Influence of the Brønsted Acidity on the Ring Opening of Decalin for Pt-USY Catalysts
Abstract
:1. Introduction
2. Results and Discussion
2.1. Characterization of Catalysts
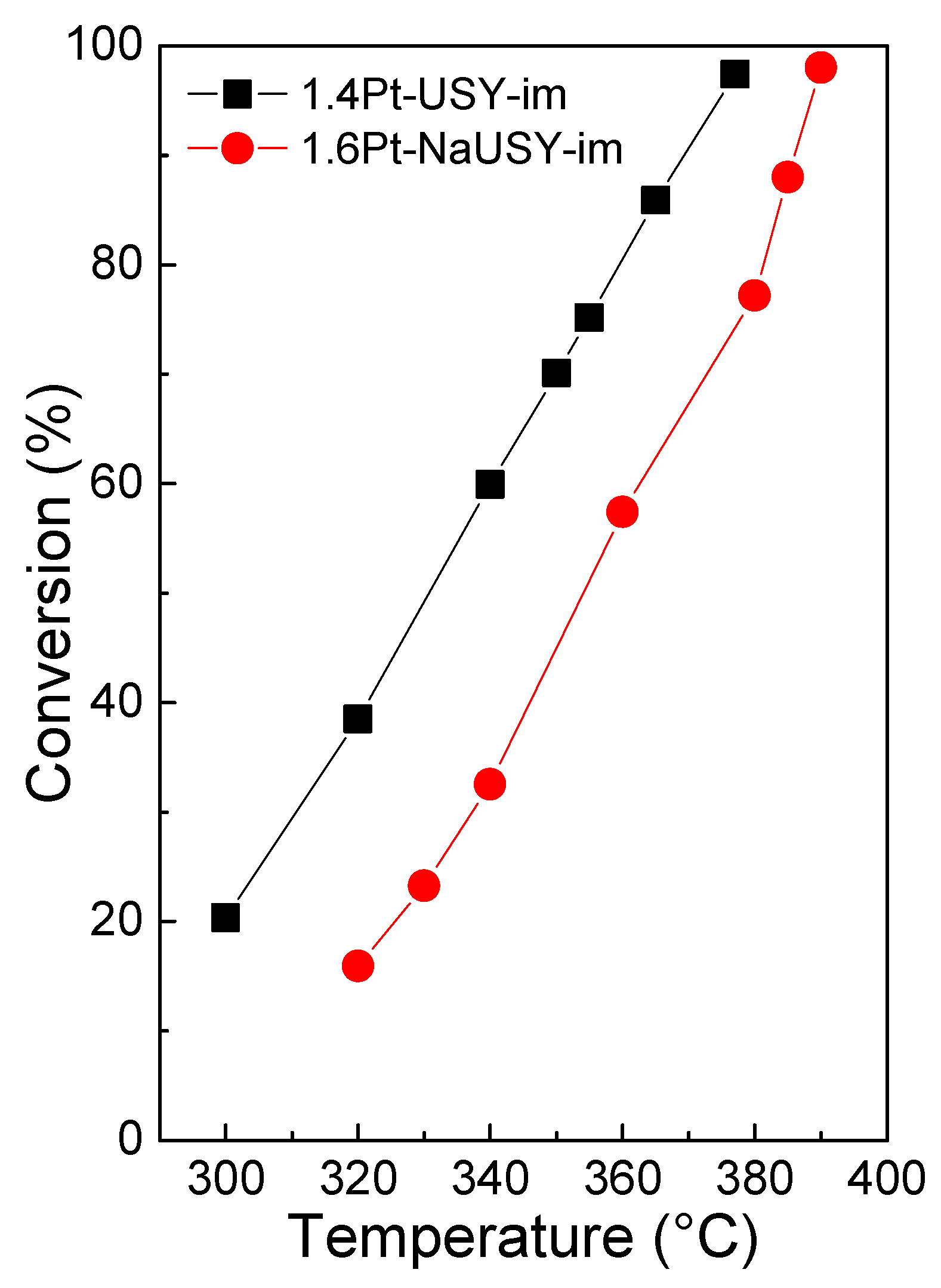
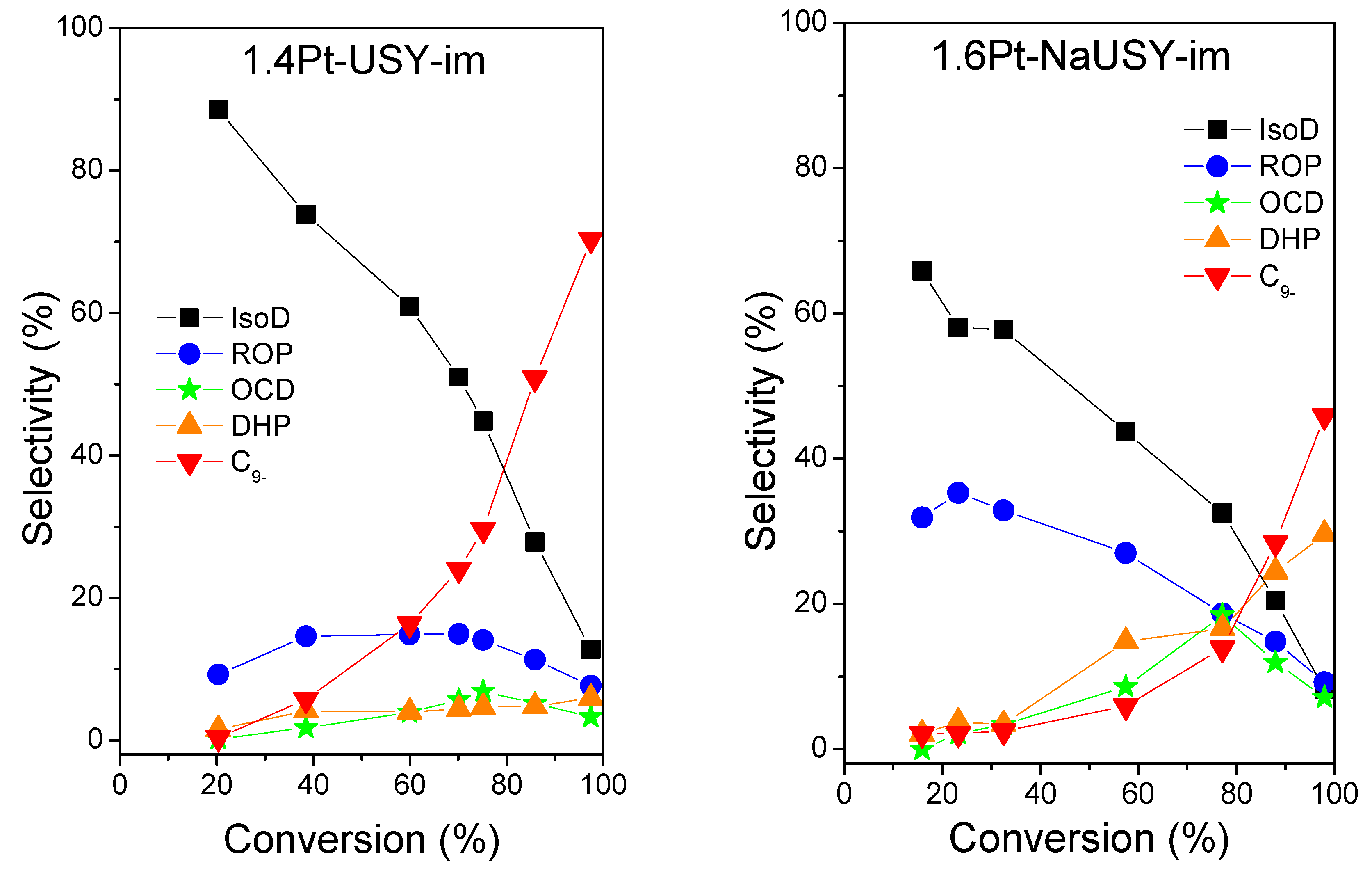
2.2. Catalytic Results
3. Materials and Methods
3.1. Preparation of the Catalysts
3.2. Characterization Techniques
3.3. Catalytic Experiments
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Martínez, A.; Arribas, M.A.; Pergher, S.B.C. Bifunctional noble metal/zeolite catalysts for upgrading low-quality diesel fractions via selective opening of naphthenic rings. Catal. Sci. Technol. 2016, 6, 2528–2542. [Google Scholar] [CrossRef]
- McVicker, G.B.; Touvelle, M.S.; Hudson, C.W.; Vaughan, D.E.W.; Daage, M.; Hantzer, S.; Klein, D.P.; Ellis, E.S.; Cook, B.R.; Feeley, O.C.; et al. Process for Selectively Opening Naphthenic Rings. U.S. Patent 5,763,731, 9 June 1998. [Google Scholar]
- McVicker, G.B.; Daage, M.; Touvelle, M.S.; Hudson, C.W.; Klein, D.P.; Baird, W.C., Jr.; Cook, B.R.; Chen, J.G.; Hantzer, S.; Vaughan, D.E.W.; et al. Selective Ring Opening of Naphthenic Molecules. J. Catal. 2002, 210, 137–148. [Google Scholar] [CrossRef]
- Kubička, D.; Kumar, N.; Mäki-Arvela, P.; Tiitta, M.; Niemi, V.; Salmi, T.; Murzin, D.Y. Ring opening of decalin over zeolites: I. Activity and selectivity of proton-form zeolites. J. Catal. 2004, 222, 65–79. [Google Scholar]
- Santikunaporn, M.; Herrera, J.E.; Jongpatiwut, S.; Resasco, D.E.; Alvarez, W.E.; Sughrue, E.L. Ring opening of decalin and tetralin on HY and Pt/HY zeolite catalysts. J. Catal. 2004, 228, 100–113. [Google Scholar] [CrossRef]
