Control of Microstructure for Co-Cr-Mo Fibers Fabricated by Unidirectional Solidification
Abstract
1. Introduction
2. Materials and Methods
3. Experimental Procedure
4. Results and Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chiba, A.; Kumagai, K.; Nomura, N.; Miyakawa, S. Pin-on-disk wear behavior in a like-on-like configuration in a biological environment of high carbon cast and low carbon forged Co-29Cr-6Mo alloys. Acta Mater. 2007, 55, 1309–1318. [Google Scholar] [CrossRef]
- Buford, A.; Goswami, T. Review of wear mechanisms in hip implants: Paper I—General. Mater. Des. 2004, 25, 385–393. [Google Scholar] [CrossRef]
- Yamanaka, K.; Mori, M.; Chiba, A. Assessment of precipitation behavior in dental castings of a Co-Cr-Mo alloy. J. Mech. Behav. Biomed. Mater. 2015, 50, 268–276. [Google Scholar] [CrossRef] [PubMed]
- Charnley, J. Arthroplasty of the Hip: A New Operation. Clin. Orthopaed. Relat. Res. 1973, 95, 4–8. [Google Scholar] [CrossRef]
- Ingham, E.; Fisher, J. Biological reactions to wear debris in total joint replacement, Proceedings of the Institution of Mechanical Engineers, Part H. J. Eng. Med. 2000, 214, 21–37. [Google Scholar] [CrossRef]
- Mani, G.; Feldman, M.; Patel, D.; Agrawal, C. Coronary stents: A materials perspective. Biomaterials 2007, 28, 1689–1710. [Google Scholar] [CrossRef]
- Wataha, J.C. Alloys for prosthodontic restorations. J. Prosthet. Dent. 2002, 87, 351–363. [Google Scholar] [CrossRef]
