Effect of Molecular Structure of Organic Acids on the Crystal Habit of α-CaSO4·0.5H2O from Phosphogypsum
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of α-CaSO4·0.5H2O Crystals
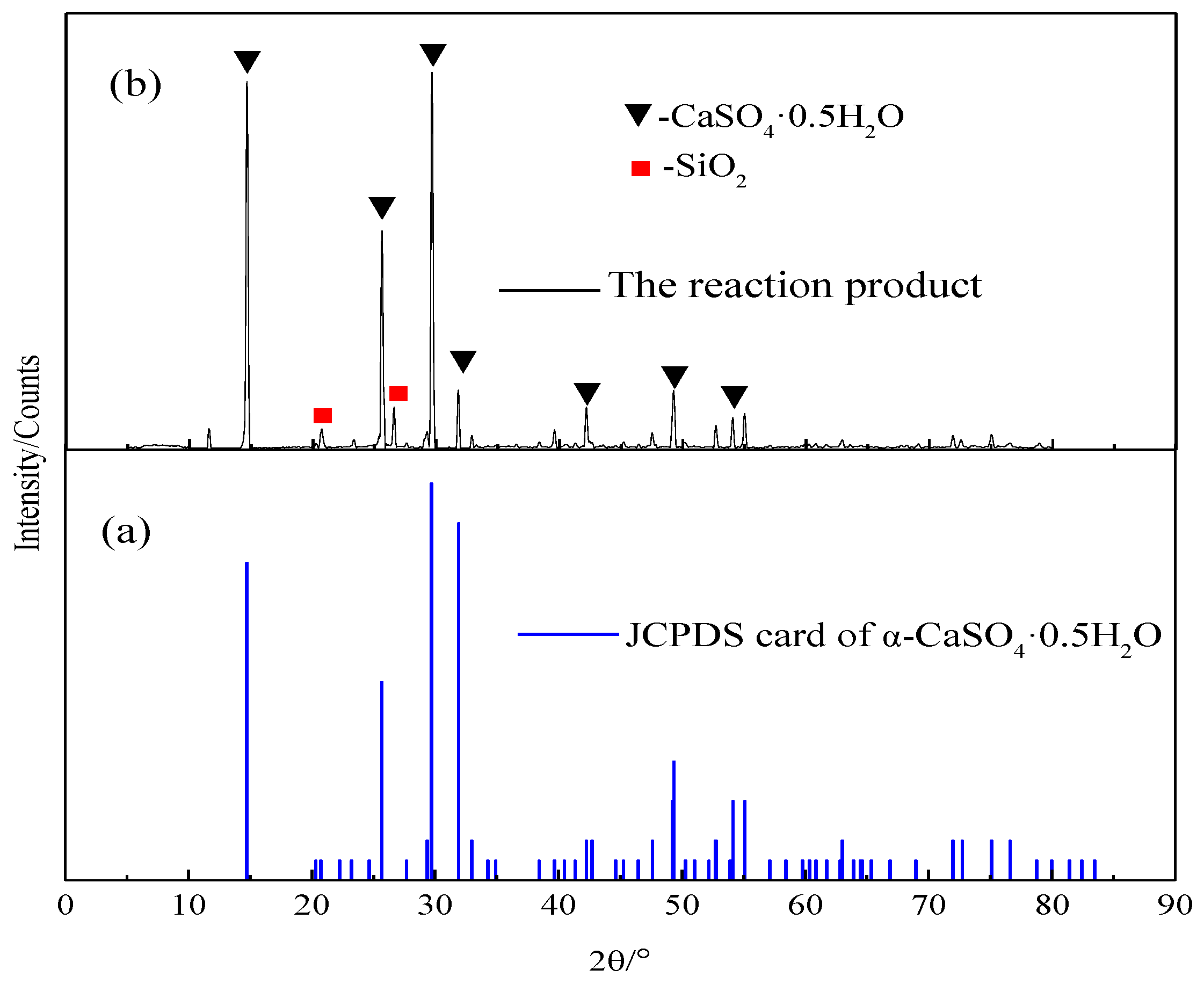
2.3. Characterization Methods
3. Results and Discussion
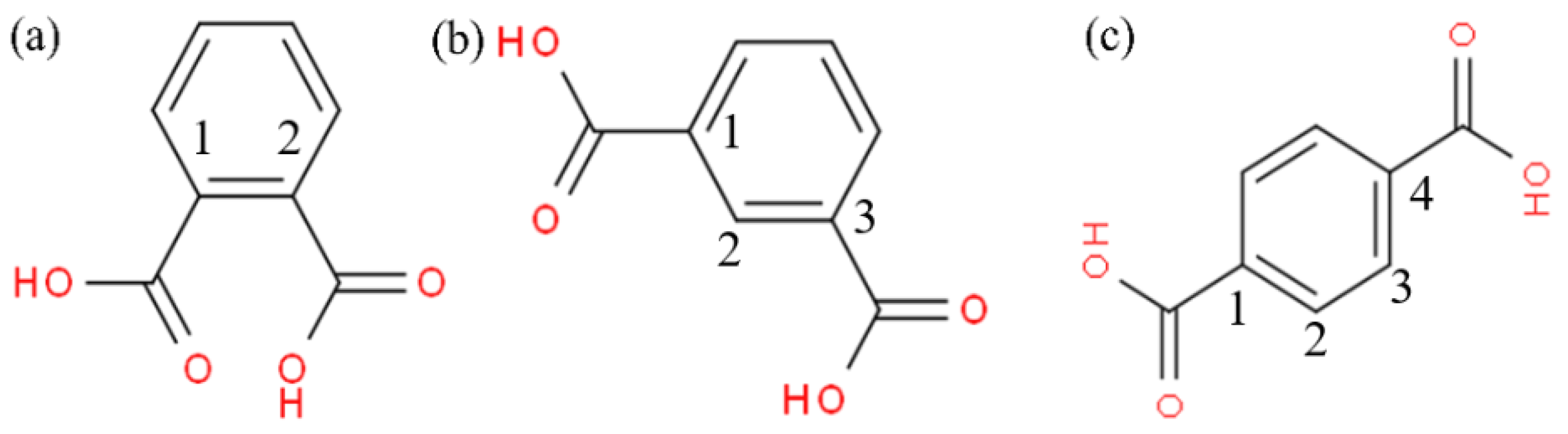
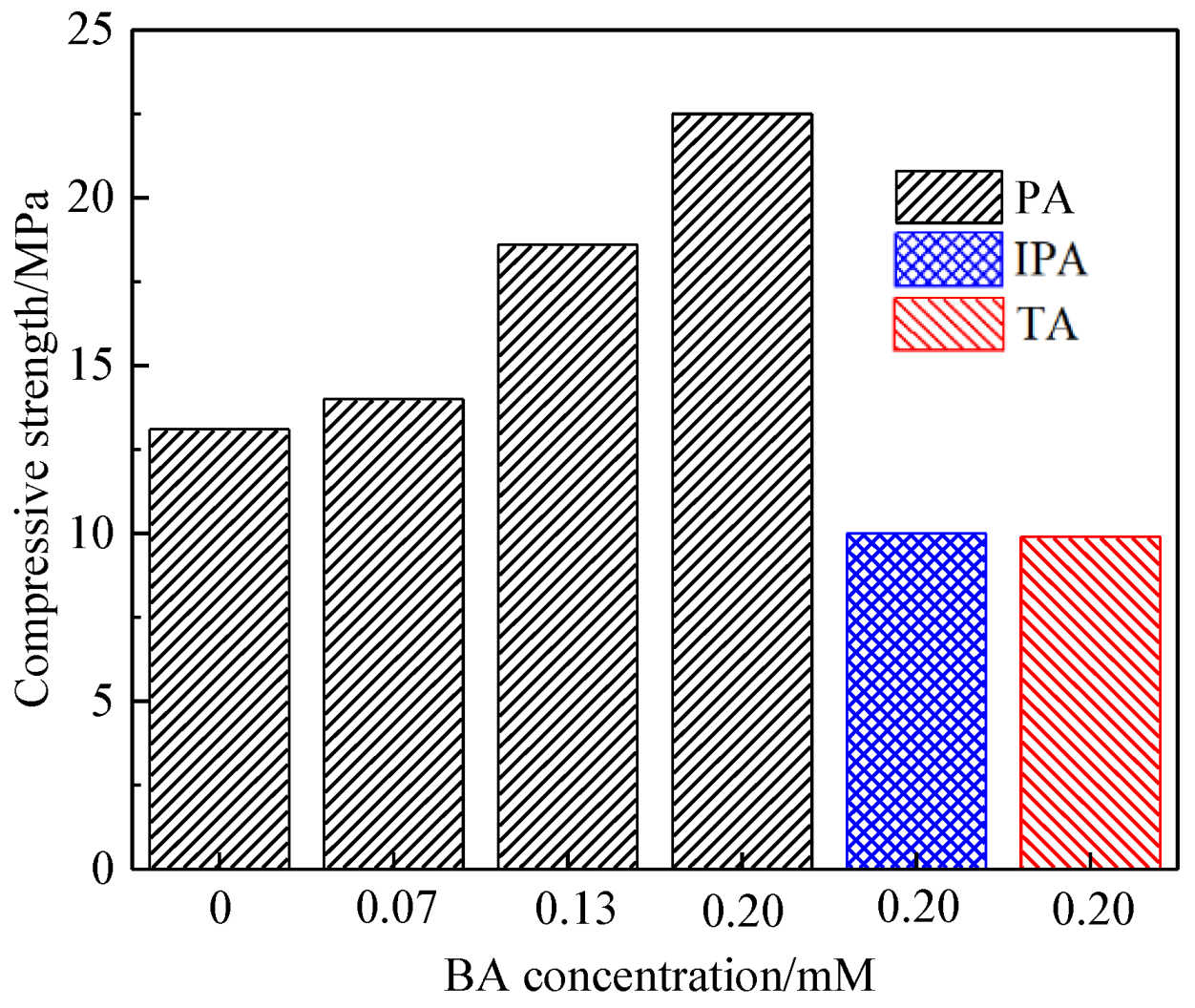
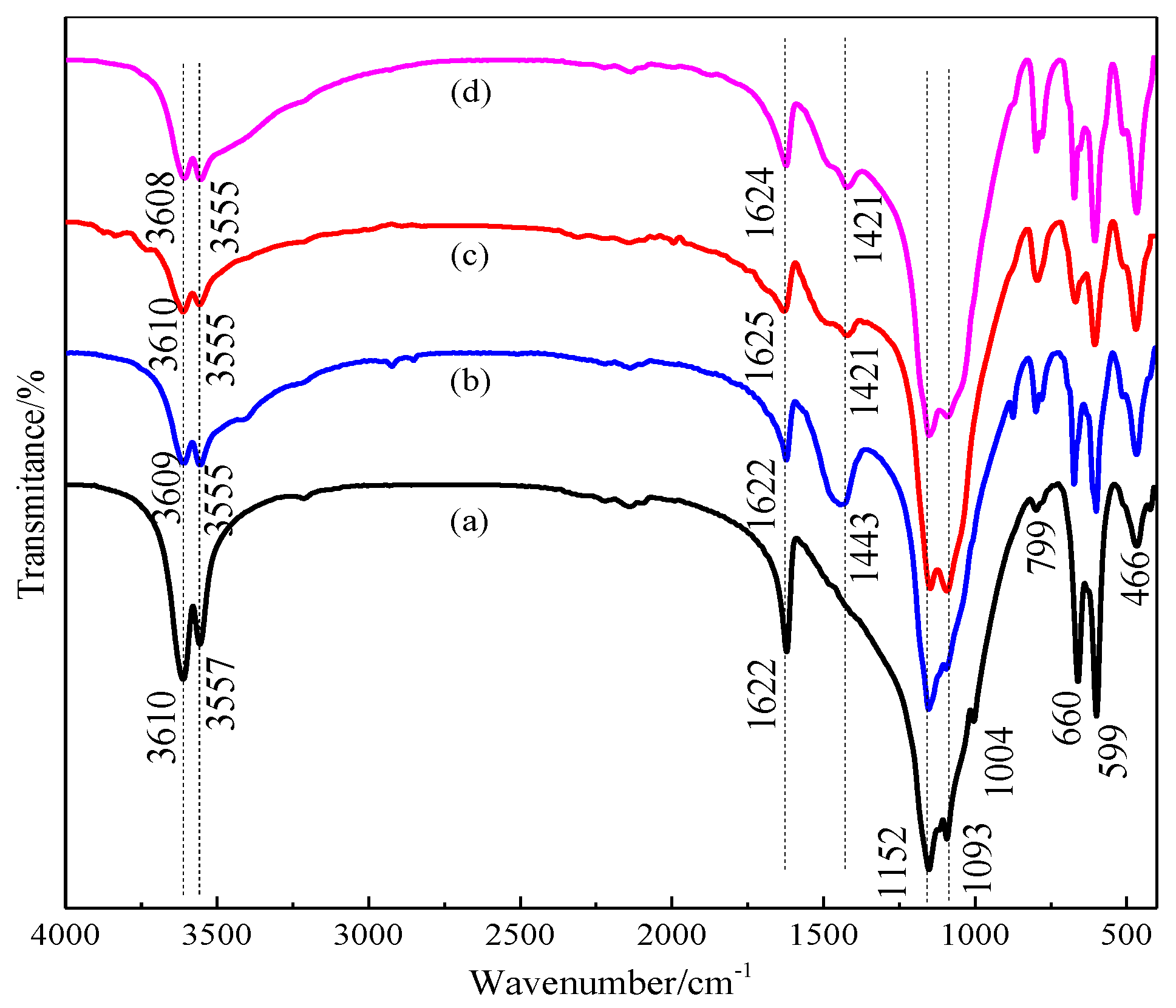
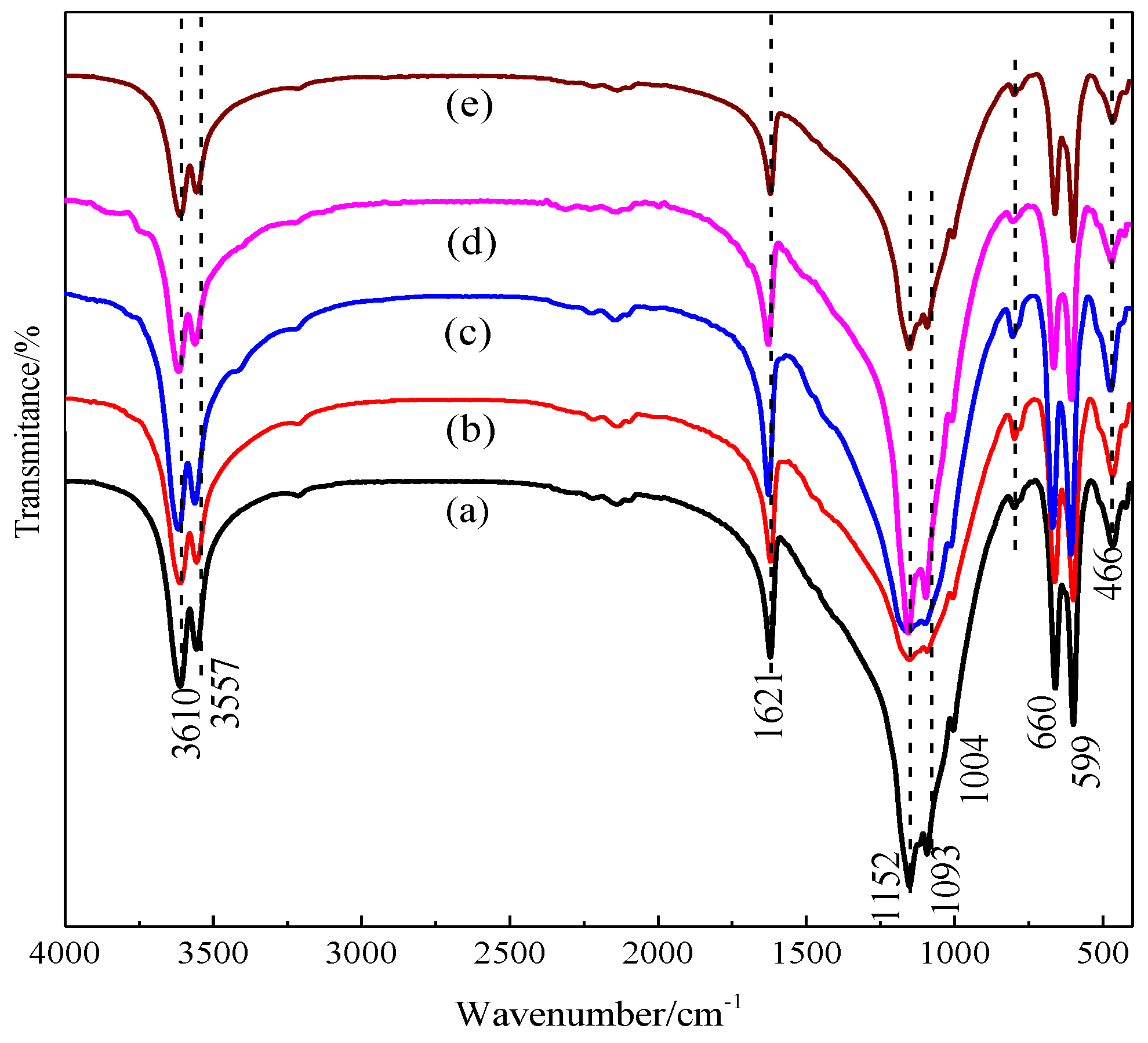
3.1. Effect of Benzenedicarboxylic Acid on the Crystal Habit of α-CaSO4·0.5H2O
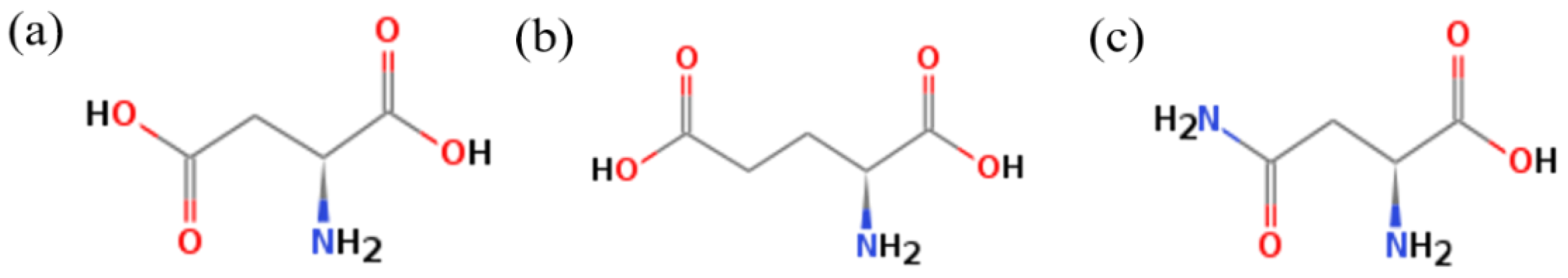
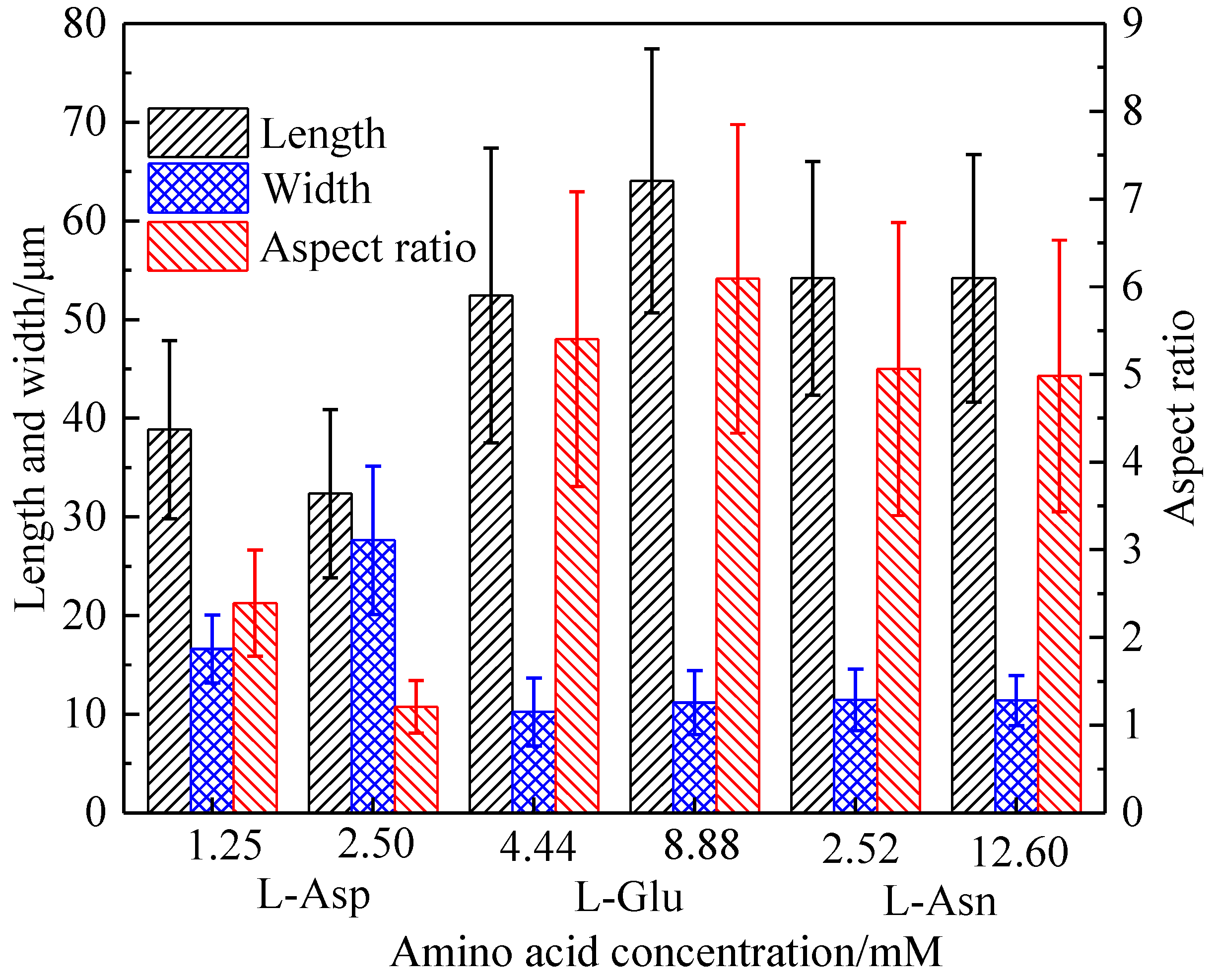
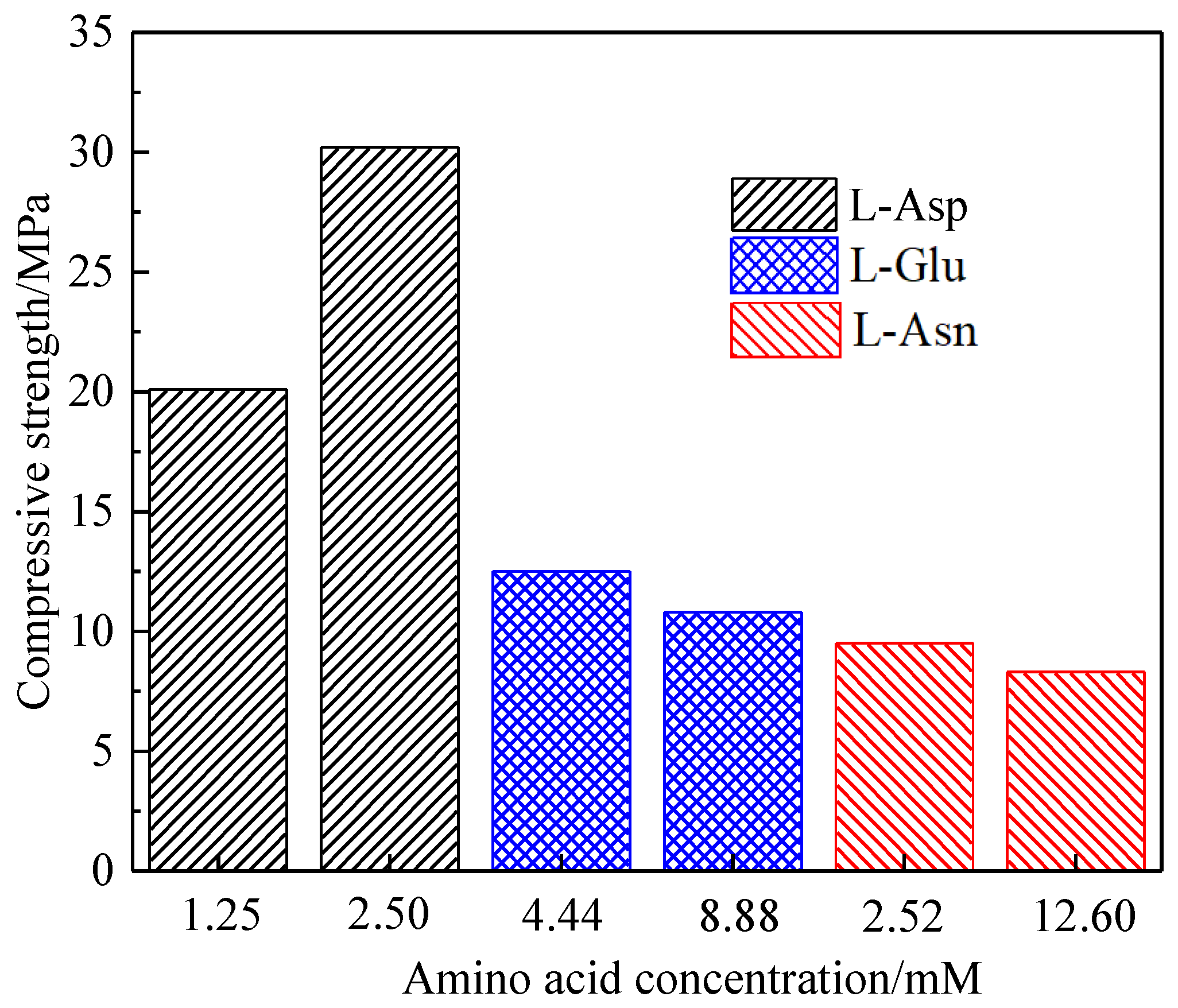
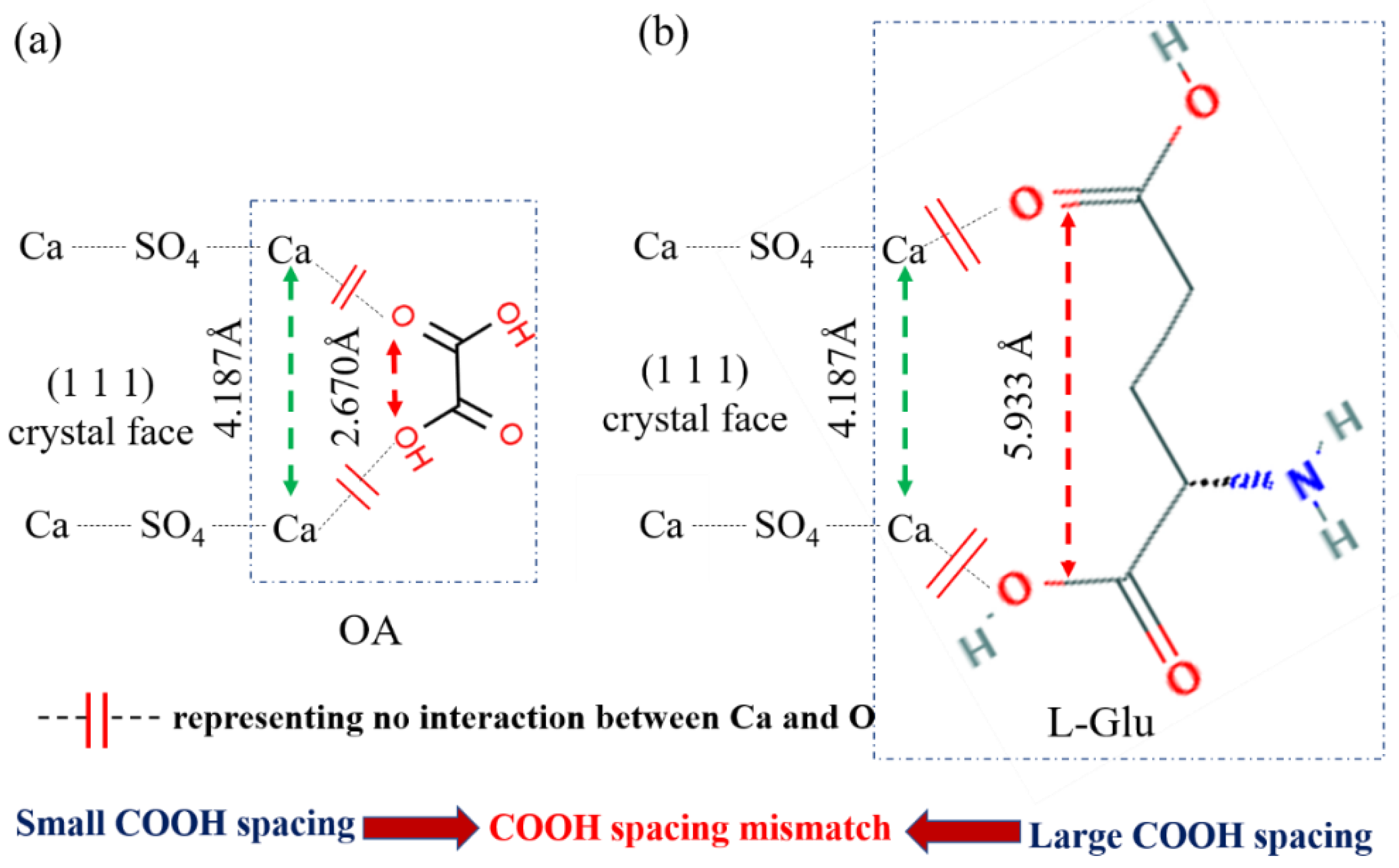
3.2. Effect of Amino Acid on the Crystal Habit of α-CaSO4·0.5H2O
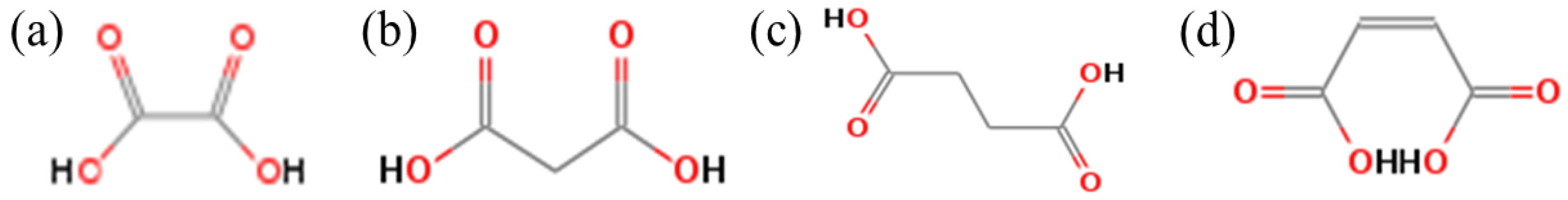
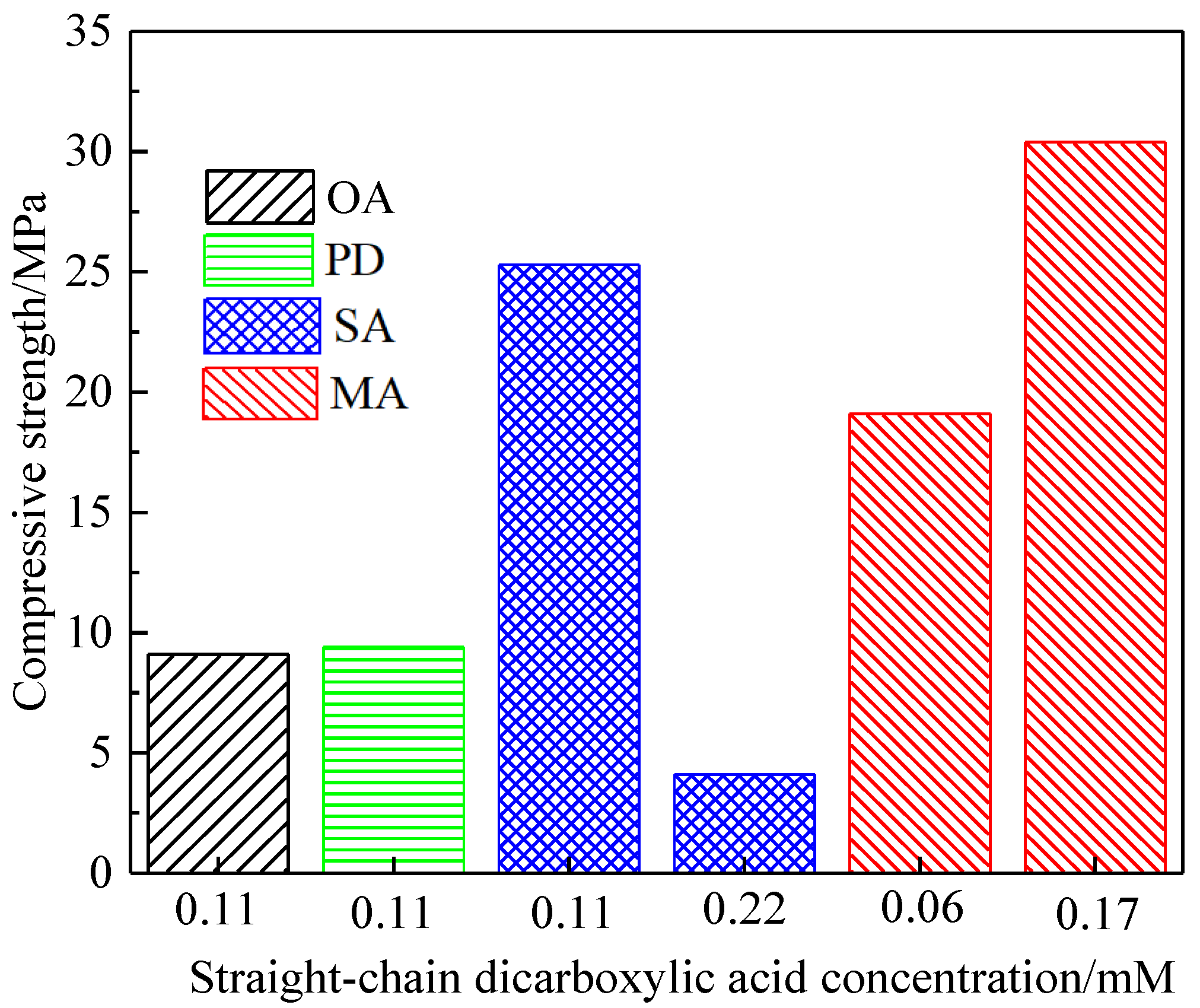
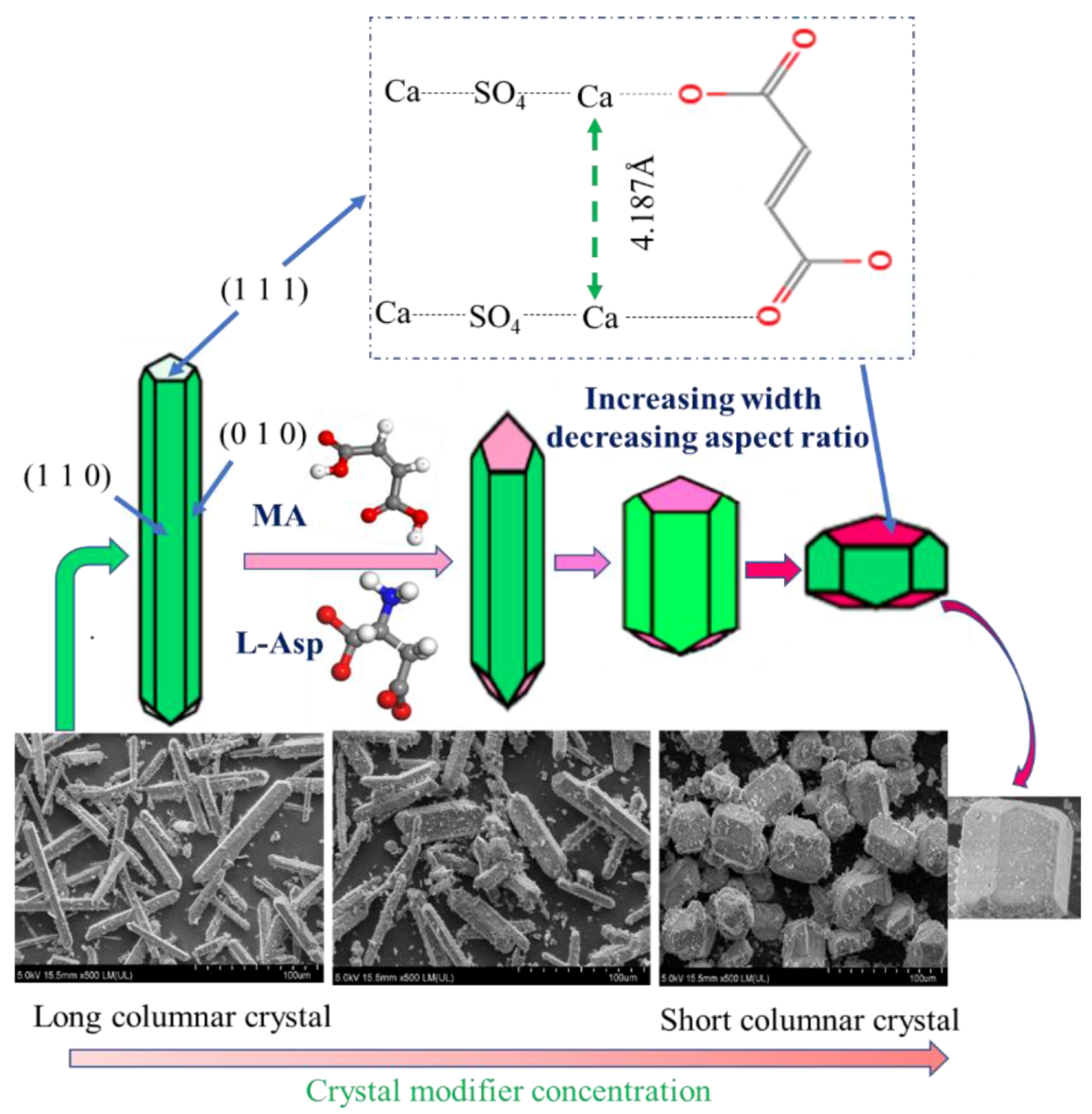
3.3. Effect of Straight-Chain Dicarboxylic Acid on the Crystal Habit of α-CaSO4·0.5H2O
3.4. Control Mechanism of Organic Acids for the Crystal Habit of α-CaSO4·0.5H2O
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Hammas, I.; Horchani-Naifer, K.; Férid, M. Solubility study and valorization of phosphogypsum salt solution. Int. J. Miner. Process. 2013, 123, 87–93. [Google Scholar] [CrossRef]
