Nitrogen-Doped Porous Carbon Derived from Bamboo Shoot as Solid Base Catalyst for Knoevenagel Condensation and Transesterification Reactions
Abstract
1. Introduction
2. Results and Discussion
2.1. Catalyst Activity of the Carbon Materials for Knoevenagel Condensation
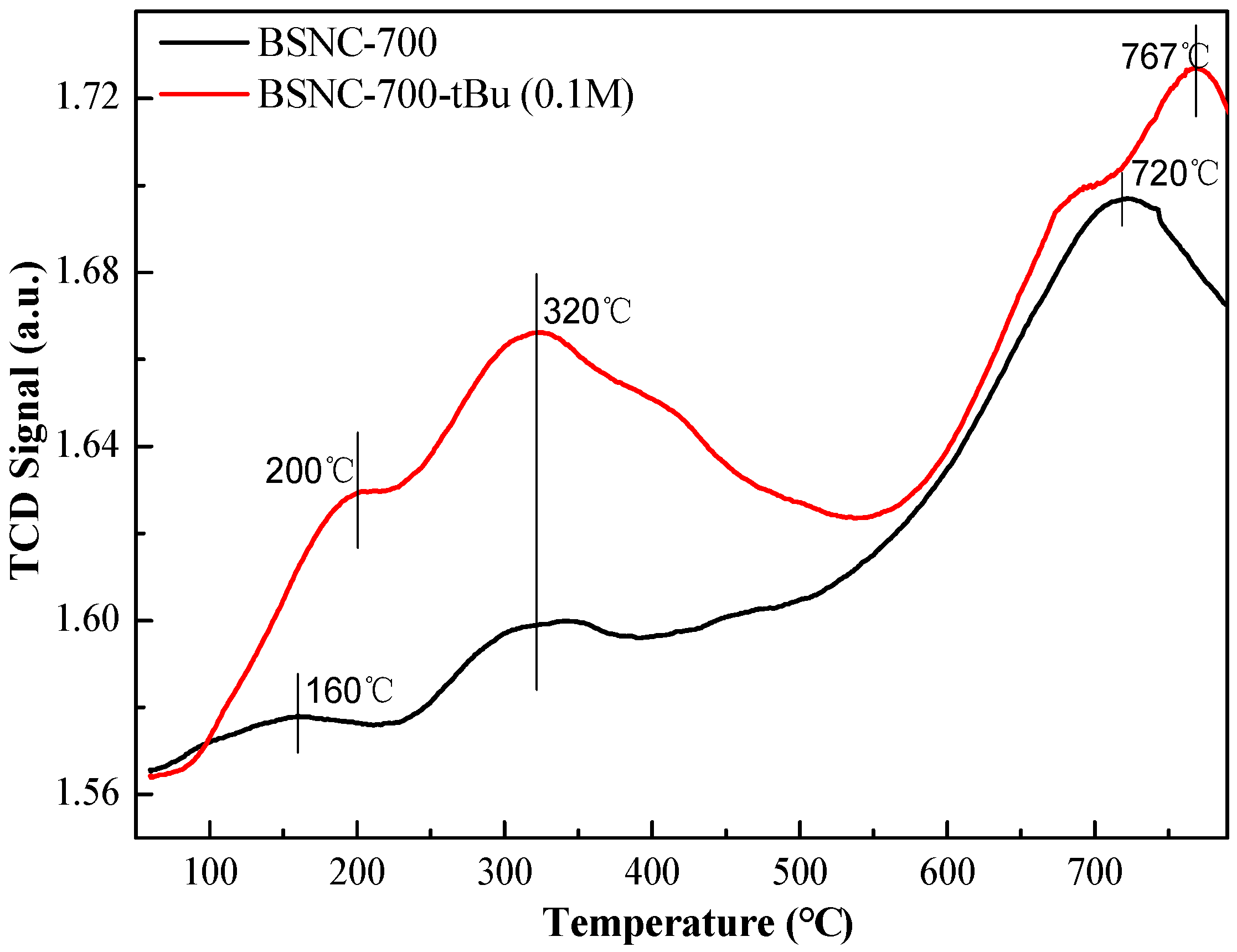
2.2. Catalyst Activity of the Deprotonation of BSNC-700
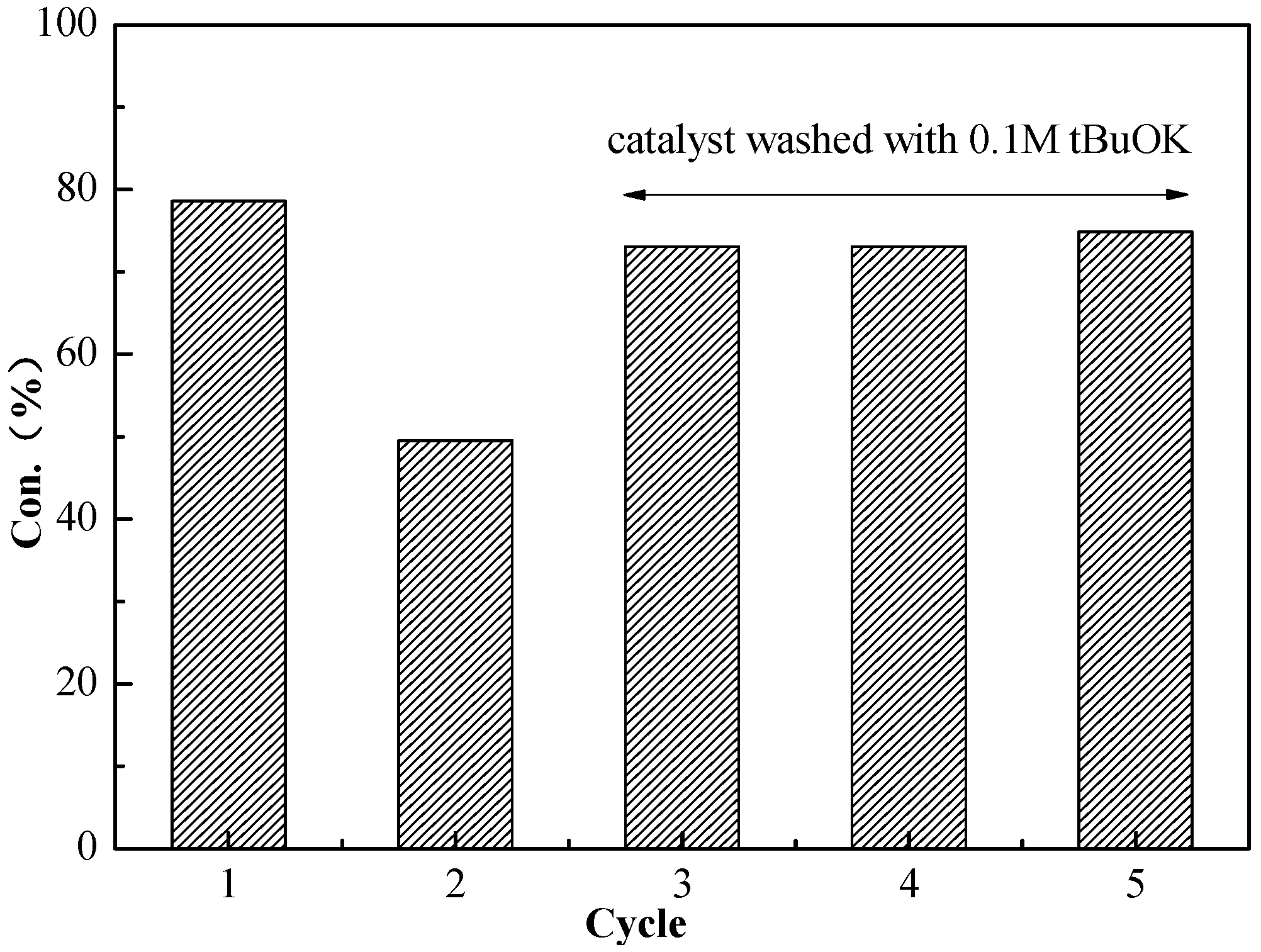
2.3. Recyclability of BSNC-700-tBu (0.1 M)
2.4. The Catalyst Activities of BSNC-700-tBu (0.1 M) for Transesterification Reactions
3. Materials and Methods
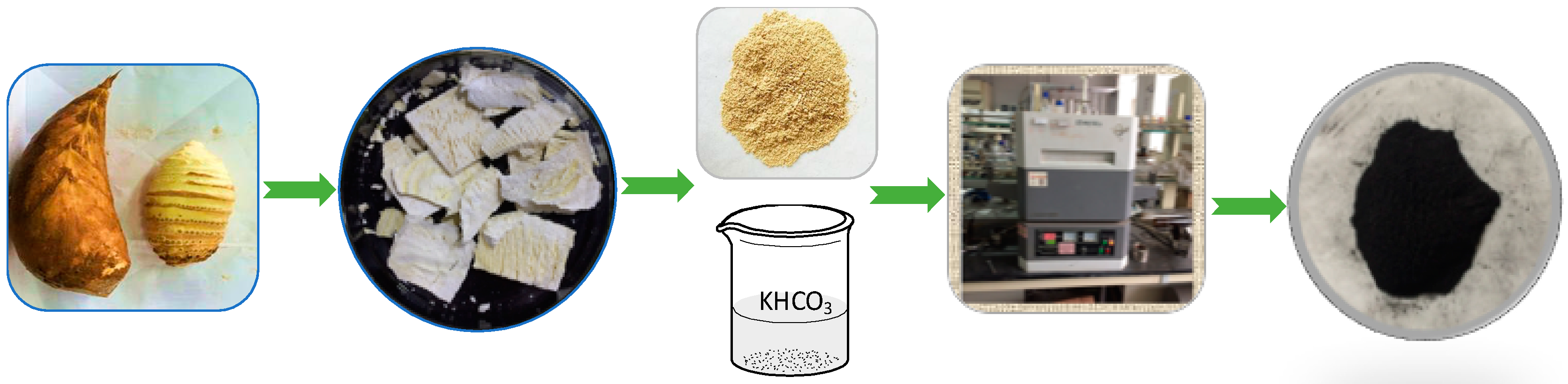
3.1. Preparation of BSNCs
3.2. Catalytic Test
3.3. Characterization
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Ying, A.; Liu, L.; Wu, G.; Chen, X.; Ye, W. Knoevenagel condensation catalyzed by DBU Br(ö)nsted ionic liquid without solvent. Chem. Res. Chin. Univ. 2009, 25, 876–881. [Google Scholar]
- Siddiqui, Z.N.; Tarannum, S. TsOH-SiO2, as an efficient and eco-friendly catalyst for Knoevenagel condensation. Tetrahedron Lett. 2014, 55, 2612–2617. [Google Scholar] [CrossRef]
- Singh, M.S.; Raghuvanshi, K. Recent advances in InCl3-catalyzed one-pot organic synthesis. Tetrahedron 2012, 68, 8683–8697. [Google Scholar] [CrossRef]
- Siddiqui, Z.N.; Khan, K. [Et3NH][HSO4]-catalyzed efficient, eco-friendly, and sustainable synthesis of quinoline derivatives via Knoevenagel condensation. ACS Sustain. Chem. Eng. 2014, 2, 1187–1194. [Google Scholar] [CrossRef]
