Phase Transitions and Crystal Structures of Ni(II) Complexes Determined with X-ray Powder Diffraction Data
Abstract
1. Introduction
2. Experimental and Data Analysis
2.1. Sample Preparation
2.2. Intensity Data Collection
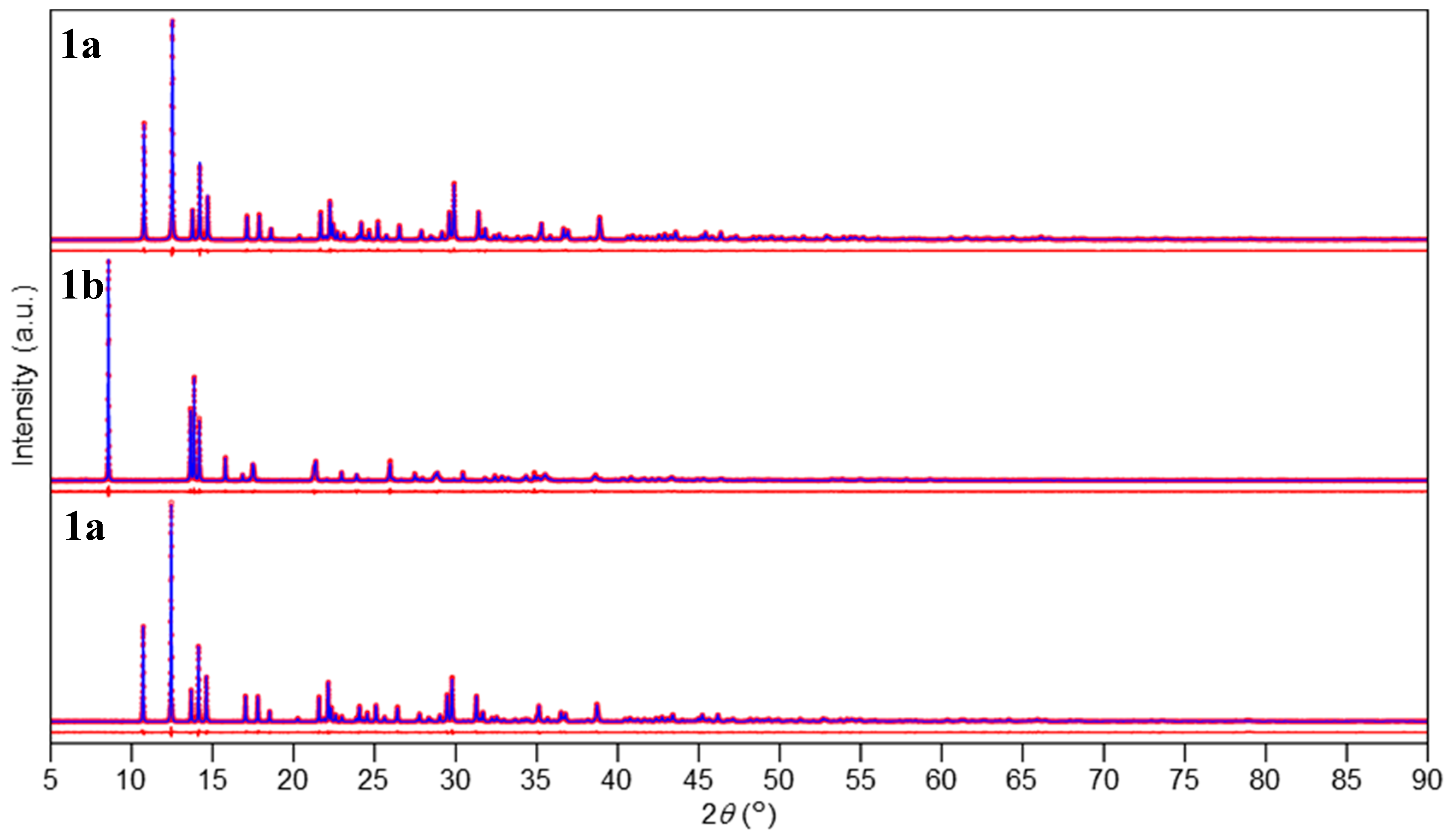
2.3. Data Analysis
3. Results and Discussion
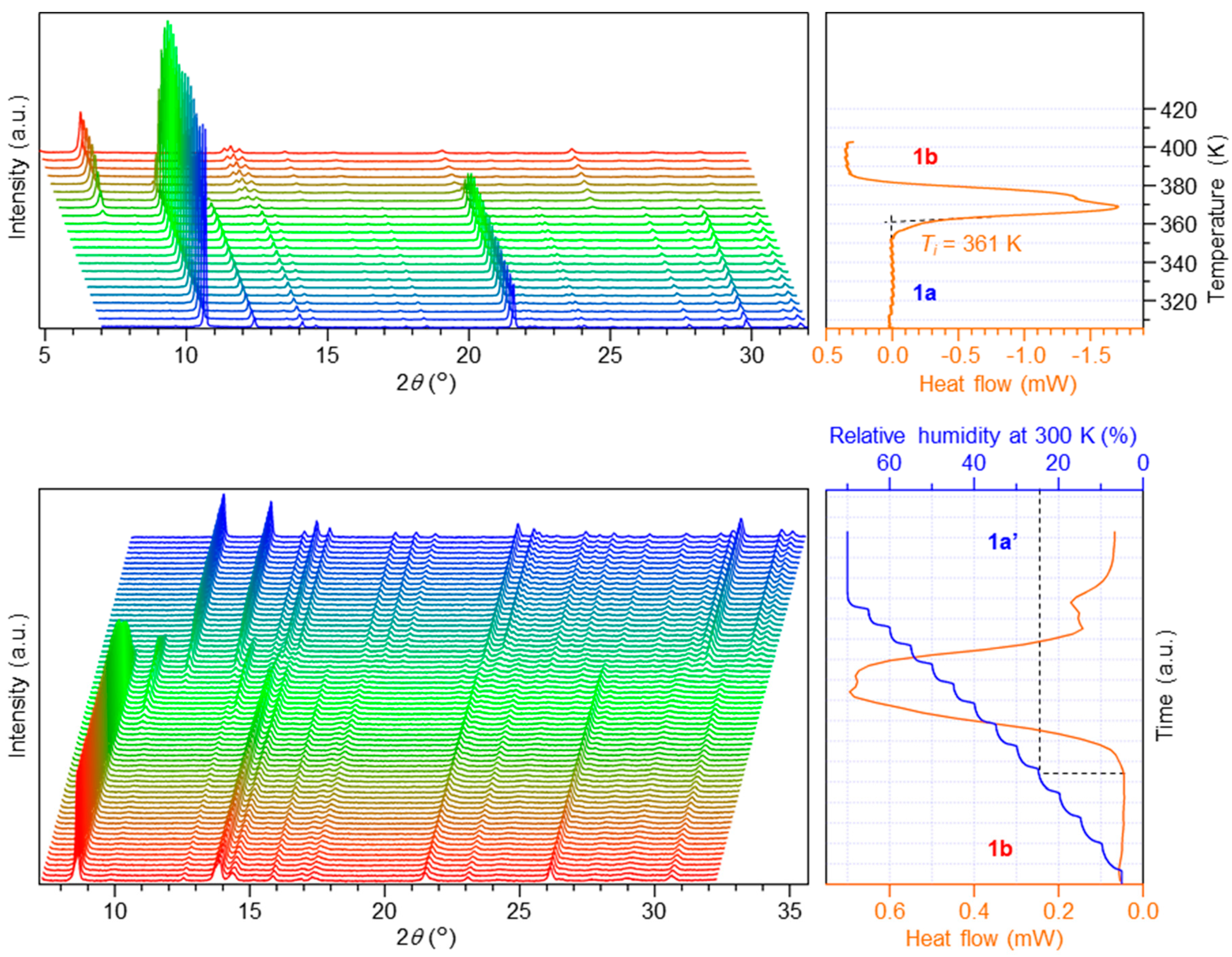
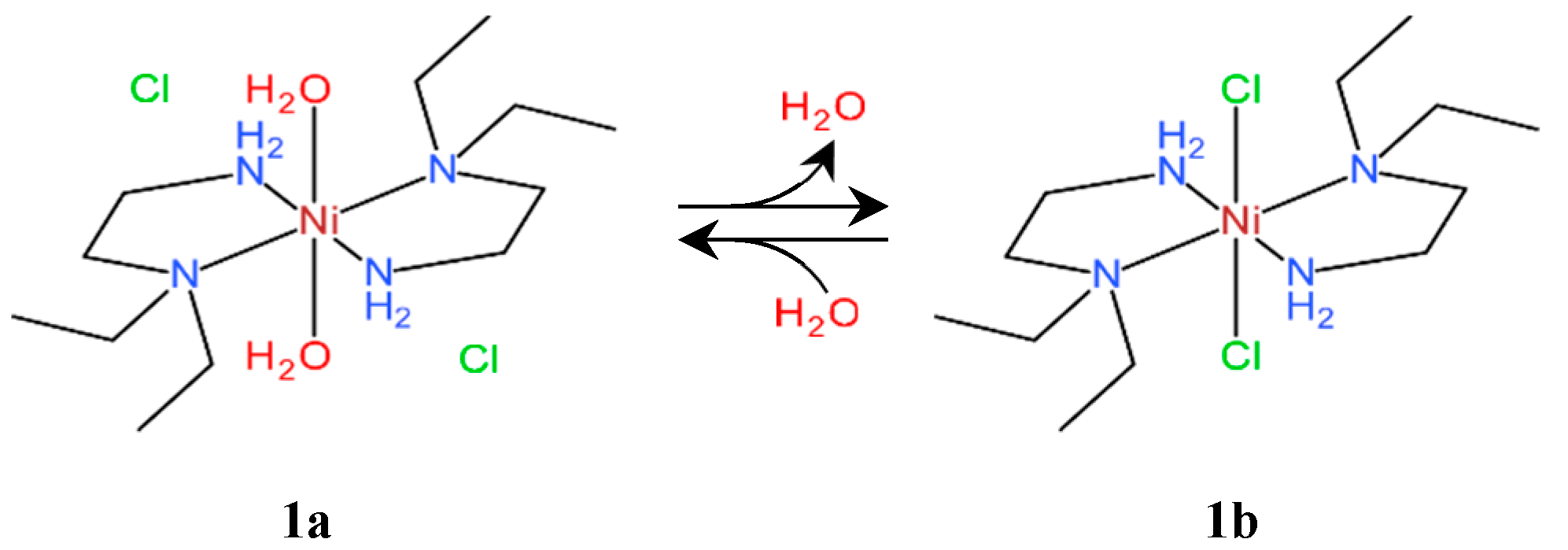
3.1. Phase Transition Behaviors of Chloride Complex 1a
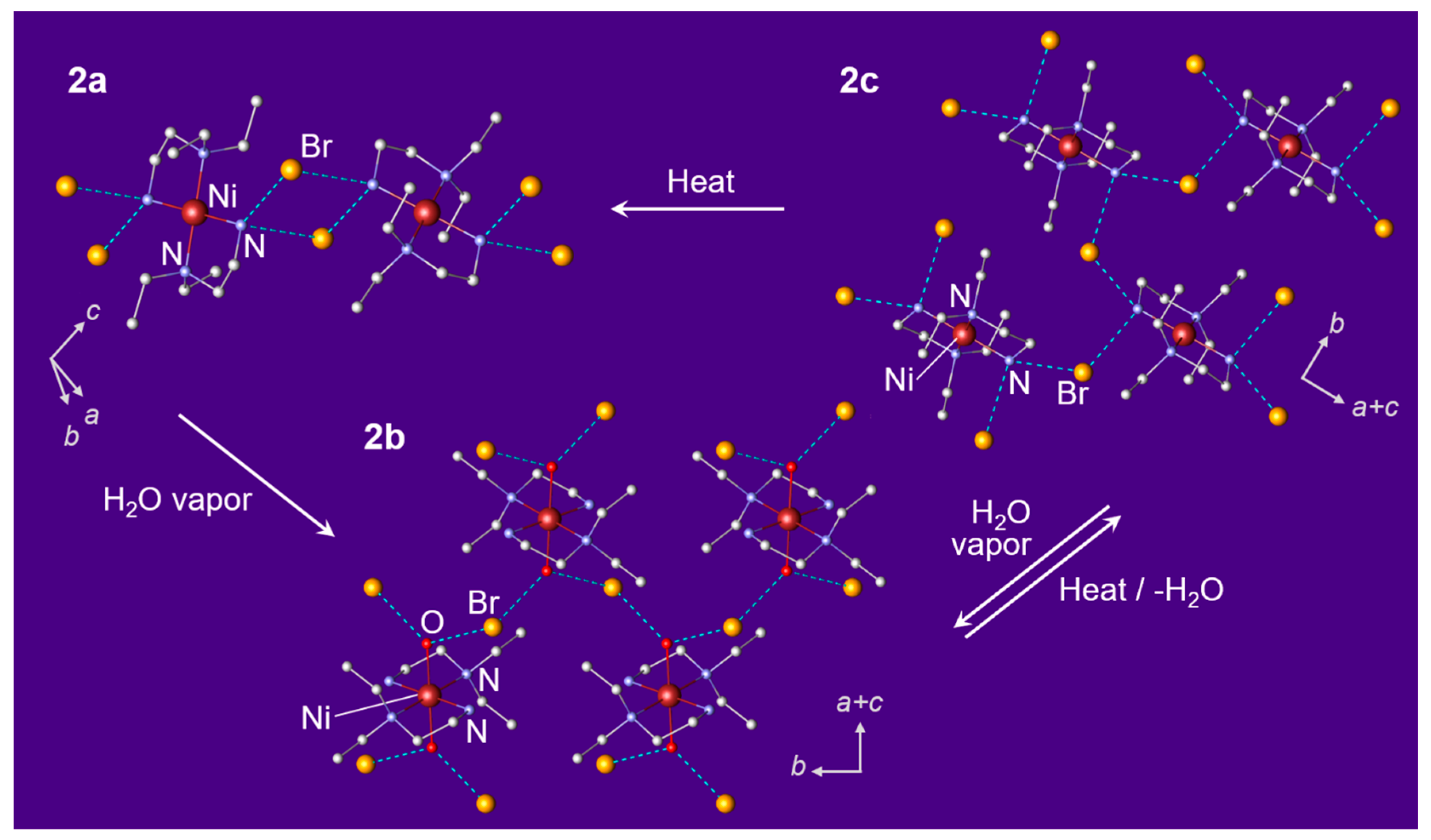
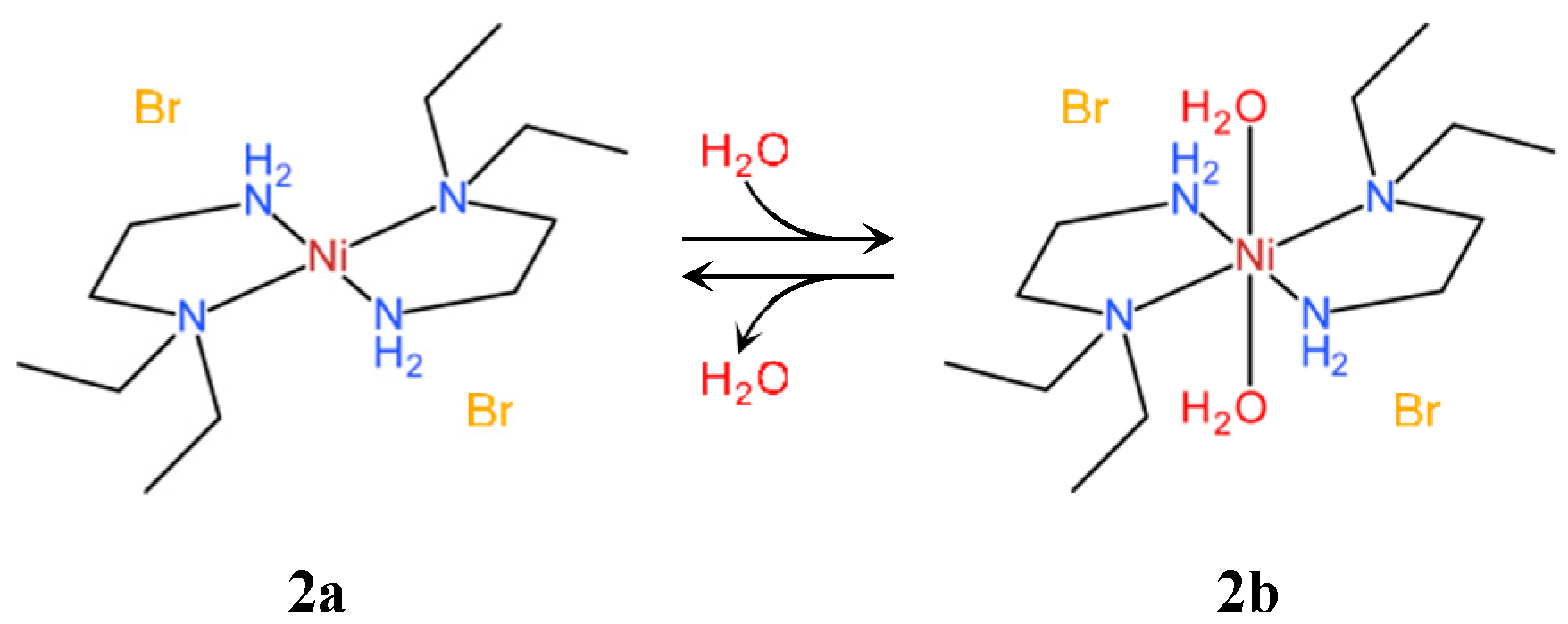
3.2. Phase Transition Behaviors of Bromide Complex 2a
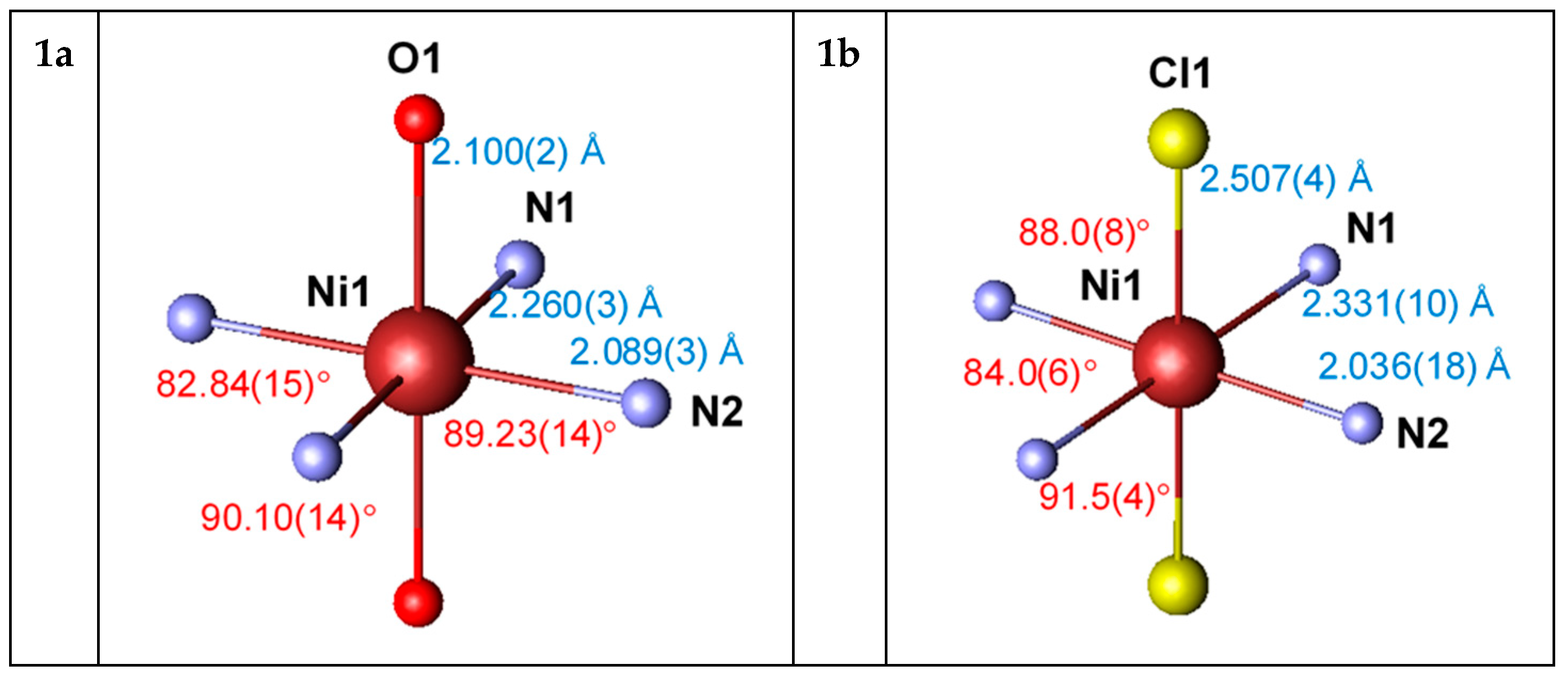
3.3. Crystal Structures of Chloride Complexes
3.4. Crystal Structures of Bromide Complexes
4. Summary
Author Contributions
Funding
Conflicts of Interest
References