- Romero-Hermida, I.; Morales-Flórez, V.; Santos, A.; Villena, A.; Esquivias, L. Technological proposals for recycling industrial wastes for environmental applications. Minerals-Basel 2014, 4, 746–757. [Google Scholar] [CrossRef]
- Rashad, A.M. Phosphogypsum as a construction material. J. Clean. Prod. 2017, 166, 732–743. [Google Scholar] [CrossRef]
- Islam, G.M.S.; Chowdhury, F.H.; Raihan, M.T.; Amit, S.K.S.; Islam, M.R. Effect of phosphogypsum on the properties of portland cement. Procedia Eng. 2017, 171, 744–751. [Google Scholar] [CrossRef]
- Hua, S.D.; Wang, K.J.; Yao, X. Developing high performance phosphogypsum-based cementitious materials for oil-well cementing through a step-by-step optimization method. Cem. Concr. Compos. 2016, 72, 299–308. [Google Scholar] [CrossRef]
- Yang, L.; Zhang, Y.S.; Yan, Y. Utilization of original phosphogypsum as raw material for the preparation of self-leveling mortar. J. Clean. Prod. 2016, 127, 204–213. [Google Scholar] [CrossRef]
- Ye, X.D. Current situation, existing problems and suggestions of phosphogypsum utilization in China in 2018. Phosphate Compound Fert. 2018, 34, 1–4. [Google Scholar]
- Jiang, G.M.; Wang, H.; Chen, Q.S.; Zhang, X.M.; Wu, Z.B.; Guan, B.H. Preparation of alpha-calcium sulfate hemihydrate from FGD gypsum in chloride-free Ca(NO3)2 solution under mild conditions. Fuel 2016, 174, 235–241. [Google Scholar] [CrossRef]