- Akova, T.; Ucar, Y.; Tukay, A.; Balkaya, M.C.; Brantley, W.A. Comparison of the bond strength of laser-sintered and cast base metal dental alloys to porcelain. Dent. Mater. 2008, 24, 1400–1404. [Google Scholar] [CrossRef]
- Jevremovic, D.; Puskar, T.; Kosec, B.; Vukelic, D.; Budak, I.; Aleksandrovic, S.; Egbeer, D.; Williams, R. The analysis of the mechanical properties of F75 Co–Cr alloy for use in selective laser melting (SLM) manufacturing of removable partial dentures (RPD). Metalurgija 2012, 51, 171–174. [Google Scholar]
- Takaichi, A.; Suyalatu; Nakamoto, T.; Joko, N.; Nomura, N.; Tsutsumi, Y.; Migita, S.; Doi, H.; Kurosu, S.; Chiba, A.; et al. Microstructures and mechanical properties of Co-29Cr-6Mo alloy fabricated by selective laser melting process for dental applications. J. Mech. Behav. Biomed. Mater. 2013, 21, 67–76. [Google Scholar] [CrossRef]
- Zhou, X.; Wang, D.; Liu, X.; Zhang, D.D.; Qu, S.; Ma, J.; London, G.; Shen, Z.; Liu, W. 3D-imaging of selective laser melting defects in a Co-Cr-Mo alloy by synchrotron radiation micro-CT. Acta Mater. 2015, 98, 1–16. [Google Scholar] [CrossRef]
- Sun, S.H.; Koizumi, Y.; Kurosu, S.; Li, Y.P.; Matsumoto, H.; Chiba, A. Build direction dependence of microstructure and high-temperature tensile property of Co-Cr-Mo alloy fabricated by electron beam melting. Acta Mater. 2014, 64, 154–168. [Google Scholar] [CrossRef]
- Sun, S.H.; Koizumi, Y.; Kurosu, S.; Li, Y.; Chiba, A. Phase and grain size inhomogeneity and their influences on creep behavior of Co–Cr–Mo alloy additive manufactured by electron beam melting. Acta Mater. 2015, 86, 305–318. [Google Scholar] [CrossRef]
