Rhodamine B Removal of TiO2@SiO2 Core-Shell Nanocomposites Coated to Buildings
Abstract
1. Introduction
2. Experimental Details
2.1. Materials
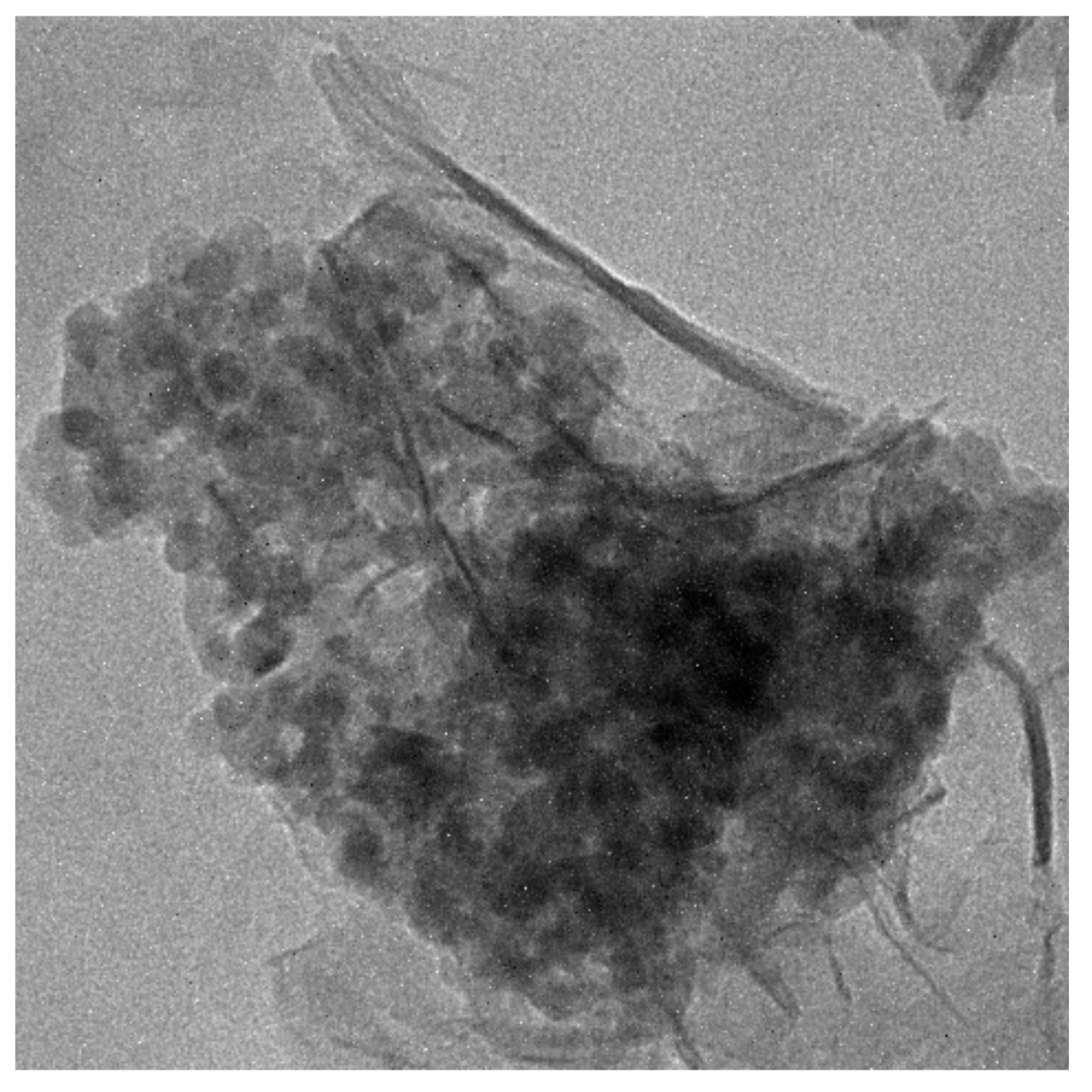
2.2. Synthesis of TiO2@SiO2 Nanocomposites
2.3. Cement Paste Preparation, Curing and Surface Treatment
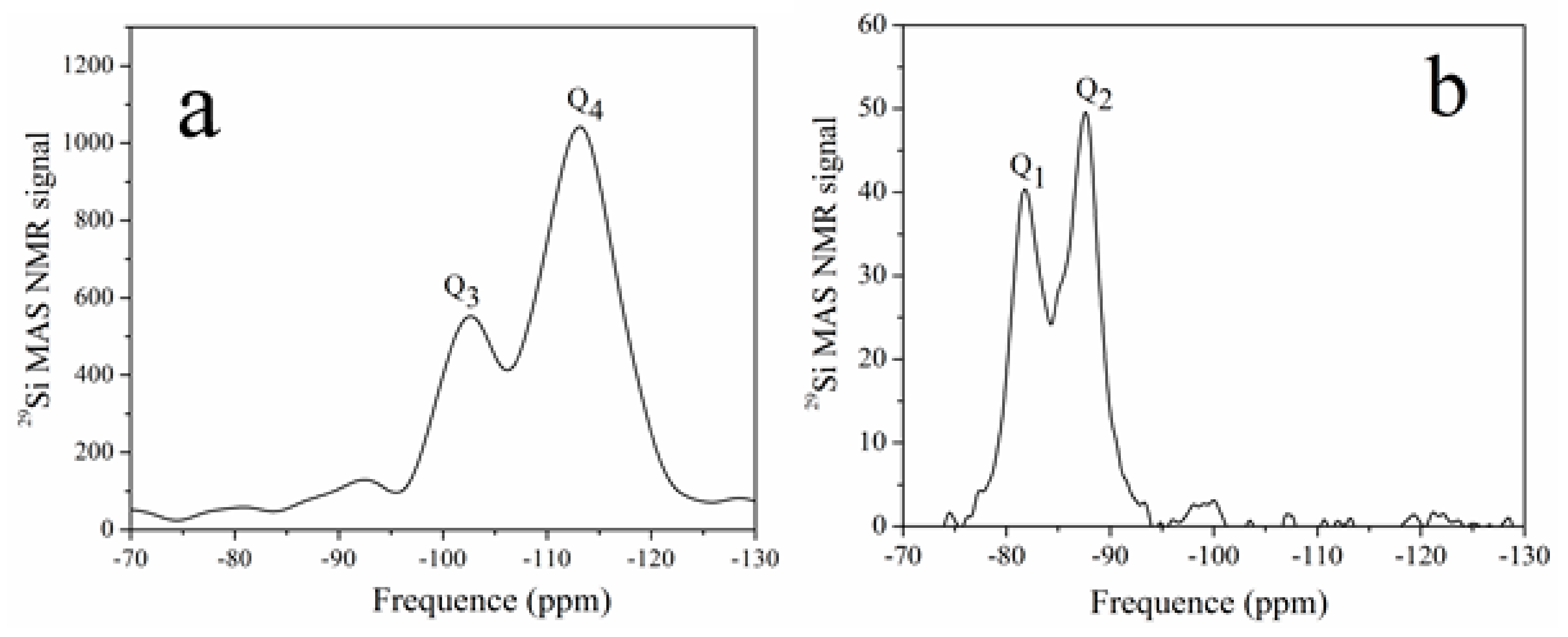
2.4. Characterization
2.5. Photocatalytic Degradation of RhB
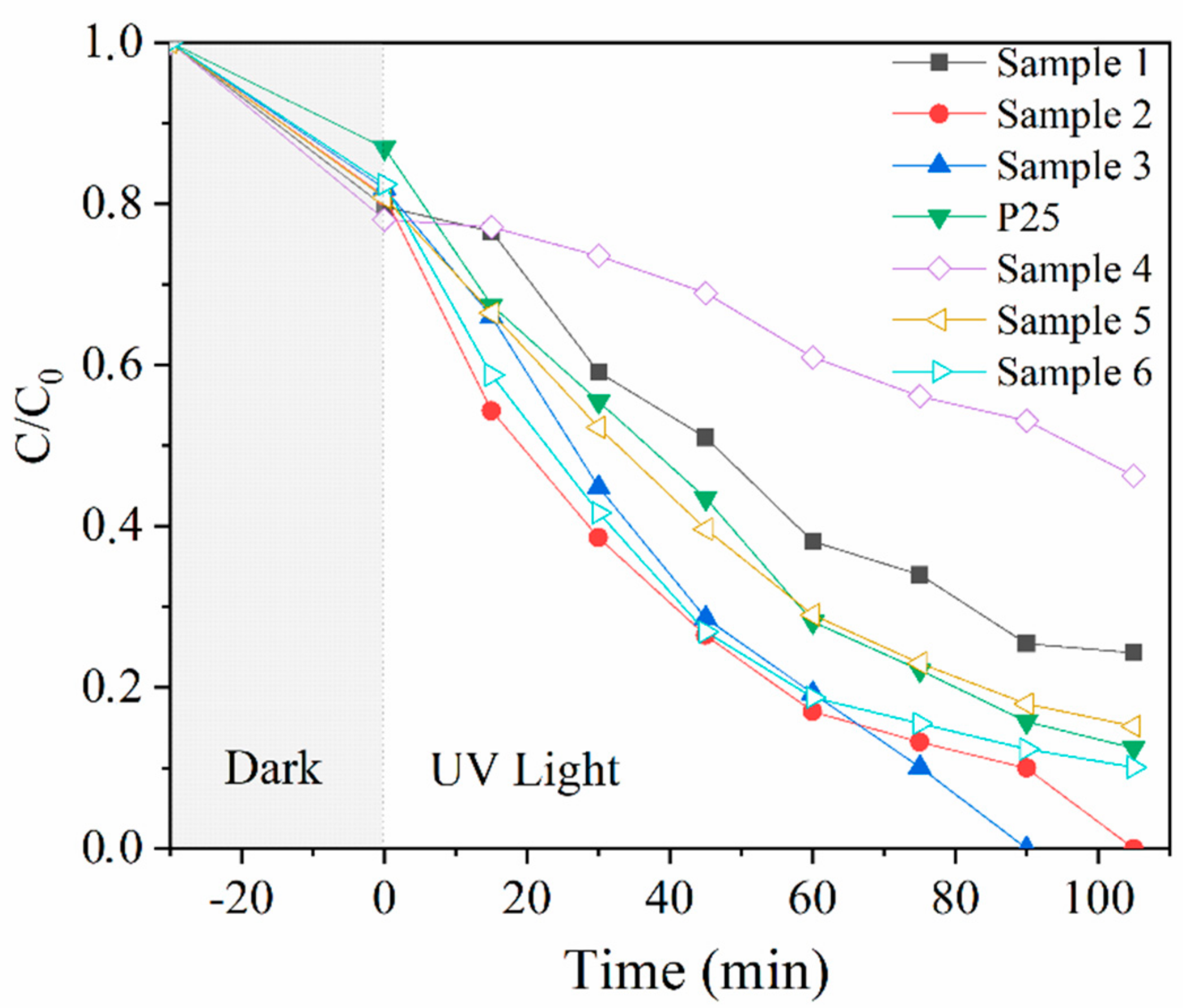
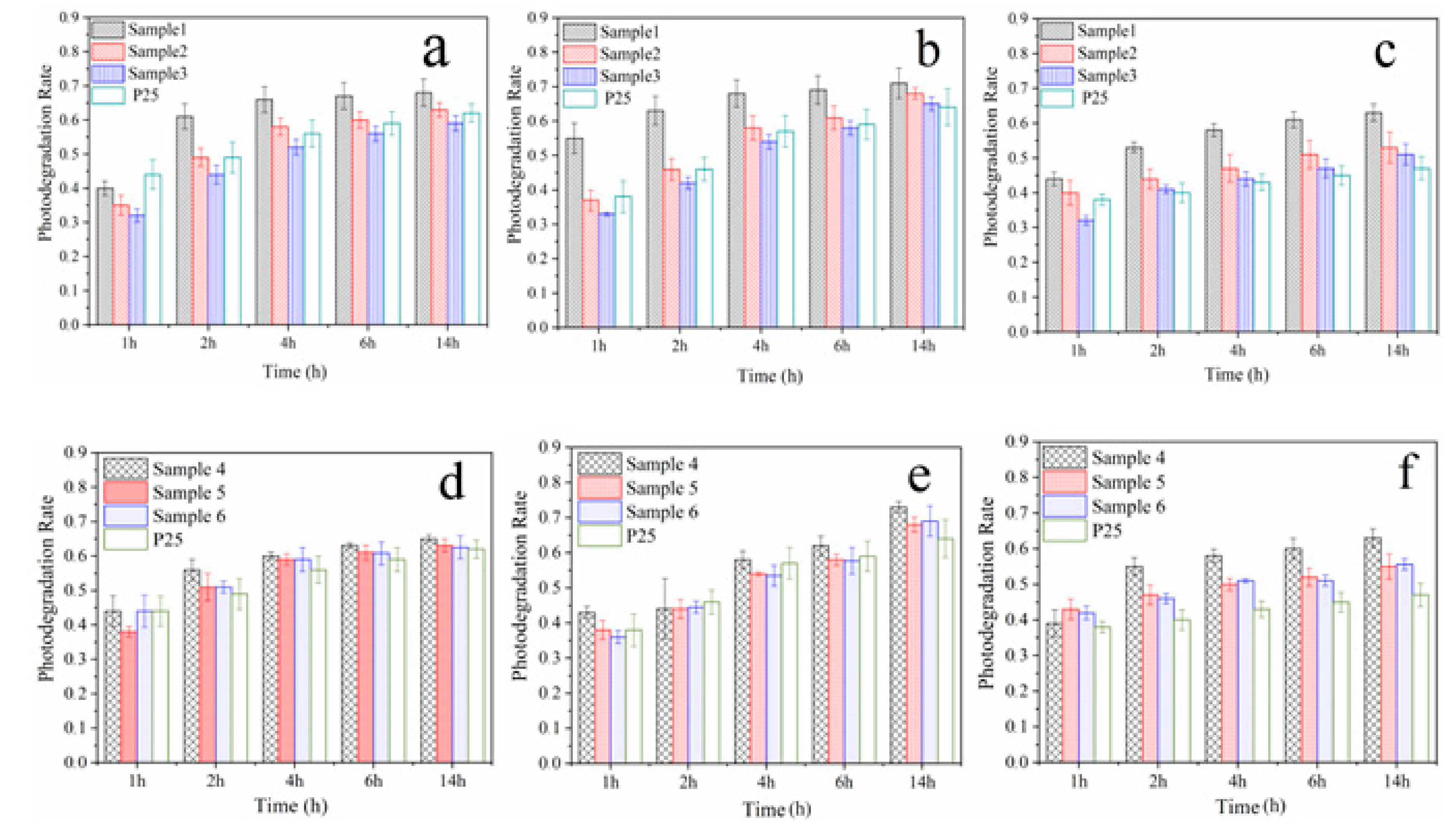
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Guo, M.Z.; Maury-Ramirez, A.; Poon, C.S. Self-cleaning ability of titanium dioxide clear paint coated architectural mortar and its potential in field application. J. Clean Prod. 2016, 112, 3583–3588. [Google Scholar] [CrossRef]
- Chen, M.; Chu, J.W. NOx photocatalytic degradation on active concrete road surface-from experiment to real-scale application. J. Clean. Prod. 2011, 19, 1266–1272. [Google Scholar] [CrossRef]
- Guo, M.Z.; Maury-Ramirez, A.; Poon, C.S. Versatile photocatalytic functions of self-compacting architectural glass mortars and their inter-relationship. Mater. Des. 2015, 88, 1260–1268. [Google Scholar] [CrossRef]
- Yousefi, A.; Allahverdi, A.; Hejazi, P. Effective dispersion of nano-TiO2 powder for enhancement of photocatalytic properties in cement mixes. Constr. Build Mater. 2013, 41, 224–230. [Google Scholar] [CrossRef]