- Shu, K.; Masaharu, S.; Hidetoshi, K.; William, L. ChemInform Abstract: Rare-earth metal triflates in organic synthesis. Chem. Rev. 2002, 102, 2227–2302. [Google Scholar]
- Mohite, A.R.; Bhat, R.G. A practical and convenient protocol for the synthesis of (E)-α, β-unsaturated acids. Org. Lett. 2013, 15, 4564–4567. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Cortés-Concepción, J.A.; Mustian, M.; Amiridis, M.D. Effect of basic properties of MgO on the heterogeneous synthesis of flavanone. Appl. Catal. 2006, 302, 232–236. [Google Scholar] [CrossRef]
- Li, M.; Tang, M.; Deng, J.; Wang, Y. Nitrogen-doped flower-like porous carbon materials directed by in situ hydrolysed MgO: Promising support for Ru nanoparticles in catalytic hydrogenations. Nano Res. 2016, 9, 3129–3140. [Google Scholar] [CrossRef]
- Sakthivel, B.; Dhakshinamoorthy, A. Chitosan as a reusable solid base catalyst for Knoevenagel condensation reaction. J. Colloid Interface Sci. 2017, 485, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Thitsartarn, W.; Kawi, S. An active and stable CaO-CeO2 catalyst for transesterification of oil to biodiesel. Green Chem. 2011, 13, 3423–3430. [Google Scholar] [CrossRef]
- Boey, P.L.; Maniam, G.P.; Hamid, S.A. Performance of calcium oxide as a heterogeneous catalyst in biodiesel production: A review. Chem. Eng. J. 2011, 168, 15–22. [Google Scholar] [CrossRef]