- Yokota, Y.; Nihei, T.; Tanaka, K.; Sakairi, K.; Chani, V.; Ohashi, Y.; Kurosawa, S.; Kamada, K.; Yoshikawa, A. Fabrication of Metallic Fibers with High Melting Point and Poor Workability by Unidirectional Solidification. Adv. Eng. Mater. 2018, 20. [Google Scholar] [CrossRef]
- Yoshikawa, A.; Nikl, M.; Boulon, G.; Fukuda, T. Challenge and study for developing of novel single crystalline optical materials using micro-pulling-down method. Opt. Mater. 2007, 30, 6–10. [Google Scholar] [CrossRef]
- Yokota, Y.; Kurosawa, S.; Shoji, Y.; Ohashi, Y.; Kamada, K.; Yoshikawa, A. Development of novel growth methods for halide single crystals. Opt. Mater. 2017, 65, 46–51. [Google Scholar] [CrossRef]
- Yokota, Y.; Fujimoto, Y.; Yanagida, T.; Takahashi, H.; Yonetani, M.; Hayashi, K.; Park, I.; Kawaguchi, N.; Fukuda, K.; Yamaji, A.; et al. Crystal growth of Na-Co-doped Ce: LiCaAlF6 single crystals and their optical, scintillation, and physical properties. Cryst. Growth Des. 2011, 11, 4775–4779. [Google Scholar] [CrossRef]
- Yoshikawa, A.; Satonaga, T.; Kamada, K.; Sato, H.; Nikl, M.; Solovieva, N.; Fukuda, T. Crystal growth of Ce: PrF3 by micro-pulling-down method. J. Cryst. Growth 2004, 270, 427–432. [Google Scholar] [CrossRef]
- Murakami, R.; Kamada, K.; Shoji, Y.; Yokota, Y.; Yoshino, M.; Kurosawa, S.; Ohashi, Y.; Yamaji, A.; Yoshikawa, A. Fabrication of flexible Ir and Ir-Rh wires and application for thermocouple. J. Cryst. Growth 2018, 487, 72–77. [Google Scholar] [CrossRef]
