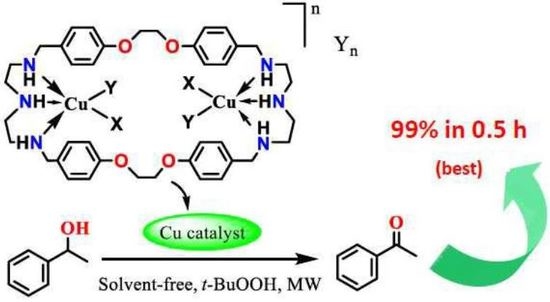
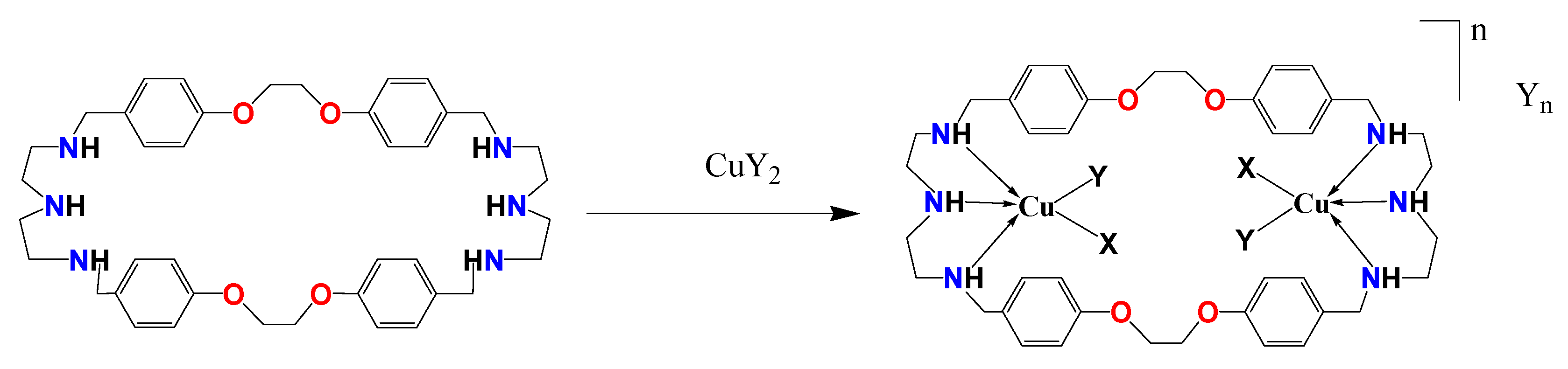
Synthesis and Structure of Copper Complexes of a N6O4 Macrocyclic Ligand and Catalytic Application in Alcohol Oxidation
Abstract
1. Introduction
2. Results and Discussion
2.1. X-Ray Diffraction Analysis
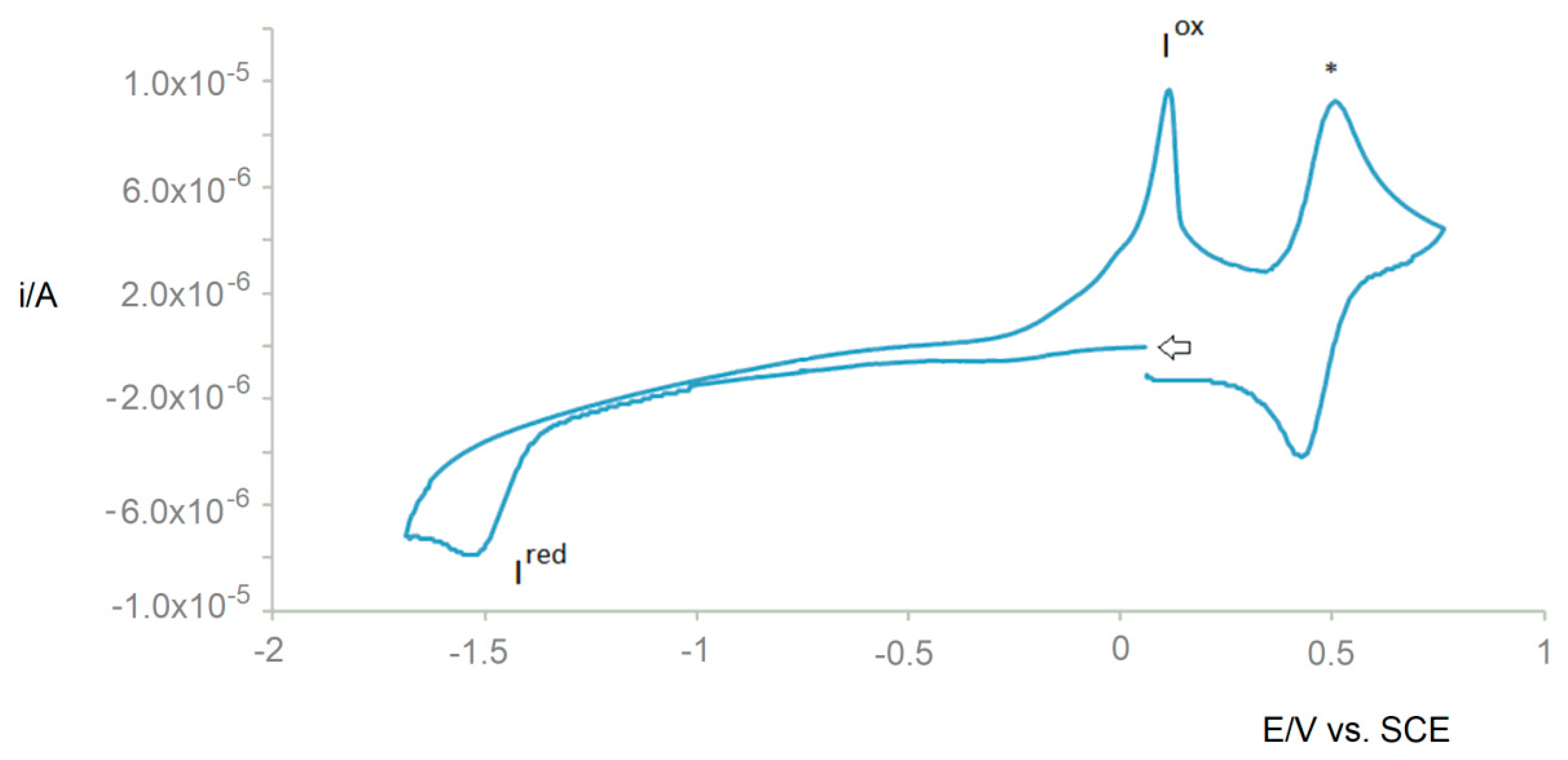
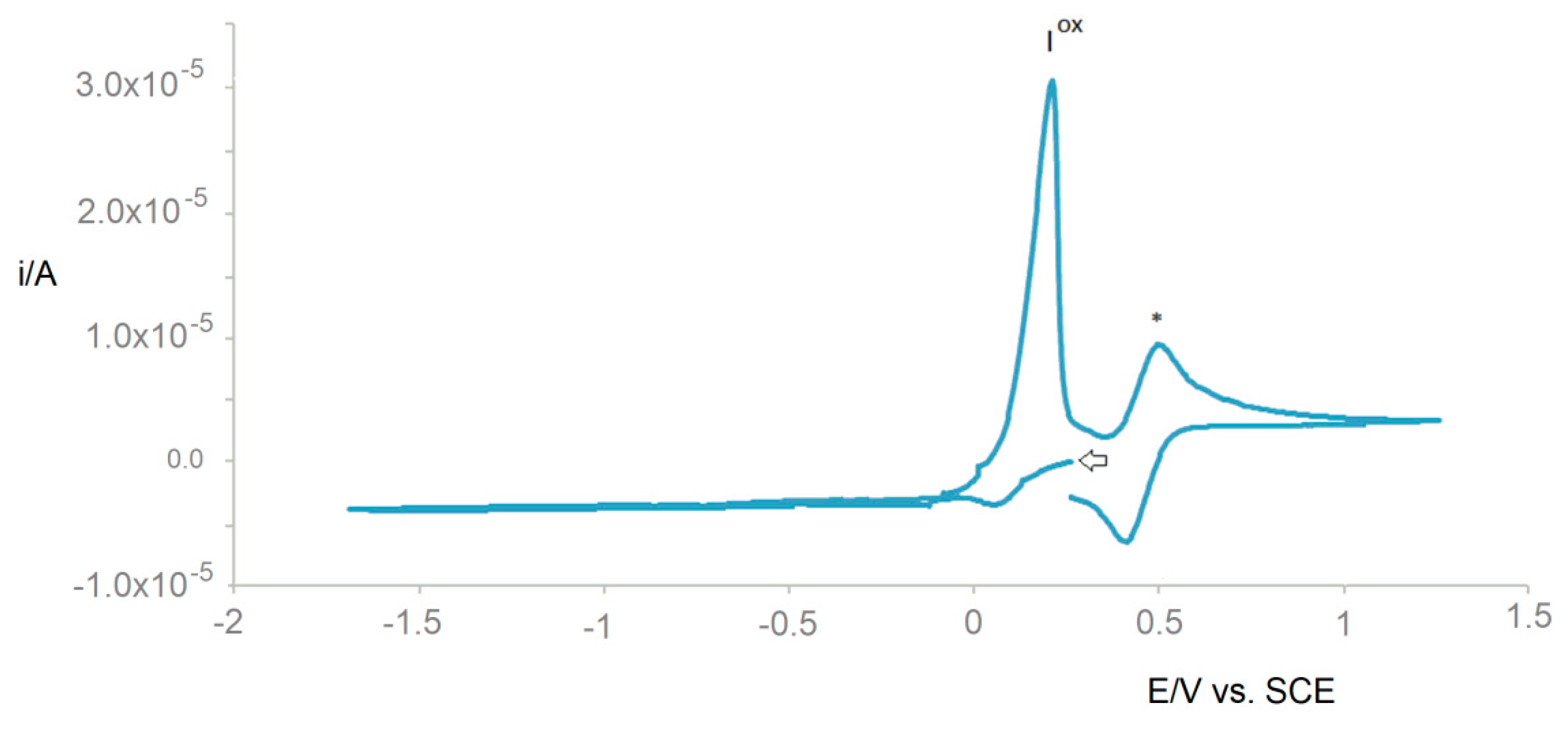
2.2. Electrochemical Behavior
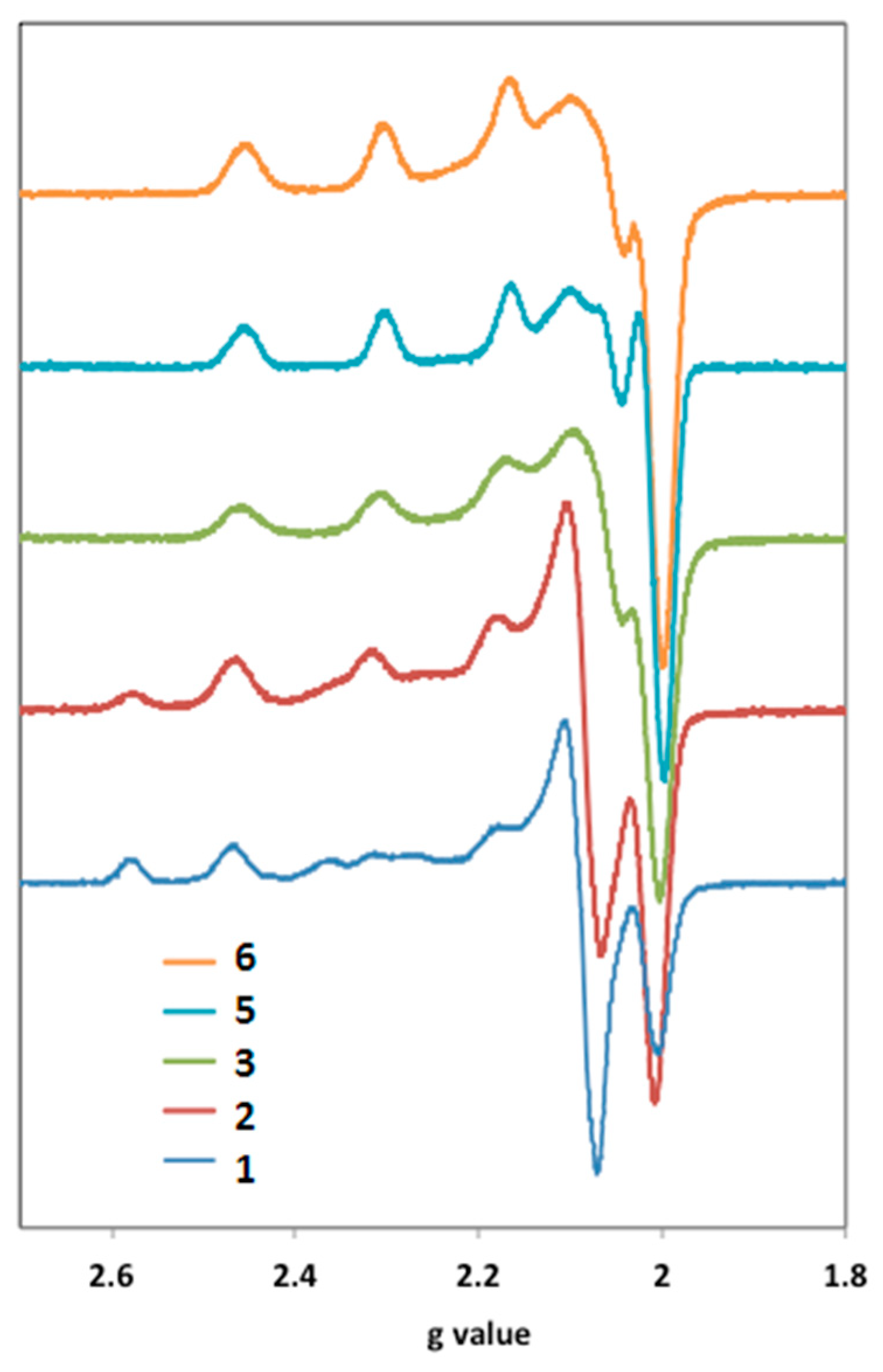
2.3. EPR Characterization
2.4. Thermal Properties
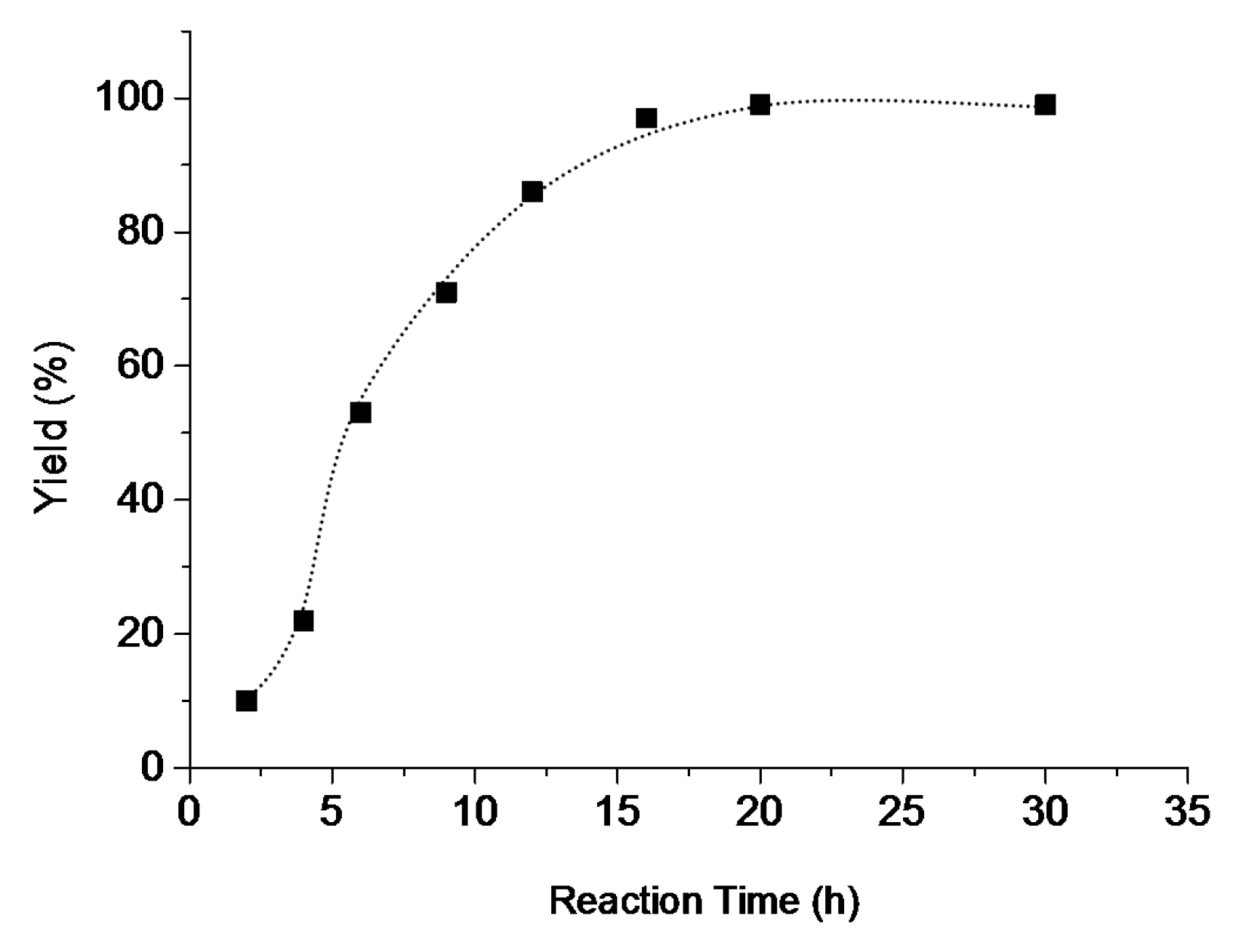
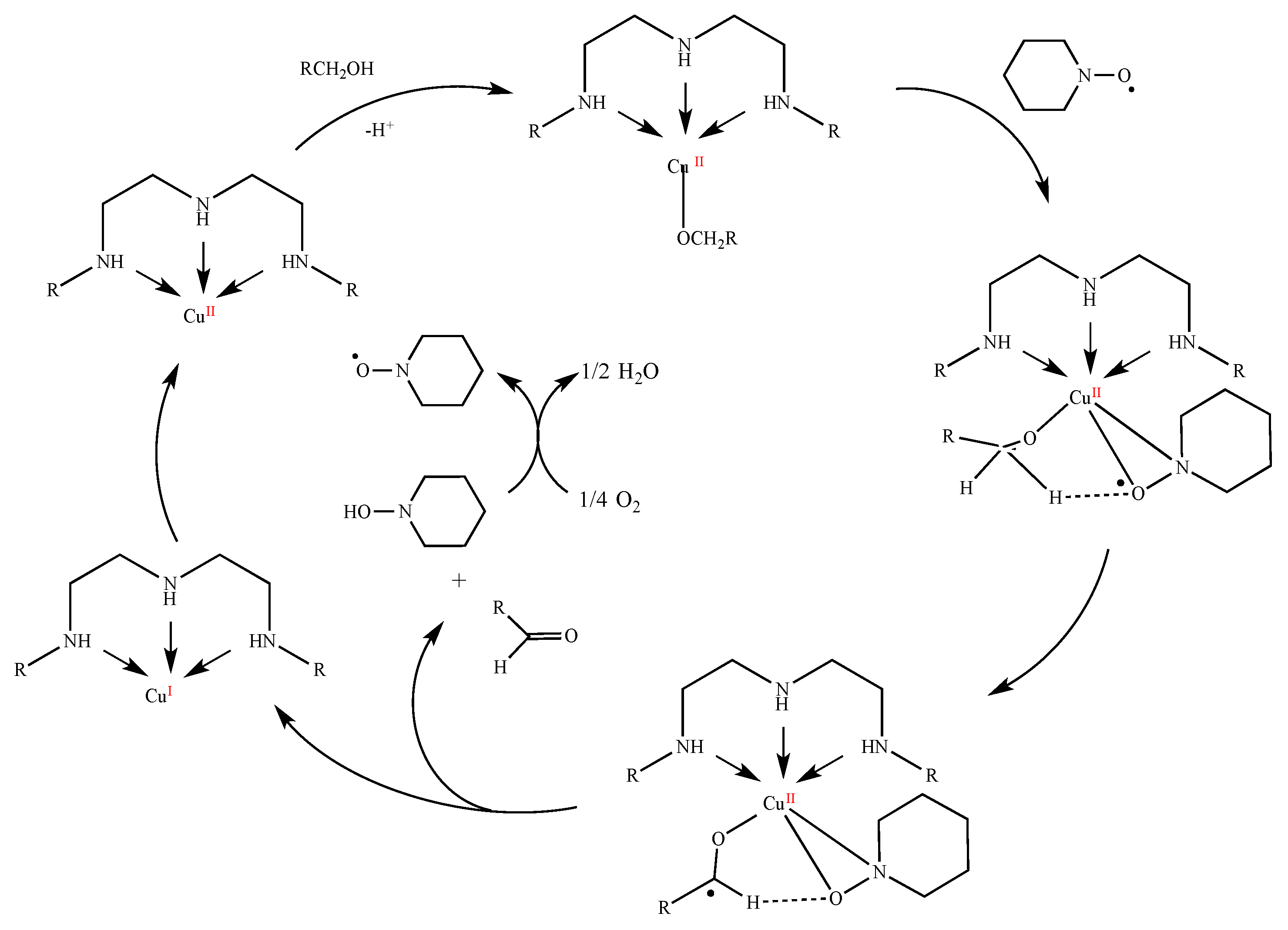
2.5. Catalytic Behavior of Compounds 1–6
3. Materials and Methods
3.1. Crystal Structure Determinations
3.2. Electrochemical Studies
3.3. Catalytic Studies
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Amouri, H.; Desmarets, C.; Moussa, J. Confined Nanospaces in Metallocages: Guest Molecules, Weakly Encapsulated Anions, and Catalyst Sequestration. Chem. Rev. 2012, 112, 2015–2041. [Google Scholar] [CrossRef]
- Alibrandi, G.; Arena, C.G.; Lando, G.; Vecchio, C.L.; Parisi, M.F. {[1.1.1]Cryptand/Imidazole}: A Prototype Composite Kinetic Molecular Device for Automatic NMR Variable pH Reaction Monitoring. Chem. Eur. J. 2011, 17, 1419–1422. [Google Scholar] [CrossRef] [PubMed]
- Garcia, J.; Kuda-Wedagedara, A.N.W.; Allen, M.J. Physical Properties of Eu2+-Containing Cryptates as Contrast Agents for Ultrahigh-Field Magnetic Resonance Imaging. Eur. J. Inorg. Chem. 2012, 2012, 2135–2140. [Google Scholar] [CrossRef] [PubMed]
- Nelson, J.; Mckee, V.; Morgan, G. Coordination Chemistry of Azacryptands. Prog. Inorg. Chem. 2007, 47, 167–317. [Google Scholar]
- Geoghegan, P.; O′ Leary, P. Hydroxyamide-Based Ligands and Their Use in the Asymmetric Catalysis of Key Organic Transformations. ACS Catal. 2012, 2, 573–591. [Google Scholar] [CrossRef]
- Mateus, P.; Delgado, R.; Brandão, P.; Félix, V. Dicarboxylate Recognition by Two Macrobicyclic Receptors: Selectivity for Fumarate over Maleate. J. Org. Chem. 2012, 77, 4611–4621. [Google Scholar] [CrossRef]
- Zhu, K.; Wu, L.; Yan, X.; Zheng, B.; Zhang, M.; Huang, F. Anion-Assisted Complexation of Paraquat by Cryptands Based on Bis(m-phenylene)-[32]crown-10. Chem. Eur. J. 2010, 16, 6088–6098. [Google Scholar] [CrossRef]
- Alibrandi, G.; Vecchio, C.L.; Lando, G. [1.1.1]Cryptand: A Molecular Automatic Titrator. Angew. Chem. Int. Ed. 2009, 48, 6332–6334. [Google Scholar] [CrossRef]
- Xu, Z.; Huang, X.; Liang, J.; Zhang, S.; Zhou, S.; Chen, M.; Tang, M.; Jiang, L. Efficient Syntheses of Novel Cryptands Based on Bis(m-phenylene)-26-crown-8 and Their Complexation with Paraquat. Eur. J. Org. Chem. 2010, 2010, 1904–1911. [Google Scholar] [CrossRef]
- Gamage, N.H.; Mei, Y.; Garcia, J.; Allen, M.J. Oxidatively Stable, Aqueous Europium(II) Complexes through Steric and Electronic Manipulation of Cryptand Coordination Chemistry. Angew. Chem. Int. Ed. 2010, 49, 8923–8925. [Google Scholar] [CrossRef] [PubMed]
- Chaloner, L.; Askari, M.S.; Kutteh, A.; Schindler, S.; Ottenwaelder, X. Formation and Reactivity of a Biomimetic Hydroperoxocopper(II) Cryptate. Eur. J. Inorg. Chem. 2011, 2011, 4204–4211. [Google Scholar] [CrossRef]
- Liu, M.; Li, S.; Hu, M.; Wang, F.; Huang, F. Selectivity Algorithm for the Formation of Two Cryptand/Paraquat Catenanes. Org. Lett. 2010, 12, 760–763. [Google Scholar] [CrossRef]
- Hao, H.G.; Zheng, X.D.; Lu, T.B. Photoinduced Catalytic Reaction by a Fluorescent Active Cryptand Containing an Anthracene Fragment. Angew. Chem. Int. Ed. 2010, 49, 8148–8151. [Google Scholar] [CrossRef]
- Alliger, G.E.; Müller, P.; Do, L.H.; Cummins, C.C.; Nocera, D.G. Family of Cofacial Bimetallic Complexes of a Hexaanionic Carboxamide Cryptand. Inorg. Chem. 2011, 50, 4107–4115. [Google Scholar] [CrossRef]