- Guan, B.H.; Yang, L.; Fu, H.L.; Kong, B.; Li, T.Y.; Yang, L.C. α-calcium sulfate hemihydrate preparation from FGD gypsum in recycling mixed salt solutions. Chem. Eng. J. 2011, 174, 296–303. [Google Scholar] [CrossRef]
- Zhang, R.; Li, Y.J.; Liu, J.; Zhao, Y.Y.; Jiang, Y.D. Utilization of phosphogypsum and treatment of the impure elements. Conserv. Utilization Miner. Resour. 2015, 50–54. [Google Scholar] [CrossRef]
- Guan, Q.J.; Sun, W.; Hu, Y.H.; Yin, Z.G.; Zhang, C.H.; Guan, C.P.; Zhu, X.N.; Ahmed Khoso, S. Simultaneous control of particle size and morphology of α-CaSO4·½H2O with organic additives. J. Am. Ceram. Soc. 2019, 102, 2441–2449. [Google Scholar]
- Shen, Z.X.; Guan, B.H.; Fu, H.L.; Yang, L.C. Effect of potassium sodium tartrate and sodium citrate on the preparation of α-calcium sulfate hemihydrate from flue gas desulfurization gypsum in a concentrated electrolyte solution. J. Am. Ceram. Soc. 2009, 92, 2894–2899. [Google Scholar] [CrossRef]
- Duan, Z.Y.; Li, J.X.; Li, T.G.; Zheng, S.R.; Han, W.M.; Geng, Q.Y.; Guo, H.B. Influence of crystal modifier on the preparation of α-hemihydrate gypsum from phosphogypsum. Constr. Build. Mater. 2017, 133, 323–329. [Google Scholar] [CrossRef]
- Li, F.; Liu, J.L.; Yang, G.Y.; Pan, Z.Y.; Ni, X.; Xu, H.; Huang, Q. Effect of pH and succinic acid on the morphology of α-calcium sulfate hemihydrate synthesized by a salt solution method. J. Cryst. Growth 2013, 374, 31–36. [Google Scholar] [CrossRef]
