Recent Advances in the Direct Synthesis of Hydrogen Peroxide Using Chemical Catalysis—A Review
Abstract
1. Introduction
1.1. Industrial H2O2 Manufacture—A Historical Perspective
1.2. State of the Art in the Industrial Production of H2O2
1.3. Why Is There an Increased Interest in the Direct Synthesis of Hydrogen Peroxide?
- excessive use of solvents for the process
- a negative environmental impact owing to the production of unwanted waste
- complex and multi-step process
- mass transfer limitations and low efficiency
- transport limitations of reactants between reactors
2. The Direct Synthesis Approach to H2O2 Production Using Chemical Catalysis
2.1. Pros and Cons of the Direct Synthesis Approach to H2O2 Synthesis
2.2. Mechanism of the Direct Synthesis of H2O2
2.3. Series of Elementary Steps in H2O2 Synthesis
- Free energies of H2 and O2 adsorption are negligible under saturation conditions
- The adsorption and desorption of the H2O2 species is unrestricted
2.4. Process Conditions for the Direct Synthesis of H2O2 from H2 and O2
2.4.1. Ratio of the Gaseous Mixture
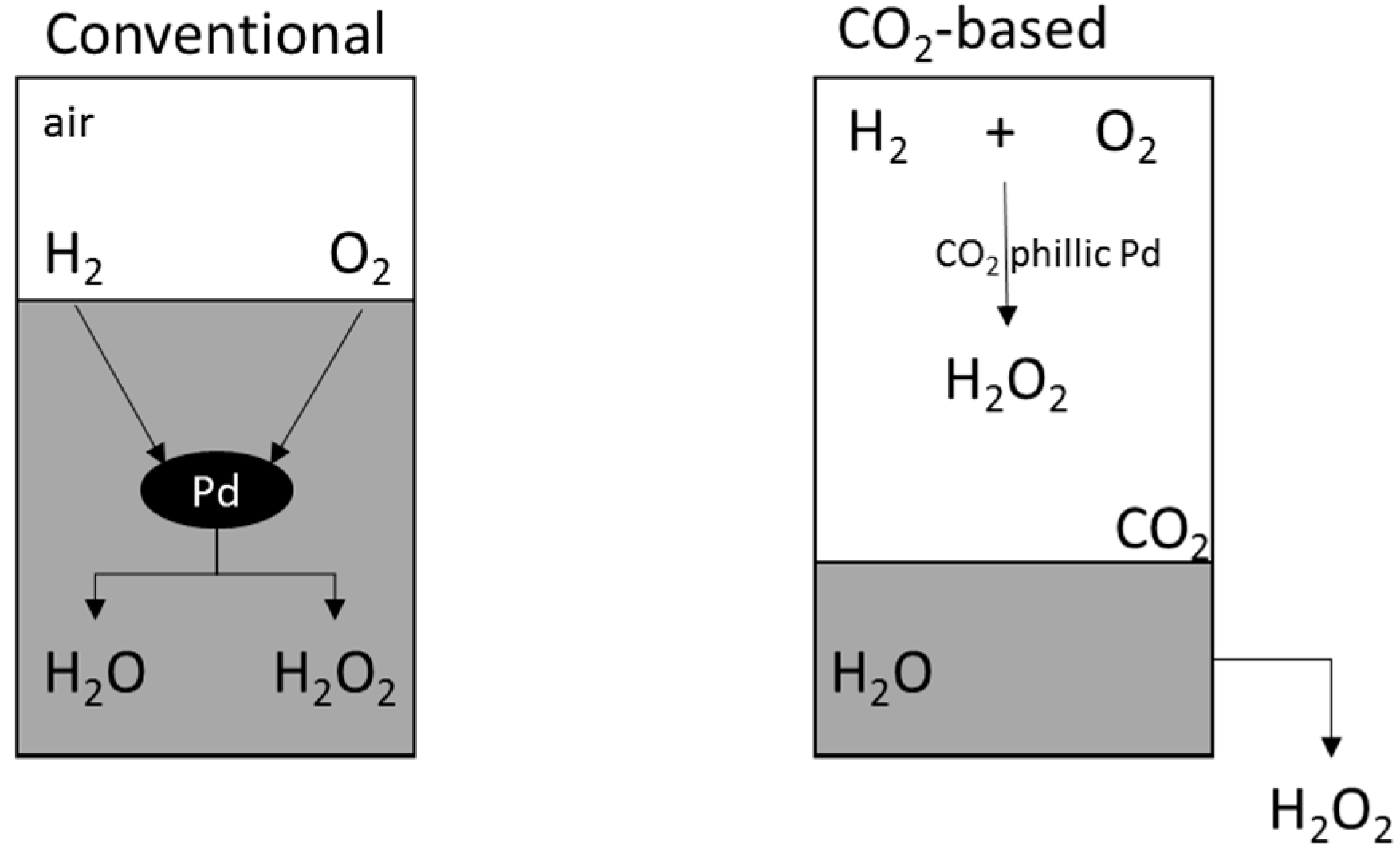
2.4.2. Reaction Medium
2.4.3. Additives/Promoters
- (i)
- oxyacids such as acetic acid, perchloric acid, phosphoric acid (H3PO4), nitric acid, and sulphuric acid (H2SO4)
- (ii)
- halide acids such as hydrochloric acid (HCl), hydrobromic acid (HBr), and hydroiodic acid (HI).
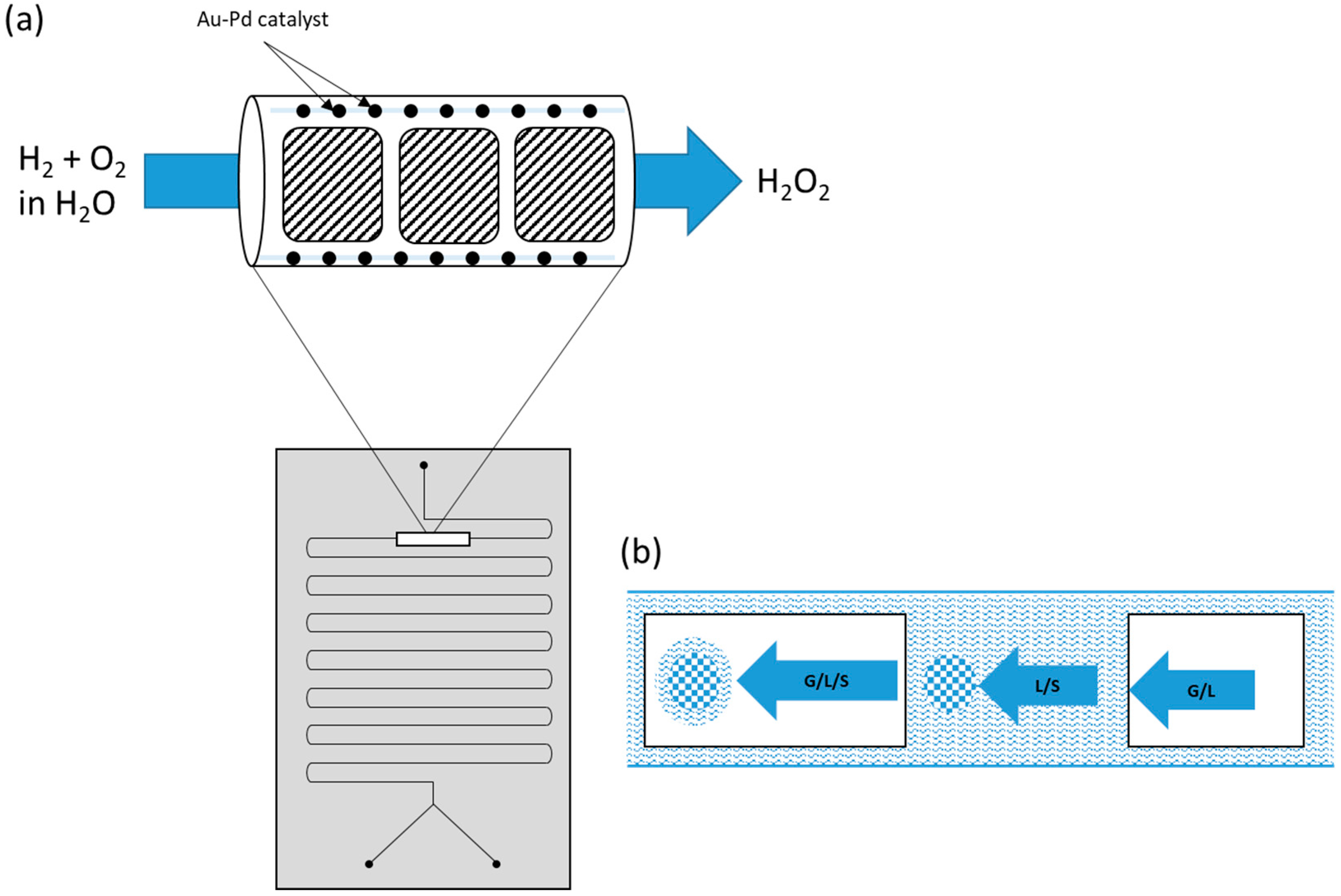
2.4.4. Reactor Design
2.4.5. Influence of the Catalytic Material
3. Summary, Conclusions and Future Perspectives
Acknowledgments
Conflicts of Interest
References
- Goor, G. Hydrogen Peroxide: Manufacture and Industrial Use for Production of Organic Chemicals. In Catalytic Oxidations with Hydrogen Peroxide as Oxidant; Strukul, G., Ed.; Springer: Dordrecht, The Netherlands, 1992; pp. 13–43. [Google Scholar]
- Eul, W.; Moeller, A.; Steiner, N. Hydrogen Peroxide. In Kirk-Othmer Encyclopedia of Chemical Technology; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2000. [Google Scholar] [CrossRef]
- Goor, G.; Glunneber, J.; Jacobi, S. Hydrogen Peroxide. In Ullmann’s Encyclopedia of Industrial Chemistry; Wiley-VCH: Weinheim, Germany, 2012. [Google Scholar] [CrossRef]
- Selinsek, M.; Deschner, B.J.; Doronkin, D.E.; Sheppard, T.L.; Grunwaldt, J.-D.; Dittmeyer, R. Revealing the Structure and Mechanism of Palladium during Direct Synthesis of Hydrogen Peroxide in Continuous Flow Using Operando Spectroscopy. ACS Catal. 2018, 8, 2546–2557. [Google Scholar] [CrossRef]
- Rodríguez-Gómez, A.; Platero, F.; Caballero, A.; Colón, G. Improving the direct synthesis of hydrogen peroxide from hydrogen and oxygen over Au-Pd/SBA-15 catalysts by selective functionalization. Mol. Catal. 2018, 445, 142–151. [Google Scholar] [CrossRef]
- Yi, Y.; Wang, L.; Li, G.; Guo, H. A review on research progress in the direct synthesis of hydrogen peroxide from hydrogen and oxygen: Noble-metal catalytic method, fuel-cell method and plasma method. Catal. Sci. Technol. 2016, 6, 1593–1610. [Google Scholar] [CrossRef]
- Campos-Martin, J.M.; Blanco-Brieva, G.; Fierro, J.L.G. Hydrogen Peroxide Synthesis: An Outlook beyond the Anthraquinone Process. Angew. Chem. Int. Ed. 2006, 45, 6962–6984. [Google Scholar] [CrossRef] [PubMed]
- Jones, C.W.; Clark, J.H. Introduction to the preparation and properties of hydrogen peroxide. In Applications of Hydrogen Peroxide and Derivatives; Jones, C.W., Clark, J.H., Eds.; The Royal Society of Chemistry: London, UK, 1999; pp. 1–36. [Google Scholar]
- Thénard, L.J. Observations sur des combinasions nouvelles entre l’oxigène et divers acides. Ann. Chim. Phys. 1818, 8, 306–313. [Google Scholar]
- Thènard, L.J. Nouvelles Observations sur les Acides et les Oxides oxigénés. Ann. Chim. Phys. 1818, 9, 51–56. [Google Scholar]
- Meidinger, H. Ueber voltametrische Messungen. Justus Liebigs Annalen der Chemie 1853, 88, 57–81. [Google Scholar] [CrossRef]
- Berthelot, M. Sur la formation de l’eau oxygénée, de l’ozone et de l’acide persulfurique pendant l’électrolyse. Comptes Rendus de l’Académie des Sciences 1878, 86, 71–76. [Google Scholar]
- Manchot, W. Ueber Sauerstoffactivirung. Justus Liebigs Annalen der Chemie 1901, 314, 177–199. [Google Scholar] [CrossRef]
- Manchot, W.; Herzog, J. Ueber die Oxydation des Indigweisses durch Sauerstoffgas. Justus Liebigs Annalen der Chemie 1901, 316, 318–330. [Google Scholar] [CrossRef]
- Manchot, W.; Herzog, J. Die Autoxydation des Hydrazobenzols. Justus Liebigs Annalen der Chemie 1901, 316, 331–332. [Google Scholar] [CrossRef]
- Comyns, A.E. Encyclopedic Dictionary of Named Processes in Chemical Technology, 4th ed.; CRC Press: Boca Raton, FL, USA, 2014. [Google Scholar]
- Wendt, H.; Kreysa, G. Industrial Processes. In Electrochemical Engineering: Science and Technology in Chemical and Other Industries; Wendt, H., Kreysa, G., Eds.; Springer: Berlin/Heidelberg, Germany, 1999; pp. 290–369. [Google Scholar]
- Henkel, H.; Weber, W. Manufacture of Hydrogen Peroxide. U.S. Patent 1,108,752, 25 August 1914. [Google Scholar]
- Walton, J.H.; Filson, G.W. The Direct Preparation of Hydrogen Peroxide in a High Concentration. J. Am. Chem. Soc. 1932, 54, 3228–3229. [Google Scholar] [CrossRef]