- Yan, Y.; Miao, J.; Yang, Z.; Xiao, F.X.; Yang, H.B.; Liu, B.; Yang, Y. Carbon nanotube catalysts: Recent advances in synthesis, characterization and applications. Chem. Soc. Rev. 2015, 44, 3295–3346. [Google Scholar] [CrossRef] [PubMed]
- Sebti, S.; Solhy, A.; Tahir, R.; Smahi, A. Modified hydroxyapatite with sodium nitrate: An efficient new solid catalyst for the claisen-schmidt condensation. Appl. Catal. A 2002, 235, 273–281. [Google Scholar] [CrossRef]
- Wang, X.; Tseng, Y.H.; Chan, J.C.C.; Cheng, S. Catalytic applications of aminopropylated mesoporous silica prepared by a template-free route in flavanones synthesis. J. Catal. 2005, 233, 266–275. [Google Scholar] [CrossRef]
- Marchetti, J.M.; Errazu, A.F. Biodiesel production from acid oils and ethanol using a solid basic resin as catalyst. Biomass Bioenergy 2010, 34, 272–277. [Google Scholar] [CrossRef]
- Liu, F.; Li, W.; Sun, Q.; Zhu, L.; Meng, X.; Guo, Y.H.; Xiao, F.S. Transesterification to biodiesel with superhydrophobic porous solid base catalysts. ChemSusChem 2011, 4, 1059–1062. [Google Scholar] [CrossRef] [PubMed]
- Calvino-Casilda, V.; Lopez-Peinado, A.J.; Duran-Valle, C.J.; Martin-Aranda, R.M. Cheminform abstract: Last decade of research on activated carbons as catalytic support in chemical processes. Catal. Rev. 2010, 41, 325–380. [Google Scholar] [CrossRef]
- Jin, X.; Balasubramanian, V.; Selvan, S.; Sawant, D.; Chari, M.; Lu, G.; Vinu, A. Highly ordered mesoporous carbon nitride nanoparticles with high nitrogen content: A metal-free basic catalyst. Angew. Chem. 2009, 48, 7884–7887. [Google Scholar] [CrossRef] [PubMed]
