Hydrotreating of Light Cycle Oil over Supported on Porous Aromatic Framework Catalysts
Abstract
1. Introduction
2. Results and Discussion
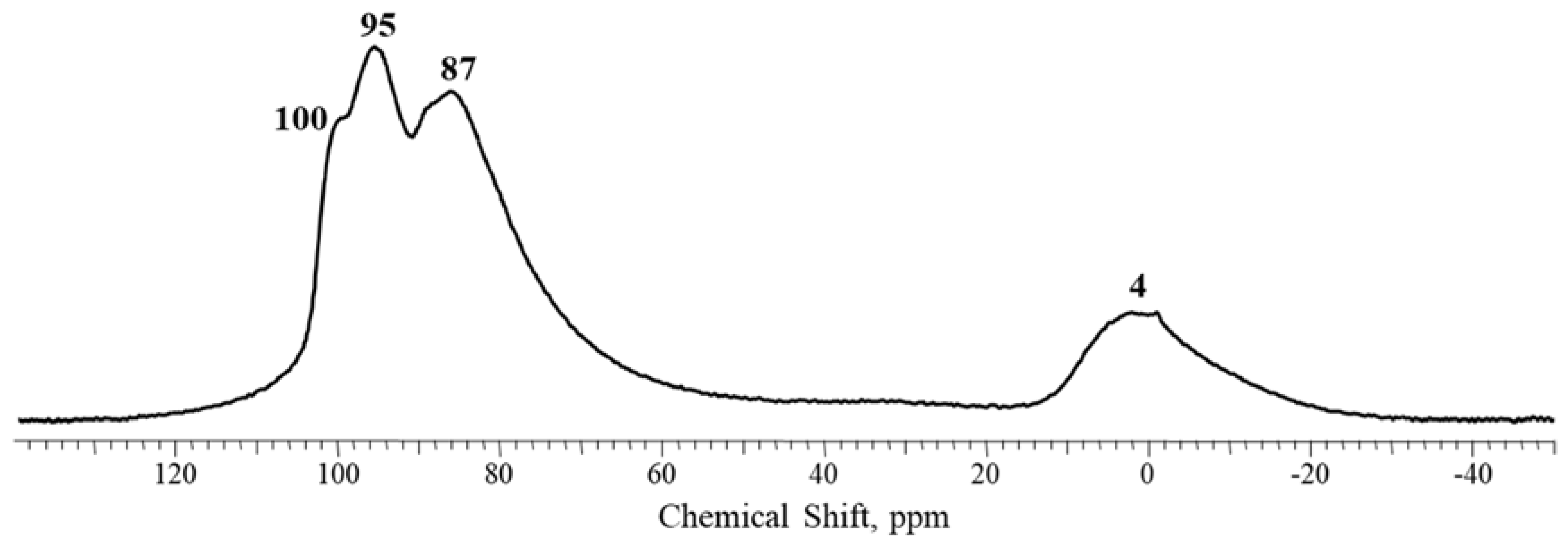
2.1. Characterization of Precursors and Catalysts
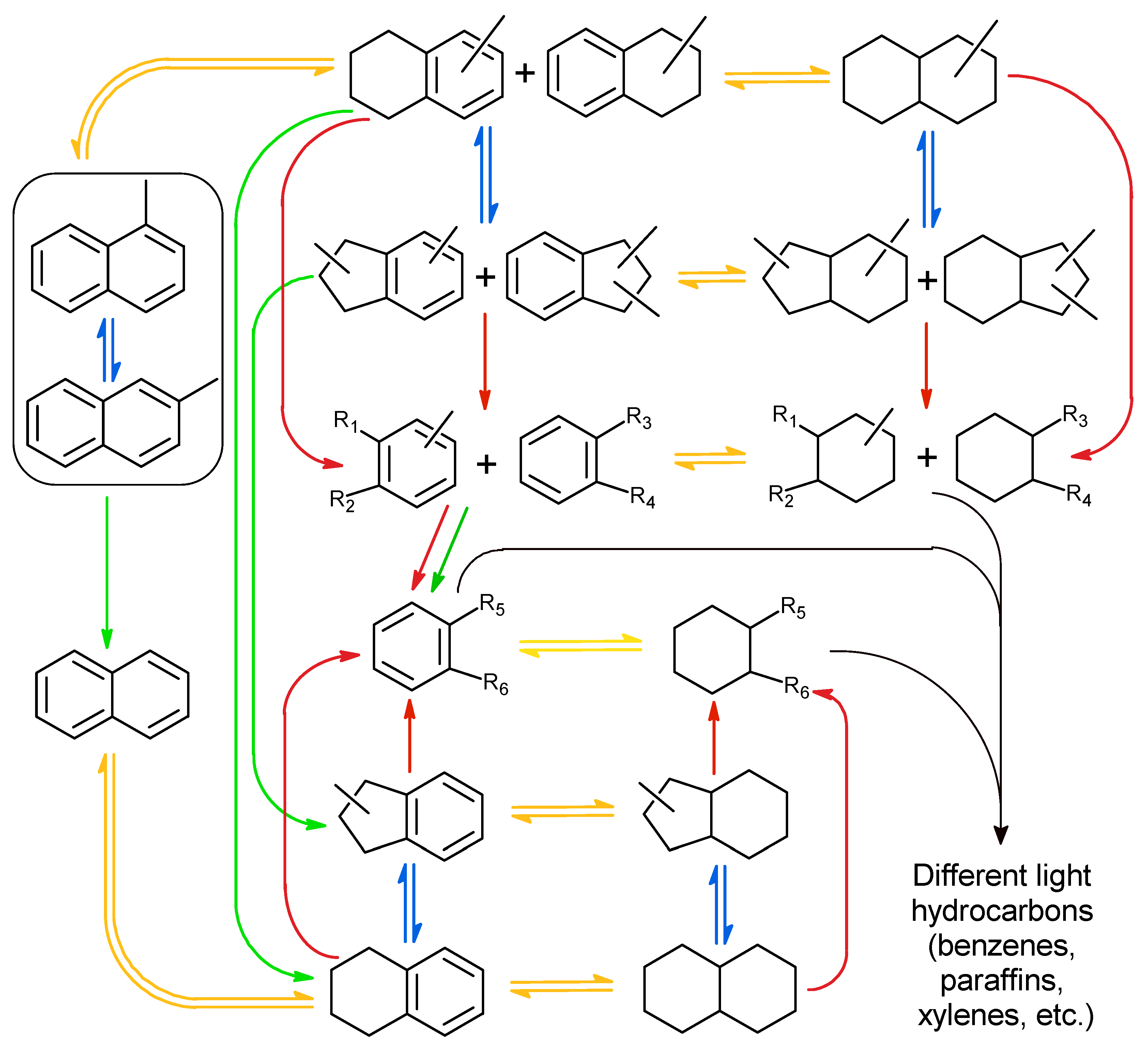
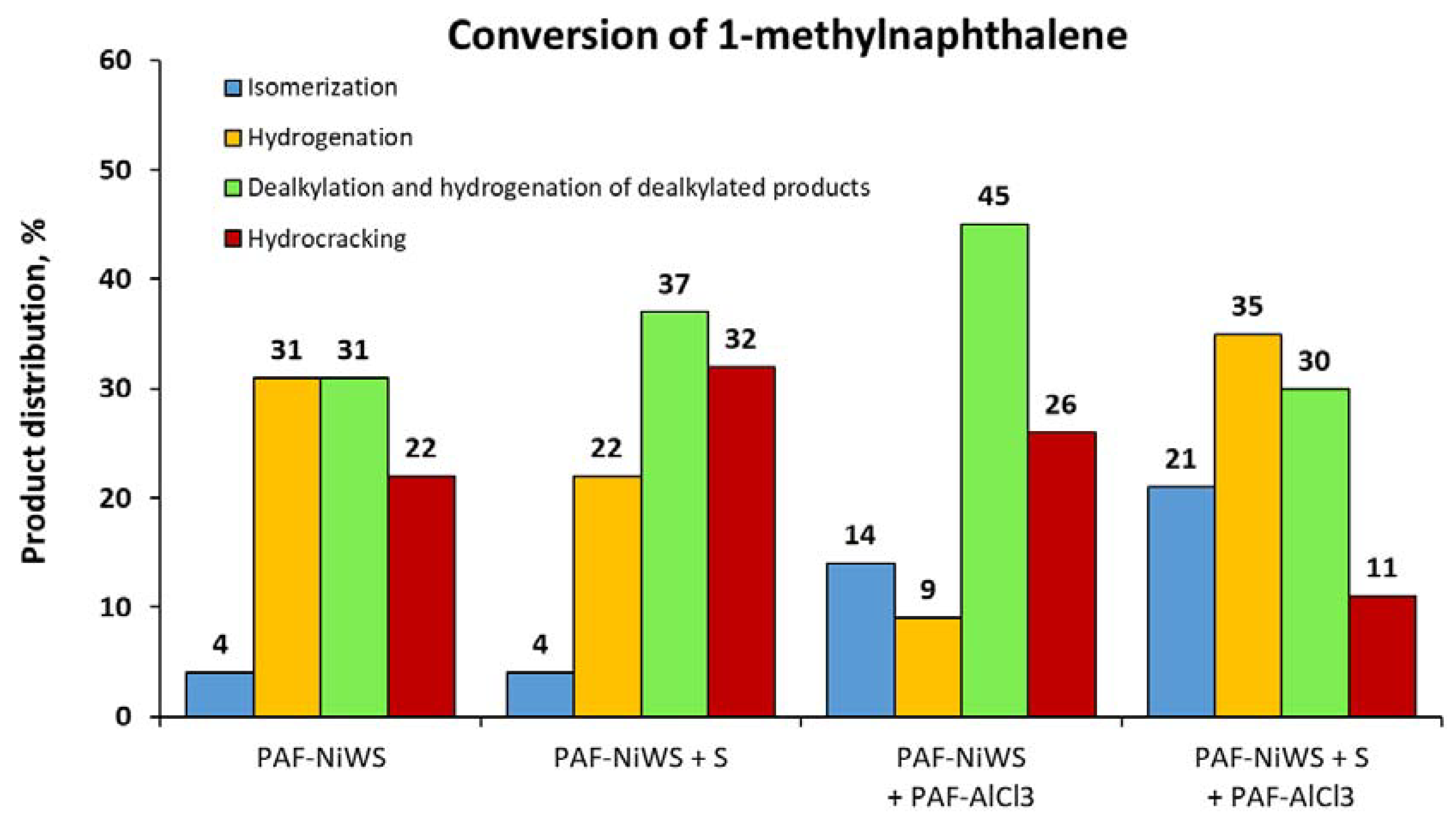
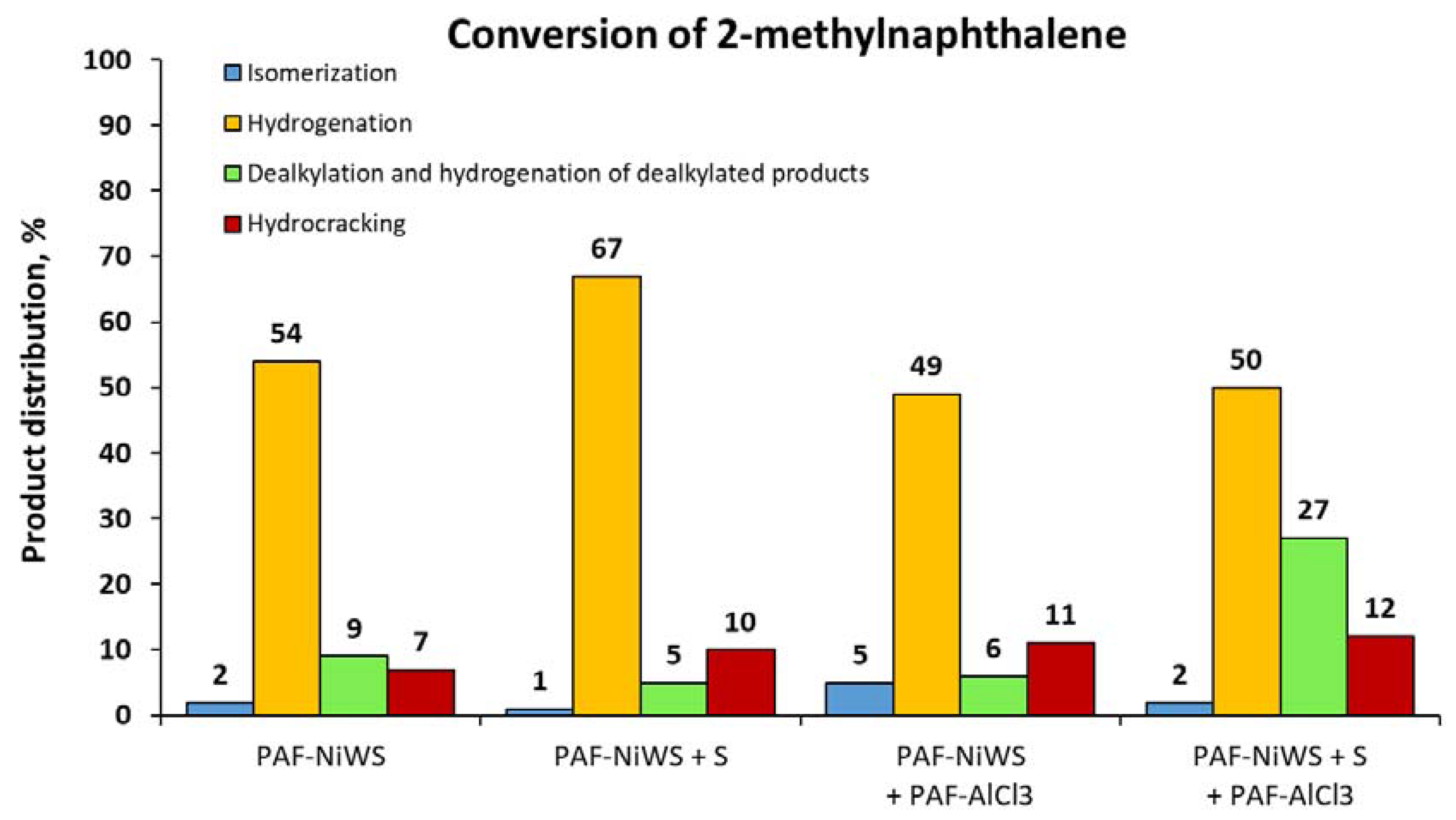
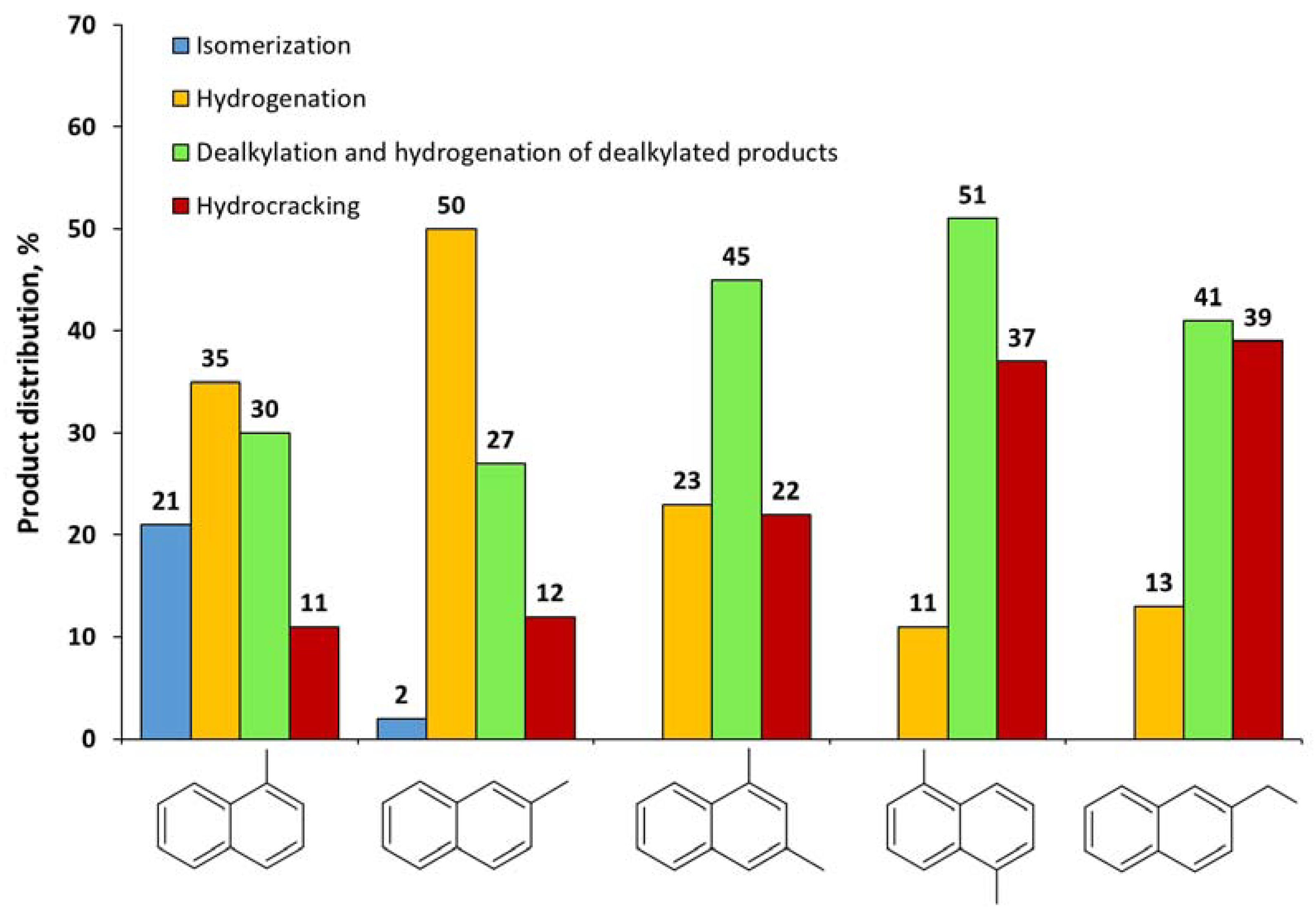
2.2. Catalytic Experiments with Model Substrates
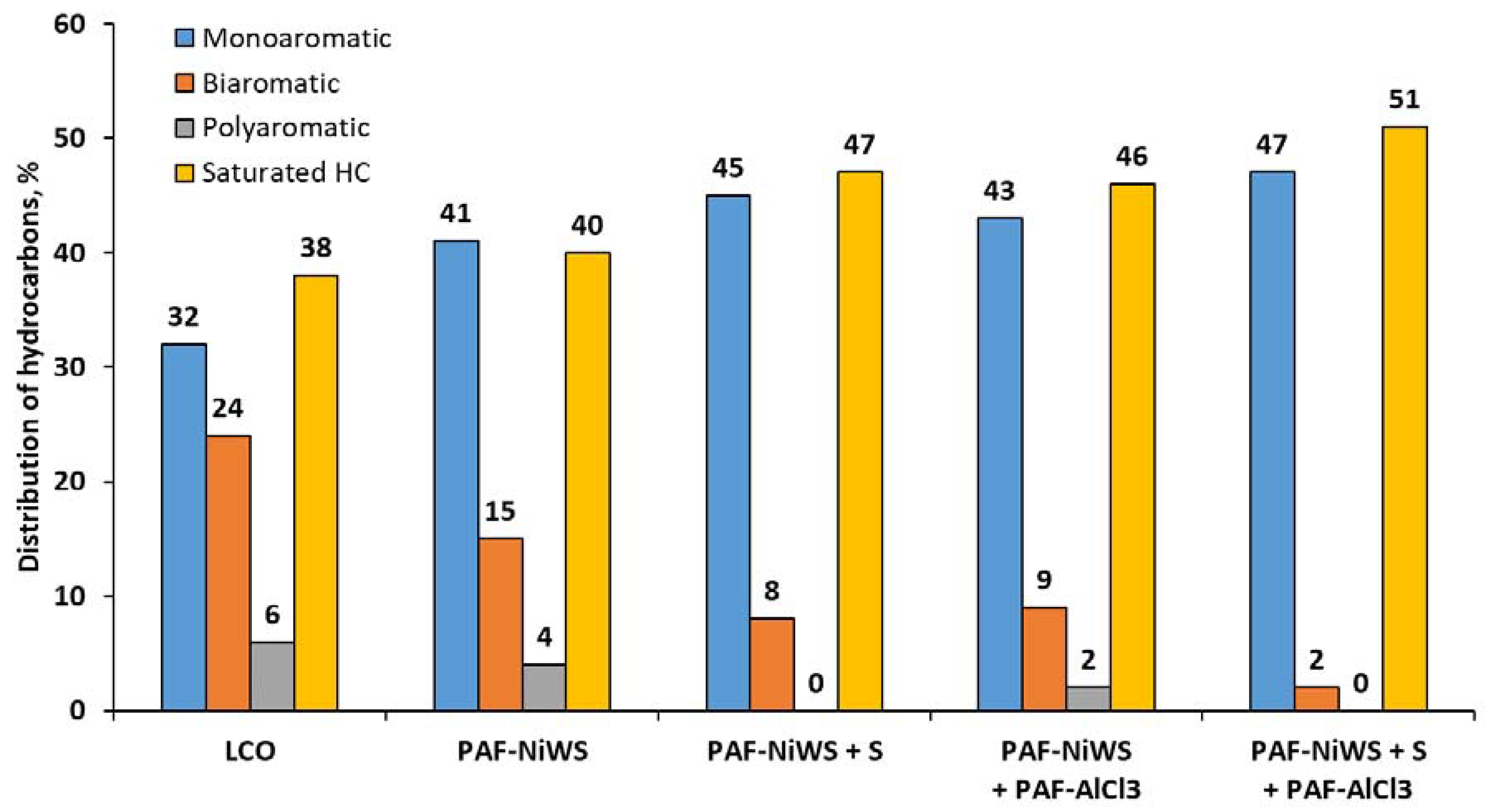
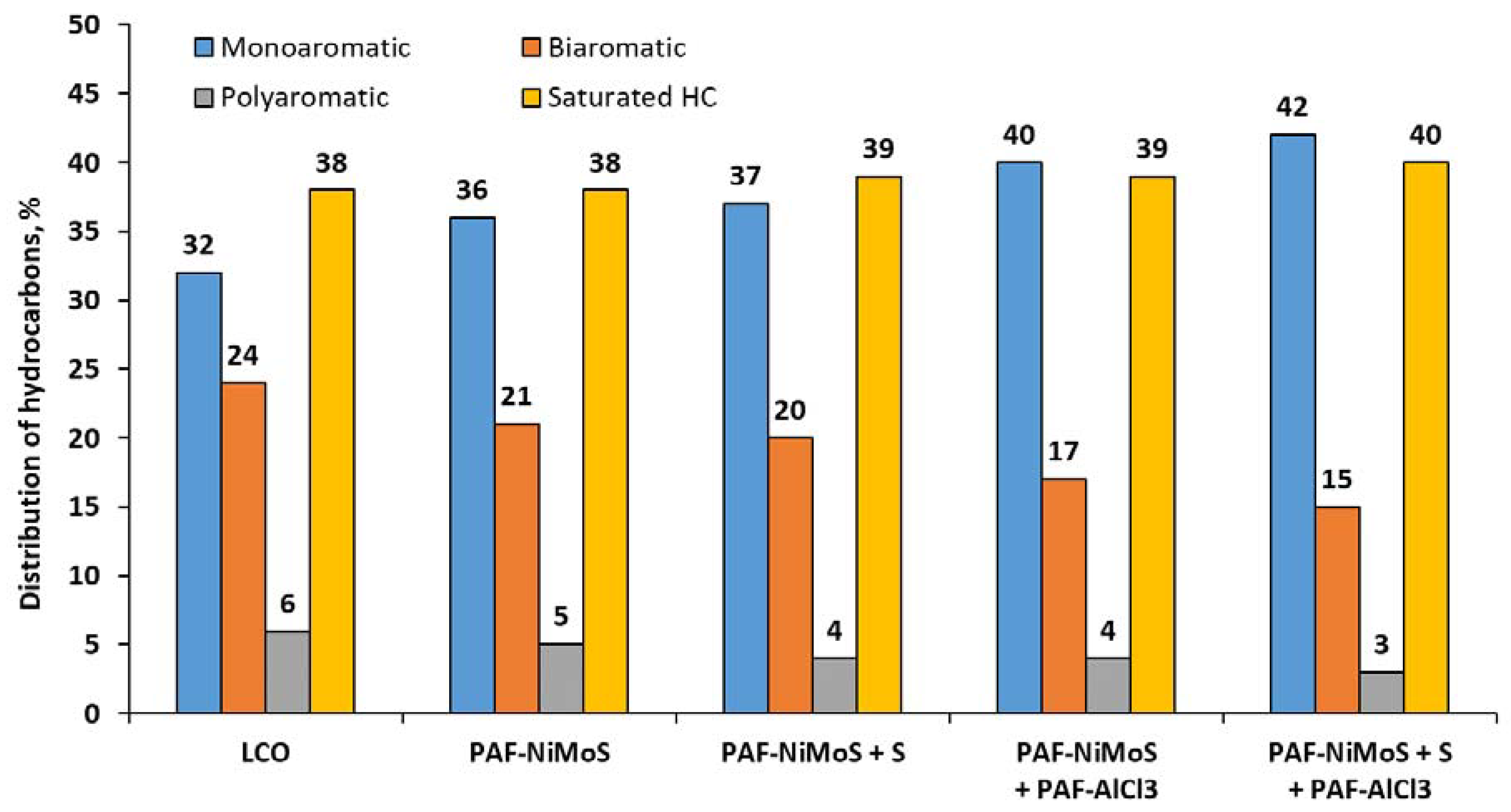
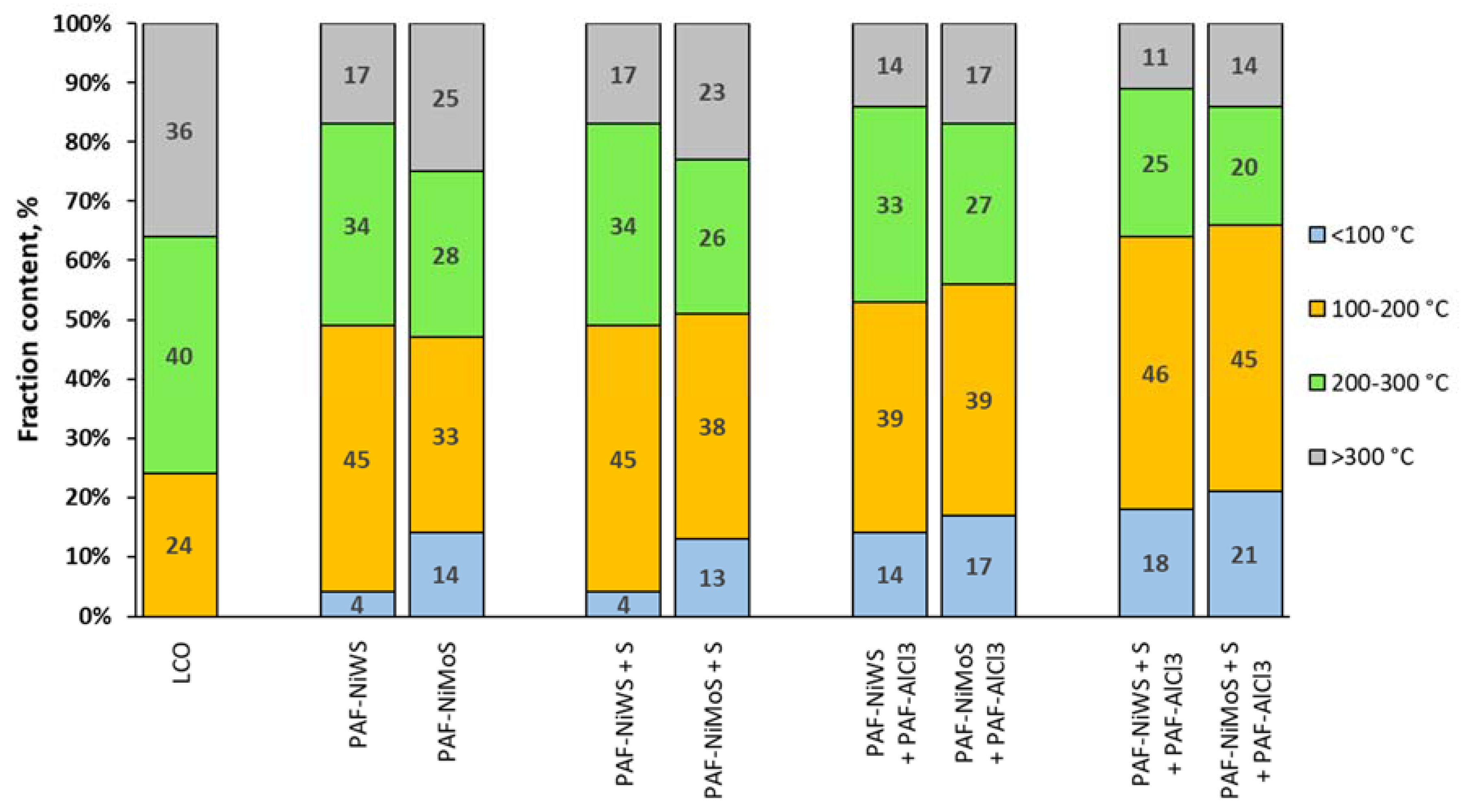
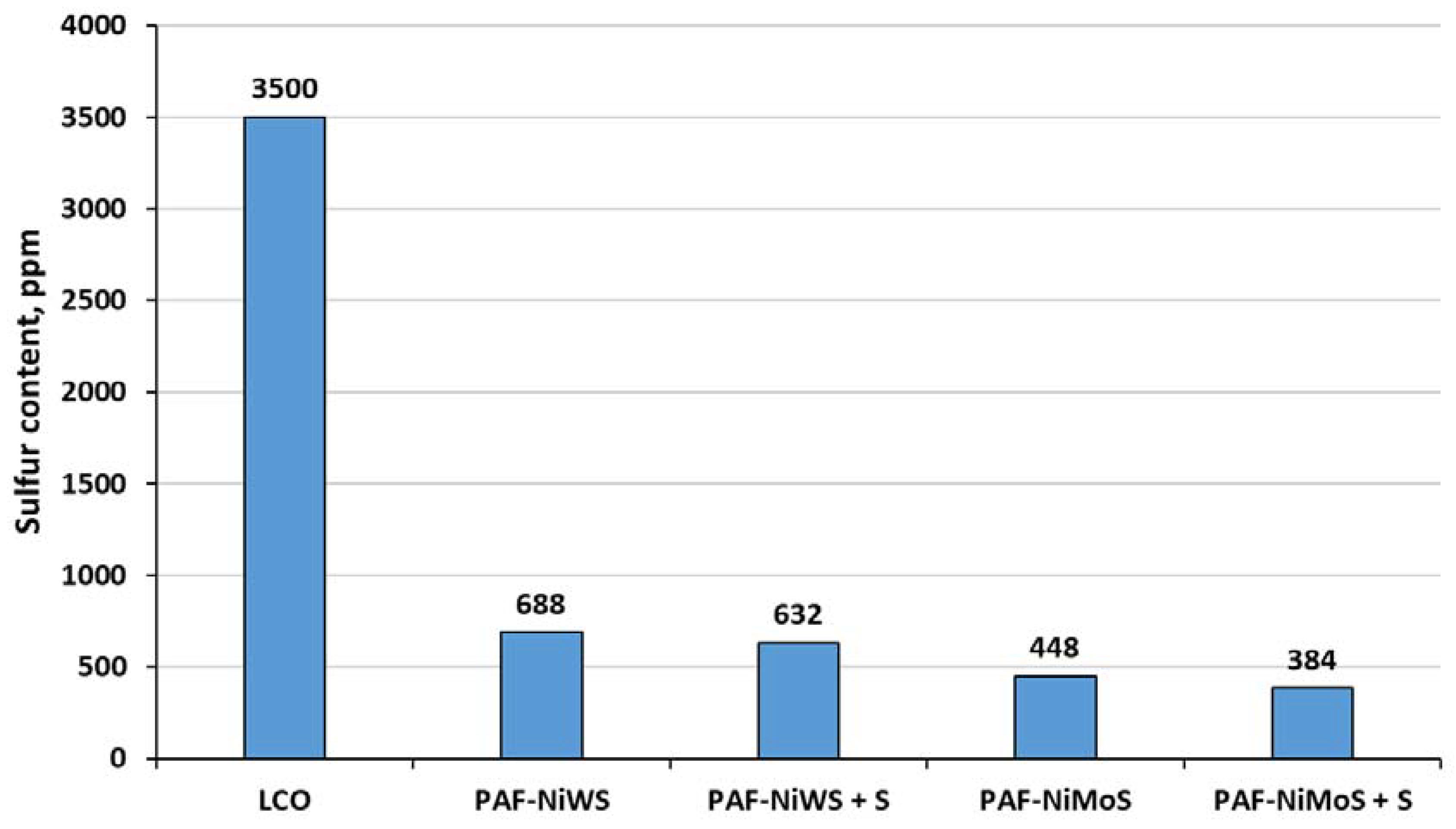
2.3. Catalytic Experiments with Light Cycle Oil (LCO)
3. Materials and Methods
3.1. Materials
3.2. Synthesis of Precursors and Catalysts
3.3. Characterization of Catalysts
3.4. Catalytic Testing Procedure
3.5. Gas Chromatography
3.6. X-ray Fluorescence Analysis
3.7. High Performance Liquid Chromatography
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Primo, A.; Garcia, H. Zeolites as Catalysts in Oil Refining. Chem. Soc. Rev. 2014, 43, 7548–7561. [Google Scholar] [CrossRef] [PubMed]
- Ennaert, T.; Van Aelst, J.; Dijkmans, J.; De Clercq, R.; Schutyser, W.; Dusselier, M.; Verboekend, D.; Sels, B.F. Potential and Challenges of Zeolite Chemistry in the Catalytic Conversion of Biomass. Chem. Soc. Rev. 2016, 45, 584–611. [Google Scholar] [CrossRef] [PubMed]
- Wan, Y.; Zhao, D. On the Controllable Soft-Templating Approach to Mesoporous Silicates. Chem. Rev. 2007, 107, 2821–2860. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, R. Synthesis and Applications of Ordered and Disordered Mesoporous Zeolites: Present and Future Prospective. Catal. Today 2018, 309, 172–188. [Google Scholar] [CrossRef]
