Effect of Dilute Acid and Alkali Pretreatments on the Catalytic Performance of Bamboo-Derived Carbonaceous Magnetic Solid Acid
Abstract
1. Introduction
2. Results and Discussion
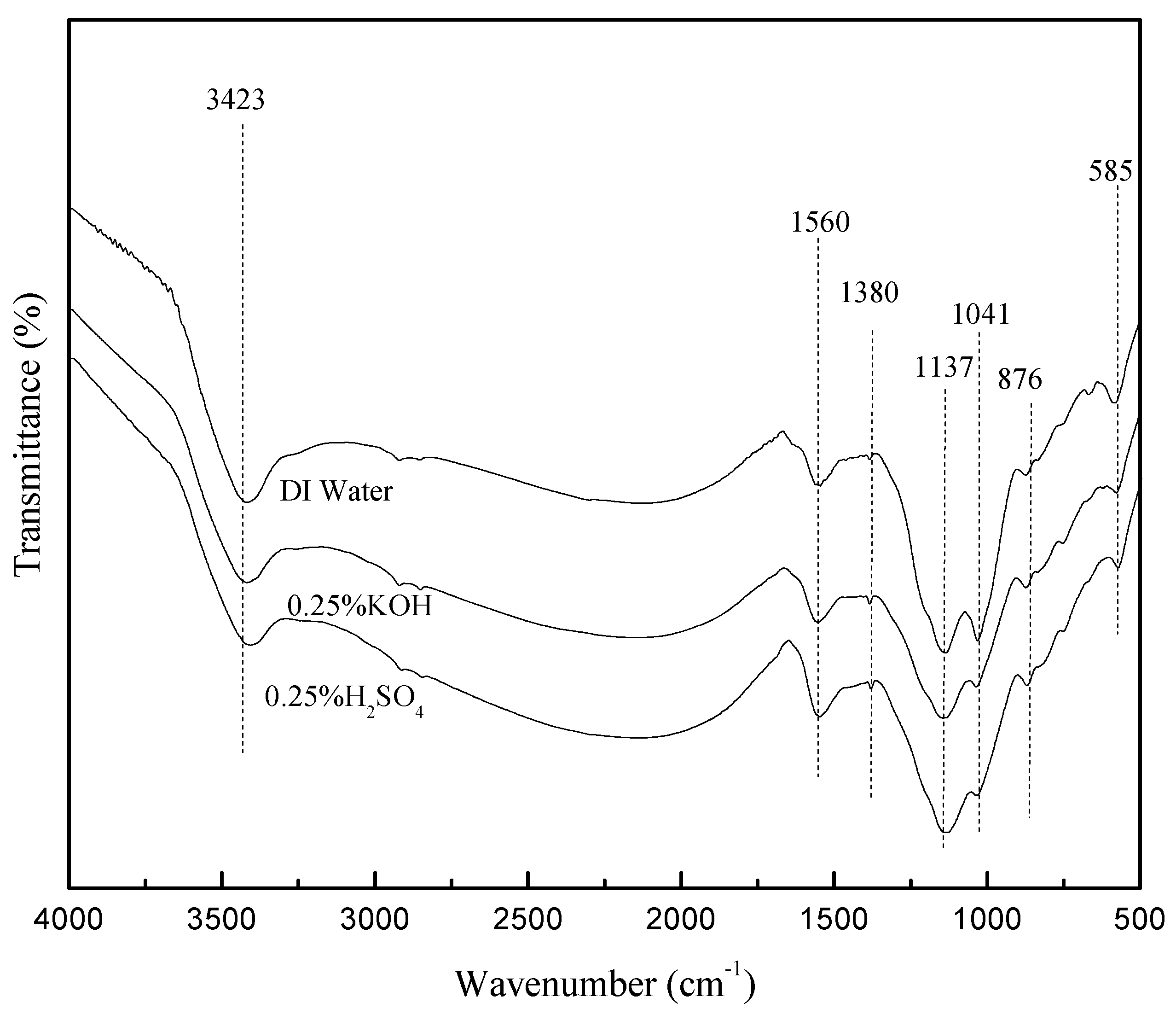
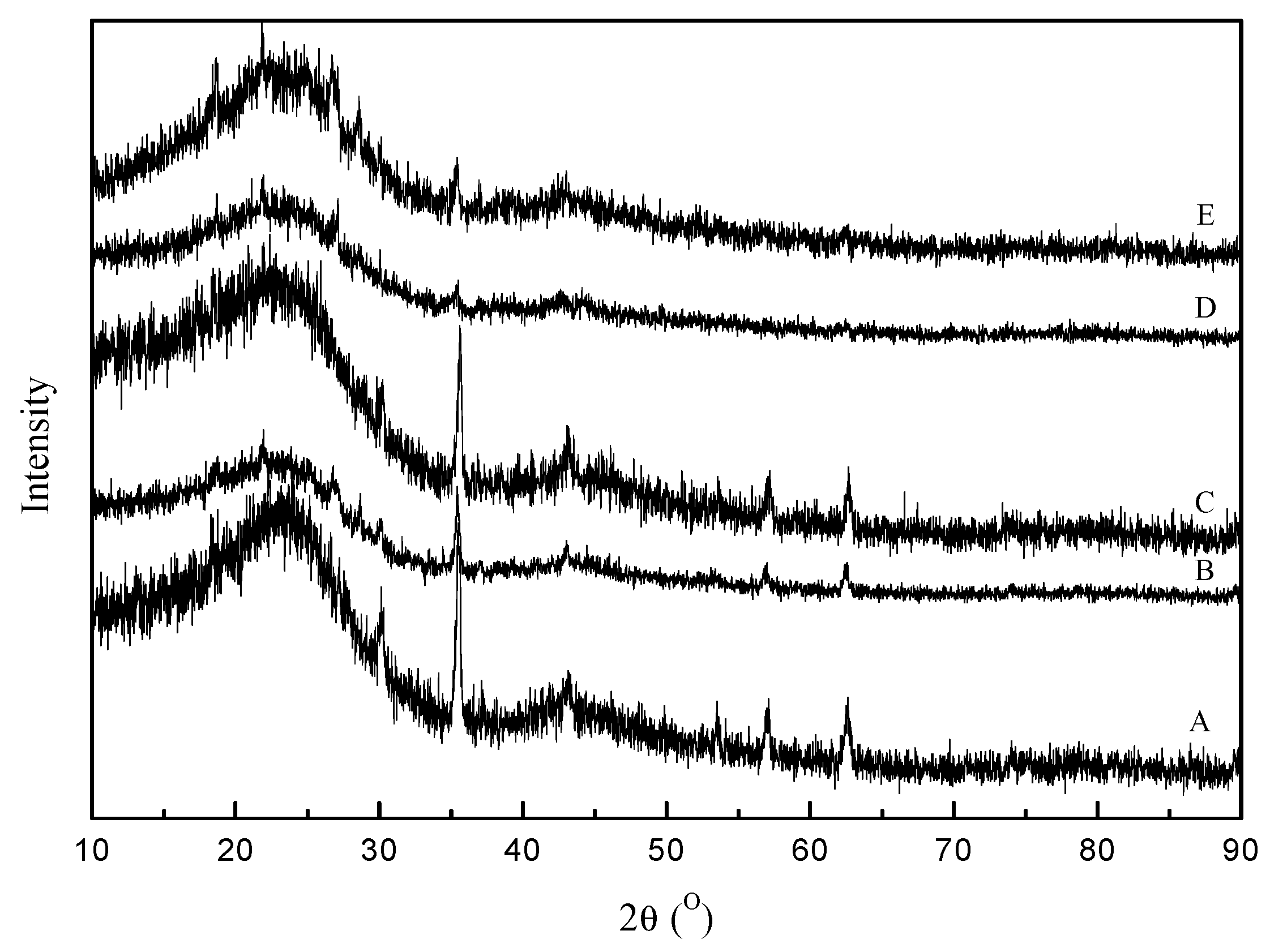
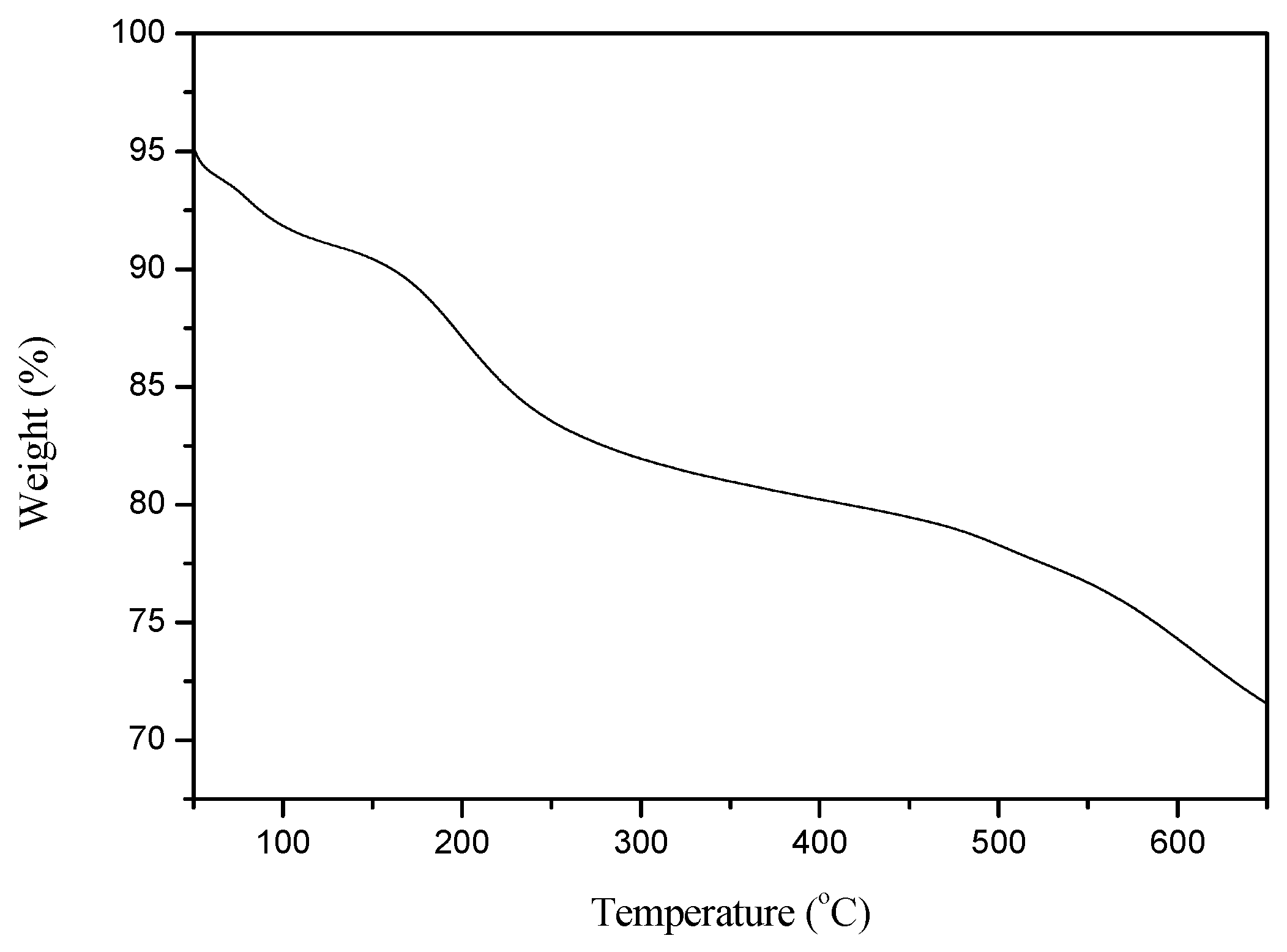
2.1. Characterization of the Catalyst
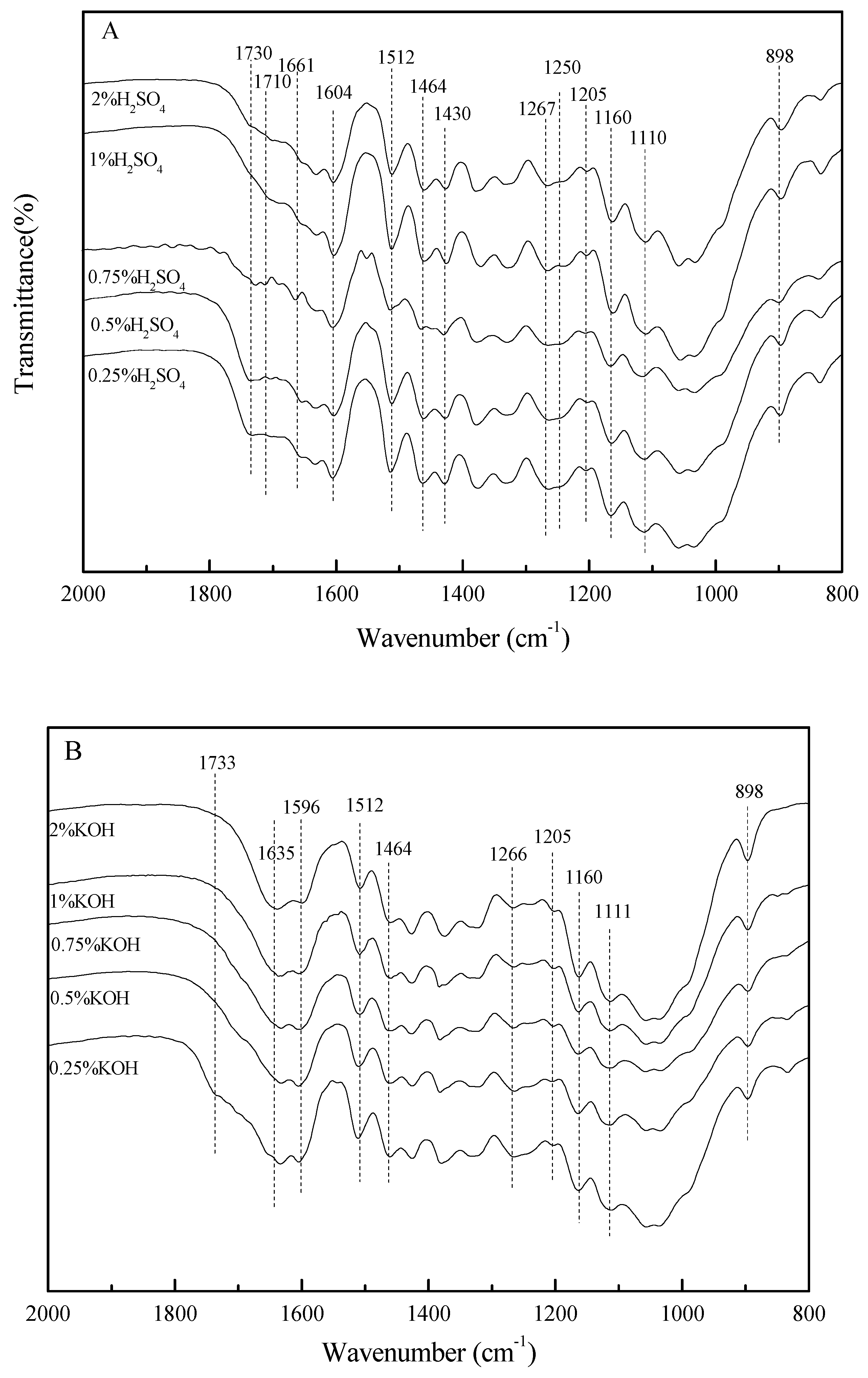
2.2. Effects of Pretreatment Conditions on the Chemical Composition of Bamboo
2.3. Hydrolysis of Corncob with Bamboo-Derived Carbonaceous Magnetic Solid Acid
3. Materials and Methods
3.1. Materials
3.2. Methods
3.2.1. Catalyst Characterization
3.2.2. Two-Step Preparation of Bamboo-Derived Magnetic Carbonaceous Solid Acid
3.2.3. Hydrolysis of Corncob by Bamboo-Derived Magnetic Solid Acid
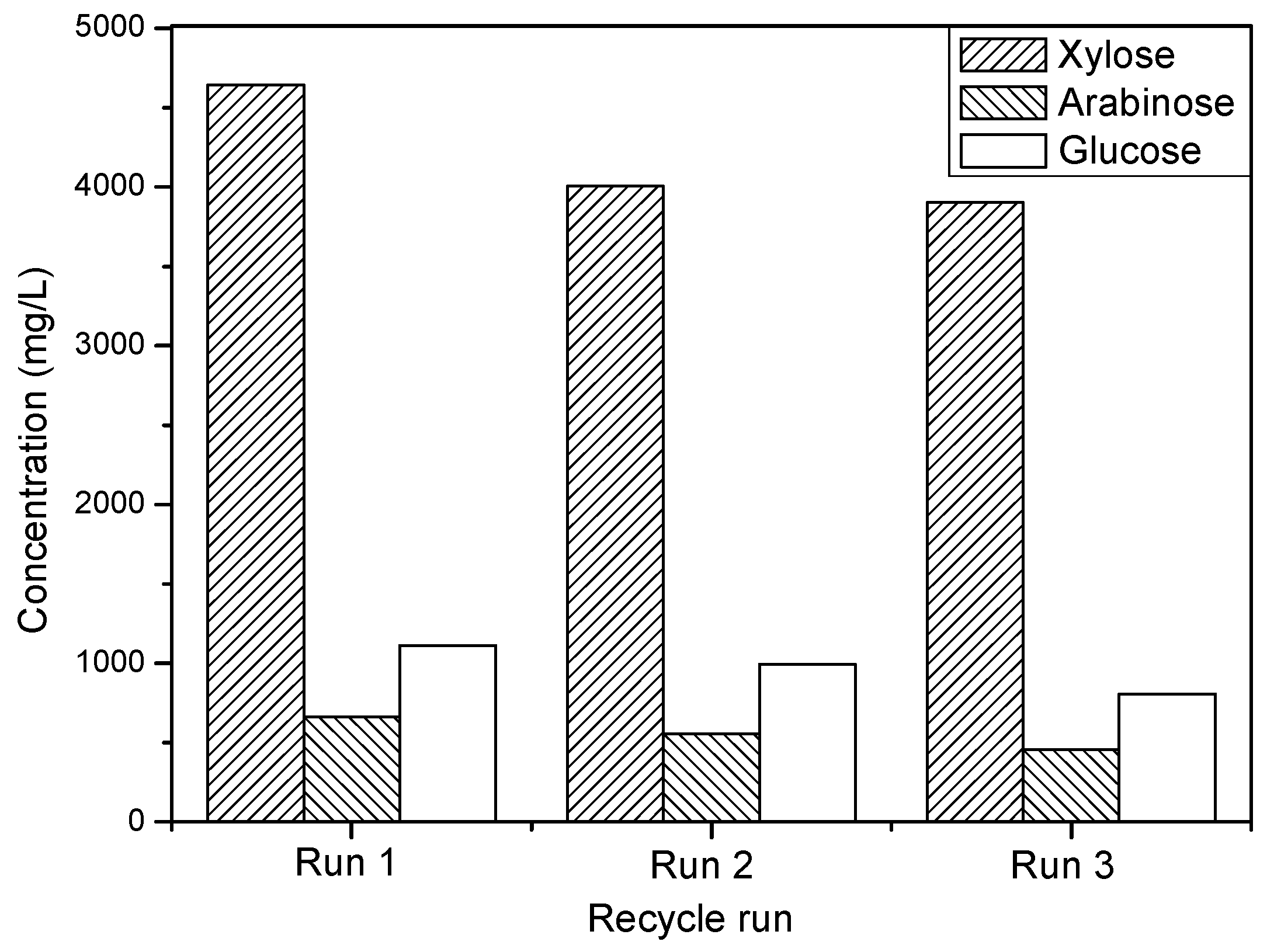
3.2.4. Recyclability of Catalyst
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Schmitt, C.; Belén, M.; Reolon, G.; Zimmermann, M.; Raffelt, K.; Grunwaldt, J.-D.; Dahmen, N. Synthesis and regeneration of nickel-based catalysts for hydrodeoxygenation of beech wood fast pyrolysis bio-oil. Catalysts 2018, 8, 449–477. [Google Scholar] [CrossRef]
- Xin, D.; Yang, Z.; Liu, F.; Xu, X.; Zhang, J. Comparison of aqueous ammonia and dilute acid pretreatment of bamboo fractions: Structure properties and enzymatic hydrolysis. Bioresour. Technol. 2015, 175, 529–536. