Chiral Phase-Transfer Catalysts with Hydrogen Bond: A Powerful Tool in the Asymmetric Synthesis
Abstract
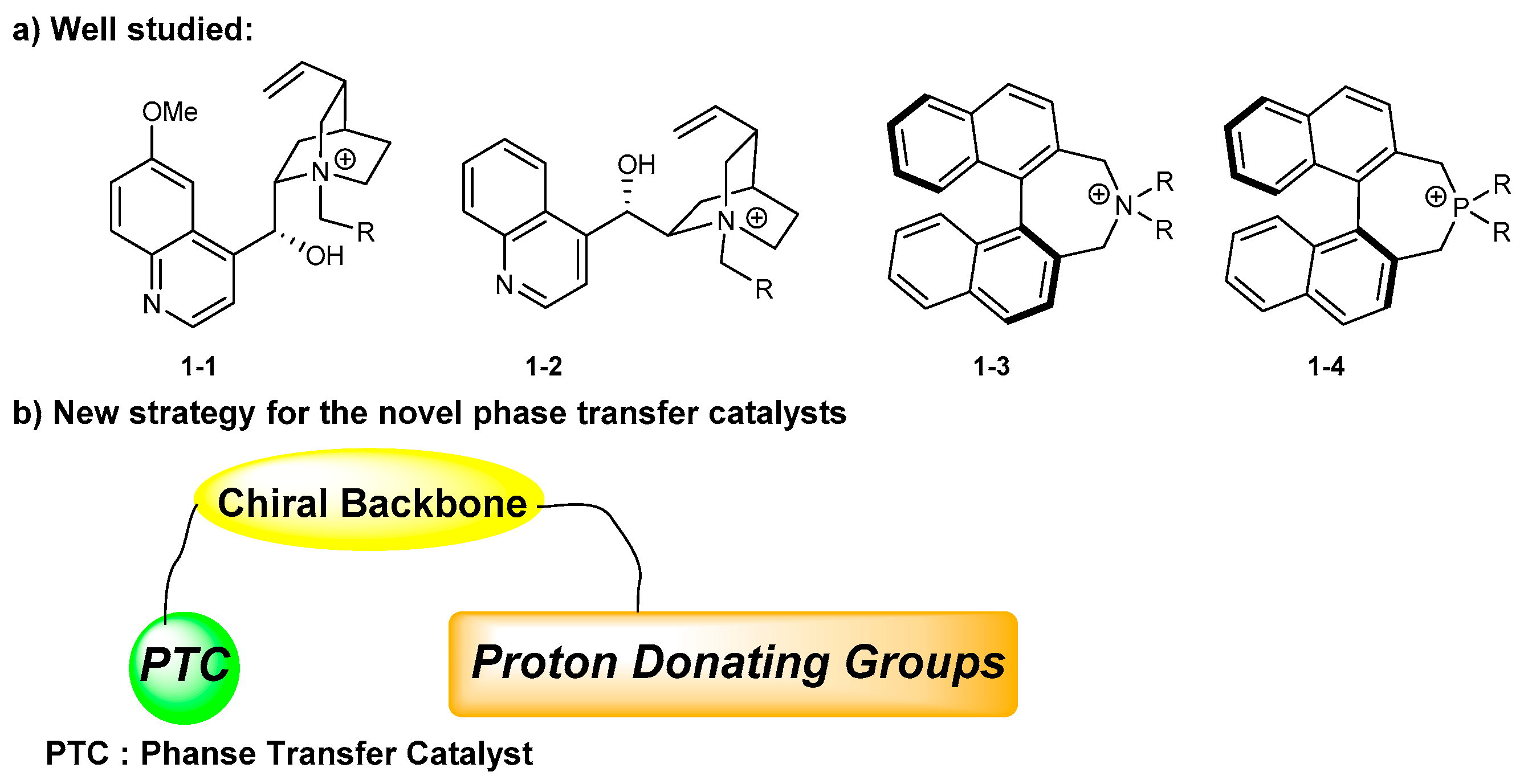
1. Introduction
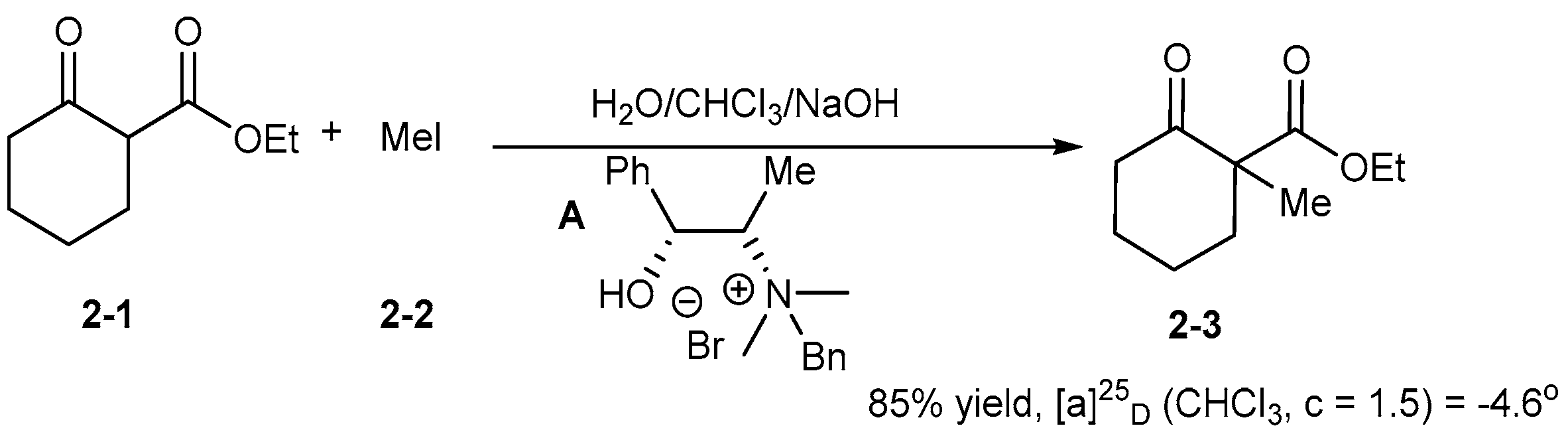
2. Early Examples
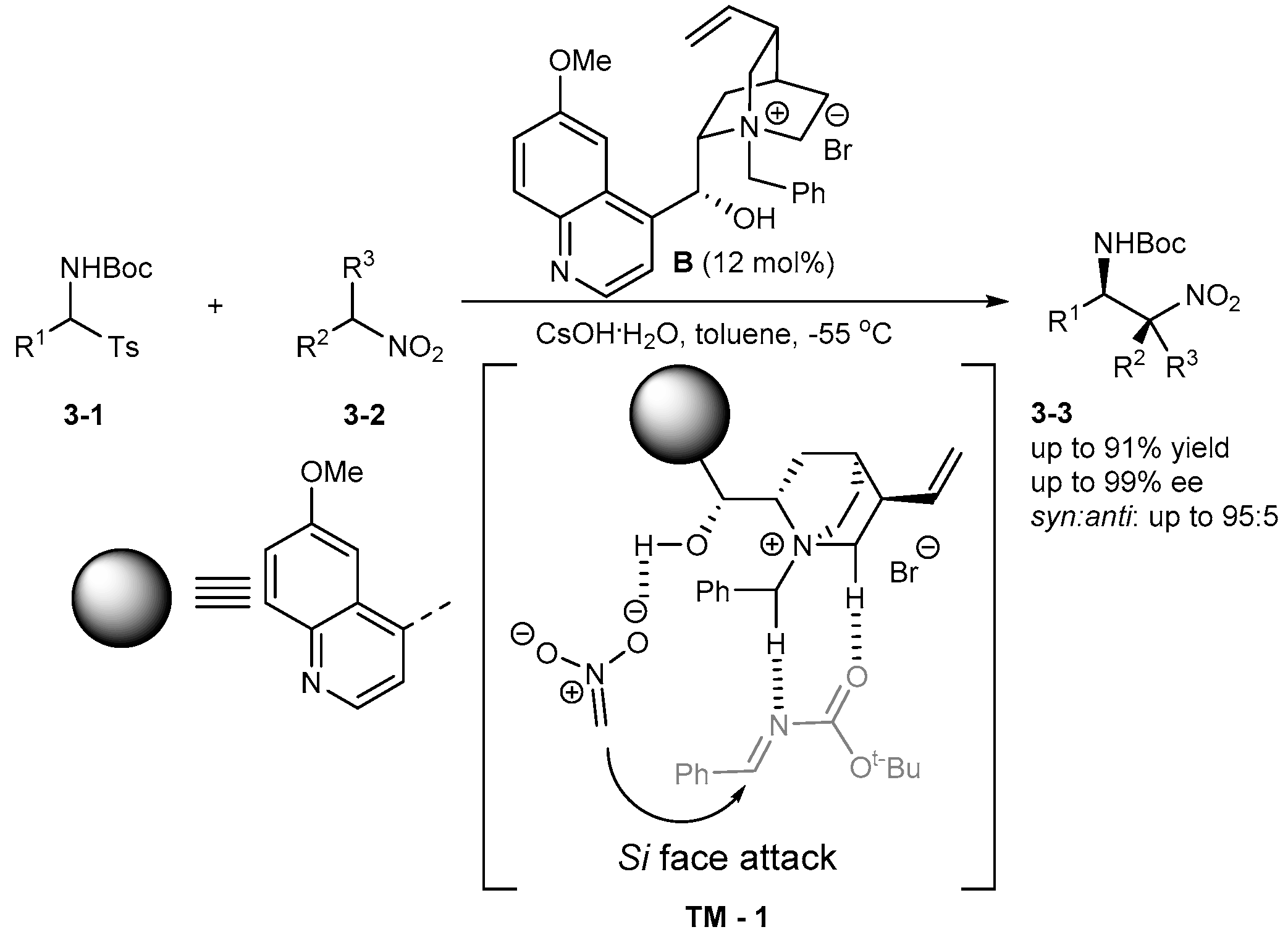
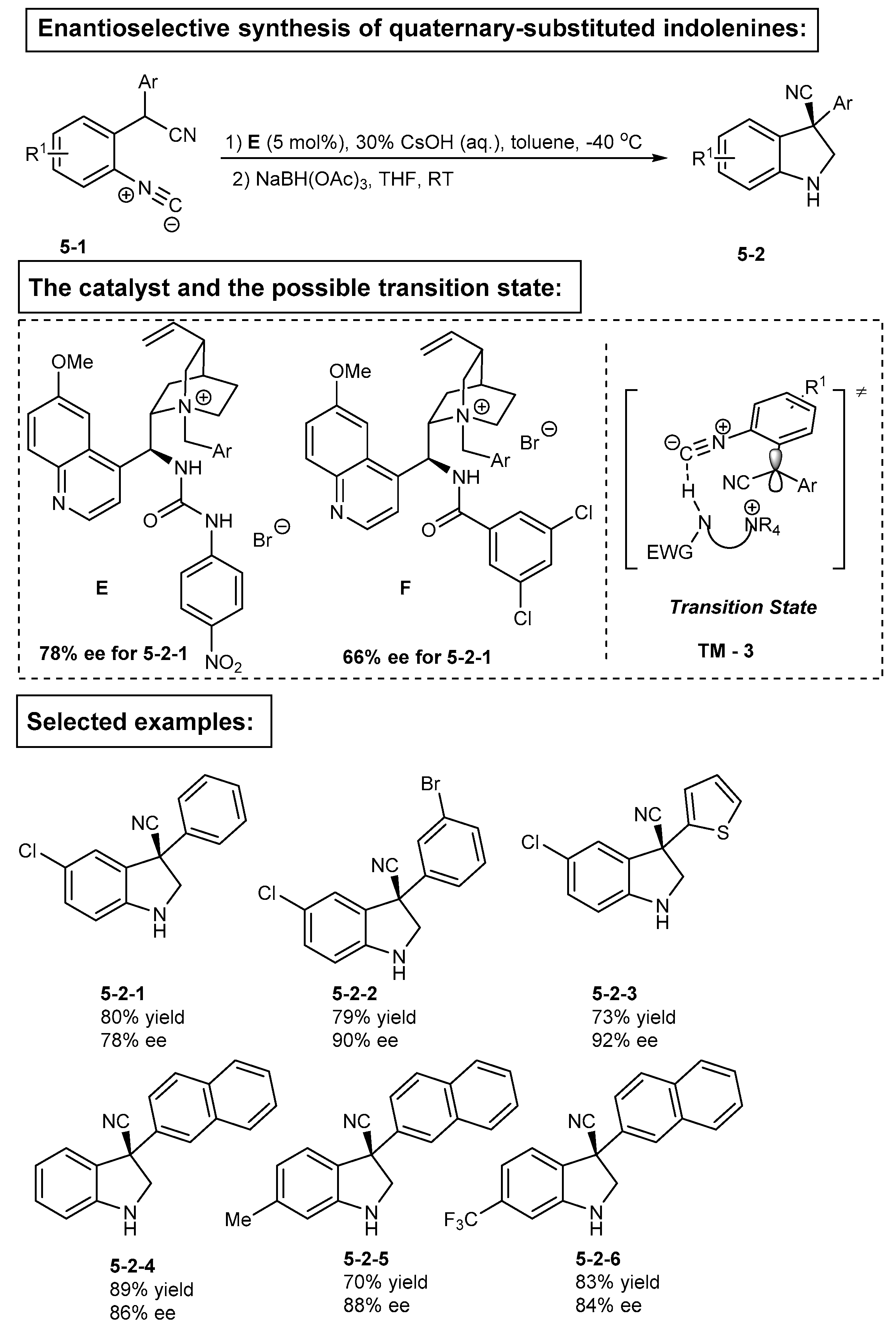
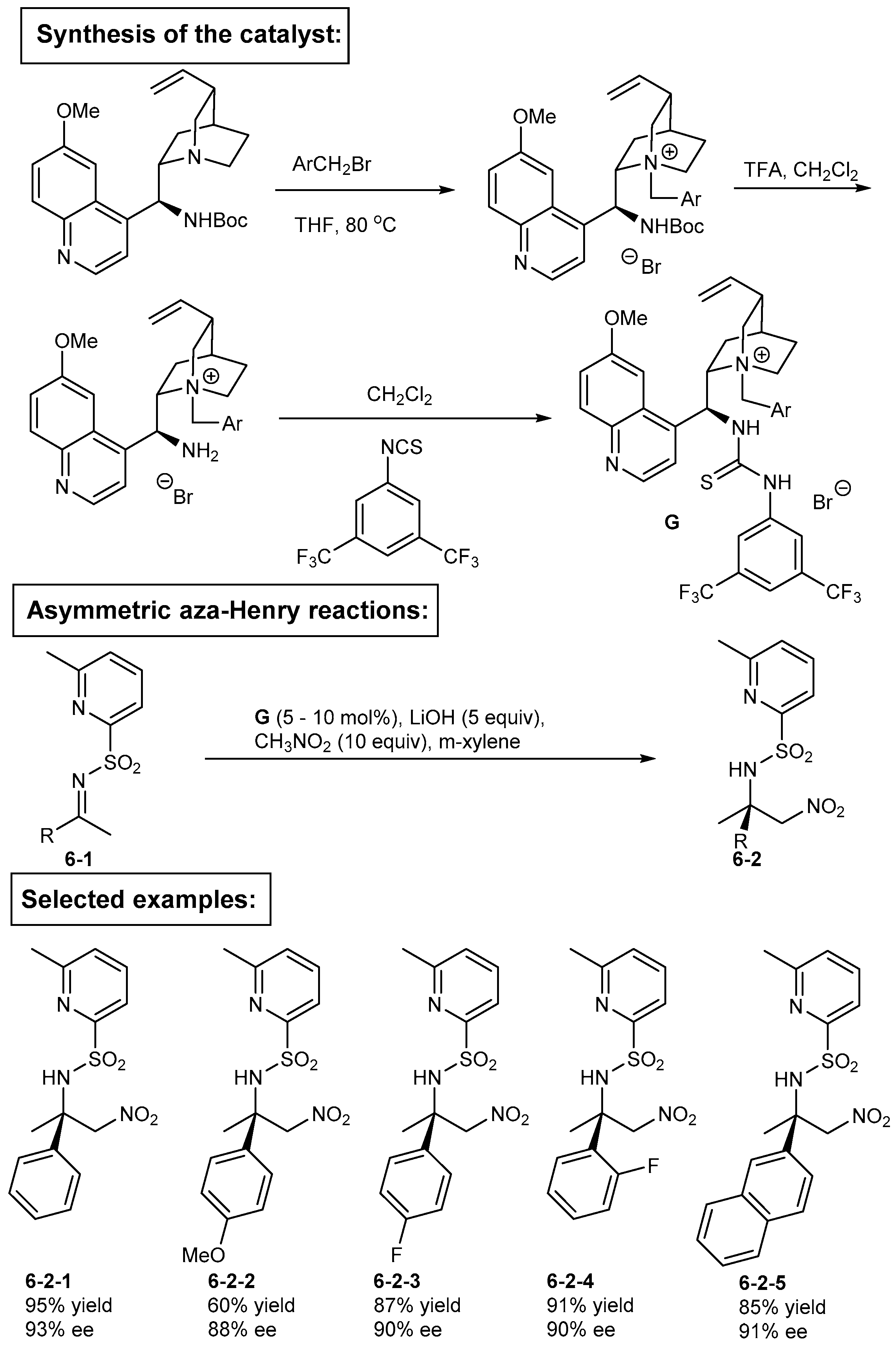
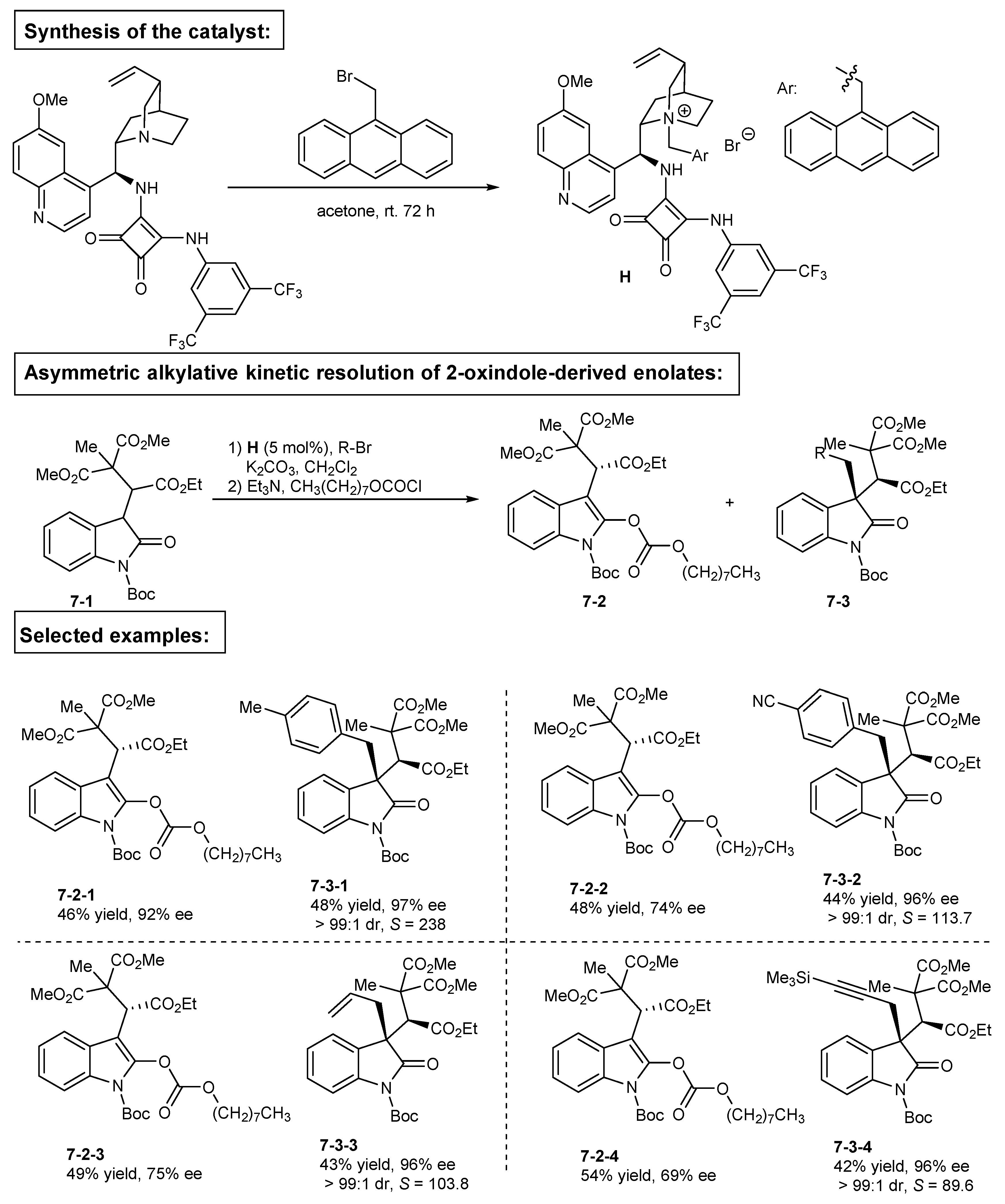
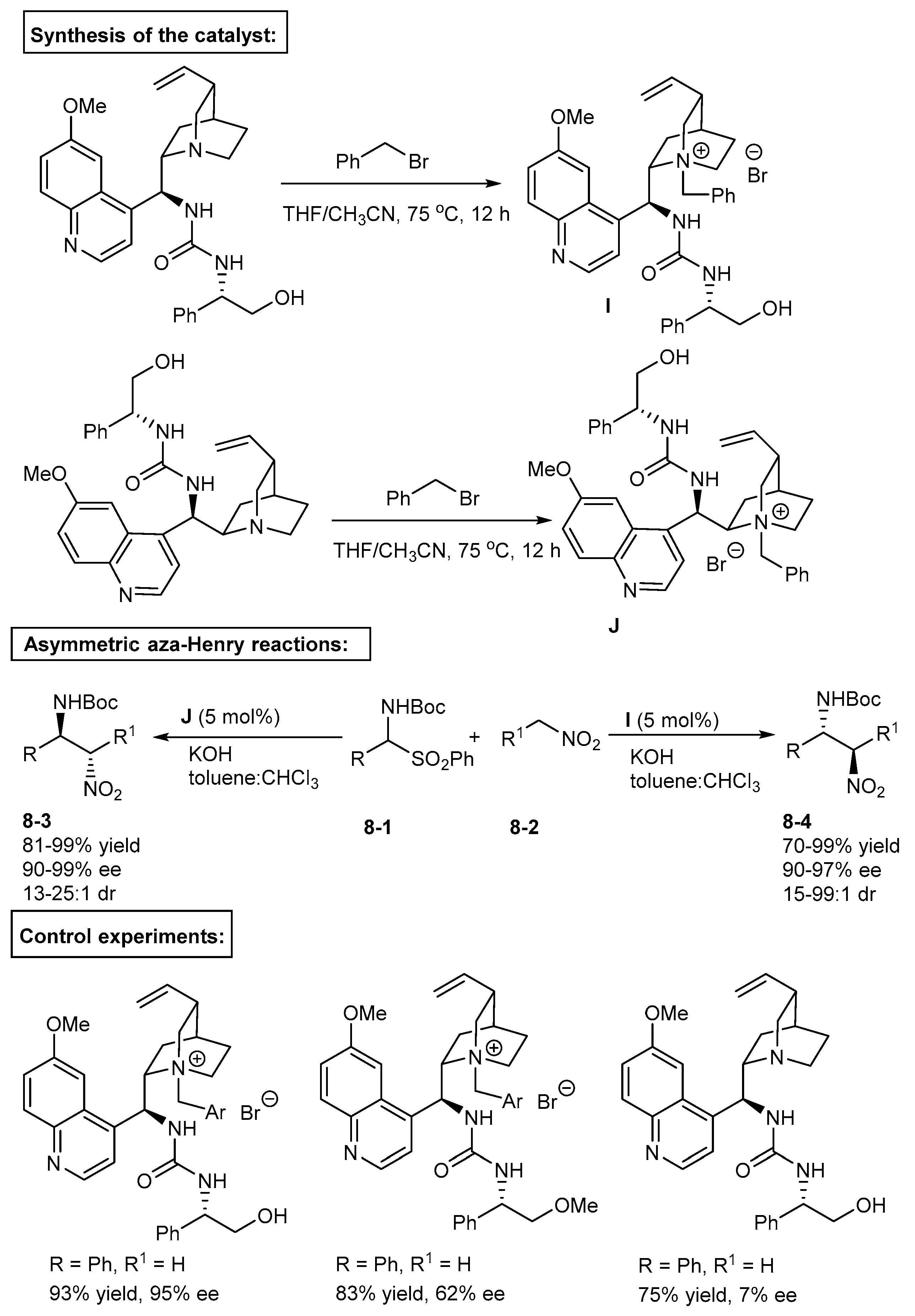
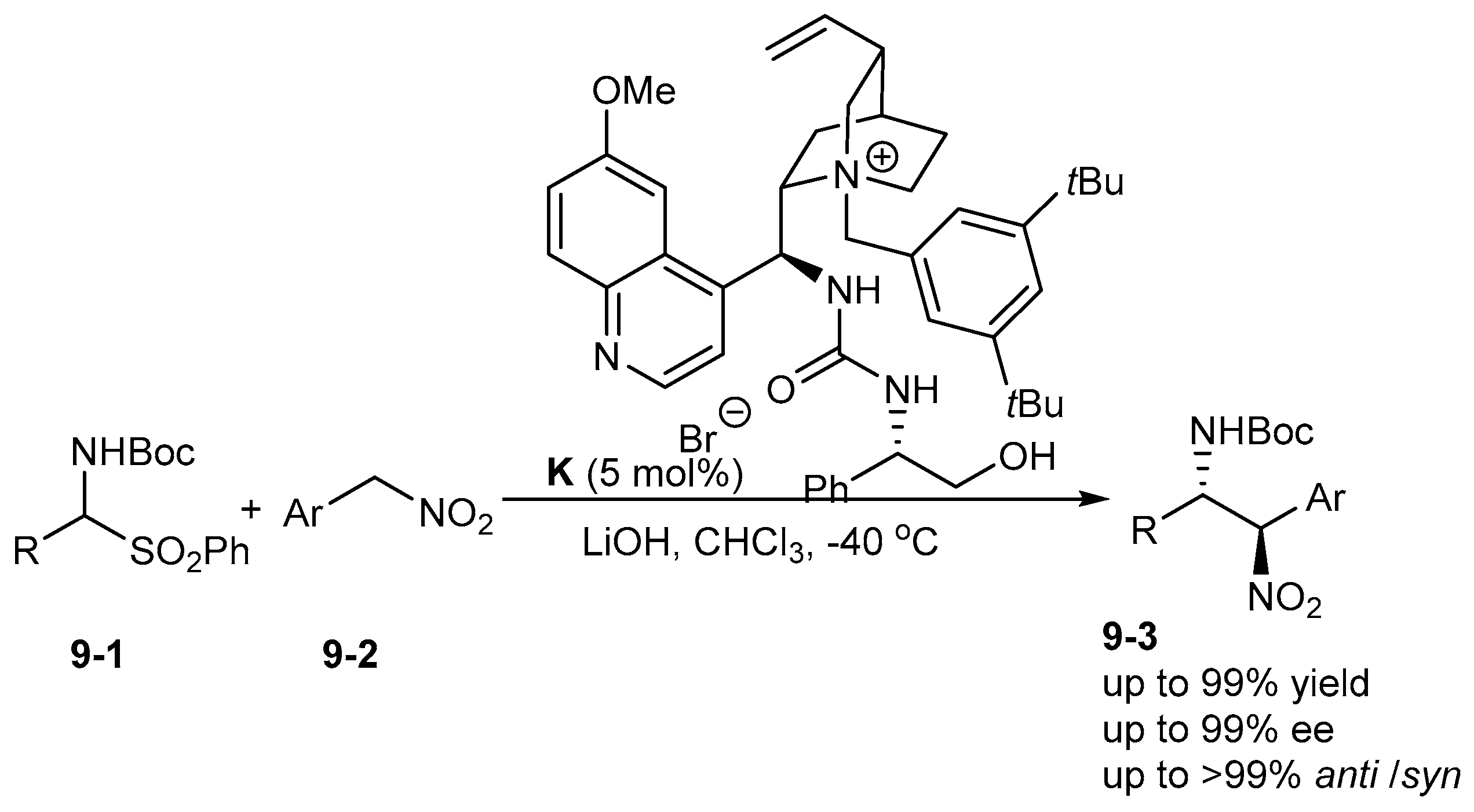
3. Bifunctional Phase-Transfer catalysts derived from Cinchona alkaloids
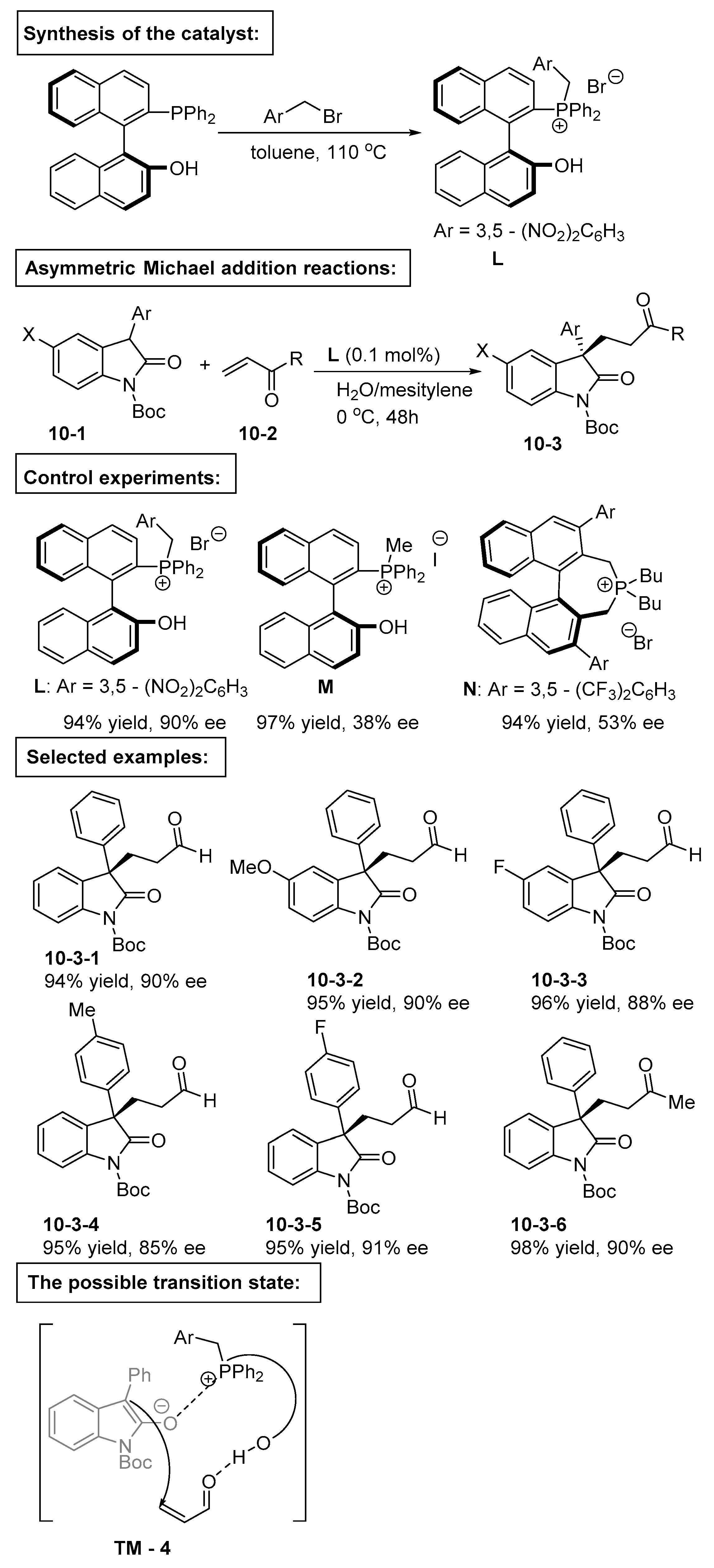
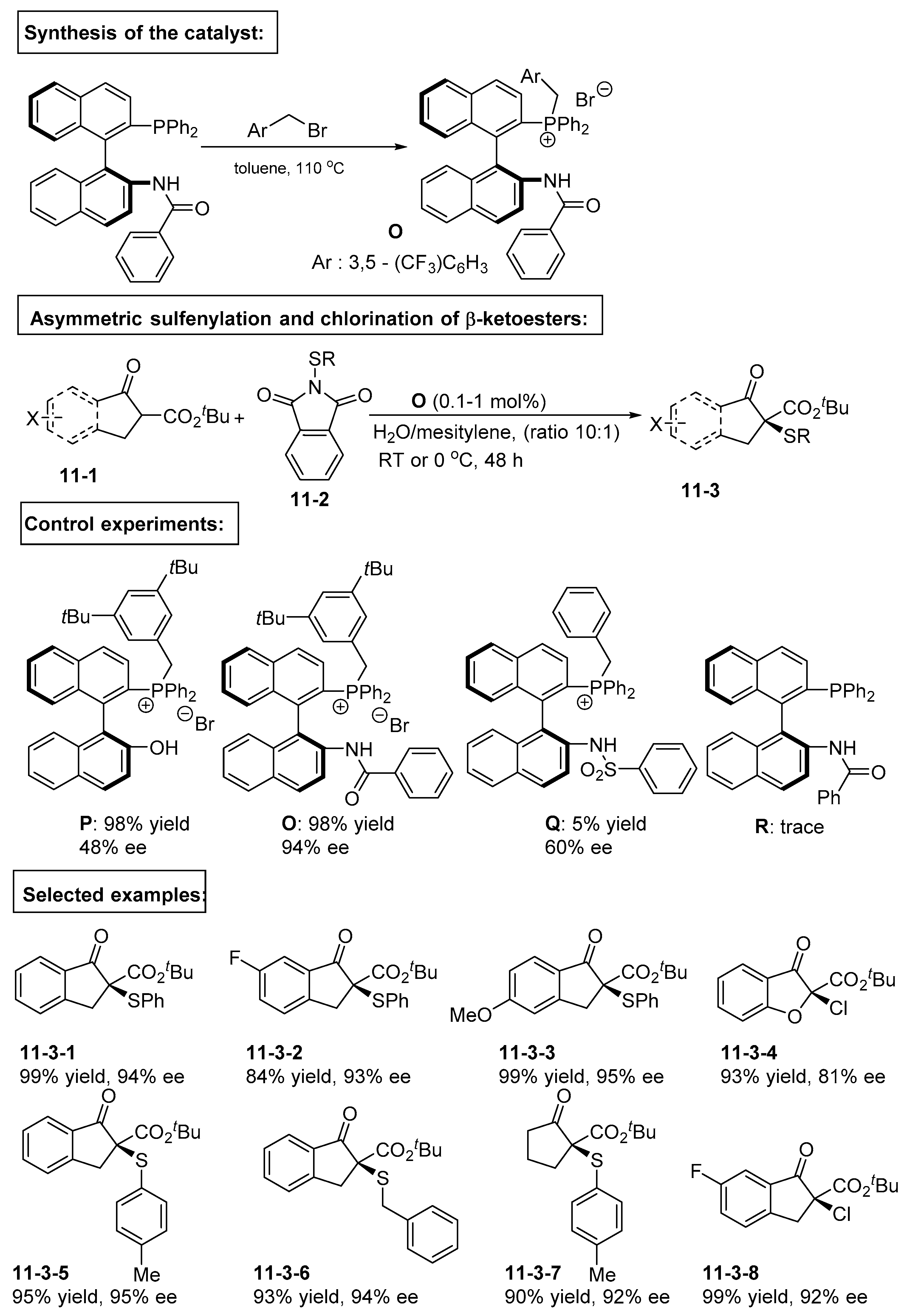
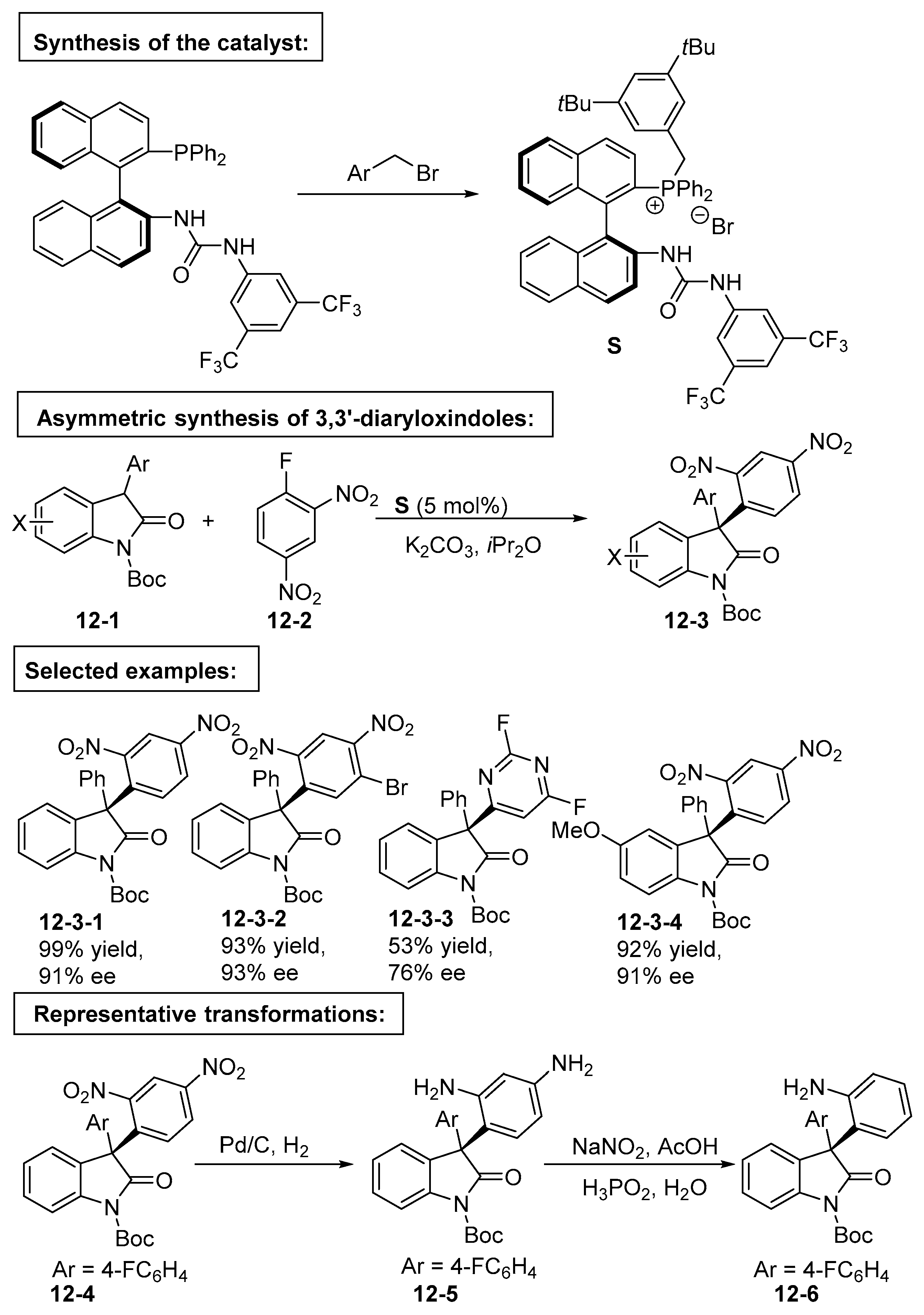
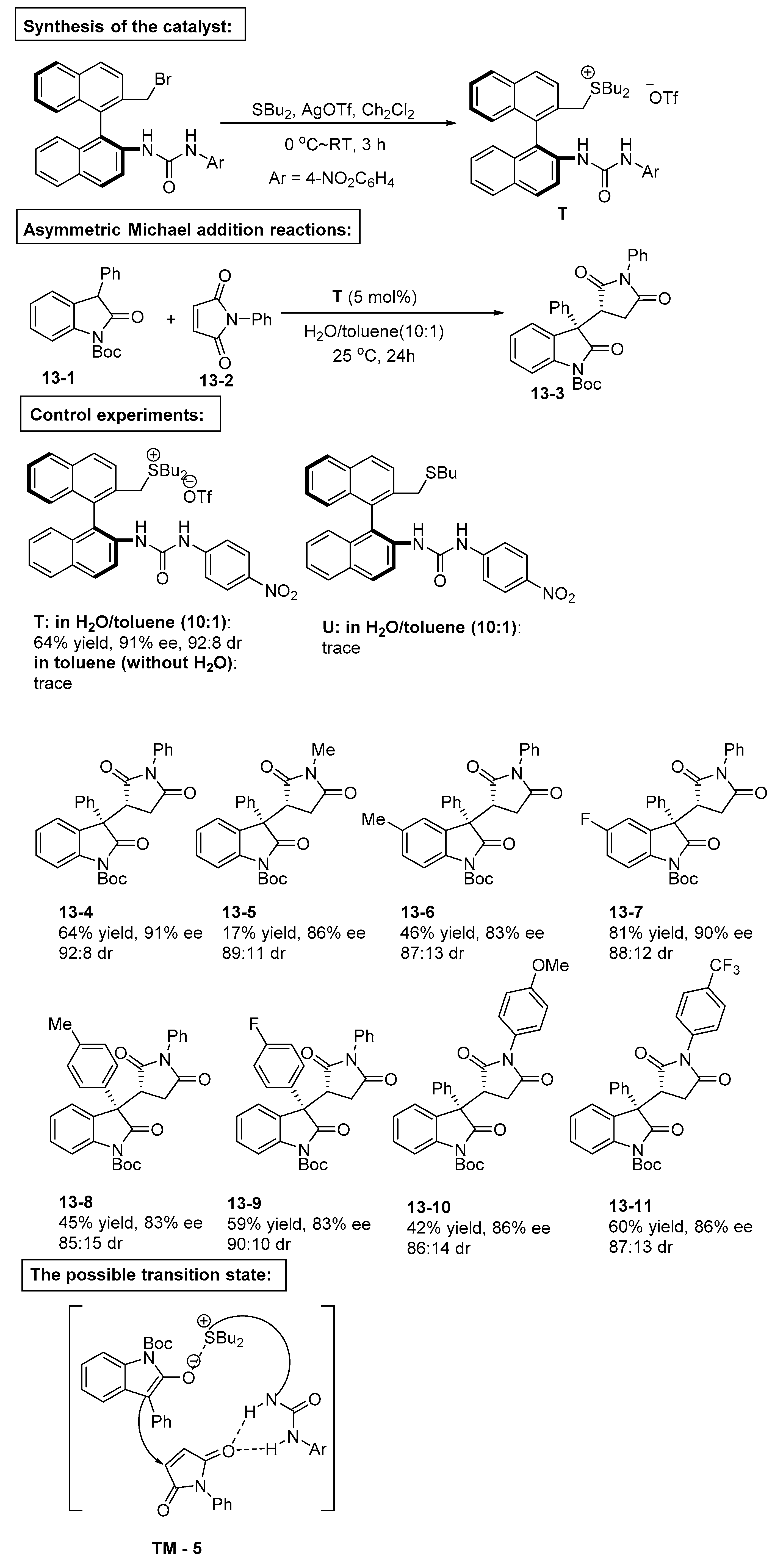
4. Bifunctional Phase-Transfer catalysts derived from Binaphthyl
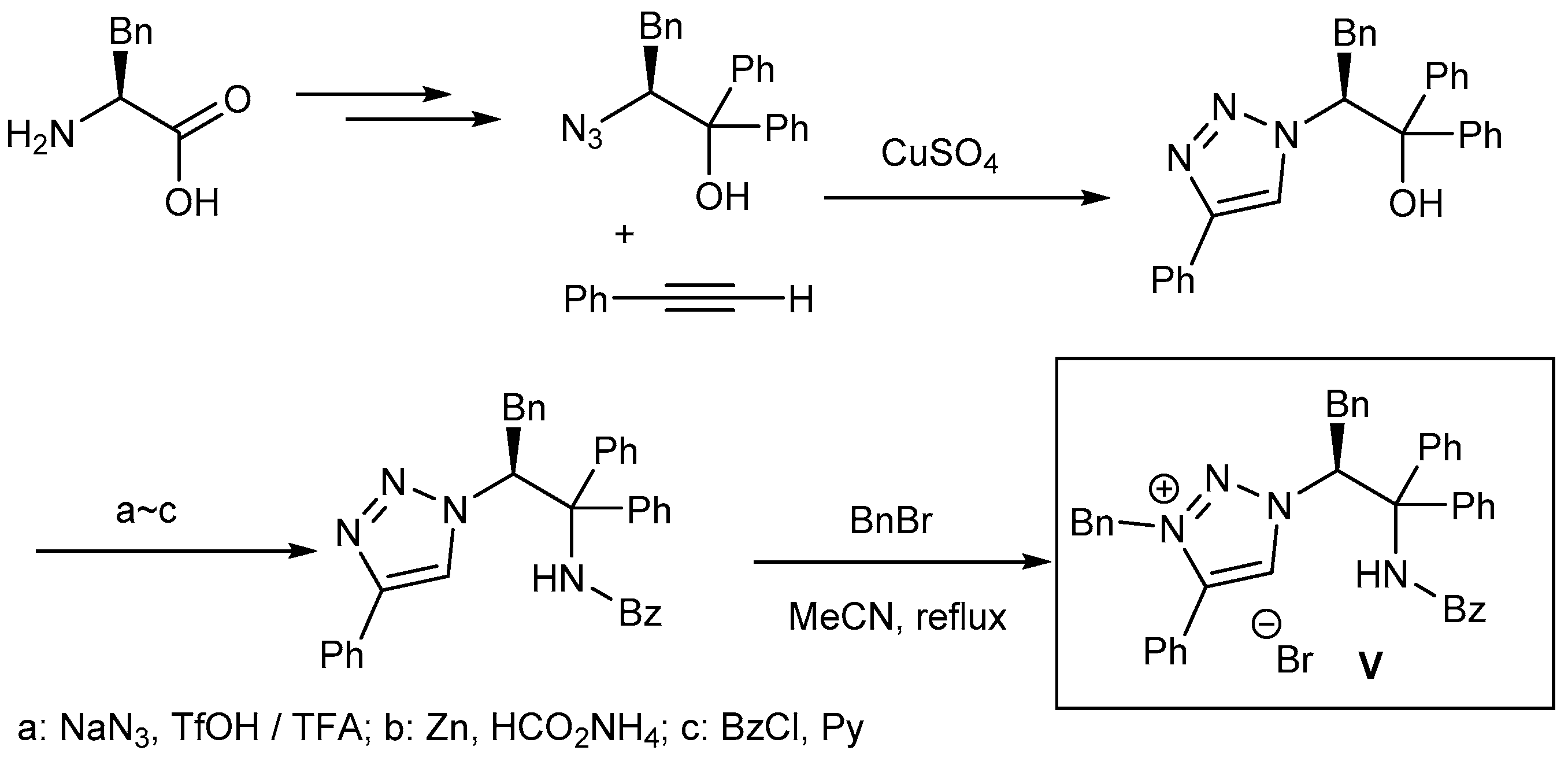
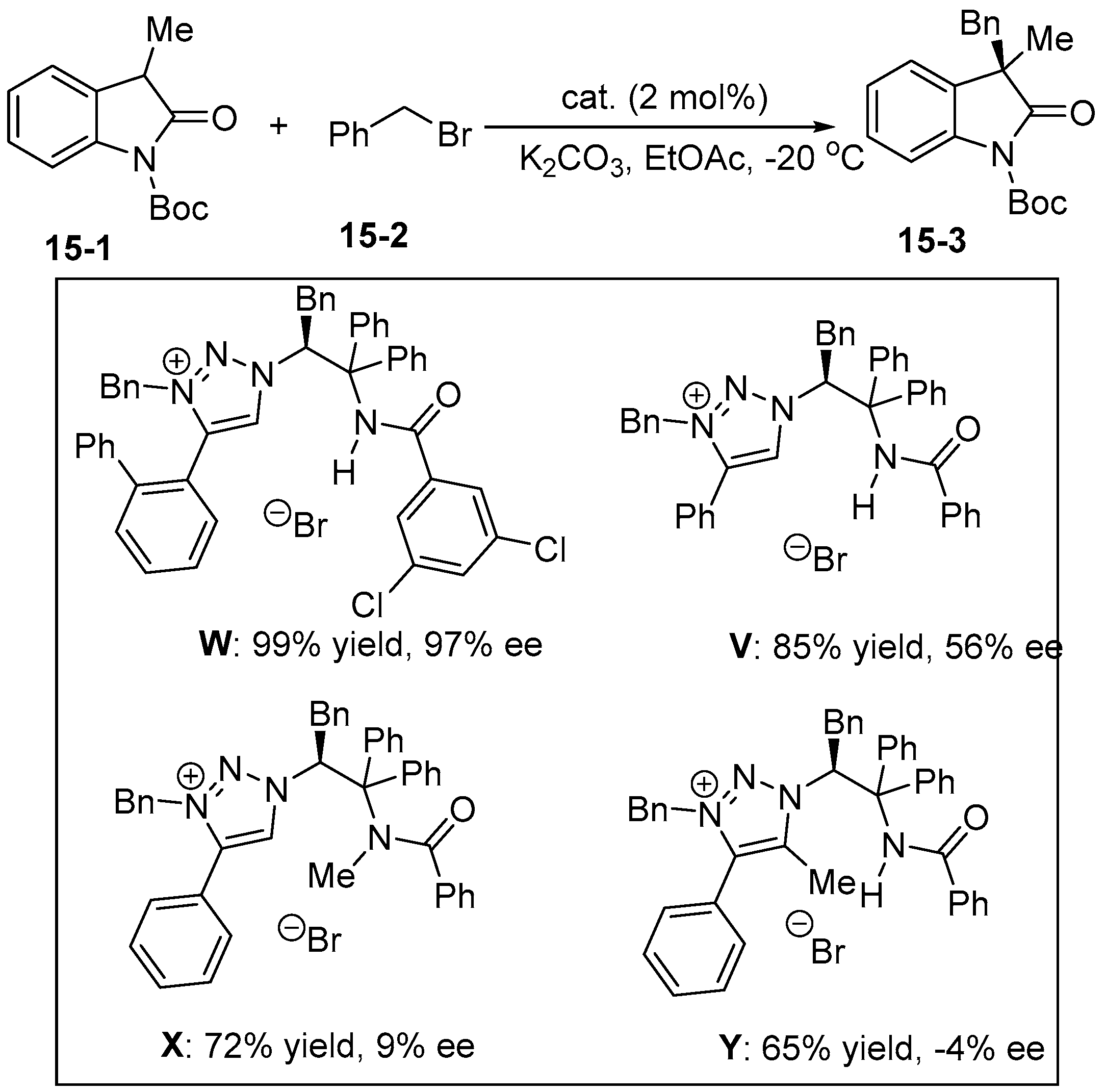
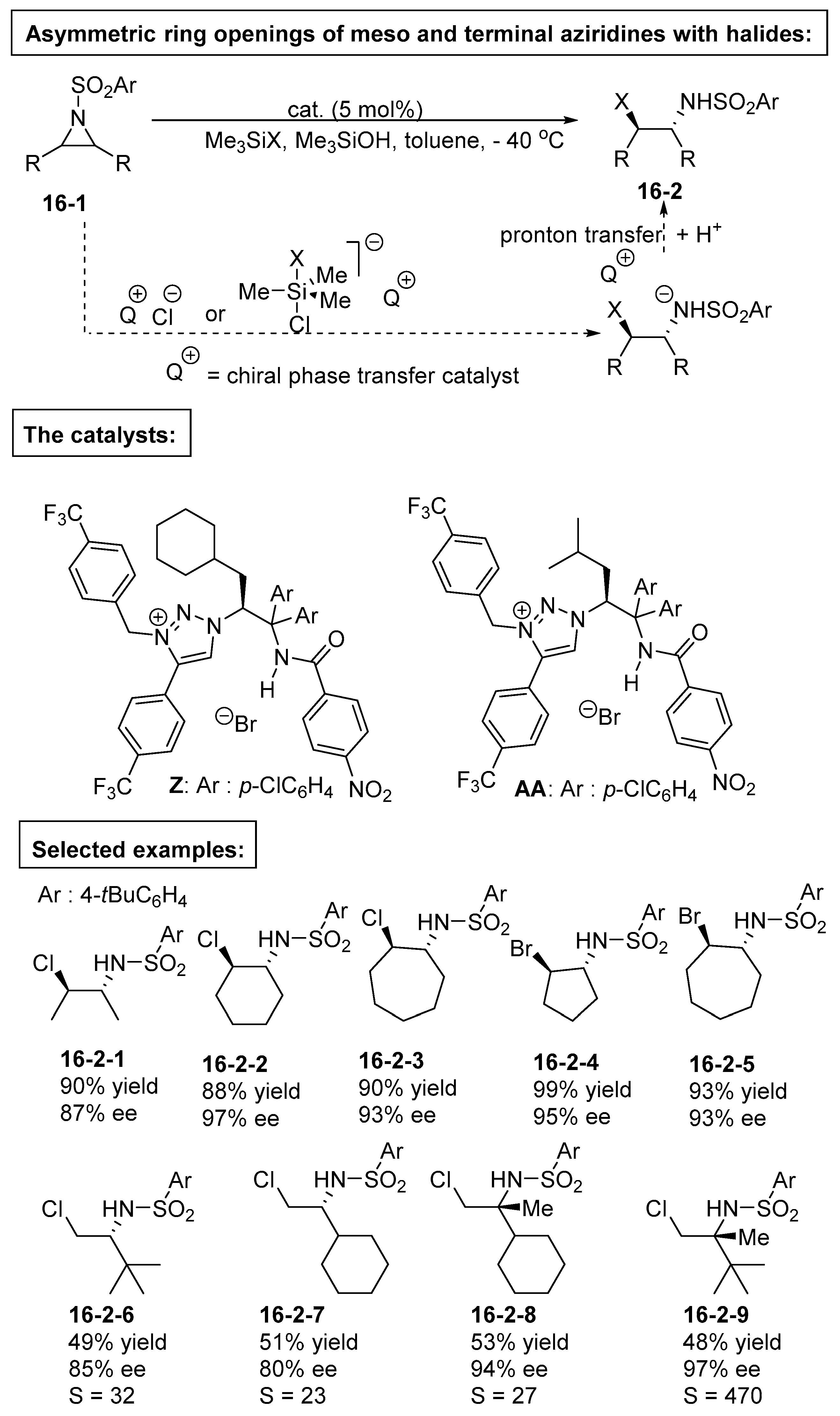
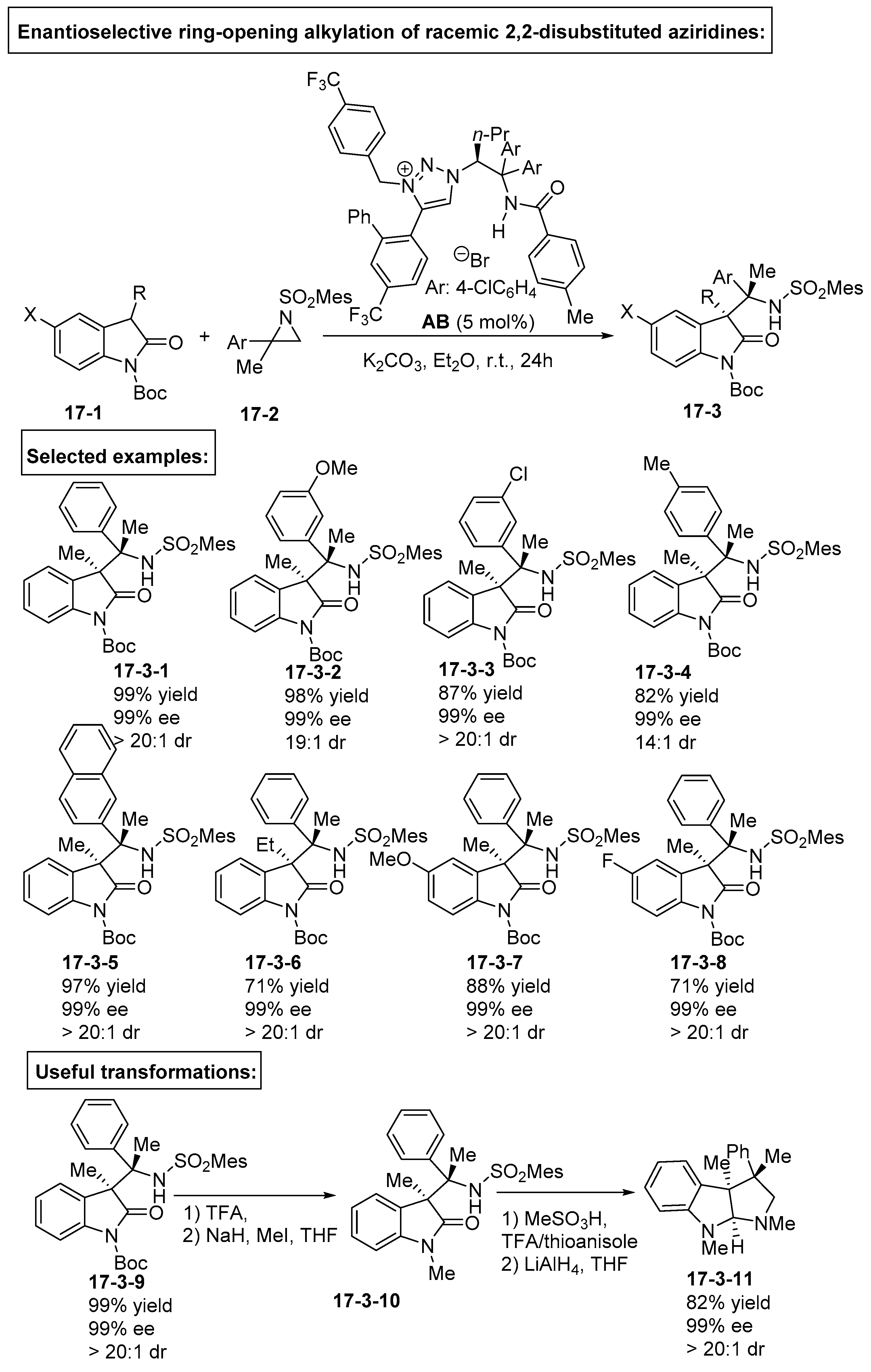
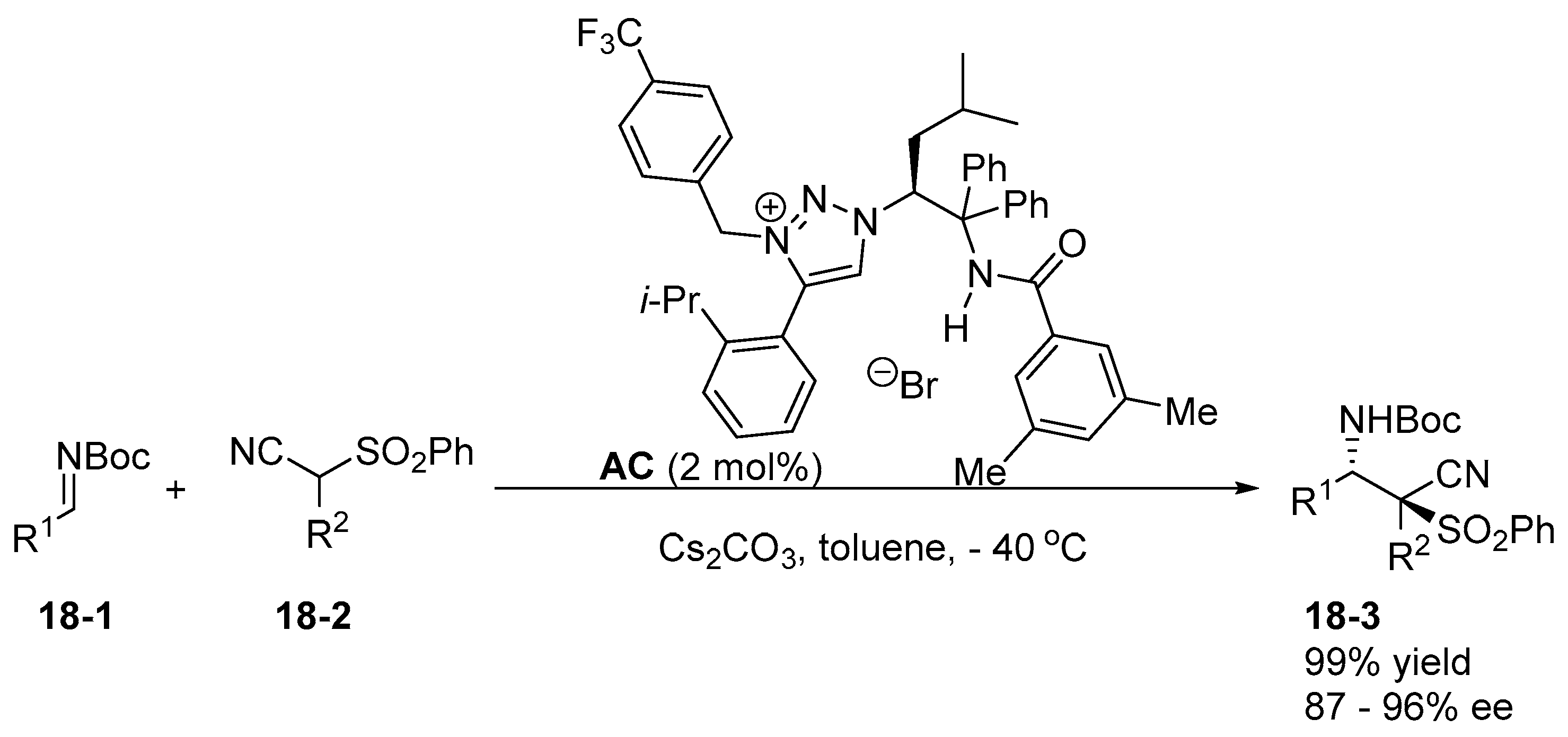
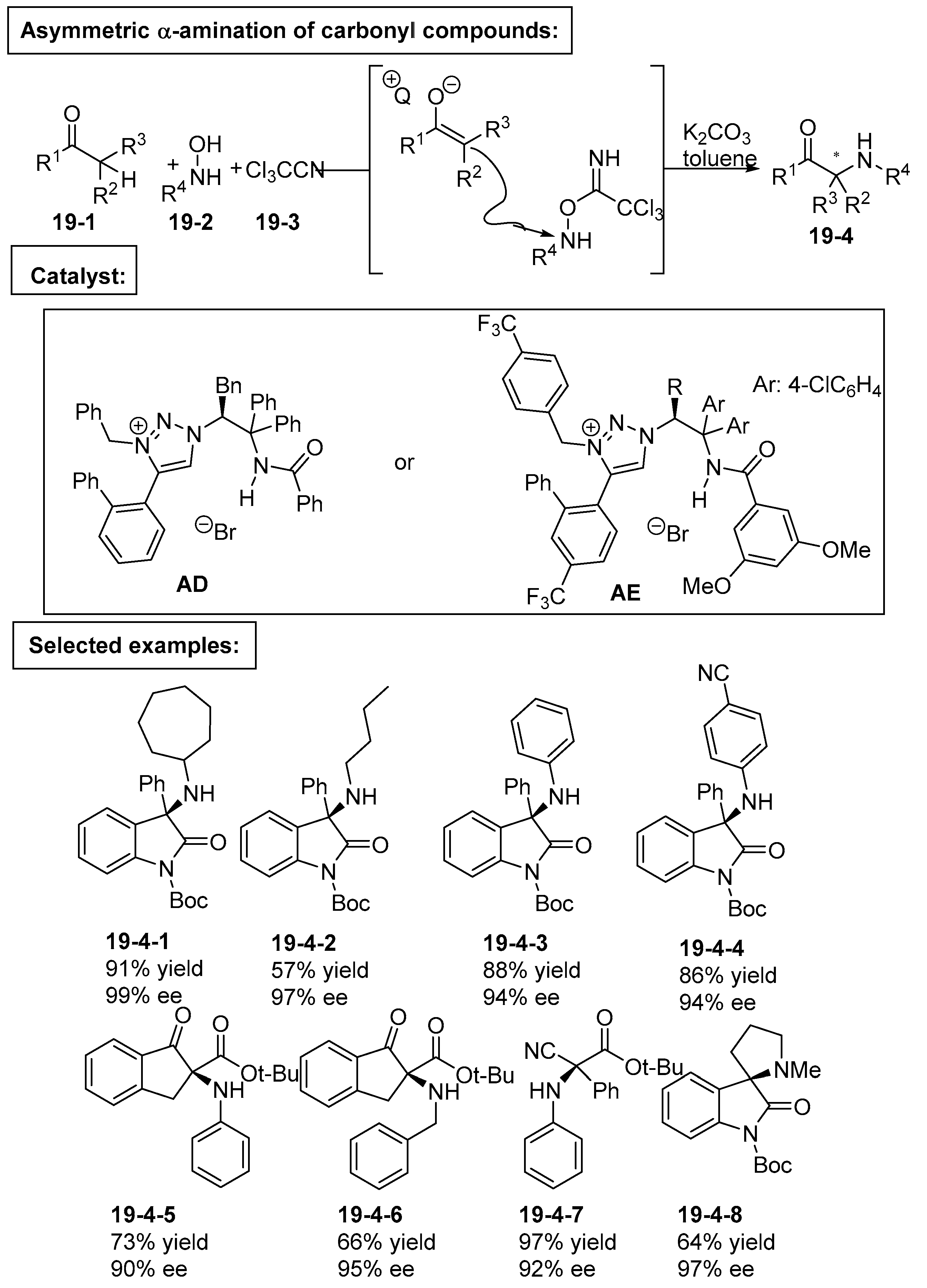
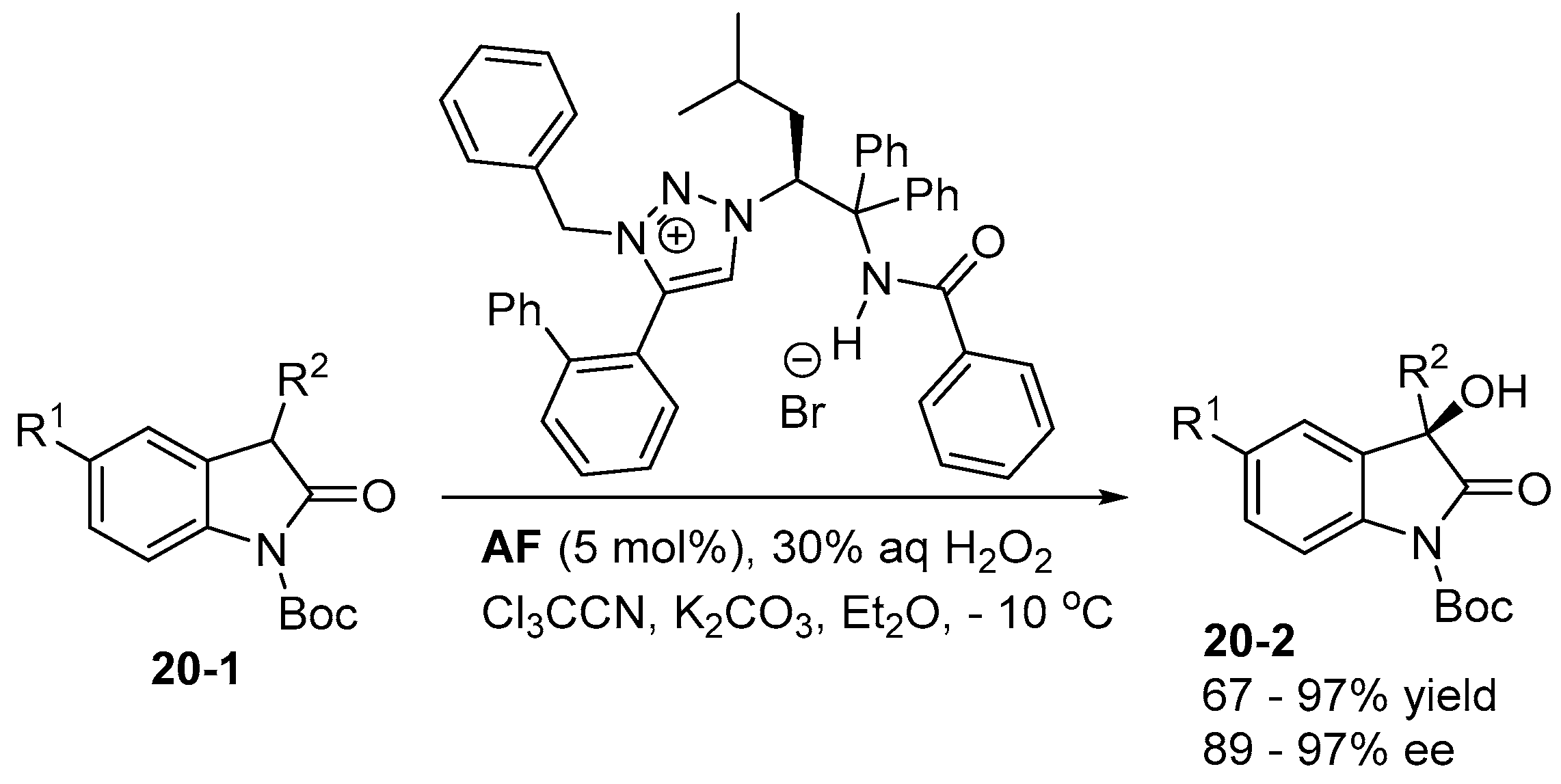
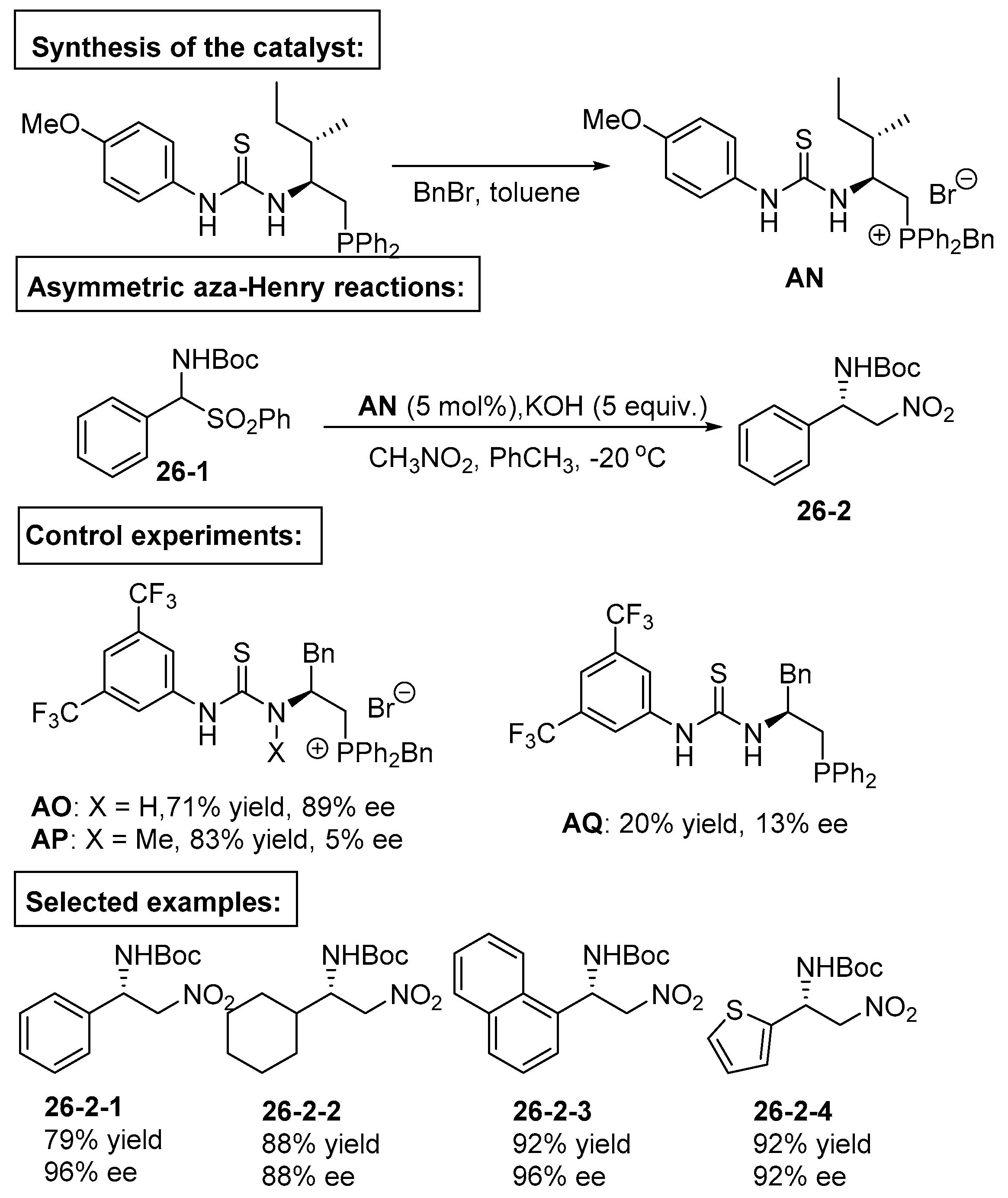
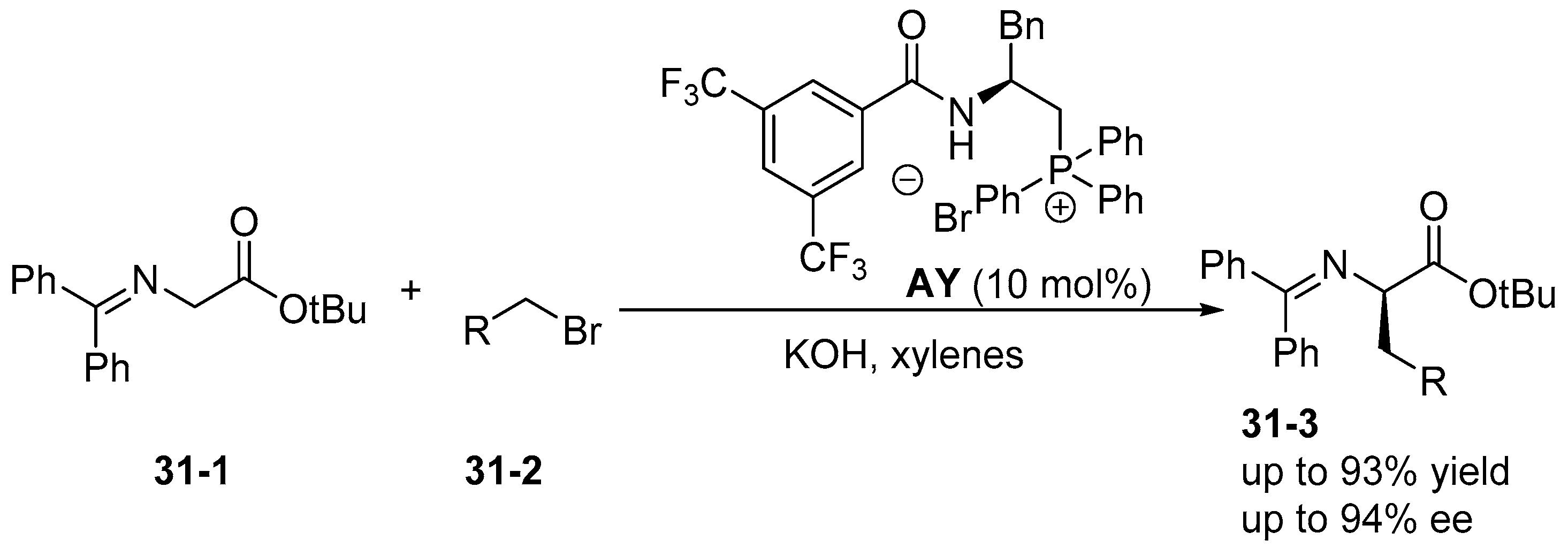
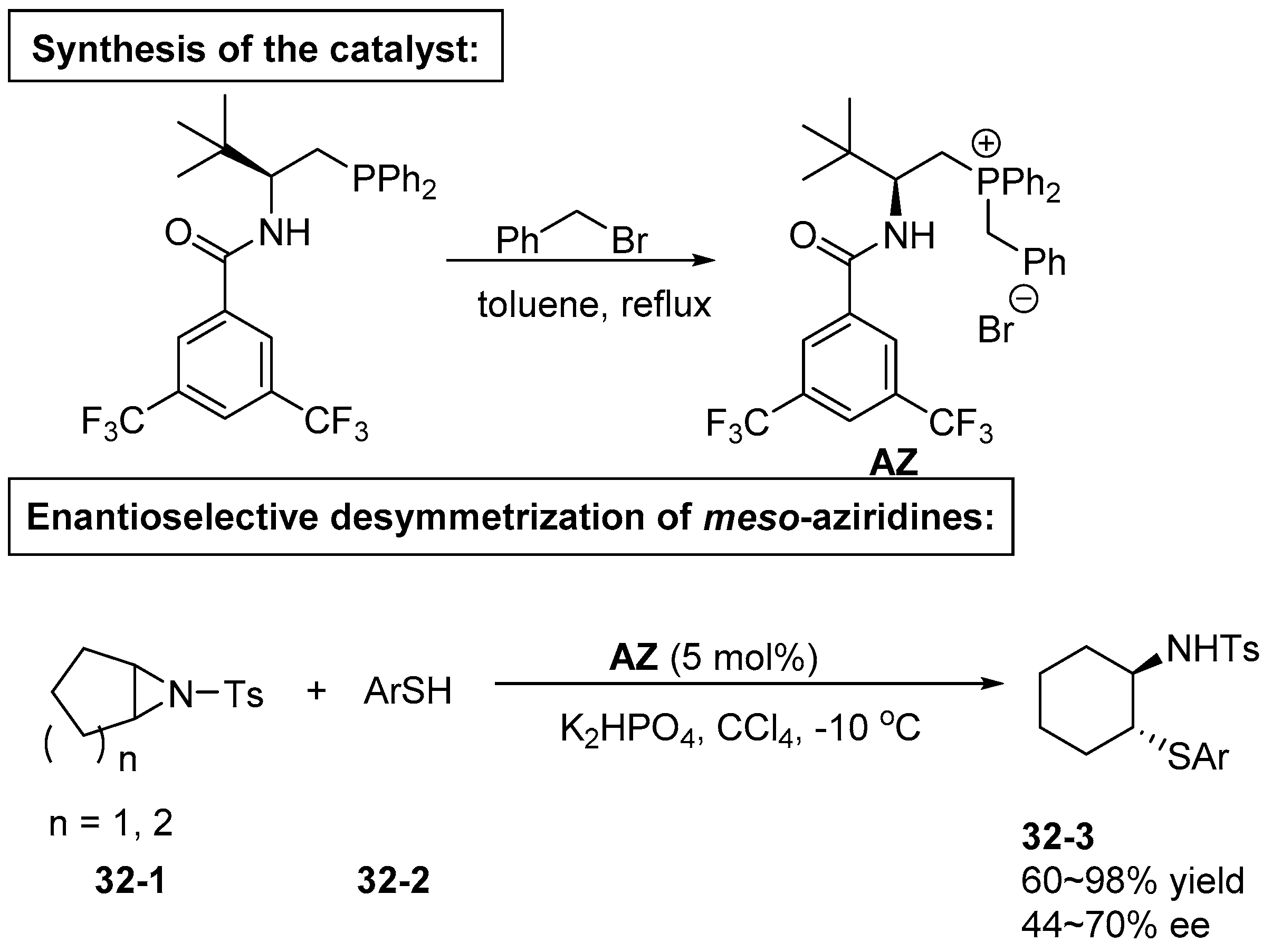
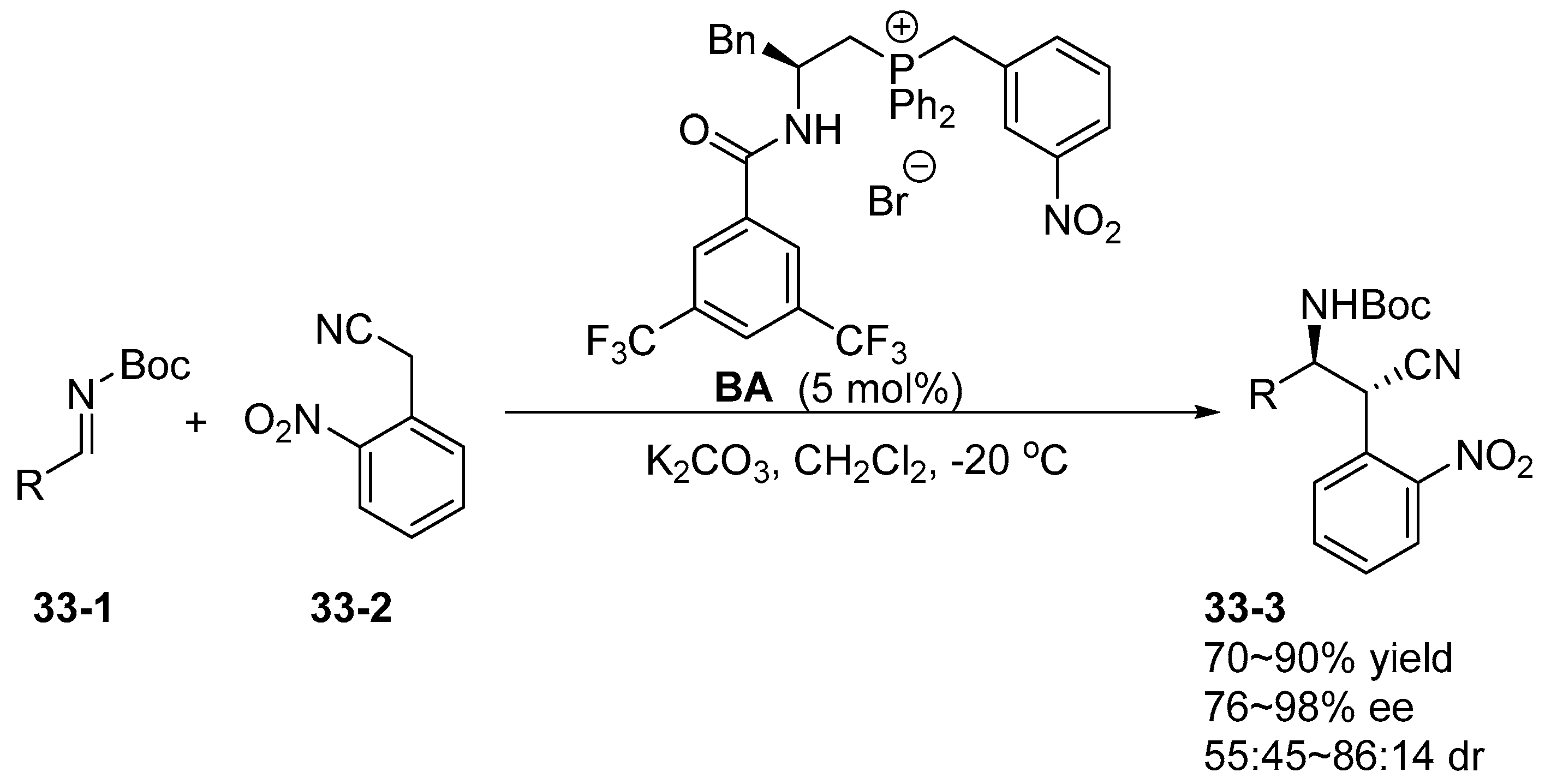
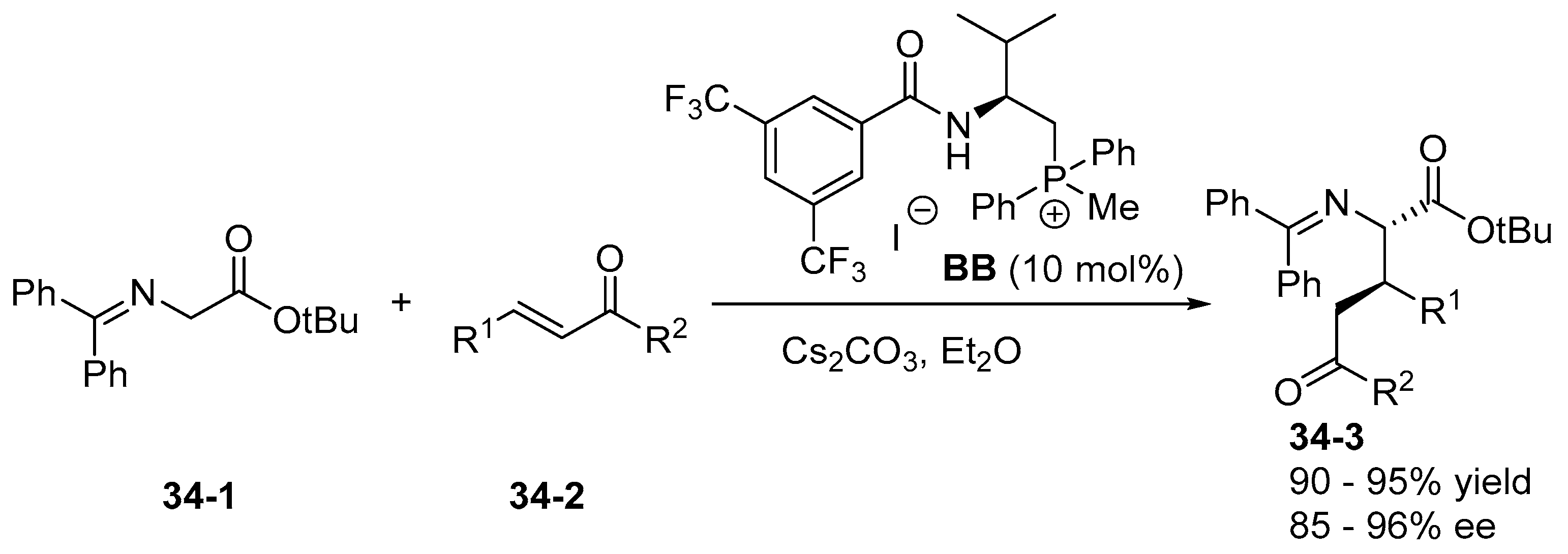
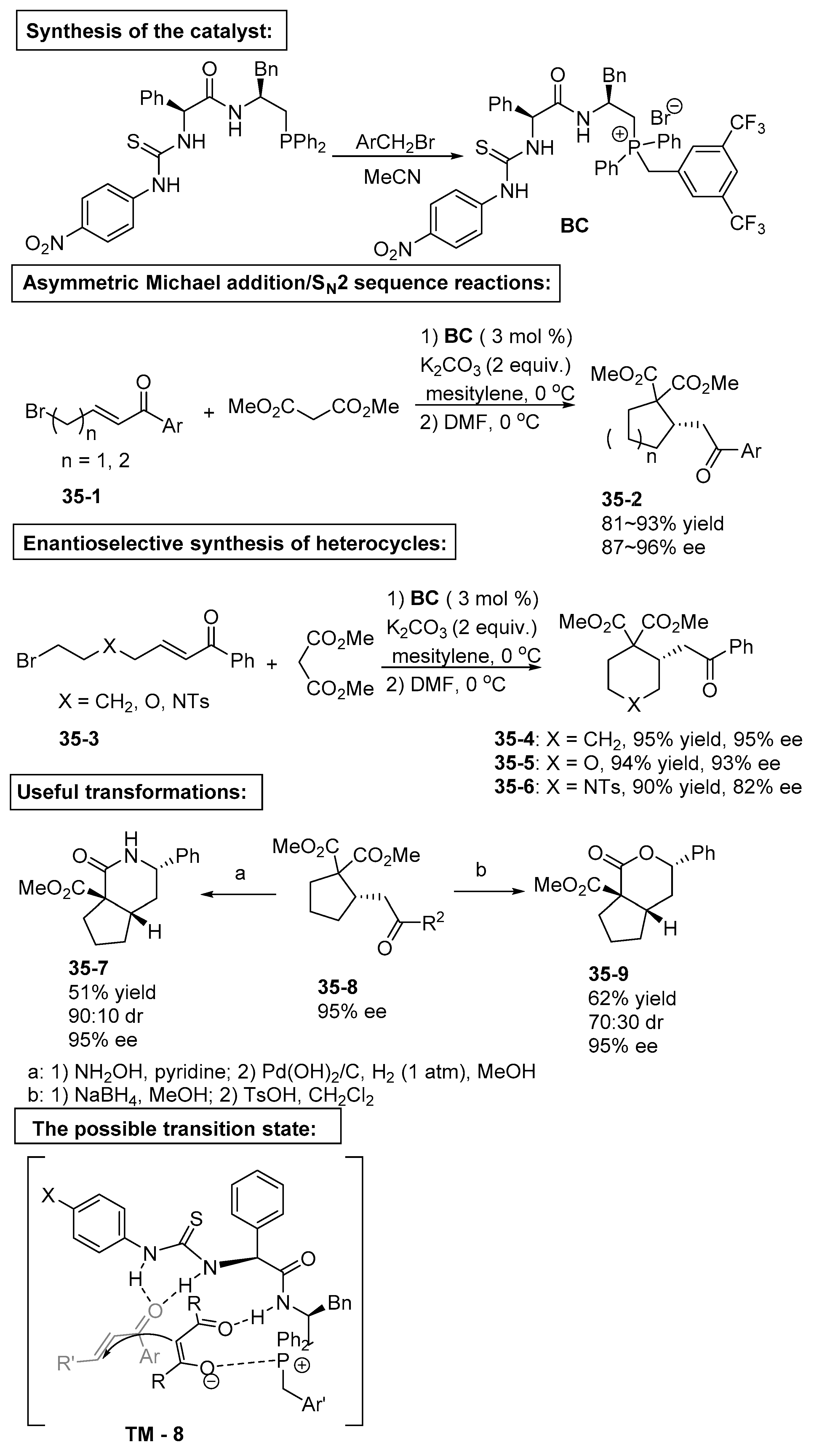
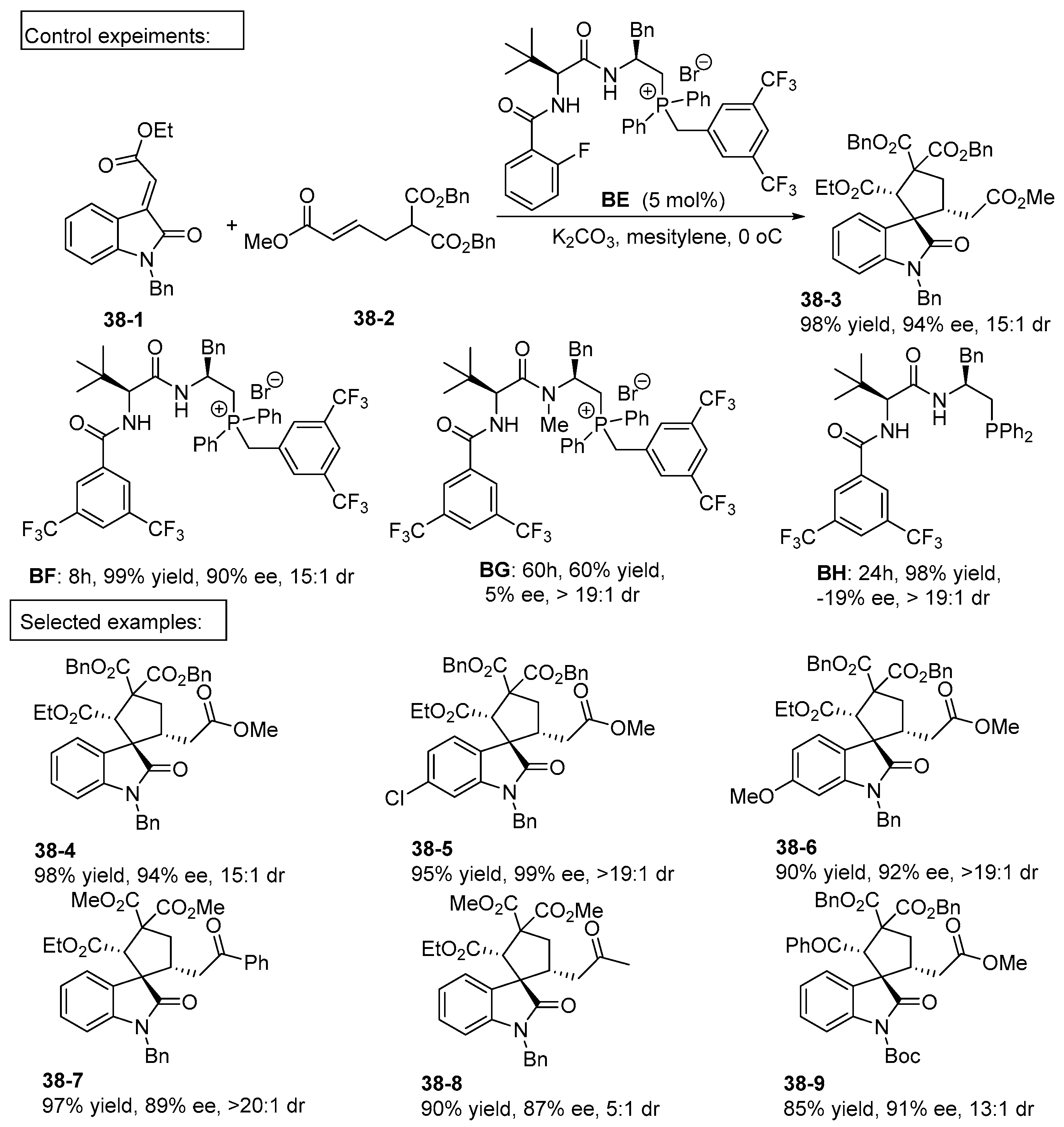
5. Bifunctional Phase-Transfer catalysts derived from Amino Acid
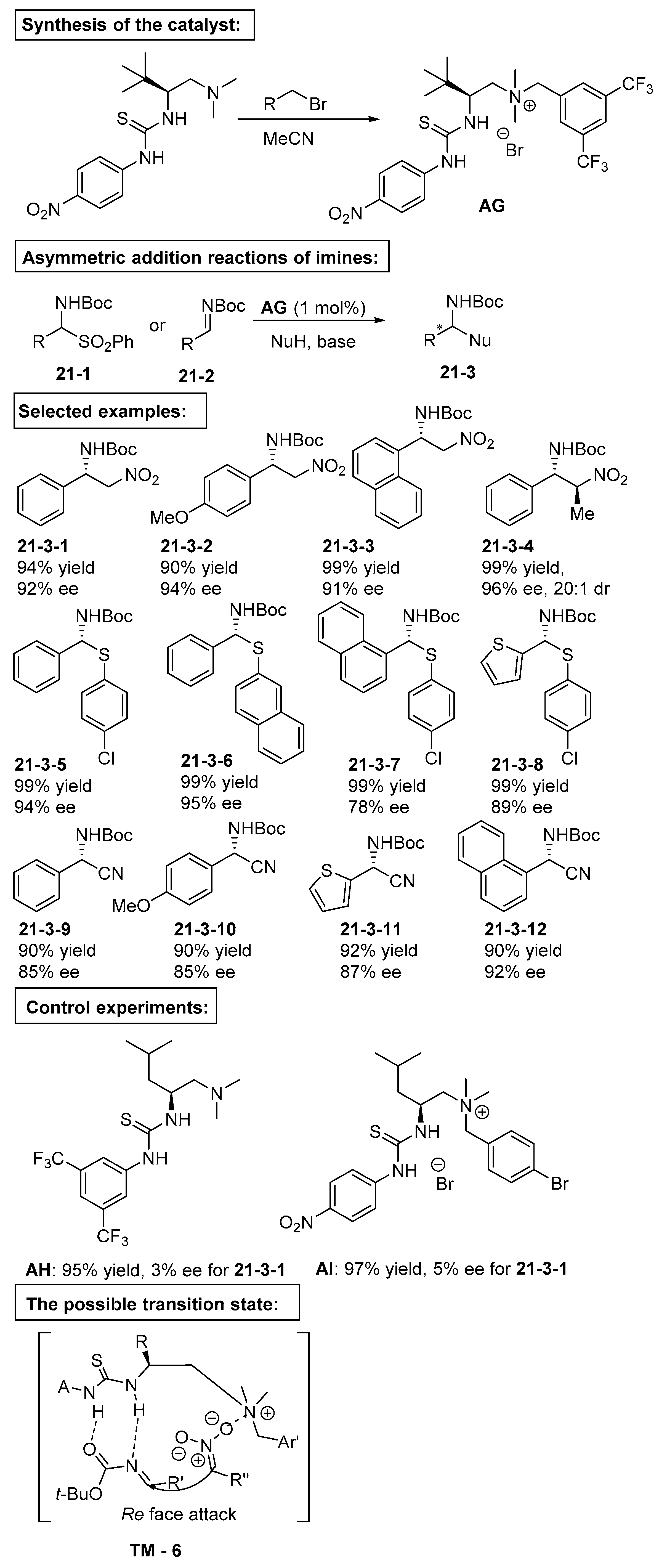
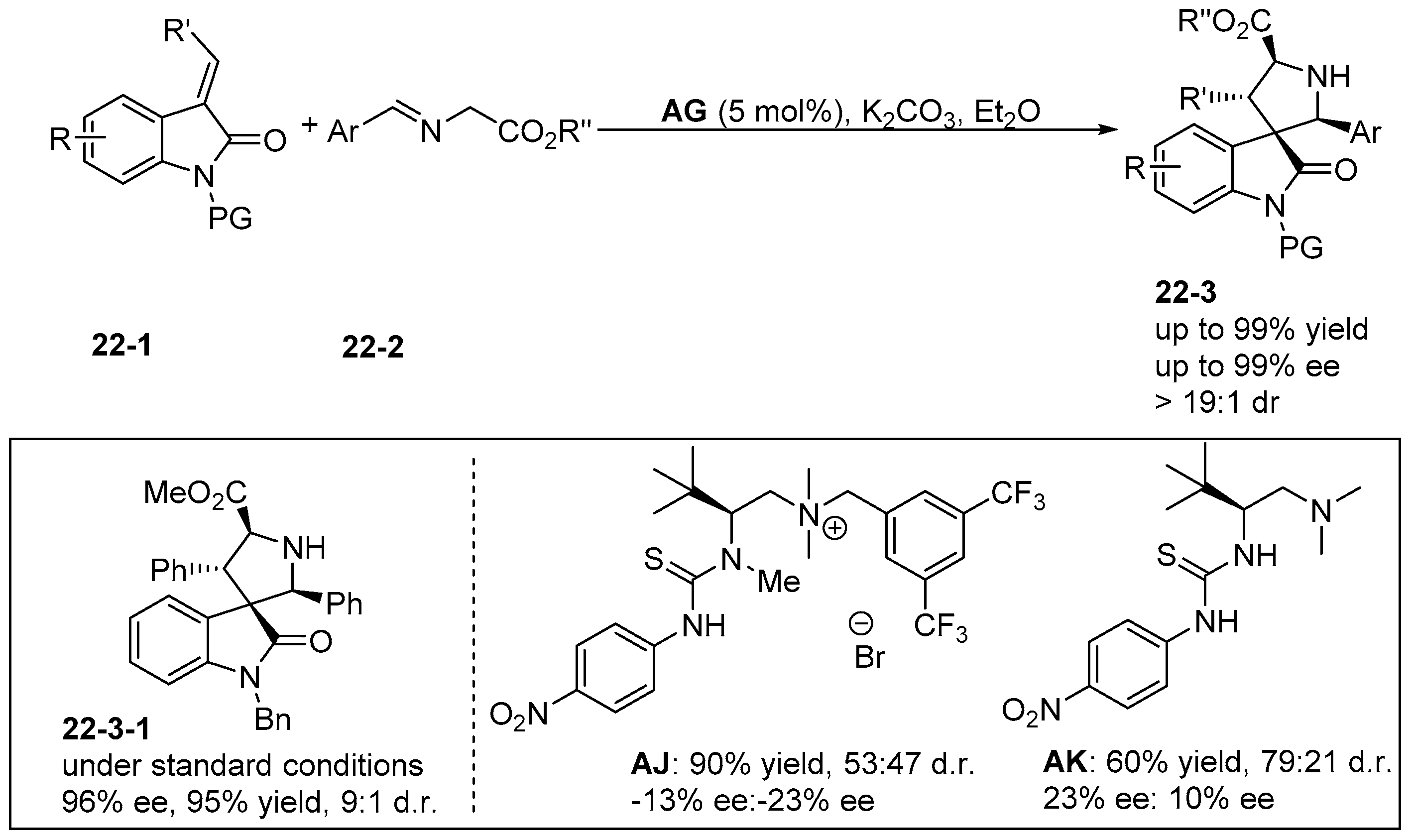
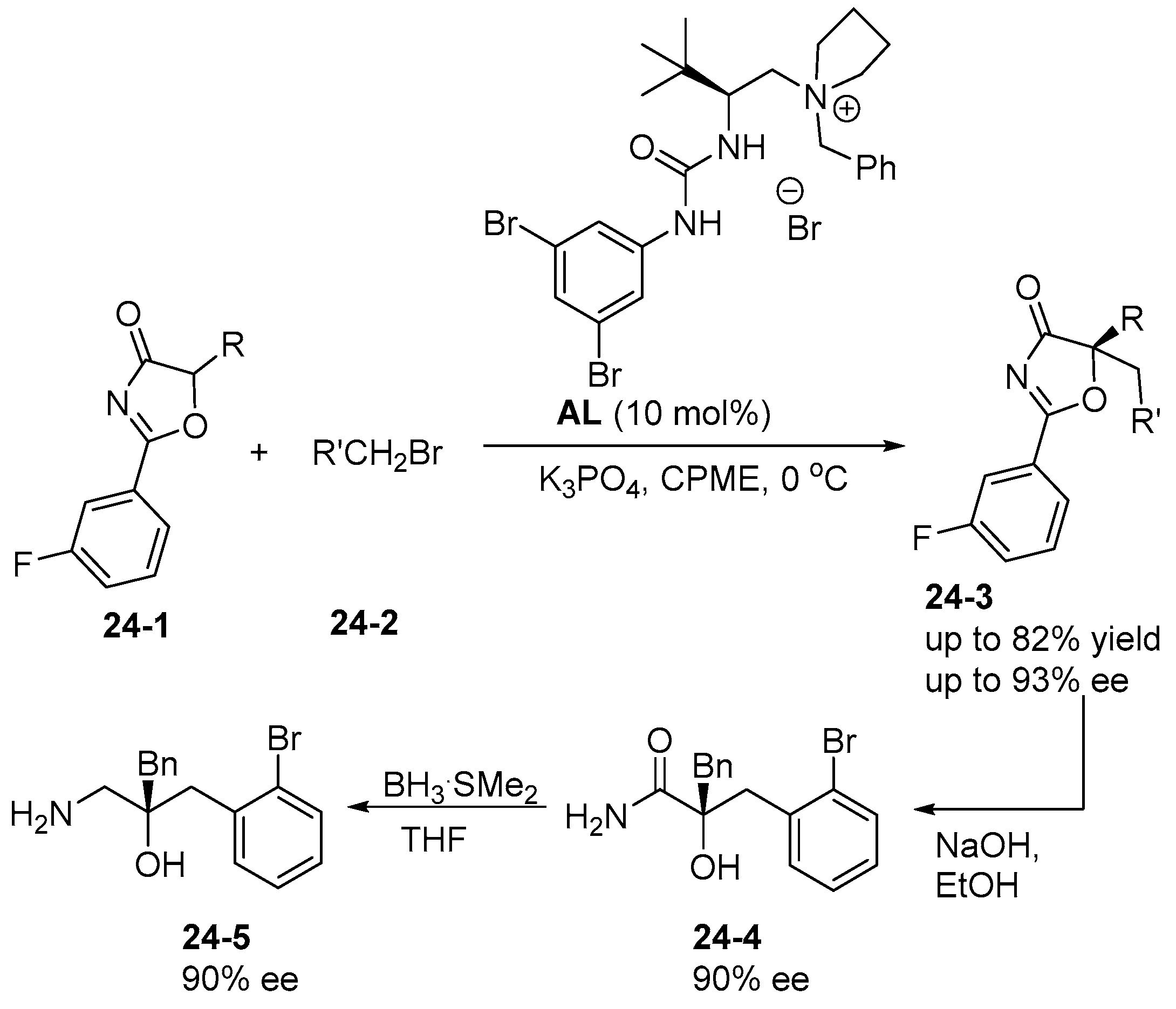
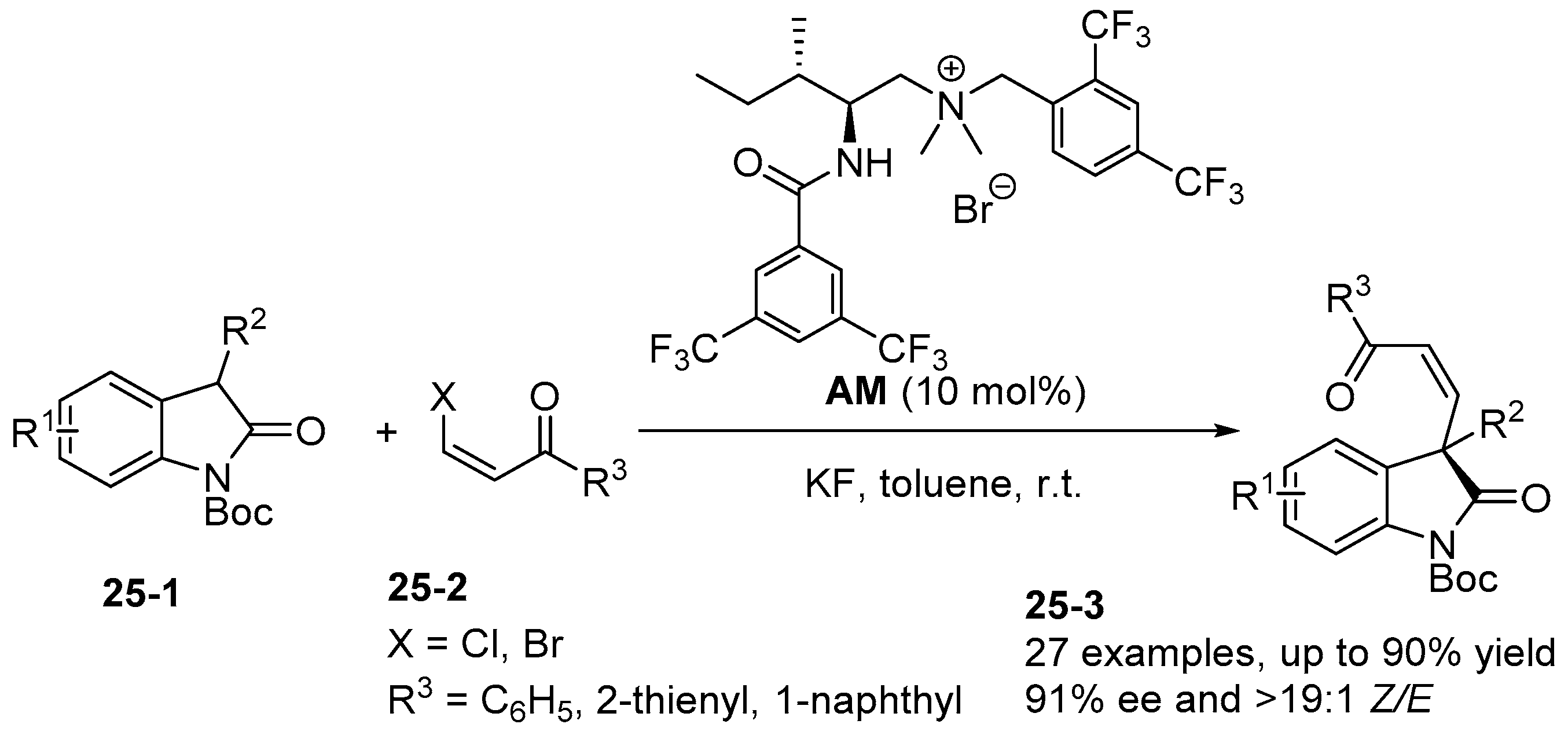
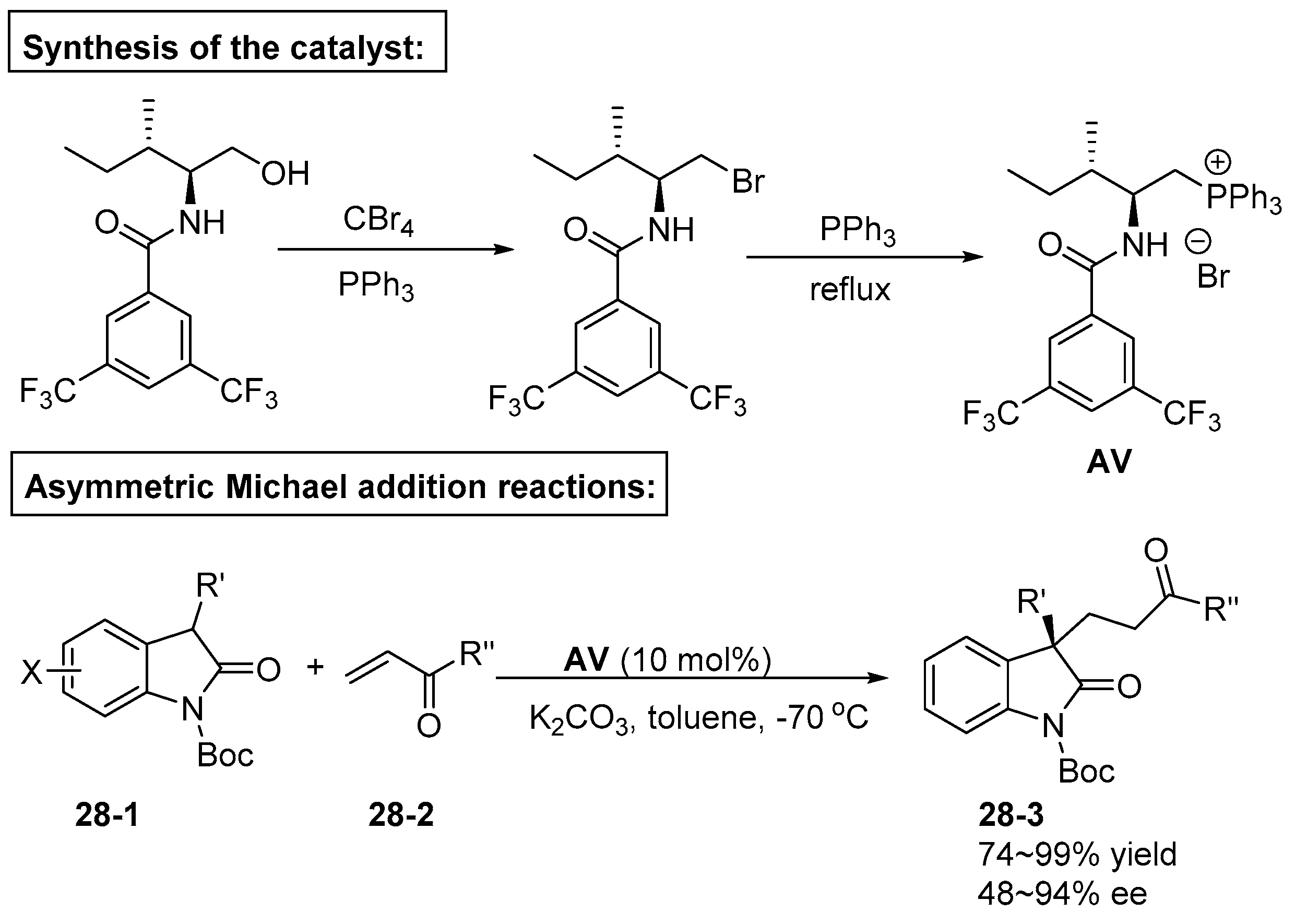
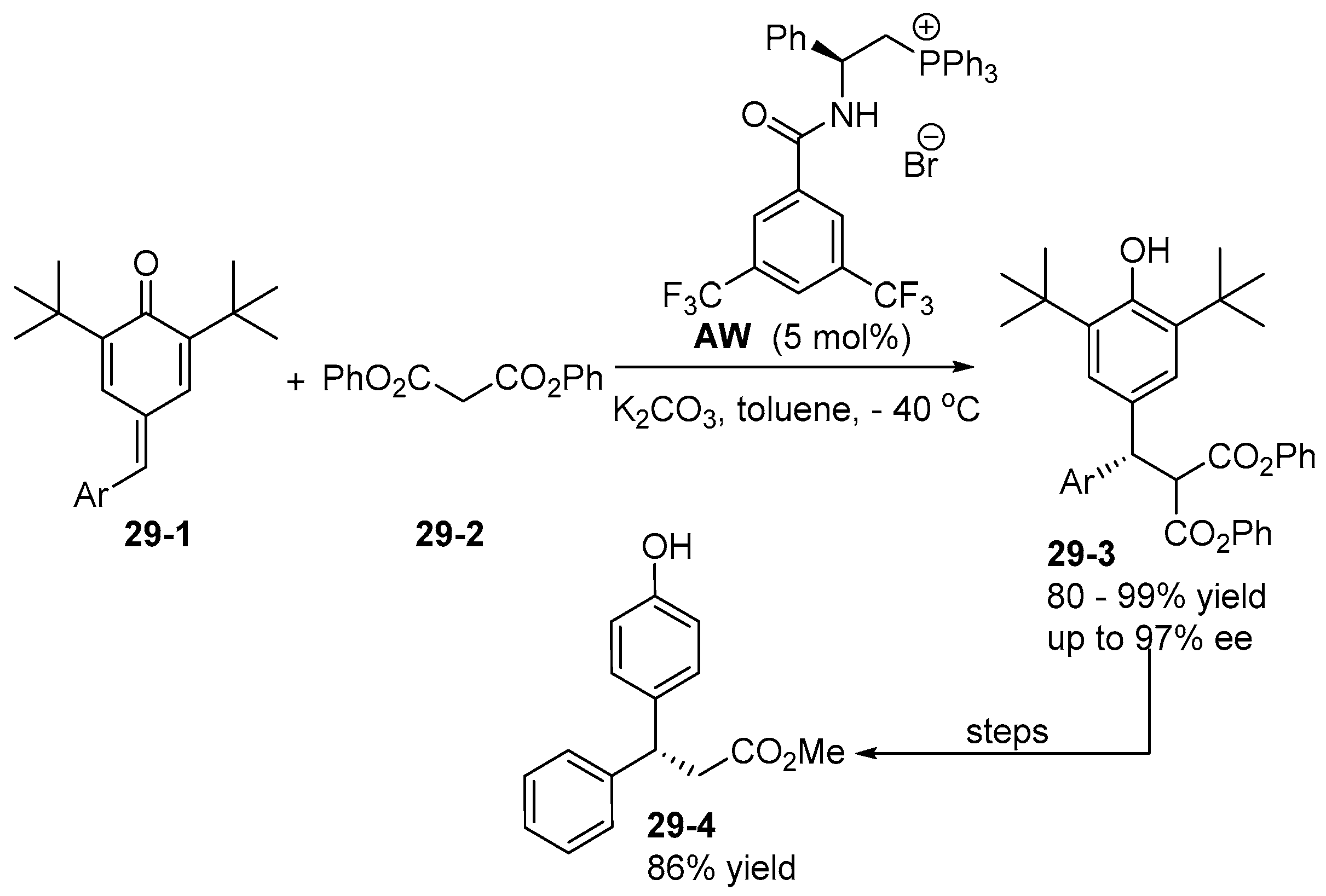
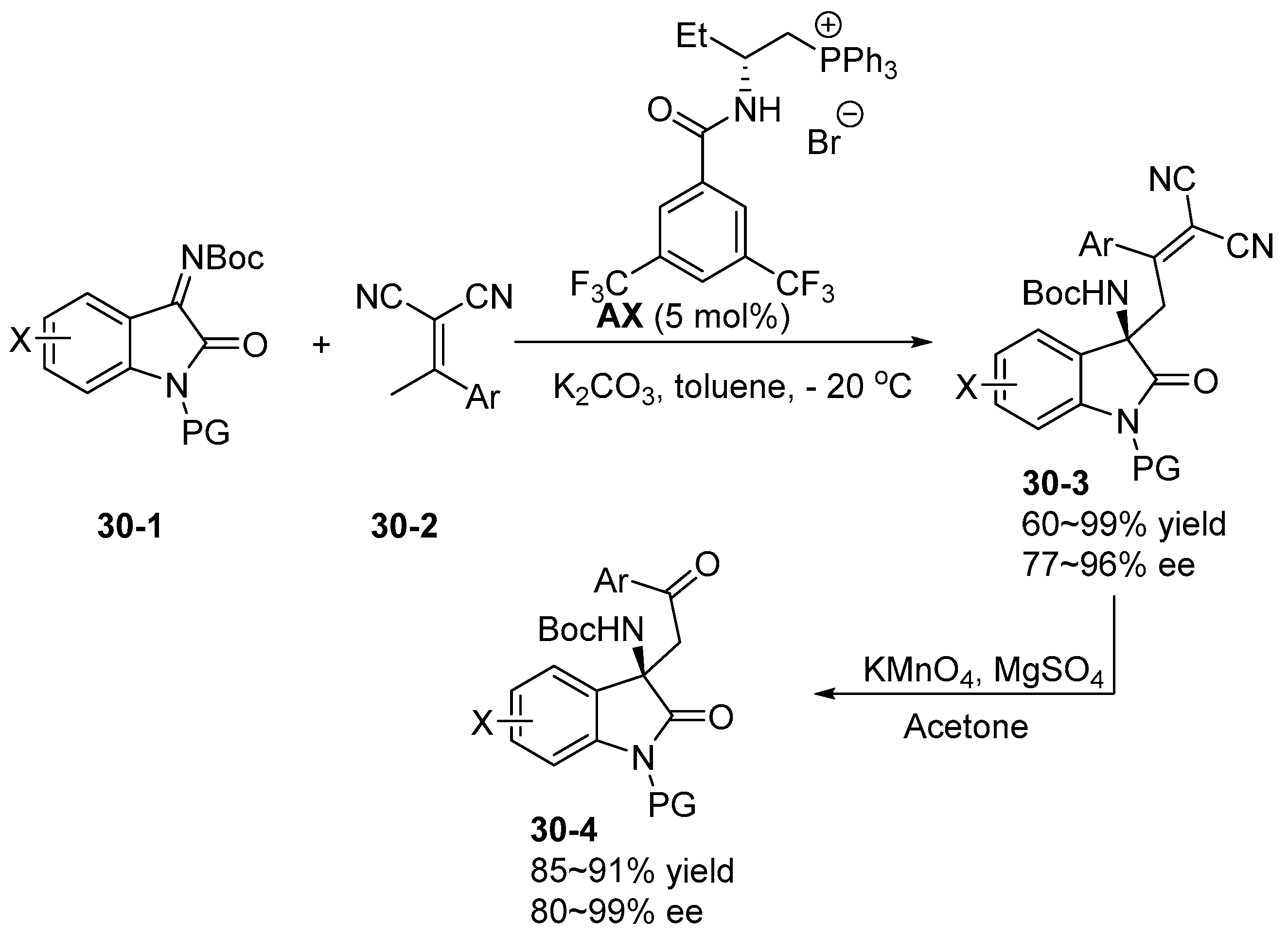
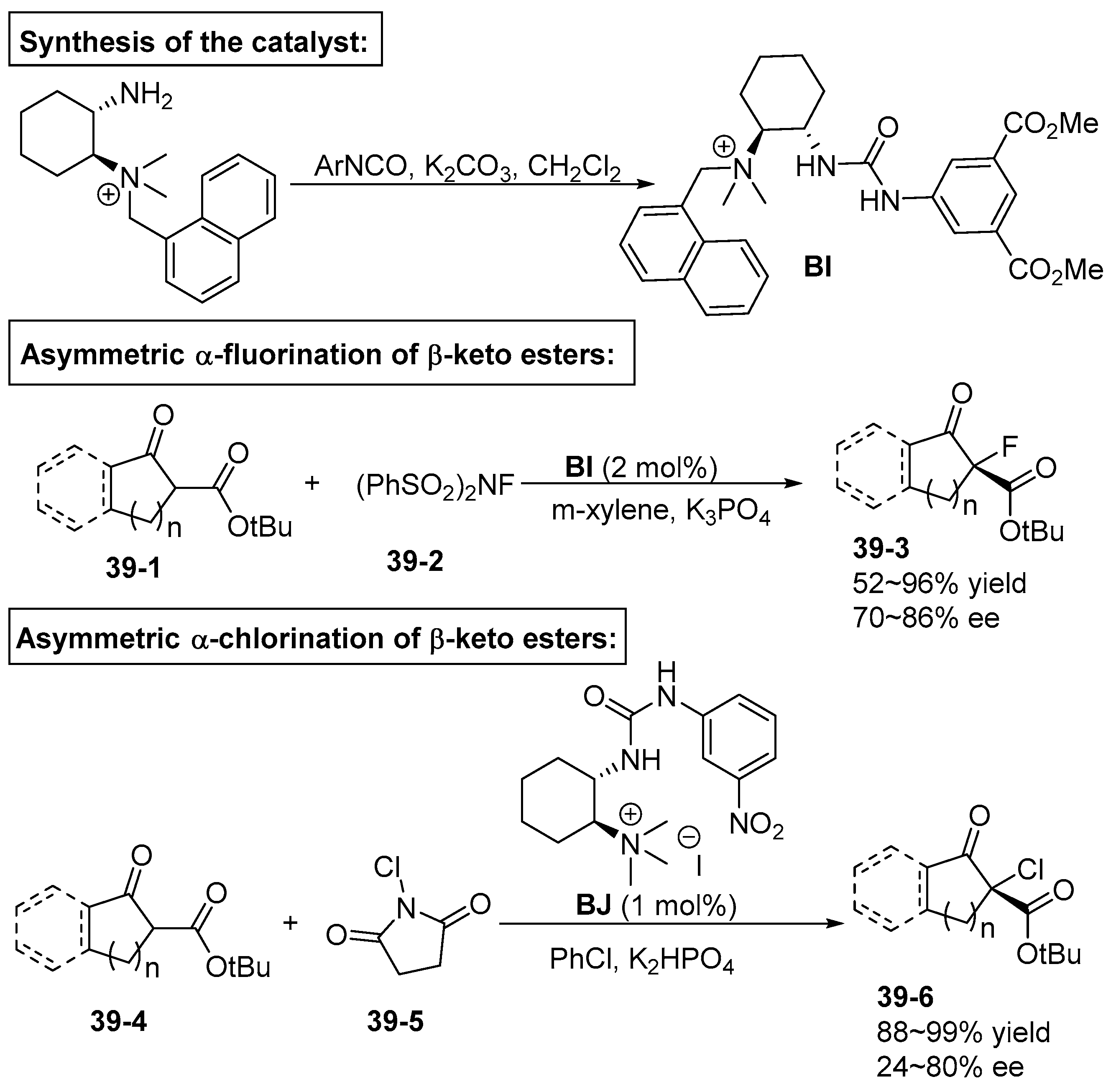
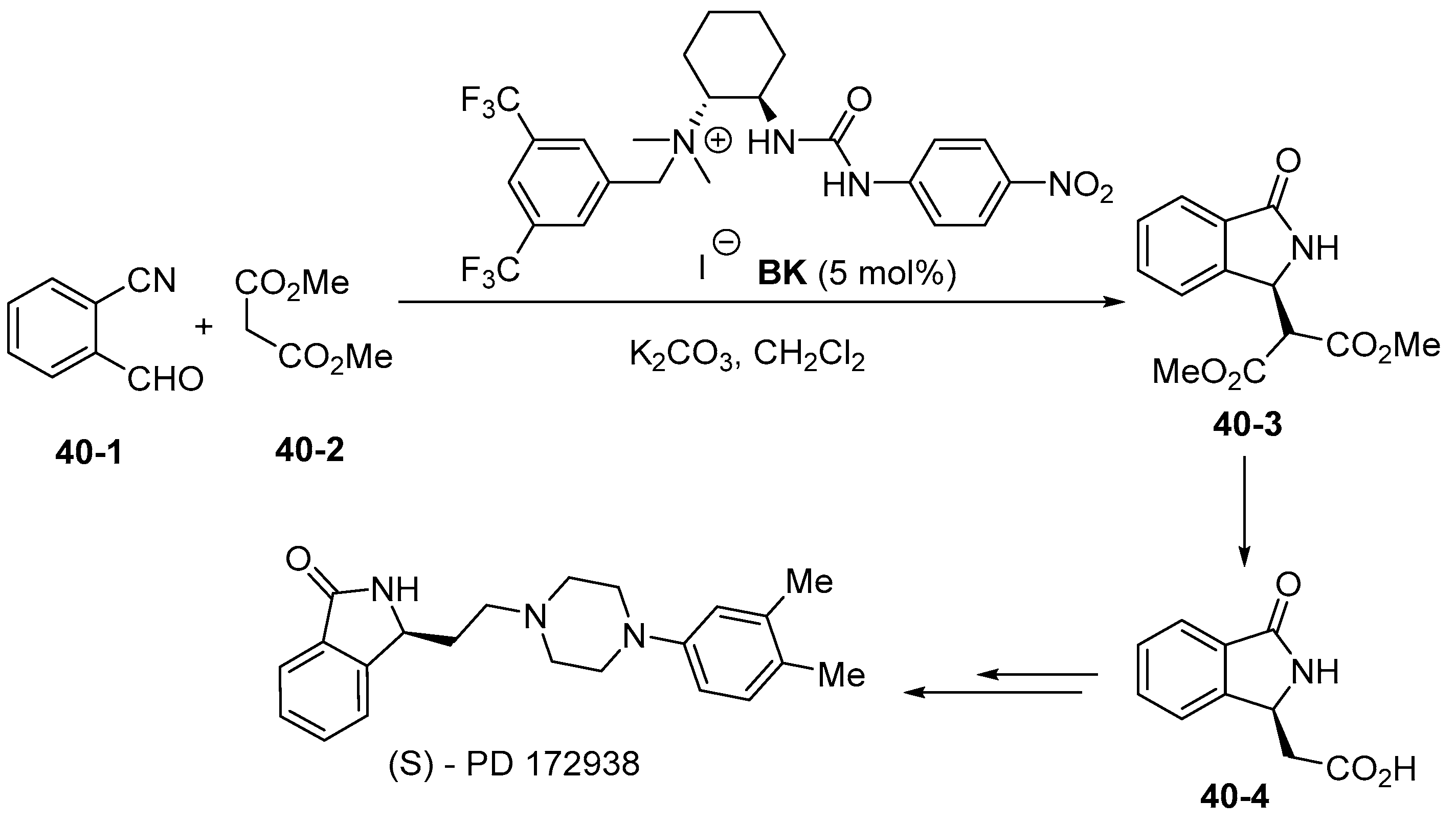
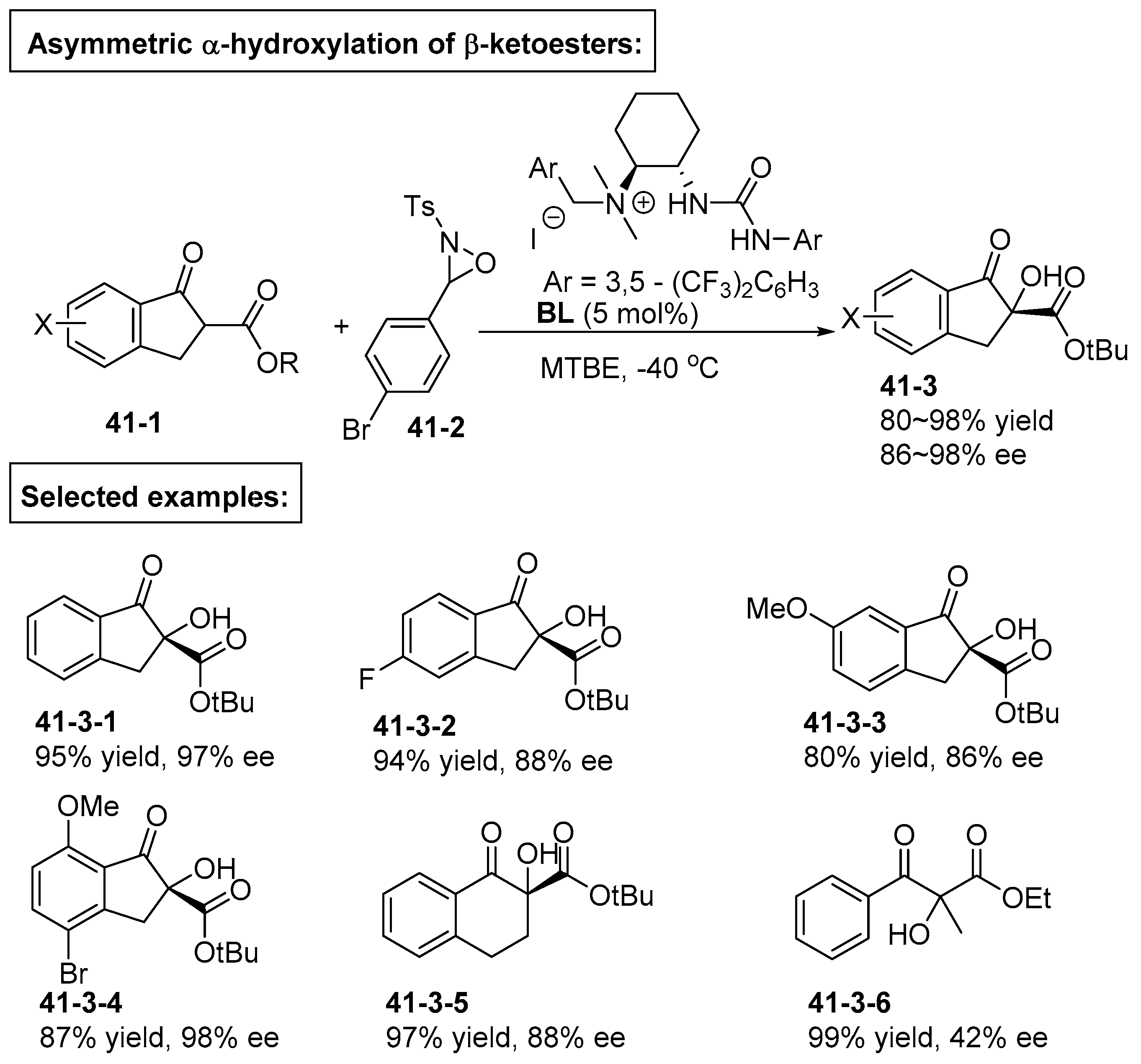
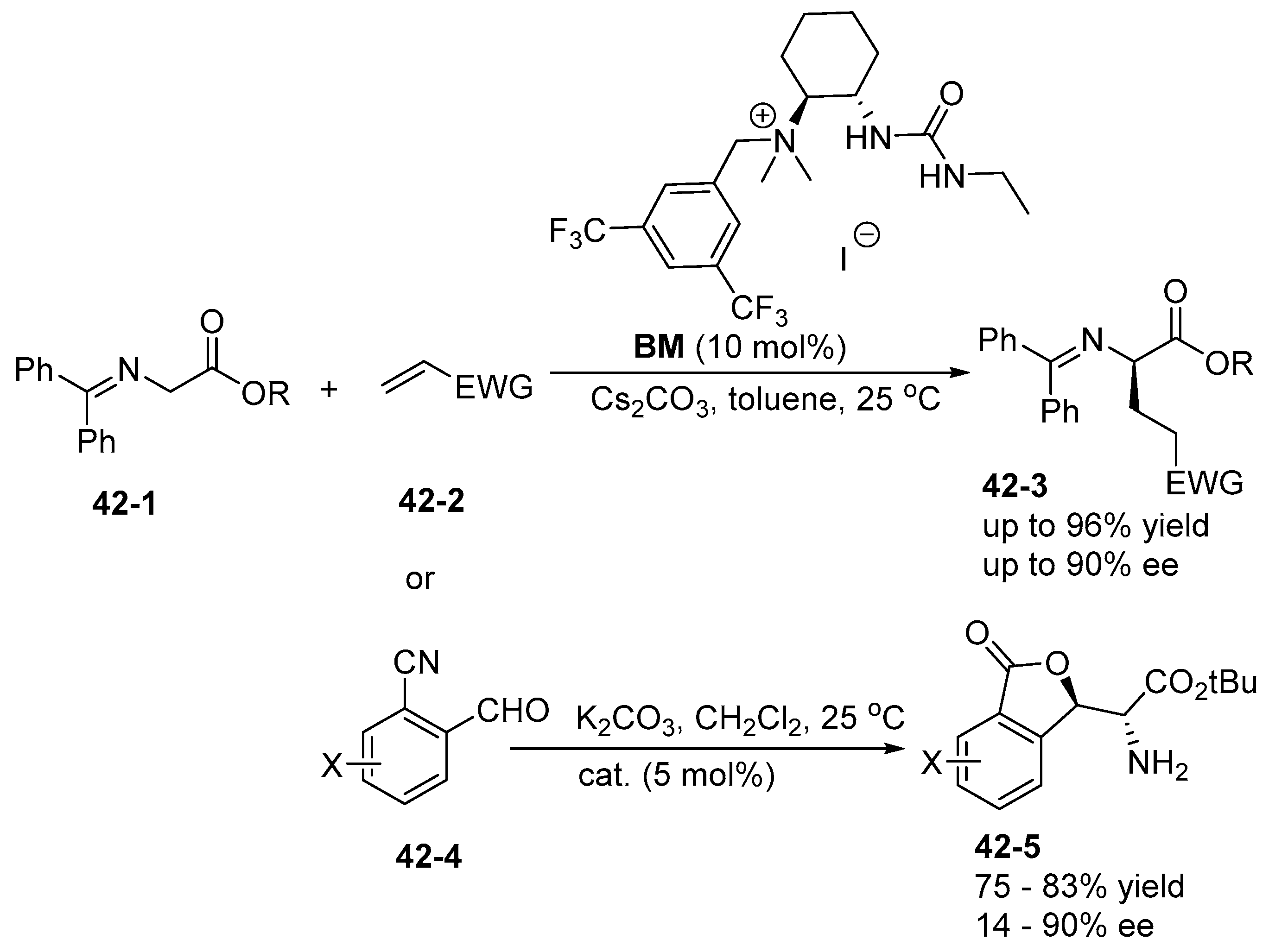
6. Derived from Cyclohexane
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Starks, C.M. Phase-transfer catalysis. I. Heterogeneous reactions involving anion transfer by quaternary ammonium and phosphonium salts. J. Am. Chem. Soc. 1971, 93, 195–199. [Google Scholar] [CrossRef]
- Maruoka, K. (Ed.) Asymmetric Phase Transfer Catalysis; Wiley: Weinheim, Germanny, 2008. [Google Scholar]
