Decomposition of Al2O3-Supported PdSO4 and Al2(SO4)3 in the Regeneration of Methane Combustion Catalyst: A Model Catalyst Study
Abstract
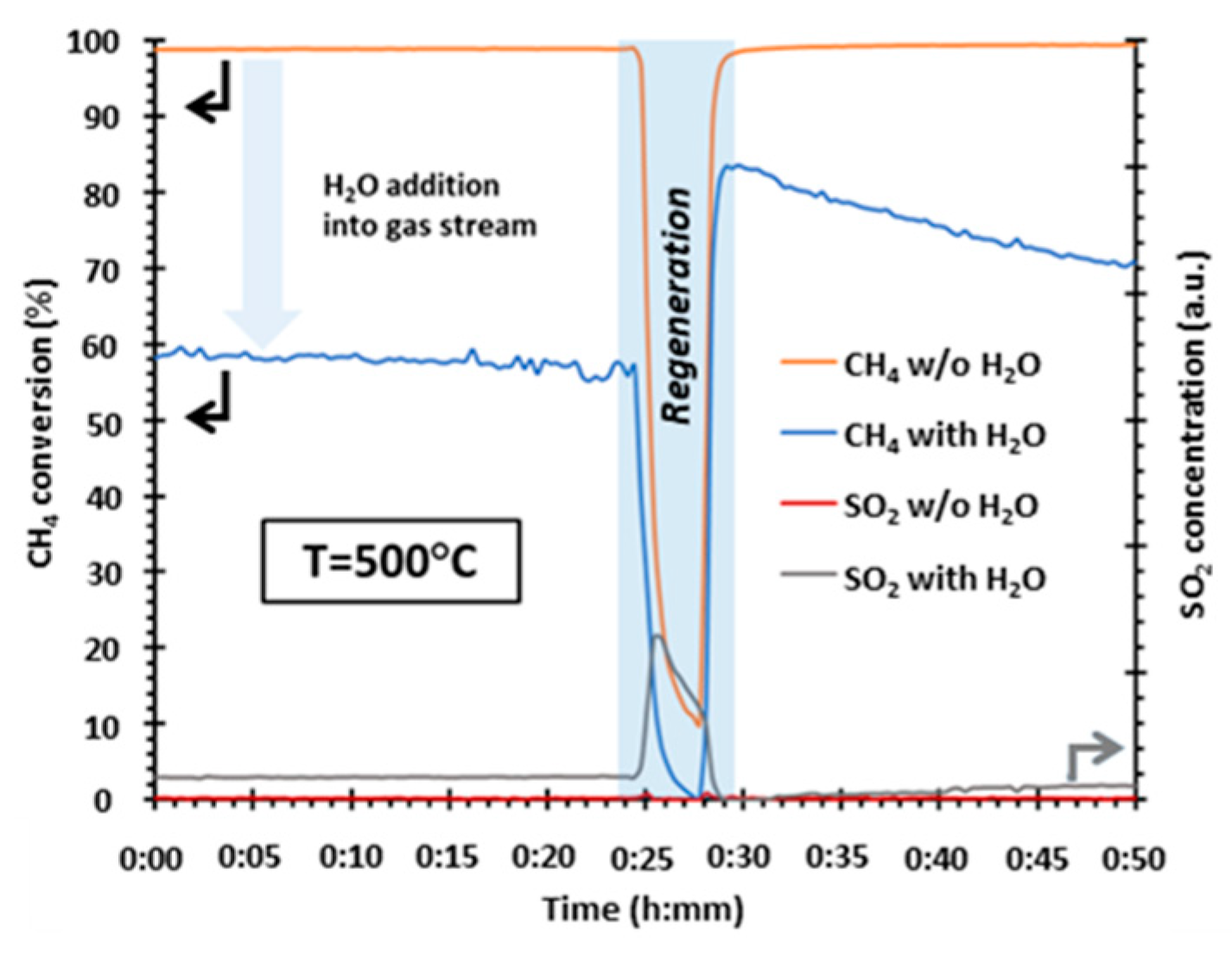
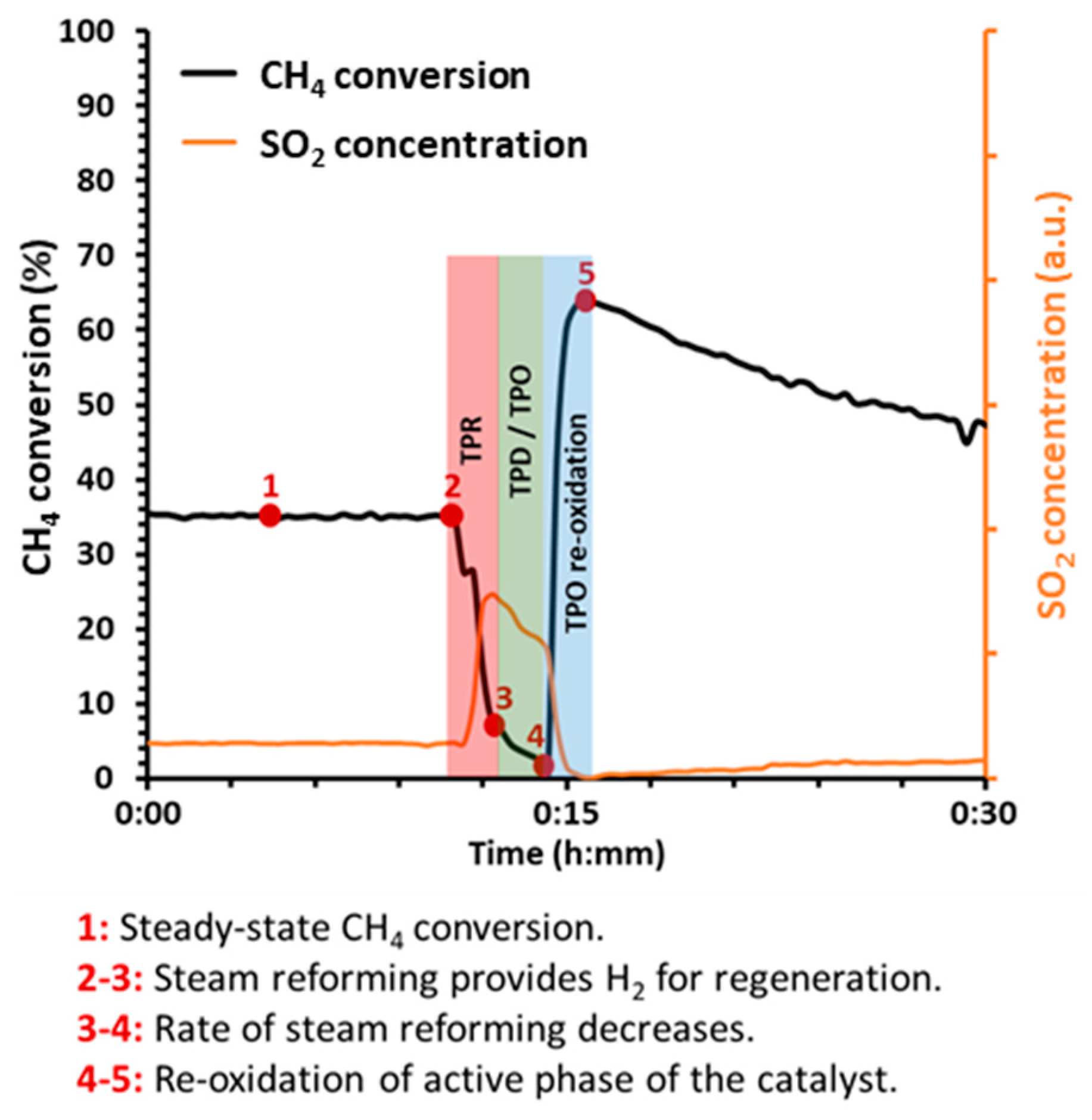
:1. Introduction
2. Results and Discussion
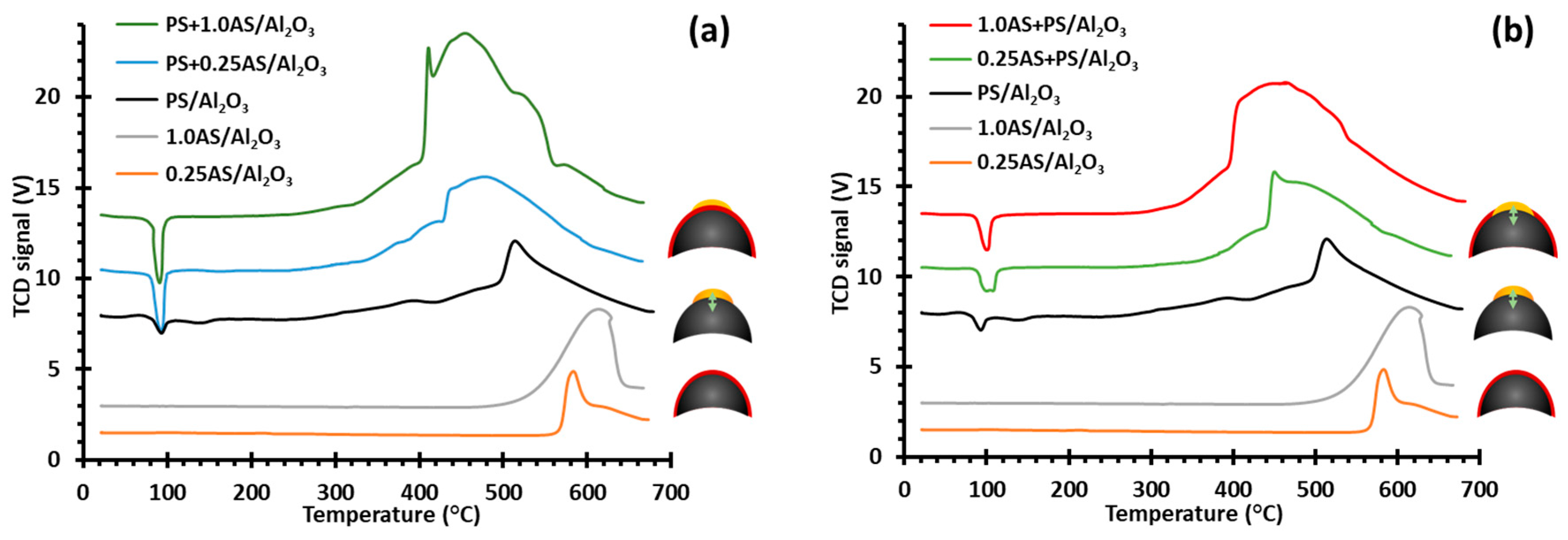
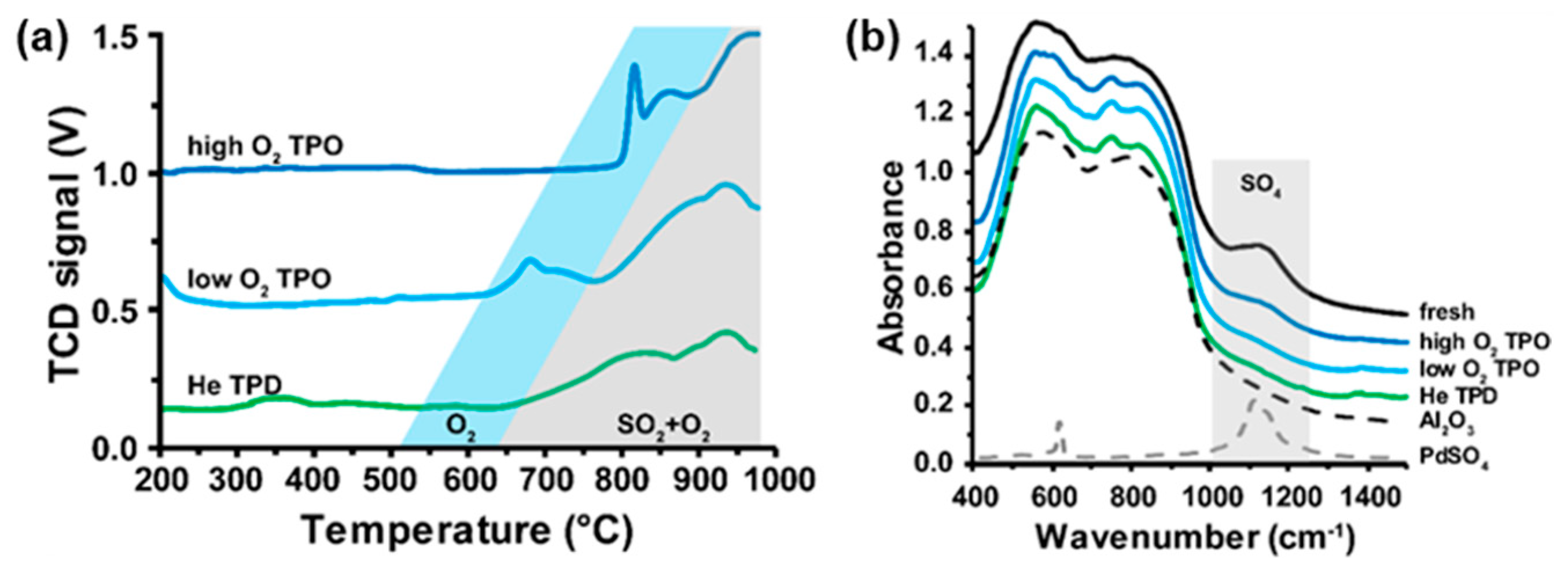
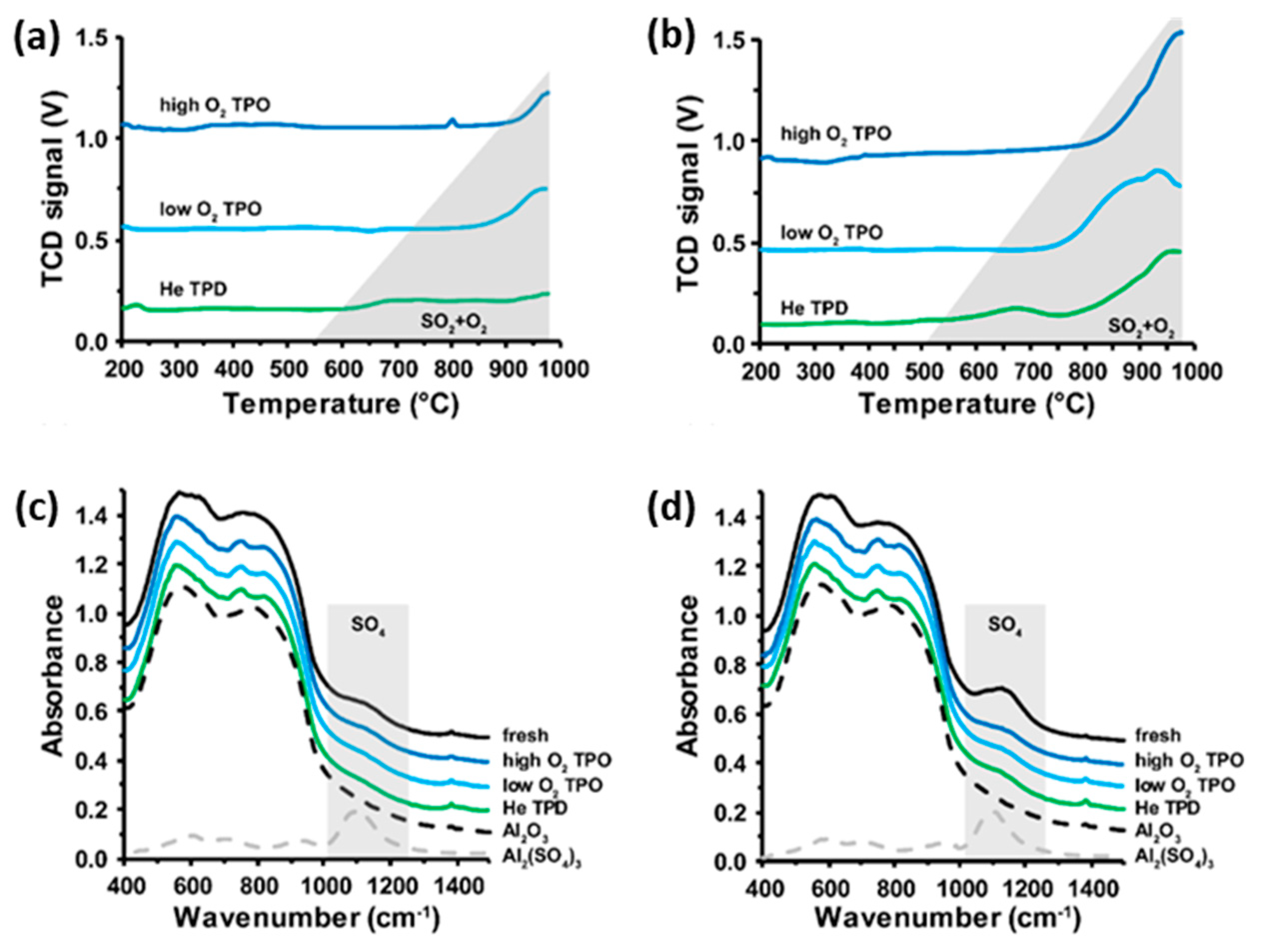
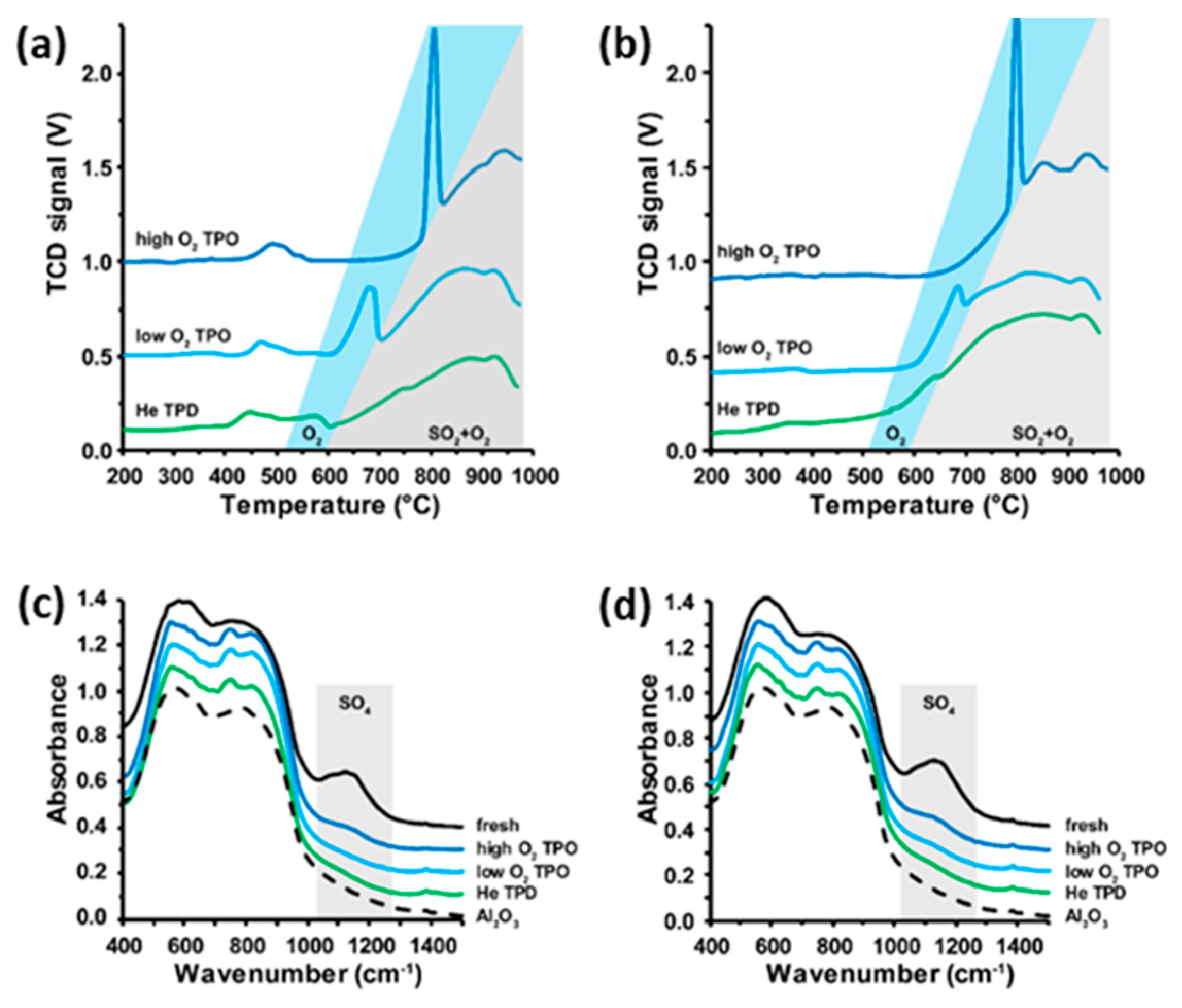
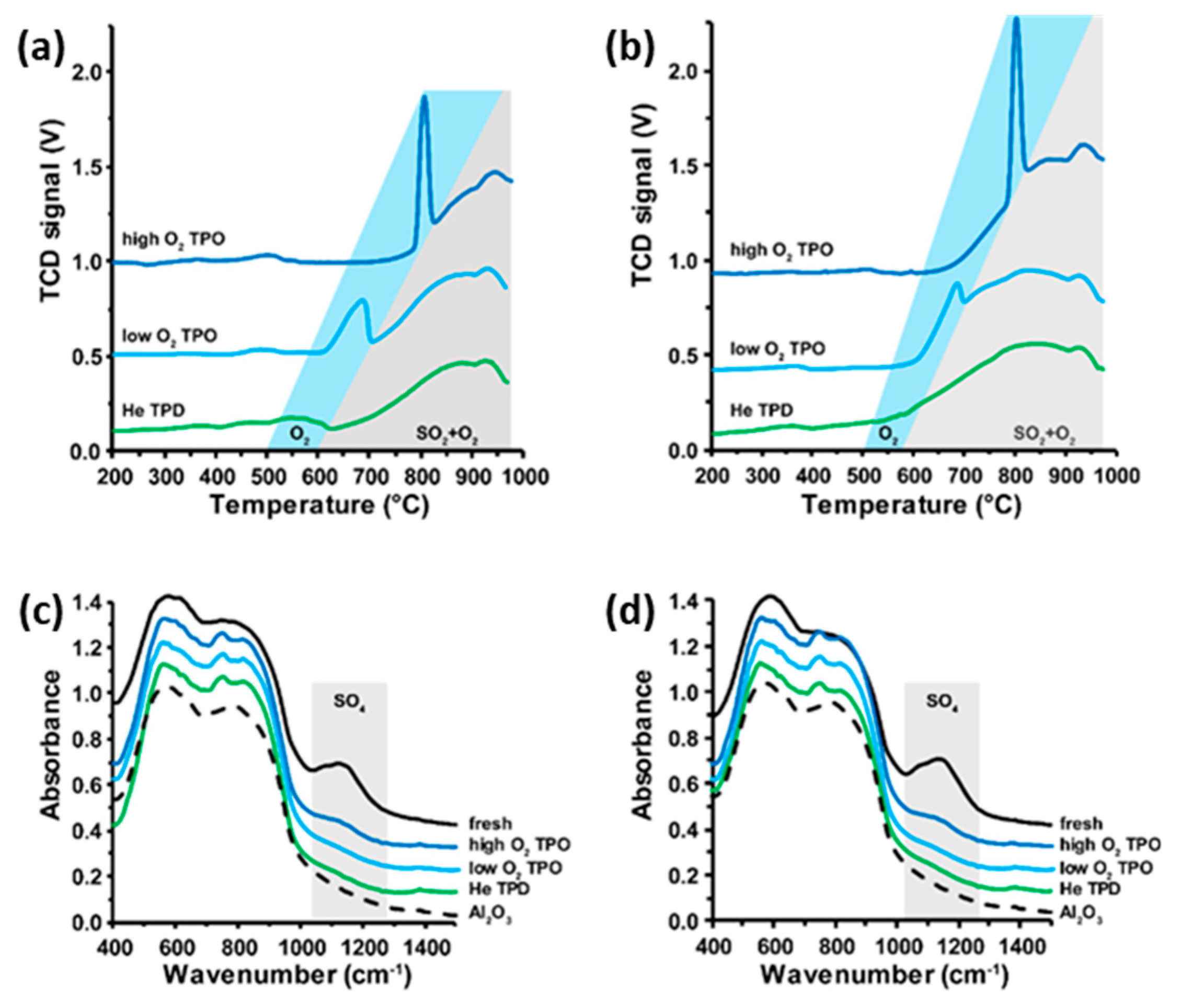
2.1. Thermal Decomposition of Al2O3-Supported PdSO4 and Al2(SO4)3 under Hydrogen Gas
2.2. Thermal Decomposition of Al2O3-Supported PdSO4 and Al2(SO4)3
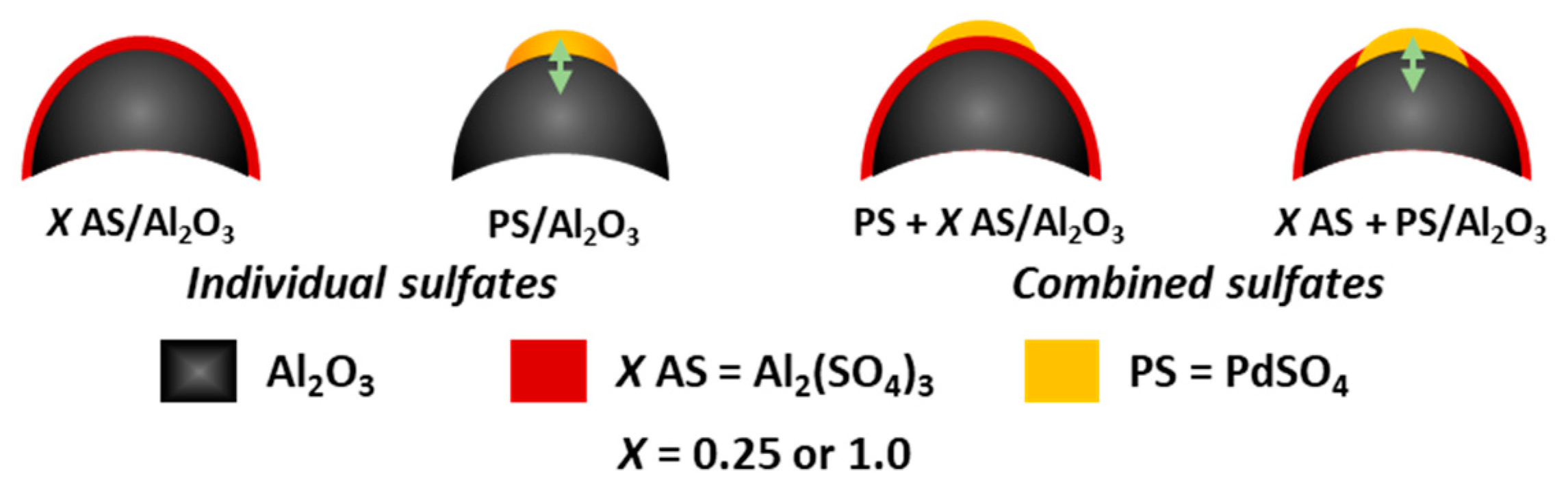
2.3. Thermal Decomposition of Al2O3-Supported PdSO4 and Al2(SO4)3 Combinations
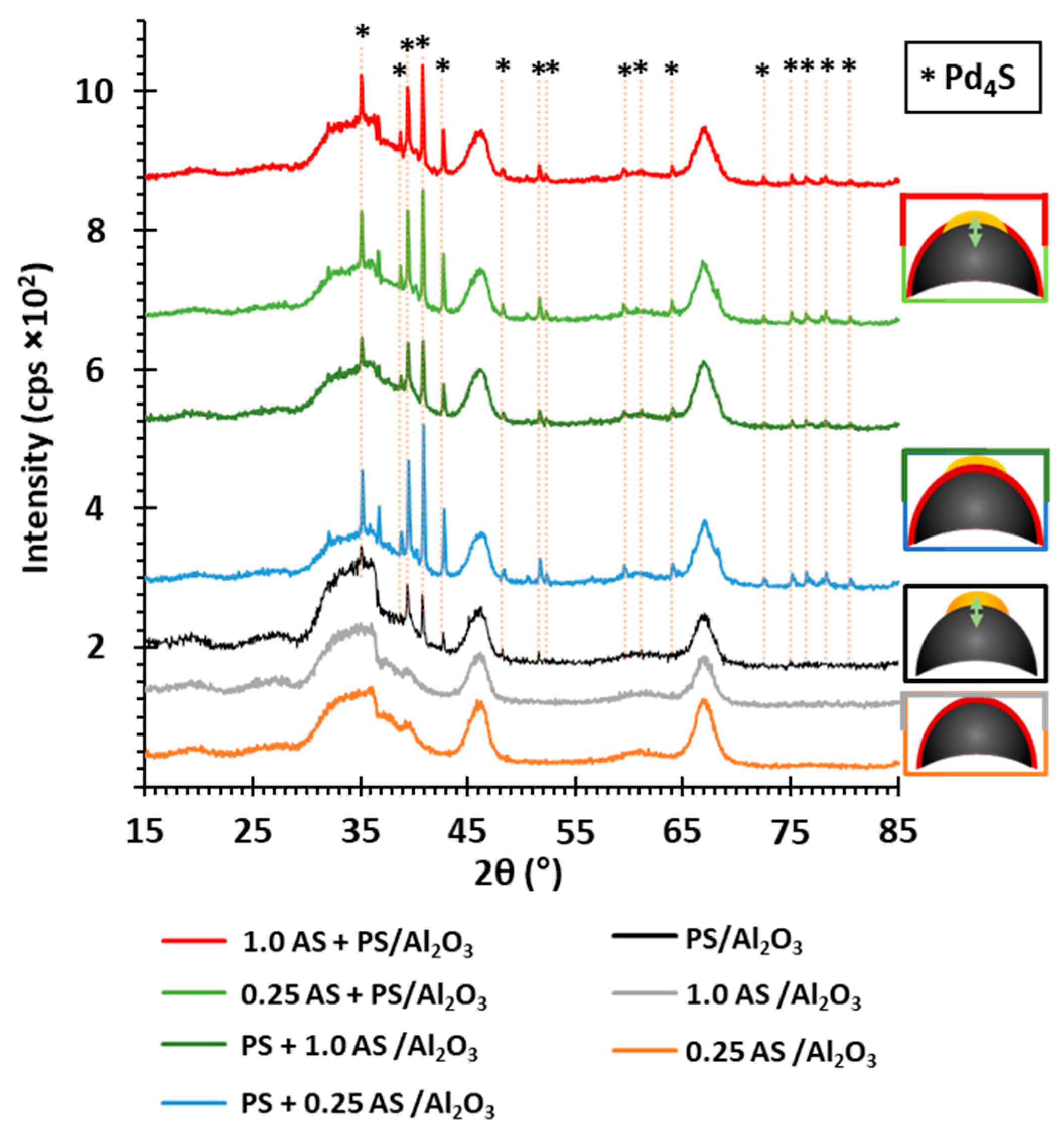
2.4. State of Palladium after Thermal Decomposition of Al2O3-Supported PdSO4 and Al2(SO4)3
3. Materials and Methods
3.1. Catalysts
3.2. Characterization Techniques
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Keenan, M. Exhaust Emissions Control: 60 Years of Innovation and Development. SAE Tech. Pap. 2017. [Google Scholar] [CrossRef]
- Gélin, P.; Primet, M. Complete oxidation of methane at low temperature over noble metal based catalysts: A review. Appl. Catal. B Environ. 2002, 39, 1–37. [Google Scholar] [CrossRef]