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Deng, A.; Ren, J.; Liu, C.; Lu, Q.; Zhong, L.; Peng, F.; Sun, R. Catalytic hydrothermal pretreatment of corncob into xylose and furfural via solid acid catalyst. Bioresour. Technol. 2014, 158, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Gupta, P.; Paul, S. Solid acids: Green alternatives for acid catalysis. Catal. Today 2014, 236, 153–170. [Google Scholar] [CrossRef]
- Lee, J. Biological conversion of lignocellulosic biomass to ethanol. J. Biotechnol. 1997, 56, 1–24. [Google Scholar] [CrossRef]
- Börjesson, M.; Larsson, A.; Westman, G.; Ström, A. Periodate oxidation of xylan-based hemicelluloses and its effect on their thermal properties. Carbohydr. Polym. 2018, 202, 280–287. [Google Scholar] [CrossRef] [PubMed]
- Pellera, F.-M.; Gidarakos, E. Chemical pretreatment of lignocellulosic agroindustrial waste for methane production. Waste Manag. 2018, 71, 689–703. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Ren, J.; Zhong, L.; Sun, R.; Liang, L. Production of furfural from xylose, water-insoluble hemicelluloses and water-soluble fraction of corncob via a tin-loaded montmorillonite solid acid catalyst. Bioresour. Technol. 2015, 176, 176–183. [Google Scholar] [CrossRef] [PubMed]
- Zaccheria, F.S.F.; Iftitah, E.D.; Ravasio, N. Brønsted and Lewis Solid Acid Catalysts in the Valorization of Citronellal. Catalysts 2018, 8, 410–411. [Google Scholar] [CrossRef]
