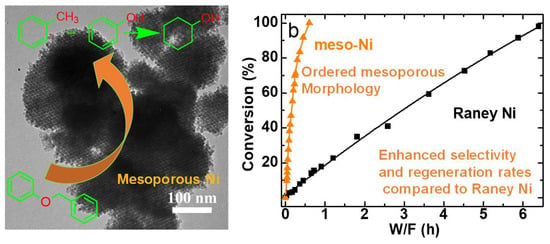
Enhanced Selective Production of Arenes and Regenerating Rate in Aryl Ether Hydrogenolysis over Mesoporous Nickel in Plug-Flow Reactors
Abstract
1. Introduction
2. Results and Discussion
2.1. Characterizations of the Catalysts
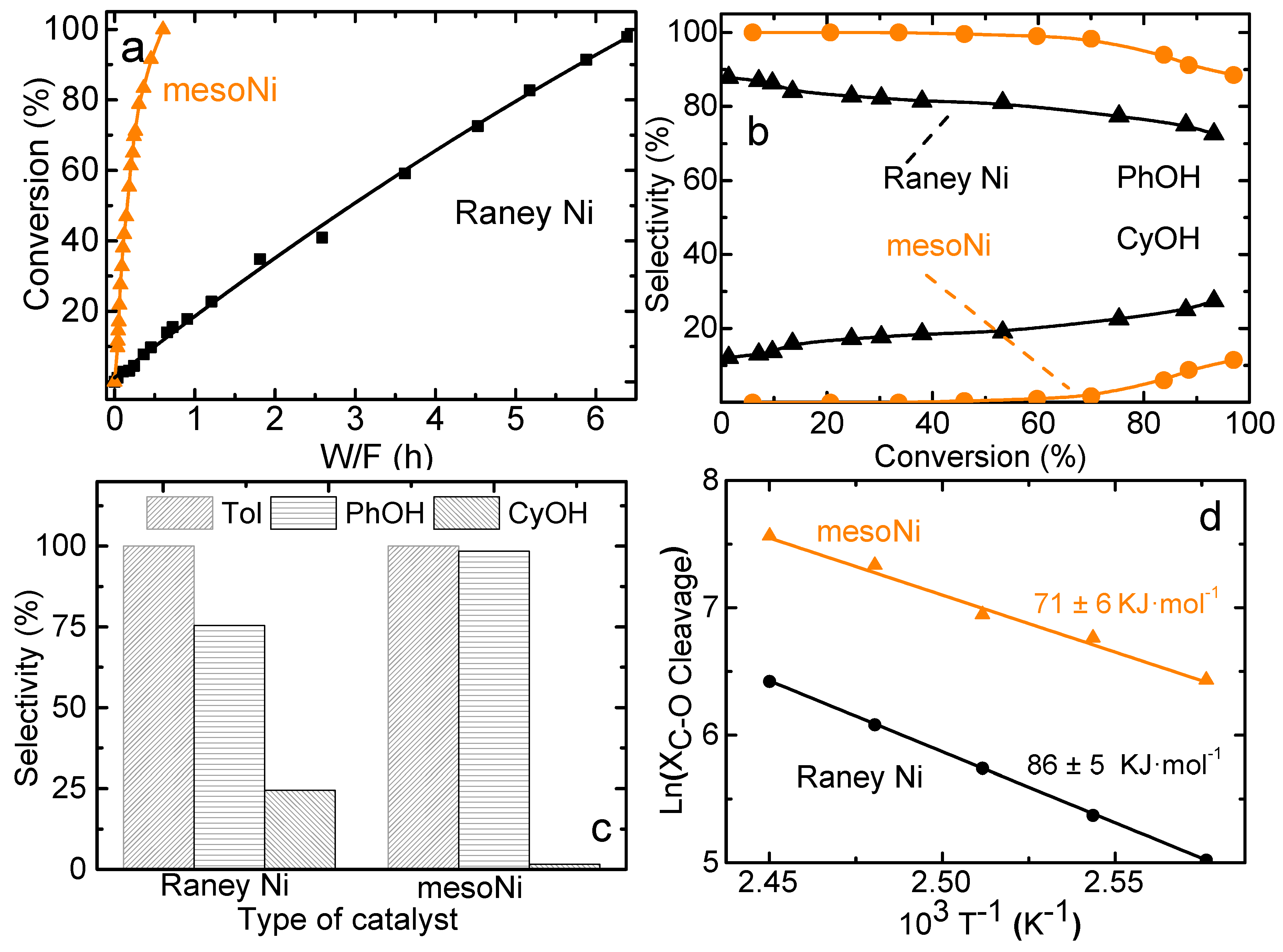
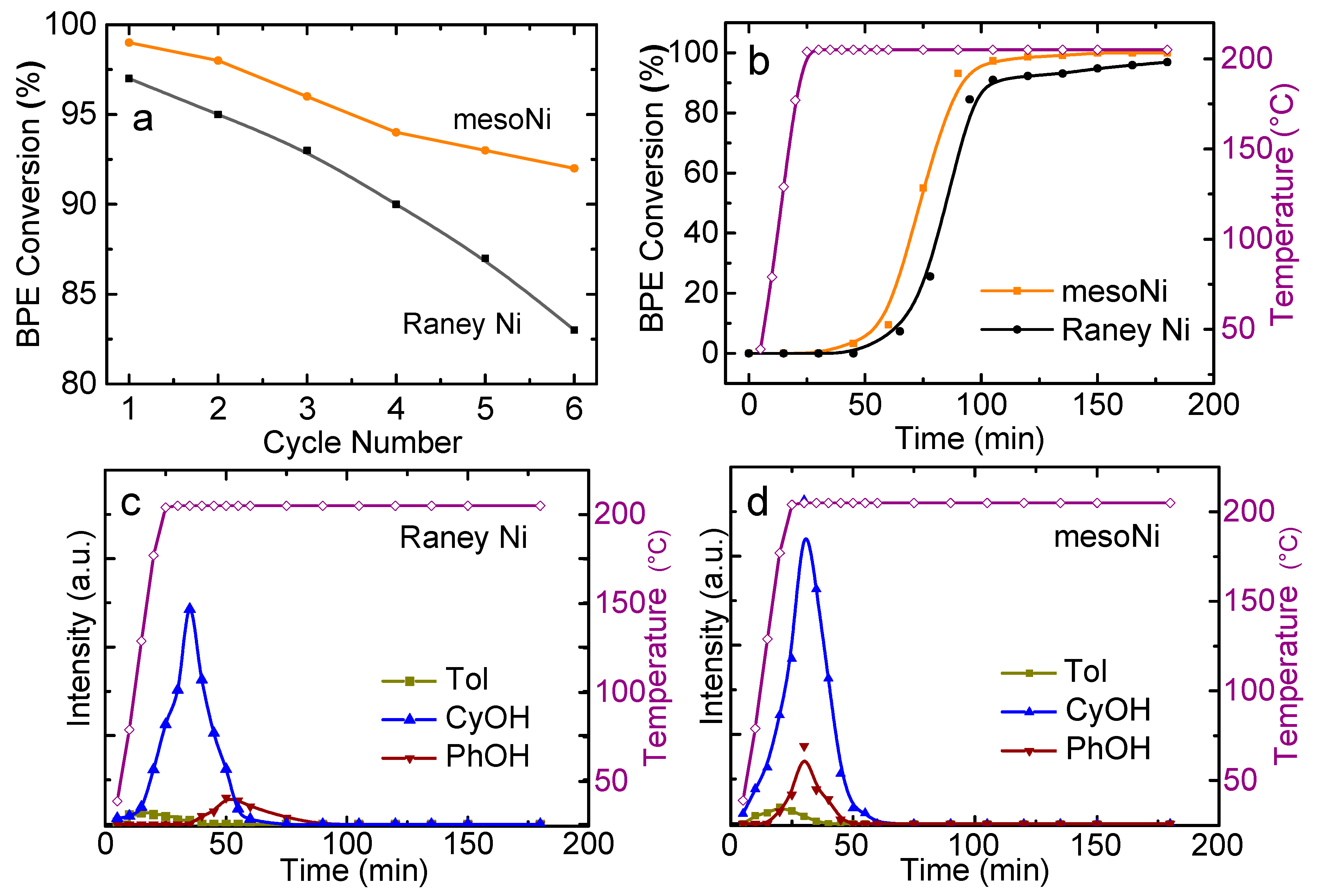
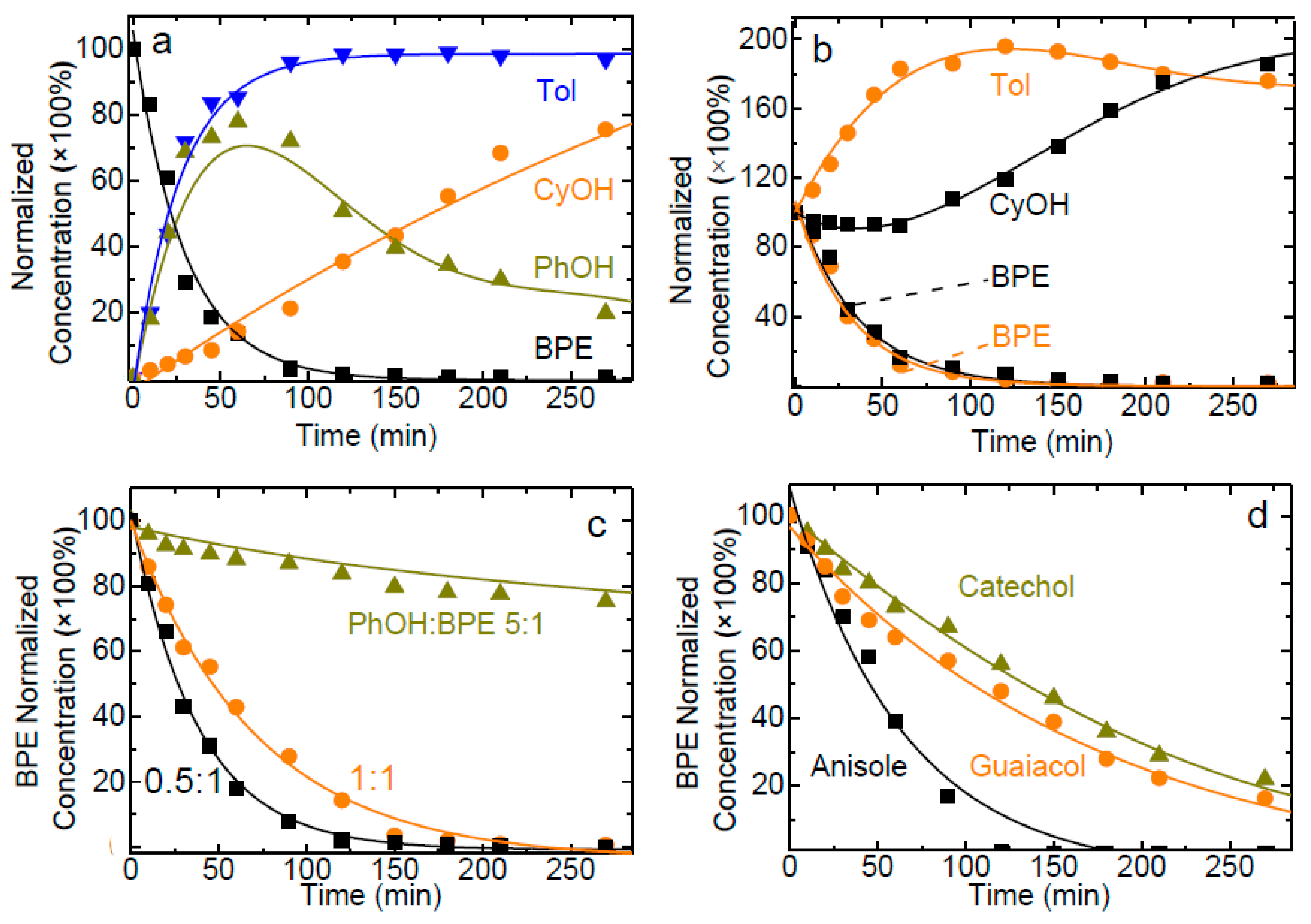
2.2. Catalytic Hydrogenolysis of BPE
3. Materials and Methods
3.1. Preparation of Catalysts
3.2. Hydrogenolysis of BPE
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Deuss, P.J.; Barta, K. From models to lignin: Transition metal catalysis for selective bond cleavage reactions. Coord. Chem. Rev. 2015, 306, 510–532. [Google Scholar] [CrossRef]
- Joseph, Z.; Bruijnincx, P.C.A.; Jongerius, A.L.; Weckhuysen, B.M. The catalytic valorization of lignin for the production of renewable chemicals. Chem. Rev. 2013, 110, 3552–3599. [Google Scholar]
- Carrott, P.J.M.S.; Carrott, M.M.L.R. Lignin—From natural adsorbent to activated carbon: A review. Bioresour. Technol. 2007, 98, 2301–2312. [Google Scholar]
- Sun, Z.; Fridrich, B.; de Santi, A.; Elangovan, S.; Barta, K. Bright side of lignin depolymerization: Toward new platform chemicals. Chem. Rev. 2018, 118, 614–678. [Google Scholar] [CrossRef] [PubMed]
- Upton, B.M.; Kasko, A.M. Strategies for the conversion of lignin to high-value polymeric materials: Review and perspective. Chem. Rev. 2016, 116, 2275–2306. [Google Scholar] [CrossRef] [PubMed]