- Bingre, R.; Louis, B.; Nguyen, P. An Overview on Zeolite Shaping Technology and Solutions to Overcome Diffusion Limitations. Catalysts 2018, 8, 163. [Google Scholar] [CrossRef]
- Kaneko, T.; Nagata, F.; Kugimiya, S.; Kato, K. Morphological Control of Mesoporous Silica Particles by Dual Template Method. Ceram. Int. 2018. [Google Scholar] [CrossRef]
- Albertini, F.; Ribeiro, T.; Alves, S.; Baleizão, C.; Farinha, J.P.S. Boron-Chelating Membranes Based in Hybrid Mesoporous Silica Nanoparticles for Water Purification. Mater. Des. 2018, 141, 407–413. [Google Scholar] [CrossRef]
- Pal, N.; Bhaumik, A. Soft Templating Strategies for the Synthesis of Mesoporous Materials: Inorganic, Organic–inorganic Hybrid and Purely Organic Solids. Adv. Colloid Interface Sci. 2013, 189–190, 21–41. [Google Scholar] [CrossRef] [PubMed]
- Topsøe, H. The Role of Co–Mo–S Type Structures in Hydrotreating Catalysts. Appl. Catal. A Gen. 2007, 322, 3–8. [Google Scholar] [CrossRef]
- Raybaud, P.; Toulhoat, H. Catalysis by Transition Metal Sulfides: From Molecular Theory to Industrial Application; Editions TECHNIP: Paris, France, 2013; ISBN 9782710809913. [Google Scholar]
- Díaz, U.; Corma, A. Ordered Covalent Organic Frameworks, COFs and PAFs. From Preparation to Application. Coord. Chem. Rev. 2016, 311, 85–124. [Google Scholar] [CrossRef]