- Guan, Q.J.; Sun, W.; Hu, Y.H.; Yin, Z.G.; Guan, C.P. Synthesis of α-CaSO4·0.5H2O from flue gas desulfurization gypsum regulated by C4H4O4Na2·6H2O and NaCl in glycerol-water solution. RSC Adv. 2017, 7, 27807–27815. [Google Scholar] [CrossRef]
- Guan, Q.J.; Tang, H.H.; Sun, W.; Hu, Y.H.; Yin, Z.G. Insight into influence of glycerol on preparing α-CaSO4·1/2H2O from flue gas desulfurization gypsum in glycerol–water solutions with succinic acid and NaCl. Ind. Eng. Chem. Res. 2017, 56, 9831–9838. [Google Scholar] [CrossRef]
- Mi, Y.; Chen, D.Y.; Wang, S.Z. Utilization of phosphogypsum for the preparation of α-calcium sulfate hemihydrate in chloride-free solution under atmospheric pressure. J. Chem. Technol. Biotechnol. 2018, 93, 2371–2379. [Google Scholar] [CrossRef]
- Shao, D.D.; Zhao, B.; Zhang, H.Q.; Wang, Z.G.; Shi, C.J.; Cao, J.L. Preparation of large-grained α-high strength gypsum with FGD gypsum. Cryst. Res. Technol. 2017, 52, 1700078. [Google Scholar] [CrossRef]
- Lu, W.D.; Ma, B.G.; Su, Y.; He, X.Y.; Jin, Z.; Qi, H.H. Preparation of α-hemihydrate gypsum from phosphogypsum in recycling CaCl2 solution. Constr. Build. Mater. 2019, 214, 399–412. [Google Scholar] [CrossRef]