- Gélin, P.; Urfels, L.; Primet, M.; Tena, E. Complete oxidation of methane at low temperature over Pt and Pd catalysts for the abatement of lean-burn natural gas fuelled vehicles emissions: Influence of water and sulphur containing compounds. Catal. Today 2003, 83, 45–57. [Google Scholar] [CrossRef]
- Villamaina, R.; Nova, I.; Tronconi, E.; Maunula, T.; Keenan, M. The Effect of CH4 on NH3-SCR Over Metal-Promoted Zeolite Catalysts for Lean-Burn Natural Gas Vehicles. Top. Catal. 2018, 61, 1974–1982. [Google Scholar] [CrossRef]
- Maunula, T.; Kallinen, K.; Savimäki, A.; Wolff, T. Durability Evaluations and Rapid Ageing Methods in Commercial Emission Catalyst Development for Diesel, Natural Gas and Gasoline Applications. Top. Catal. 2016, 59, 1049–1053. [Google Scholar] [CrossRef]
- Kinnunen, N.M.; Hirvi, J.T.; Suvanto, M.; Pakkanen, T.A. Methane combustion activity of Pd–PdOx–Pt/Al2O3 catalyst: The role of platinum promoter. J. Mol. Catal. A Chem. 2012, 356, 20–28. [Google Scholar] [CrossRef]
- Roth, D.; Gélin, P.; Primet, M.; Tena, E. Catalytic behaviour of Cl-free and Cl-containing Pd/Al2O3 catalysts in the total oxidation of methane at low temperature. Appl. Catal. A Gen. 2000, 203, 37–45. [Google Scholar] [CrossRef]
- European standard for diesel fuel, EN 590:2009; European Commission Standard; 2009. Available online: https://ec.europa.eu/growth/single-market/european-standards_en (accessed on 7 May 2019).
- Jones, J.M.; Dupont, V.A.; Brydson, R.; Fullerton, D.J.; Nasri, N.S.; Ross, A.B.; Westwood, A.V.K. Sulphur poisoning and regeneration of precious metal catalysed methane combustion. Catal. Today 2003, 81, 589–601. [Google Scholar] [CrossRef]