- Cárdenas, C.; Tobón, J.I.; García, C.; Vila, J. Functionalized building materials: Photocatalytic abatement of NOx by cement pastes blened with TiO2 nanoparticles. Constr. Build Mater. 2012, 36, 820–825. [Google Scholar]
- Chen, J.; Kou, S.C.; Poon, C.S. Photocatalytic cement-based materials: Comparison of nitrogen oxides and toluene removal potentials and evaluation of self-cleaning performance. Build Environ. 2011, 46, 1827–1833. [Google Scholar] [CrossRef]
- Sugrañez, R.; Álvarez, J.I.; Cruz-Yusta, M.; Mármol, I.; Morales, J.; Vila, J.; Sánchez, L. Enhanced photocatalytic degradation of NOx gases by regulating the microstructure of mortar cement modified with titanium dioxide. Build Environ. 2013, 69, 55–63. [Google Scholar] [CrossRef]
- Sun, J.F.; Xu, K.; Shi, C.Q.; Ma, J.; Li, W.F.; Shen, X.D. Influence of core/shell TiO2@SiO2 nanoparticles on cement hydration. Constr. Build Mater. 2017, 156, 114–122. [Google Scholar] [CrossRef]
- Tiainen, H.; Wiedmer, D.; Haugen, H.J. Processing of highly porous TiO2 bone scaffolds with improved compressive strength. J. Eur. Ceram Soc. 2013, 33, 15–24. [Google Scholar] [CrossRef]