- Solinas, A.; Taddei, M. Solid-supported reagents and catch-and-release techniques in organic synthesis. Synthesis 2007, 38, 2409–2453. [Google Scholar] [CrossRef]
- Sebti, S.D.; Tahir, R.; Nazih, R.; Boulaajaj, S.D. Comparison of different Lewis acid supported on hydroxyapatite as new catalysts of Friedel-Crafts alkylation. Appl. Catal. A Gen. 2001, 218, 25–30. [Google Scholar] [CrossRef]
- Tanabe, K.; Yamaguchi, T. Acid-base bifunctional catalysis by ZrO2 and its mixed oxides. Catal. Today 1994, 20, 185–197. [Google Scholar] [CrossRef]
- Hara, M.; Yoshida, T.; Takagaki, A.; Takata, T.; Kondo, J.; Hayashi, S.; Domen, K. Carbon material as a strong protonic acid. Angew. Chem. Int. Ed. 2004, 43, 2955–2958. [Google Scholar] [CrossRef] [PubMed]
- Oregui, M.; Miletic, N.; Hao, W.; Björnerbäck, F.; Rosnes, M.; Garitaonandia, J.; Hedin, N.; Arias, P.L.; Barth, T. High-performance magnetic activated carbon from solid waste from lignin conversion processes. Part II: Their use as NiMo catalyst supports for lignin conversion. Energy Procedia 2017, 114, 6272–6296. [Google Scholar]
- Liu, F.; Rotaru, A.E.; Shrestha, P.M.; Malvankar, N.S.; Nevin, K.P.; Lovley, D.R. Promoting direct interspecies electron transfer with activated carbon. Energy Environ. Sci. 2012, 10, 8982–8989. [Google Scholar] [CrossRef]
- Li, X.; Lei, T.; Wang, Z.; Li, X.; Wen, M.; Yang, M.; Chen, G.; He, X.; Xu, H.; Guan, Q.; et al. Catalytic pyrolysis of corn straw with magnetic solid acid catalyst to prepare levulinic acid by response surface methodology. Ind. Crop. Prod. 2018, 116, 73–80. [Google Scholar] [CrossRef]
