Assessment of the Thermal Properties of Aromatic Esters as Novel Phase Change Materials
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Characterization
2.2.1. Density
2.2.2. Differential Scanning Calorimetry (DSC)
2.2.3. Thermal Cycling with EasyMax
2.2.4. Thermogravimetric Analysis (TGA)
2.2.5. Transient Hot Bridge (THB)
3. Results and Discussion
3.1. Density
3.2. Phase Change Temperatures
3.3. Phase Change Enthalpies
3.4. Degradation Temperatures
3.5. Thermal Conductivity
3.6. Natural Occurrence and Toxicity
4. Conclusions and Outlook
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| 2,6-DMNDC | Dimethyl-2,6-Naphthalene Dicarboxylate |
| AnAc | Anisyl Acetate |
| BEB | Benzyl Benzoate |
| DBO | Dibenzyl Oxalate |
| DiMeOHTe | Dimethyl Terephthalate |
| DSC | Differential Scanning Calorimetry |
| I | Measuring current [mA] (for Transient Hot Bridge) |
| LHS | Latent Heat Storage |
| Lit. | Literature |
| Me-2-Fu | Methyl-2-Furoate |
| Me-2-MeBe | Methyl-2-methoxybenzoate |
| MW | Molecular Weight [g/mol] |
| NA | Not Available |
| PCM | Phase Change Material |
| PET | Polyethylene Terephthalate |
| PEN | Polyethylene Naphthalate |
| PHB | Phenyl Benzoate |
| RT | Room Temperature |
| SNSF | Swiss National Science Foundation |
| Tdegr endset | Endset degradation temperature [C] |
| Tdegr onset | Onset degradation temperature [C] |
| TES | Thermal Energy Storage |
| TGA | Thermogravimetric Analysis |
| THB | Transient Hot Bridge |
| Tc | Onset crystallization temperature [C] |
| Tm | Onset melting temperature [C] |
| Greek Symbols | |
| ΔH | Gravimetric [J/g], volumetric [J/mL] or molar enthalpy of fusion [kJ/mol] |
| Thermal conductivity [W/(m·K)] | |
| Density [g/mL] |
Appendix A. DSC curves of AnAc, BEB and Me-2-MeBe



Appendix B. EasyMax Curves of BEB

Appendix C. TGA Curves


References
- International Energy Agency IEA. Tracking Energy Integration, Paris. 2019. Available online: https://www.iea.org/reports/tracking-energy-integration (accessed on 20 August 2020).
- International Energy Agency IEA. World Energy Outlook 2019, Paris. 2019. Available online: https://www.iea.org/reports/world-energy-outlook-2019 (accessed on 20 August 2020).
- International Energy Agency IEA. Renewables 2019, Paris. 2019. Available online: https://www.iea.org/reports/renewables-2019 (accessed on 20 August 2020).
- Li, G.; Zheng, X. Thermal energy storage system integration forms for a sustainable future. Renew. Sustain. Energy Rev. 2016, 62, 736–757. [Google Scholar] [CrossRef]
- Cabeza, L.; Mehling, H. Heat and Cold Storage with PCM: An up to Date Introduction into Basics and Applications; Springer: Berlin/Heidelberg, Germany, 2008; ISBN 9783540685562. [Google Scholar]
- Magendran, S.S.; Khan, F.S.A.; Mubark, N.M.; Vaka, M.; Rashmi, W.; Khalid, M.; Abdullah, E.C.; Nizamuddin, S.; Karri, R.R. Synthesis of organic phase change materials (PCM) for energy storage applications: A review. Nano-Struct. Nano-Objects 2019, 20, 100399. [Google Scholar] [CrossRef]
- Gmelin, L. Handbuch der Chemie, Vol. 4: Handbuch der organischen Chemie; K. Winter: Heidelberg, Germany, 1848; ISBN 9781176030664. [Google Scholar]
- Sari, A.; Bicer, A. Thermal energy storage properties and thermal reliability of some fatty acid esters/building material composites as novel form-stable PCMs. Sol. Energy Mater. Sol. Cells 2012, 101, 114–122. [Google Scholar] [CrossRef]