- Ooi, T.; Maruoka, K. Recent advances in asymmetric phase-transfer catalysis. Angew. Chem. Int. Ed. 2007, 46, 4222–4266. [Google Scholar] [CrossRef] [PubMed]
- Shirakawa, S.; Maruoka, K. Recent developments in asymmetric phase-transfer reactions. Angew. Chem. Int. Ed. 2013, 52, 4312–4348. [Google Scholar] [CrossRef] [PubMed]
- Novacek, J.; Waser, M. Bifunctional Chiral Quaternary Ammonium Salt Catalysts: A Rapidly Emerging Class of Powerful Asymmetric Catalysts. Eur. J. Org. Chem. 2013, 2013, 637–648. [Google Scholar] [CrossRef]
- Kaneko, S.; Kumatabara, Y.; Shirakawa, S. A new generation of chiral phase-transfer catalysis. Org. Biomol. Chem. 2016, 14, 5367–5376. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Kumatabara, Y.; Shirakawa, S. Chiral quaternary phosphonium salts as phase-transfer catalysts for environmentally benign asymmetric transformations. Green Chem. 2016, 18, 331–341. [Google Scholar] [CrossRef]
- Golandaj, A.; Ahmad, A.; Ramjugernath, D. Phosphonium Salts in Asymmetric Catalysis: A Journey in a Decade’s Extensive Research Work. Adv. Synth. Catal. 2017, 359, 3676–3706. [Google Scholar] [CrossRef]
- Schörgenhumer, J.; Tiffner, M.; Waser, M. Chiral phase-transfer catalysis in the asymmetric α-heterofunctionalization of prochiral nucleophiles. Beilstein J. Org. Chem. 2017, 13, 1753–1769. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.; Yasuda, N. Contemporary asymmetric phase transfer catalysis: Large-scale industrial applications. Org. Process Res. Dev. 2015, 19, 1731–1746. [Google Scholar] [CrossRef]
- Gururaja, G.; Waser, M. Asymmetric phase-transfer catalysis as a powerful tool in the synthesis of biologically active chiral complex natural products. Stud. Nat. Prod. Chem. 2014, 43, 409–435. [Google Scholar]
- Shirakawa, S.; Maruoka, K. Chiral onium salts (Phase-transfer reactions). Compr. Enant. Organocatalysis 2013, 2, 365–379. [Google Scholar]
- Shirakawa, S.; Moteki, S.A.; Maruoka, K. Asymmetric phase-transfer catalysis. In Modern Tools for the Synthesis of Complex Bioactive Molecules; John Wiley & Sons. Inc.: Hoboken, NJ, USA, 2012; pp. 213–242. [Google Scholar]
- Ager, D. Alkaloid derived auxiliaries: Cinchona alkaloid and derivatives. Compr. Chirality 2012, 3, 223–247. [Google Scholar]
- Brak, K.; Jacobsen, E.N. Asymmetric ion-pairing catalysis. Angew. Chem. Int. Ed. 2013, 52, 534–561. [Google Scholar] [CrossRef] [PubMed]