- Karakhanov, E.; Maximov, A.; Boronoev, M.; Kulikov, L.; Terenina, M. Mesoporous Organo-Inorganic Hybrid Materials as Hydrogenation Catalysts. Pure Appl. Chem. 2017, 89, 1157–1166. [Google Scholar] [CrossRef]
- Kaur, P.; Hupp, J.T.; Nguyen, S.T. Porous Organic Polymers in Catalysis: Opportunities and Challenges. ACS Catal. 2011, 1, 819–835. [Google Scholar] [CrossRef]
- Karakhanov, E.; Kardasheva, Y.; Kulikov, L.; Maximov, A.; Zolotukhina, A.; Vinnikova, M.; Ivanov, A. Sulfide Catalysts Supported on Porous Aromatic Frameworks for Naphthalene Hydroprocessing. Catalysts 2016, 6, 122. [Google Scholar] [CrossRef]
- Karakhanov, E.; Boronoev, M.; Ignatyeva, V.; Maximov, A.; Filippova, T.; Kardasheva, Y. Hydroprocessing of Aromatics Using Sulfide Catalysts Supported on Ordered Mesoporous Phenol-Formaldehyde Polymers. J. Inorg. Organomet. Polym. Mater. 2016, 26, 1253–1258. [Google Scholar] [CrossRef]
- Boronoev, M.P.; Vinnikova, M.A.; Ignat’eva, V.I.; Kulikov, L.A.; Putilin, F.N.; Maksimov, A.L.; Karakhanov, E.A. Bimetallic Sulfide Catalysts Based on Mesoporous Organic Supports in the Hydrofining of Light Cycle Oil. Pet. Chem. 2017, 57, 855–858. [Google Scholar] [CrossRef]
- Alonso, G.; Petranovskii, V.; Del Valle, M.; Cruz-Reyes, J.; Fuentes, S. Catalytic Properties of WS2 Catalysts Prepared by in Situ Decomposition of Tetraalkyl-Ammonium Thiotungstates. Stud. Surf. Sci. Catal. 1999, 127, 351–355. [Google Scholar]
- Alonso, G.; Del Valle, M.; Cruz, J.; Petranovskii, V.; Licea-Claverie, A.; Fuentes, S. Preparation of MoS2 Catalysts by in Situ Decomposition of Tetraalkylammonium Thiomolybdates. Catal. Today 1998, 43, 117–122. [Google Scholar] [CrossRef]
