSC Adv. 2016, 6, 108164–108179. [Google Scholar] [CrossRef]
- Martinez-Felipe, A.; Brebner, F.; Zaton, D.; Concellon, A.; Ahmadi, S.; Piñol, M.; Oriol, L. Molecular recognition via hydrogen bonding in supramolecular complexes: A Fourier transform infrared spectroscopy study. Molecules 2018, 23, 2278. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Felipe, A.; Imrie, C.T. The role of hydrogen bonding in the phase behaviour of supramolecular liquid crystal dimers. J. Mol. Struct. 2015, 1100, 429–437. [Google Scholar] [CrossRef]
- Abdy, M.J.; Murdoch, A.; Martinez-Felipe, A. New insights into the role of hydrogen bonding on the liquid crystal behaviour of 4-alkoxybenzoic acids: A detailed IR spectroscopy study. Liq. Cryst. 2016, 43, 2191–2207. [Google Scholar] [CrossRef]
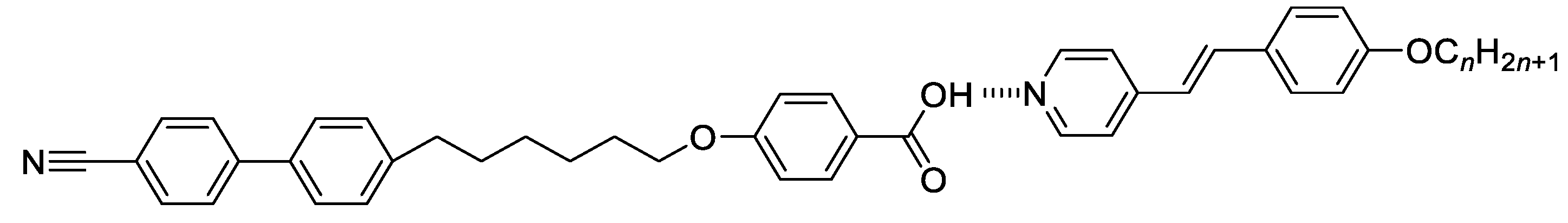
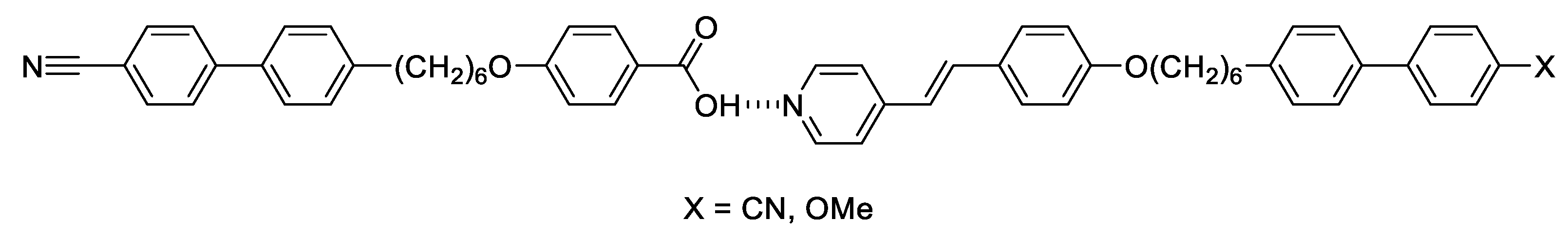


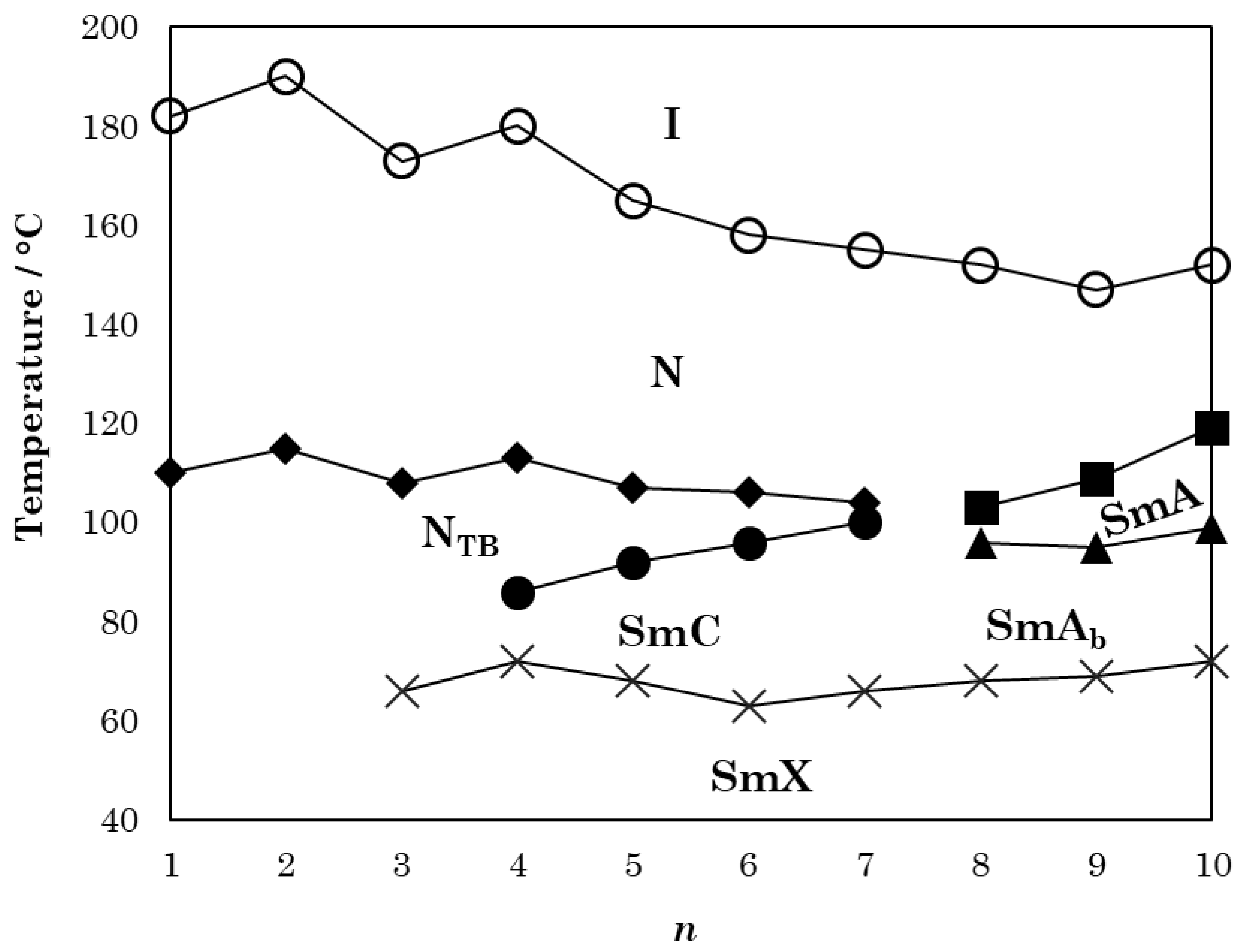

| n | TCr-/°C | TSmX/°C | TSmAbSmA/°C | TSmCNTB/°C | TSmAN/°C | TNTBN/°C | TNI/°C |
| 1 | 130 | 110 † | 182 | ||||
| 2 | 119 | 115 † | 190 | ||||
| 3 | 109 | 66 * | 108 † | 173 | |||
| 4 | 108 | 70 * | 86 * | 113 † | 180 | ||
| 5 | 121 | 68 * | 92 * | 107 † | 165 | ||
| 6 | 105 | 63 * | 96 * | 106 † | 158 | ||
| 7 | 100 | 66 * | 100 * | 104 † | 155 | ||
| 8 | 99 | 68 * | 96 * | 103 | 152 | ||
| 9 | 101 | 67 * | 95 * | 109 | 147 | ||