- Arribas, M.A.A.; Concepción, P.; Martínez, A.N. The role of metal sites during the coupled hydrogenation and ring opening of tetralin on bifunctional Pt(Ir)/USY catalysts. Appl. Catal. A Gen. 2004, 267, 111–119. [Google Scholar] [CrossRef]
- Moraes, R.; Thomas, K.; Thomas, S.; Donk, S.V.; Grasso, G.; Gilson, J.-P.; Houalla, M. Ring opening of decalin and methylcyclohexane over alumina-based monofunctional WO3/Al2O3 and Ir/Al2O3 catalysts. J. Catal. 2012, 286, 62–77. [Google Scholar] [CrossRef]
- Gault, F.G. Mechanisms of Skeletal Isomerization of Hydrocarbons on Metals. In Advances in Catalysis; Eley, D.D., Pines, H., Weisz, P.B., Eds.; Academic Press: Cambridge, MA, USA, 1981; pp. 1–95. [Google Scholar]
- Santi, D.; Holl, T.; Calemma, V.; Weitkamp, J. High-performance ring-opening catalysts based on iridium-containing zeolite Beta in the hydroconversion of decalin. Appl. Catal. A Gen. 2013, 455, 46–57. [Google Scholar] [CrossRef]
- Djeddi, A.; Fechete, I.; Garin, F. Selective ring opening of methylcyclopentane over Pt/γ-Al2O3, Ir/γ-Al2O3 and Pt-Ir/γ-Al2O3 catalysts with hydrogen at atmospheric pressure. Appl. Catal. A Gen. 2012, 413–414, 340–349. [Google Scholar] [CrossRef]
- Galperin, L.B.; Bricker, J.C.; Holmgren, J.R. Effect of support acid—Basic properties on activity and selectivity of Pt catalysts in reaction of methylcyclopentane ring opening. Appl. Catal. A Gen. 2003, 239, 297–304. [Google Scholar] [CrossRef]
- Onyestyák, G.; Pál-Borbély, G.; Beyer, H.K. Cyclohexane conversion over H-zeolite supported platinum. Appl. Catal. A Gen. 2002, 229, 65–74. [Google Scholar]
- D’Ippolito, S.A.; Benitez, V.M.; Reyes, P.; Rangel, M.C.; Pieck, C.L. Selective ring opening of decalin with Pt–Ir/Al2O3 catalyst prepared by catalytic reduction. Catal. Today 2011, 172, 177–182. [Google Scholar] [CrossRef]