- Cangelosi, V.M.; Carter, T.G.; Crossland, J.L.; Zakharov, L.N.; Johnson, D.W. Self-Assembled E2L3 Cryptands (E = P, As, Sb, Bi): Transmetalation, Homo- and Heterometallic Assemblies, and Conformational Isomerism. Inorg. Chem. 2010, 49, 9985–9992. [Google Scholar] [CrossRef]
- Yan, X.; Wei, P.; Zhang, M.; Chi, X.; Liu, J.; Huang, F. [2]Pseudorotaxanes Based on the Recognition of Cryptands to Vinylogous Viologens. Org. Lett. 2011, 13, 6370–6373. [Google Scholar] [CrossRef]
- Saeed, M.A.; Wong, B.M.; Fronczek, F.R.; Venkatraman, R.; Hossain, M.A. Formation of an Amine-Water Cyclic Pentamer: A New Type of Water Cluster in a Polyazacryptand. Cryst. Growth Des. 2010, 10, 1486–1488. [Google Scholar] [CrossRef]
- Ma, Z.; Cao, R. An N8O6 cryptand: The host for Ag+ guests and H2O molecule. J. Mol. Struct. 2005, 738, 137–142. [Google Scholar] [CrossRef]
- Alliger, G.E.; Müller, P.; Cummins, C.C.; Nocera, D.G. Cofacial Dicobalt Complex of a Binucleating Hexacarboxamide Cryptand Ligand. Inorg. Chem. 2010, 49, 3697–3699. [Google Scholar] [CrossRef]
- Bischof, C.; Wahsner, J.; Scholten, J.; Trosien, S.; Seitz, M. Quantification of C−H Quenching in Near-IR Luminescent Ytterbium and Neodymium Cryptates. J. Am. Chem. Soc. 2010, 132, 14334–14335. [Google Scholar] [CrossRef]
- Cangelosi, V.M.; Zakharov, L.N.; Johnson, D.W. Supramolecular “Transmetalation” Leads to an Unusual Self-Assembled P2L3 Cryptand. Angew. Chem. Int. Ed. 2010, 49, 1248–1251. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, A.; Nakagawa, Y.; Uehara, K.; Hikichi, S.; Mizuno, N. Inorganic Cryptand: Size-Selective Strong Metallic Cation Encapsulation by a Disilicoicosatungstate (Si2W20) Polyoxometalate. Angew. Chem. Int. Ed. 2009, 48, 7055–7058. [Google Scholar] [CrossRef] [PubMed]
- Shen, Z.; Jordan, R.F. Self-Assembled Tetranuclear Palladium Catalysts That Produce High Molecular Weight Linear Polyethylene. J. Am. Chem. Soc. 2010, 132, 52–53. [Google Scholar] [CrossRef] [PubMed]
- Mateus, P.; Delgado, R.; Brandão, P.; Félix, V. Recognition of Oxalate by a Copper(II) Polyaza Macrobicyclic Complex. Chem. Eur. J. 2011, 17, 7020–7031. [Google Scholar] [CrossRef] [PubMed]
- Hossain, M.A.; Saeed, M.A.; Fronczek, F.R.; Wong, B.M.; Dey, K.R.; Mendy, J.S.; Gibson, D. Charge-Assisted Encapsulation of Two Chlorides by a Hexaprotonated Azamacrocycle. Cryst. Growth Des. 2010, 10, 1478–1481. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Shi, H.; Deng, X.; Guedes da Silva, M.F.C.; Martins, L.M.D.R.S.; Pombeiro, A.J.L. Silver coordination polymers with tri- and hexacyanoethyl-functionalized macrocyclic ligands. Dalton Trans. 2015, 44, 1388–1396. [Google Scholar] [CrossRef]
- Ghosh, M.; Weyhermuller, T.; Wieghardt, K. Electronic structure of the members of the electron transfer series [NiL]z (z = 3+, 2+, 1+, 0) and [NiL(X)]n (X = Cl, CO, P(OCH3)3) species containing a tetradentate, redox-noninnocent, Schiff base macrocyclic ligand L: An experimental and density functional theoretical study. Dalton Trans. 2010, 39, 1996–2007. [Google Scholar] [PubMed]
- Gamez, P.; Aubel, P.G.; Driessen, W.L.; Reedijk, J. Homogeneous bio-inspired copper-catalyzed oxidation reactions. Chem. Soc. Rev. 2001, 30, 376–385. [Google Scholar] [CrossRef]
- Ma, Z.; Zhang, B.; Guedes da Silva, M.F.C.; Silva, J.; Mendo, A.S.; Baptista, P.V.; Fernandes, A.R.; Pombeiro, A.J.L. Synthesis, characterization, thermal properties and antiproliferative potential of copper(II) 4’-phenylterpyridine compounds. Dalton Trans. 2016, 45, 5339–5355. [Google Scholar] [CrossRef]
- Lewis, E.A.; Tolman, W.B. Reactivity of Dioxygen−Copper Systems. Chem. Rev. 2004, 104, 1047–1076. [Google Scholar] [CrossRef]
- Itoh, S. Comprehensive Coordination Chemistry, 2nd ed.; McCleverty, J.A., Meyer, T.J., Eds.; Elsevier: Amsterdam, The Netherlands, 2003; Volume 8, Chapter 8.15; p. 369. [Google Scholar]