- Stamatiou, A.; Obermeyer, M.; Fischer, L.J.; Schuetz, P.; Worlitschek, J. Investigation of unbranched, saturated, carboxylic esters as phase change materials. Renew. Energy 2017, 108, 401–409. [Google Scholar] [CrossRef]
- Aydin, A.A.; Aydin, A. High-chain fatty acid esters of 1-hexadecanol for low temperature thermal energy storage with phase change materials. Sol. Energy Mater. Sol. Cells 2012, 96, 93–100. [Google Scholar] [CrossRef]
- Ravotti, R.; Fellmann, O.; Lardon, N.; Fischer, L.J.; Stamatiou, A.; Worlitschek, J. Synthesis and Investigation of Thermal Properties of Highly Pure Carboxylic Fatty Esters to Be Used as PCM. Appl. Sci. 2018, 8, 1069. [Google Scholar] [CrossRef] [Green Version]
- Ravotti, R.; Fellmann, O.; Lardon, N.; Fischer, L.J.; Stamatiou, A.; Worlitschek, J. Analysis of Bio-Based Fatty Esters PCM’s Thermal Properties and Investigation of Trends in Relation to Chemical Structures. Appl. Sci. 2019, 9, 225. [Google Scholar] [CrossRef] [Green Version]
- Lutton, E.S.; Fehl, A.J. The polymorphism of odd and even saturated single acid triglycerides, C8–C22. Lipids 1970, 5, 90–99. [Google Scholar] [CrossRef]
- Hernqvist, L. Polymorphism of Triglycerides a Crystallographic Review. J. Food Struct. 1990, 9, 39–44. [Google Scholar]
- Moorty, A.S.; Liu, R.; Mazzanti, G.; Wesdorp, L.H.; Marangoni, A.G. Estimating Thermodynamic Properties of Pure Triglyceride Systems Using the Triglyceride Property Calculator. J. Am. Oil Chem. Soc. 2016, 94, 187–199. [Google Scholar] [CrossRef]
- Hunter, C.A.; Sanders, J.K.M. The nature of π-π interactions. J. Am. Chem. Soc. 1990, 112, 5525–5534. [Google Scholar] [CrossRef]
- Ravotti, R.; Fellmann, O.; Fischer, L.J.; Stamatiou, A.; Worlitschek, J. Investigation of the Thermal Properties of Diesters from Methanol, 1-Pentanol, and 1-Decanol as Sustainable Phase Change Materials. Materials 2020, 13, 810. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Burdock, G.A. Fenaroli’s Handbook of Flavor Ingredients, 6th ed.; CRC Press: Boca Raton, FL, USA, 2009; ISBN 9781420090772. [Google Scholar]
- Fahlbusch, K.G.; Hammerschmidt, F.J.; Panten, J.; Pickenhagen, W.; Schatkowski, D.; Bauer, K.; Garbe, D.; Surburg, H. Flavors and Fragrances. In Ullmann’s Encyclopedia of Industrial Chemistry; Wiley-VCH Verlag GmbH and Co.: Weinheim, Germany, 2003; ISBN 9783527306732. [Google Scholar]
- Kurokawa, H.; Ohshima, M.; Sugiyama, K.; Miura, H. Methanolysis of polyethylene terephthalate (PET) in the presence of aluminium tiisopropoxide catalyst to form dimethyl terephthalate and ethylene glycol. Polym. Degrad. Stab. 2003, 79, 529–533. [Google Scholar] [CrossRef]
- Lillwitz, L.D. Production of dimethyl-2,6-naphthalenedicarboxylate: Precursor to polyethylene naphthalate. J. Am. Chem. Soc. 1990, 221, 337–358. [Google Scholar] [CrossRef]
- Otake, S.; Yoda, S. Decomposition of Polyethylene 2,6-Naphthalene Dicarboxylate to Constituent Monomers Using Supercritical Methanol. Polym. J. 1999, 31, 714–716. [Google Scholar]
- Castellón, C.; Günther, E.; Mehling, H.; Hiebler, S.; Cabeza, L.F. Determination of the enthalpy of PCM as a function of temperature using a heat-flux DSC—A study of different measurement procedures and their accuracy. Int. J. Energy Res. 2008, 32, 1258–1265. [Google Scholar] [CrossRef]
- Perkin Elmer Guide. Introduction to Thermogravimetric Analysis. Available online: https://www.perkinelmer.com/lab-solutions/resources/docs/faq_beginners-guide-to-thermogravimetric-analysis_009380c_01.pdf (accessed on 21 August 2020).