- Goodgame, D.M.L.; Venanzi, L.M. Diamine Complexes of Nickel(II). Part I. Complexes with NN-Diethylethylenediamine. J. Chem. Soc. 1963, 616–627. [Google Scholar] [CrossRef]
- Tsuchiya, R.; Joba, S.; Uehara, A.; Kyuno, E. Derivatographic Studies on Transition Metal Complexes. X. Thermal Octahedral-to-Square Planar Transformation of Ni(NN-deen)2X2·nH2O in Solid Phase. Bull. Chem. Soc. Jpn. 1973, 46, 1454–1456. [Google Scholar] [CrossRef]
- Fabbrizzi, L.; Micheloni, M.; Paoletti, P. Continuous and Discontinuous Thermochromism of Copper(II) and Nickel(II) Complexes with N,N-Diethylethylenediamine. Inorg. Chem. 1974, 13, 3019–3021. [Google Scholar] [CrossRef]
- Ferraro, J.R.; Basile, L.J.; G-Ineguez, L.R.; Paoletti, P.; Fabbrizzi, L. Concerning the Thermochromic Mechanism of Copper(II) and Nickel(II) Complexes of N,N-Diethylethylenediamine. Inorg. Chem. 1976, 15, 2342–2345. [Google Scholar] [CrossRef]