- Lee, D.H. Comprehensive Coordination Chemistry, 2nd ed.; McCleverty, J.A., Meyer, T.J., Eds.; Elsevier: Amsterdam, The Netherlands, 2003; Volume 8, Chapter 8.17; p. 437. [Google Scholar]
- Fraústo da Silva, J.J.R.; Williams, R.J.P. The Biological Chemistry of the Elements; Oxford University Press: Oxford, UK, 2001. [Google Scholar]
- Solomon, E.I.; Sundaram, U.M.; Machonkin, T.E. Multicopper Oxidases and Oxygenases. Chem. Rev. 1996, 96, 2563–2606. [Google Scholar] [CrossRef] [PubMed]
- Klinman, J.P. Mechanisms Whereby Mononuclear Copper Proteins Functionalize Organic Substrates. Chem. Rev. 1996, 96, 2541–2562. [Google Scholar] [CrossRef]
- Ayala, M.; Torres, E. Enzymatic activation of alkanes: Constraints and prospective. Appl. Catal. A Gen. 2004, 272, 1–13. [Google Scholar] [CrossRef]
- Elliot, S.J.; Zhu, M.; Tso, L.; Nguyen, H.-H.T.; Yip, J.H.-K.; Chan, S.I. Regio- and Stereoselectivity of Particulate Methane Monooxygenase from Methylococcus capsulatus (Bath). J. Am. Chem. Soc. 1997, 119, 9949–9955. [Google Scholar] [CrossRef]
- Lieberman, R.L.; Rosenzweig, A.C. Biological methane oxidation: Regulation, biochemistry, and active site structure of particulate methane monooxygenase. Crit. Rev. Biochem. Mol. Biol. 2004, 39, 147–164. [Google Scholar] [CrossRef]
- Lieberman, R.L.; Rosenzweig, A.C. Crystal structure of a membrane-bound metalloenzyme that catalyses the biological oxidation of methane. Nature 2005, 434, 177–182. [Google Scholar] [CrossRef]
- Kopylovich, M.N.; Ribeiro, A.P.C.; Alegria, E.C.B.A.; Martins, N.M.R.; Martins, L.M.D.R.S.; Pombeiro, A.J.L. Catalytic oxidation of alcohols: Recent advances. Adv. Organometal. Chem. 2015, 63, 91–174. [Google Scholar]
- Pombeiro, A.J.L.; Guedes da Silva, M.F.C.; Lemos, M.A.N.D.A. Electron-Transfer Induced Isomerizations of Coordination Compounds. Coord. Chem. Rev. 2001, 219, 53–80. [Google Scholar] [CrossRef]
- Addison, A.W.; Rao, T.N.; Reedijk, J.; van Rijn, J.; Verschoor, G.C. Synthesis, structure, and spectroscopic properties of copper(II) compounds containing nitrogen–sulphur donor ligands; the crystal and molecular structure of aqua[1,7-bis(N-methylbenzimidazol-2-yl)-2,6-dithiaheptane]copper(II) perchlorate. J. Chem. Soc. Dalton Trans. 1984, 1349–1356. [Google Scholar] [CrossRef]
- Di Nicola, C.; Garau, F.; Karabach, Y.Y.; Martins, L.M.D.R.S.; Monari, M.; Pandolfo, L.; Pettinari, C.; Pombeiro, A.J.L. Trinuclear Triangular Copper(II) Clusters-Synthesis, Electrochemical Studies and Catalytic Peroxidative Oxidation of Cycloalkanes. Eur. J. Inorg. Chem. 2009, 2009, 666–676. [Google Scholar] [CrossRef]
- Lewis, W.B.; Alei, M., Jr.; Morgan, L.O. Magnetic resonance studies on copper(II) complex ions in solution. I. Temperature dependences of the 17O NMR and copper(II) EPR linewidths of Cu(H2O)62+. J. Chem. Phys. 1966, 44, 2409–2417. [Google Scholar] [CrossRef]
- Sakaguchi, U.; Addison, A.W. Spectroscopic and redox studies of some copper(II) complexes with biomimetic donor atoms: Implications for protein copper centres. J. Chem. Soc. Dalton Trans. 1979, 1979, 600–608. [Google Scholar] [CrossRef]
- Kopylovich, M.N.; Mahmudov, K.T.; Guedes da Silva, M.F.C.; Kuznetsov, M.L.; Figiel, P.J.; Karabach, Y.Y.; Luzyanin, K.V.; Pombeiro, A.J.L. Ortho-hydroxyphenylhydrazo-β-diketones: Tautomery, Coordination Ability and Catalytic Activity of Their Copper(II) Complexes toward Oxidation of Cyclohexane and Benzylic Alcohols. Inorg. Chem. 2011, 50, 918–931. [Google Scholar] [CrossRef]
- Sheldon, R.A.; Arends, I.W.C.E. Organocatalytic Oxidations Mediated by Nitroxyl Radicals. Adv. Synth. Catal. 2004, 346, 1051–1071. [Google Scholar] [CrossRef]
- de Mico, A.; Margarita, R.; Mariani, A.; Piancatelli, G. Radical additions to olefins in the presence of iodobenzenediacetate: An easy route to alkyl dithiocyanates. Tetrahedron Lett. 1996, 37, 1889–1892. [Google Scholar] [CrossRef]