- Nichols, J.M.; Bishop, L.M.; Bergman, R.G.; Ellman, J.A. Catalytic c-o bond cleavage of 2-aryloxy-1-arylethanols and its application to the depolymerization of lignin-related polymers. J. Am. Chem. Soc. 2010, 132, 12554–12555. [Google Scholar] [CrossRef] [PubMed]
- Son, S.; Toste, F.D. Non-oxidative vanadium-catalyzed c–o bond cleavage: Application to degradation of lignin model compounds. Angew. Chem. Int. Ed. Engl. 2010, 49, 3791–3794. [Google Scholar] [CrossRef]
- Sergeev, A.G.; Hartwig, J.F. Selective, nickel-catalyzed hydrogenolysis of aryl ethers. Science 2011, 42, 439. [Google Scholar] [CrossRef]
- Jiayue, H.; Chen, Z.; Lercher, J.A. Ni-catalyzed cleavage of aryl ethers in the aqueous phase. J. Am. Chem. Soc. 2012, 134, 20768. [Google Scholar]
- Parsell, T.H.; Owen, B.C.; Klein, I.; Jarrell, T.M.; Marcum, C.L.; Haupert, L.J.; Amundson, L.M.; Kenttämaa, H.I.; Ribeiro, F.; Miller, J.T.; et al. Cleavage and hydrodeoxygenation (hdo) of c–o bonds relevant to lignin conversion using pd/zn synergistic catalysis. Chem. Sci. 2013, 4, 806–813. [Google Scholar] [CrossRef]
- Shao, Y.; Xia, Q.; Dong, L.; Liu, X.; Han, X.; Parker, S.F.; Cheng, Y.; Daemen, L.L.; Ramirezcuesta, A.J.; Yang, S. Selective production of arenes via direct lignin upgrading over a niobium-based catalyst. Nat. Commun. 2017, 8, 16104. [Google Scholar] [CrossRef] [PubMed]