- Nihei, T.; Yokota, Y.; Arakawa, M.; Ohashi, Y.; Kurosawa, S.; Kamada, K.; Chani, V.; Yoshikawa, A. Growth of platinum fibers using the micro-pulling-down method. J. Cryst. Growth 2017, 468, 403–406. [Google Scholar] [CrossRef]
- Yokota, Y.; Nihei, T.; Abe, S.; Yoshino, M.; Yamaji, A.; Toyoda, S.; Sato, H.; Ohashi, Y.; Kurosawa, S.; Kamada, K.; et al. Direct Fabrication of Co-Cr-Mo fiber from the Melt by Unidirectional Solidification. Sci. Tech. Adv. Mater. Submitted.
- Mani, A.; Salinas-Rodriguez; Lopez, H.F. Deformation induced FCC to HCP transformation in a Co-27Cr-5Mo-0.05C alloy. Mater. Sci. Eng. A 2011, 528, 3037–3043. [Google Scholar] [CrossRef]
- Immarigeon, J.P.; Rajan, K.; Wallace, W. Microstructural changes during isothermal foging of a Co-Cr-Mo alloy. Metall. Trans. A 1984, 15, 339–345. [Google Scholar] [CrossRef]
- Lippared, H.E.; Kennedy, R.L. Process Metallurgy of Wrought CoCrMo Alloy; ASTM: West Conshohocken, PA, USA, 1999; pp. 98–107. [Google Scholar]
- Lopez, H.F.; Saldvar-Garcia, A.J. Martensitic transformation in a cast Co-Cr-Mo-C alloy. Metall. Mater. Trans. A 2008, 39, 8–18. [Google Scholar] [CrossRef]
- Kurosu, S.; Matsumoto, H.; Chiba, A. Grain refinement of biomedical Co-27Cr-5Mo-0.16N alloy by reverse transformation. Mater. Lett. 2010, 64, 49–52. [Google Scholar] [CrossRef]
- Chiba, A.; Li, X.G.; Kim, M.S. High work-hardening rate and deformation twinning of Co-Ni-based superalloy at elevated temperatures. Philos. Mag. A 1999, 79, 1533–1554. [Google Scholar] [CrossRef]