- Von Schickh, O. Herstellung von Peroxyden durch Autoxydation. Geschichtliche Entwicklung. Chem. Ing. Tech. 1960, 32, 462. [Google Scholar] [CrossRef]
- Hans-Joachim, R.; Georg, P. Production of Hydrogen Peroxide. U.S. Patent 2,215,883, 24 September 1940. [Google Scholar]
- Rust, F.F. Manufacture of Hydrogen Peroxide. U.S. Patent 2,871,104, 27 January 1959. [Google Scholar]
- Berl, E. A new cathodic process for the production of H2O2. Trans. Electrochem. Soc. 1939, 76, 359–369. [Google Scholar] [CrossRef]
- Li, H.; Zheng, B.; Pan, Z.; Zong, B.; Qiao, M. Advances in the slurry reactor technology of the anthraquinone process for H2O2 production. Front. Chem. Sci. Eng. 2017, 12, 124–131. [Google Scholar] [CrossRef]
- Ciriminna, R.; Albanese, L.; Meneguzzo, F.; Pagliaro, M. Hydrogen Peroxide: A Key Chemical for Today’s Sustainable Development. ChemSusChem 2016, 9, 3374–3381. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Serna, J.; Moreno, T.; Biasi, P.; Cocero, M.J.; Mikkola, J.-P.; Salmi, T.O. Engineering in direct synthesis of hydrogen peroxide: Targets, reactors and guidelines for operational conditions. Green Chem. 2014, 16, 2320–2343. [Google Scholar] [CrossRef]
- Centi, G.; Perathoner, S.; Abate, S. Direct Synthesis of Hydrogen Peroxide: Recent Advances. In Modern Heterogeneous Oxidation Catalysis; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2009; pp. 253–287. [Google Scholar]
- Hancu, D.; Beckman, E.J. Generation of hydrogen peroxide directly from H2 and O2 using CO2 as the solvent. Green Chem. 2001, 3, 80–86. [Google Scholar] [CrossRef]
- Poliakoff, M.; Fitzpatrick, J.M.; Farren, T.R.; Anastas, P.T. Green Chemistry: Science and Politics of Change. Science 2002, 297, 807–810. [Google Scholar] [CrossRef] [PubMed]
- Linthorst, J.A. An overview: Origins and development of green chemistry. Found. Chem. 2010, 12, 55–68. [Google Scholar] [CrossRef]
- Centi, G.; Perathoner, S. Catalysis and sustainable (green) chemistry. Catal. Today 2003, 77, 287–297. [Google Scholar] [CrossRef]
- Centi, G.; Perathoner, S. From Green to Sustainable Industrial Chemistry. In Sustainable Industrial Chemistry; Cavani, F., Centi, G., Perathoner, S., Trifiro, F., Eds.; Wiley-VCH: Weinheim, Germany, 2009. [Google Scholar]
- Sheldon, R. Atom Utilization, E factors and the Catalytic Solution. Surf. Chem. Catal. 2000, 3, 541–551. [Google Scholar]
- Li, C.-J.; Trost, B.M. Green chemistry for chemical synthesis. Proc. Natl. Acad. Sci. USA 2008, 105, 13197–13202. [Google Scholar] [CrossRef] [PubMed]
- Sheldon, R.A. Fundamentals of green chemistry: Efficiency in reaction design. Chem. Soc. Rev. 2012, 41, 1437–1451. [Google Scholar] [CrossRef] [PubMed]
- Crole, D.A.; Freakley, S.J.; Edwards, J.K.; Hutchings, G.J. Direct synthesis of hydrogen peroxide in water at ambient temperature. Proc. R. Soc. A 2016, 472. [Google Scholar] [CrossRef] [PubMed]
- Freakley, S.J.; He, Q.; Harrhy, J.H.; Lu, L.; Crole, D.A.; Morgan, D.J.; Ntainjua, E.N.; Edwards, J.K.; Carley, A.F.; Borisevich, A.Y.; et al. Palladium-tin catalysts for the direct synthesis of H2O2 with high selectivity. Science. 2016, 351, 965. [Google Scholar] [CrossRef] [PubMed]
- Flaherty, D.W. Direct Synthesis of H2O2 from H2 and O2 on Pd Catalysts: Current Understanding, Outstanding Questions, and Research Needs. ACS Catal. 2018, 8, 1520–1527. [Google Scholar] [CrossRef]
- Wilson, N.M.; Priyadarshini, P.; Kunz, S.; Flaherty, D.W. Direct synthesis of H2O2 on Pd and AuxPd1 clusters: Understanding the effects of alloying Pd with Au. J. Catal. 2018, 357, 163–175. [Google Scholar] [CrossRef]
- Wilson, N.M.; Flaherty, D.W. Mechanism for the Direct Synthesis of H2O2 on Pd Clusters: Heterolytic Reaction Pathways at the Liquid–Solid Interface. J. Am. Chem. Soc. 2016, 138, 574–586. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Lunsford, J.H. Controlling factors in the direct formation of H2O2 from H2 and O2 over a Pd/SiO2 catalyst in ethanol. Appl. Catal. A 2006, 314, 94–100. [Google Scholar] [CrossRef]
- Moreno, T.; Garcia-Serna, J.; Cocero, M.J. Direct synthesis of hydrogen peroxide in methanol and water using scCO2 and N2 as diluents. Green Chem. 2010, 12, 282–289. [Google Scholar] [CrossRef]
- Gervasini, A.; Carniti, P.; Desmedt, F.; Miquel, P. Liquid Phase Direct Synthesis of H2O2: Activity and Selectivity of Pd-Dispersed Phase on Acidic Niobia-Silica Supports. ACS Catal. 2017, 7, 4741–4752. [Google Scholar] [CrossRef]
- Khan, Z.; Dummer, N.F.; Edwards, J.K. Silver–palladium catalysts for the direct synthesis of hydrogen peroxide. Philos. Trans. R. Soc. A 2018, 376. [Google Scholar] [CrossRef] [PubMed]
- Seo, M.; Lee, D.-W.; Han, S.S.; Lee, K.-Y. Direct Synthesis of Hydrogen Peroxide from Hydrogen and Oxygen over Mesoporous Silica-Shell-Coated, Palladium-Nanocrystal-Grafted SiO2 Nanobeads. ACS Catal. 2017, 7, 3039–3048. [Google Scholar] [CrossRef]
- Bianchi, D.; Bortolo, R.; D’Aloisio, R.; Ricci, M. Biphasic Synthesis of Hydrogen Peroxide from Carbon Monoxide, Water, and Oxygen Catalyzed by Palladium Complexes with Bidentate Nitrogen Ligands. Angew. Chem. Int. Ed. 1999, 38, 706–708. [Google Scholar] [CrossRef]