- Lecarpentier, S.; van Gestel, J.; Thomas, K.; Gilson, J.-P.; Houalla, M. Study of Ir/WO3/ZrO2–SiO2 ring-opening catalysts: Part II. Reaction network, kinetic studies and structure–activity correlation. J. Catal. 2008, 254, 49–63. [Google Scholar] [CrossRef]
- Santikunaporn, M.; Alvarez, W.E.; Resasco, D.E. Ring contraction and selective ring opening of naphthenic molecules for octane number improvement. Appl. Catal. A Gen. 2007, 325, 175–187. [Google Scholar] [CrossRef]
- Di Felice, L.; Catherin, N.; Piccolo, L.; Laurenti, D.; Blanco, E.; Leclerc, E.; Geantet, C.; Calemma, V. Decalin ring opening over NiWS/SiO2-Al2O3 catalysts in the presence of H2S. Appl. Catal. A Gen. 2016, 512, 43–51. [Google Scholar] [CrossRef]
- Nassreddine, S.; Massin, L.; Aouine, M.; Geantet, C.; Piccolo, L. Thiotolerant Ir/SiO2–Al2O3 bifunctional catalysts: Effect of metal–acid site balance on tetralin hydroconversion. J. Catal. 2011, 278, 253–265. [Google Scholar] [CrossRef]
- Nassreddine, S.; Massin, L.; Aouine, M.; Geantet, C.; Piccolo, L. Thiotolerant Ir/SiO2–Al2O3 bifunctional catalysts: Effect of support acidity on tetralin hydroconversion. Catal. Sci. Technol. 2011, 1, 408–412. [Google Scholar] [CrossRef]
- Piccolo, L.; Nassreddine, S.; Toussaint, G.; Geantet, C. Mechanism of Tetralin Ring Opening and Contraction over Bifunctional Ir/SiO2-Al2O3 Catalysts. ChemSusChem 2012, 5, 1717–1723. [Google Scholar] [CrossRef]
- Corma, A.; González-Alfaro, V.; Orchillés, A.V. Decalin and Tetralin as Probe Molecules for Cracking and Hydrotreating the Light Cycle Oil. J. Catal. 2001, 200, 34–44. [Google Scholar] [CrossRef]