- Honkanen, M.; Kärkkäinen, M.; Kolli, T.; Heikkinen, O.; Viitanen, V.; Zeng, L.; Jiang, H.; Kallinen, K.; Huuhtanen, M.; Keiski, R.L.; et al. Accelerated deactivation studies of the natural-gas oxidation catalyst—Verifying the role of sulfur and elevated temperature in catalyst aging. Appl. Catal. B Environ. 2016, 182, 439–448. [Google Scholar] [CrossRef]
- Honkanen, M.; Wang, J.; Kärkkäinen, M.; Huuhtanen, M.; Jiang, H.; Kallinen, K.; Keiski, R.L.; Akola, J.; Vippola, M. Regeneration of sulfur-poisoned Pd-based catalyst for natural gas oxidation. J. Catal. 2018, 358, 253–265. [Google Scholar] [CrossRef]
- Mowery, D.L.; McCormick, R.L. Deactivation of alumina supported and unsupported PdO methane oxidation catalyst: The effect of water on sulfate poisoning. Appl. Catal. B Environ. 2001, 34, 287–297. [Google Scholar] [CrossRef]
- Hoyos, L.J.; Praliaud, H.; Primet, M. Catalytic combustion of methane over palladium supported on alumina and silica in presence of hydrogen sulfide. Appl. Catal. A Gen. 1993, 98, 125–138. [Google Scholar] [CrossRef]
- Venezia, A.M.; Di Carlo, G.; Pantaleo, G.; Liotta, L.F.; Melaet, G.; Kruse, N. Oxidation of CH4 over Pd supported on TiO2-doped SiO2: Effect of Ti(IV) loading and influence of SO2. Appl. Catal. B Environ. 2009, 88, 430–437. [Google Scholar] [CrossRef]