- Zudin, V.; Likholobov, V.; Ermakov, Y.I. Catalytic synthesis of hydrogen peroxide from oxygen and water in the presence of carbon monoxide and phosphine complexes of palladium. Kinet. Catal. (USSR) (Engl. Transl.) 1980, 20, 1599–1600. [Google Scholar]
- Thiel Werner, R. New Routes to Hydrogen Peroxide: Alternatives for Established Processes? Angew. Chem. Int. Ed. 1999, 38, 3157–3158. [Google Scholar] [CrossRef]
- Stahl, S.S.; Thorman, J.L.; Nelson, R.C.; Kozee, M.A. Oxygenation of Nitrogen-Coordinated Palladium(0): Synthetic, Structural, and Mechanistic Studies and Implications for Aerobic Oxidation Catalysis. J. Am. Chem. Soc. 2001, 123, 7188–7189. [Google Scholar] [CrossRef] [PubMed]
- Chinta, S.; Lunsford, J.H. A mechanistic study of H2O2 and H2O formation from H2 and O2 catalyzed by palladium in an aqueous medium. J. Catal. 2004, 225, 249–255. [Google Scholar] [CrossRef]
- Plauck, A.; Stangland, E.E.; Dumesic, J.A.; Mavrikakis, M. Active sites and mechanisms for H2O2 decomposition over Pd catalysts. Proc. Natl. Acad. Sci. USA 2016, 113, E1973–E1982. [Google Scholar] [CrossRef] [PubMed]
- Yi, Y.; Xu, C.; Wang, L.; Yu, J.; Zhu, Q.; Sun, S.; Tu, X.; Meng, C.; Zhang, J.; Guo, H. Selectivity control of H2/O2 plasma reaction for direct synthesis of high purity H2O2 with desired concentration. Chem. Eng. J. 2017, 313, 37–46. [Google Scholar] [CrossRef]
- Dittmeyer, R.; Grunwaldt, J.D.; Pashkova, A. A review of catalyst performance and novel reaction engineering concepts in direct synthesis of hydrogen peroxide. Catal. Today 2015, 248, 149–159. [Google Scholar] [CrossRef]
- Samanta, C. Direct synthesis of hydrogen peroxide from hydrogen and oxygen: An overview of recent developments in the process. Appl. Catal. A 2008, 350, 133–149. [Google Scholar] [CrossRef]
- Edwards, J.K.; Freakley, S.J.; Lewis, R.J.; Pritchard, J.C.; Hutchings, G.J. Advances in the direct synthesis of hydrogen peroxide from hydrogen and oxygen. Catal. Today 2015, 248, 3–9. [Google Scholar] [CrossRef]
- Kolehmainen, E.; Turunen, I. Direct synthesis of hydrogen peroxide in microreactors. Russ. J. Gen. Chem. 2012, 82, 2100–2107. [Google Scholar] [CrossRef]
- Lewis, B.; von Elbe, G. The Reaction between Hydrogen and Oxygen. In Combustion, Flames and Explosions of Gases, 3rd ed.; Academic Press: San Diego, CA, USA, 1987; pp. 25–77. [Google Scholar]
- Biasi, P.; Sterchele, S.; Bizzotto, F.; Manzoli, M.; Lindholm, S.; Ek, P.; Bobacka, J.; Mikkola, J.-P.; Salmi, T. Application of the Catalyst Wet Pretreatment Method (CWPM) for catalytic direct synthesis of H2O2. Catal. Today 2015, 246, 207–215. [Google Scholar] [CrossRef]
- Edwards, J.K.; Solsona, B.; Ntainjua, E.; Carley, A.F.; Herzing, A.A.; Kiely, C.J.; Hutchings, G.J. Switching Off Hydrogen Peroxide Hydrogenation in the Direct Synthesis Process. Science 2009, 323, 1037–1041. [Google Scholar] [CrossRef] [PubMed]
- Chung, Y.-M.; Lee, Y.-R.; Ahn, W.-S. Direct Synthesis of Hydrogen Peroxide from Hydrogen and Oxygen over Pd-supported Metal-Organic Framework Catalysts. Bull. Korean Chem. Soc. 2015, 36, 1378–1383. [Google Scholar] [CrossRef]
- Lari, G.M.; Puértolas, B.; Shahrokhi, M.; López, N.; Pérez-Ramírez, J. Hybrid Palladium Nanoparticles for Direct Hydrogen Peroxide Synthesis: The Key Role of the Ligand. Angew. Chem. 2017, 129, 1801–1805. [Google Scholar] [CrossRef]
- Seo, M.; Kim, H.J.; Han, S.S.; Lee, K.-Y. Direct Synthesis of Hydrogen Peroxide from Hydrogen and Oxygen Using Tailored Pd Nanocatalysts: A Review of Recent Findings. Catal. Surv. Asia 2017, 21, 1–12. [Google Scholar] [CrossRef]
- Urban, S.; Weltin, A.; Flamm, H.; Kieninger, J.; Deschner, B.J.; Kraut, M.; Dittmeyer, R.; Urban, G.A. Electrochemical multisensor system for monitoring hydrogen peroxide, hydrogen and oxygen in direct synthesis microreactors. Sens. Actuators, B 2018, 273, 973–982. [Google Scholar] [CrossRef]
- Lu, Z.; Chen, G.; Siahrostami, S.; Chen, Z.; Liu, K.; Xie, J.; Liao, L.; Wu, T.; Lin, D.; Liu, Y.; et al. High-efficiency oxygen reduction to hydrogen peroxide catalysed by oxidized carbon materials. Nat. Catal. 2018, 1, 156–162. [Google Scholar] [CrossRef]
- Abate, S.; Perathoner, S.; Centi, G. Deactivation mechanism of Pd supported on ordered and non-ordered mesoporous silica in the direct H2O2 synthesis using CO2-expanded methanol. Catal. Today 2012, 179, 170–177. [Google Scholar] [CrossRef]
- Hâncu, D.; Green, J.; Beckman, E.J. H2O2 in CO2: Sustainable Production and Green Reactions. Acc. Chem. Res. 2002, 35, 757–764. [Google Scholar] [CrossRef] [PubMed]
- Landon, P.; Collier, P.J.; Carley, A.F.; Chadwick, D.; Papworth, A.J.; Burrows, A.; Kiely, C.J.; Hutchings, G.J. Direct synthesis of hydrogen peroxide from H2 and O2 using Pd and Au catalysts. Phys. Chem. Chem. Phys. 2003, 5, 1917–1923. [Google Scholar] [CrossRef]
- Gallina, G.; García-Serna, J.; Salmi, T.O.; Canu, P.; Biasi, P. Bromide and Acids: A Comprehensive Study on Their Role on the Hydrogen Peroxide Direct Synthesis. Ind. Eng. Chem. Res. 2017, 56, 13367–13378. [Google Scholar] [CrossRef]
- Lewis, R.J.; Edwards, J.K.; Freakley, S.J.; Hutchings, G.J. Solid Acid Additives as Recoverable Promoters for the Direct Synthesis of Hydrogen Peroxide. Ind. Eng. Chem. Res. 2017, 56, 13287–13293. [Google Scholar] [CrossRef]