- Guan, B.H.; Kong, B.; Fu, H.L.; Yu, J.; Jiang, G.M.; Yang, L. Pilot scale preparation of α-calcium sulfate hemihydrate from FGD gypsum in Ca–K–Mg aqueous solution under atmospheric pressure. Fuel 2012, 98, 48–54. [Google Scholar] [CrossRef]
- Li, X.B.; Zhang, Q.; Ke, B.L.; Wang, X.C.; Li, L.J.; Li, X.H.; Mao, S. Insight into the effect of maleic acid on the preparation of α-hemihydrate gypsum from phosphogypsum in Na2SO4 solution. J. Cryst. Growth 2018, 493, 34–40. [Google Scholar] [CrossRef]
- Tan, H.; Dong, F. Morphological regulation of calcium sulfate hemihydrate from phosphogypsum. Materialwissenschaft und Werkstofftechnik 2017, 48, 1191–1196. [Google Scholar] [CrossRef]
- Ma, B.G.; Lu, W.D.; Su, Y.; Li, Y.B.; Gao, C.; He, X.Y. Synthesis of α-hemihydrate gypsum from cleaner phosphogypsum. J. Clean. Prod. 2018, 195, 396–405. [Google Scholar] [CrossRef]
- Li, X.B.; Zhang, Q.; Shen, Z.H.; Li, L.J.; Li, X.H.; Mao, S. L-aspartic acid: A crystal modifier for preparation of hemihydrate from phosphogypsum in CaCl2 solution. J. Cryst. Growth 2019, 511, 48–55. [Google Scholar] [CrossRef]
- Wang, X.S.; Zhou, M.; Ke, X.; Tan, R.Q.; Chen, Y.C.; Hou, H.B. Synthesis of alpha hemihydrate particles with lithium and carboxylates via the hydrothermal method. Powder Technol. 2017, 317, 293–300. [Google Scholar] [CrossRef]