- Haas, A.; Rabl, S.; Ferrari, M.; Calemma, V.; Weitkamp, J. Ring opening of decalin via hydrogenolysis on Ir/-and Pt/silica catalysts. Appl. Catal. A Gen. 2012, 425–426, 97–109. [Google Scholar] [CrossRef]
- Kubička, D.; Kangas, M.; Kumar, N.; Tiitta, M.; Lindblad, M.; Murzin, D.Y. Ring Opening of Decalin Over Zeolite-Supported Iridium Catalysts. Top. Catal. 2010, 53, 1438–1445. [Google Scholar] [CrossRef]
- Kumar, N.; Mäki-Arvela, P.; Musakka, N.; Kubicka, D.; Kangas, M.; Tiitta, M.; Österholm, H.; Leino, A.-R.; Kordás, K.; Heikkilä, T.; et al. On the way to improve cetane number in diesel fuels: Ring opening of decalin over Ir-modified embedded mesoporous materials. Catal. Ind. 2013, 5, 105–122. [Google Scholar] [CrossRef]
- Ma, H.; Yang, X.; Wen, G.; Tian, G.; Wang, L.; Xu, Y.; Wang, B.; Tian, Z. Coupledhydrogenation and ring opening of tetralin on potassium modified Pt/USY catalysts. Catal. Lett. 2007, 116, 149–154. [Google Scholar] [CrossRef]
- Rabl, S.; Haas, A.; Santi, D.; Flego, C.; Ferrari, M.; Calemma, V.; Weitkamp, J. Ring opening of cis-decalin on bifunctional Ir/- and Pt/La-X zeolite catalysts. Appl. Catal. A Gen. 2011, 400, 131–141. [Google Scholar] [CrossRef]
- Arribas, M.A.; Corma, A.; Díaz-Cabañas, M.J.; Martínez, A. Hydrogenation and ring opening of Tetralin over bifunctional catalysts based on the new ITQ-21 zeolite. Appl. Catal. A Gen. 2004, 273, 277–286. [Google Scholar] [CrossRef]
- Arribas, M.A.; Martínez, A. Production of high cetane diesel fuels by simultaneous hydrogenation of aromatics and ring opening of naphthenes over bifunctional molecular sieves based catalysts. In Studies in Surface Science and Catalysis; Corma, A., Melo, F.V., Mendioroz, S., Fierro, J.L.G., Eds.; Elsevier: Amsterdam, The Netherlands, 2000; pp. 2585–2590. [Google Scholar]