- Dijksman, A.; Arends, I.W.C.E.; Sheldon, R.A. The ruthenium/TEMPO-catalysed aerobic oxidation of alcohols. Platin. Metals Rev. 2001, 45, 15–19. [Google Scholar]
- Mahmudov, K.T.; Kopylovich, M.N.; Guedes da Silva, M.F.C.; Figiel, P.J.; Karabach, Y.Y.; Pombeiro, A.J.L. New copper(II) dimer with 3-(2-hydroxy-4-nitro-phenylhydrazo)pentane-2,4-dione and its catalytic activity in cyclohexane and benzyl alcohol oxidations. J. Mol. Catal. A Chem. 2010, 318, 44–50. [Google Scholar] [CrossRef]
- Soroceanu, A.; Cazacu, M.; Shova, S.; Turta, C.; Kožíšek, J.; Gall, M.; Breza, M.; Rapta, P.; Mac Leod, T.C.O.; Pombeiro, A.J.L.; et al. Copper(II) Complexes with Schiff Bases Containing a Disiloxane Unit: Synthesis, Structure, Bonding Features and Catalytic Activity for Aerobic Oxidation of Benzyl Alcohol. Eur. J. Inorg. Chem. 2013, 2013, 1458–1474. [Google Scholar] [CrossRef]
- Arends, I.W.C.A.; Sheldon, R.A. Modern Oxidation Methods; Bäckvall, J.-E., Ed.; Wiley: Weinheim, Germany, 2004; p. 83. [Google Scholar]
- Michel, C.; Belanzoni, P.; Gamez, P.; Reedijk, J.; Baerends, E.J. Activation of the C−H Bond by Electrophilic Attack: Theoretical Study of the Reaction Mechanism of the Aerobic Oxidation of Alcohols to Aldehydes by the Cu(bipy)2+/2,2,6,6-Tetramethylpiperidinyl-1-oxy Cocatalyst System. Inorg. Chem. 2009, 48, 11909–11920. [Google Scholar] [CrossRef]
- Sutradhar, M.; Martins, L.M.D.R.S.; Guedes da Silva, M.F.C.; Alegria, E.C.B.A.; Liu, C.-M.; Pombeiro, A.J.L. Dinuclear Mn(II,II) complexes: Magnetic properties and microwave assisted oxidation of alcohols. Dalton Trans. 2014, 43, 3966–3977. [Google Scholar] [CrossRef]
- Arion, V.B.; Platzer, S.; Rapta, P.; Machata, P.; Breza, M.; Vegh, D.; Dunsch, L.; Telser, J.; Shova, S.; Mac Leod, T.C.O.; et al. Marked stabilization of redox states and enhanced catalytic activity in galactose oxidase models based on transition metal S-methylisothiosemicarbazonates with –SR group in ortho-position to the phenolic oxygen. Inorg. Chem. 2013, 52, 7524–7540. [Google Scholar] [CrossRef]
- Sabbatini, A.; Martins, L.M.D.R.S.; Mahmudov, K.T.; Kopylovich, M.N.; Drew, M.G.B.; Pettinari, C.; Pombeiro, A.J.L. Microwave-assisted and solvent-free peroxidative oxidation of 1-phenylethanol to acetophenone with a CuII–TEMPO catalytic system. Catal. Commun. 2014, 48, 69–72. [Google Scholar] [CrossRef]
- Sheldon, R.A. E factors, green chemistry and catalysis: An odyssey. Chem. Commun. 2008, 29, 3352−3365. [Google Scholar] [CrossRef]
- Sutradhar, M.; Alegria, E.C.B.A.; Mahmudov, K.T.; Guedes da Silva, M.F.C.; Pombeiro, A.J.L. Iron(III) and cobalt(III) complexes with both tautomeric (keto and enol) forms of aroylhydrazone ligands: Catalysts for the microwave assisted oxidation of alcohols. RSC Adv. 2016, 6, 8079–8088. [Google Scholar] [CrossRef]
- Dronova, M.S.; Bilyachenko, A.N.; Yalymov, A.I.; Kozlov, Y.N.; Shul’pina, L.S.; Korlyukov, A.A.; Arkhipov, D.E.; Levitsky, M.M.; Shubina, E.S.; Shul’pin, G.B. Solvent-controlled synthesis of tetranuclear cage-like copper(II) silsesquioxanes. Remarkable features of the cage structures and their high catalytic activity in oxidation with peroxides. Dalton Trans. 2014, 43, 872–882. [Google Scholar] [CrossRef]
- Sutradhar, M.; Martins, L.M.D.R.S.; Carabineiro, S.A.C.; Guedes da Silva, M.F.C.; Buijnsters, J.G.; Figueiredo, J.L.; Pombeiro, A.J.L. Oxidovanadium(V) complexes anchored on carbon materials as catalysts for the oxidation of 1-phenylethanol. ChemCatChem 2016, 8, 2254–2266. [Google Scholar] [CrossRef]
- Rockenbauer, A.; Korecz, L. Automatic computer simulations of ESR spectra. Appl. Magn. Reson. 1996, 10, 29–43. [Google Scholar] [CrossRef]
- CrysAlisPRO, version 171.36.20; Oxford Diffraction/Agilent Technologies UK Ltd.: Yarnton, Oxfordshire, UK, 2011.