| 10 | 105 | 72 * | 100 * | 119 | 152 | ||
| n | ΔSCr-/R | ΔSSmX-/R | ΔSSmAbSmA/R | ΔSSmCNTB/R | ΔSSmAN/R | ΔSNTBN/R | ΔSNI/R |
| 1 | 8.95 | - | 0.67 | ||||
| 2 | 9.64 | - | 0.94 | ||||
| 3 | 8.19 | 0.12 | - | 0.89 | |||
| 4 | 11.63 | 0.90 | 0.23 | - | 0.70 | ||
| 5 | 13.51 | 0.96 | 0.24 | - | 0.94 | ||
| 6 | 13.07 | 1.00 | 0.30 | - | 0.91 | ||
| 7 | 13.01 | 1.11 | 0.48 | - | 1.02 | ||
| 8 | 12.46 | 0.87 | 0.58 ⁑ | ⁑ | 1.16 | ||
| 9 | 13.12 | 0.52 | 0.20 | 0.04 | 0.67 | ||
| 10 | 13.43 | 0.77 | 0.31 | 0.07 | 1.08 |
| n | Phase | Complex Length/l | Layer Thickness/Longitudinal Distance, d/Å | Distance Between Mesogens/Å | d/l |
|---|---|---|---|---|---|
| 2 | N | 37.8 | 12.3 † | 4.4 | 0.3 |
| NTB | 44.5, 19.4, 12.3 | 4.4 | 1.2, 0.5, 0.3 | ||
| 3 | SmX | 39.0 | 45.4, 22.8, 12.6 | 4.5, 4.1 | 1.4, 0.6, 0.3 |
| 4 | NTB | 40.1 | 21.6, 12.8 † | 4.9 | 0.5, 0.3 |
| 5 | N | 41.3 | 52.9, 23.1, 13.7 | 4.5 | 1.3, 0.6, 0.3 |
| NTB | 52.6, 22.9, 13.6 | 4.5 | 1.3, 0.6, 0.3 | ||
| SmC | 50.6, 23.7, 13.1 | 4.3 | 1.2, 0.6, 0.3 | ||
| SmX | 47.2, 23.7, 13.1 | 4.6, 4.2 | 1.1, 0.6, 0.3 | ||
| 6 | SmC | 42.4 | 52.2, 25.9, 13.5 | 4.4 | 1.2, 0.6, 0.3 |
| 7 | NTB | 47.2, 24.7 | 4.4 | 1.1, 0.6 | |
| SmC | 43.6 | 52.5, 26.0 | 4.4 | 1.2, 0.6 | |
| SmX | 49.6, 25.8 | 4.6, 4.1 | 1.1, 0.6 | ||
| 10 | SmA | 47.1 | 51.9 | 4.3 | 1.1 |