- Ruot, B.; Plassais, A.; Olive, F.; Guillot, L.; Bonafous, L. TiO2-containing cement pastes and mortars: Measurements of the photocatalytic efficiency using a rhodamine B-based colourimetric test. Sol. Energy 2009, 83, 1794–1801. [Google Scholar] [CrossRef]
- Folli, A.; Pade, C.; Hansen, T.B.; Marco, T.D.; Macphee, D.E. TiO2 photocatalysis in cementitious systems: Insights into self-cleaning and depollution chemistry. Cem. Concr. Res. 2012, 42, 539–548. [Google Scholar] [CrossRef]
- Rachel, A.; Subrahmanyam, M.; Boule, P. Comparison of photocatalytic efficiencies of TiO2 in suspended and immobilised form for the photocatalytic degradation of nitrobenzenesulfonic acids. Appl. Cat. B Environ. 2002, 37, 301–308. [Google Scholar] [CrossRef]
- Martinez, T.; Bertron, A.; Escadeillas, G.; Ringot, E.; Simon, V. BTEX abatement by photocatalytic TiO2-bearing coatings applied to cement mortars. Build Environ. 2014, 71, 186–192. [Google Scholar] [CrossRef]
- Kamaruddin, S.; Stephan, D. Quartz-titania composites for the photocatalytical modification of construction materials. Cem. Concr. Comp. 2013, 36, 109–115. [Google Scholar] [CrossRef]
- Kamaruddin, S.; Stephan, D. Sol-gel Mediated Coating and Characterization of Photocatalytic Sand and Fumed Silica for Environmental Remediation. Water Air Soil Pollut. 2014, 225, 1948. [Google Scholar] [CrossRef]