- Schutyser, W.; Renders, T.; Van den Bosch, S.; Koelewijn, S.F.; Beckham, G.T.; Sels, B.F. Chemicals from lignin: An interplay of lignocellulose fractionation, depolymerisation, and upgrading. Chem. Soc. Rev. 2018, 47, 852–908. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Zhao, X.; Wang, A.; Huber, G.W.; Zhang, T. Catalytic transformation of lignin for the production of chemicals and fuels. Chem. Rev. 2015, 115, 11559–11624. [Google Scholar] [CrossRef] [PubMed]
- Lama, S.M.G.; Pampel, J.; Fellinger, T.-P.; Beskoski, V.P.; Slavkovic-Beskoski, L.; Antonietti, M.; Molinari, V. Efficiency of ni nanoparticles supported on hierarchical porous nitrogen-doped carbon for hydrogenolysis of kraft lignin in flow and batch systems. ACS Sustain. Chem. Eng. 2017, 5, 2415–2420. [Google Scholar] [CrossRef]
- Anderson, E.M.; Stone, M.L.; Hulsey, M.J.; Beckham, G.T.; Roman-Leshkov, Y. Kinetic studies of lignin solvolysis and reduction by reductive catalytic fractionation decoupled in flow-through reactors. ACS Sustain. Chem. Eng. 2018, 6, 7951–7959. [Google Scholar] [CrossRef]
- Yang, S.; Jeong, S.; Ban, C.; Kim, H.; Kim, D.H. Catalytic cleavage of ether bond in a lignin model compound over carbon-supported noble metal catalysts in supercritical ethanol. Catalysts 2019, 9. [Google Scholar] [CrossRef]
- Molinari, V.; Clavel, G.; Graglia, M.; Antonietti, M.; Esposito, D. Mild continuous hydrogenolysis of kraft lignin over titanium nitride-nickel catalyst. ACS Catal. 2016, 6, 1663–1670. [Google Scholar] [CrossRef]