- Doyle, A.G.; Jacobsen, E.N. Small-molecule H-bond donors in asymmetric catalysis. Chem. Rev. 2007, 107, 5713–5743. [Google Scholar] [CrossRef] [PubMed]
- Fiaud, J.-C. Asymmetric alkylation reaction by means of a chiral phase transfer catalyst. Tetrahedron Lett. 1975, 16, 3495–3496. [Google Scholar] [CrossRef]
- Gomez-Bengoa, E.; Linden, A.; López, R.; Múgica-Mendiola, I.; Oiarbide, M.; Palomo, C. Asymmetric aza-Henry reaction under phase transfer catalysis: An experimental and theoretical study. J. Am. Chem. Soc. 2008, 130, 7955–7966. [Google Scholar] [CrossRef] [PubMed]
- Bernal, P.; Fernández, R.; Lassaletta, J.M. Organocatalytic asymmetric cyanosilylation of nitroalkenes. Chem. Eur. J. 2010, 16, 7714–7718. [Google Scholar] [CrossRef] [PubMed]
- Johnson, K.M.; Rattley, M.S.; Sladojevich, F.; Barber, D.M.; Nuñez, M.G.; Goldys, A.M.; Dixon, D.J. A new family of cinchona-derived bifunctional asymmetric phase-transfer catalysts: Applications to the enantio- and diastereoselective nitro-Mannich reaction of amidosulfones. Org. Lett. 2012, 14, 2492–2495. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Woods, P.A.; Smith, M.D. Cation-directed enantioselective synthesis of quaternary-substituted indolenines. Chem. Sci. 2013, 4, 2907–2911. [Google Scholar] [CrossRef]
- Wang, B.; Xu, T.; Zhu, L.; Lan, Y.; Wang, J.; Lu, N.; Wei, Z.; Lin, Y.; Duan, H. Highly enantioselective nitro-Mannich reaction of ketimines under phase-transfer catalysis. Org. Chem. Front. 2017, 4, 1266–1271. [Google Scholar] [CrossRef]
- Sorrentino, E.; Connon, S.J. Enantioselective Alkylative Kinetic Resolution of 2-Oxindole-Derived Enolates Promoted by Bifunctional Phase Transfer Catalysts. Org. Lett. 2016, 18, 5204–5207. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Liu, Y.; Sun, C.; Wei, Z.; Cao, J.; Liang, D.; Lin, Y.; Duan, H. Asymmetric Phase-Transfer Catalysts Bearing Multiple Hydrogen-Bonding Donors: Highly Efficient Catalysts for Enantio- and Diastereoselective Nitro-Mannich Reaction of Amidosulfones. Org. Lett. 2014, 16, 6432–6435. [Google Scholar] [CrossRef] [PubMed]
- Lu, N.; Li, R.; Wei, Z.; Cao, J.; Liang, D.; Lin, Y.; Duan, H.J. Enantio- and Diastereoselective Nitro-Mannich Reaction of α-Aryl Nitromethanes with Amidosulfones Catalyzed by Phase-Transfer Catalysts. Org. Chem. 2017, 82, 4668–4676. [Google Scholar] [CrossRef] [PubMed]