- Hendrix, Y.; Lazaro, A.; Yu, Q.; Brouwers, J. Titania-Silica Composites: A Review on the Photocatalytic Activity and Synthesis Methods. World J. Nano Sci. Eng. 2015, 5, 161–177. [Google Scholar] [CrossRef]
- Kamaruddin, S.; Stephan, D. The preparation of silica-titania core-shell particles and their impact as an alternative material to pure nano-titania photocatalysts. Catal. Today 2011, 161, 53–55. [Google Scholar] [CrossRef]
- Mendoza, C.; Vallea, A.; Castellote, M.; Bahamondea, A.; Faraldosa, M. TiO2 and TiO2-SiO2 coated cement: Comparison of mechanic and photocatalytic properties. Appl. Catal. B Environ. 2015, 178, 155–164. [Google Scholar] [CrossRef]
- Sikora, P.; Cendrowski, K.; Markowska-Szczupak, A.; Horszczaruk, E.; Mijowska, E. The effects of silica/titania nanocomposite on the mechanical and bactericidal properties of cement mortars. Constr. Build Mater. 2017, 150, 738–746. [Google Scholar] [CrossRef]
- Wang, D.; Hou, P.K.; Yang, P.; Cheng, X. BiOBr@SiO2 flower-like nanospheres chemically-bonded on cement-based materials for photocatalysis. Appl. Surf. Sci. 2018, 430, 539–548. [Google Scholar] [CrossRef]
- Wang, D.; Hou, P.K.; Zhang, L.N.; Yang, P.; Cheng, X. Photocatalytic and hydrophobic activity of cement-based materials from benzyl-terminated-TiO2 spheres with core-shell structures. Constr. Build Mater. 2017, 148, 176–183. [Google Scholar] [CrossRef]