- Chen, X.; Guan, W.; Tsang, C.-W.; Hu, H.; Liang, C. Lignin valorizations with ni catalysts for renewable chemicals and fuels productions. Catalysts 2019, 9. [Google Scholar] [CrossRef]
- Kumaniaev, I.; Subbotina, E.; Sävmarker, J.; Larhed, M.; Galkin, M.V.; Samec, J. Lignin depolymerization to monophenolic compounds in a flow-through system. Green Chem. 2017, 19. [Google Scholar] [CrossRef]
- Yang, F.; Liu, D.; Zhao, Y.; Wang, H.; Han, J.; Ge, Q.; Zhu, X. Size dependence of vapor phase hydrodeoxygenation of m-cresol on ni/sio2 catalysts. ACS Catal. 2018, 8, 1672–1682. [Google Scholar] [CrossRef]
- Qi, S.-C.; Zhang, L.; Einaga, H.; Kudo, S.; Norinaga, K.; Hayashi, J.-i. Nano-sized nickel catalyst for deep hydrogenation of lignin monomers and first-principles insight into the catalyst preparation. J. Mater. Chem. A 2017, 5, 3948–3965. [Google Scholar] [CrossRef]
- Verhaak, M.J.F.M.; Vandillen, A.J.; Geus, J.W. The deactivation of nickel catalysts in the hydrogenation of acetonitrile. J. Catal. 1993, 143, 187–200. [Google Scholar] [CrossRef]
- Baiker, A.; Monti, D.; Fan, Y.S. Deactivation of copper, nickel, and cobalt catalysts by interaction with aliphatic amines. J. Catal. 1984, 88, 81–88. [Google Scholar] [CrossRef]
- Sun, X.; Hao, H.; Ji, H.; Li, X.; Cai, S.; Zheng, C. Nanocasting synthesis of in2o3 with appropriate mesostructured ordering and enhanced gas-sensing property. ACS Appl. Mater. Interfaces 2014, 6, 401–409. [Google Scholar] [CrossRef]