- Yoda, K.; Suyalatu; Takaichi, A.; Nomura, N.; Tsutsumi, Y.; Doi, H.; Kurosu, S.; Chiba, A.; Igarashi, Y.; Hanawa, T. Effects of chromium and nitrogen content on the microstructures and mechanical properties of as-cast Co-Cr-Mo alloys for dental applications. Acta Biomater. 2012, 8, 2856–2862. [Google Scholar] [CrossRef]
- Yamanaka, K.; Mori, M.; Chiba, A. Nanoarchitectured Co-Cr-Mo orthopedic implant alloys: Nitrogen-enhanced nanostructural evolution and its effect on phase stability. Acta Biomater. 2013, 9, 6259–6267. [Google Scholar] [CrossRef]
- Kurosu, S.; Matsumoto, H.; Chiba, A. Isothermal Phase Transformation in Biomedical Co-29Cr-6Mo Alloy without Addition of Carbon or Nitrogen. Metall. Mater. Trans. A 2010, 41, 2613–2625. [Google Scholar] [CrossRef]
- Dobbs, H.S.; Robertson, J.L.M. Heat treatment of cast Co−Cr−Mo for orthopaedic implant use. J. Mater. Sci. 1983, 18, 391–401. [Google Scholar] [CrossRef]
- Liao, Y.; Pourzal, R.; Stemmer, P.; Wimmer, M.A.; Jacobs, J.J.; Fischer, A.; Marks, L.D. New insights into hard phases of CoCrMo metal-on-metal hip replacements. J. Mech. Behav. Biomed. Mater. 2012, 12, 39–49. [Google Scholar] [CrossRef] [PubMed]
- Okazaki, Y. Effects of heat treatment and hot forging on microstructure and mechanical properties of Co–Cr–Mo alloy for surgical implants. Mater. Trans. 2008, 49, 817–823. [Google Scholar] [CrossRef]
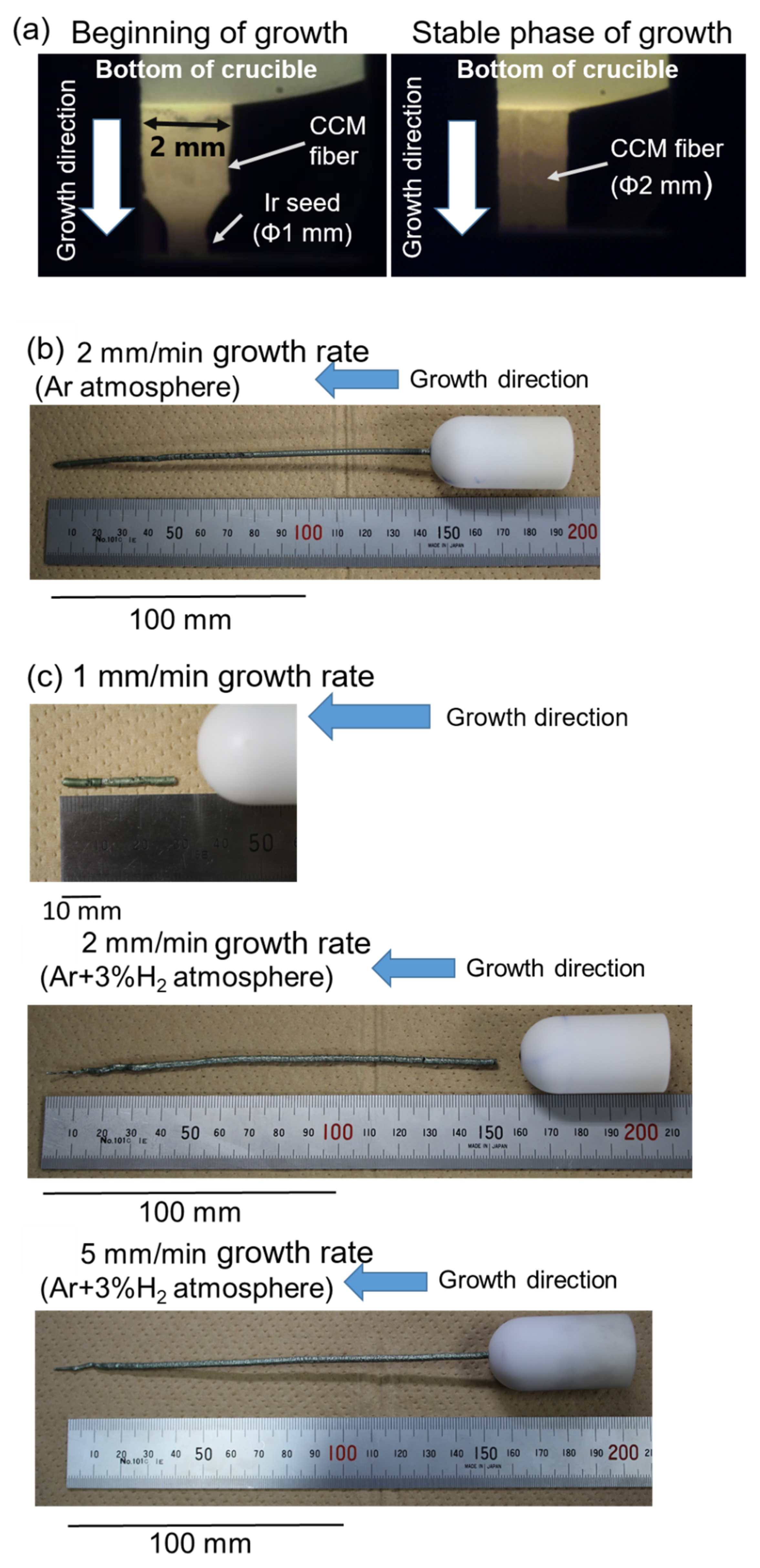

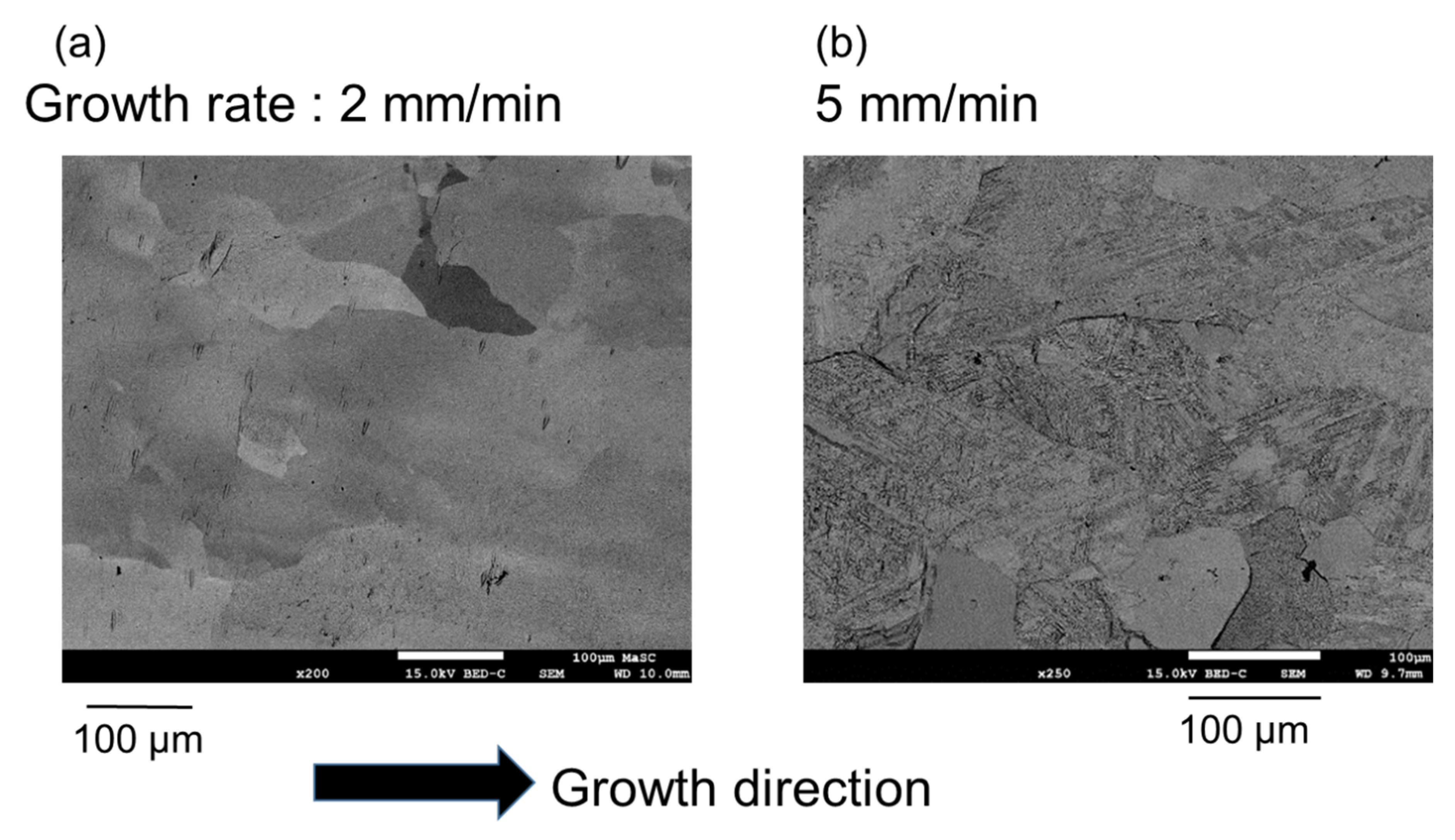