- Vinogradova, E.; Estrada, M.; Moreno, A. Colloidal aggregation phenomena: Spatial structuring of TEOS-derived silica aerogels. J. Colloid Interface Sci. 2006, 298, 209–212. [Google Scholar] [CrossRef] [PubMed]
- Humbert, B. Estimation of hydroxyl density at the surface of pyrogenic silicas by complementary NMR and Raman experiments. J. Non-Cryst. Solids 1995, 191, 29–37. [Google Scholar] [CrossRef]


| Constituent | CaO | SiO2 | SO3 | Al2O3 | MgO | Fe2O3 | TiO2 | K2O | Na2O |
|---|---|---|---|---|---|---|---|---|---|
| Mass (wt. %) | 65.44 | 20.09 | 4.07 | 2.55 | 1.77 | 0.25 | 0.33 | 0.24 | 0.71 |
| Parameters TiO2@SiO2 | P25 (g) | TEOS (mL) | Water (g) | Ethanol (mL) | NH3·H2O (mL) | Temperature (°C) | Main Thickness of the Shells (nm) | Surface Areas (m2∙g−1) |
|---|---|---|---|---|---|---|---|---|
| Sample 1 | 0.05 | 0.3 | 100 | 80 | 1 | 25 | 3.92 | 96.63 |
| Sample 2 | 0.10 | 0.3 | 100 | 80 | 1 | 25 | 2.25 | 46.63 |
| Sample 3 | 0.15 | 0.3 | 100 | 80 | 1 | 25 | 1.95 | 46.49 |
| Sample 4 | 0.05 | 0.3 | 100 | 80 | 1 | 0 | 6.13 | 110.42 |
| Sample 5 | 0.10 | 0.3 | 100 | 80 | 1 | 0 | 4.17 | 99.75 |
| Sample 6 | 0.15 | 0.3 | 100 | 80 | 1 | 0 | 3.70 | 91.42 |