- Watts, P.; Wiles, C. Recent advances in synthetic micro reaction technology. Chem. Commun. 2007, 443–467. [Google Scholar] [CrossRef] [PubMed]
- Shang, M.; Hessel, V. Synthesis and Application of H2O2 in Flow Reactors. In Sustainable Flow Chemistry; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2017; pp. 43–72. [Google Scholar]
- Kanungo, S.; Paunovic, V.; Schouten, J.C.; Neira D’Angelo, M.F. Facile Synthesis of Catalytic AuPd Nanoparticles within Capillary Microreactors Using Polyelectrolyte Multilayers for the Direct Synthesis of H2O2. Nano Lett. 2017, 17, 6481–6486. [Google Scholar] [CrossRef] [PubMed]
- Paunovic, V.; Schouten, J.C.; Nijhuis, T.A. Direct synthesis of hydrogen peroxide in a wall-coated microchannel reactor over Au–Pd catalyst: A performance study. Catal. Today 2015, 248, 160–168. [Google Scholar] [CrossRef]
- Voloshin, Y.; Lawal, A. Overall kinetics of hydrogen peroxide formation by direct combination of H2 and O2 in a microreactor. Chem. Eng. Sci. 2010, 65, 1028–1036. [Google Scholar] [CrossRef]
- Ng, J.F.; Nie, Y.; Chuah, G.K.; Jaenicke, S. A wall-coated catalytic capillary microreactor for the direct formation of hydrogen peroxide. J. Catal. 2010, 269, 302–308. [Google Scholar] [CrossRef]
- Biasi, P.; Menegazzo, F.; Pinna, F.; Eränen, K.; Canu, P.; Salmi, T.O. Hydrogen Peroxide Direct Synthesis: Selectivity Enhancement in a Trickle Bed Reactor. Ind. Eng. Chem. Res. 2010, 49, 10627–10632. [Google Scholar] [CrossRef]
- Biasi, P.; Menegazzo, F.; Pinna, F.; Eränen, K.; Salmi, T.O.; Canu, P. Continuous H2O2 direct synthesis over PdAu catalysts. Chem. Eng. J. 2011, 176–177, 172–177. [Google Scholar] [CrossRef]
- Biasi, P.; Gemo, N.; Hernández Carucci, J.R.; Eränen, K.; Canu, P.; Salmi, T.O. Kinetics and Mechanism of H2O2 Direct Synthesis over a Pd/C Catalyst in a Batch Reactor. Ind. Eng. Chem. Res. 2012, 51, 8903–8912. [Google Scholar] [CrossRef]
- Biasi, P.; Garcia-Serna, J.; Bittante, A.; Salmi, T. Direct synthesis of hydrogen peroxide in water in a continuous trickle bed reactor optimized to maximize productivity. Green Chem. 2013, 15, 2502–2513. [Google Scholar] [CrossRef]
- Biasi, P.; Serna, J.G.; Salmi, T.O.; Mikkola, J.-M. Hydrogen Peroxide Direct Synthesis: Enhancement of Selectivity and Production with non-Conventional Methods. Chem. Eng. Trans. 2013, 32, 673–678. [Google Scholar]
- Abejón, R.; Abejón, A.; Biasi, P.; Gemo, N.; Garea, A.; Salmi, T.; Irabien, J.A. Hydrogen peroxide obtained via direct synthesis as alternative raw material for ultrapurification process to produce electronic grade chemical. J. Chem. Technol. Biotechnol. 2016, 91, 1136–1148. [Google Scholar] [CrossRef]
- Edwards, J.K.; Carley, A.F.; Herzing, A.A.; Kiely, C.J.; Hutchings, G.J. Direct synthesis of hydrogen peroxide from H2 and O2 using supported Au-Pd catalysts. Faraday Discuss. 2008, 138, 225–239. [Google Scholar] [CrossRef] [PubMed]
- Ntainjua, E.N.; Freakley, S.J.; Hutchings, G.J. Direct Synthesis of Hydrogen Peroxide Using Ruthenium Catalysts. Top. Catal. 2012, 55, 718–722. [Google Scholar] [CrossRef]
- Gu, J.; Wang, S.; He, Z.; Han, Y.; Zhang, J. Direct synthesis of hydrogen peroxide from hydrogen and oxygen over activated-carbon-supported Pd-Ag alloy catalysts. Catal. Sci. Technol. 2016, 6, 809–817. [Google Scholar] [CrossRef]
- Tian, P.; Xu, X.; Ao, C.; Ding, D.; Li, W.; Si, R.; Tu, W.; Xu, J.; Han, Y.-F. Direct and Selective Synthesis of Hydrogen Peroxide over Palladium–Tellurium Catalysts at Ambient Pressure. ChemSusChem 2017, 10, 3342–3346. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Gao, K.; Li, W.; Zhang, J. Effect of Zn addition on the direct synthesis of hydrogen peroxide over supported palladium catalysts. Appl. Catal. A 2017, 531, 89–95. [Google Scholar] [CrossRef]
- Xu, H.; Cheng, D.; Gao, Y. Design of High-Performance Pd-Based Alloy Nanocatalysts for Direct Synthesis of H2O2. ACS Catal. 2017, 7, 2164–2170. [Google Scholar] [CrossRef]
- Tian, P.; Ouyang, L.; Xu, X.; Ao, C.; Xu, X.; Si, R.; Shen, X.; Lin, M.; Xu, J.; Han, Y.-F. The origin of palladium particle size effects in the direct synthesis of H2O2: Is smaller better? J. Catal. 2017, 349, 30–40. [Google Scholar] [CrossRef]
- Howe, A.; Miedziak, P.; Morgan, D.J.; He, Q.; Strasser, P.; Edwards, J. One pot microwave synthesis of highly stable AuPd@Pd supported core-shell nanoparticles. Faraday Discuss. 2018. [Google Scholar] [CrossRef] [PubMed]
- Desmedt, F.; Vlasselaer, Y.; Miquel, P. Method for the Direct Synthesis of Hydrogen Peroxide. U.S. Pantent 9,663,365, 30 May 2017. [Google Scholar]
- Inoue, T.; Tanaka, Y.; Pacheco Tanaka, D.A.; Suzuki, T.M.; Sato, K.; Nishioka, M.; Hamakawa, S.; Mizukami, F. Direct production of hydrogen peroxide from oxygen and hydrogen applying membrane-permeation mechanism. Chem. Eng. Sci. 2010, 65, 436–440. [Google Scholar] [CrossRef]
- Park, S.; Park, D.R.; Choi, J.H.; Kim, T.J.; Chung, Y.-M.; Oh, S.-H.; Song, I.K. Direct synthesis of hydrogen peroxide from hydrogen and oxygen over insoluble Cs2.5H0.5PW12O40 heteropolyacid supported on Pd/MCF. J. Mol. Catal. A 2010, 332, 76–83. [Google Scholar] [CrossRef]
- Pashkova, A.; Dittmeyer, R.; Kaltenborn, N.; Richter, H. Experimental study of porous tubular catalytic membranes for direct synthesis of hydrogen peroxide. Chem. Eng. J. 2010, 165, 924–933. [Google Scholar] [CrossRef]