- Laperdrix, E.; Justin, I.; Costentin, G.; Saur, O.; Lavalley, J.; Aboulayt, A.; Ray, J.; Nédez, C. Comparative study of CS2 hydrolysis catalyzed by alumina and titania. Appl. Catal. B Environ. 1998, 17, 167–173. [Google Scholar] [CrossRef]
- Laperdrix, E.; Sahibed-dine, A.; Costentin, G.; Saur, O.; Bensitel, M.; Nédez, C.; Mohamed Saad, A.B.; Lavalley, J.C. Reduction of sulfate species by H2S on different metal oxides and promoted aluminas. Appl. Catal. B Environ. 2000, 26, 71–80. [Google Scholar] [CrossRef]
- Arosio, F.; Colussi, S.; Groppi, G.; Trovarelli, A. Regeneration of S-poisoned Pd/Al2O3 catalysts for the combustion of methane. Catal. Today 2006, 117, 569–576. [Google Scholar] [CrossRef]
- Castellazzi, P.; Groppi, G.; Forzatti, P.; Finocchio, E.; Busca, G. Activation process of Pd/Al2O3 catalysts for CH4 combustion by reduction/oxidation cycles in CH4-containing atmosphere. J. Catal. 2010, 275, 218–227. [Google Scholar] [CrossRef]
- Ordóñez, S.; Hurtado, P.; Diez, F.V. Methane catalytic combustion over Pd/Al2O3 in presence of sulphur dioxide: Development of a regeneration procedure. Catal. Lett. 2005, 100, 27–34. [Google Scholar] [CrossRef]
- Yu, T.-C.; Shaw, H. The effect of sulfur poisoning on methane oxidation over palladium supported on γ-alumina catalysts. Appl. Catal. B Environ. 1998, 18, 105–114. [Google Scholar] [CrossRef]
- Bounechada, D.; Groppi, G.; Forzatti, P.; Kallinen, K.; Kinnunen, T. Effect of periodic lean/rich switch on methane conversion over a Ce–Zr promoted Pd-Rh/Al2O3 catalyst in the exhausts of natural gas vehicles. Appl. Catal. B Environ. 2012, 119, 91–99. [Google Scholar] [CrossRef]