| Sample | Sample 1 | Sample 2 | Sample 3 | Sample 4 | Sample 5 | Sample 6 | P25 |
|---|---|---|---|---|---|---|---|
| Direct measurement | 68.1% | 63.1% | 59.4% | 65.0% | 63.1% | 62.5% | 62.4% |
| After curing | 71.5% | 68.1% | 65.0% | 73.4% | 68.0% | 69.1% | 64.0% |
| After weather process | 62.9% | 53.4% | 51.0% | 63.3% | 55.0% | 55.6% | 47.4% |
| Reduction | 7.6% | 15.4% | 14.1% | 2.6% | 12.8% | 11.0% | 24.0% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, D.; Geng, Z.; Hou, P.; Yang, P.; Cheng, X.; Huang, S. Rhodamine B Removal of TiO2@SiO2 Core-Shell Nanocomposites Coated to Buildings. Crystals 2020, 10, 80. https://doi.org/10.3390/cryst10020080
Wang D, Geng Z, Hou P, Yang P, Cheng X, Huang S. Rhodamine B Removal of TiO2@SiO2 Core-Shell Nanocomposites Coated to Buildings. Crystals. 2020; 10(2):80. https://doi.org/10.3390/cryst10020080
Chicago/Turabian StyleWang, Dan, Zhi Geng, Pengkun Hou, Ping Yang, Xin Cheng, and Shifeng Huang. 2020. "Rhodamine B Removal of TiO2@SiO2 Core-Shell Nanocomposites Coated to Buildings" Crystals 10, no. 2: 80. https://doi.org/10.3390/cryst10020080
APA StyleWang, D., Geng, Z., Hou, P., Yang, P., Cheng, X., & Huang, S. (2020). Rhodamine B Removal of TiO2@SiO2 Core-Shell Nanocomposites Coated to Buildings. Crystals, 10(2), 80. https://doi.org/10.3390/cryst10020080