- Shirakawa, S.; Atsuyuki, K.; Tokuda, T.; Maruoka, K. Efficient approach for the design of effective chiral quaternary phosphonium salts in asymmetric conjugate additions. Chem. Sci. 2013, 4, 2248–2252. [Google Scholar] [CrossRef]
- Shirakawa, S.; Tokashi, T.; Kasai, A.; Maruoka, K. Design of chiral bifunctional quaternary phosphonium bromide catalysts possessing an amide moiety. Org. Lett. 2013, 15, 3350–3353. [Google Scholar] [CrossRef] [PubMed]
- Shirakawa, S.; Koga, K.; Tokuda, T.; Yamamoto, K.; Maruoka, K. Catalytic asymmetric synthesis of 3,3′-diaryloxindoles as triarymethanes with a chiral all-carbon quaternary center: Phase-transfer-catalyzed SNAr reaction. Angew. Chem. Int. Ed. 2014, 53, 6220–6223. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Maruoka, K.; Shirakawa, S. Chiral tertiary sulfonium salts as effective catalysts for asymmetric base-free neutral phase-transfer reactions. Angew. Chem. Int. Ed. 2017, 56, 4819–4923. [Google Scholar] [CrossRef] [PubMed]
- Hughes, A.B. (Ed.) Amino Acids, Peptides and Proteins in Organic Chemistry: Modified Amino Acids, Oranocatalysis and Enzymes; Wiely: Weinheim, Germanny, 2011; Volume 2. [Google Scholar]
- Chai, Z.; Zhao, G. Efficient organocatalysts derived from simple chiral acyclic amino acids in asymmetric catalysis. Catal. Sci. Technol. 2012, 2, 29–41. [Google Scholar] [CrossRef]
- Xu, L.-W.; Lu, Y. Primary amino acids: Privileged catalysts in enantioselective organocatalysis. Org. Biomol. Chem. 2008, 6, 2047–2053. [Google Scholar] [CrossRef] [PubMed]
- Ohmatsu, K.; Kiyokawa, M.; Ooi, T. Chiral 1,2,3-triazoliums as new cationic organic catalysts with anion-recognition ability: Application to asymmetric alkylation of oxindoles. J. Am. Chem. Soc. 2011, 133, 1307–1309. [Google Scholar] [CrossRef] [PubMed]
- Ohmatsu, K.; Hamajima, Y.; Ooi, T. Catalytic asymmetric ring opening of meso an dterminal aziridines with halides mediated by chiral 1,2,3-triazolium silicates. J. Am. Chem. Soc. 2012, 134, 8794–8797. [Google Scholar] [CrossRef] [PubMed]
- Ohmatsu, K.; Ando, Y.; Ooi, T. Asymmetric Substitution at the Tetrasubstituted Chiral Carbon: Catalytic Ring-Opening Alkylation of Racemic 2,2-Disubstituted Aziridines with 3-Substituted Oxindoles. J. Am. Chem. Soc. 2013, 135, 18706–18709. [Google Scholar] [CrossRef] [PubMed]