- Chen, T.; Peng, L.; Yu, X.; He, L. Magnetically recyclable cellulose-derived carbonaceous solid acid catalyzed the biofuel 5-ethoxymethylfurfural synthesis from renewable carbohydrates. Fuel 2018, 219, 344–352. [Google Scholar] [CrossRef]
- Guo, Y.; Rockstraw, D.A. Physical and chemical properties of carbons synthesized from xylan, cellulose, and Kraft lignin by H3PO4 activation. Carbon 2006, 44, 1464–1475. [Google Scholar] [CrossRef]
- Zhu, M.Q.; Wang, Z.W.; Wen, J.L.; Qiu, L.; Zhu, Y.H.; Su, Y.Q.; Wei, Q.; Sun, R.C. The effects of autohydrolysis pretreatment on the structural characteristics, adsorptive and catalytic properties of the activated carbon prepared from Eucommia ulmoides Oliver based on a biorefinery process. Bioresour. Technol. 2017, 232, 159–167. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.; Chen, X.; Shekiro, J.; Kuhn, E.; Wang, W.; Ji, Y.; Kozliak, E.; Himmel, M.E.; Tucker, M.P. Kinetic modelling and experimental studies for the effects of Fe2+ ions on xylan hydrolysis with dilute-acid pretreatment and subsequent enzymatic hydrolysis. Catalysts 2018, 8, 39–57. [Google Scholar] [CrossRef]