- Badens, E.; Veesler, S.; Boistelle, R. Crystallization of gypsum from hemihydrate in presence of additives. J. Cryst. Growth 1999, 198, 704–709. [Google Scholar] [CrossRef]
- Teng, W.; Wang, J.; Wu, J.; Du, Y.; Jia, X.; Li, H.; Wang, T. Rapid synthesis of alpha calcium sulfate hemihydrate whiskers in glycerol-water solution by using flue-gas-desulfurization gypsum solid waste. J. Cryst. Growth 2018, 496–497, 24–30. [Google Scholar] [CrossRef]
- Jia, C.Y.; Chen, Q.S.; Zhou, X.; Wang, H.; Jiang, G.M.; Guan, B.H. Trace NaCl and Na2EDTA mediated synthesis of α-calcium sulfate hemihydrate in glycerol–water solution. Ind. Eng. Chem. Res. 2016, 55, 9189–9194. [Google Scholar] [CrossRef]
- Liu, X.F.; Peng, J.H.; Zhang, J.X.; Qu, J.D.; Li, M. Effect of organic diacid carbon chain length on crystal morphology of α-calcium sulfate hemihydrate in preparation from flue gas desulphurization gypsum. Appl. Mech. Mater. 2012, 253–255, 542–545. [Google Scholar] [CrossRef]
- Zhao, Z.M.; Luan, Y.; Quan, S.C.; Wu, J.L. The effect of functional groups for the organic acid on the crystal transition of phosphogypsum crystal. J. Build. Mater. 2018, 21, 247–252. [Google Scholar]
- Peng, J.H.; Chen, M.F.; Zhang, J.X.; Qu, J.D.; Zou, C.Y. Influnce of organic acid structure on crystal morphology of α-hemihydrate desulfurization gypsum and Its crystal modification mechanism. J. Sichuan Univ. (Eng. Sci. Ed.) 2012, 44, 116–172. [Google Scholar]