- Ohmatsu, K.; Goto, A.; Ooi, T. Catalytic asymmetric Mannich-type reactions of α-cyano α-sulfonyl carbanions. Chem. Commun. 2012, 48, 7913–7915. [Google Scholar] [CrossRef] [PubMed]
- Ohmatsu, K.; Ando, Y.; Nakashima, T.; Ooi, T. A Modular Strategy for the Direct Catalytic Asymmetric a-Amination of Carbonyl Compounds. Chem 2016, 1, 802–810. [Google Scholar] [CrossRef]
- Ohmatsu, K.; Ando, Y.; Ooi, T. In Situ Electrophilic Activation of Hydrogen Peroxide for Catalytic Asymmetric α-Hydroxylation of 3-Substituted Oxindoles. Synlett 2017, 28, 1291–1294. [Google Scholar]
- Wang, H.-Y.; Chai, Z.; Zhao, G. Novel bifunctional thioureaeammonium salt catalysts derived from amino acids: Application to highly enantio- and diastereoselective aza-Henry reaction. Tetrahedron 2013, 69, 5104–5111. [Google Scholar] [CrossRef]
- Wang, H.-Y.; Zhang, J.-X.; Cao, D.-D.; Zhao, G. Enantioselective Addition of Thiols to Imines Catalyzed by Thiourea-Quaternary Ammonium Salts. ACS Catal. 2013, 3, 2218–2221. [Google Scholar] [CrossRef]
- Zhang, J.-X.; Wang, H.-Y.; Jin, Q.-W.; Zheng, C.-W.; Zhao, G.; Shang, Y.-J. Thiourea–quaternary ammonium salt catalyzed asymmetric 1,3-dipolar cycloaddition of imino esters to construct spiro [pyrrolidin-3,3′-oxindoles]. Org. Lett. 2016, 18, 4774–4777. [Google Scholar] [CrossRef] [PubMed]
- Du, T.; Li, Z.; Zheng, C.; Fang, G.; Yu, L.; Liu, J.; Zhao, G. Highly enantioselective 1,3-dipolar cycloaddition of imino esters with benzofuranone derivatives catalyzed by thiourea−quaternary ammonium salt. Tetrahedron 2018, 74, 7485–7494. [Google Scholar] [CrossRef]
- Coppola, G.M.; Schuster, H.F. (Eds.) α–Hydroxy Acids in Enantioselective Synthesis; Wiely: Weinheim, Germany, 1997. [Google Scholar]
- Duan, S.; Li, S.; Ye, X.; Du, N.-N.; Tan, C.-H.; Jiang, Z. Enantioselective Synthesis of Dialkylated α-Hydroxy Carboxylic Acids through Asymmetric Phase-Transfer Catalysis. J. Org. Chem. 2015, 80, 7770–7778. [Google Scholar] [CrossRef] [PubMed]
- Jin, Q.; Zheng, C.; Zhao, G.; Zou, G. Bifunctional quaternary ammonium salts catalyzed stereoselective conjugate addition of oxindoles to electron-deficient β–haloalkenes. J. Org. Chem. 2017, 82, 4840–4850. [Google Scholar] [CrossRef] [PubMed]