- Safari, A.; Karimi, K.; Shafiei, M. Dilute alkali pretreatment of softwood pine: A biorefinery approach. Bioresour. Technol. 2017, 234, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Shi, Y.; Zhou, G.; Xu, X.; Liu, E.; Zhou, Y.; Zhang, F.; Li, C.; Fang, H.; Chen, L. Structural development and carbon dynamics of Moso bamboo forests in Zhejiang Province, China. For. Ecol. Manag. 2018, 409, 479–488. [Google Scholar] [CrossRef]
- Sluiter, J.B.; Ruiz, R.O.; Scarlata, C.J.; Sluiter, A.D.; Templeton, D.W. Compositional analysis of lignocellulosic feedstocks: Review and description of methods. J. Agric. Food Chem. 2010, 58, 9043–9053. [Google Scholar] [CrossRef] [PubMed]
- Qi, X.; Watanabe, M.; Aida, T.M.; Smith, R.L., Jr. Sulfated zirconia as a solid acid catalyst for the dehydration of fructose to 5-hydroxymethylfurfural. Catal. Commun. 2009, 10, 1771–1775. [Google Scholar] [CrossRef]
- Li, H.; Deng, A.; Ren, J.; Liu, C.; Wang, W.; Peng, F.; Sun, R. A modified biphasic system for the dehydration of d-xylose into furfural using SO42−/TiO2−ZrO2/La3+ as a solid catalyst. Catal. Today 2014, 234, 251–256. [Google Scholar] [CrossRef]
- Hu, L.; Tang, X.; Wu, Z.; Lin, L.; Xu, J.; Xu, N.; Dai, B. Magnetic lignin-derived carbonaceous catalyst for the dehydration of fructose into 5-hydroxymethylfurfural in dimethylsulfoxide. Chem. Eng. J. 2015, 263, 299–308. [Google Scholar] [CrossRef]
- Jia, X.; Dai, R.; Sun, Y.; Song, H.; Wu, X. One-step hydrothermal synthesis of Fe3O4/g-C3N4 nanocomposites with improved photocatalytic activities. J. Mater. Sci. 2016, 27, 3791–3798. [Google Scholar] [CrossRef]