- Mandal, P.K.; Mandal, T.K. Anion water in gypsum (CaSO4·2H2O) and hemihydrate (CaSO4·1/2H2O). Cem. Concr. Res. 2002, 32, 313–316. [Google Scholar] [CrossRef]
- Mao, J.W.; Jiang, G.M.; Chen, Q.S.; Guan, B.H. Influences of citric acid on the metastability of α-calcium sulfate hemihydrate in CaCl2 solution. Colloid. Surface. A Physicochem. Eng. Asp. 2014, 443, 265–271. [Google Scholar] [CrossRef]



| Items | Radioactivity Nuclide Specific Activity | Internal Exposure Index | External Exposure Index | ||
|---|---|---|---|---|---|
| 226Ra | 232Th | 40K | |||
| PG | 61.0 | 2.3 | 3.3 | 0.31 | 0.17 |
| Main materials for building | ≤1.0 | ≤1.0 | |||
| Class A decorative materials | ≤1.0 | ≤1.3 | |||
| BA Concentration/(mM) | Average Length/μm | Average Width/μm | Aspect Ratio | |
|---|---|---|---|---|
| PA | 0 | 59.04 ± 14.08 | 11.04 ± 3.64 | 5.74 ± 1.84 |
| 0.07 | 42.56 ± 10.02 | 16.09 ± 3.55 | 2.72 ± 0.70 | |
| 0.13 | 39.39 ± 9.63 | 18.02 ± 4.91 | 2.36 ± 0.91 | |
| 0.20 | 32.19 ± 6.87 | 20.73± 5.26 | 1.61 ± 0.39 | |
| IPA | 0.20 | 41.83 ± 12.72 | 10.60 ± 3.81 | 4.24 ± 1.43 |
| TA | 0.20 | 40.30 ± 11.51 | 9.46 ± 2.96 | 4.54 ± 1.60 |
| Dicarboxylic Acids Concentration/mM | Average Length/μm | Average Width /μm | Aspect Ratio | |
|---|---|---|---|---|
| OA | 0.11 | 51.31 ± 12.17 | 9.86 ± 2.74 | 5.58 ± 1.85 |