- Sakamoto, Y.; Kim, T.W.; Ryoo, R.; Terasaki, O. Three-dimensional structure of large-pore mesoporous cubic ia(3)over-bard silica with complementary pores and its carbon replica by electron crystallography. Angew. Chem.-Int. Ed. 2004, 43, 5231–5234. [Google Scholar] [CrossRef]
- Hafizi, H.; Chermahini, A.N.; Saraji, M.; Mohammadnezhad, G. The catalytic conversion of fructose into 5-hydroxymethylfurfural over acid-functionalized kit-6, an ordered mesoporous silica. Chem. Eng. J. 2016, 294, 380–388. [Google Scholar] [CrossRef]
- Wang, X.; Rinaldi, R. Solvent effects on the hydrogenolysis of diphenyl ether with raney nickel and their implications for the conversion of lignin. ChemSusChem 2012, 5, 1455–1466. [Google Scholar] [CrossRef]
- Kleitz, F.; Choi, S.H.; Ryoo, R. Cubic ia3d large mesoporous silica: Synthesis and replication to platinum nanowires, carbon nanorods and carbon nanotubes. Chem. Commun. 2003, 2136–2137. [Google Scholar] [CrossRef]
- Chen, P.; Zhang, Q.; Shu, R.; Xu, Y.; Ma, L.; Wang, T. Catalytic depolymerization of the hydrolyzed lignin over mesoporous catalysts. Bioresour. Technol. 2017, 226, 125–131. [Google Scholar] [CrossRef]
- Sun, X.; Shi, Y.; Zhang, P.; Zheng, C.; Zheng, X.; Zhang, F.; Zhang, Y.; Guan, N.; Zhao, D.; Stucky, G.D. Container effect in nanocasting synthesis of mesoporous metal oxides. J. Am. Chem. Soc. 2011, 133, 14542–14545. [Google Scholar] [CrossRef]
- Zaheer, M.; Hermannsdoerfer, J.; Kretschmer, W.P.; Motz, G.; Kempe, R. Robust heterogeneous nickel catalysts with tailored porosity for the selective hydrogenolysis of aryl ethers. ChemCatChem 2014, 6, 91–95. [Google Scholar] [CrossRef]