- Goyal, R.; Sarkar, B.; Sameer, S.; Singhal, N.; Bordoloi, A. Role of pyridinic nitrogen on base catalyzed knoevenagel condensation over pristine CNx. ChemistrySelect 2017, 2, 8086–8090. [Google Scholar] [CrossRef]
- Fujita, S.I.; Katagiri, A.; Watanabe, H.; Asano, S.; Yoshida, H.; Arai, M. Preparation of nitrogen-doped carbon from polyacrylonitrile and its application as a solid-base catalyst. ChemCatChem 2015, 7, 2965–2970. [Google Scholar] [CrossRef]
- Makowski, P.; Weber, J.; Thomas, A.; Goettmann, F. A mesoporous poly(benzimidazole) network as a purely organic heterogeneous catalyst for the Knoevenagel condensation. Catal. Commun. 2009, 10, 243–247. [Google Scholar] [CrossRef]
- Wei, Z.; Chen, Y.; Wang, J.; Su, D.; Tang, M.; Mao, S.; Wang, Y. Cobalt encapsulated in N-doped graphene layers: An efficient and stable catalyst for hydrogenation of quinoline compounds. ACS Catal. 2016, 6, 5816–5822. [Google Scholar] [CrossRef]
- Ansari, M.B.; Jin, H.; Parvin, M.N.; Park, S.E. Mesoporous carbon nitride as a metal-free base catalyst in the microwave assisted Knoevenagel condensation of ethylcyanoacetate with aromatic aldehydes. Catal. Today 2012, 185, 211–216. [Google Scholar] [CrossRef]
- Kan-Nari, N.; Okamura, S.; Fujita, S.I.; Ozaki, J.I.; Arai, M. Nitrogen-doped carbon materials prepared by ammoxidation as solid base catalysts for Knoevenagel condensation and transesterification reactions. Adv. Synth. Catal. 2010, 352, 1476–1484. [Google Scholar] [CrossRef]
