Host-Guest Engineering of Layered Double Hydroxides towards Efficient Oxygen Evolution Reaction: Recent Advances and Perspectives
Abstract
:1. Introduction
2. The Engineering of Host Layer
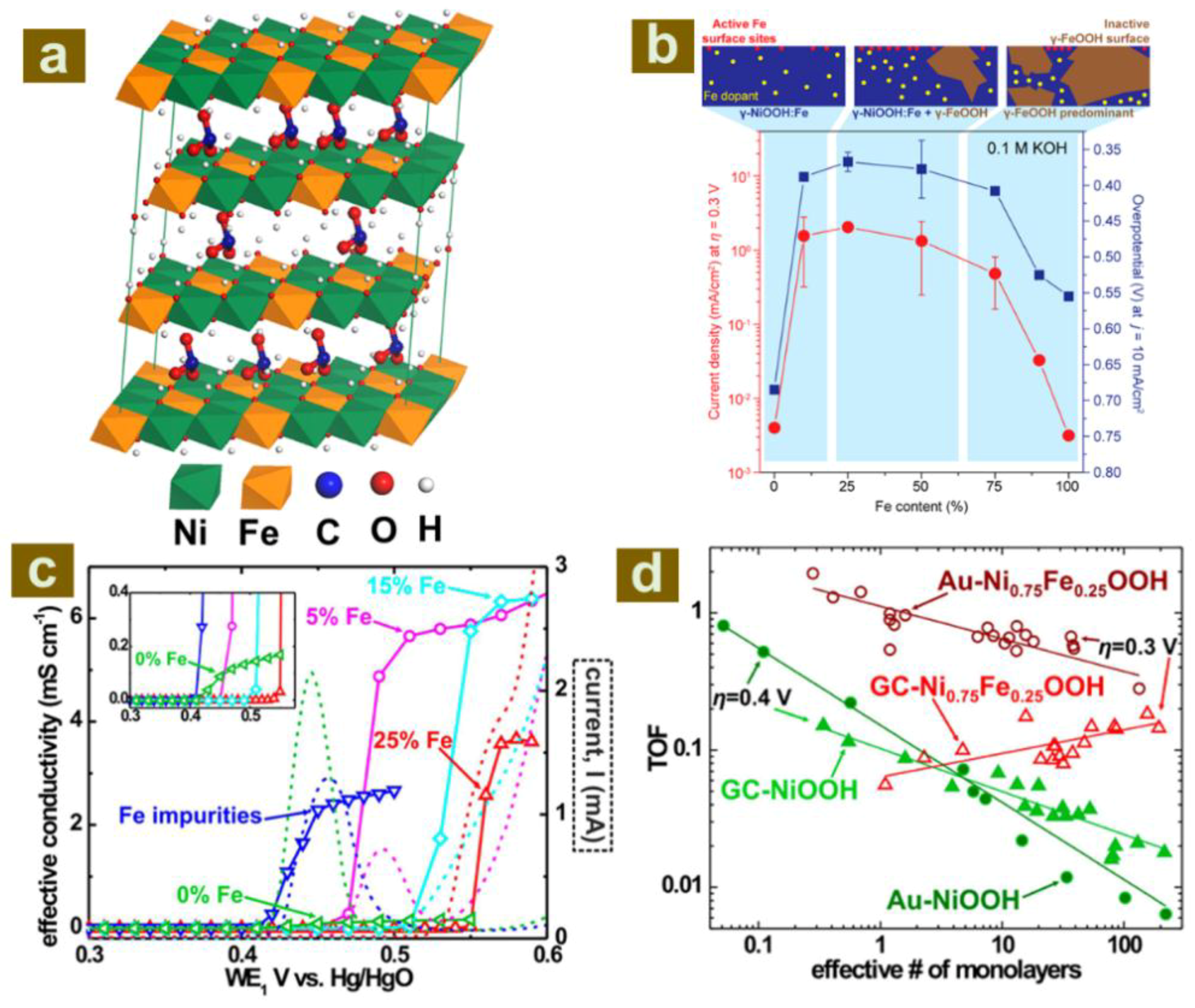
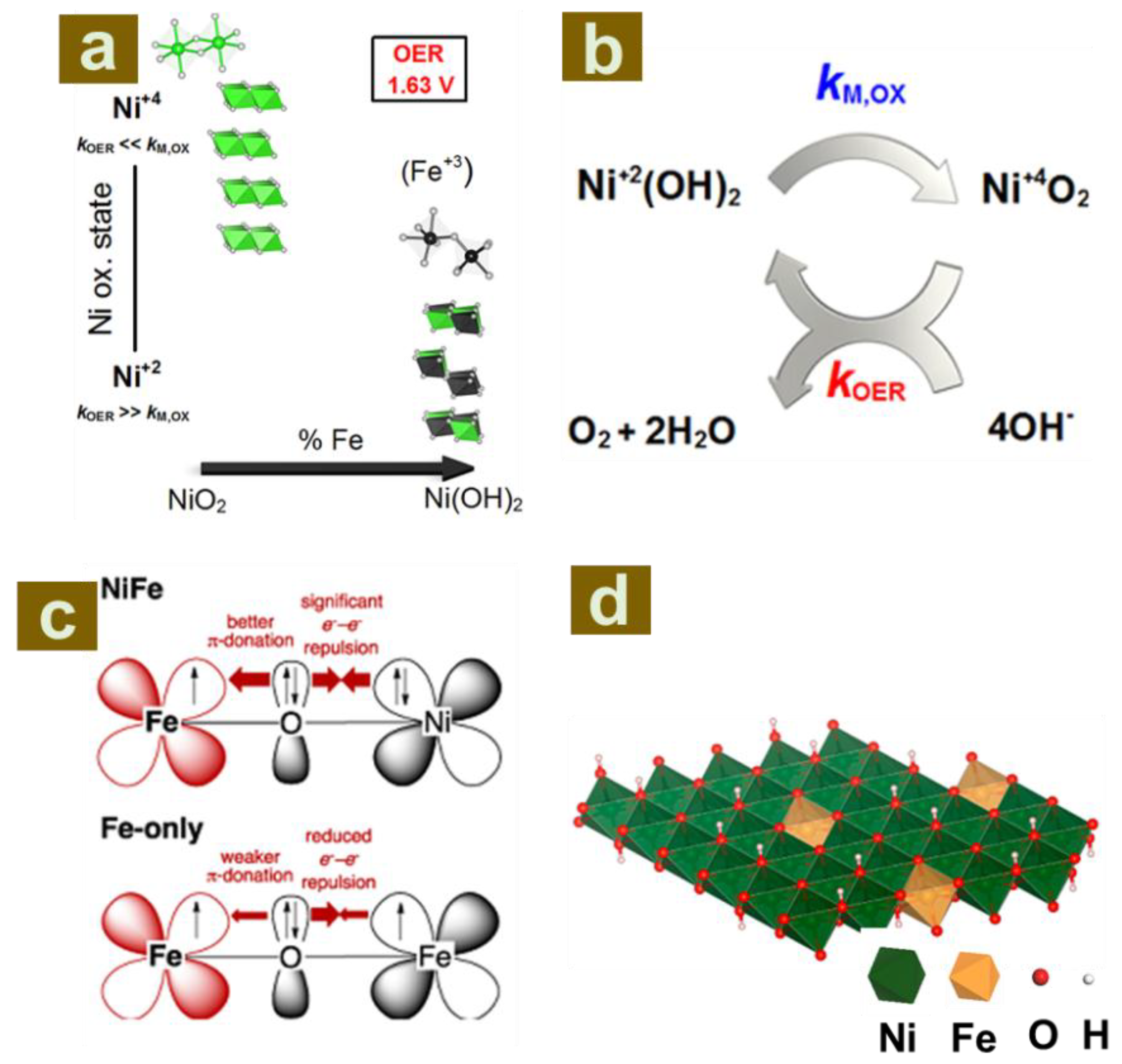
2.1. The Role of Host Layer Metal Ions
2.2. The Engineering of LDH Host Layers
2.2.1. The Exfoliation of LDHs
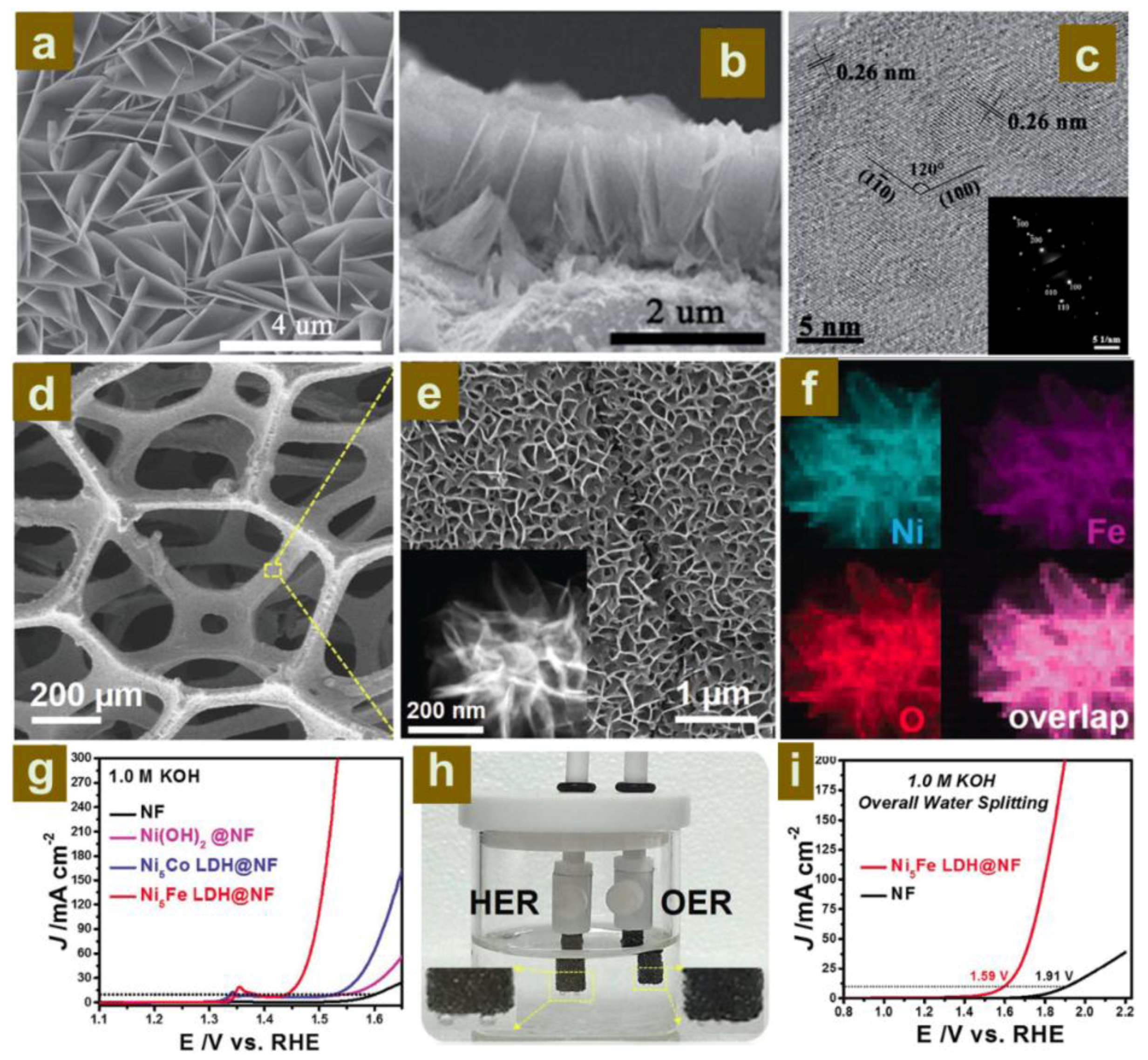
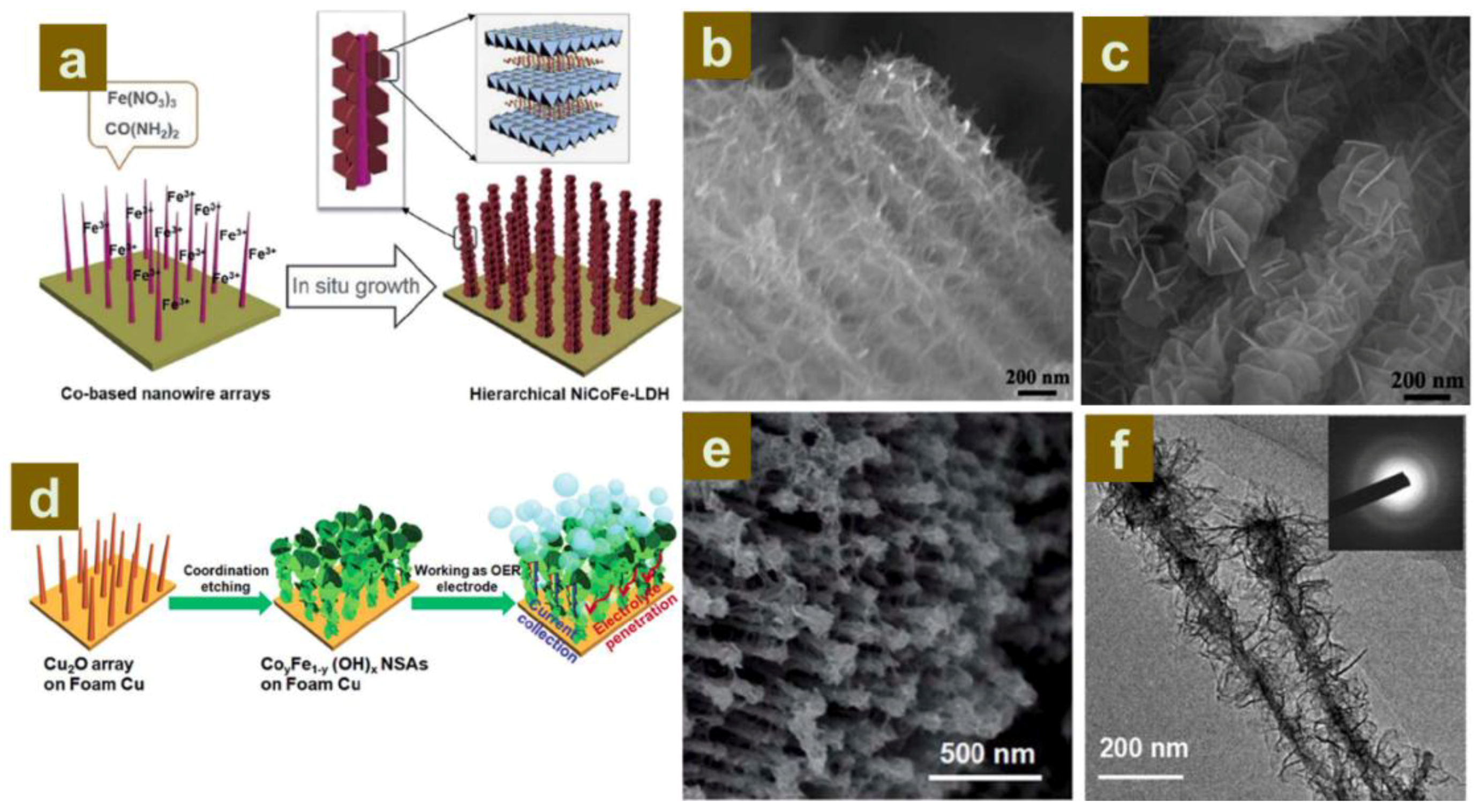
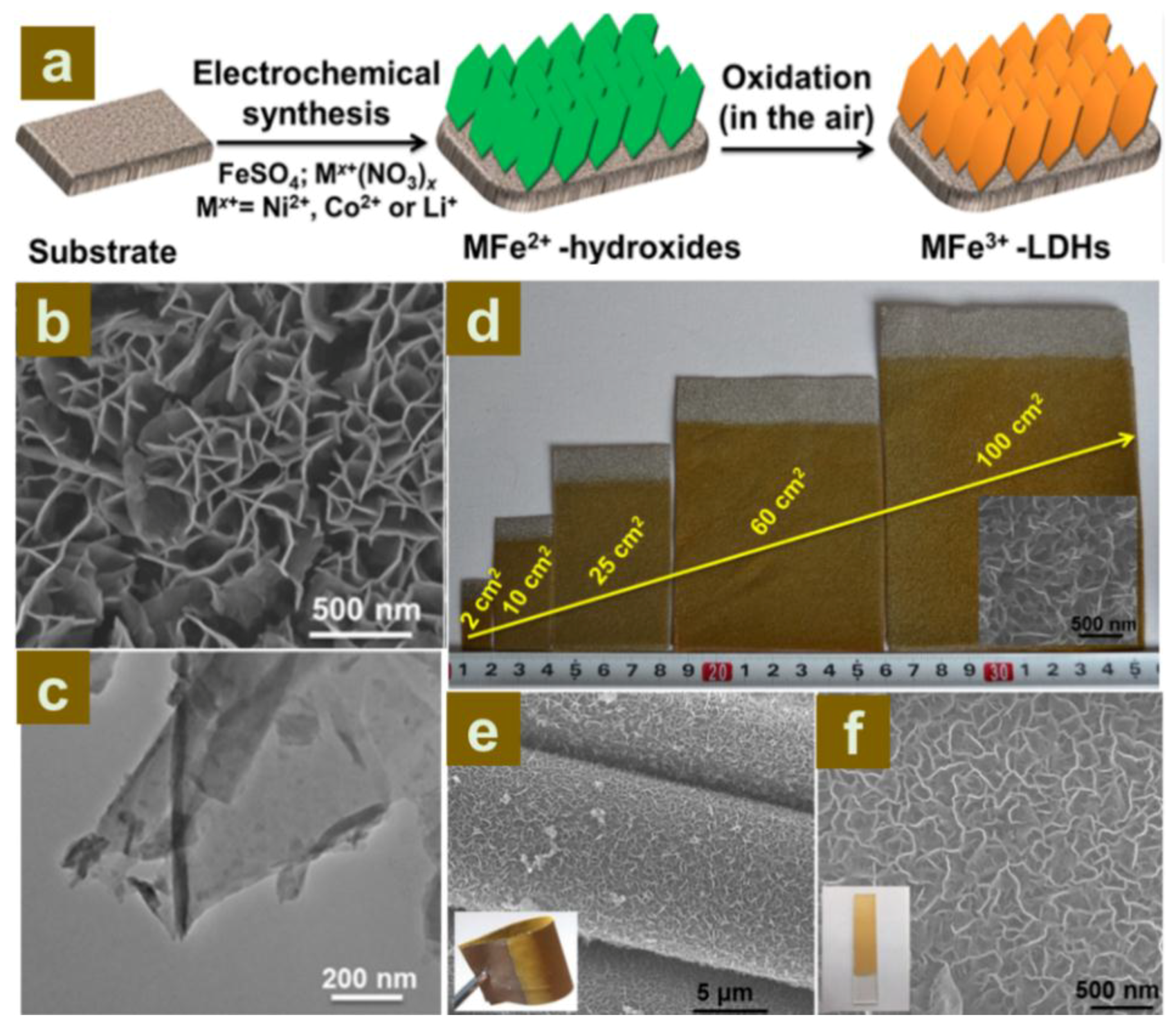
2.2.2. Construction of LDH Nanoarrays
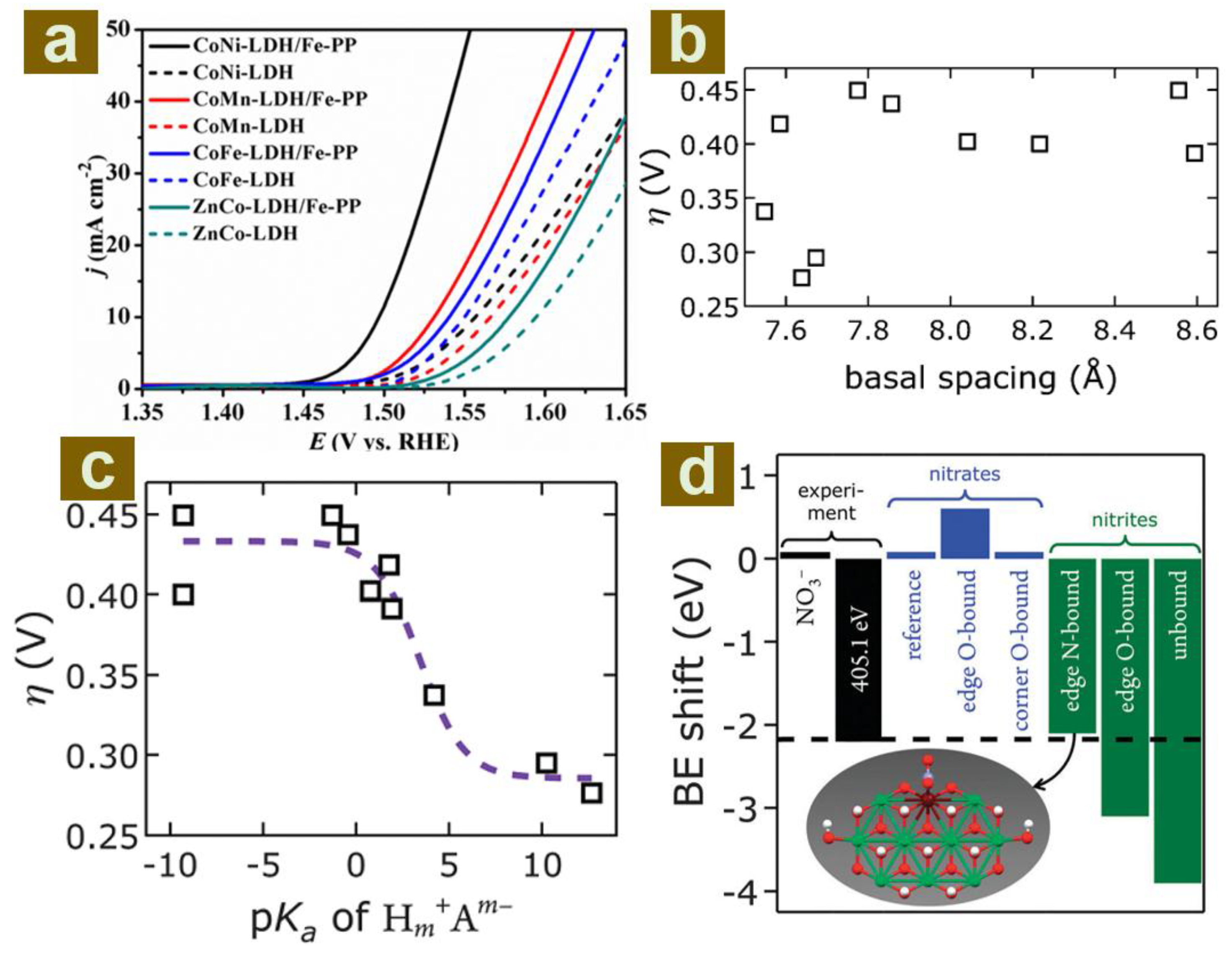
3. The Engineering of Interlayer Guests
4. Conclusions and Perspectives
Acknowledgments
Conflicts of Interest
References
- Menezes, P.W.; Indra, A.; Das, C.; Walter, C.; Göbel, C.; Vitaly, G.V.; Schmeiβer, D.; Driess, M. Uncovering the nature of active species of nickel phosphide catalysts in high–performance electrochemical overall water splitting. ACS Catal. 2017, 7, 103–109. [Google Scholar] [CrossRef]
- Hou, Y.; Qiu, M.; Zhang, T.; Ma, J.; Liu, S.H.; Zhuang, X.D.; Yuan, C.; Feng, X.L. Efficient electrochemical and photoelectrochemical water splitting by a 3D nanostructured carbon supported on flexible exfoliated graphene foil. Adv. Mater. 2017, 29, 1604480. [Google Scholar]
- Ryu, S.J.; Hoffmann, M.R. Mixed-metal semiconductor anodes for electrochemical water splitting and reactive chlorine species generation: Implications for electrochemical wastewater treatment. Catalysts 2016, 6, 59. [Google Scholar] [CrossRef]
- Li, X.M.; Hao, X.G.; Abudula, A.; Guan, G.Q. Nanostructured catalysts for electrochemical water splitting: Current state and prospects. J. Mater. Chem. A 2016, 4, 11973–12000. [Google Scholar]
- Reier, T.; Pawolek, Z.; Cherevko, S.; Bruns, M.; Jones, T.; Teschner, D.; Selve, S.; Bergmann, A.; Nong, H.N.; Schlögl, R.; et al. Molecular insight in structure and activity of highly efficient, low–Ir Ir–Ni oxide catalysts for electrochemical water splitting. J. Am. Chem. Soc. 2015, 137, 13031–13040. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.Y.; Li, L.J.; Ahn, H.S.; Manthiram, A. Delineating the roles of Co3O4 and N–doped carbon nanoweb (CNW) in bifunctional Co3O4/CNW catalysts for oxygen reduction and oxygen evolution reactions. J. Mater. Chem. A 2015, 3, 11615–11623. [Google Scholar] [CrossRef]
- Li, P.X.; Ma, R.G.; Zhou, Y.; Chen, Y.F.; Zhou, Z.Z.; Liu, G.H.; Liu, Q.; Peng, G.H.; Liang, Z.H.; Wang, J.C. In situ growth of spinel CoFe2O4 nanoparticles on rod–like ordered mesoporous carbon for bifunctional electrocatalysis of both oxygen reduction and oxygen evolution. J. Mater. Chem. A 2015, 3, 15598–15606. [Google Scholar] [CrossRef]
- Hutchings, G.S.; Zhang, Y.; Li, J.; Yonemoto, B.Y.; Zhou, X.G.; Zhu, K.K.; Jiao, F. In situ formation of cobalt oxide nanocubanes as efficient oxygen evolution catalysts. J. Am. Chem. Soc. 2015, 137, 4223–4229. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.H.; Li, Q.; Wu, C.H.; Zhu, H.Y.; Garcia, A.M.; Shen, B.; Guo, J.H.; Sun, S.H. Stable cobalt nanoparticles and their monolayer array as an efficient electrocatalyst for oxygen evolution reaction. J. Am. Chem. Soc. 2015, 137, 7071–7074. [Google Scholar] [CrossRef] [PubMed]