- Resende, K.A.; Hori, C.E.; Noronha, F.B.; Shi, H.; Gutierrez, O.Y.; Camaioni, D.M.; Lercher, J.A. Aqueous phase hydrogenation of phenol catalyzed by pd and pdag on zro2. Appl. Catal. A-Gen. 2017, 548, 128–135. [Google Scholar] [CrossRef]
- Paone, E.; Espro, C.; Pietropaolo, R.; Mauriello, F. Selective arene production from transfer hydrogenolysis of benzyl phenyl ether promoted by a co-precipitated pd/fe3o4 catalyst. Catal. Sci. Technol. 2016, 6, 7937–7941. [Google Scholar] [CrossRef]
- Sawhney, B.L. Vapor-phase sorption and polymerization of phenols by smectite in air and nitrogen. Clay Clay Miner. 1985, 33, 123–127. [Google Scholar] [CrossRef]
- Luo, Z.; Zheng, Z.; Wang, Y.; Sun, G.; Jiang, H.; Zhao, C. Hydrothermally stable ru/hzsm-5-catalyzed selective hydrogenolysis of lignin-derived substituted phenols to bio-arenes in water. Green Chem. 2016, 18, 5845–5858. [Google Scholar] [CrossRef]
- Li, K.; Wang, R.; Chen, J. Hydrodeoxygenation of anisole over silica-supported ni2p, mop, and nimop catalysts. Energ Fuel 2011, 25, 854–863. [Google Scholar] [CrossRef]
- Walter, E.D.; Qi, L.; Chamas, A.; Mehta, H.S.; Sears, J.A.; Scott, S.L.; Hoyt, D.W. Operando mas nmr reaction studies at high temperatures and pressures. J. Phys. Chem. C 2018, 122, 8209–8215. [Google Scholar] [CrossRef]