- Linseis Workshop. Measuring Thermal Conductivity of PCMs. Available online: https://thermalmaterials.org/workshops/workshop_thermalconductivity2015/presentations/transient-hot-bridge/at_download/file (accessed on 15 March 2020).
- Dreisbach, R.R. Physical Properties of Chemical Compounds. Adv. Chem. 1961, 22, 3–486. [Google Scholar] [CrossRef] [Green Version]
- Bunn, C.W. The melting points of chain polymers. J. Polym. Sci. 1996, 34, 799–819. [Google Scholar] [CrossRef]
- Clar, E.; Kemp, W.; Stewart, D.G. The significance of Kekulé structures for the stability of aromatic systems. Tetrahedron 1957, 3, 325–333. [Google Scholar] [CrossRef]
- Sarbu, I.; Sebarchievici, C. A Comprehensive Review of Thermal Energy Storage. Sustainability 2018, 10, 191. [Google Scholar] [CrossRef] [Green Version]
- Trahanovsky, S.W.; Ong, C.C.; Pataky, J.G.; Weitl, F.L.; Muellen, P.W.; Clardy, J.C.; Hansen, R.S. Organic oxalates. VI. Pyrolysis of di(.alpha.-substituted)benzyl oxalates. J. Org. Chem. 1971, 36, 3575–3579. [Google Scholar] [CrossRef]
- Zmywaczyk, J.; Zbi´nkowski, P.; Smogór, H.; Olejnik, A.; Koniorczyk, P. Cooling of High-Power LED Lamp Using a Commercial Paraffin Wax. Int. J. Thermophys. 2017, 38, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Wei, X.; Luo, T. Chain length effect on thermal transport in amorphous polymers and a structure–thermal conductivity relation. Phys. Chem. Chem. Phys. 2019, 21, 15523–15530. [Google Scholar] [CrossRef] [PubMed]
- Speight, J.G.; Lange, N.A. Lange’s Handbook of Chemistry, 16th ed.; McGraw-Hill: New York, NY, USA, 2005; pp. 2758–2807. ISBN 0-07-143220-5. [Google Scholar]
- Bogatov, G.F.; Lapin, V.B.; Rastorguev Yu, L. Thermal Conductivity of Naphthalene Hydrocarbons in the Liquid State. I. Naphthalene and Methylnaphthalene. Viniti 1987, 1–15. Available online: http://www.ddbst.com/en/EED/PCP/TCN_C123.php (accessed on 10 September 2020).
- Sun, X.; Liu, L.; Mo, Y.; Li, J.; Li, C. Enhanced thermal energy storage of a paraffin-based phase change material (PCM) using nano carbons. Appl. Therm. Eng. 2020, 181, 115992. [Google Scholar] [CrossRef]
- Lazzarin, R.M.; Mancin, S.; Noro, M.; Righetti, G. Hybrid PCM-aluminium foams’ thermal storages: An experimental study. Int. J. Low-Carbon Technol. 2018, 13, 286–291. [Google Scholar] [CrossRef]
- Delgado, W.; Stamatiou, A.; Maranda, S.; Waser, R.; Worlitschek, J. Assessment of High Power Latent Heat Storage Solutions. In Proceedings of the Eurotherm Seminar 112 Advances in Thermal Energy Storage, Lleida, Spain, 15–17 May 2019; ISBN 978-84-9144-155-7. [Google Scholar]
- Amagour, M.E.H.; Rachek, A.; Bennajah, M.; Ebn Touhami, M. Experimental investigation and comparative performance analysis of a compact finned-tube heat exchanger uniformly filled with a phase change material for thermal energy storage. Energy Convers. Manag. 2018, 165, 137–151. [Google Scholar] [CrossRef]
- Waser, R.; Maranda, S.; Stamatiou, A.; Zaglio, M.; Worlitschek, J. Modeling of solidification including supercooling effects in a fin-tube heat exchanger based latent heat storage. Sol. Energy 2020, 200, 10–21. [Google Scholar] [CrossRef]
- Agency for Toxic Substances and Disease Registry (ATSDR). Case Studies in Environmental Medicine Toxicity of Polycyclic Aromatic Hydrocarbons (PAHs). 2009. Available online: https://www.atsdr.cdc.gov/csem/pah/docs/pah.pdf (accessed on 5 May 2020).
