Titanium and Vanadium Catalysts with 2-Hydroxyphenyloxazoline and Oxazine Ligands for Ethylene-Norbornene (co)Polymerization
Abstract
1. Introduction
2. Results and Discussion
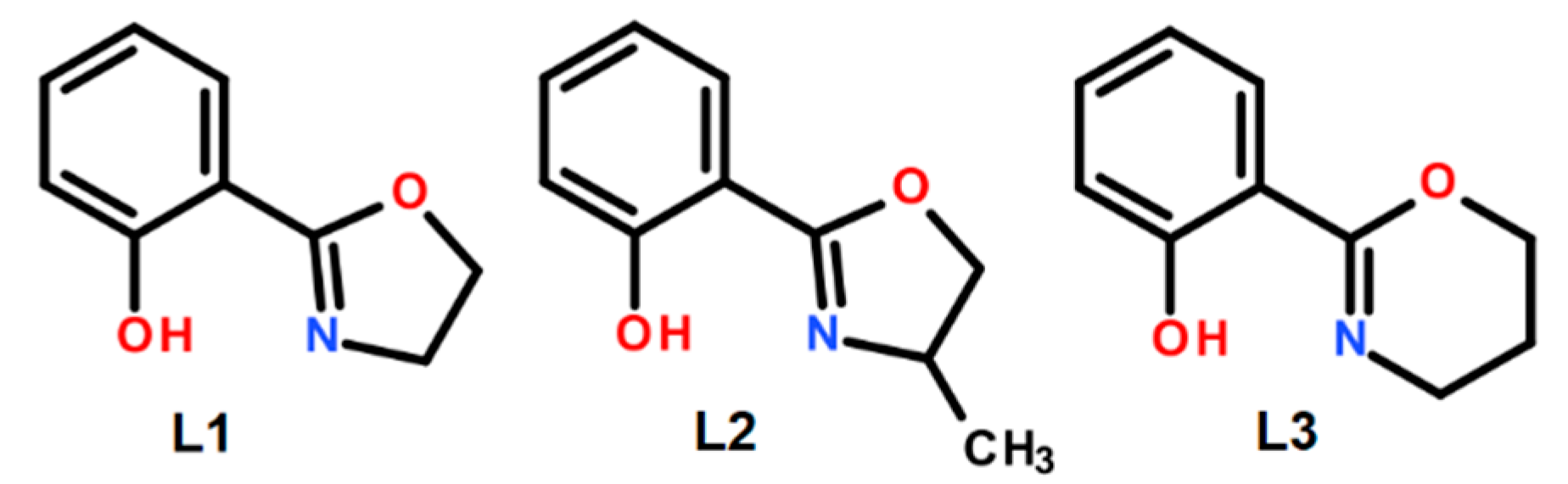
2.1. Synthesis and Analysis of the Catalysts
2.2. Ethylene Polymerization
2.3. Ethylene-Norbornene Copolymerization
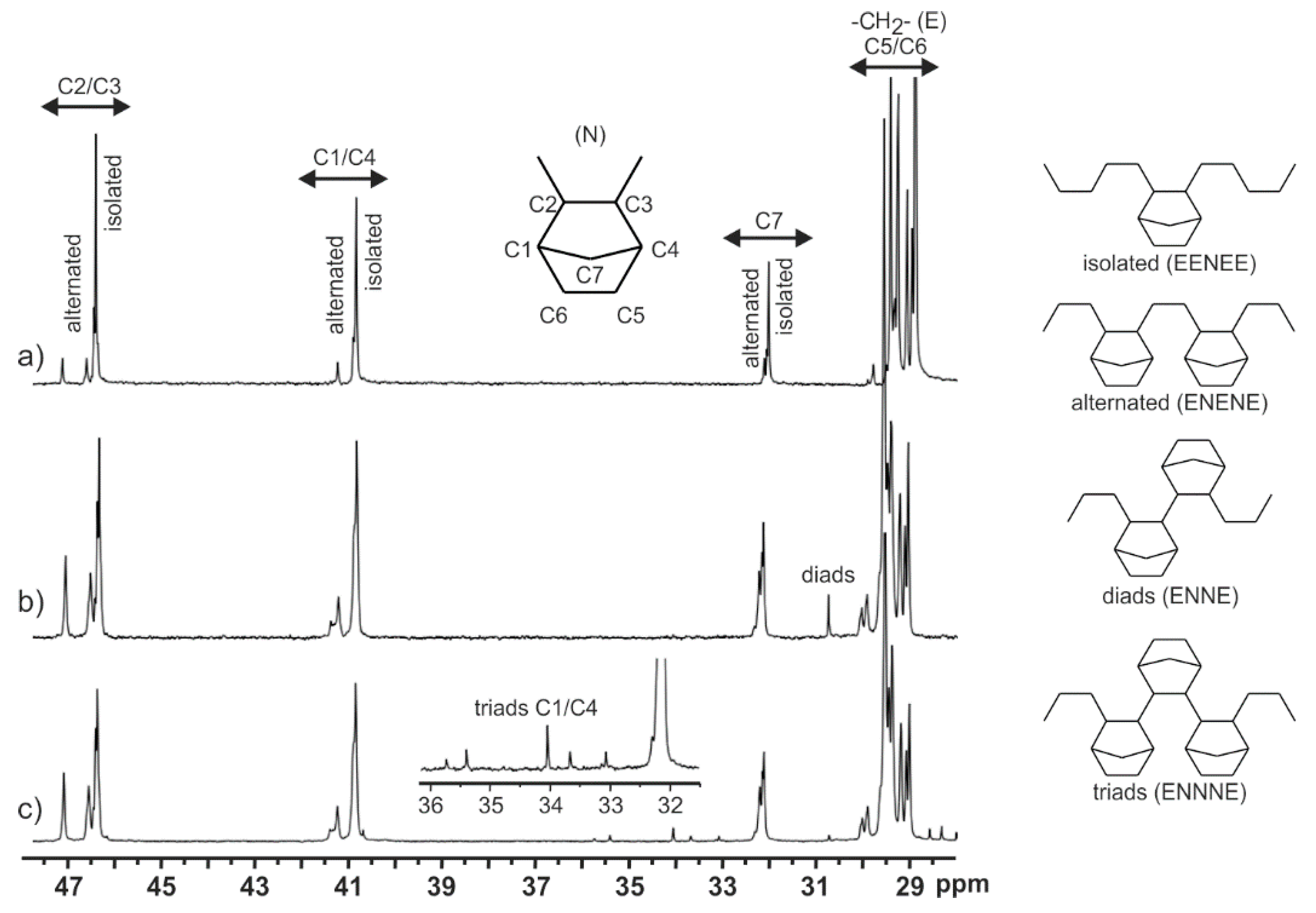
2.4. Norbornene Incorporation
3. Materials and Methods
3.1. Materials
3.2. Ligands Synthesis
3.3. Catalysts Preparation
3.4. (Co)Polymerization Procedure
3.5. Polymer Sample Purification
3.6. Instruments
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Forsyth, J.F.; Scrivani, T.; Benavente, R.; Marestin, C.; Perena, J.M. Thermal and dynamic mechanical behavior of ethylene/norbornene copolymers with medium norbornene contents. J. Appl. Polym. Sci. 2001, 82, 2159–2165. [Google Scholar] [CrossRef]
- Lamonte, R.R.; McNally, D. Cyclic olefin copolymers. Adv. Mater. Process. 2001, 3, 33–36. [Google Scholar]
- Nunes, S.P.; Ohlsson, P.D.; Ordeig, O.; Kutter, P.J. Cyclic olefin polymers: Emerging materials for lab-on-a-chip applications. Microfluid. Nanofluid. 2010, 9, 145–161. [Google Scholar] [CrossRef]
- Mitsui Chemicals America, INC. Available online: www.mitsuichemicals.com (accessed on 7 December 2019).
- TOPAS Advanced Polymers. Available online: www.topas.com/products-topas_coc (accessed on 7 December 2019).