- He, G.W.; Zhang, W.; Deng, Y.D.; Zhong, C.; Hu, W.B.; Han, X.P. Engineering pyrite-type bimetallic Ni-doped CoS2 nanoneedle arrays over a wide compositional range for enhanced oxygen and hydrogen electrocatalysis with flexible property. Catalysts 2017, 7, 366. [Google Scholar] [CrossRef]
- Cherevko, S.; Geiger, S.; Kasian, O.; Kulyk, O.; Grote, J.P.; Savan, A.; Shrestha, B.R.; Merzlikin, S.; Breitbach, B.; Ludwig, A.; et al. Oxygen and hydrogen evolution reactions on Ru, RuO2, Ir, and IrO2 thin film electrodes in acidic and alkaline electrolytes: A comparative study on activity and stability. Catalysis Today 2016, 262, 170–180. [Google Scholar] [CrossRef]
- Lee, Y.M.; Suntivich, J.; May, K.J.; Perry, E.E.; Yang, S.H. Synthesis and activities of rutile IrO2 and RuO2 nanoparticles for oxygen evolution in acid and alkaline solutions. J. Phys. Chem. Lett. 2012, 3, 399–404. [Google Scholar] [CrossRef] [PubMed]
- Bates, M.K.; Jia, Q.Y.; Doan, H.; Liang, W.T.; Mukerjee, S. Charge–transfer effects in Ni–Fe and Ni–Fe–Co. mixed–metal oxides for the alkaline oxygen evolution reaction. ACS Catal. 2016, 6, 155–161. [Google Scholar] [CrossRef]
- Weng, B.C.; Xu, F.H.; Wang, C.L.; Meng, W.W.; Grice, C.R.; Yan, Y.F. A layered Na1−xNiyFe1−yO2 double oxide oxygen evolution reaction electrocatalyst for highly efficient water–splitting. Energy Environ. Sci. 2017, 10, 121–128. [Google Scholar] [CrossRef]
- Han, X.L.; Yu, Y.F.; Huang, Y.; Liu, D.L.; Zhang, B. Photogenerated carriers boost water splitting activity over transition–metal/semiconducting metal oxide bifunctional electrocatalysts. ACS Catal. 2017, 7, 6464–6470. [Google Scholar] [CrossRef]
- Lu, F.; Zhou, M.; Zhou, Y.X.; Zeng, X.H. First–row transition metal based catalysts for the oxygen evolution reaction under alkaline conditions: Basic principles and recent advances. Small 2017, 13, 1701931. [Google Scholar] [CrossRef] [PubMed]
- Stevens, M.B.; Enman, L.J.; Batchellor, A.S.; Cosby, M.R.; Vise, A.E.; Trang, C.D.M.; Boettcher, S.W. Measurement techniques for the study of thin film heterogeneous water oxidation electrocatalysts. Chem. Mater. 2017, 29, 120–140. [Google Scholar] [CrossRef]
- Han, L.; Dong, S.J.; Wang, E.K. Transition–metal (CO, Ni, and Fe)–based electrocatalysts for the water oxidation reaction. Adv. Mater. 2016, 28, 9266–9291. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.L.; Cao, X.H.; Wu, X.J.; He, Q.Y.; Yang, J.; Zhang, X.; Chen, J.Z.; Zhao, W.; Han, S.K.; Nam, G.H.; et al. Recent advances in ultrathin two-dimensional nanomaterials. Chem. Rev. 2017, 117, 6225–6331. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.L.; Tang, C.; Wang, H.F.; Zhang, Q.; Yang, C.H.; Wei, F. Dual-sized NiFe layered double hydroxides in situ grown on oxygen-decorated self-dispersal nanocarbon as enhanced water oxidation catalysts. J. Mater. Chem. A 2015, 3, 24540–24546. [Google Scholar] [CrossRef]
- Zhou, L.; Shao, M.F.; Li, J.B.; Jiang, S.; Wei, M.; Duan, X. Two–dimensional ultrathin arrays of CoP: Electronic modulation toward high performance overall water splitting. Nano Energy 2017, 41, 583–590. [Google Scholar] [CrossRef]