- Cao, D.; Chai, Z.; Zhang, J.; Ye, Z.; Xiao, H.; Wang, H.; Chen, J.; Wu, X.; Zhao, G. Thiourea-phosphonium salts from amino acids: Cooperative phase-transfer catalysts in the enantioselective aza-Henry reaction. Chem. Commun. 2013, 49, 5972–5974. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Wang, K.; Ren, Y.; Li, N.; Tang, B.; Zhao, G. Asymmetric Strecker reactions catalyzed by thiourea phosphonium and ammonium salts. Adv. Synth. Catal. 2017, 359, 1819–1824. [Google Scholar] [CrossRef]
- Wu, X.-Y.; Liu, Q.; Liu, Y.; Wang, Q.; Zhang, Y.; Chen, J.; Cao, W.; Zhao, G. Amino acid-derived phosphonium salts-catalyzed Michael addition of 3-substituted oxindoles. Adv. Synth. Catal. 2013, 355, 2701–2706. [Google Scholar] [CrossRef]
- Chu, W.-D.; Zhang, L.-F.; Bao, X.; Zhao, X.-H.; Zeng, C.; Du, J.-Y.; Zhang, G.-B.; Wang, F.-X.; Ma, X.-Y.; Fan, C.-A. Asymmetric catalytic 1,6-conjugate addition /aromatization of para-quinone methides: Enantioselective introduction of functionalized diarylmethine stereogenic centers. Angew. Chem. Int. Ed. 2013, 52, 9229–9233. [Google Scholar] [CrossRef] [PubMed]
- Ge, L.; Lu, X.; Cheng, C.; Chen, J.; Cao, W.; Wu, X.; Zhao, G. Amide-Phosphonium Salt as Bifunctional Phase Transfer Catalyst for Asymmetric 1,6-Addition of Malonate Esters to para-Quinone Methides. J. Org. Chem. 2016, 81, 9315–9325. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.; Lu, X.; Ge, L.; Chen, J.; Cao, W.; Wu, X.; Zhao, G. Effective asymmetric vinylogous Mannich reaction of isatin imines with α,α-dicyanoolefins in the presence of a simple chiral amide phosphonium bifunctional phase transfer catalyst. Org. Chem. Front. 2017, 4, 101–114. [Google Scholar] [CrossRef]
- Wen, S.; Li, X.; Lu, Y. Enantioselective alkylation of glycine imine promoted by amino-acid-derived phosphonium salts. Asian J. Org. Chem. 2016, 5, 1457–1460. [Google Scholar] [CrossRef]
- Zhang, J.; Cao, D.; Wang, H.; Zhao, G.; Shang, Y. Enantioselective desymmetrization of meso-aziridines with aromatic thiols catalyzed by chiral bifunctional quaternary phosphonium salts derived from α–amino acids. Tetrahedron 2015, 71, 1785–1791. [Google Scholar] [CrossRef]
- Pan, R.; Zhang, J.; Zheng, C.; Wang, H.; Cao, D.; Cao, W.; Zhao, G. Enantioselective direct Mannich reaction of functionalized acetonitrile to N-Boc imines catalyzed by quaternary phosphonium catalysis. Tetrahedron 2017, 73, 2349–2358. [Google Scholar] [CrossRef]