- Ishii, T.; Hiroi, T.; Thomas, J.R. Feruloylated xyloglucan and p-coumaroyl arabinoxylan oligosaccharides from bamboo shoot cell-walls. Phytochemistry 1990, 29, 1999–2003. [Google Scholar] [CrossRef]
- Xu, F.; Yu, J.; Tesso, T.; Dowell, F.; Wang, D. Qualitative and quantitative analysis of lignocellulosic biomass using infrared techniques: A mini-review. Appl. Energ. 2013, 104, 801–809. [Google Scholar] [CrossRef]
- Faix, O. Classification of lignins from different botanical origins by FT-IR Spectroscopy. Holzforschung 1991, 45, 21–28. [Google Scholar] [CrossRef]
- Li, J.; Li, K.; Zhang, T.; Wang, S.; Jiang, Y.; Bao, Y.; Tie, M. Development of activated carbon from windmill palm sheath fiber by KOH activation. Fiber. Polym. 2016, 17, 880–887. [Google Scholar] [CrossRef]
- He, C.; Sasaki, T.; Shimizu, Y.; Koshizaki, N. Synthesis of ZnO nanoparticles using nanosecond pulsed laser ablation in aqueous media and their self-assembly towards spindle-like ZnO aggregates. Appl. Surf. Sci. 2008, 254, 2196–2202. [Google Scholar] [CrossRef]
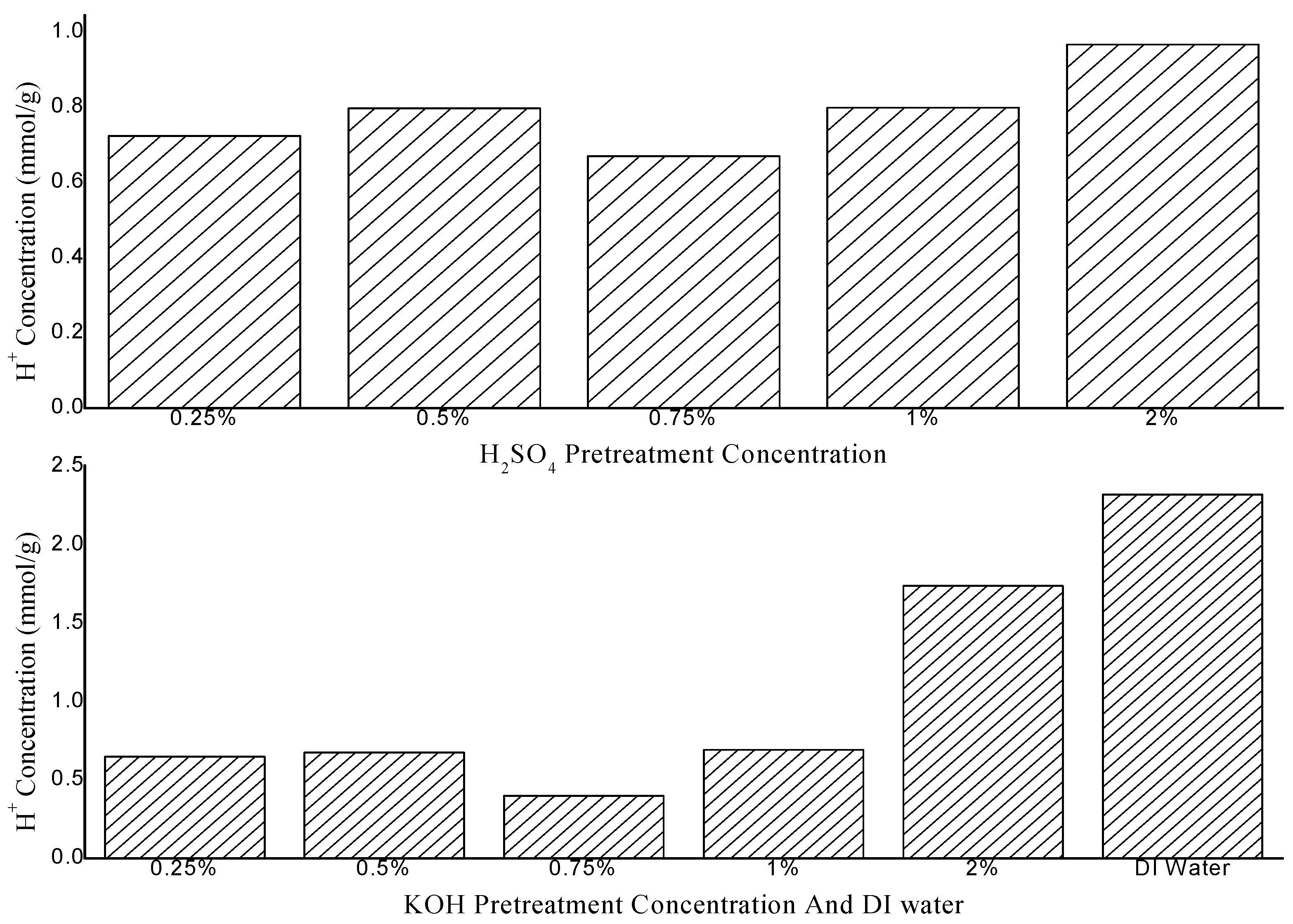
| Entry | Sample | Xylose | Arabinose | Glucose |
|---|---|---|---|---|
| 1 | 0.25% H2SO4 | 1995.35 | 510.50 | 166.85 |
| 2 | 0.5% H2SO4 | 2157.45 | 582.70 | 243.45 |
| 3 | 0.75% H2SO4 | 2805.00 | 605.05 | 266.75 |
| 4 | 1% H2SO4 | 6278.50 | 665.25 | 417.15 |
| 5 | 2% H2SO4 | 8258.90 | 711.00 | 613.30 |