- Van, D.S.; Jong, K.P.D.; Bitter, J.H. Nitrogen-containing carbon nanotubes as solid base catalysts. Chem. Commun. 2006, 46, 4859–4861. [Google Scholar]
- Sun, Y.B.; Cao, C.; Huang, P.; Yang, S.; Song, W. Amines functionalized c60 as solid base catalysts for knoevenagel condensation with high activity and stability. RSC Adv. 2015, 5, 86082–86087. [Google Scholar] [CrossRef]
- Karagoz, S.; Tay, T.; Ucar, S.M. Activated carbons from waste biomass by sulfuric acid activation and their use on methylene blue adsorption. Bioresour. Technol. 2008, 99, 6214–6222. [Google Scholar] [CrossRef] [PubMed]
- Kai, W.; Ning, Z.; Lei, S.; Rui, Y.; Tian, X.; Wang, J.; Song, Y.; Xu, D.; Guo, Q.; Liu, L. Promising biomass-based activated carbons derived from willow catkins for high performance supercapacitors. Electrochim. Acta 2015, 166, 1–11. [Google Scholar]
- Guo, S.; Dong, X.; Zhu, C.; Han, Y.; Ma, F.; Wu, T. Pyrolysis behaviors and thermodynamics properties of hydrochar from bamboo (phyllostachys heterocycla cv. pubescens) shoot shell. Bioresour. Technol. 2017, 233, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Ding, Z.Q.; Gao, Q.; Xun, H.; Tang, F.; Xia, E.D. Major chemical constituents of bamboo shoots (Phyllostachys pubescens): Qualitative and quantitative research. J. Agric. Food Chem. 2016, 64, 2498–2505. [Google Scholar] [CrossRef] [PubMed]