- Wen, S.; Li, X.; Yao, W.; Waheed, A.; Ullah, N.; Lu, Y. Highly enantioselective conjugate addition of glycine imines to activated alkenes catalyzed by amino-acid-derived chiral phosphonium salts. Eur. J. Org. Chem. 2016, 2016, 4298–4301. [Google Scholar] [CrossRef]
- Cao, D.; Zhang, J.; Wang, H.; Zhao, G. Dipeptide-Derived Multifunctional Quaternary Phosphonium Salt Catalyzed Asymmetric Cyclizations via a Tandem Michael Addition/SN2 Sequence. Chem. Eur. J. 2015, 21, 9998–10002. [Google Scholar] [CrossRef] [PubMed]
- Moyano, A.; Rios, R. Asymmetric organocatalytic cyclization and cycloaddition reactions. Chem. Rev. 2011, 111, 4703–4832. [Google Scholar] [CrossRef] [PubMed]
- Volla, C.M.R.; Atodiresei, I.; Rurping, M. Catalytic C-C bond-forming multi-component cascade or domino reactions: Pushing the boundaries of complexity in asymmetric organocatalysis. Chem. Rev. 2014, 114, 2390–2431. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Lu, H.; Xu, P.-F. Asymmetric catalytic cascade reactions for constructing diverse scaffolds and complex molecules. Acc. Chem. Res. 2015, 48, 1832–1844. [Google Scholar] [CrossRef] [PubMed]
- Cao, D.; Fang, G.; Zhang, J.; Wang, H.; Zheng, C.; Zhao, G. Enantioselective Michael Addition of Malonates to Chalcone Derivatives Catalyzed by Dipeptide-derived Multifunctional Phosphonium Salts. J. Org. Chem. 2016, 81, 9973–9982. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Cao, D.; Zhang, J.; Wang, H.; Zou, G.; Zhao, G. Dipeptide-derived multifunctional phosphonium salt as a catalyst to synthesize highly functionalized chiral cyclopentanes. Tetrahedron 2016, 72, 4141–4150. [Google Scholar] [CrossRef]
- Zhang, J.; Cao, D.; Wang, H.; Zheng, C.; Zhao, G.; Shang, Y. Enantioselective Construction of Spirocyclic Oxindoles via Tandem Michael/Michael Reactions Catalyzed by Multifunctional Quaternary Phosphonium Salt. J. Org. Chem. 2016, 81, 10558–10568. [Google Scholar] [CrossRef] [PubMed]
- Bennani, Y.L.; Hanessian, S. Trans-1,2-Diaminocyclohexane Derivatives as Chiral Reagents, Scaffolds, and Ligands for Catalysis: Applications in Asymmetric Synthesis and Molecular Recognition. Chem. Rev. 1997, 97, 3161–3196. [Google Scholar] [CrossRef] [PubMed]
- Kizirian, J.-C. Chiral tertiary diamine in asymmetric synthesis. Chem. Rev. 2008, 108, 140–205. [Google Scholar] [CrossRef] [PubMed]