- Bruker. APEX2; Bruker AXS Inc.: Madison, WI, USA, 2012. [Google Scholar]
- Sheldrick, G.M. SADABS. Program for Empirical Absorption Correction; University of Gottingen: Gottingen, Germany, 1996. [Google Scholar]
- Sheldrick, G.M. SHELX97. Programs for Crystal Structure Analysis (Release 97-2); University of Göttingen: Göttingen, Germany, 1997. [Google Scholar]
- Farrugia, L.J. WinGX suite for small-molecule single-crystal crystallography. J. Appl. Crystallogr. 1999, 32, 837–838. [Google Scholar] [CrossRef]
- Guedes da Silva, M.F.C.; Pombeiro, A.J.L. Redox Potential Parameterization in Coordination Compounds with Polydentate Scorpionate and Benzene Ligands. Electrochim. Acta 2012, 82, 478–483. [Google Scholar] [CrossRef]
- Ma, Z.; Liu, S.-X. Synthesis and Characterization of a Macrocyclic Compound, C40H46N6O4. Chin. J. Struct. Chem. 2003, 22, 553–557. [Google Scholar]
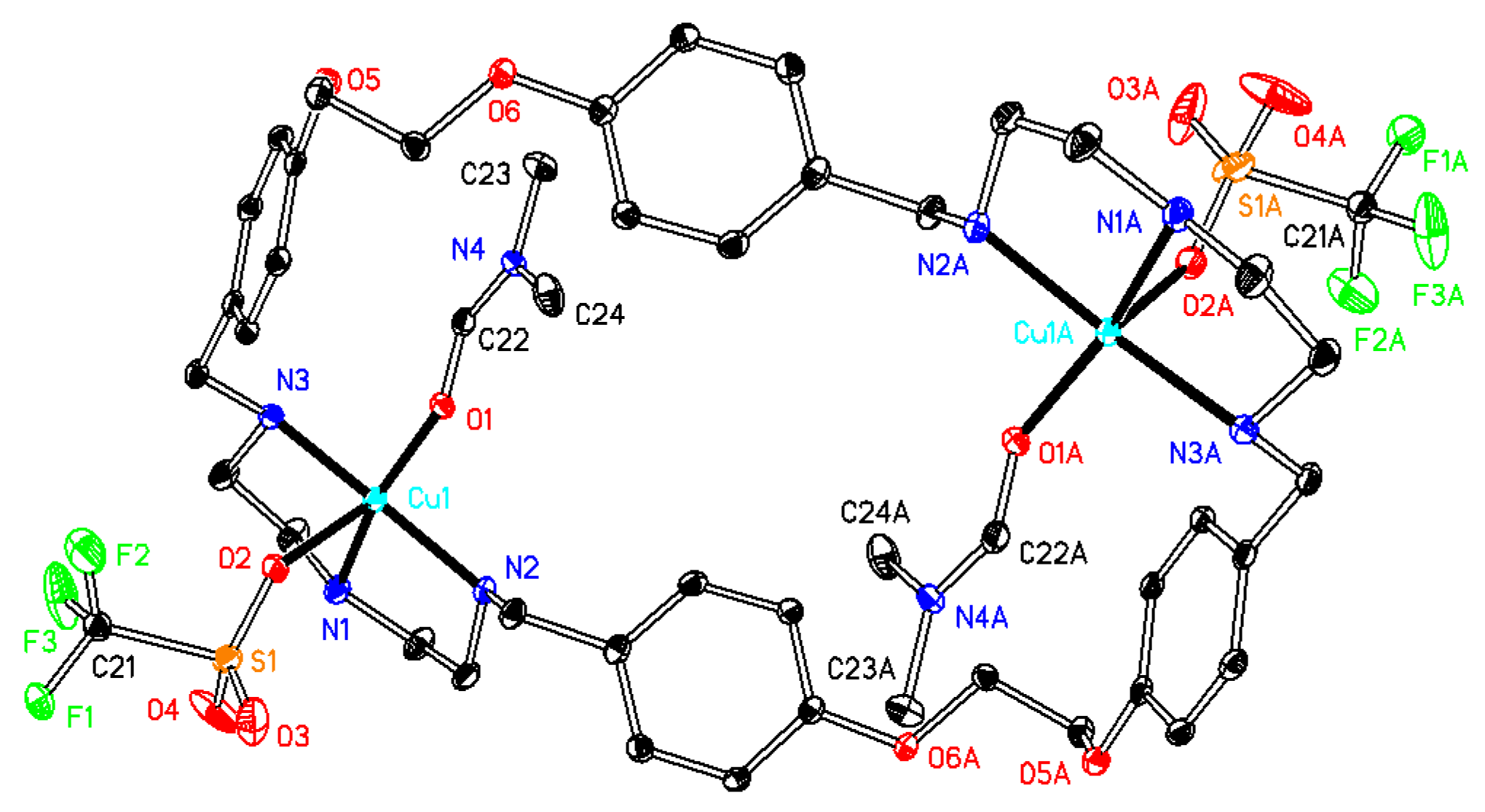
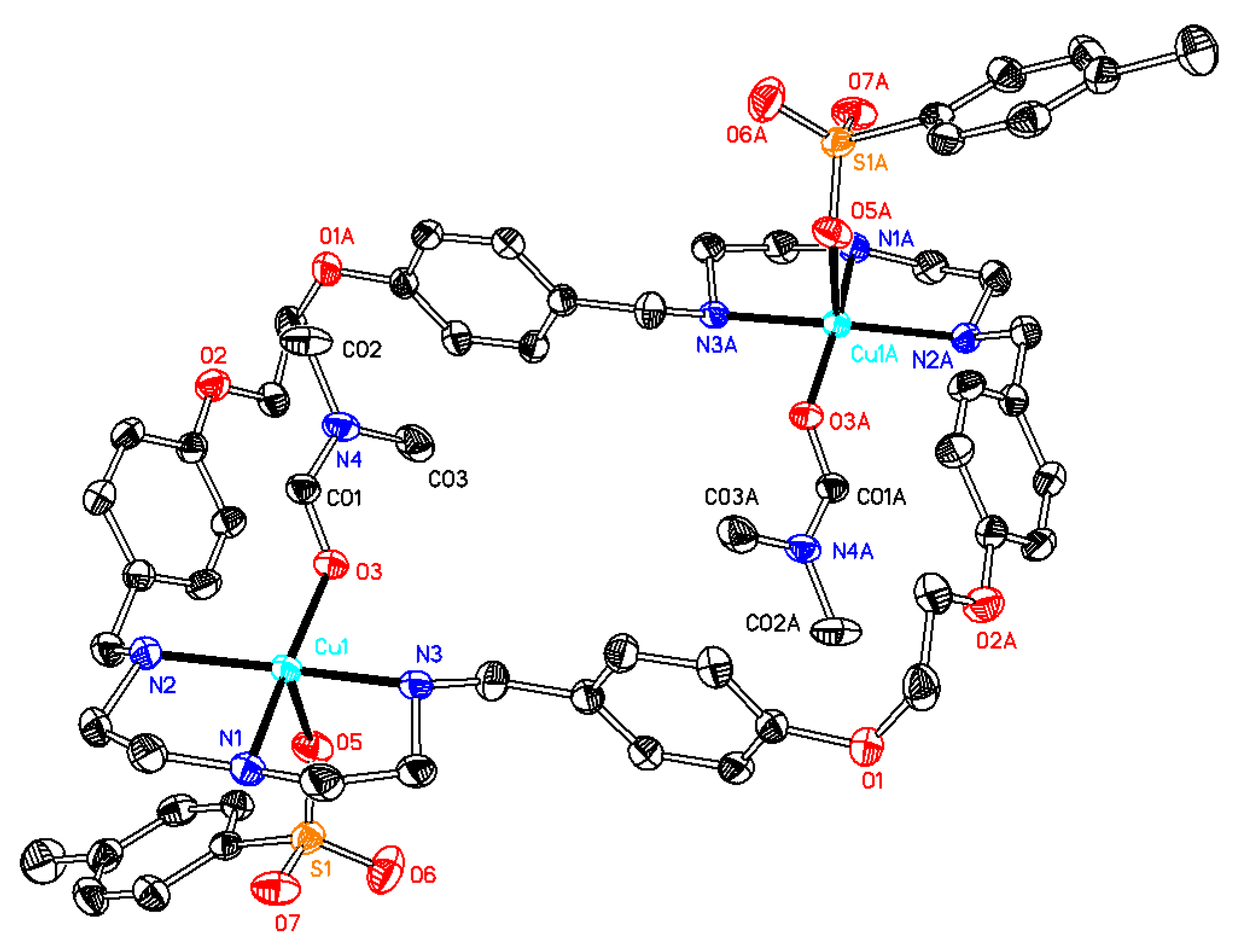
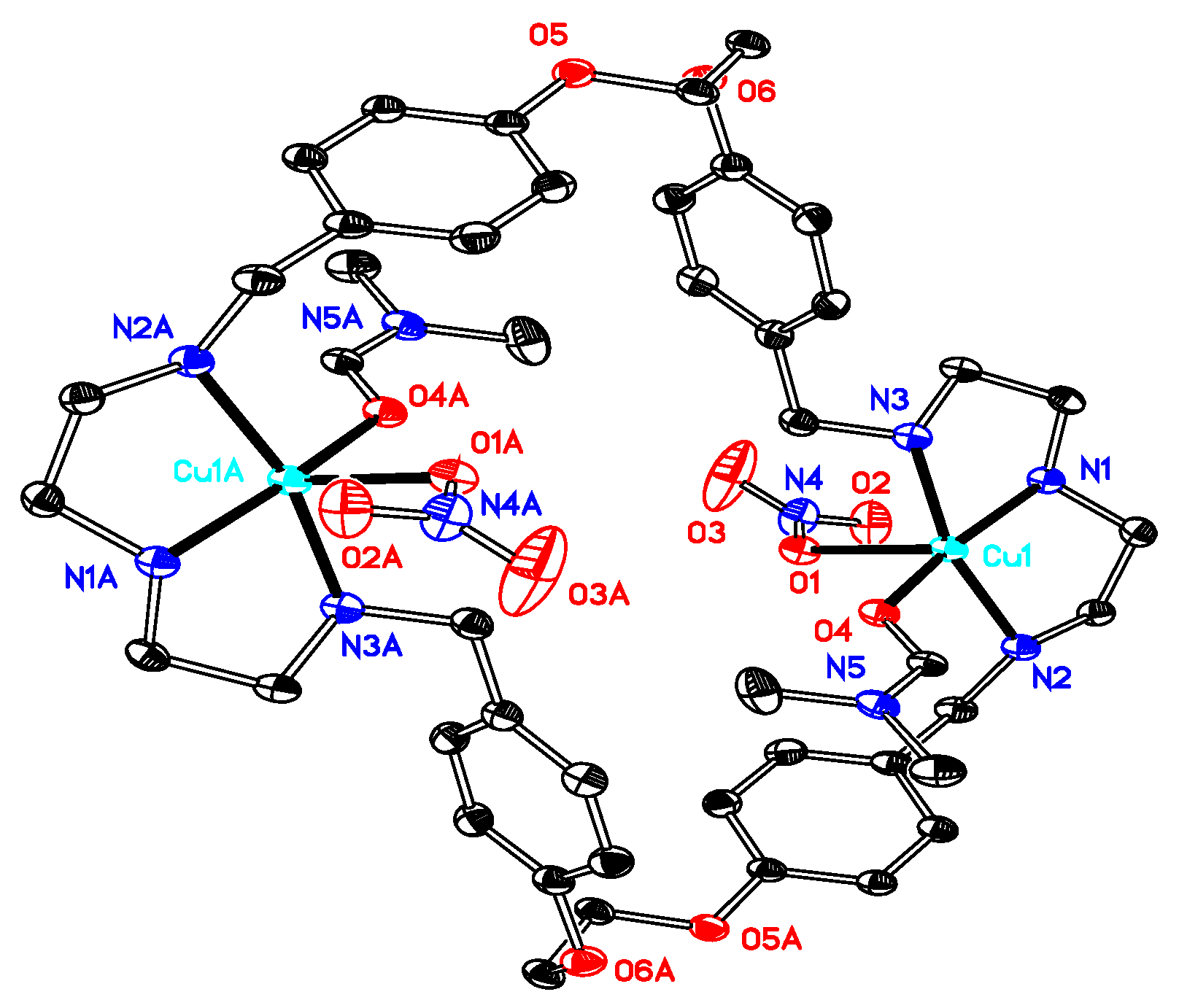
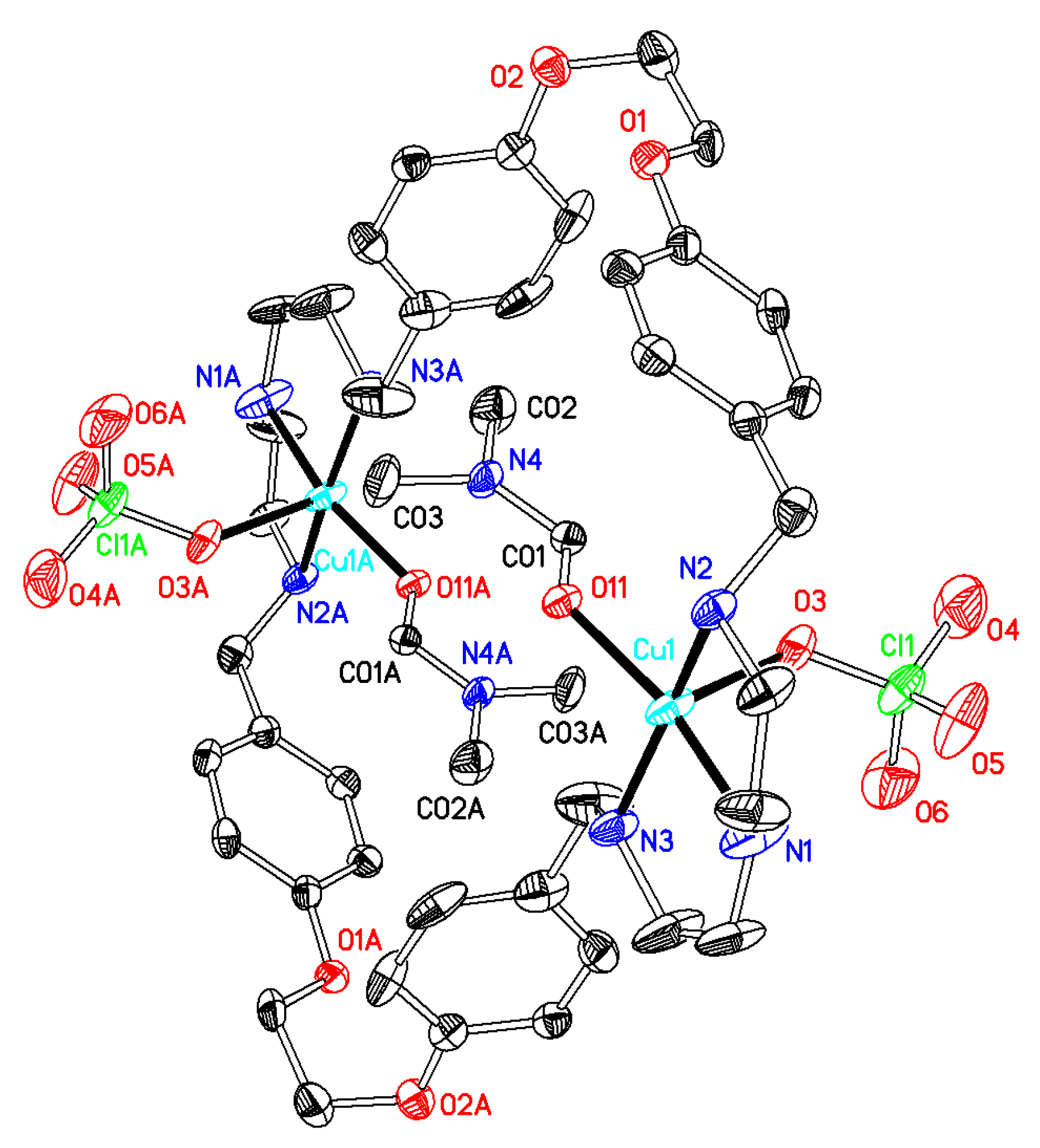
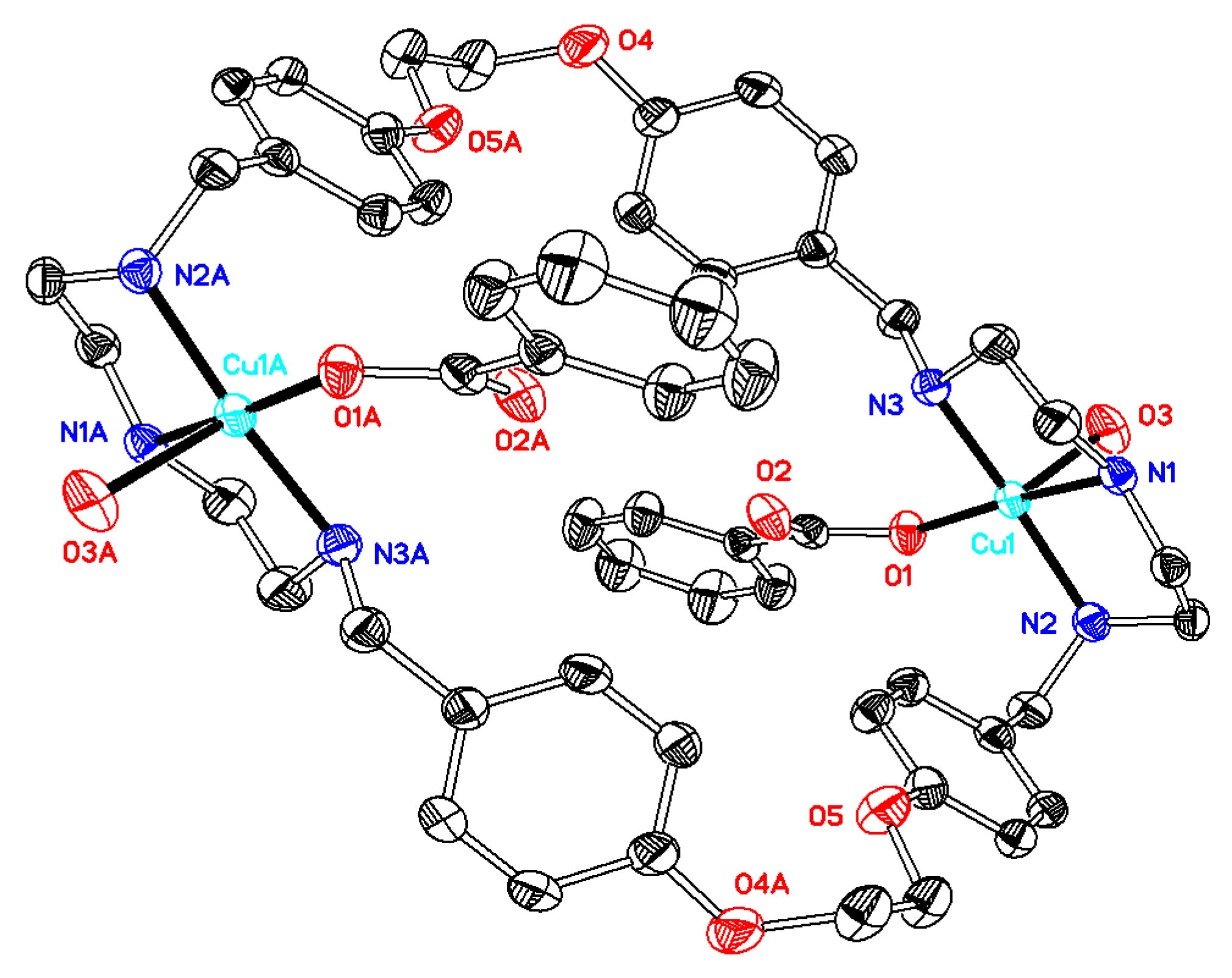
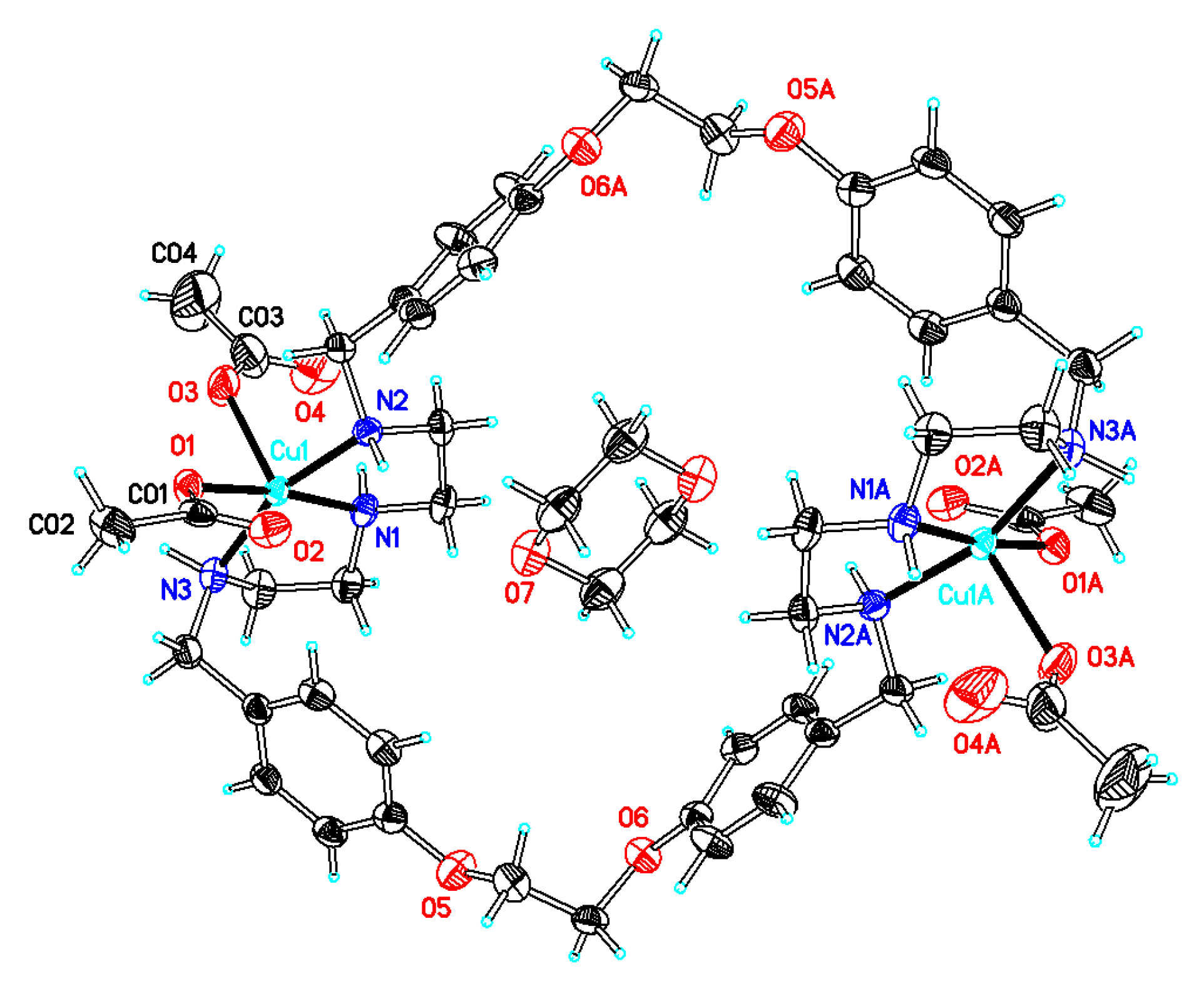
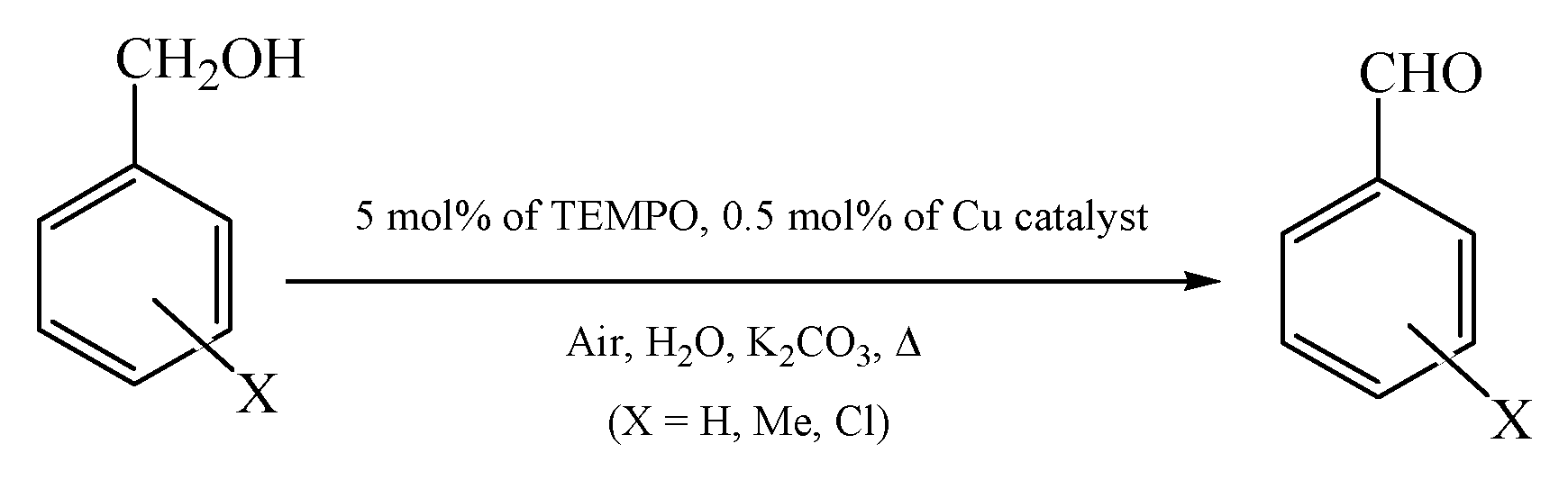

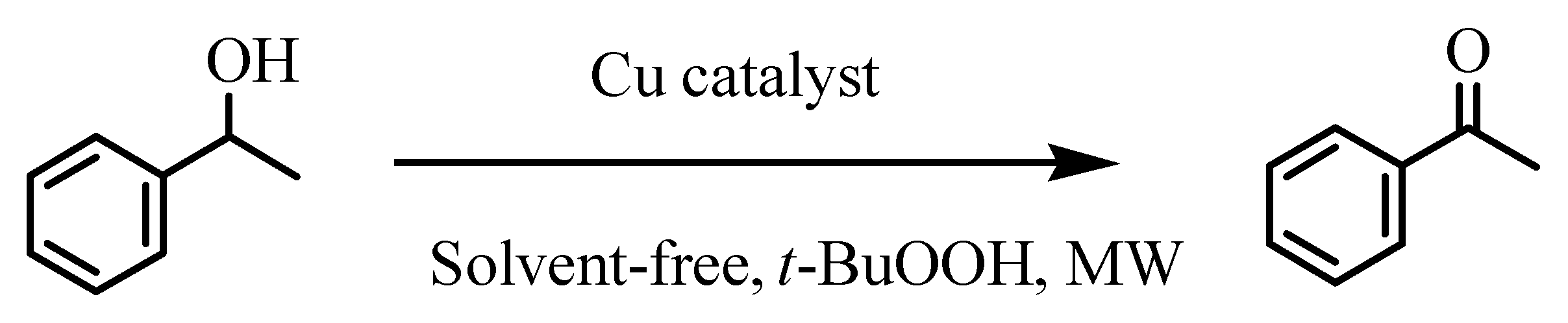
| 1·0.5H2O | 2·2DMF·2H2O | 3·6DMF·0.5CH4O | 4·2H2O | 5·C4H8O2·2H2O | 6·C4H8O2·5H2O | |
|---|---|---|---|---|---|---|
| empirical formula | C50H69Cu2F12N8O18.5S4 | C80H114Cu2N10O22S4 | C64.5H106Cu2N18O24.5 | C46H72Cl4Cu2N8O24 | C72H90Cu2N6O18 | C52H84Cu2N6O19 |
| fw | 1561.51 | 1823.13 | 1652.77 | 1390.00 | 1454.60 | 1224.35 |
| cryst syst | Triclinic | Triclinic | Triclinic | Monoclinic | Triclinic | Triclinic |
| T (K) | 120 | 150 | 120 | 150 | 298 | 150 |
| Space group | P | P | P | P21/n | P | P |
| a (Å) | 10.832(2) | 13.0363(19) | 12.9020(6) | 17.7983(6) | 9.1949(18) | 9.5711(19) |
| b (Å) | 12.331(3) | 13.801(2) | 13.3540(6) | 8.9329(3) | 11.473(2) | 9.823(2) |
| c (Å) | 13.336(3) | 14.449(3) | 14.4500(8) | 18.9816(6) | 18.881(4) | 18.144(4) |
| α (deg) | 79.37(3) | 100.354(8) | 109.270(5) | 90 | 73.07(3) | 78.20(3) |