- Kulikov, L.A.; Boronoev, M.P.; Makeeva, D.A.; Nenasheva, M.V.; Egazar’yants, S.V.; Karakhanov, E.A. Hydroconversion of Naphthalene in the Presence of NiMoS/NiWS-AlCl3 Catalyst Systems Derived from Mesoporous Aromatic Frameworks. Chem. Technol. Fuels Oils 2018, 53, 1–6. [Google Scholar] [CrossRef]
- Poisot, M.; Bensch, W. Decomposition of Tetraalkylammonium Thiotungstates Characterized by Thermoanalysis, Mass Spectrometry, X-ray Diffractometry and Scanning Electron Microscopy. Thermochim. Acta 2007, 453, 42–51. [Google Scholar] [CrossRef]
- Poisot, M.; Bensch, W.; Fuentes, S.; Alonso, G. Decomposition of Tetra-Alkylammonium Thiomolybdates Characterised by Thermoanalysis and Mass Spectrometry. Thermochim. Acta 2006, 444, 35–45. [Google Scholar] [CrossRef]
- Yuan, Y.; Sun, F.; Ren, H.; Jing, X.; Wang, W.; Ma, H.; Zhao, H.; Zhu, G. Targeted Synthesis of a Porous Aromatic Framework with a High Adsorption Capacity for Organic Molecules. J. Mater. Chem. 2011, 21, 13498–13502. [Google Scholar] [CrossRef]
- Li, P.; Chen, Y.; Zhang, C.; Baokun, H.; Liu, X.; Liu, T.; Jiang, Z.; Li, C. Highly Selective Hydrodesulfurization of Gasoline on Unsupported Co-Mo Sulfide Catalysts: Effect of MoS2 Morphology. Appl. Catal. A Gen. 2017, 533, 99–108. [Google Scholar] [CrossRef]
- Le, Z.; Afanasiev, P.; Li, D.; Long, X.; Vrinat, M. Solution Synthesis of the Unsupported Ni–W Sulfide Hydrotreating Catalysts. Catal. Today 2008, 130, 24–31. [Google Scholar] [CrossRef]