- Zhang, G.; Wang, G.C.; Liu, Y.; Liu, H.J.; Qu, J.H.; Li, J.H. Highly active and stable catalysts of phytic acid–derivative transition metal phosphides for full water splitting. J. Am. Chem. Soc. 2016, 138, 14686–14693. [Google Scholar] [CrossRef] [PubMed]
- Tan, Y.W.; Wang, H.; Liu, P.; Shen, Y.H.; Cheng, C.; Hirata, A.; Fujita, T.; Tang, Z.; Chen, M.W. Versatile nanoporous bimetallic phosphides towards electrochemical water splitting. Energy Environ. Sci. 2016, 9, 2257–2261. [Google Scholar] [CrossRef]
- Li, W.; Zhang, S.L.; Fan, Q.N.; Zhang, F.Z.; Xu, S.L. Hierarchically scaffolded CoP/CoP2 nanoparticles: Controllable synthesis and their application as a well-matched bifunctional electrocatalyst for overall water splitting. Nanoscale 2017, 9, 5677–5685. [Google Scholar] [CrossRef] [PubMed]
- Corrigan, D.A. The catalysis of the oxygen evolution reaction by iron impurities in thin film nickel oxide electrodes. J. Electrochem. Soc. 1987, 134, 377–384. [Google Scholar] [CrossRef]
- Gong, M.; Li, Y.G.; Wang, H.L.; Liang, Y.Y.; Wu, J.Z.; Zhou, J.G.; Wang, J.; Regier, T.; Wei, F.; Dai, H.J. An advanced Ni–Fe layered double hydroxide electrocatalyst for water oxidation. J. Am. Chem. Soc. 2013, 135, 8452–8455. [Google Scholar] [CrossRef] [PubMed]
- Shao, M.F.; Ning, F.Y.; Zhao, J.W.; Wei, M.; Evans, D.G.; Duan, X. Preparation of Fe3O4@SiO2@layered double hydroxide core–shell microspheres for magnetic separation of proteins. J. Am. Chem. Soc. 2012, 134, 1071–1077. [Google Scholar] [CrossRef] [PubMed]
- Evans, D.G.; Duan, X. Preparation of layered double hydroxides and their applications as additives in polymers, as precursors to magnetic materials and in biology and medicine. Chem. Commun. 2006, 0, 485–496. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; O’Hare, D. Recent advances in the synthesis and application of layered double hydroxide (LDH) nanosheets. Chem. Rev. 2012, 7, 4124–4155. [Google Scholar] [CrossRef] [PubMed]
- Ning, F.Y.; Shao, M.F.; Xu, S.M.; Fu, Y.; Zhang, R.K.; Wei, M.; Evans, D.G.; Duan, X. TiO2/Graphene/NiFe-layered double hydroxide nanorod array photoanodes for efficient photoelectrochemical water splitting. Energy Environ. Sci. 2016, 9, 2633–2643. [Google Scholar] [CrossRef]
- Zhang, H.J.; Li, X.P.; Hahnel, A.; Naumann, V.; Lin, C.; Azimi, S.; Schweizer, S.L.; Maijenburg, A.W.; Wehrspohn, R.B. Bifunctional heterostructure assembly of NiFe LDH nanosheets on NiCoP nanowires for highly efficient and stable overall water splitting. Adv. Funct. Mater. 2018, 28, 1706847. [Google Scholar] [CrossRef]
- Lu, Z.Y.; Xu, W.W.; Zhu, W.; Yang, Q.; Lei, X.D.; Liu, J.F.; Li, Y.P.; Sun, X.M.; Duan, X. Three-dimensional NiFe layered double hydroxide film for high- efficiency oxygen evolution reaction. Chem. Commun. 2014, 50, 6479–6482. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.; Lohe, M.R.; Zhang, J.; Liu, S.H.; Zhuang, X.D.; Feng, X.L. Vertically oriented cobalt selenide/NiFe layered-double-hydroxide nanosheets supported on exfoliated graphene foil: An efficient 3D electrode for overall water splitting. Energy Environ. Sci. 2016, 9, 478–483. [Google Scholar] [CrossRef]