| β (deg) | 70.16(3) | 99.297(8) | 95.630(4) | 95.854(2) | 78.86(3) | 79.53(3) |
| γγ (deg) | 85.05(3) | 116.479(8) | 112.130(5) | 90 | 80.88(3) | 61.04(3) |
| V (Å3) | 1646.3(6) | 2201.0(6) | 2105.41(18) | 3002.15(17) | 1858.5(6) | 1454.2(5) |
| Z | 1 | 1 | 1 | 2 | 1 | 1 |
| Dcalc (Mg m−3) | 1.575 | 1.375 | 1.304 | 1.538 | 1.300 | 1.398 |
| μ (Mo Kα) (mm−1) | 0.880 | 0.654 | 0.585 | 0.971 | 0.643 | 0.808 |
| Independent reflections | 5801 | 11065 | 7615 | 5220 | 9219 | 6338 |
| Observed reflections | 5239 | 8164 | 6375 | 2934 | 7292 | 5381 |
| Rint | 0.0223 | 0.0368 | 0.0310 | 0.0539 | 0.0208 | 0.0241 |
| R a | 0.0733 | 0.0453 | 0.0780 | 0.0736 | 0.0555 | 0.0364 |
| wR b | 0.1828 | 0.1278 | 0.2239 | 0.1839 | 0.1632 | 0.0921 |
| 1 | 2 | 3 | 4 | 5 | 6 | |
|---|---|---|---|---|---|---|
| τ550 | 0.03 | 0.08 | 0.04 | 0.03 | 0.15 | 0.15 |
| d(Cu2+) relative to l.s. basal plane | −0.1785 (18) | −0.2043(3) | 0.6311(14) | −0.2457 (11) | 0.1671(11) | −0.1898(3) |
| Cu–Oequatorial | 1.955(3) | 1.9703(15) | 1.979(2) | 1.927(4) | 1.9561(19) | 1.9461(18) |
| Cu–Oaxial | 2.275(3) | 2.2518(17) | 2.240(3) | 2.352(5) | 2.264(2) | 2.211(2) |
| Cu–N | 2.000(4) | 1.9942(19) | 1.997(4) | 1.986(6) | 2.004(2) | 2.002(2) |
| 2.024(3) | 2.0470(18) | 2.040(3) | 2.011(5) | 2.044(2) | 2.025(2) | |
| 2.036(3) | 2.0729(18) | 2.042(4) | 2.031(5) | 2.046(2) | 2.032(2) | |
| ∠ N2–Cu–N3 | 168.07(14) | 163.46(8) | 165.69 | 163.5(2) | 164.30 | 163.01(8) |
| Intramolecular Cu⋅⋅⋅Cu | 12.518(5) | 10.888(2) | 11.032(1) | 10.990(2) | 12.019(4) | 11.941(4) |
| Largest intramolecular N⋅⋅⋅N | 15.858(9) Å | 14.215(5) | 14.350(6) | 14.42(1) | 12.246(6) | 11.316(5) |
| Chirality at N2, N3 (N2i, N3i) | SR(RS) | RS(SR) | SR(RS) | SR(RS) | RR(SS) | RR(SS) |
| Compound | redEp (Ired) | oxEp (Iox) b |
|---|---|---|
| 1 | −1.78 | −0.11 |
| 2 | −1.67 | −0.01 |
| 3 | −1.53 | 0.10 |
| 4 | −1.44 | 0.24 |
| 5 | −1.60 | −0.02 |
| 6 | −1.87 | −0.05 |
| Compound | gx | gy | gz | Ax [×10−4cm−1] | Ay [×10−4cm−1] | Az [×10−4cm−1] | % | gz/Az [×10−4cm] |