| PD | 0.11 | 51.87 ± 10.17 | 10.53 ± 2.75 | 5.19 ± 1.45 |
| SA | 0.11 | 50.14 ± 10.87 | 17.67 ± 6.71 | 3.12 ± 1.10 |
| 0.22 | 13.89 ± 7.33 | 1.47 ± 0.54 | 9.40 ± 3.10 | |
| MA | 0.06 | 59.19 ± 11.11 | 13.68 ± 4.10 | 4.68 ± 1.52 |
| 0.17 | 39.35 ± 14.69 | 18.15 ± 7.24 | 2.42 ± 1.09 | |
| Organic Acid | OA | PD | SA | MA | PA | IPA | TA | L-Asp | L-Asn | L-Glu |
|---|---|---|---|---|---|---|---|---|---|---|
| COOH separation /Å | 2.670 | 2.865 | 4.327 | 3.785 | 3.946 | 5.120 | 7.018 | 4.154 | 4.073 | 5.933 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, X.; Zhang, Q. Effect of Molecular Structure of Organic Acids on the Crystal Habit of α-CaSO4·0.5H2O from Phosphogypsum. Crystals 2020, 10, 24. https://doi.org/10.3390/cryst10010024
Li X, Zhang Q. Effect of Molecular Structure of Organic Acids on the Crystal Habit of α-CaSO4·0.5H2O from Phosphogypsum. Crystals. 2020; 10(1):24. https://doi.org/10.3390/cryst10010024
Chicago/Turabian StyleLi, Xianbo, and Qin Zhang. 2020. "Effect of Molecular Structure of Organic Acids on the Crystal Habit of α-CaSO4·0.5H2O from Phosphogypsum" Crystals 10, no. 1: 24. https://doi.org/10.3390/cryst10010024
APA StyleLi, X., & Zhang, Q. (2020). Effect of Molecular Structure of Organic Acids on the Crystal Habit of α-CaSO4·0.5H2O from Phosphogypsum. Crystals, 10(1), 24. https://doi.org/10.3390/cryst10010024