- Wang, Q.; Shang, L.; Shi, R.; Zhang, X.; Zhao, Y.F.; Waterhouse, G.I.N.; Wu, L.Z.; Tung, C.H.; Zhang, T.Z. NiFe layered double hydroxide nanoparticles on Co., N-codoped carbon nanoframes as efficient bifunctional catalysts for rechargeable zinc–air batteries. Adv. Energy Mater. 2017, 7, 1700467. [Google Scholar] [CrossRef]
- Li, Y.G.; Gong, M.; Liang, Y.Y.; Feng, J.; Kim, J.E.; Wang, H.L.; Hong, G.S.; Zhang, B.; Dai, H.J. Advanced zinc-air batteries based on high-performance hybrid electrocatalysts. Nat. Commun. 2013, 4, 1805. [Google Scholar] [CrossRef] [PubMed]
- Tang, D.; Liu, J.; Wu, X.Y.; Liu, R.H.; Han, X.; Han, Y.Z.; Huang, H.; Liu, Y.; Kang, Z.H. Carbon quantum dot/NiFe layered double-hydroxide composite as a highly efficient electrocatalyst for water oxidation. ACS Appl. Mater. Interfaces 2014, 6, 7918–7925. [Google Scholar] [CrossRef] [PubMed]
- Ma, W.; Ma, R.Z.; Wang, C.X.; Liang, J.B.; Liu, X.H.; Zhou, K.C.; Sasaki, T. A superlattice of alternately stacked Ni–Fe hydroxide nanosheets and graphene for efficient splitting of water. ACS Nano 2015, 9, 1977–1984. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.; Wang, H.S.; Wang, H.F.; Zhang, Q.; Tian, G.L.; Nie, J.Q.; Wei, F. Spatially confined hybridization of nanometer-sized NiFe hydroxides into nitrogen-doped graphene frameworks leading to superior oxygen evolution reactivity. Adv. Mater. 2015, 27, 4516–4522. [Google Scholar] [CrossRef] [PubMed]
- Tang, D.; Han, Y.Z.; Ji, W.B.; Qiao, S.; Zhou, X.; Liu, R.H.; Han, X.; Huang, H.; Liu, Y.; Kang, Z.H. A high-performance reduced grapheme oxide/ZnCo layered double hydroxide electrocatalyst for efficient water oxidation. Dalton Trans. 2014, 43, 15119–15125. [Google Scholar] [CrossRef] [PubMed]
- Long, X.; Li, J.K.; Xiao, S.; Yan, K.Y.; Wang, Z.L.; Chen, H.N.; Yang, S.H. A strongly coupled graphene and FeNi double hydroxide hybrid as an excellent electrocatalyst for the oxygen evolution reaction. Angew. Chem. Int. Ed. 2014, 53, 7584–7588. [Google Scholar] [CrossRef] [PubMed]
- Ma, W.; Ma, R.Z.; Wu, J.H.; Sun, P.Z.; Liu, X.H.; Zhou, K.C.; Sasaki, T. Development of efficient electrocatalysts via molecular hybridization of NiMn layered double hydroxide nanosheets and grapheme. Nanoscale 2016, 8, 10425–10432. [Google Scholar] [CrossRef] [PubMed]
- Vargas, G.; Vazquez, S.J.; Oliver, T.M.A.; Ramos, S.L.; Flores, M.G.; Reguera, E. Influence on the electrocatalytic water oxidation of M2+/M3+ cation arrangement in NiFe LDH: Experimental and theoretical DFT evidences. Electrocatalysis 2017, 8, 383–391. [Google Scholar] [CrossRef]
- Han, N.; Zhao, F.P.; Li, Y.G. Ultrathin nickel–iron layered double hydroxide nanosheets intercalated with molybdate anions for electrocatalytic water oxidation. J. Mater. Chem. A 2015, 3, 16348–16353. [Google Scholar] [CrossRef]
- Gao, R.; Yan, D.P. Fast formation of single-unit-cell-thick and defect-rich layered double hydroxide nanosheets with highly enhanced oxygen evolution reaction for water splitting. Nano Res. 2018, 11, 1883–1894. [Google Scholar] [CrossRef]