- Salaün, M.; Kouakou, A.; Da Costa, S.; Da Costa, P. Synthetic gas bench study of a natural gas vehicle commercial catalyst in monolithic form: On the effect of gas composition. Appl. Catal. B Environ. 2009, 88, 386–397. [Google Scholar] [CrossRef]
- Gremminger, A.T.; de Carvalho, H.W.P.; Popescu, R.; Grunwaldt, J.-D.; Deutschmann, O. Influence of gas composition on activity and durability of bimetallic Pd-Pt/Al2O3 catalysts for total oxidation of methane. Catal. Today 2015, 258, 470–480. [Google Scholar] [CrossRef]
- Arosio, F.; Colussi, S.; Trovarelli, A.; Groppi, G. Effect of alternate CH4-reducing/lean combustion treatments on the reactivity of fresh and S-poisoned Pd/CeO2/Al2O3 catalysts. Appl. Catal. B Environ. 2008, 80, 335–342. [Google Scholar] [CrossRef]
- Kinnunen, N.M.; Kallinen, K.; Maunula, T.; Keenan, M.; Suvanto, M. Fundamentals of sulfate species in methane combustion catalyst operation and regeneration—A simulated exhaust gas study. Catalysts 2019, 9, 417. [Google Scholar] [CrossRef]
- Nissinen, V.H.; Kinnunen, N.M.; Suvanto, M. Regeneration of a sulfur-poisoned methane combustion catalyst: Structural evidence of Pd4S formation. Appl. Catal. B Environ. 2018, 237, 110–115. [Google Scholar] [CrossRef]