- Tritto, I.; Boggioni, L.; Ferro, D.R. Metallocene catalyzed ethene- and propene co-norbornene polymerization: Mechanisms from a detailed microstructural analysis. Coord. Chem. Rev. 2006, 250, 212–241. [Google Scholar] [CrossRef]
- Blank, F.; Janiak, C. Metal catalysts for the vinyl/addition polymerization of norbornene. Coord. Chem. Rev. 2009, 253, 827–861. [Google Scholar] [CrossRef]
- Pei, L.; He, S.; Gao, J.; Liao, H.; Gao, H. Homo- and copolymerizations of ethylene and norbornene using bis(β-ketoamino) titanium catalysts containing pyrazolone rings. Polymers 2017, 9, 262. [Google Scholar] [CrossRef] [PubMed]
- Huo, P.; Liu, W.; He, X.; Wang, H.; Chen, Y. Nickel(II) complexes with three-dimensional geometry α-diimine ligands: Synthesis and catalytic activity toward copolymerization of norbornene. Organometallics 2013, 32, 2291–2298. [Google Scholar] [CrossRef]
- Manteghi, A.; Arabi, H.; Jahani, Y. Synthesis, characterization, rheological and thermal behavior of metallocene ethylene-norbornene copolymers with low norbornene content using pentafluorophenol modified methylaluminoxane. Polym. Int. 2015, 64, 900–906. [Google Scholar] [CrossRef]
- He, X.; Deng, Y.; Jiang, X.; Wang, Z.; Yang, Y.; Han, Z.; Chen, D. Copolymerization of norbornene and butyl methacrylate at elevated temperatures by a single centre nickel catalyst bearing bulky bis (α-diimine) ligand with strong electron-with drawing groups. Polym. Chem. 2017, 8, 2390–2396. [Google Scholar] [CrossRef]
- Zhao, W.; Nomura, K. Design of efficient molecular catalysts for synthesis of cyclic olefin copolymers (COC) by copolymerization of ethylene and α-olefins with norbornene or tetracyclododecene. Catalysts 2016, 6, 175. [Google Scholar] [CrossRef]
- Leone, G.; Pierro, I.; Zanchin, G.; Forni, A.; Bertini, F.; Rapallo, A.; Ricci, G. Vanadium(III)—Catalyzed copolymerization of ethylene with norbornene: Microstructure at tetrad level and reactivity ratios. J. Mol. Catal. A Chem. 2016, 424, 220–231. [Google Scholar] [CrossRef]
- Zanchin, G.; Gavezzoliy, A.; Bertini, F.; Ricci, G.; Leone, G. Homo- and Copolymerization of Ethylene with Norbornene Catalyzed by Vanadium(III) Phosphine Complexes. Molecules 2019, 24, 2088. [Google Scholar] [CrossRef] [PubMed]
- Zanchin, G.; Vendier, L.; Pierro, I.; Bertini, F.; Ricci, G.; Lorber, C.; Leone, G. Homo- and co-polymerization of ethylene with cyclic olefins catalyzed by phosphine adducts of (imido)vanadium(IV) complexes. Organometallics 2018, 37, 3181–3195. [Google Scholar] [CrossRef]
- Zanchin, G.; Pierro, I.; Parisini, E.; Martí-Rujas, J.; Ricci, G.; Leone, G. Synthesis, structure and behavior of vanadium(III) diphosphine complexes in the homo- and co-polymerization of ethylene with norbornene: The ligand donor strength and bite angle make the difference. J. Organomet. Chem. 2018, 861, 142–150. [Google Scholar] [CrossRef]
- Yan, Q.; Sun, Z.L.; Zhang, W.J.; Nomura, K.; Sun, W.H. Vanadyl Di (5-t-butyl-2-(aryliminomethyl)quinolin-8-olate): Synthesis, Characterization, and Ethylene (Co-)Polymerization. Macromol. Chem. Phys. 2014, 215, 1744–1752. [Google Scholar] [CrossRef]