- Salim, S.S.; Bell, A.T. Effects of Lewis Acid Catalysts on the Hydrogenation and Cracking of Three-Ring Aromatic and Hydroaromatic Structures Related to Coal. Fuel 1984, 63, 469–476. [Google Scholar] [CrossRef]
- Saidi, M.R.; Pourshojaei, Y.; Aryanasab, F. Highly Efficient Michael Addition Reaction of Amines Catalyzed by Silica-Supported Aluminum Chloride. Synth. Commun. 2009, 39, 1109–1119. [Google Scholar] [CrossRef]
- Hall, G.E.; Johnson, E.A. The Reaction of Benzene with Aluminium Chloride. J. Chem. Soc. C Org. 1966, 2043–2047. [Google Scholar] [CrossRef]
- Merino, E.; Verde-Sesto, E.; Maya, E.M.; Corma, A.; Iglesias, M.; Sánchez, F. Mono-Functionalization of Porous Aromatic Frameworks to Use as Compatible Heterogeneous Catalysts in One-Pot Cascade Reactions. Appl. Catal. A Gen. 2014, 469, 206–212. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, B.; Williams, K.; Gao, W.-Y.; Ma, S. A New Microporous Carbon Material Synthesized via Thermolysis of a Porous Aromatic Framework Embedded with an Extra Carbon Source for Low-Pressure CO2 Uptake. Chem. Commun. 2013, 49, 10269–10271. [Google Scholar] [CrossRef] [PubMed]
- Lu, W.; Yuan, D.; Sculley, J.; Zhao, D.; Krishna, R.; Zhou, H.-C. Sulfonate-Grafted Porous Polymer Networks for Preferential CO2 Adsorption at Low Pressure. J. Am. Chem. Soc. 2011, 133, 18126–18129. [Google Scholar] [CrossRef] [PubMed]
- Nakayama, Y.; Senda, Y.; Kawasaki, H.; Koshitani, N.; Hosoi, S.; Kudo, Y.; Morioka, H.; Nagamine, M. Sulfone-Based Electrolytes for Aluminium Rechargeable Batteries. Phys. Chem. Chem. Phys. 2015, 17, 5758–5766. [Google Scholar] [CrossRef] [PubMed]