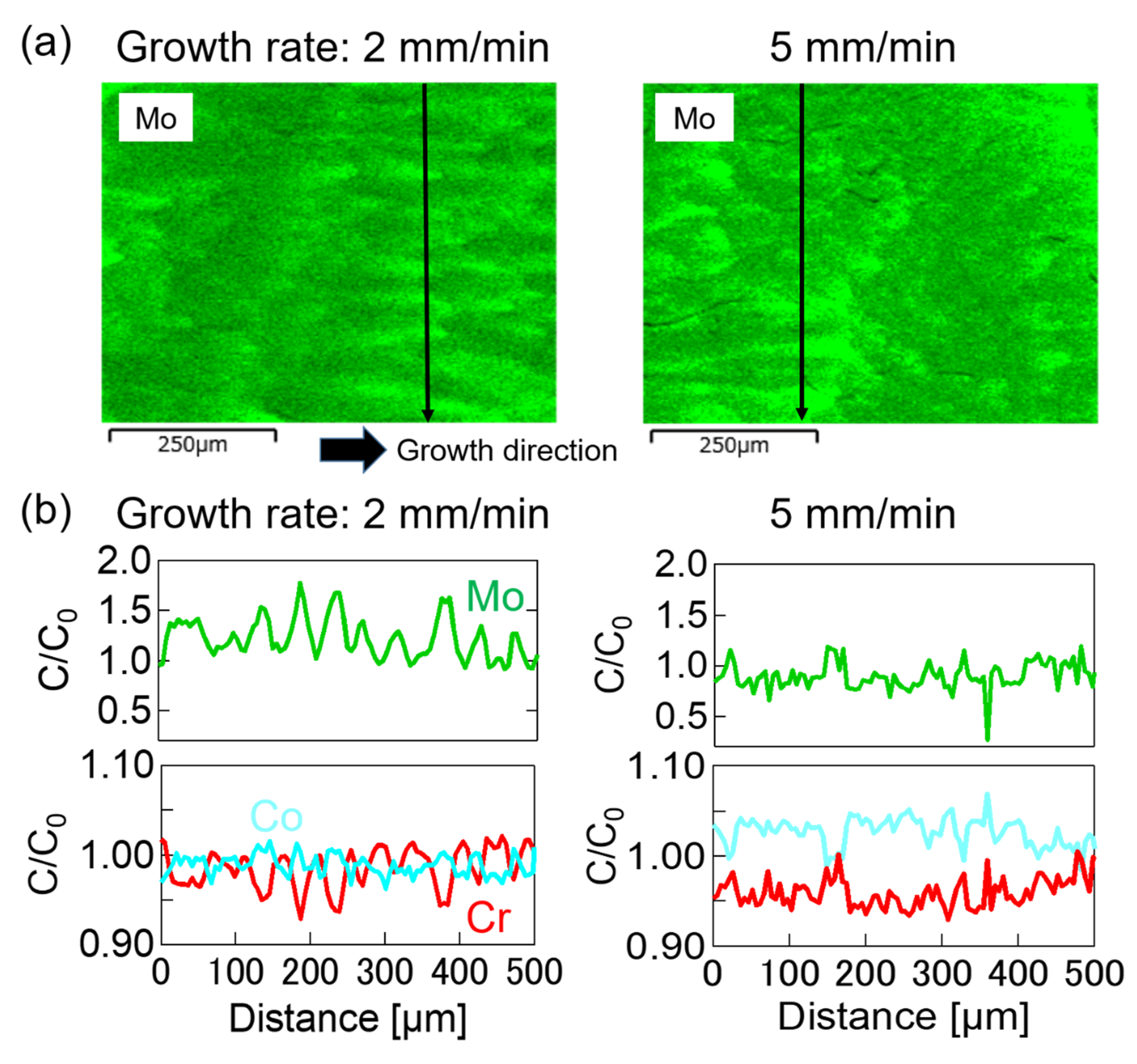
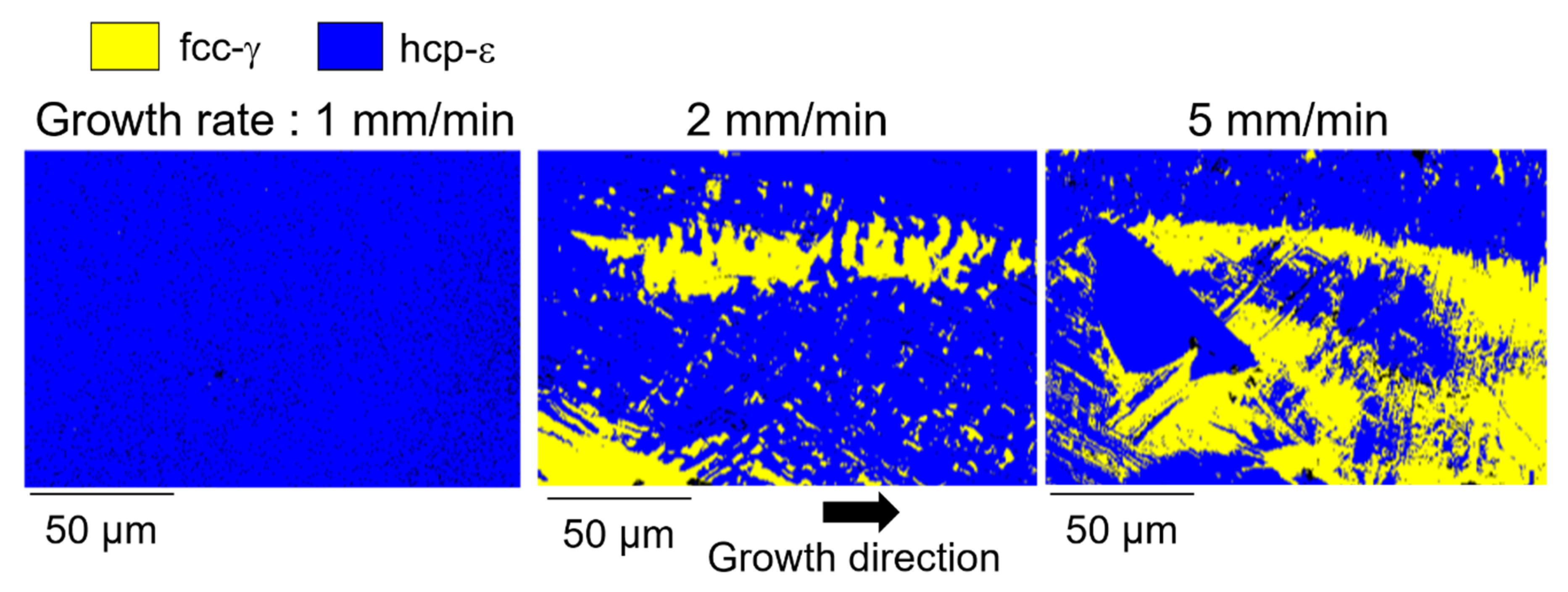
| Phase | 1 mm/min | 2 mm/min | 5 mm/min |
|---|---|---|---|
| fcc-γ | 0.01% | 23.49% | 36.51% |
| hcp-ε | 96.01% | 75.42% | 61.76% |
| Zero solution | 3.98% | 1.09% | 1.73% |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abe, S.; Yokota, Y.; Nihei, T.; Yoshino, M.; Yamaji, A.; Toyoda, S.; Sato, H.; Ohashi, Y.; Kurosawa, S.; Kamada, K.; et al. Control of Microstructure for Co-Cr-Mo Fibers Fabricated by Unidirectional Solidification. Crystals 2020, 10, 11. https://doi.org/10.3390/cryst10010011
Abe S, Yokota Y, Nihei T, Yoshino M, Yamaji A, Toyoda S, Sato H, Ohashi Y, Kurosawa S, Kamada K, et al. Control of Microstructure for Co-Cr-Mo Fibers Fabricated by Unidirectional Solidification. Crystals. 2020; 10(1):11. https://doi.org/10.3390/cryst10010011
Chicago/Turabian StyleAbe, Shoki, Yuui Yokota, Takayuki Nihei, Masao Yoshino, Akihiro Yamaji, Satoshi Toyoda, Hiroki Sato, Yuji Ohashi, Shunsuke Kurosawa, Kei Kamada, and et al. 2020. "Control of Microstructure for Co-Cr-Mo Fibers Fabricated by Unidirectional Solidification" Crystals 10, no. 1: 11. https://doi.org/10.3390/cryst10010011
APA StyleAbe, S., Yokota, Y., Nihei, T., Yoshino, M., Yamaji, A., Toyoda, S., Sato, H., Ohashi, Y., Kurosawa, S., Kamada, K., & Yoshikawa, A. (2020). Control of Microstructure for Co-Cr-Mo Fibers Fabricated by Unidirectional Solidification. Crystals, 10(1), 11. https://doi.org/10.3390/cryst10010011