- Ochędzan-Siodlak, W.; Bihun-Kisiel, A.; Siodlak, D.; Poliwoda, A.; Dziuk, B. Titanium and vanadium catalysts with oxazoline ligands for ethylene-norbornene (co)polymerization. Eur. Polym. J. 2018, 106, 148–155. [Google Scholar] [CrossRef]
- Boggioni, L.; Sidari, D.; Losio, S.; Stehling, U.M.; Auriemma, F.; Malafronte, A.; Girolamo, R.D.; Rosa, C.D.; Tritto, I. Ethylene-co-norbornene copolymerization using a dual catalyst system in the presence of a chain transfer agent. Polymers 2019, 11, 554. [Google Scholar] [CrossRef]
- Wang, H.; Cheng, H.; Tanaka, R.; Shiono, T.; Cai, Z. Efficient control of ethylene-norbornene copolymerization behavior of a fluorenylamido-ligated titanium complex: Substituent effects of the amido ligand and copolymer properties. Polym. Chem. 2018, 9, 4492–4497. [Google Scholar] [CrossRef]
- Wu, J.Q.; Li, Y.S. Well-defined vanadium complexes as the catalysts for olefin polymerization. Coord. Chem. Rev. 2011, 255, 2303–2314. [Google Scholar] [CrossRef]
- Langeslay, R.R.; Kaphan, D.M.; Marshall, C.L.; Stair, P.C.; Sattelberger, A.P.; Delferro, M. Catalytic applications of vanadium: A mechanistic perspective. Chem. Rev. 2019, 119, 2128–2191. [Google Scholar] [CrossRef]
- Budagumpi, S.; Keri, R.S.; Biffis, A.; Patil, S.A. Olefin poly/oligomerizations by metal precatalysts bearing non-heterocyclic N-donor ligands. Appl. Catal. A Gen. 2017, 535, 32–60. [Google Scholar] [CrossRef]
- Diteepeng, N.; Tang, X.; Hou, X.; Li, Y.S.; Phomphrai, K.; Nomura, K. Ethylene polymerisation and ethylene/norbornene copolymerisation by using aryloxo-modified vanadium(V) complexes containing 2,6-difluoro-, dichloro-phenylimido complexes. Dalton Trans. 2015, 44, 12273–12281. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Xiang, H.X.; Wang, K.T.; Wu, G.; Li, Y.B. Efficient preparation of cyclic olefin copolymers with unreacted double bonds by using thermal stable non-metallocene vanadium catalytic system. Macromol. Chem. Phys. 2019, 220, 1900008–1900017. [Google Scholar] [CrossRef]
- Wang, W.; Nomura, K. Notable effects of aluminum alkyls and solvents for highly efficient ethylene (co)polymerizations catalyzed by (arylimido)-(aryloxo)vanadium complexes. Adv. Synth. Catal. 2006, 348, 743–750. [Google Scholar] [CrossRef]
- Wu, J.Q.; Mu, J.S.; Zhang, S.W.; Li, Y.S. Vanadium(V) complexes containing tetradentate amine trihydroxy ligands as catalysts for copolymerization of cyclic olefins. J. Polym. Sci. A Polym. Chem. 2010, 48, 1122–1132. [Google Scholar] [CrossRef]
- Desimoni, G.; Faita, G.; Jorgensen, K.A. C2-Symmetric chiral bis (oxazoline) ligands in asymmetric catalysis. Chem. Rev. 2006, 106, 3561–3651. [Google Scholar] [CrossRef]
- Yang, G.; Zhang, W. Renaissance of pyridine-oxazolines as chiral ligands for asymmetric catalysis. Chem. Soc. Rev. 2018, 47, 1783–1810. [Google Scholar] [CrossRef]
- Cozzi, P.G.; Gallo, E.; Floriani, C.; Chiesi-Villa, A.; Rizzoli, C. (Hydroxyphenyl) oxazoline: A novel and remarkably facile entry into the area of chiral cationic alkylzirconium complexes which serve as polymerization catalysts. Organometallics 1995, 14, 4994–4996. [Google Scholar] [CrossRef]