- Nöth, H.; Rurlaender, R.; Wolfgardt, P. An Investigation of AlCl3 Solutions in Ethers by 27Al NMR Spectroscopy. Z. Naturforsch. B 1982, 37, 29–37. [Google Scholar] [CrossRef]
- Černý, Z.; Macháček, J.; Fusek, J.; Čásenský, B.; Křiž, O.; Tuck, D.G. 27Al NMR Studies of the Hydrolysis of Aluminium(III) Chloride in Non-Aqueous Media. Inorg. Chim. Acta 2000, 300–302, 556–564. [Google Scholar] [CrossRef]
- Sugumaran, V.; Biswas, H.; Yadav, A.; Christopher, J.; Kagdiyal, V.; Patel, M.B.; Basu, B. Molecular-Level Characterization of Refinery Streams by High-Resolution Mass Spectrometry. Energy Fuels 2015, 29, 2940–2950. [Google Scholar] [CrossRef]
- Kabe, T.; Aoyama, Y.; Wang, D.; Ishihara, A.; Qian, W.; Hosoya, M.; Zhang, Q. Effects of H2S on Hydrodesulfurization of Dibenzothiophene and 4,6-Dimethyldibenzothiophene on Alumina-Supported NiMo and NiW Catalysts. Appl. Catal. A Gen. 2001, 209, 237–247. [Google Scholar] [CrossRef]
- Halachev, T.; Nava, R.; Dimitrov, L. Catalytic Activity of (P)NiMo/Ti-HMS and (P)NiW/Ti-HMS Catalysts in the Hydrogenation of Naphthalene. Appl. Catal. A Gen. 1998, 169, 111–117. [Google Scholar] [CrossRef]
- Ward, J.W. Hydrocracking Processes and Catalysts. Fuel Process. Technol. 1993, 35, 55–85. [Google Scholar] [CrossRef]
- Silva-Rodrigo, R.; Calderón-Salas, C.; Melo-Banda, J.A.; Domínguez, J.M.; Vázquez-Rodríguez, A. Synthesis, Characterization and Comparison of Catalytic Properties of NiMo- and NiW/Ti-MCM-41 Catalysts for HDS of Thiophene and HVGO. Catal. Today 2004, 98, 123–129. [Google Scholar] [CrossRef]