- Toledano, A.; Serrano, L.; Pineda, A.; Romero, A.A.; Luque, R.; Labidi, J. Microwave-assisted depolymerisation of organosolv lignin via mild hydrogen-free hydrogenolysis: Catalyst screening. Appl. Catal. B-Environ. 2014, 145, 43–55. [Google Scholar] [CrossRef]
- Mauriello, F.; Paone, E.; Pietropaolo, R.; Balu, A.M.; Luque, R. Catalytic transfer hydrogenolysis of lignin-derived aromatic ethers promoted by bimetallic pd/ni systems. ACS Sustain. Chem. Eng. 2018, 6, 9269–9276. [Google Scholar] [CrossRef]
- Friend, C.M.; Muetterties, E.L. Coordination chemistry of metal surfaces. 3. Benzene and toluene interactions with nickel surfaces. J. Am. Chem. Soc. 1981, 103, 773–779. [Google Scholar] [CrossRef][Green Version]
- Xia, J.; He, G.; Zhang, L.; Sun, X.; Wang, X. Hydrogenation of nitrophenols catalyzed by carbon black-supported nickel nanoparticles under mild conditions. Appl. Catal. B-Environ. 2016, 180, 408–415. [Google Scholar] [CrossRef]
- Luc, W.; Jiao, F. Synthesis of nanoporous metals, oxides, carbides, and sulfides: Beyond nanocasting. Acc. Chem. Res. 2016, 49, 1351–1358. [Google Scholar] [CrossRef] [PubMed]
- Tian, C.; Li, J.; Ma, C.; Wang, P.; Sun, X.; Fang, J. An ordered mesoporous ag superstructure synthesized via a template strategy for surface-enhanced raman spectroscopy. Nanoscale 2015, 7, 12318–12324. [Google Scholar] [CrossRef] [PubMed]
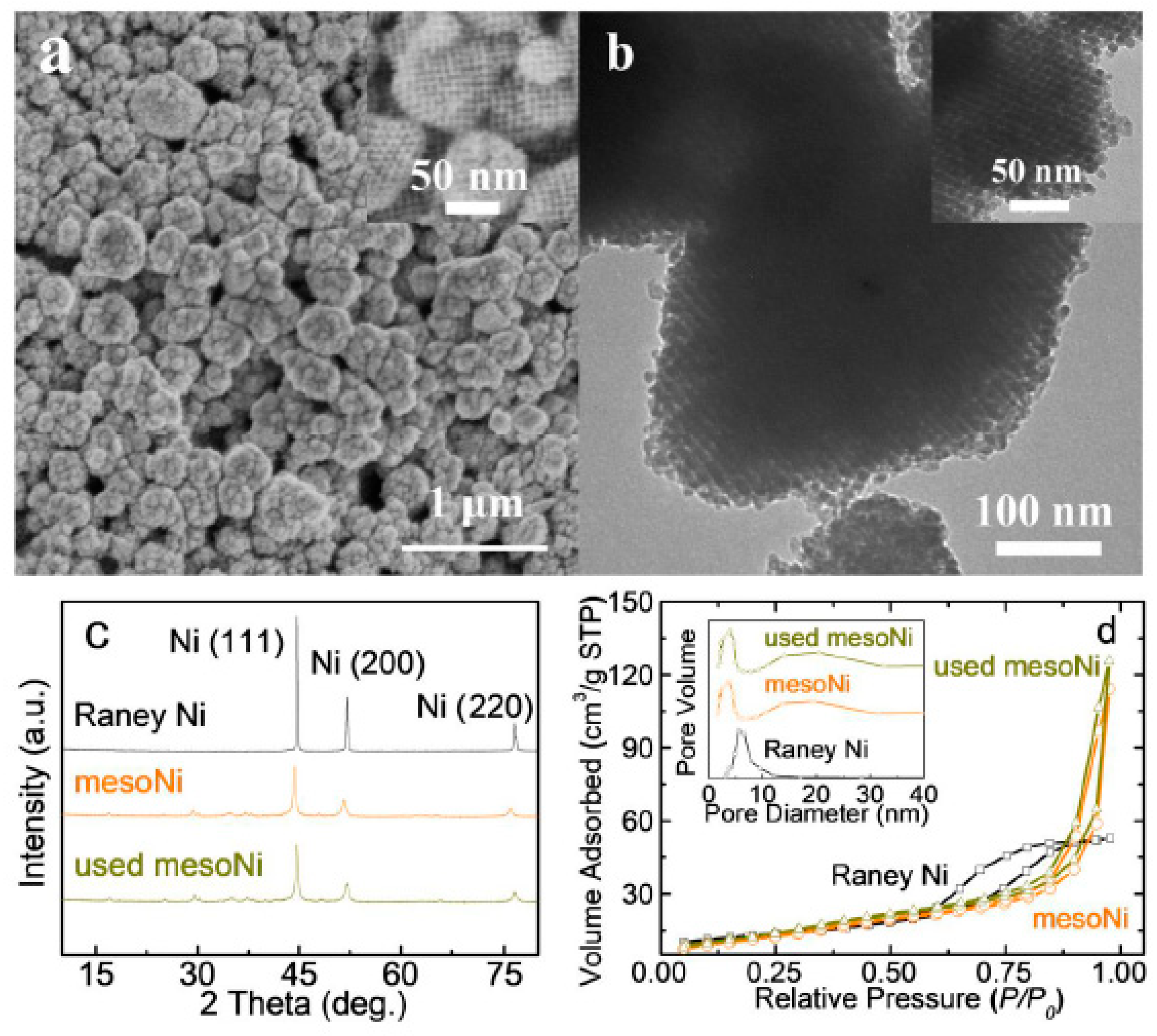
| Catalysts | SBET a (m2·g−1) | Dp b (nm) | Pore Volume (cm3·g−1) | dXRD (nm) | dTEM (nm) | Na c wt% | Si wt% | Ni Leaching d (mg·L−1) |
|---|---|---|---|---|---|---|---|---|