|---|---|---|---|---|---|---|---|---|
| 1 | 2.082 | 2.082 | 2.410 | 0.0 | 0.0 | 132.8 | 55 | 181.5 |
| 2.042 | 2.069 | 2.242 | 29.2 | 26.9 | 181.1 | 44 | 123.8 | |
| 2 | 2.080 | 2.080 | 2.410 | 0.0 | 0.0 | 132.8 | 31 | 181.5 |
| 2.042 | 2.070 | 2.243 | 29.2 | 26.9 | 181.2 | 68 | 123.8 | |
| 3 | 2.033 | 2.063 | 2.239 | 24.1 | 34.3 | 180.8 | 100 | 123.8 |
| 5 | 2.032 | 2.060 | 2.229 | 24.0 | 33.6 | 183.9 | 100 | 121.2 |
| 6 | 2.032 | 2.060 | 2.230 | 22.4 | 35.8 | 181.4 | 100 | 122.9 |
| Entry | Complex | Substrate | Product | Time (h) | Yield b (%) | TON [TOF(h−1)] c |
|---|---|---|---|---|---|---|
| 1 d | - | benzyl alcohol | benzaldehyde | 20 | 5 | 10 (1) |
| 2 e | L | benzyl alcohol | benzaldehyde | 20 | 4 | 9 (1) |
| 3 | 1 | benzyl alcohol | benzaldehyde | 20 | 97 | 197 (10) |
| 4 | 2 | benzyl alcohol | benzaldehyde | 20 | 99 | 201 (10) |
| 5 | 3 | benzyl alcohol | benzaldehyde | 20 | 94 | 191 (10) |
| 6 | 4 | benzyl alcohol | benzaldehyde | 20 | 79 | 160 (8) |
| 7 | 5 | benzyl alcohol | benzaldehyde | 20 | 84 | 168 (8) |
| 8 | 6 | benzyl alcohol | benzaldehyde | 20 | 99 | 232 (12) |
| 9 f | 6 | benzyl alcohol | benzaldehyde | 20 | 7 | 14 (1) |
| 10 g | 6 | benzyl alcohol | benzaldehyde | 20 | 27 | 55 (3) |
| 11 | 6 | benzyl alcohol | benzaldehyde | 6 | 54 | 110 (18) |
| 12 | 6 | benzyl alcohol | benzaldehyde | 9 | 64 | 130 (14) |
| 13 h | 6 | benzyl alcohol | benzaldehyde | 20 | 3 | 9 (1) |
| 14 i | 6 | benzyl alcohol | benzaldehyde | 20 | 26 | 53 (3) |
| 15 j | 6 | benzyl alcohol | benzaldehyde | 20 | 86 | 166 (8) |
| 16 | 6 | 2-Me-benzyl alcohol | 2-Me-benzaldehyde | 20 | 64 | 129 (14) |
| 17 | 6 | 4-Me-benzyl alcohol | 4-Me-benzaldehyde | 20 | 88 | 171 (9) |
| 18 | 6 | 2-Cl-benzyl alcohol | 2-Cl benzaldehyde | 20 | 72 | 146 (7) |
| 19 | 6 | 4-Cl-benzyl alcohol | 4-Cl-benzaldehyde | 20 | 91 | 185 (9) |
| 20 | 6 | cinnamyl alcohol | cinnamaldehyde | 20 | 57 | 108 (5) |
| 21 | 6 | 1-phenylethanol | acetophenone | 20 | 63 | 126 (6) |
| Entry | Catalyst Precursor | Reaction Time (h) | Temperature (°C) | Additive (µmol) | Yield b (%) | TON [TOF(h−1)] c |