- Luo, M.; Cai, Z.; Wang, C.; Bi, Y.M.; Qian, L.; Hao, Y.C.; Li, L.; Kuang, Y.; Li, Y.P.; Lei, X.D.; et al. Phosphorus oxoanion-intercalated layered double hydroxides for high-performance oxygen evolution. Nano Res. 2017, 10, 1732–1739. [Google Scholar] [CrossRef]
- Ge, X.; Gu, C.D.; Wang, X.L.; Tu, J.P. Ionothermal synthesis of cobalt iron layered double hydroxides (LDHs) with expanded interlayer spacing as advanced electrochemical materials. J. Mater. Chem. A 2014, 2, 17066–17076. [Google Scholar] [CrossRef]
- Jin, H.Y.; Mao, S.J.; Zhan, G.P.; Xu, F.; Bao, X.B.; Wang, Y. Fe incorporated α-Co(OH)2 nanosheet with remarkably improved activity towards oxygen evolution reaction. J. Mater. Chem. A 2017, 5, 1078–1084. [Google Scholar] [CrossRef]
- Wang, D.Y.; Costa, F.R.; Vyalikh, A.; Leuteritz, A.; Scheler, U.; Jehnichen, D.; Wagenknech, U.; Häussler, L.; Heinrich, G. One-step synthesis of organic LDH and its comparison with regeneration and anion exchange method. Chem. Mater. 2009, 21, 4490–4497. [Google Scholar] [CrossRef]
- Xu, H.J.; Wang, B.K.; Shan, C.F.; Xi, P.X.; Liu, W.S.; Tang, Y. Ce-doped NiFe-layered double hydroxide ultrathin nanosheets/nanocarbon hierarchical nanocomposite as an efficient oxygen evolution catalyst. ACS Appl. Mater. Interfaces 2018, 10, 6336–6345. [Google Scholar] [CrossRef] [PubMed]
- Trotochaud, L.; Young, S.L.; Ranney, J.K.; Boettcher, S.W. Nickel−iron oxyhydroxide oxygen-evolution electrocatalysts: The role of intentional and incidental iron incorporation. J. Am. Chem. Soc. 2014, 136, 6744–6753. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.Y.C.; Dang, L.; Liang, H.F.; Bi, W.L.; Gerken, J.B.; Jin, S.; Alp, E.E.; Stahl, S.S. Operando analysis of NiFe and Fe oxyhydroxide electrocatalysts for water oxidation: Detection of Fe4+ by Mössbauer spectroscopy. J. Am. Chem. Soc. 2015, 137, 15090–15093. [Google Scholar] [CrossRef] [PubMed]
- Gong, M.; Dai, H.J. A mini review of NiFe-based materials as highly active oxygen evolution reaction electrocatalysts. Nano Res. 2015, 8, 23–39. [Google Scholar] [CrossRef]
- Shao, M.F.; Zhang, R.K.; Li, Z.H.; Wei, M.; Evans, D.G.; Duan, X. Layered double hydroxides toward electrochemical energy storage and conversion: Design, synthesis and applications. Chem. Commun. 2015, 51, 15880–15893. [Google Scholar] [CrossRef] [PubMed]
- Dionigi, F.; Strasser, P. NiFe-based (oxy)hydroxide catalysts for oxygen evolution reaction in non-acidic electrolytes. Adv. Energy Mater. 2016, 6, 1600621. [Google Scholar] [CrossRef]
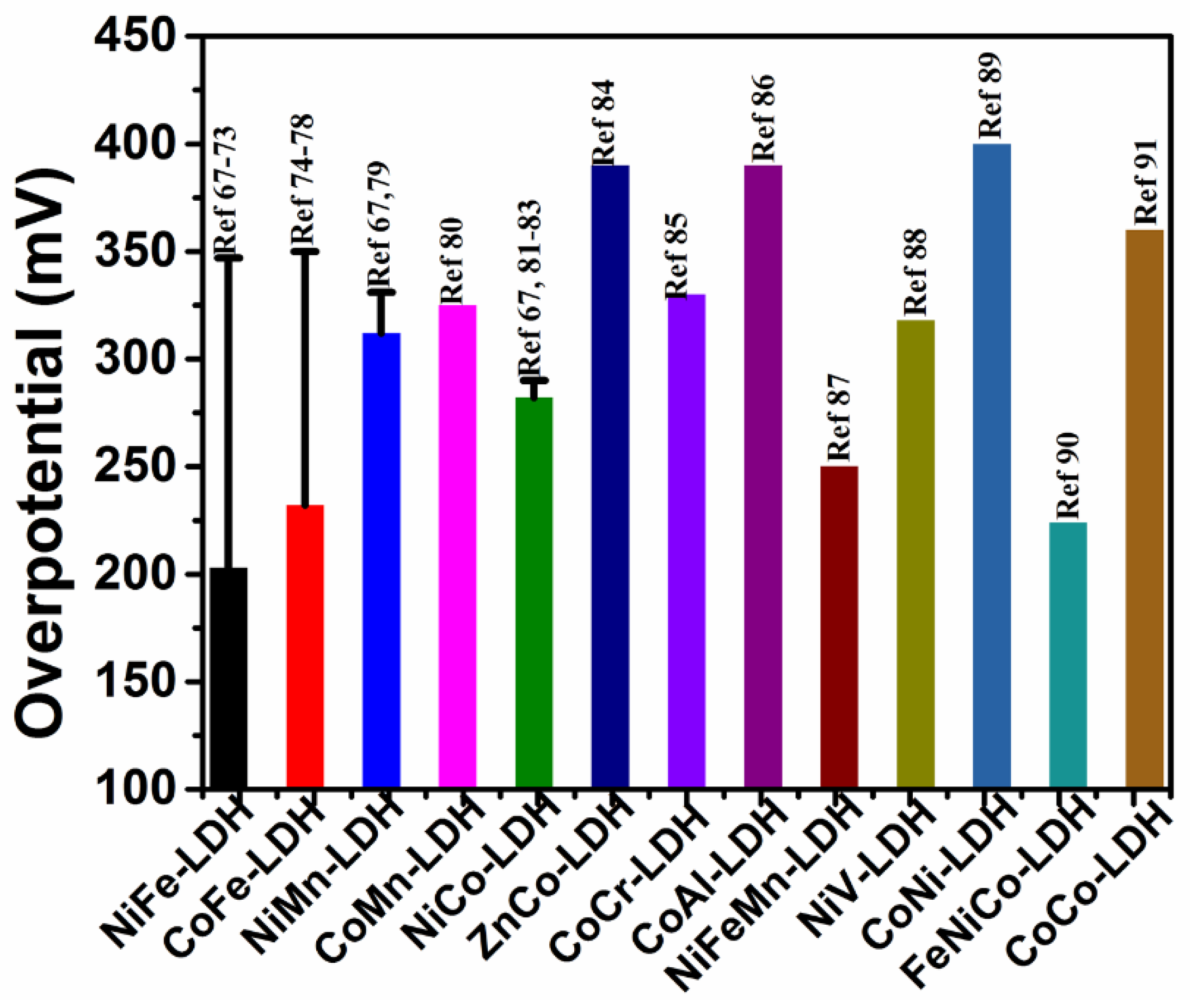
- Burke, M.S.; Enman, L.J.; Batchellor, A.S.; Zou, S.H.; Boettcher, S.W. Oxygen evolution reaction electrocatalysis on transition metal oxides and (oxy)hydroxides: Activity trends and design principles. Chem. Mater. 2015, 27, 7549–7558. [Google Scholar] [CrossRef]
- Tang, C.; Titirici, M.M.; Zhang, Q. A review of nanocarbons in energy electrocatalysis: Multifunctional substrates and highly active sites. J. Energy Chem. 2017, 26, 1077–1093. [Google Scholar] [CrossRef]