- Piccolo, L.; Gigantiello, N.; Parker, W.O.; Calemma, V., Jr. Discussion on “A comprehensive two-dimensional gas chromatography coupled with quadrupole mass spectrometry approach for identification of C10 derivatives from decalin” by C. Flego, N. Gigantiello, WO Parker, Jr., V. Calemma [J. Chromatogr. A 1216 (2009) 2891]. J. Chromatogr. A 2010, 1217, 5872–5873. [Google Scholar] [PubMed]
- Blanco, E.; Felice, L.D.; Catherin, N.; Piccolo, L.; Laurenti, D.; Lorentz, C.; Geantet, C.; Calemma, V. Understanding the Mechanisms of Decalin Hydroprocessing Using Comprehensive Two-Dimensional Chromatography. Ind. Eng. Chem. Res. 2016, 55, 12516–12523. [Google Scholar] [CrossRef]
- Emeis, C.A. Determination of Integrated Molar Extinction Coefficients for Infrared Absorption Bands of Pyridine Adsorbed on Solid Acid Catalysts. J. Catal. 1993, 141, 347–354. [Google Scholar] [CrossRef]
- Flego, C.; Gigantiello, N.; Parker, W.O.; Calemma, V., Jr. A comprehensive two-dimensional gas chromatography coupled with quadrupole mass spectrometry approach for identification of C10 derivatives from decalin. J. Chromatogr. A 2009, 1216, 2891–2899. [Google Scholar] [CrossRef] [PubMed]
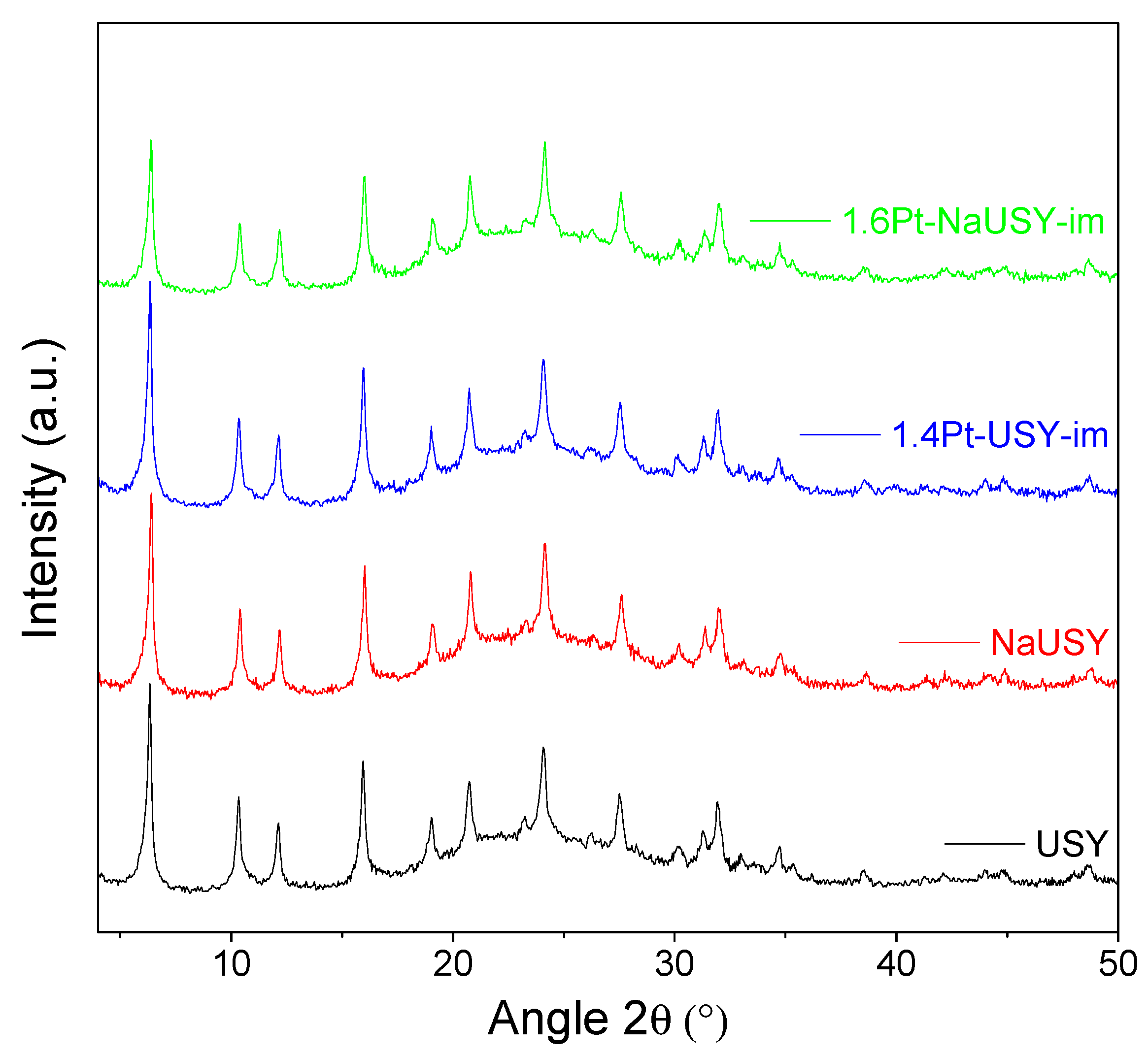


| Zeolite | Si:Al a | Na b (wt%) | Cryst.c (%) | Pt d (wt%) | DPt e (%) | AMSA f (m2/g) | dPt g (nm) |
|---|---|---|---|---|---|---|---|
| USY | 26.5 | 0.02 | 100 | ||||
| NaUSY | 27.0 | 0.39 | 99 | ||||
| 1.4Pt-USY-im | 26.0 | 0.02 | 96 | 1.4 | 21.4 | 0.8 | 5.28 |
| 1.6Pt-NaUSY-im | 27.0 | 0.39 | 92 | 1.6 | 27.7 | 1.1 | 4.08 |
| Zeolite | SBET (m2/g) | Smeso a (m2/g) | Vmicro b (cm3/g) | TPV c (cm3/g) | Vmeso d (cm3/g) |
|---|---|---|---|---|---|
| USY | 514.4 | 86.0 | 0.217 | 0.447 | 0.230 |