|---|---|---|---|---|---|---|
| 1 | 1 | 0.5 | 120 | - | 79 | 390 (780) |
| 2 | 2 | 0.5 | 120 | - | 66 | 326 (652) |
| 3 | 3 | 0.5 | 120 | - | 81 | 412 (824) |
| 4 | 4 | 0.5 | 120 | - | 64 | 302 (604) |
| 5 | 5 | 0.5 | 120 | - | 68 | 325 (650) |
| 6 | 6 | 0.5 | 120 | - | 99 | 542 (1.1 × 103) |
| 7 | 2 | 0.5 | 80 | - | 13 | 114 (228) |
| 8 | 2 | 6 | 80 | - | 26 | 256 (43) |
| 9 d | 2 | 0.5 | 80 | - | 42 | 105 (210) |
| 10 | 2 | 0.5 | 120 | TEMPO (30) | 99 | 496 (992) |
| 11 | 6 | 0.5 | 80 | - | 29 | 283 (566) |
| 12 | 6 | 6 | 80 | - | 97 | 1.1 × 103 (182) |
| 13 | 6 | 0.5 | 80 | TEMPO (30) | 49 | 249 (498) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, Z.; Wang, Q.; C. B. A. Alegria, E.; C. Guedes da Silva, M.F.; M. D. R. S. Martins, L.; Telo, J.P.; Correia, I.; J. L. Pombeiro, A. Synthesis and Structure of Copper Complexes of a N6O4 Macrocyclic Ligand and Catalytic Application in Alcohol Oxidation. Catalysts 2019, 9, 424. https://doi.org/10.3390/catal9050424
Ma Z, Wang Q, C. B. A. Alegria E, C. Guedes da Silva MF, M. D. R. S. Martins L, Telo JP, Correia I, J. L. Pombeiro A. Synthesis and Structure of Copper Complexes of a N6O4 Macrocyclic Ligand and Catalytic Application in Alcohol Oxidation. Catalysts. 2019; 9(5):424. https://doi.org/10.3390/catal9050424
Chicago/Turabian StyleMa, Zhen, Qijun Wang, Elisabete C. B. A. Alegria, M. Fátima C. Guedes da Silva, Luísa M. D. R. S. Martins, João P. Telo, Isabel Correia, and Armando J. L. Pombeiro. 2019. "Synthesis and Structure of Copper Complexes of a N6O4 Macrocyclic Ligand and Catalytic Application in Alcohol Oxidation" Catalysts 9, no. 5: 424. https://doi.org/10.3390/catal9050424
APA StyleMa, Z., Wang, Q., C. B. A. Alegria, E., C. Guedes da Silva, M. F., M. D. R. S. Martins, L., Telo, J. P., Correia, I., & J. L. Pombeiro, A. (2019). Synthesis and Structure of Copper Complexes of a N6O4 Macrocyclic Ligand and Catalytic Application in Alcohol Oxidation. Catalysts, 9(5), 424. https://doi.org/10.3390/catal9050424