- Suen, N.T.; Hung, S.F.; Quan, Q.; Zhang, N.; Xu, Y.J.; Chen, H.M. Electrocatalysis for the oxygen evolution reaction: recent development and future perspectives. Chem. Soc. Rev. 2017, 46, 337–365. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.B.; Zhao, C. Iron-doped nickel phosphate as synergistic electrocatalyst for water oxidation. Chem. Mater. 2016, 28, 5659–5666. [Google Scholar] [CrossRef]
- Xi, W.; Ren, Z.Y.; Kong, L.J.; Wu, J.; Du, S.C.; Zhu, J.Q.; Xue, Y.Z.; Meng, H.Y.; Fu, H.G. Dual-valence nickel nanosheets covered with thin carbon as bifunctional electrocatalysts for full water splitting. J. Mater. Chem. A 2016, 4, 7297–7304. [Google Scholar] [CrossRef]
- Pfrommer, J.; Azarpira, A.; Steigert, A.; Olech, K.; Menezes, P.W.; Duarte, R.F.; Liao, X.X.; Wilks, R.G.; Bär, M.; Thomas, S.N.; et al. Active and stable nickel-based electrocatalysts based on the ZnO:Ni system for water oxidation in alkaline media. ChemCatChem 2017, 9, 672–676. [Google Scholar] [CrossRef]
- Luo, P.; Zhang, H.J.; Liu, L.; Zhang, Y.; Deng, J.; Xu, C.H.; Hu, N.; Wang, Y. Targeted synthesis of unique nickel sulfide (NiS, NiS2) microarchitectures and the applications for the enhanced water splitting system. ACS Appl. Mater. Interfaces 2017, 9, 2500–2508. [Google Scholar] [CrossRef] [PubMed]
- Friebel, D.; Louie, M.W.; Bajdich, M.; Sanwald, K.E.; Cai, Y.; Wise, A.M.; Cheng, M.J.; Sokaras, D.; Weng, T.C.; Mori, R.A.; et al. Identification of highly active Fe sites in (Ni,Fe)OOH for electrocatalytic water splitting. J. Am. Chem. Soc. 2015, 137, 1305–1313. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.; Wang, H.F.; Wang, H.S.; Wei, F.; Zhang, Q. Guest–host modulation of multi-metallic (oxy)hydroxides for superb water oxidation. J. Mater. Chem. A 2016, 4, 3210–3216. [Google Scholar] [CrossRef]
- Gorlin, M.; Chernev, P.; Araujo, J.F.; Reier, T.; Dresp, S.; Paul, B.; Krahnert, R.; Dau, H.; Strasser, P. Oxygen evolution reaction dynamics, faradaic charge efficiency, and the active metal redox states of Ni–Fe oxide water splitting electrocatalysts. J. Am. Chem. Soc. 2016, 138, 5603–5614. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.J.; Zhou, J.; Wu, C.Q.; Wang, C.D.; Zhang, Y.K.; Liu, D.B.; Lin, Y.X.; Jiang, H.L.; Song, L. Integrated flexible electrode for oxygen evolution reaction: layered double hydroxide coupled with single-walled carbon nanotubes film. ACS Sustainable Chem. Eng. 2018, 6, 2911–2915. [Google Scholar] [CrossRef]
- Yang, Y.; Dang, L.; Shearer, M.J.; Sheng, H.Y.; Li, W.J.; Chen, J.; Xiao, P.; Zhang, Y.H.; Hamers, R.J.; Jin, S. Highly active trimetallic NiFeCr layered double hydroxide electrocatalysts for oxygen evolution reaction. Adv. Energy Mater. 2018, 1703189. [Google Scholar] [CrossRef]
- Sun, X.H.; Shao, Q.; Pi, Y.C.; Guo, J.; Huang, X.Q. A general approach to synthesise ultrathin NiM(M=Fe, Co, Mn) hydroxide nanosheets as high performance low-cost electrocatalysts for overall water splitting. J. Mater. Chem. A 2017, 5, 7769–7775. [Google Scholar] [CrossRef]
- Li, Z.H.; Shao, M.F.; An, H.L.; Wang, Z.X.; Xu, S.M.; Wei, M.; Evans, D.G.; Duan, X. Fast electrosynthesis of Fe-containing layered double hydroxide arrays toward highly efficient electrocatalytic oxidation reactions. Chem. Sci. 2015, 6, 6624–6631. [Google Scholar] [CrossRef] [PubMed]