- Okino, T.; Hoashi, Y.; Takemoto, Y.J. Enantioselective Michael reaction of malonates to nitroolefins catalyzed by bifunctional organocatalysts. J. Am. Chem. Soc. 2003, 125, 12672–12673. [Google Scholar] [CrossRef] [PubMed]
- Novacek, J.; Waser, M. Syntheses and Applications of (Thio)Urea-Containing Chiral Quaternary Ammonium Salt Catalysts. Eur. J. Org. Chem. 2014, 2014, 802–809. [Google Scholar] [CrossRef] [PubMed]
- Novacek, J.; Monkowius, U.; Himmelsbach, M.; Waser, M. Asymmetric α-chlorination of β-ketoesters using bifunctional ammonium salt catalysis. Monatsh. Chem. 2016, 147, 533–538. [Google Scholar] [CrossRef]
- Mola, A.D.; Tiffner, M.; Scorzelli, F.; Palombi, L.; Filosa, R.; Caprariis, P.D.; Waser, M.; Massa, A. Bifunctional phase-transfer catalysis in the asymmetric synthesis of biologically active isoindolinones. Beilstein J. Org. Chem. 2015, 11, 2591–2599. [Google Scholar] [CrossRef] [PubMed]
- Novacek, J.; Izzo, J.A.; Vetticatt, M.J.; Waser, M. Bifunctional Ammonium Salt Catalyzed Asymmetric α-Hydroxylation of β-Ketoesters by Simultaneous Resolution of Oxaziridines. Chem. Eur. J. 2016, 22, 17339–17344. [Google Scholar] [CrossRef] [PubMed]
- Tiffner, M.; Novacek, J.; Busillo, A.; Gratzer, K.; Massa, A.; Waser, M. Design of chiral urea-quaternary ammonium salt hybrid catalysts for asymmetric reactions of glycine Schiff bases. RSC Adv. 2015, 5, 78941–78949. [Google Scholar] [CrossRef] [PubMed]

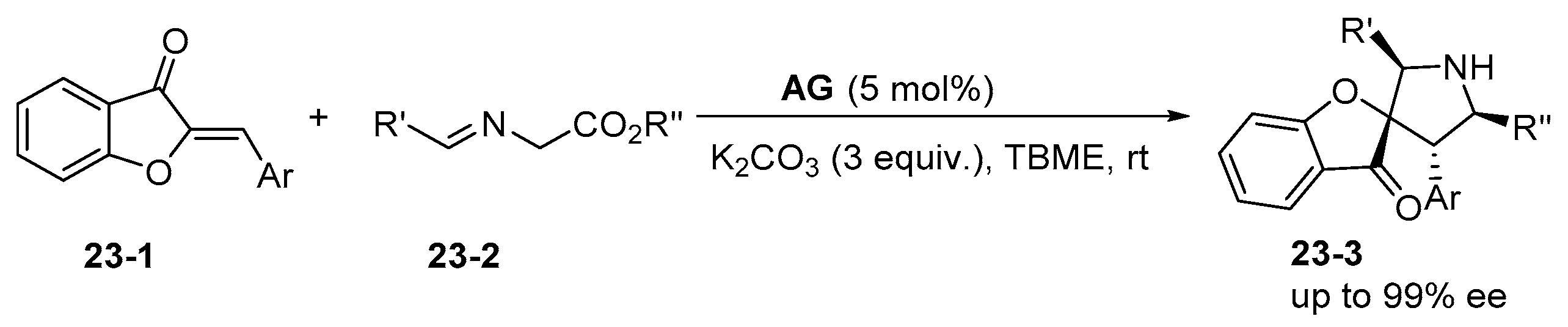

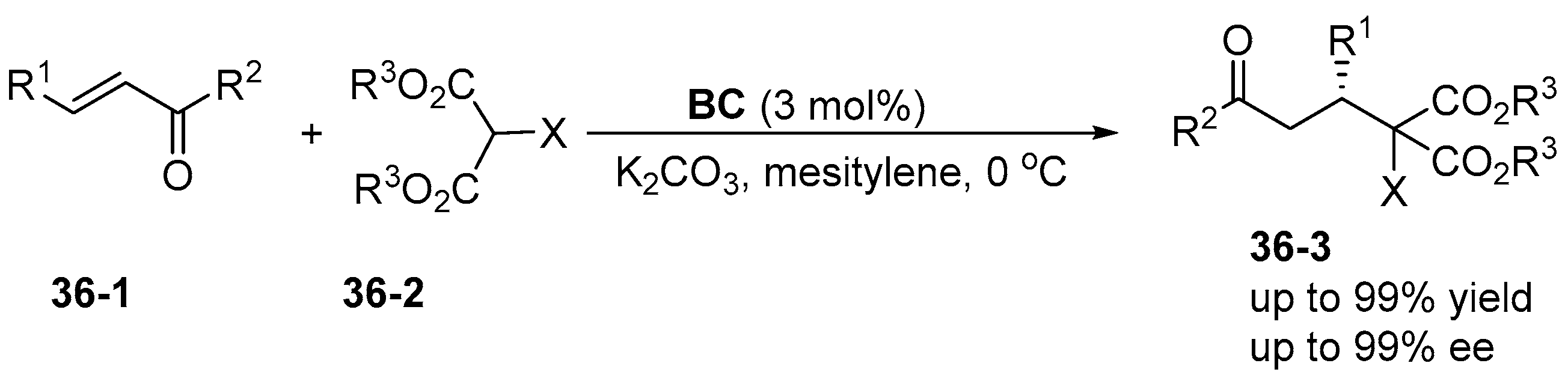

© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, H. Chiral Phase-Transfer Catalysts with Hydrogen Bond: A Powerful Tool in the Asymmetric Synthesis. Catalysts 2019, 9, 244. https://doi.org/10.3390/catal9030244
Wang H. Chiral Phase-Transfer Catalysts with Hydrogen Bond: A Powerful Tool in the Asymmetric Synthesis. Catalysts. 2019; 9(3):244. https://doi.org/10.3390/catal9030244
Chicago/Turabian StyleWang, Hongyu. 2019. "Chiral Phase-Transfer Catalysts with Hydrogen Bond: A Powerful Tool in the Asymmetric Synthesis" Catalysts 9, no. 3: 244. https://doi.org/10.3390/catal9030244
APA StyleWang, H. (2019). Chiral Phase-Transfer Catalysts with Hydrogen Bond: A Powerful Tool in the Asymmetric Synthesis. Catalysts, 9(3), 244. https://doi.org/10.3390/catal9030244