- Cozzi, P.G.; Floriani, C.; Chiesi-Villa, A.; Rizzoli, C. Oxazoline early transition metal complexes: Functionalizable achiral titanium(IV), titanium(III), zirconium(IV), vanadium(III), and chiral zirconium(IV) bis(oxazoline) complexes. Inorg. Chem. 1995, 34, 2921–2930. [Google Scholar] [CrossRef]
- Makio, H.; Terao, H.; Iwashita, A.; Fujita, T. FI catalysts for olefin polymerization-a comprehensive treatment. Chem. Rev. 2011, 111, 2363–2449. [Google Scholar] [CrossRef]
- Ishii, S.; Nakano, T.; Kawamura, K.; Kinoshita, S.; Ichikawa, S.; Fujita, T. Development of new selective ethylene trimerization catalysts based on highly active ethylene polymerization catalysts. Catal. Today 2018, 303, 263–270. [Google Scholar] [CrossRef]
- Long, Y.Y.; Wang, Y.X.; Li, B.X.; Li, Y.G.; Li, Y.S. Quasi-living copolymerization of ethylene with 1-hexene by heteroligated (salicylaldiminato β-enaminoketonato) titanium complexes. J. Polym. Sci. A Polym. Chem. 2017, 55, 2787–2797. [Google Scholar] [CrossRef]
- Sarada, G.; Sim, B.; Cho, W.; Yoon, J.; Gal, Y.S.; Kim, J.J.; Jin, S.H. New sky-blue and bluishegreen emitting Ir(III) complexes containing an azoline ancillary ligand for highly efficient PhOLEDs. Dye. Pigment. 2016, 131, 60–68. [Google Scholar] [CrossRef]
- Traar, P.; Schachner, J.A.; Stanje, B.; Belaj, F.; Mösch-Zanetti, N.C. Dioxomolybdenum(VI) complexes with naphtholate-oxazoline ligands in catalytic epoxidation of olefins. J. Mol. Catal. A Chem. 2014, 385, 54–60. [Google Scholar] [CrossRef]
- Elagab, H.A.; Alt, H.G. Structure-property-relationship studies with ethylene polymerization catalysts of Ti, Zr and V containing heterocyclic ligands. Inorg. Chim. Acta 2015, 437, 26–35. [Google Scholar] [CrossRef]
- Wang, F.Z.; Tian, S.S.; Li, R.P.; Li, W.M.; Chen, C.L. Ligand steric effects on naphthyl-α-diimine nickel catalyzed α-olefin polymerization. Chin. J. Polym. Sci. 2018, 36, 157–162, (English Edition). [Google Scholar] [CrossRef]
- Dai, S.; Zhou, S.; Zhang, W.; Chen, C. Systematic investigations of ligand steric effects on α-diimine palladium catalyzed olefin polymerization and copolymerization. Macromolecules 2016, 49, 8855–8862. [Google Scholar] [CrossRef]
- Nomura, K.; Bahuleyan, B.K.; Zhang, S.; Sharma, P.M.V.; Katao, S.; Igarashi, A.; Inagaki, A.; Tamm, M. Synthesis and structural analysis of (imido)vanadium(V) dichloride complexes containing imidazolin-2-iminato- and imidazolidin-2-iminato ligands, and their use as catalyst precursors for ethylene (co)polymerization. Inorg. Chem. 2014, 53, 607–623. [Google Scholar] [CrossRef]
- Forsyth, J.; Perena, J.M.; Benavente, R.; Perez, E.; Tritto, I.; Boggioni, L.; Brintzinger, H.H. Influence of the polymer microstructure on the thermal properties of cycloolefin copolymers with high norbornene contents. Macromol. Chem. Phys. 2001, 202, 614–620. [Google Scholar] [CrossRef]
- Boggioni, L.; Losio, S.; Tritto, I. Microstructure of copolymers of norbornene based on assignments of 13C NMR spectra: Evolution of a methodology. Polymers 2018, 10, 647. [Google Scholar] [CrossRef]
- Krasuska, A.; Białek, M.; Czaja, K. Ethylene polymerization with FI complexes having novel phenoxy-imine ligands: Effect of metal type and complex immobilization. J. Polym. Sci. A Polym. Chem. 2011, 49, 1644–1654. [Google Scholar] [CrossRef]