- Li, X.M.; Hao, X.G.; Wang, Z.D.; Abudula, A.; Guan, G.Q. In-situ intercalation of NiFe LDH materials: An efficient approach to improve electrocatalytic activity and stability for water splitting. J. Power Sources 2017, 347, 193–200. [Google Scholar] [CrossRef]
- Yang, H.D.; Luo, S.; Bao, Y.; Luo, Y.T.; Jin, J.; Ma, J.T. In situ growth of ultrathin Ni–Fe LDH nanosheets for high performance oxygen evolution reaction. Inorg. Chem. Front. 2017, 4, 1173–1181. [Google Scholar] [CrossRef]
- Xu, Y.Q.; Hao, Y.C.; Zhang, G.X.; Lu, Z.Y.; Han, S.; Li, Y.P.; Sun, X.M. Room-temperature synthetic NiFe layered double hydroxide with different anions intercalation as an excellent oxygen evolution catalyst. RSC Adv. 2015, 5, 55131–55135. [Google Scholar] [CrossRef]
- Zhang, Y.; Shao, Q.; Pi, Y.C.; Guo, J.; Huang, X.Q. A cost-effiient bifunctional ultrathin nanosheets array for electrochemical overall water splitting. Small 2017, 13, 1700355. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.Q.; Liu, Y.H.; Xiao, D.; Sun, J. Ultrafast and large scale preparation of superior catalyst for oxygen evolution reaction. J. Power Sources 2017, 365, 320–326. [Google Scholar] [CrossRef]
- Zhou, T.T.; Cao, Z.; Wang, H.; Gao, Z.; Lia, L.; Mab, H.Y.; Zhao, Y.F. Ultrathin Co–Fe hydroxide nanosheet arrays for improved oxygen evolution during water splitting. RSC Adv. 2017, 7, 22818–22824. [Google Scholar] [CrossRef]
- Feng, L.X.; Li, A.R.; Li, Y.X.; Liu, J.; Wang, L.D.Y.; Huang, L.Y.; Wang, Y.; Ge, X.B. A highly active CoFe layered double hydroxide for water splitting. ChemPlusChem 2017, 82, 483–488. [Google Scholar] [CrossRef]
- Zhou, P.; Wang, Y.Y.; Xie, C.; Chen, C.; Liu, H.W.; Chen, R.; Huo, J.; Wang, S.Y. Acid-etched layered double hydroxides with rich defects for enhancing the oxygen evolution reaction. Chem. Commun. 2017, 53, 11778–11781. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.F.; Yang, S.; Zhang, B.; Yang, H.G. Defect-rich ultrathin cobalt−iron layered double hydroxide for electrochemical overall water splitting. ACS Appl. Mater. Interfaces 2016, 8, 34474–34481. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.Y.; Zhang, Y.Q.; Liu, Z.J.; Xie, C.; Feng, S.; Liu, D.D.; Shao, M.F.; Wang, S.Y. Layered double hydroxide nanosheets with multiple vacancies obtained by dry exfoliation as highly efficient oxygen evolution electrocatalysts. Angew. Chem. Int. Ed. 2017, 56, 5867–5871. [Google Scholar] [CrossRef] [PubMed]
- Sumboja, A.; Chen, J.W.; Zong, Y.; Lee, P.S.; Liu, Z.L. NiMn layered double hydroxide as efficient electrocatalyst for oxygen evolution reaction and its application in rechargeable Zn air batteries. Nanoscale 2017, 9, 774–780. [Google Scholar] [CrossRef] [PubMed]
- Song, F.; Hu, X.L. Ultrathin cobalt–manganese layered double hydroxide is an efficient oxygen evolution catalyst. J. Am. Chem. Soc. 2014, 136, 16481–16484. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Zhang, A.L.; Li, L.L.; Ai, L.H. Nickel−cobalt layered double hydroxide nanosheets as high performance electrocatalyst for oxygen evolution reaction. J. Power Sources 2015, 278, 445–451. [Google Scholar] [CrossRef]