- Mitra, S.; De, G.; Chaudhuri, N.R. Thermal Investigation of Diamine Complexes of Nickel(II) in the Solid Phase. Thermochimica Acta 1983, 66, 187–195. [Google Scholar] [CrossRef]
- Ikeda, R.; Kotani, K.; Ohki, H.; Ishimaru, S.; Okamoto, K.; Ghosh, A. Studies on 2H NMR and single crystal X-ray diffraction of thermochromic bis(N,N-diethyl-1,2-ethanediamine) nickel(II) complexes with and without structural phase transitions. J. Mol. Struct. 1995, 345, 159–165. [Google Scholar] [CrossRef]
- Hayami, S.; Urakami, D.; Sato, S.; Kojima, Y.; Inoue, K.; Ohba, M. Structures and Dielectric Properties in Thermochromic Nickel(II) Compounds. Chem. Lett. 2009, 38, 490–491. [Google Scholar] [CrossRef]
- Ihara, Y.; Satake, Y.; Fujimoto, Y.; Senda, H.; Suzuki, M.; Uehara, A. X-Ray Crystal Structures of Octahedral Diaquabis(N,N-dialkylethylenediamine)nickel(II) Complexes Possessing Elongated Nickel(II)-Nitrogen Bonds along Axial Direction. Bull. Chem. Soc. Jpn. 1991, 64, 2349–2352. [Google Scholar] [CrossRef]
- Ferrara, S.J.; Mague, J.T.; Donahue, J.P. trans-Bis(N,N-diethylethylenediamine)-nickel(II) dibromide. Acta Cryst. 2011, E67, m48–m49. [Google Scholar] [CrossRef]
- Werner, P.-E.; Eriksson, L.; Westdahl, M. TREOR, a semi-exhaustive trial-and-erro poweder indexing program for all symmetries. J. Appl. Cryst. 1985, 18, 367–370. [Google Scholar] [CrossRef]
- Altomare, A.; Camalli, M.; Cuocci, C.; Giacovazzo, C.; Moliterni, A.; Rizzi, R. EXPO2009: Structure solution by powder data in direct and reciprocal space. J. Appl. Cryst. 2009, 42, 1197–1202. [Google Scholar] [CrossRef]
- Falcioni, M.; Deem, M.W. A biased Monte Carlo scheme for zeolite structure solution. J. Chem. Phys. 1999, 110, 1754–1766. [Google Scholar] [CrossRef]