- ASTM D2887-16a. Standard Test Method for Boiling Range Distribution of Petroleum Fractions by Gas Chromatography; ASTM International: West Conshohocken, PA, USA, 2016. [Google Scholar]
- ASTM D4294-16e1. Standard Test Method for Sulfur in Petroleum and Petroleum Products by Energy Dispersive X-ray Fluorescence Spectrometry; ASTM International: West Conshohocken, PA, USA, 2016. [Google Scholar]
- GOST R EN 12916-2008. Petroleum Products. Determination of Aromatic Hydrocarbon Types in Middle Distillates. High Performance Liquid Chromatography Method with Refractive Index Detection; Standartinform: Moscow, Russia, 2008. [Google Scholar]
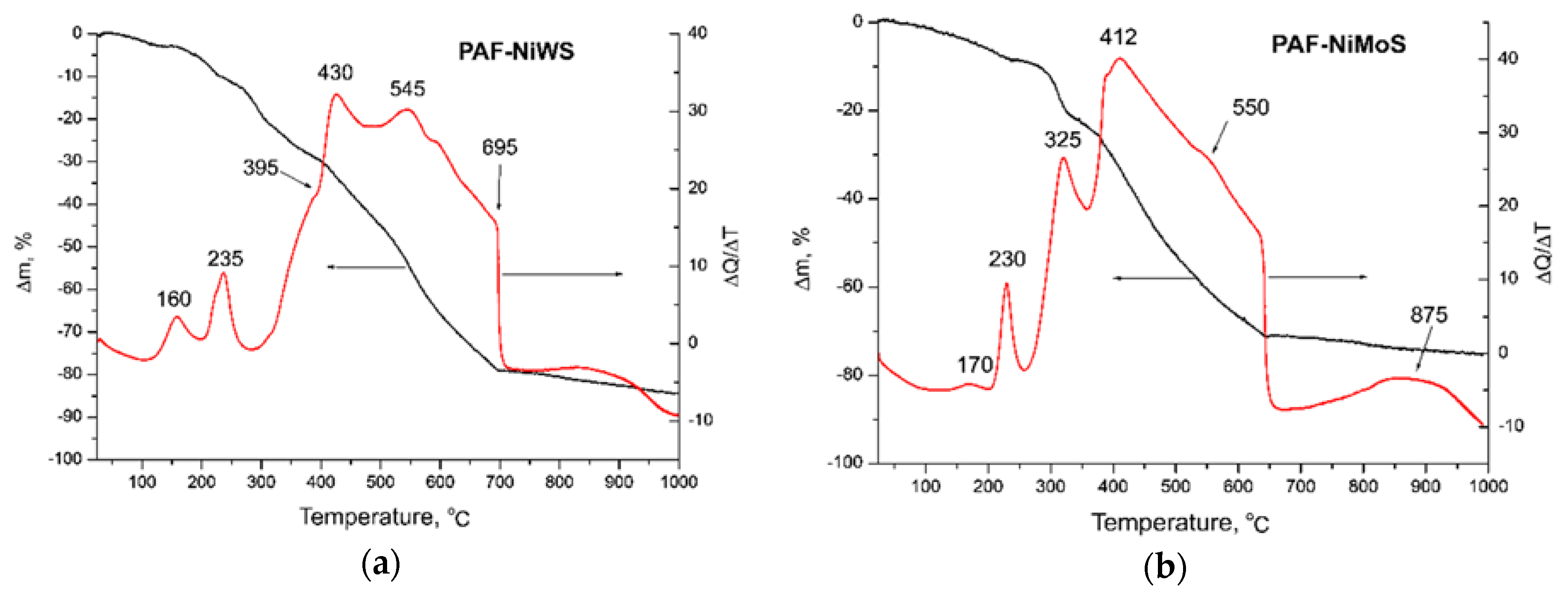
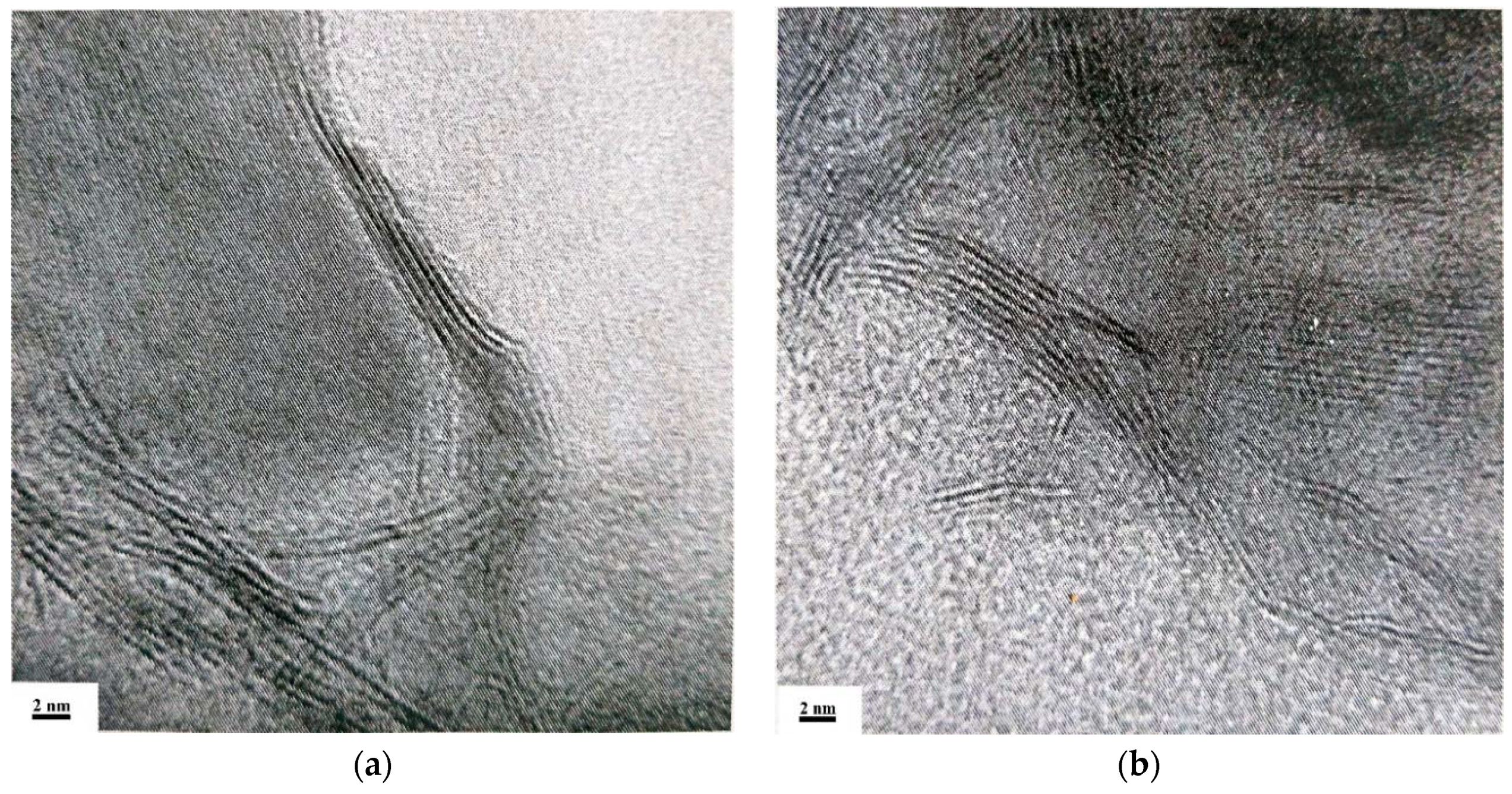
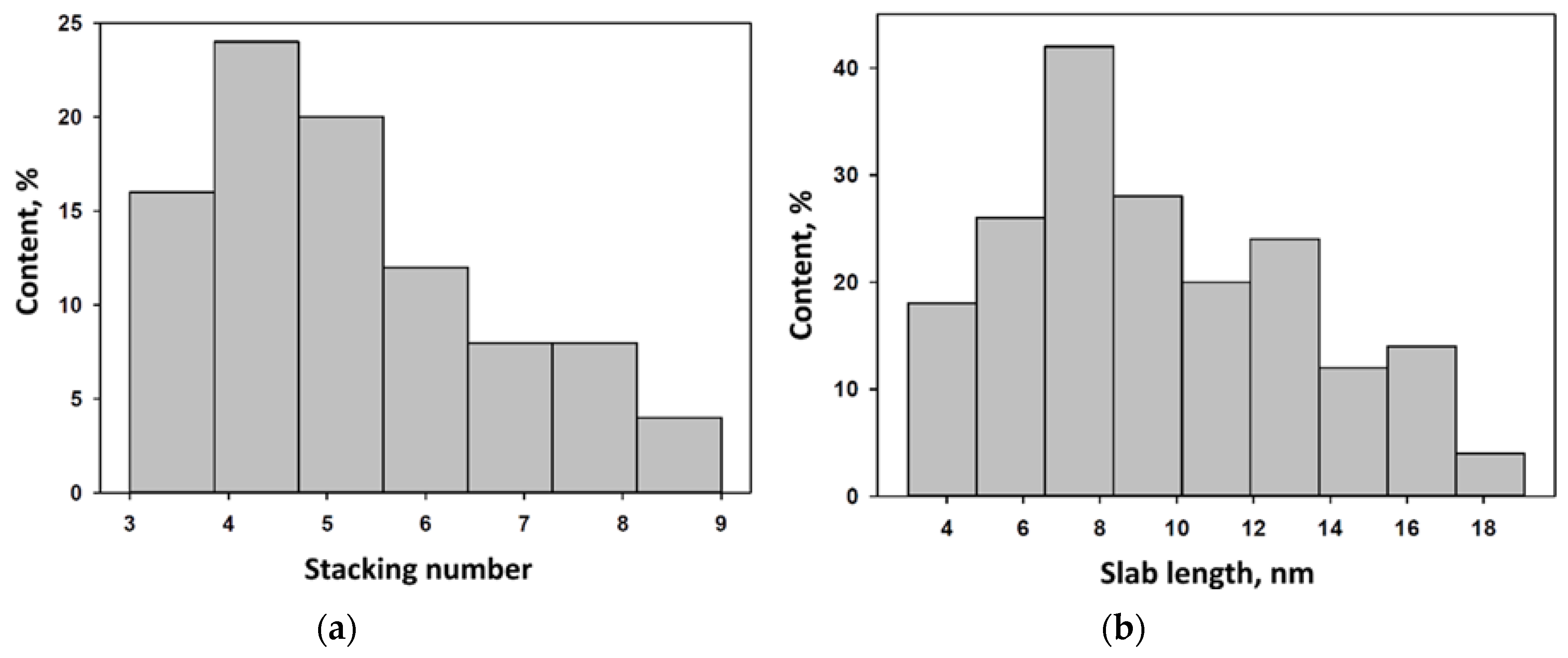
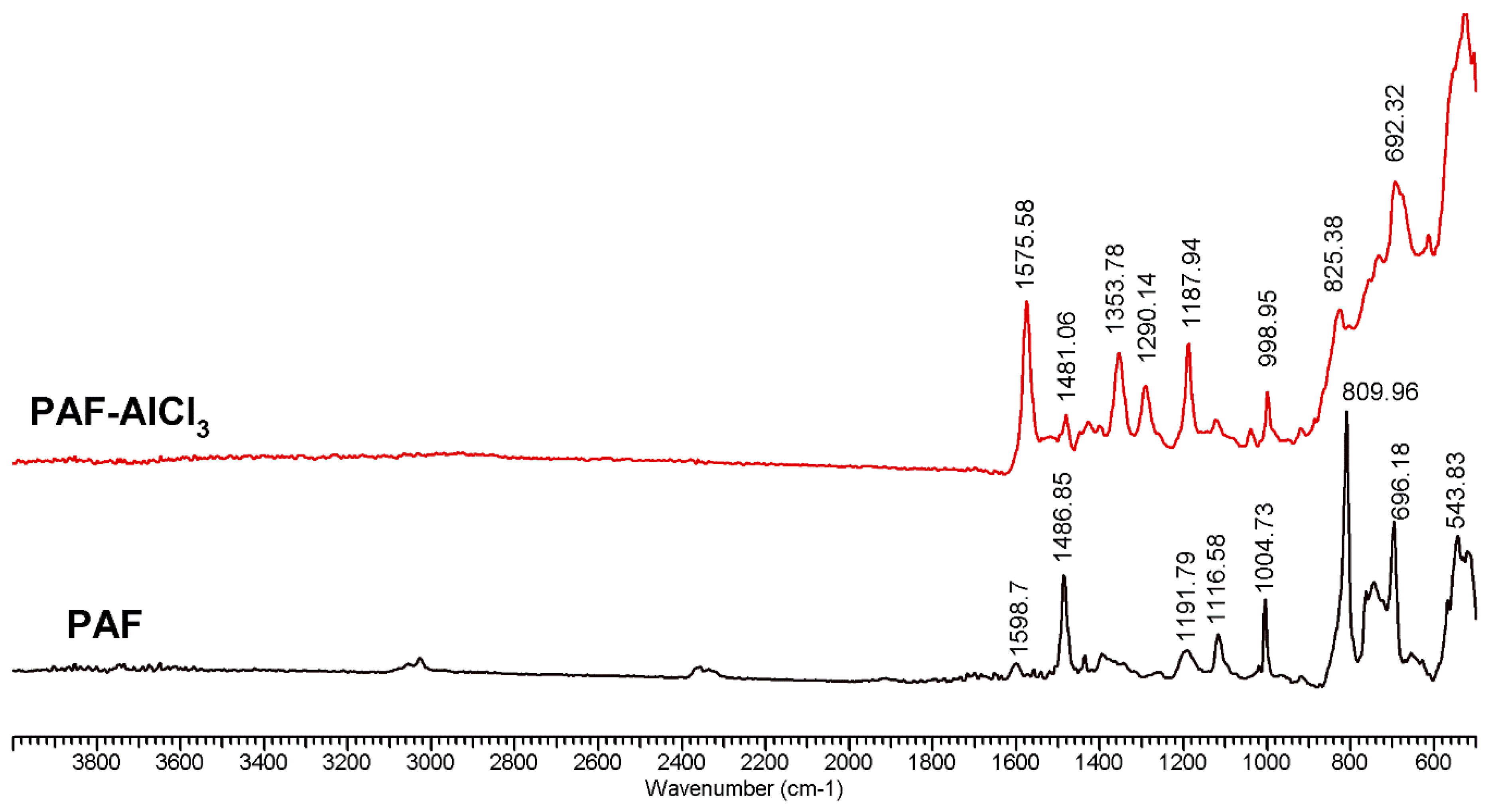
| Catalyst | , (nm) | D | fe/fc | |
|---|---|---|---|---|
| PAF-NiWS [6] | 15.2 ± 11.3 | 4.4 | 0.037 | 22.3 |
| PAF-NiWS (current work) | 9.5 ± 7.4 | 5.3 | 0.041 | 21.5 |
| Catalyst | Parameter | MeS2 | MeSxOy | Me6+ |
|---|---|---|---|---|
| PAF-NiWS | Binding energy (eV) | 32.6–4f7/2 34.8–4f5/2 | 32.9–4f7/2 35.1–4f5/2 | 36.0–4f7/2 38.1–4f5/2 |
| Weight fraction (%) | 44% | 36% | 20% | |
| PAF-NiWS [14] | Binding energy (eV) | 32.4–4f7/2 34.4–4f5/2 | 33.5–4f7/2 35.5–4f5/2 | 35.9–4f7/2 37.8–4f5/2 |
| Weight fraction (%) | 53% (13%–WSx) | 7% | 40% | |
| PAF-NiMoS | Binding energy (eV) | 229.3–3d5/2 232.3–3d3/2 | 229.6–3d5/2 233.5–3d3/2 | 232.5–3d5/2 235.7–3d3/2 |
| Weight fraction (%) | 41% | 41% | 18% | |