- Kinnunen, N.M.; Hirvi, J.T.; Kallinen, K.; Maunula, T.; Keenan, M.; Suvanto, M. Case study of a modern lean-burn methane combustion catalyst for automotive applications: What are the deactivation and regeneration mechanisms? Appl. Catal. B Environ. 2017, 207, 114–119. [Google Scholar] [CrossRef]
- Neyestanaki, A.K.; Klingstedt, F.; Salmi, T.; Murzin, D.Y. Deactivation of postcombustion catalysts, a review. Fuel 2004, 83, 395–408. [Google Scholar] [CrossRef]
- Ciuparu, D.; Lyubovsky, M.R.; Altman, E.; Pfefferle, L.D.; Datye, A. Catalytic Combustion of Methane over Palladium-Based Catalysts. Catal. Rev. 2002, 44, 593–649. [Google Scholar] [CrossRef]
- Euzen, P.; Le Gal, J.H.; Rebours, B.; Martin, G. Deactivation of palladium catalyst in catalytic combustion of methane. Catal. Today 1999, 47, 19–27. [Google Scholar] [CrossRef]
- Grunwaldt, J.D.; van Vegten, N.; Baiker, A. Insight into the structure of supported palladium catalysts during the total oxidation of methane. Chem. Commun. 2007, 4635–4637. [Google Scholar] [CrossRef]
- Mighell, A.D. TOPAS V2.0: General Profile Analysis Software for Powder Diffraction Data; Bruker AXS: Karlsruhe, Germany, 2000. [Google Scholar]
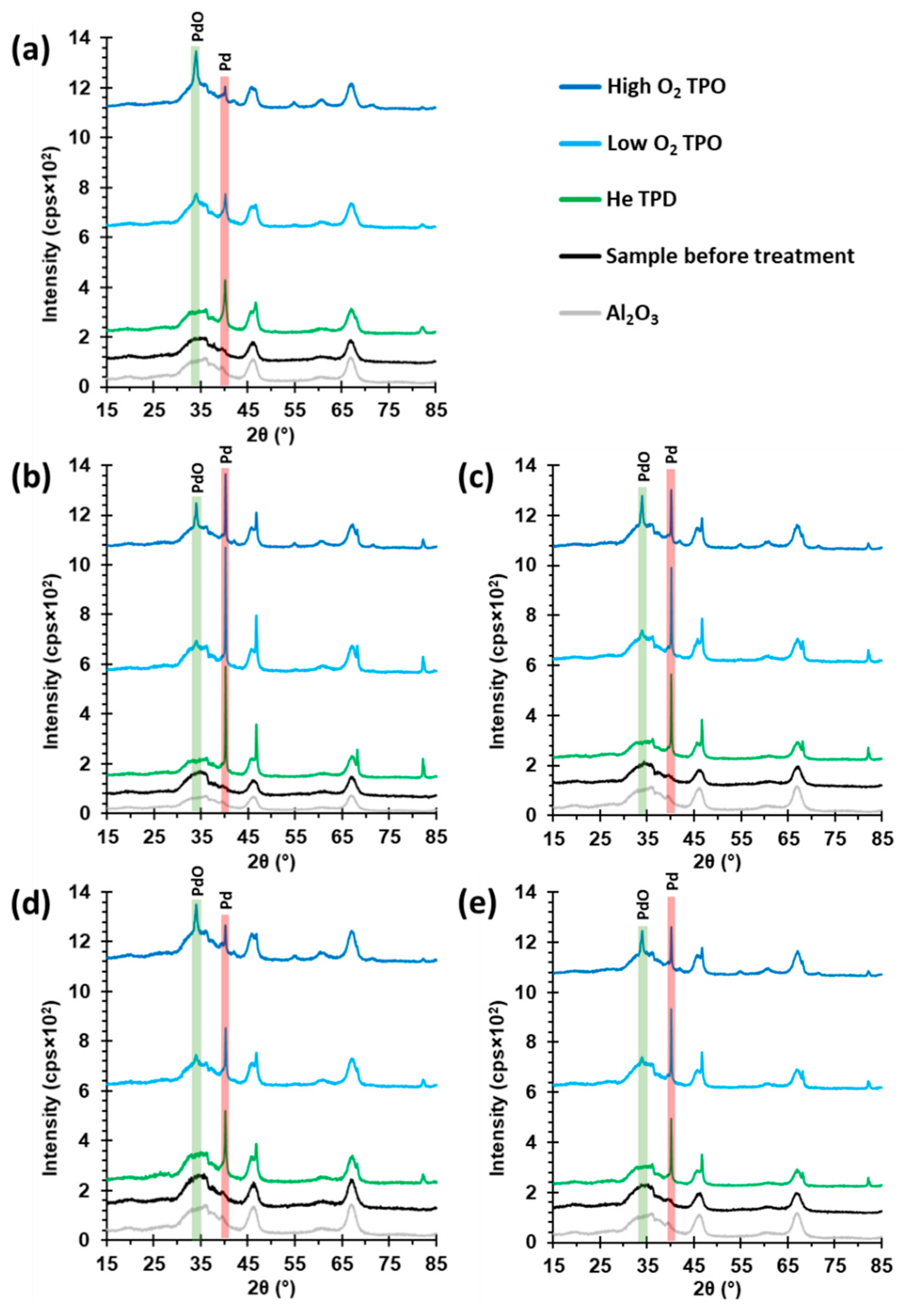
| Catalyst | Treatment | Sulfur Content (wt.-%) 1 | Relative Sulfur Content (%) 2 | PdO Peak Area | PdO Crystallite Size (nm) | Pd Peak Area | Pd Crystallite Size (nm) |
|---|---|---|---|---|---|---|---|
| Prepared | 0.88 | 100 | - | - | - | - | |
| 10% O2/He | 0.41 | 47 | 148.1 | 12.1 | 71.5 | 12.3 | |