- Rietveld, H.M. A profile refinement method for nuclear and magnetic structures. J. Appl. Cryst. 1969, 2, 65–71. [Google Scholar] [CrossRef]
- Sasaki, A.; Himeda, A.; Konaka, H.; Muroyama, N. Ab initio crystal structure analysis based on powder diffraction data using PDXL. Rigaku J. 2010, 26, 10–14. [Google Scholar]




| Sample | 1a | 1b | 1a’ |
|---|---|---|---|
| Crystal system | Monoclinic | Triclinic | Monoclinic |
| Space group | P21/n | P-1 | P21/n |
| a (Å) | 9.2046(1) | 6.5439(3) | 9.1783(1) |
| b (Å) | 8.7403(1) | 6.8553(3) | 8.7153(1) |
| c (Å) | 12.4664(1) | 10.7084(5) | 12.4321(2) |
| α (°) | 90 | 74.339(1) | 90 |
| β (°) | 104.4432(4) | 82.765(1) | 104.4694(4) |
| γ (°) | 90 | 77.643(2) | 90 |
| V (Å3) | 971.242(13) | 450.64(3) | 962.91(3) |
| Z | 2 | 1 | 2 |
| Z’ | 0.5 | 0.5 | 0.5 |
| Rp | 4.29% | 5.89% | 3.49% |
| Rwp | 5.99% | 8.21% | 4.51% |
| S | 1.39 | 1.28 | 1.08 |
| Sample | 2a | 2b | 2c | 2a’ |
|---|---|---|---|---|
| Crystal system | Monoclinic | Monoclinic | Monoclinic | Monoclinic |
| Space group | C2/c | P21/n | P21/n | C2/c |
| a (Å) | 13.0152(3) | 9.2587(2) | 9.3036(4) | 13.0882(3) |
| b (Å) | 11.5547(3) | 8.9071(2) | 8.0564(4) | 11.7373(2) |
| c (Å) | 12.9689(4) | 12.7873(2) | 12.6451(6) | 12.8290(3) |
| α (°) | 90 | 90 | 90 | 90 |
| β (°) | 105.7195(6) | 104.1356(4) | 100.1845(14) | 105.1115(4) |
| γ (°) | 90 | 90 | 90 | 90 |
| V (Å3) | 1877.41(9) | 1022.61(3) | 932.86(8) | 1902.63(7) |
| Z | 4 | 2 | 2 | 4 |
| Z’ | 0.5 | 0.5 | 0.5 | 0.5 |
| Rp | 3.82% | 3.24% | 3.27% | 4.30% |
| Rwp | 5.02% | 4.24% | 4.21% | 5.57% |
| S | 1.01 | 1.16 | 1.31 | 1.12 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Konaka, H.; Sasaki, A. Phase Transitions and Crystal Structures of Ni(II) Complexes Determined with X-ray Powder Diffraction Data. Crystals 2020, 10, 106. https://doi.org/10.3390/cryst10020106
Konaka H, Sasaki A. Phase Transitions and Crystal Structures of Ni(II) Complexes Determined with X-ray Powder Diffraction Data. Crystals. 2020; 10(2):106. https://doi.org/10.3390/cryst10020106
Chicago/Turabian StyleKonaka, Hisashi, and Akito Sasaki. 2020. "Phase Transitions and Crystal Structures of Ni(II) Complexes Determined with X-ray Powder Diffraction Data" Crystals 10, no. 2: 106. https://doi.org/10.3390/cryst10020106
APA StyleKonaka, H., & Sasaki, A. (2020). Phase Transitions and Crystal Structures of Ni(II) Complexes Determined with X-ray Powder Diffraction Data. Crystals, 10(2), 106. https://doi.org/10.3390/cryst10020106