| 6 | 0.25% KOH | ND a | ND | ND |
| 7 | 0.5% KOH | ND | ND | ND |
| 8 | 0.75% KOH | ND | ND | ND |
| 9 | 1% KOH | ND | ND | ND |
| 10 | 2% KOH | ND | ND | ND |
| 11 | DI Water | ND | ND | ND |
| Entry | Sample | Xylose | Arabinose | Glucose |
|---|---|---|---|---|
| 1 | 0.25% H2SO4 | 9.61 | 0.058 | 38.02 |
| 2 | 0.5% H2SO4 | 6.08 | 0.051 | 39.93 |
| 3 | 0.75% H2SO4 | 5.71 | ND a | 42.05 |
| 4 | 1% H2SO4 | 4.17 | ND | 44.81 |
| 5 | 2% H2SO4 | 4.83 | ND | 48.31 |
| 6 | 0.25% KOH | 9.69 | 0.69 | 40.61 |
| 7 | 0.5% KOH | 12.14 | 0.84 | 40.55 |
| 8 | 0.75% KOH | 11.79 | 0.88 | 43.14 |
| 9 | 1% KOH | 14.74 | 1.15 | 50.23 |
| 10 | 2% KOH | 12.05 | 0.74 | 51.82 |
| 11 | DI Water | 10.93 | 0.74 | 33.94 |
| Entry | Sample | Xylose | Arabinose | Glucose |
|---|---|---|---|---|
| 1 | 0.25% H2SO4 | 4643.40 | 662.80 | 1111.30 |
| 2 | 0.5% H2SO4 | 3100.05 | 492.15 | 1100.90 |
| 3 | 0.75% H2SO4 | 2284.05 | 360.20 | 683.6 |
| 4 | 1% H2SO4 | 3356.75 | 497.80 | 991.40 |
| 5 | 2% H2SO4 | 2259.40 | 318.40 | 219.50 |
| 6 | 0.25% KOH | 1856.20 | 278.40 | 672.50 |
| 7 | 0.5% KOH | 2642.85 | 366.55 | 668.50 |
| 8 | 0.75% KOH | 2358.30 | 320.90 | 562.90 |
| 9 | 1% KOH | 2034.75 | 302.4 | 850.80 |
| 10 | 2% KOH | 2601.00 | 402.70 | 1226.20 |
| 11 | DI Water | 286.90 | 25.55 | 386.45 |
| Entry | Sample | Xylose | Arabinose | Glucose |
|---|---|---|---|---|
| 1 | 0.25% H2SO4 | 22.35 | 0.00 | 1172.05 |
| 2 | 0.5% H2SO4 | 46.30 | 0.00 | 1288.85 |