- Pritchard, J.C.; He, Q.; Ntainjua, E.N.; Piccinini, M.; Edwards, J.K.; Herzing, A.A.; Carley, A.F.; Moulijn, J.A.; Kiely, C.J.; Hutchings, G.J. The effect of catalyst preparation method on the performance of supported Au-Pd catalysts for the direct synthesis of hydrogen peroxide. Green Chem. 2010, 12, 915–921. [Google Scholar] [CrossRef]
- Biasi, P.; Canu, P.; Menegazzo, F.; Pinna, F.; Salmi, T.O. Direct Synthesis of Hydrogen Peroxide in a Trickle Bed Reactor: Comparison of Pd-Based Catalysts. Ind. Eng. Chem. Res. 2012, 51, 8883–8890. [Google Scholar] [CrossRef]
- Gemo, N.; Biasi, P.; Canu, P.; Salmi, T.O. Mass transfer and kinetics of H2O2 direct synthesis in a batch slurry reactor. Chem. Eng. J. 2012, 207–208, 539–551. [Google Scholar] [CrossRef]
- Kilpiö, T.; Biasi, P.; Bittante, A.; Salmi, T.; Wärnå, J. Modeling of Direct Synthesis of Hydrogen Peroxide in a Packed-Bed Reactor. Ind. Eng. Chem. Res. 2012, 51, 13366–13378. [Google Scholar] [CrossRef]
- Kim, J.; Chung, Y.-M.; Kang, S.-M.; Choi, C.-H.; Kim, B.-Y.; Kwon, Y.-T.; Kim, T.J.; Oh, S.-H.; Lee, C.-S. Palladium Nanocatalysts Immobilized on Functionalized Resin for the Direct Synthesis of Hydrogen Peroxide from Hydrogen and Oxygen. ACS Catal. 2012, 2, 1042–1048. [Google Scholar] [CrossRef]
- Menegazzo, F.; Signoretto, M.; Frison, G.; Pinna, F.; Strukul, G.; Manzoli, M.; Boccuzzi, F. When high metal dispersion has a detrimental effect: Hydrogen peroxide direct synthesis under very mild and nonexplosive conditions catalyzed by Pd supported on silica. J. Catal. 2012, 290, 143–150. [Google Scholar] [CrossRef]
- Park, S.; Lee, J.; Song, J.H.; Kim, T.J.; Chung, Y.-M.; Oh, S.-H.; Song, I.K. Direct synthesis of hydrogen peroxide from hydrogen and oxygen over Pd/HZSM-5 catalysts: Effect of Brönsted acidity. J. Mol. Catal. A 2012, 363–364, 230–236. [Google Scholar] [CrossRef]
- Piccinini, M.; Edwards, J.K.; Moulijn, J.A.; Hutchings, G.J. Influence of reaction conditions on the direct synthesis of hydrogen peroxide over AuPd/carbon catalysts. Catal. Sci. Technol. 2012, 2, 1908–1913. [Google Scholar] [CrossRef]
- Rossi, U.; Zancanella, S.; Artiglia, L.; Granozzi, G.; Canu, P. Direct synthesis of H2O2 on model Pd surfaces. Chem. Eng. J. 2012, 207–208, 845–850. [Google Scholar] [CrossRef]
- Freakley, S.J.; Piccinini, M.; Edwards, J.K.; Ntainjua, E.N.; Moulijn, J.A.; Hutchings, G.J. Effect of Reaction Conditions on the Direct Synthesis of Hydrogen Peroxide with a AuPd/TiO2 Catalyst in a Flow Reactor. ACS Catal. 2013, 3, 487–501. [Google Scholar] [CrossRef]
- Gudarzi, D.; Ratchananusorn, W.; Turunen, I.; Salmi, T.; Heinonen, M. Preparation and Study of Pd Catalysts Supported on Activated Carbon Cloth (ACC) for Direct Synthesis of H2O2 from H2 and O2. Top. Catal. 2013, 56, 527–539. [Google Scholar] [CrossRef]
- Abate, S.; Arrigo, R.; Perathoner, S.; Centi, G. Role of Feed Composition on the Performances of Pd-Based Catalysts for the Direct Synthesis of H2O2. Top. Catal. 2014, 57, 1208–1217. [Google Scholar] [CrossRef]
- Arrigo, R.; Schuster, M.E.; Abate, S.; Wrabetz, S.; Amakawa, K.; Teschner, D.; Freni, M.; Centi, G.; Perathoner, S.; Hävecker, M.; et al. Dynamics of Palladium on Nanocarbon in the Direct Synthesis of H2O2. ChemSusChem 2014, 7, 179–194. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Lee, D.-W.; Lee, K.-Y. Direct synthesis of hydrogen peroxide from hydrogen and oxygen over single-crystal cubic palladium on silica catalysts. J. Mol. Catal. A 2014, 383–384, 64–69. [Google Scholar] [CrossRef]
- Ratchananusorn, W.; Gudarzi, D.; Turunen, I. Catalytic direct synthesis of hydrogen peroxide in a novel microstructured reactor. Chem. Eng. Process. 2014, 84, 24–30. [Google Scholar] [CrossRef]
- Torrente-Murciano, L.; He, Q.; Hutchings, G.J.; Kiely, C.J.; Chadwick, D. Enhanced Au-Pd Activity in the Direct Synthesis of Hydrogen Peroxide using Nanostructured Titanate Nanotube Supports. ChemCatChem 2014, 6, 2531–2534. [Google Scholar] [CrossRef]
- Inoue, T.; Adachi, J.; Ohtaki, K.; Lu, M.; Murakami, S.; Sun, X.; Wang, D.F. Direct hydrogen peroxide synthesis using glass microfabricated reactor–Paralleled packed bed operation. Chem. Eng. J. 2015, 278, 517–526. [Google Scholar] [CrossRef]
- Paunovic, V.; Ordomsky, V.V.; Sushkevich, V.L.; Schouten, J.C.; Nijhuis, T.A. Direct Synthesis of Hydrogen Peroxide over Au-Pd Catalyst—The Effect of Co-Solvent Addition. ChemCatChem 2015, 7, 1161–1176. [Google Scholar] [CrossRef]
- Paunovic, V.; Schouten, J.C.; Nijhuis, T.A. Direct synthesis of hydrogen peroxide using concentrated H2 and O2 mixtures in a wall-coated microchannel–kinetic study. Appl. Catal. A 2015, 505, 249–259. [Google Scholar] [CrossRef]
- Akram, A.; Freakley, S.J.; Reece, C.; Piccinini, M.; Shaw, G.; Edwards, J.K.; Desmedt, F.; Miquel, P.; Seuna, E.; Willock, D.J.; et al. Gas phase stabiliser-free production of hydrogen peroxide using supported gold-palladium catalysts. Chem. Sci. 2016, 7, 5833–5837. [Google Scholar] [CrossRef] [PubMed]
- Blanco-Brieva, G.; Montiel-Argaiz, M.; Desmedt, F.; Miquel, P.; Campos-Martin, J.M.; Fierro, J.L.G. Direct synthesis of hydrogen peroxide with no ionic halides in solution. RSC Adv. 2016, 6, 99291–99296. [Google Scholar] [CrossRef]