- Baali, A.; Yahyaoui, A. Polycyclic Aromatic Hydrocarbons (PAHs) and Their Influence to Some Aquatic Species. In Biochemical Toxicology—Heavy Metals and Nanomaterials; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef] [Green Version]
- Wilderbeek, H.T.A.; Van der Meer, M.G.M.; Jansen, M.A.G.; Nelissen, L.; Fischer, H.R.; Van Es, S.; Bastiaansen, C.W.M.; Lub, J.; Broer, D. Synthesis and properties of phenyl benzoate-based and biphenyl-based liquid crystalline thiol-ene monomers. Liq. Cryst. Chem. 2010, 1, 93–108. [Google Scholar] [CrossRef]
- World Health Organization. World Health Organization Model List of Essential Medicines: 21st List; World Health Organization: Geneva, Switzerland, 2019; Available online: https://apps.who.int/iris/bitstream/handle/10665/325771/WHO-MVP-EMP-IAU-2019.06-eng.pdf?sequence=1&isAllowed=y (accessed on 1 February 2020).
- Fakhrpour, G.; Bagheri, S.; Golriz, M.; Shekari, M. Degradation kinetics of PET/PEN blend nanocomposites using differential isoconversional and differential master plot approaches. J. Therm. Anal. Calorim. 2016, 124, 917–924. [Google Scholar] [CrossRef]
- Farid, M.; Khudhair, S.; Razack, S.; Al-Hallaj, S. A review on phase change energy storage: Materials and applications. Energy Convers. Manag. 2004, 45, 1597–1615. [Google Scholar] [CrossRef]
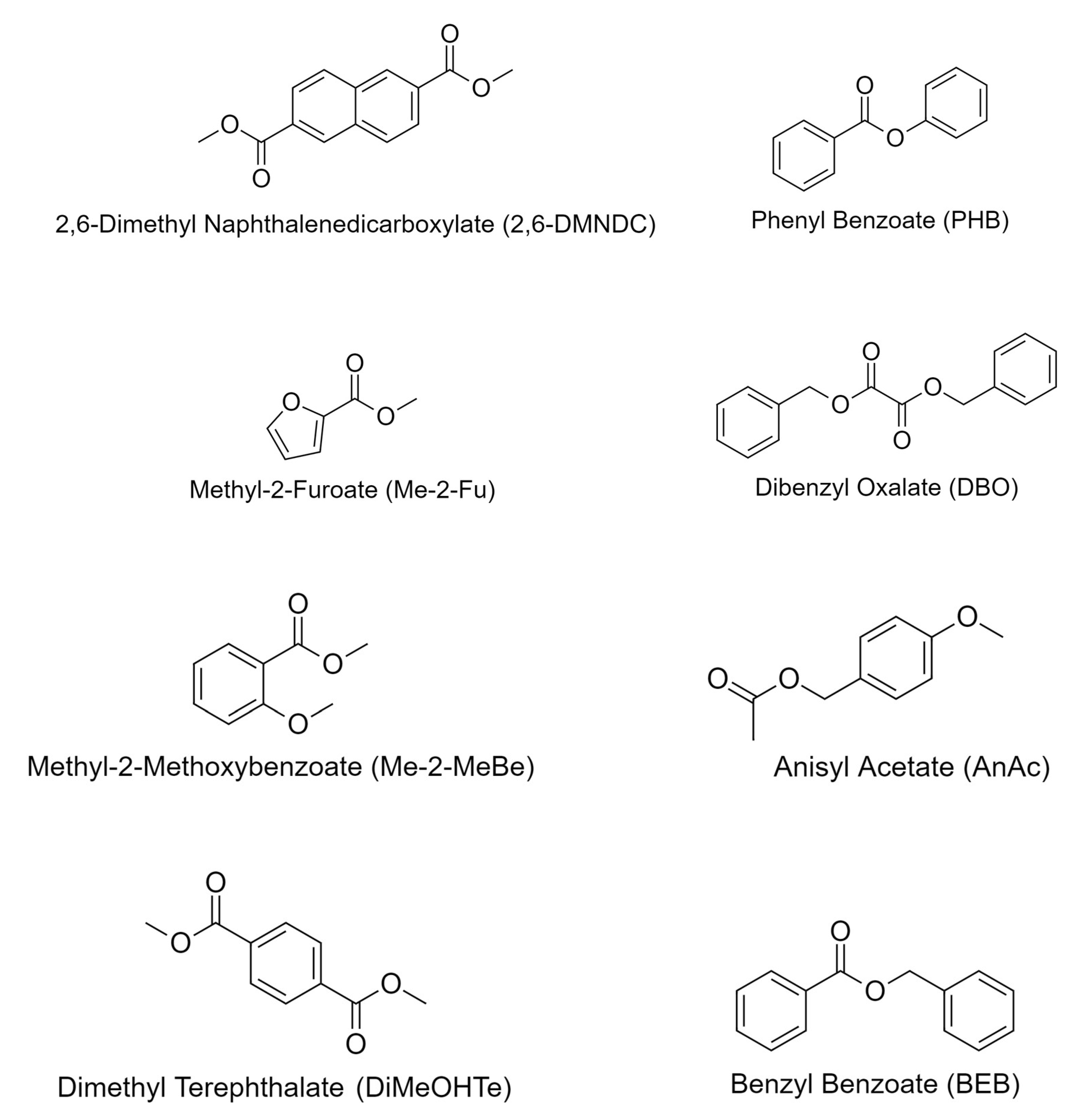



| Ester | Structure | Carbon Number | Measuring T ( C) | [g/mL] | lit. [g/mL] |
|---|---|---|---|---|---|
| DBO | (CO2CH2C6H5)2 | 16 | 90 | 1.22 ± 0.01 | NA |
| 2,6-DMNDC | C10H6(CO2CH3)2 | 14 | 210 | 1.09 ± 0.01 | NA |
| BEB | C6H5COOCH2C6H5 | 14 | RT | 1.12 ± <0.01 | 1.12 (20 C) |