| 3 | 0.75% H2SO4 | 205.85 | 0.10 | 874.35 |
| 4 | 1% H2SO4 | 39.20 | 0.00 | 1150.00 |
| 5 | 2% H2SO4 | 63.55 | 0.00 | 1193.85 |
| 6 | 0.25% KOH | 42.00 | 0.00 | 1038.50 |
| 7 | 0.5% KOH | 44.30 | 0.00 | 1073.10 |
| 8 | 0.75% KOH | 105.30 | 0.65 | 959.90 |
| 9 | 1% KOH | 197.15 | 0.50 | 817.05 |
| 10 | 2% KOH | 0.00 | 0.00 | 1004.10 |
| 11 | DI Water | 0.00 | 0.00 | 999.05 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhu, Y.; Huang, J.; Sun, S.; Wu, A.; Li, H. Effect of Dilute Acid and Alkali Pretreatments on the Catalytic Performance of Bamboo-Derived Carbonaceous Magnetic Solid Acid. Catalysts 2019, 9, 245. https://doi.org/10.3390/catal9030245
Zhu Y, Huang J, Sun S, Wu A, Li H. Effect of Dilute Acid and Alkali Pretreatments on the Catalytic Performance of Bamboo-Derived Carbonaceous Magnetic Solid Acid. Catalysts. 2019; 9(3):245. https://doi.org/10.3390/catal9030245
Chicago/Turabian StyleZhu, Yikui, Jiawei Huang, Shaolong Sun, Aimin Wu, and Huiling Li. 2019. "Effect of Dilute Acid and Alkali Pretreatments on the Catalytic Performance of Bamboo-Derived Carbonaceous Magnetic Solid Acid" Catalysts 9, no. 3: 245. https://doi.org/10.3390/catal9030245
APA StyleZhu, Y., Huang, J., Sun, S., Wu, A., & Li, H. (2019). Effect of Dilute Acid and Alkali Pretreatments on the Catalytic Performance of Bamboo-Derived Carbonaceous Magnetic Solid Acid. Catalysts, 9(3), 245. https://doi.org/10.3390/catal9030245