| SmAb | 53.5, 27.2 | 4.3 | 1.1, 0.6 | ||
| SmX | 52.4, 26.6 | 4.6, 4.2 | 1.1, 0.6 |
| Complex | TCr-/°C | TNTBN/°C | TNI/°C | ΔSCr-/R | ΔSNTBN/R | ΔSNI/R |
|---|---|---|---|---|---|---|
| CB6OBA:1OB6OS | 140 | 142* | 184 | 12.03 | 0.04 | 0.60 |
| CB6OBA:CB6OS | 127 | 143 | 186 | 12.34 | 0.07 | 1.24 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Walker, R.; Pociecha, D.; Martinez-Felipe, A.; Storey, J.M.; Gorecka, E.; Imrie, C.T. Twist-Bend Nematogenic Supramolecular Dimers and Trimers Formed by Hydrogen Bonding. Crystals 2020, 10, 175. https://doi.org/10.3390/cryst10030175
Walker R, Pociecha D, Martinez-Felipe A, Storey JM, Gorecka E, Imrie CT. Twist-Bend Nematogenic Supramolecular Dimers and Trimers Formed by Hydrogen Bonding. Crystals. 2020; 10(3):175. https://doi.org/10.3390/cryst10030175
Chicago/Turabian StyleWalker, Rebecca, Damian Pociecha, Alfonso Martinez-Felipe, John MD Storey, Ewa Gorecka, and Corrie T Imrie. 2020. "Twist-Bend Nematogenic Supramolecular Dimers and Trimers Formed by Hydrogen Bonding" Crystals 10, no. 3: 175. https://doi.org/10.3390/cryst10030175
APA StyleWalker, R., Pociecha, D., Martinez-Felipe, A., Storey, J. M., Gorecka, E., & Imrie, C. T. (2020). Twist-Bend Nematogenic Supramolecular Dimers and Trimers Formed by Hydrogen Bonding. Crystals, 10(3), 175. https://doi.org/10.3390/cryst10030175