| PHB | C6H5CO2C6H5 | 13 | 90 | 1.19 ± 0.01 | NA |
| DiMeOHTe | C6H4-1,4-(CO2CH3)2 | 10 | 155 | 1.09 ± 0.01 | NA |
| AnAc | CH3CO2CH2C6H4-4-(OCH3) | 9 | RT | 1.11 ± <0.01 | 1.11 (25 C) |
| Me-2-MeBe | CH3OC6H4C02CH3 | 9 | RT | 1.16 ± 0.01 | 1.16 (25 C) |
| Me-2-Fu | C6H6O3 | 6 | RT | 1.18 ± 0.01 | 1.18 (25 C) |
| Ester | Structure | Carbon Number | MW [g/mol] | Tc [Onset, C] | Tm [Onset, C] | Supercooling [C] | H [J/g] | H [kJ/mol] | Volumetric H [J/mL] | Tdegr onset [C] | Tdegr endset [C] |
|---|---|---|---|---|---|---|---|---|---|---|---|
| DBO | (CO2CH2C6H5)2 | 16 | 270.3 | 50.8 ± 10.6 | 78.5 ± 0.3 | 27.7 ± 10.4 | 131.0 ± 7.0 | 35.4 ± 1.9 | 159.8 ± 8.6 | 175 ± 5 | 530 ± 15 |
| 2,6-DMNDC | C10H6(CO2CH3)2 | 14 | 244.2 | 177.0 ± 1.2 | 190.2 ± 0.8 | 13.2 ± 1.3 | 158.4 ± 3.9 | 38.7 ± 0.9 | 172.7 ± 4.2 | 220 ± 13 | 325 ± 10 |
| BEB | C6H5COOCH2C6H5 | 14 | 212.2 | −19.1 * ± 1.6 | 18.1 ** ± 1.2 | 37.2 ± 2.1 | NA | NA | NA | 148 ± 3 | 282 ± 13 |
| PHB | C6H5CO2C6H5 | 13 | 198.2 | 19.0 ± 1.4 | 68.3 ± 0.1 | 49.3 ± 1.3 | 102.0 ± 4.5 | 20.2 ± 0.9 | 121.4 ± 5.3 | 143 ± 13 | 273 ± 10 |
| DiMeOHTe | C6H4-1,4-(CO2CH3)2 | 10 | 194.2 | 131.4 ± 1.9 | 140.2 ± 0.1 | 8.7 ± 1.9 | 154.6 ± 1.5 | 30.0 ± 0.3 | 168.5 ± 1.6 | 150 ± 5 | 244 ± 5 |
| AnAc | CH3CO2CH2C6H4-4-(OCH3) | 9 | 180.2 | NA | −15.7 ** ± 0.5 | NA | NA | NA | NA | 123 ± 4 | 237 ± 6 |
| Me-2-MeBe | CH3OC6H4C02CH3 | 9 | 166.2 | NA | NA | NA | NA | NA | NA | 122 ± 3 | 217 ± 8 |
| Me-2-Fu | C6H6O3 | 6 | 126.1 | −46.9 ± 1.3 | −3.8 ± 0.3 | 43.1 ± 1.5 | 97.7 ± 5.9 | 12.3 ± 0.7 | 115.2 ± 7.0 | 67 ± 21 | 213 ± 49 |
| Ester | Structure | Carbon Number | MW [g/mol] | Tm lit. (Chemical Suppliers) |
|---|---|---|---|---|