- Sierra-Salazar, A.F.; Li, W.S.J.; Bathfield, M.; Ayral, A.; Abate, S.; Chave, T.; Nikitenko, S.I.; Hulea, V.; Perathoner, S.; Lacroix-Desmazes, P. Hierarchically porous Pd/SiO2 catalyst by combination of miniemulsion polymerisation and sol-gel method for the direct synthesis of H2O2. Catal. Today 2016, 306, 16–22. [Google Scholar] [CrossRef]
- Han, G.-H.; Seo, M.; Cho, Y.-H.; Han, S.S.; Lee, K.-Y. Highly dispersed Pd catalysts prepared by a sonochemical method for the direct synthesis of hydrogen peroxide. Mol. Catal. 2017, 429, 43–50. [Google Scholar] [CrossRef]
- Hirama, H.; Yoshioka, H.; Matsumoto, Y.; Amada, T.; Hori, Y.; Ohtaki, K.; Lu, M.; Inoue, T. Design, Fabrication, and Performance of an Optimized Flow Reactor with Parallel Micropacked Beds. Ind. Eng. Chem. Res. 2017, 56, 14200–14206. [Google Scholar] [CrossRef]
- Tu, R.; Li, L.; Zhang, S.; Chen, S.; Li, J.; Lu, X. Carbon-Modified Mesoporous Anatase/TiO2(B) Whisker for Enhanced Activity in Direct Synthesis of Hydrogen Peroxide by Palladium. Catalysts 2017, 7, 175. [Google Scholar] [CrossRef]
- Yook, S.; Kwon, H.C.; Kim, Y.-G.; Choi, W.; Choi, M. Significant Roles of Carbon Pore and Surface Structure in AuPd/C Catalyst for Achieving High Chemoselectivity in Direct Hydrogen Peroxide Synthesis. ACS Sustain. Chem. Eng. 2017, 5, 1208–1216. [Google Scholar] [CrossRef]
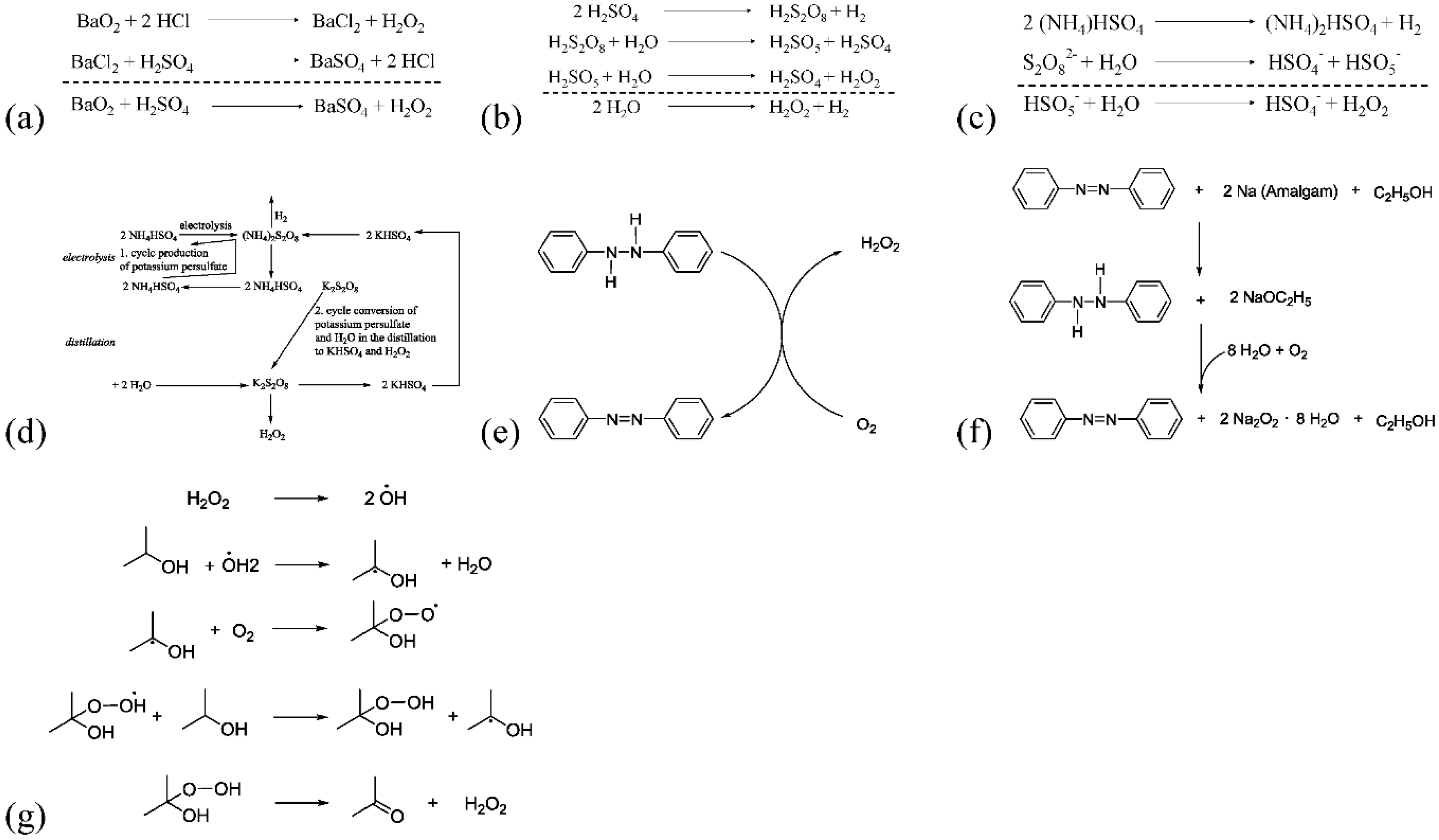
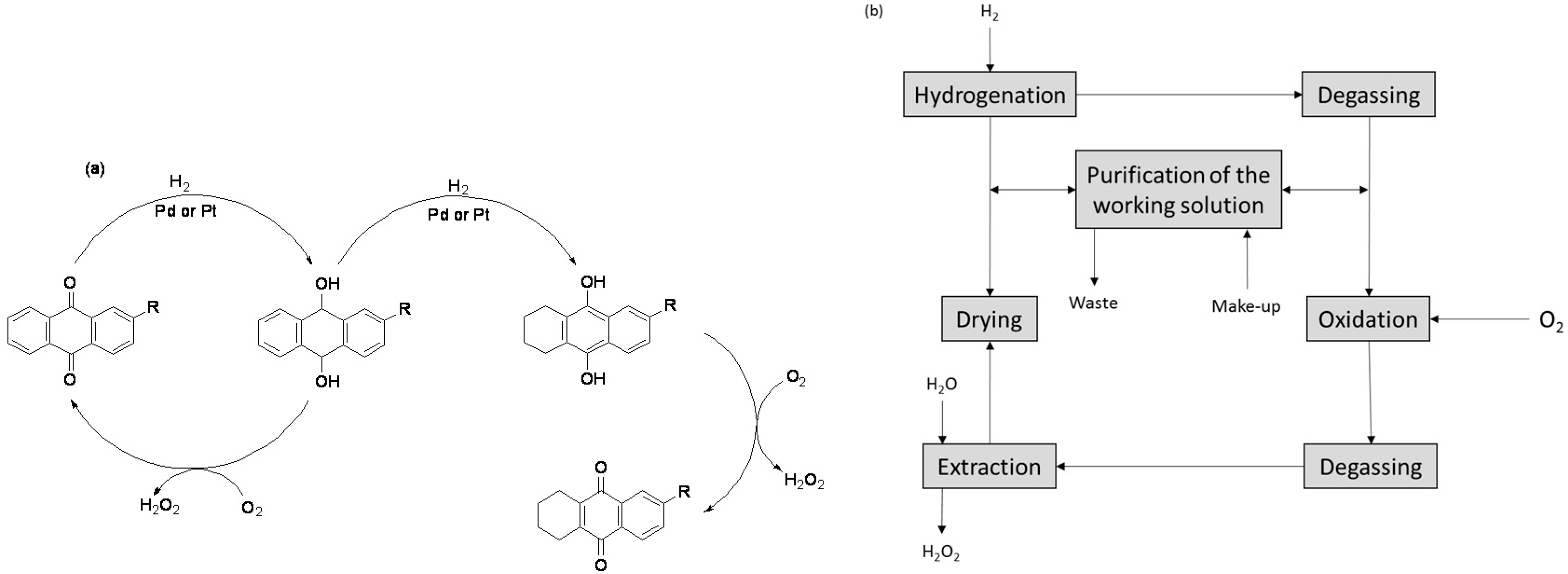
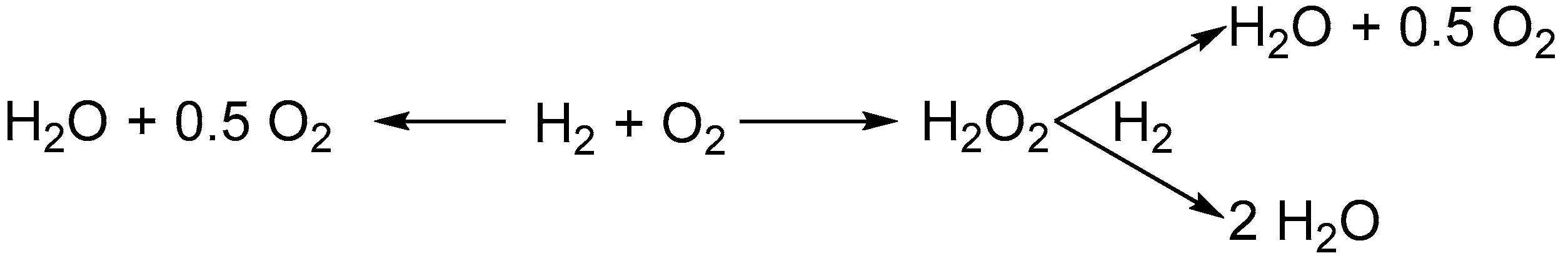

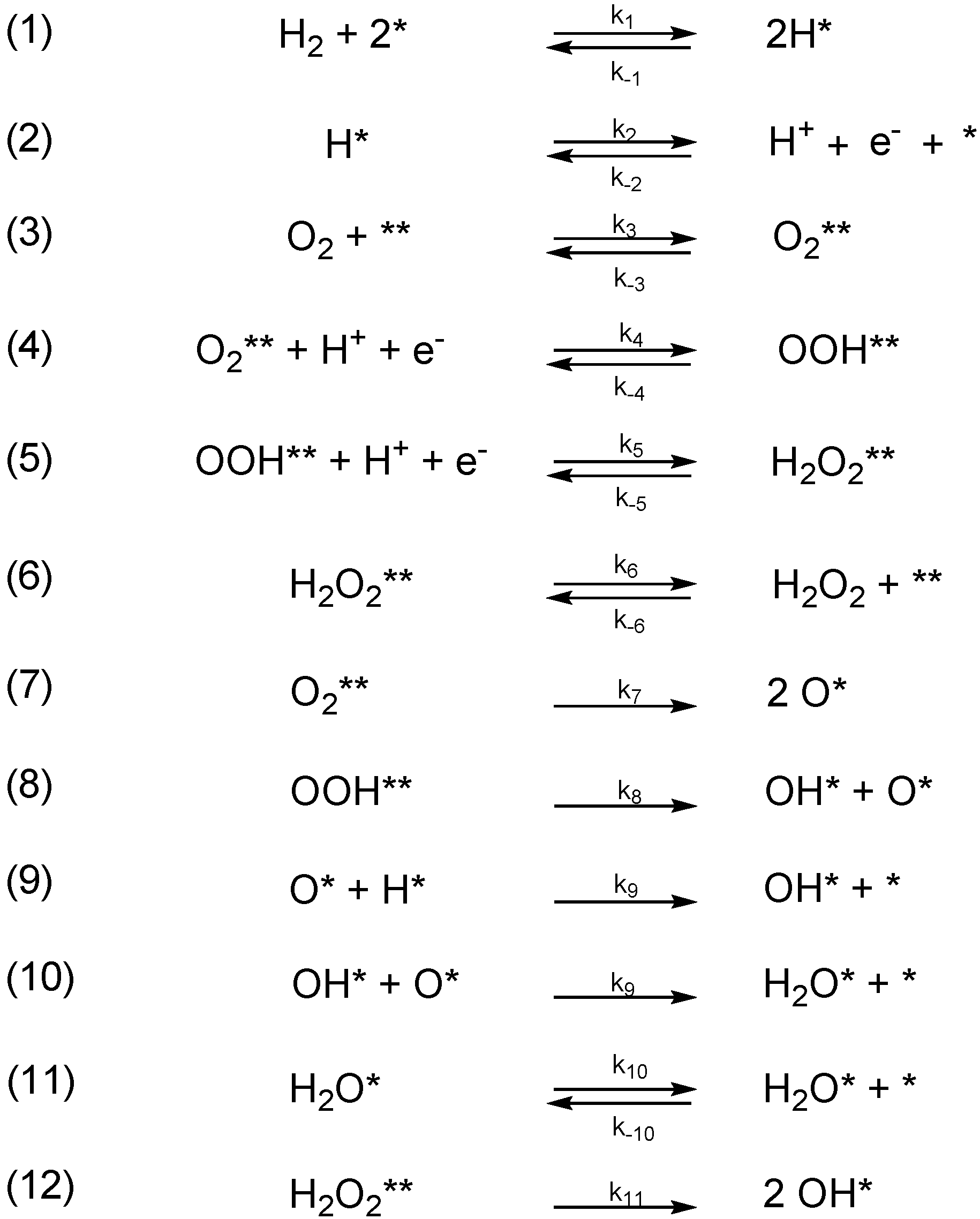

| Year | Inventor | Catalysis Type | Details |
|---|---|---|---|
| 1818 | L.J. Thenard | Chemical | Barium peroxide reacts with hydrochloric acid to form barium chloride and H2O2. Yield of 3% H2O2 was 2000 tons/annum (t/a) (Scheme 1a) [9,10] |
| 1853 | H. Meidinger | Electrochemical | Electrolysis of sulphuric acid to yield H2O2 [11] (Scheme 1b) |
| 1878 | M. Berthelot | Electrochemical | Elucidated the mechanism of sulphuric acid electrolysis. Reported the formation of peroxodisulphuric acid as an intermediate [12] (Scheme 1b) |
| 1901 | W. Manchot | Chemical | Autoxidation of hydroquinones and hydrazobenezenes under alkaline conditions in the presence of molecular oxygen to yield H2O2 [13,14,15] (Scheme 1e) |
| 1908 | Degussa-Weissenstein Process | Electrochemical | First production plant set up in Wiessenstein, Austria [16,17] |
| 1910 | Münchner Process or the Pietzsch-Adolph Process | Electrochemical | Developed by Pietzsch and Adolph at the Elektrochemische Werke, Munich. Used potassium peroxodisulphate instead of sulphuric acid to produce H2O2 [16,17] (Scheme 1c) |
| 1914 | Hugo Henkel and Walter Weber | Chemical | First account of direct synthesis of hydrogen peroxide using its constituent gases [18] |
| 1924 | Reidl-Löwenstein Process | Electrochemical | Similar to the Pietzsch-Adolph Process; used ammonium peroxodisulphate to produce H2O2 by electrolysis (Scheme 1d). Yield of 100% H2O2 was 35 kt [16,17] |
| 1932 | Walton and Filson | Chemical autoxidation | Published their work on the alternate oxidation and reduction of hydrazobenzenes to produce H2O2 [19] (Scheme 1e) |
| 1935 | Pfleiderer, Baden Aniline and Soda Factory (BASF) | Chemical autoxidation | Alkaline autoxidation of hydrazobenzenes to form sodium peroxide, later hydrolysed to form H2O2 [20] (Scheme 1f) |
| 1935–1945 | Riedl and Pfleiderer | Chemical autoxidation | Anthraquinone autoxidation process (AO) set up in two different cities each with 2000 t capacity [16,21] (Scheme 2a) |
| 1953 | E.I. du Pont de Nemors | Chemical autoxidation | Commercial plant setup with based on the Riedl and Pfleiderer process [1,3,8] (Scheme 2a) |
| 1957–1980 | Shell process | Chemical autoxidation | Oxidation of 2-propanol to yield H2O2 at a capacity of 15 kt [22] (Scheme 1g) |
| Advantages | Disadvantages |
|---|---|
| Absence of organic substrates such as anthraquinones or organic solvents | Unselective reactions leading to simultaneous side products other than H2O2, namely water (H2O) |
| Usage of green solvents like water, methanol, or ethanol | Complex process with mass transfer limitations involving three phases: gas (H2/O2), liquid (reaction medium), and solid (catalyst) |
| Economical because of fewer downstream operations to produce H2O2 | Safety: explosive nature of the H2 and O2 mixture over a wide range of concentrations (4 mol %–94 mol %) |
| The whole process can be accomplished with a single reactor system | Presence of chloride and/or bromide ions in the reaction medium |
| Serial No. (S/N) | Additive Added | Outcome | ||||
|---|---|---|---|---|---|---|
| NaBr (M *) | H3PO4 (M) | H2SO4 (M) | H2 Conversion (%) | H2O2 Selectivity (%) | H2O2 Productivity (molH2O2·Kg(Pd)−1·h−1) | |
| 1 | 0 | 0 | 0 | 100 | 0 | 0 |
| 2 | 0 | 0.003 | 100 | 0 | 0 | |
| 3 | 0.0005 | 0 | 0 | 92 | 50 | 740.1 |
| 4 | 0.0005 | 0.003 | 0 | 85 | 61 | 891 |
| 5 | 0.0005 | 0.005 | 0.025 | 79 | 65 | 830 |
| Temperature and Pressure (K and MPa) | Catalyst | Reactor Type | Solvent(s) | Promoters | Conversion and Selectivity (%) | Literature Reference |
|---|---|---|---|---|---|---|
| 263 and 2 | Palladium-CeS | Trickle bed reactor with Teflon lining | Methanol | None | No data, 80 | Biasi et al. [76] |