- Białek, M.; Bisz, E. Polypropylene and poly (ethylene-co-1-octene) effective synthesis with diamine-bis (phenolate) complexes: Effect of complex structure on catalyst activity and product microstructure. Polym. Sci. A Polym. Chem. 1017, 55, 2467–2476. [Google Scholar] [CrossRef]
| Item | Catalyst | Al/Mt a | Yield (g) | Activity b (kg PE/molMt) | Tm (°C) | X DSC (%) |
|---|---|---|---|---|---|---|
| 1 | (L1)2TiCl2 | 1500 | 0.68 | 136 | 118.3 | 48.2 |
| 2 | 1000 | 0.54 | 108 | 125.8 | 53.3 | |
| 3 | (L1)2VCl2 | 1500 | 5.37 | 1074 | 131.2 | 65.4 |
| 4 | 1000 | 6.02 | 1204 | 140.2 | 71.2 | |
| 5 c | 1000 | 18.25 | 7300 | 141.7 | 74.9 | |
| 6 | (L2)2TiCl2 | 1500 | 0.34 | 68 | 111.9 | nd |
| 7 | 1000 | 0.20 | 40 | 120.8 | 49.0 | |
| 8 | (L2)2VCl2 | 1500 | 2.49 | 498 | 123.5 | 48.9 |
| 9 | 1000 | 3.30 | 660 | 132.6 | 66.3 | |
| 10 c,d | 1000 | 11.26 | 4504 | 140.0 | 72.5 | |
| 11 | (L3)2TiCl2 | 1500 | 0.42 | 84 | 119.4 | nd |
| 12 | 1000 | 0.39 | 72 | 122.6 | 68.2 | |
| 13 | (L3)2VCl2 | 1500 | 3.16 | 632 | 138.2 | 64.8 |
| 14 c,d | 1500 | 9.24 | 3696 | 132.6 | 58.5 | |
| 15 | 1000 | 4.21 | 842 | 140.6 | 72.0 | |
| 16 c,d | 1000 | 14.45 | 5780 | 135.9 | 71.9 |
| Item | Catalyst | NB (mol/L) | Yield (g) | Activity a | Mw/105 (g/mol) b | Mw/Mn b | Tg (°C) | NB c (mol%) |
|---|---|---|---|---|---|---|---|---|
| 1 | (L1)2VCl2 | 0.5 | 20.81 | 4162 | 2.6 | 1.6 | 4.0 | 12.3 |
| 2 | 1.0 | 25.70 | 5140 | 2.0 | 1.8 | 34.2 | 22.1 | |
| 3 | 1.5 | 26.48 | 5296 | 1.8 | 1.9 | 54.0 | 27.5 | |
| 4 | (L2)2VCl2 | 0.5 | 19.10 | 3820 | 1.6 | 1.7 | 12.8 | 17.1 |
| 5 | 1.0 | 17.22 | 3444 | 1.3 | 1.9 | 27.2 | 20.7 | |
| 6 | 1.5 | 8.10 | 1620 | 0.9 | 2.2 | 52.9 | 27.2 | |
| 7 | (L3)2VCl2 | 0.5 | 17.71 | 3542 | 1.9 | 1.7 | 22.1 | 19.4 |
| 8 d | 0.5 | 13.58 | 2716 | 1.8 | 1.7 | 14.6 | 17.5 | |
| 9 | 1.0 | 23.88 | 4776 | 1.7 | 1.7 | 53.9 | 26.5 | |
| 10 d | 1.0 | 19.67 | 3934 | 1.5 | 1.7 | 43.8 | 21.8 | |
| 11 | 1.5 | 20.32 | 4064 | 1.6 | 1.9 | 61.7 | 30.1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ochędzan-Siodłak, W.; Siodłak, D.; Piontek, A.; Doležal, K. Titanium and Vanadium Catalysts with 2-Hydroxyphenyloxazoline and Oxazine Ligands for Ethylene-Norbornene (co)Polymerization. Catalysts 2019, 9, 1041. https://doi.org/10.3390/catal9121041
Ochędzan-Siodłak W, Siodłak D, Piontek A, Doležal K. Titanium and Vanadium Catalysts with 2-Hydroxyphenyloxazoline and Oxazine Ligands for Ethylene-Norbornene (co)Polymerization. Catalysts. 2019; 9(12):1041. https://doi.org/10.3390/catal9121041
Chicago/Turabian StyleOchędzan-Siodłak, Wioletta, Dawid Siodłak, Aleksandra Piontek, and Karel Doležal. 2019. "Titanium and Vanadium Catalysts with 2-Hydroxyphenyloxazoline and Oxazine Ligands for Ethylene-Norbornene (co)Polymerization" Catalysts 9, no. 12: 1041. https://doi.org/10.3390/catal9121041
APA StyleOchędzan-Siodłak, W., Siodłak, D., Piontek, A., & Doležal, K. (2019). Titanium and Vanadium Catalysts with 2-Hydroxyphenyloxazoline and Oxazine Ligands for Ethylene-Norbornene (co)Polymerization. Catalysts, 9(12), 1041. https://doi.org/10.3390/catal9121041