| DBO | (CO2CH2C6H5)2 | 16 | 270.3 | 75–78 |
| 2,6-DMNDC | C10H6(CO2CH3)2 | 14 | 244.2 | 187–193 |
| BEB | C6H5COOCH2C6H5 | 14 | 212.2 | 18 |
| PHB | C6H5CO2C6H5 | 13 | 198.2 | 71 |
| DiMeOHTe | C6H4-1,4-(CO2CH3)2 | 10 | 194.2 | 142 |
| AnAc | CH3CO2CH2C6H4-4-(OCH3) | 9 | 180.2 | NA |
| Me-2-MeBe | CH3OC6H4C02CH3 | 9 | 166.2 | NA |
| Me-2-Fu | C6H6O3 | 6 | 126.1 | NA |
| Ester | Structure | Carbon Number | I [mA] | Measuring T [C] | [W/(m·K)] |
|---|---|---|---|---|---|
| DBO | (CO2CH2C6H5)2 | 16 | 50 | 100 | 0.16 ± 0.01 |
| 2,6-DMNDC | C10H6(CO2CH3)2 | 14 | NA | 210 | NA |
| BEB | C6H5COOCH2C6H5 | 14 | 50 | 30 | 0.17 ± 0.01 |
| PHB | C6H5CO2C6H5 | 13 | 50 | 90 | 0.17 ± 0.01 |
| DiMeOHTe | C6H4-1,4-(CO2CH3)2 | 10 | NA | 160 | NA |
| AnAc | CH3CO2CH2C6H4-4-(OCH3) | 9 | 45 | 30 | 0.19 ± 0.01 |
| Me-2-MeBe | CH3OC6H4CO2CH3 | 9 | 50 | 30 | 0.19 ± 0.01 |
| Me-2-Fu | C6H6O3 | 6 | 45 | 30 | 0.20 ± 0.01 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ravotti, R.; Fellmann, O.; Fischer, L.J.; Worlitschek, J.; Stamatiou, A. Assessment of the Thermal Properties of Aromatic Esters as Novel Phase Change Materials. Crystals 2020, 10, 919. https://doi.org/10.3390/cryst10100919
Ravotti R, Fellmann O, Fischer LJ, Worlitschek J, Stamatiou A. Assessment of the Thermal Properties of Aromatic Esters as Novel Phase Change Materials. Crystals. 2020; 10(10):919. https://doi.org/10.3390/cryst10100919
Chicago/Turabian StyleRavotti, Rebecca, Oliver Fellmann, Ludger J. Fischer, Jörg Worlitschek, and Anastasia Stamatiou. 2020. "Assessment of the Thermal Properties of Aromatic Esters as Novel Phase Change Materials" Crystals 10, no. 10: 919. https://doi.org/10.3390/cryst10100919