- Torii, S.; Jimura, K.; Hayashi, S.; Kikuchi, R.; Takagaki, A. Utilization of hexagonal boron nitride as a solid acid-b bifunctional catalyst. J. Catal. 2017, 355, 176–184. [Google Scholar] [CrossRef]
- Chen, X.F.; Zhang, J.Y.; Zhang, B.; Dong, S.M.; Guo, X.C.; Mu, X.D.; Fei, B.H. A novel hierarchical porous nitrogen-doped carbon derived from bamboo shoot for high performance supercapacitor. Sci. Rep. 2017, 7, 7362. [Google Scholar] [CrossRef] [PubMed]
- Pels, J.R.; Kapteijn, F.; Moulijn, J.A.; Zhu, Q.; Thomas, K.M. Evolution of nitrogen functionalities in carbonaceous materials during pyrolysis. Carbon 1995, 33, 1641–1653. [Google Scholar] [CrossRef]
- Sevilla, M.; Parra, J.B.; Fuertes, A.B. Assessment of the role of micropore size and N-doping in CO2 capture by porous carbons. ACS Appl. Mater. Interfaces 2013, 5, 6360–6368. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, T.; Boero, M.; Huang, S.F.; Terakura, K.; Oshima, M.; Ozaki, J. Carbon alloy catalysts: Active sites for oxygen reduction reaction. J. Phys. Chem. C 2008, 112, 14706–14709. [Google Scholar] [CrossRef]
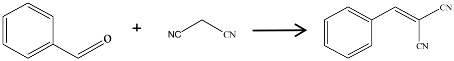
| Entry | Catalyst | Benzaldehyde Conversion (%) | Target Product Selectivity (%) |
|---|---|---|---|
| 1 | / | 54.5 | 13.3 |
| 2 | BS-700 | 2.2 | 100 |
| 3 | Commercial activated carbon | 7.5 | 0 |
| 4 | BSNC-600 | 8.3 | 100 |
| 5 | BSNC-700 | 16.1 | 100 |
| 6 | BSNC-800 | 8.2 | 100 |
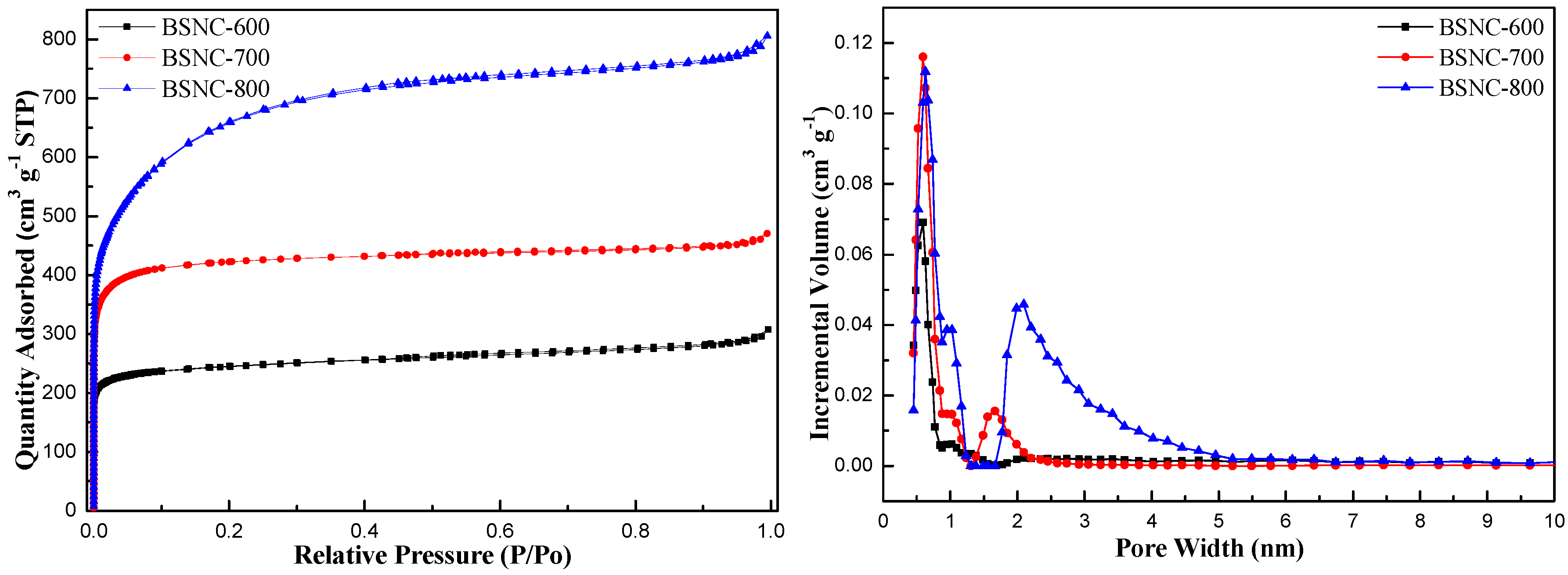
| Samples | Specific Surface Area (m2 g−1) | Pore Volume (cm3 g−1) | Pore Size (nm) | C a (wt.%) | N a (wt.%) | Surface O b (at.%) | Surface N b (at.%) | N b/O b |