| 298 and 0.1 (ambient) | Palladium on porous alumina tubing | Membrane reactor | Water | Sulphuric acid (H2SO4) sodium bromide (NaBr), phosphoric acid (H3PO4) | No data, 50 | Inoue et al. [91] |
| 283–324 and 4.6–16.7 | Palladium on carbon | Stirred slurry batch reactor | Water + scCO2 and methanol + scCO2 | H3PO4 and NaBr | No data available | Moreno et al. [42] |
| 298 and 0.1 (ambient) | Pd nanoparticles immobilized on polystyrene based polymer | Capillary microreactor | Methanol | No additive | 47 and 0.65 | Fei Ng et al. [75] |
| H2SO4, KBr | 3.9 and 77 | |||||
| 301 and 1.01 | Insoluble heteropoly acid supported on Pd immobilized on mesostructured foam (MCF) silica | Autoclave reactor | Methanol | H3PO4 and NaBr | 85 and 35 | Park et al. [92] |
| 298 and 4.5 | Metallic Pd deposited on ceramic tubes | Porous tubular membrane reactor | Methanol | H2SO4, NaBr | No data and 83 | Pashkova et al. [93] |
| 293 and 4.0 | Au-Pd on TiO2 on carbon | Stainless steel autoclave | Methanol/water | No data available | No data available | Pritchard et al. [94] |
| 315 and 2.06 | Pd on SiO2. | Microreactor | Water | H2SO4, NaBr | No data and 85 | Voloshin et al. [74] |
| 263 and 1.0 | Bimetallic Pd-Au catalyst on CeS and sulphated zirconia | Trickle-bed reactor | Methanol | No data available | 90 | Biasi et al. [77] |
| 293 and 0.65 | Pd on SiO2 | Teflon coated steel reactor | Methanol | H2SO4 | No data and 21 | Abate et al. [65] |
| Pd on mesoprous silica (SBA-15) | No data and 58 | |||||
| 263 & 1.0 | Pd-Au CeS | Trickle bed reactor | Methanol | No data available | No data and 50 | Biasi et al. [95] |
| Pd-Au on ZS | No data and 60 | |||||
| 278–313 and 2.0 | Pd/C | Batch reactor | Methanol | H2SO4 | 100 and 35 | Biasi et al. [78] |
| 298 and 2.4 | Commercial 5% Pd/C | Batch slurry reactor | Methanol | None added | No data available | Gemo et al. [96] |
| 263 and 2.0 | Pd on CeS | Packed bed reactor | Methanol | H2SO4 | No data and 70 | Kilpiö et al. [97] |
| 303 and 5 | Pd nanoparticles immobilized on a functionalised resin | Fixed bed reactor | Methanol | None added | No data and 73 | Kim et al. [98] |
| 293 and 0.1 | Pd on SiO2 | Glass stirred tank reactor | Methanol | H2SO4 | No data and 60 | Menegazzo et al. [99] |
| 275 and 4.0 | Ru-Au-Pd catalyst on TiO2 support | Stainless steel autoclave | Methanol/water | None added | No data available | Ntainjua et al. [83] |
| 301 and 1.01 | Palladium on zeolite HZSM-5 | Autoclave reactor | Methanol | H3PO4 | 90 and 16 | Park et al. [100] |
| 294 and 4.0 | AuPd/C | Stainless steel autoclave | Methanol/water | None added | No data available | Piccinini et al. [101] |
| 298 and 2.3 | Pd on sulfated zirconia and Pd on alumina | Batch autoclave reactor | Methanol | None added | No data available | Rossi et al. [102] |
| 278–308 and 2.8 | Commercial Pd/C | Trickle bed reactor | Water | NaBr | No data and 90 | Biasi et al. [79] |
| 298 and 2.4 | Commercial Pd/C | Stainless steel batch autoclave | Methanol | None added | No data and 33 | Biasi et al. [80] |
| 278 and no data | Commercial Pd/C | Trickle bed reactor | Water | H3PO4 and NaBr | No data available | |
| 278 and 1.0 | Au-Pd/TiO2 | Microreactor | Water/Methanol | None | 90 and 25 | Freakley et al. [103] |
| 273 and 3.8 | Pd on activated carbon cloth (ACC) | Stainless steel autoclave | Methanol | None added | No data and 70 | Gudarzi et al. [104] |
| 295 and 3.0 | Pd-Au on carbon nanotube (CNT) | Stainless steel autoclave with Teflon coating | Methanol and sulphuric acid | H2SO4 | No conversion values and 15–65 depending on H2/O2 flow | Abate et al. [105] |
| 295 and 3.0 | Pd on nanocarbon | Stainless steel autoclave with Teflon coating | Methanol | H2SO4 | No data and 25 | Arrigo et al. [106] |
| 293 and 0.1 | Pd nanocubes on silica | Similar to the work of Lee et al. (2011) | 10 and 25 | Kim et al. [107] | ||
| 273 and 2.0 | Pd on activated carbon cloth (ACC) | Stainless steel microreactor | Methanol | None added | No data and 23 | Ratchananusorn et al. [108] |
| 278 and 2.0 | Au-Pd on nanostructured TiO2 nanotube support | Stainless steel autoclave | Methanol | HCl | No data available | Torrente-Murciano et al. [109] |
| 275 and 2.0 | Pd/C treated with NaBr | Custom made stainless steel batch reactor | Methanol | NaBr | 95 and 1 | Biasi et al. [58] |
| 293 and 2.0 | Pd supported metal organic framework (MOF) | Autoclave reactor | Methanol | None added | No data and 26 | Chung et al. [60] |
| 296 and 1.0 | Combination of Pd/TiO2 and Au-Pd/TiO2 | Microreactor with parallel packed beds (1, 8 and 16) | Water | H2SO4, H3PO4, and NaBr | Varied depending on number of channels and the flow rate | Inoue et al. [110] |
| 303 and 2.0 | Supported Au, Pd and Au-Pd | Autoclave reactor | Water | NaBr | 15 and 50 | Paunovic et al. [111] |
| 1-pentanol | 20 and 80 | |||||
| Chloroform | 18 and 38 | |||||
| Hexane | 20 and 17 | |||||
| Methyl isobutyl ketone | 27 and 10 | |||||
| 1-butanol | 28 and 60 | |||||
| 2-butanol | 25 and 55 | |||||
| Isopropanol | 35 and 75 | |||||
| Methanol | 45 and 47 | |||||
| Ethanol | 50 and 47 | |||||
| Dimethyl sulphoxide (DMSO) | 25 and 87 | |||||
| Acetonitrile | 32 and 100 | |||||