| MesoNi | 84.2 | 8.9 | 0.191 | 13.7 | 268 ± 64 | 0.02 | 0.13 | <0.1 |
| Raney Ni | 46.3 | 5.6 | 0.082 | 19.6 | 576 ± 83 | N/D | N/D | <0.1 |
| Recycled mesoNi | 87.7 | 9.9 | 0.217 | 16.2 | 293 ± 73 | 0.01 | 0.07 | <0.1 |
| KIT-6 template | 803 | 2.3 | 0.602 | - | - | - | - | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zheng, C.; Wang, D.; Hu, X.; Ma, C.; Liu, X.; Wang, Y.; Yu, Y.; Wang, J.; Sun, X. Enhanced Selective Production of Arenes and Regenerating Rate in Aryl Ether Hydrogenolysis over Mesoporous Nickel in Plug-Flow Reactors. Catalysts 2019, 9, 904. https://doi.org/10.3390/catal9110904
Zheng C, Wang D, Hu X, Ma C, Liu X, Wang Y, Yu Y, Wang J, Sun X. Enhanced Selective Production of Arenes and Regenerating Rate in Aryl Ether Hydrogenolysis over Mesoporous Nickel in Plug-Flow Reactors. Catalysts. 2019; 9(11):904. https://doi.org/10.3390/catal9110904
Chicago/Turabian StyleZheng, Chunming, Dongxue Wang, Xudong Hu, Chao Ma, Xuan Liu, Ying Wang, Yinkui Yu, Jiaqi Wang, and Xiaohong Sun. 2019. "Enhanced Selective Production of Arenes and Regenerating Rate in Aryl Ether Hydrogenolysis over Mesoporous Nickel in Plug-Flow Reactors" Catalysts 9, no. 11: 904. https://doi.org/10.3390/catal9110904
APA StyleZheng, C., Wang, D., Hu, X., Ma, C., Liu, X., Wang, Y., Yu, Y., Wang, J., & Sun, X. (2019). Enhanced Selective Production of Arenes and Regenerating Rate in Aryl Ether Hydrogenolysis over Mesoporous Nickel in Plug-Flow Reactors. Catalysts, 9(11), 904. https://doi.org/10.3390/catal9110904