| PAF-NiMoS [14] | Binding energy (eV) | 229.0–3d5/2 232.1–3d3/2 | 230.0–3d5/2 233.6–3d3/2 | 232.8–3d5/2 235.7–3d3/2 |
| Weight fraction (%) | 55% | 26% | 19% |
| Parameter | Value |
|---|---|
| Sulfur content, ppm | 3500 |
| Content of monoaromatic hydrocarbons, % | 32 |
| Content of biaromatic hydrocarbons, % | 24 |
| Content of polyaromatic hydrocarbons, % | 6 |
| Total content of aromatics, % | 62 |
| Share of petroleum fraction (<180 °C), % | 15 |
| Share of diesel fraction (180 °C–360 °C), % | 72 |
| Share of heavy residue (>360 °C), % | 13 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karakhanov, E.; Maximov, A.; Kardasheva, Y.; Vinnikova, M.; Kulikov, L. Hydrotreating of Light Cycle Oil over Supported on Porous Aromatic Framework Catalysts. Catalysts 2018, 8, 397. https://doi.org/10.3390/catal8090397
Karakhanov E, Maximov A, Kardasheva Y, Vinnikova M, Kulikov L. Hydrotreating of Light Cycle Oil over Supported on Porous Aromatic Framework Catalysts. Catalysts. 2018; 8(9):397. https://doi.org/10.3390/catal8090397
Chicago/Turabian StyleKarakhanov, Eduard, Anton Maximov, Yulia Kardasheva, Maria Vinnikova, and Leonid Kulikov. 2018. "Hydrotreating of Light Cycle Oil over Supported on Porous Aromatic Framework Catalysts" Catalysts 8, no. 9: 397. https://doi.org/10.3390/catal8090397
APA StyleKarakhanov, E., Maximov, A., Kardasheva, Y., Vinnikova, M., & Kulikov, L. (2018). Hydrotreating of Light Cycle Oil over Supported on Porous Aromatic Framework Catalysts. Catalysts, 8(9), 397. https://doi.org/10.3390/catal8090397