| Acetone | 35 and 95 | |||||
| t-butanol | 40 and 70 | |||||
| 303 and 2.0 | Au-Pd catalyst | Autoclaved slurry reactor | Water | H2SO4 and NaBr | 5 and no data | Paunovic et al. [112] |
| 313 and 2.0 | 10 and no data | |||||
| 323 and 2.0 | 30 and no data | |||||
| 313 and 2.0 | Au-Pd colloidal nanoparticles | Microchannel-Silica capillary reactor | Water | H2SO4 and NaBr | 80 and 85 | Paunovic et al. [73] |
| 298 and 2.6 | Commercial Pd/C | Trickle bed reactor | Water | H2SO4, H3PO4, and NaBr | No data available | Abejón et al. [81] |
| 333 and 0.1 | Au-Pd/TiO2 | Fixed bed reactor | Gas phase synthesis (2% H2/air) | None | No data available | Akram et al. [113] |
| 313 and 9.5 | Pd loaded on a sulfonic acid resin | High pressure stirred reactor | Methanol | Three compounds tested: 2-bromo-2-methyl propane, 2-bromopropane, bromobenzene compared to NaBr | No conversion data; selectivity for NaBr was 80 and 75 for the rest | Blanco-Brieva et al. [114] |
| 298 and 0.5 | Au-Pd/TiO2 | Stainless steel autoclave | Methanol/water | None added | No data available | Crole et al. [36] |
| 275 and 3.7 | Pd-Sn | Stainless steel autoclave | Methanol/water | None added | 9 and 96 | Freakley et al. [37] |
| 275 and 3.0 | Pd-Ag (varying amounts) | Teflon coated stainless steel autoclave | Methanol | H2SO4 | Depended on alloy ratio | Gu et al. [84] |
| 303 and 2.0 | Porous Pd/SiO2 | Teflon coated stainless steel autoclave | Methanol | H2SO4 | No data and 46 | Sierra-Salazar et al. [115] |
| 273–305 and 0.1–3.0 | Pd/SiO2 | Packed-bed flow reactor | Methanol/water | HCl, H2SO4, and sodium bicarbonate (NaHCO3) | No data and 31 | Wilson et al. [40] |
| 288 and 2.0 | Commercial Pd/C | Trickle bed reactor | Water | H3PO4, H2SO4, and NaBr | 77 and 72 | Gallina et al. [68] |
| 278 and 5.0 | Pd particles on acidic niobia (Nb)-silica (Si) support | Slurry reactor | Methanol | None added | 38 and 78 | Gervasini et al. [43] |
| 278 and 10.0 | Water | 38 and 85 | ||||
| 293 and 0.1 | Pd/SiO2 (sonochemical approach) | Stirred glass reactor | Ethanol/water | KBr and H3PO4 | 22 and 85 | Han et al. [116] |
| Pd/SiO2 (incipient wetness approach) | 12 and 85 | |||||
| Pd/TiO2 (sonochemical approach) | 22 and 75 | |||||
| Pd/TiO2 (incipient wetness approach) | 12 and 80 | |||||
| 300 and 0.95 | Pd/TiO2 | Flow reactor comprising of 8, 16 and 32 parallel micro-packed beds | Water | H3PO4, H2SO4, and NaBr | 64 and 66 (eight glass beds) 61 and 70 (eight glass + Si beds) | Hirama et al. [117] |
| 315 and 2.0 | Au-Pd nanoparticles | Microreactor | Water | H2SO4, NaBr and acetonitrile (MeCN) | 20 and 85 | Kanungo et al. [72] |
| 273 and 4.0 | Pd on a hexadecyl-2-hydroxyethyl-dimethyl ammonium dihydrogen phosphate (HHDMA) | Stirred reactor | Methanol/water | No data available | No data and 80 | Lari et al. [61] |
| 275 and 4.0 | Au-Pd nanoparticles supported on cesium substituted phosphotungstic acid (HPA) | Stainless steel autoclave | Methanol/water | No data available | 69 and 86 | Lewis et al. [69] |
| 293 and 0.1 | Pd/SiO2 | Glass stirred reactor | Ethanol/water | KBr | 30 and 27 | Seo et al. [45] |
| 283 and 0.1 | Pd on hydroxyapatite | Slurry reactor | Ethanol | H2SO4 | 2 and 94 | Tian et al. [88] |
| 283 and 0.1 | Pd-Tellerium (Te)/TiO2 | Micro triphase reactor | Ethanol | H2SO4 | 6 and 100 | Tian et al. [85] |
| 283 and 0.1 | Pd on mesoporous anatase TiO2 | Glass triphase reactor | Water | H2SO4 | 40 and 40 | Tu et al. [118] |
| 275 and 3.0 | Pd-zinc (Zn) on alumina (Al2O3) | Stainless steel autoclave | Methanol | H2SO4 | 57 and 78.5 | Wang et al. [86] |
| 278 and 3.0 | Au-Pd supported on carbon | Stainless steel autoclave | Methanol/water | None added | 65 and 60 | Yook et al. [119] |
| 275 and 4.0 | Au-Pd/TiO2 | Stainless steel autoclave | Methanol/water | None added | No data available | Howe et al. [89] |
| No data and 4.0 | Ag-Pd/TiO2 | Stainless steel autoclave | Methanol/water | None added | No data available | Khan et al. [44] |
| 275 and 4.0 | Au-Pd/mesoporous silica (SBA-15) | Autoclave | Methanol/water | None added | No data available | Rodrigéz-Goméz et al. [5] |
| 298 and 1.1 | Pd/TiO2 | Teflon coated reaction cell | Water | NaBr | No data available | Selinsek et al. [4] |
| 273–337 and 0.1–3.1 | Au-Pd alloy | Plugged flow reactor | Methanol/water | None added | 32 and 40 | Wilson et al. [39] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ranganathan, S.; Sieber, V. Recent Advances in the Direct Synthesis of Hydrogen Peroxide Using Chemical Catalysis—A Review. Catalysts 2018, 8, 379. https://doi.org/10.3390/catal8090379
Ranganathan S, Sieber V. Recent Advances in the Direct Synthesis of Hydrogen Peroxide Using Chemical Catalysis—A Review. Catalysts. 2018; 8(9):379. https://doi.org/10.3390/catal8090379
Chicago/Turabian StyleRanganathan, Sumanth, and Volker Sieber. 2018. "Recent Advances in the Direct Synthesis of Hydrogen Peroxide Using Chemical Catalysis—A Review" Catalysts 8, no. 9: 379. https://doi.org/10.3390/catal8090379
APA StyleRanganathan, S., & Sieber, V. (2018). Recent Advances in the Direct Synthesis of Hydrogen Peroxide Using Chemical Catalysis—A Review. Catalysts, 8(9), 379. https://doi.org/10.3390/catal8090379