|---|---|---|---|---|---|---|---|---|
| Bamboo shoot | 0.4 [32] | 0.002 [32] | / | 41.67 | 4.27 | / | / | / |
| BS-700 | 4.6 | 0.01 | 7.13 | 63.36 | 4.40 | 18.3 | 6.10 | 0.333 |
| BSNC-600 | 962 | 0.48 | 1.97 | 69.18 | 4.65 | 13.31 | 4.56 | 0.343 |
| BSNC-700 | 1475 | 0.73 | 1.97 | 80.37 | 2.79 | 7.41 | 4.92 | 0.664 |
| BSNC-800 | 2271 | 1.25 | 2.19 | 43.43 | 1.06 | 10.53 | 3.73 | 0.354 |
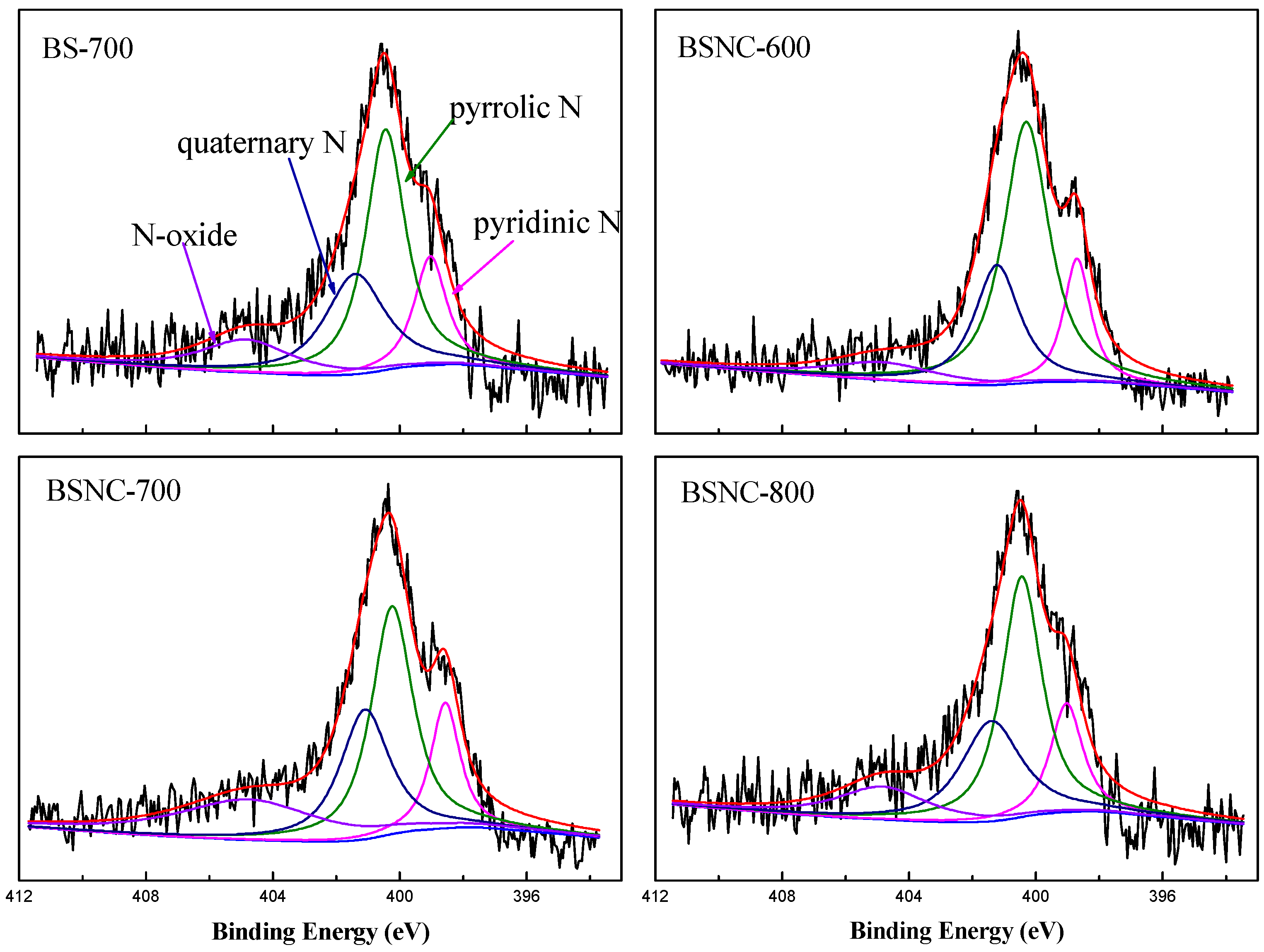
| Samples | Pyridinic N (%) 398.7 eV | Pyrrolic N (%) 400.3 eV | Quaternary N (%) 401.2 eV | N-Oxide (%) 404.7 eV |
|---|---|---|---|---|
| BS-700 | 18.1 | 43.6 | 19.9 | 18.4 |
| BSNC-600 | 14.0 | 58.8 | 19.1 | 8.1 |
| BSNC-700 | 14.4 | 37.6 | 25.4 | 22.6 |
| BSNC-800 | 15.9 | 43.5 | 27.0 | 13.6 |
| Entry | Catalysts | Benzaldehyde Conversion (%) | Target Product Selectivity (%) |
|---|---|---|---|
| 1 | BSNC-700 | 16.1 | 100 |
| 2 | BSNC-700-H (1.0 M) | 1.0 | 100 |
| 3 | BSNC-700-OH (1.0 M) | 34.4 | 100 |
| 4 | BSNC-700-OH (0.1 M) | 32.2 | 100 |
| 5 | BSNC-700-tBu (1.0 M) | 79.9 | 100 |
| 6 | BSNC-700-tBu (0.1 M) | 76.0 | 100 |
| Entry | Alcohols | β-Ketoester | Time (h) | Alcohol Conversion (%) | Target Product’s Selectivity (%) |
|---|---|---|---|---|---|
| 1 |  |  | 16 | 70.6 | 100 |
| 2 | 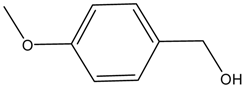 | 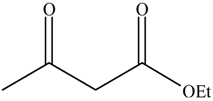 | 16 | 63.7 | 100 |
| 3 |  | 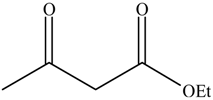 | 16 | 75.1 | 100 |