| NaUSY | 499.3 | 86.4 | 0.208 | 0.469 | 0.261 |
| 1.4Pt-USY-im | 483.9 | 88.4 | 0.200 | 0.474 | 0.274 |
| 1.6Pt-NaUSY-im | 465.3 | 85.5 | 0.191 | 0.457 | 0.266 |
| Zeolite | Brønsted | ||
|---|---|---|---|
| 250 °C | 350 °C | 400 °C | |
| USY | 67.7 | 28.0 | 4.7 |
| NaUSY | 18.7 | 3.5 | 1.2 |
| 1.4Pt-USY-im | 68.2 | 29.2 | 10.5 |
| 1.6Pt-NaUSY-im | 22.2 | 3.5 | 2.3 |
| Catalysts | T (°C) | XD a (%) | RROPs (wt%) | ROCDs, máx (wt%) | RROPs + ROCDs (wt%) | (wt%) |
|---|---|---|---|---|---|---|
| 1.4Pt-USY-im | 355 | 75.1 | 10.6 | 5.2 | 15.7 | 22.2 |
| 1.6Pt-NaUSY-im | 380 | 77.2 | 14.4 | 14.2 | 28.6 | 10.7 |
| Catalysts | T (°C) | XD (%) | SROPs (%) | SOCDs, max (%) | SROPs + SOCDs (%) | (wt%) |
|---|---|---|---|---|---|---|
| 1.4Pt-USY-im | 355 | 75.1 | 14.1 | 6.9 | 21 | 29.6 |
| 1.6Pt-NaUSY-im | 380 | 77.2 | 18.7 | 18.4 | 37.1 | 13.9 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oliveira Soares, L.W.; Castellã Pergher, S.B. Influence of the Brønsted Acidity on the Ring Opening of Decalin for Pt-USY Catalysts. Catalysts 2019, 9, 786. https://doi.org/10.3390/catal9100786
Oliveira Soares LW, Castellã Pergher SB. Influence of the Brønsted Acidity on the Ring Opening of Decalin for Pt-USY Catalysts. Catalysts. 2019; 9(10):786. https://doi.org/10.3390/catal9100786
Chicago/Turabian StyleOliveira Soares, Lech Walesa, and Sibele Berenice Castellã Pergher. 2019. "Influence of the Brønsted Acidity on the Ring Opening of Decalin for Pt-USY Catalysts" Catalysts 9, no. 10: 786. https://doi.org/10.3390/catal9100786
APA StyleOliveira Soares, L. W., & Castellã Pergher, S. B. (2019). Influence of the Brønsted Acidity on the Ring Opening of Decalin for Pt-USY Catalysts. Catalysts, 9(10), 786. https://doi.org/10.3390/catal9100786