| PS/Al2O3 3 | 0.1% O2/He | 0.11 | 13 | 121.3 | 5.7 | 118.8 | 14.4 |
| He TPD | 0.06 | 7 | - | - | 143.0 | 20.7 | |
| TPR | 0.30 | 34 | - | - | - | - | |
| Prepared | 0.29 | 100 | - | - | - | - | |
| 10% O2/He | 0.23 | 79 | - | - | - | - | |
| 0.25 AS/Al2O3 4 | 0.1% O2/He | 0.11 | 38 | - | - | - | - |
| He TPD | 0.08 | 28 | - | - | - | - | |
| TPR | 0.05 | 16 | - | - | - | - | |
| Prepared | 0.97 | 100 | - | - | - | - | |
| 10% O2/He | 0.48 | 49 | - | - | - | - | |
| 1.0 AS/Al2O3 4 | 0.1% O2/He | 0.30 | 31 | - | - | - | - |
| He TPD | 0.23 | 24 | - | - | - | - | |
| TPR | 0.07 | 7 | - | - | - | - | |
| Prepared | 1.39 | 100 | - | - | - | - | |
| 10% O2/He | 0.24 | 17 | 40.7 | 34.8 | 75.2 | 96.0 | |
| PS + 0.25 AS/Al2O3 3,4 | 0.1% O2/He | 0.05 | 4 | 18.3 | 20.1 | 114.7 | 110.4 |
| He TPD | 0.03 | 2 | - | - | 121.6 | 122.8 | |
| TPR | 0.30 | 22 | - | - | - | - | |
| Prepared | 2.10 | 100 | - | - | - | - | |
| 10% O2/He | 0.35 | 17 | 56.7 | 20.5 | 25.8 | 74.7 | |
| PS + 1.0 AS/Al2O3 3,4 | 0.1% O2/He | 0.09 | 4 | 24.5 | 17.7 | 64.7 | 66.5 |
| He TPD | 0.07 | 3 | - | - | 63.2 | 48.0 | |
| TPR | 0.30 | 14 | - | - | - | - | |
| Prepared | 1.03 | 100 | - | - | - | - | |
| 10% O2/He | 0.30 | 29 | 45.1 | 33.1 | 56.1 | 94.0 | |
| 0.25 AS + PS/Al2O3 3,4 | 0.1% O2/He | 0.10 | 10 | 16.0 | 23.4 | 85.6 | 101.2 |
| He TPD | 0.07 | 6 | - | - | 90.9 | 111.5 | |
| TPR | 0.30 | 29 | - | - | - | - | |
| Prepared | 1.71 | 100 | - | - | - | - | |