| 4 | 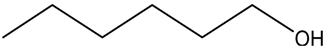 | 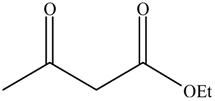 | 16 | 74.4 | 100 |
| 5 | 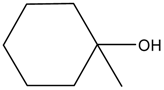 | 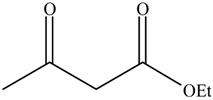 | 16 | / | / |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mi, B.; Chen, X.; Jiang, C.; Wang, J.; Chen, X.; Zhang, B.; Liu, X.; Liu, Z.; Fei, B. Nitrogen-Doped Porous Carbon Derived from Bamboo Shoot as Solid Base Catalyst for Knoevenagel Condensation and Transesterification Reactions. Catalysts 2018, 8, 232. https://doi.org/10.3390/catal8060232
Mi B, Chen X, Jiang C, Wang J, Chen X, Zhang B, Liu X, Liu Z, Fei B. Nitrogen-Doped Porous Carbon Derived from Bamboo Shoot as Solid Base Catalyst for Knoevenagel Condensation and Transesterification Reactions. Catalysts. 2018; 8(6):232. https://doi.org/10.3390/catal8060232
Chicago/Turabian StyleMi, Bingbing, Xiufang Chen, Changle Jiang, Jingxin Wang, Xiujuan Chen, Bo Zhang, Xianmiao Liu, Zhijia Liu, and Benhua Fei. 2018. "Nitrogen-Doped Porous Carbon Derived from Bamboo Shoot as Solid Base Catalyst for Knoevenagel Condensation and Transesterification Reactions" Catalysts 8, no. 6: 232. https://doi.org/10.3390/catal8060232
APA StyleMi, B., Chen, X., Jiang, C., Wang, J., Chen, X., Zhang, B., Liu, X., Liu, Z., & Fei, B. (2018). Nitrogen-Doped Porous Carbon Derived from Bamboo Shoot as Solid Base Catalyst for Knoevenagel Condensation and Transesterification Reactions. Catalysts, 8(6), 232. https://doi.org/10.3390/catal8060232