| 10% O2/He | 0.40 | 23 | 53.2 | 23.0 | 49.5 | 73.5 | |
| 1.0 AS + PS/Al2O3 3,4 | 0.1% O2/He | 0.12 | 7 | 7.7 | 38.8 | 71.5 | 85.7 |
| He TPD | 0.07 | 4 | - | - | 67.6 | 109.8 | |
| TPR | 0.30 | 18 | - | - | - | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kinnunen, N.M.; Nissinen, V.H.; Hirvi, J.T.; Kallinen, K.; Maunula, T.; Keenan, M.; Suvanto, M. Decomposition of Al2O3-Supported PdSO4 and Al2(SO4)3 in the Regeneration of Methane Combustion Catalyst: A Model Catalyst Study. Catalysts 2019, 9, 427. https://doi.org/10.3390/catal9050427
Kinnunen NM, Nissinen VH, Hirvi JT, Kallinen K, Maunula T, Keenan M, Suvanto M. Decomposition of Al2O3-Supported PdSO4 and Al2(SO4)3 in the Regeneration of Methane Combustion Catalyst: A Model Catalyst Study. Catalysts. 2019; 9(5):427. https://doi.org/10.3390/catal9050427
Chicago/Turabian StyleKinnunen, Niko M., Ville H. Nissinen, Janne T. Hirvi, Kauko Kallinen, Teuvo Maunula, Matthew Keenan, and Mika Suvanto. 2019. "Decomposition of Al2O3-Supported PdSO4 and Al2(SO4)3 in the Regeneration of Methane Combustion Catalyst: A Model Catalyst Study" Catalysts 9, no. 5: 427. https://doi.org/10.3390/catal9050427
APA StyleKinnunen, N. M., Nissinen, V. H., Hirvi, J. T., Kallinen, K., Maunula, T., Keenan, M., & Suvanto, M. (2019). Decomposition of Al2O3-Supported PdSO4 and Al2(SO4)3 in the Regeneration of Methane Combustion Catalyst: A Model Catalyst Study. Catalysts, 9(5), 427. https://doi.org/10.3390/catal9050427