- Liu, W.J.; Bao, J.; Guan, M.L.; Zhao, Y.; Lian, J.B.; Qiu, J.X.; Xu, L.; Huang, Y.P.; Qian, J.; Li, H.M. Nickel–cobalt–layered double hydroxide nanosheet arrays on Ni foam as a bifunctional electrocatalyst for overall water splitting. Dalton Trans. 2017, 46, 8372–8376. [Google Scholar] [CrossRef] [PubMed]
- Liang, H.F.; Meng, F.; Acevedo, M.C.; Li, L.C.; Forticaux, A.; Xiu, L.C.; Wang, Z.C.; Ji, S. Hydrothermal continuous flow synthesis and exfoliation of NiCo layered double hydroxide nanosheets for enhanced oxygen evolution catalysis. Nano Lett. 2015, 15, 1421–1427. [Google Scholar] [CrossRef] [PubMed]
- Qiao, C.; Zhang, Y.; Zhu, Y.Q.; Cao, C.B.; Bao, X.H.; Xu, J.Q. One–step synthesis of zinc–cobalt layered double hydroxide (Zn–Co–LDH) nanosheets for high efficiency oxygen evolution reaction. J. Mater. Chem. A 2015, 3, 6878–6883. [Google Scholar] [CrossRef]
- Dong, C.L.; Yuan, X.T.; Wang, X.; Liu, X.Y.; Dong, W.J.; Wang, R.Q.; Duan, Y.H.; Huang, F.Q. Rational design of cobalt–chromium layered double hydroxide as a highly efficient electrocatalyst for water oxidation. J. Mater. Chem. A 2016, 4, 11292–11298. [Google Scholar] [CrossRef]
- Yang, F.K.; Sliozberg, K.R.; Sinev, I.; Antoni, H.; Bähr, A.; Ollegott, K.; Xia, W.; Mas, J.; Grünert, W.; Cuenya, B.R.; et al. Synergistic effect of cobalt and iron in layered double hydroxide catalysts for the oxygen evolution reaction. ChemSusChem 2017, 10, 156–165. [Google Scholar] [CrossRef] [PubMed]
- Lu, Z.Y.; Qian, L.; Tian, Y.; Li, Y.P.; Sun, X.M.; Duan, X. Ternary NiFeMn layered double hydroxides as highly-efficient oxygen evolution catalysts. Chem. Commun. 2016, 52, 90–911. [Google Scholar] [CrossRef] [PubMed]
- Fan, K.; Chen, H.; Ji, Y.F.; Huang, H.; Claesson, P.M.; Daniel, Q.; Bertrand Philippe, B.; Rensmo, H.; Li, F.S.; Luo, Y.; et al. Nickel–vanadium monolayer double hydroxide for efficient electrochemical water oxidation. Nat. Commun. 2016, 7, 11981. [Google Scholar] [CrossRef] [PubMed]
- Yoon, S.H.; Yun, J.Y.; Lim, J.H.; Yoo, B.Y. Enhanced electrocatalytic properties of electrodeposited amorphous cobalt-nickel hydroxide nanosheets on nickel foam by the formation of nickel nanocones for the oxygen evolution reaction. J. Alloys Compd. 2017, 693, 964–969. [Google Scholar] [CrossRef]
- Long, X.; Xiao, S.; Wang, Z.L.; Zheng, X.L.; Yang, S.H. Co intake mediated formation of ultrathin nanosheets of transition metal LDH—an advanced electrocatalyst for oxygen evolution reaction. Chem. Commun. 2015, 51, 1120–1123. [Google Scholar] [CrossRef] [PubMed]
- Song, F.; Hu, X.L. Exfoliation of layered double hydroxides for enhanced oxygen evolution catalysis. Nat. Commun. 2014, 5, 4477. [Google Scholar] [CrossRef] [PubMed]
- Yan, D.F.; Li, Y.X.; Huo, J.; Chen, R.; Dai, L.M.; Wang, S.Y. Defect chemistry of nonprecious-metal electrocatalysts for oxygen reactions. Adv. Mater. 2017, 29, 1606459. [Google Scholar] [CrossRef] [PubMed]
- Dogan, F.; Long, B.R.; Croy, J.R.; Gallagher, K.G.; Iddir, H.; Russell, J.T.; Balasubramanian, M.; Key, B. Re-entrant lithium local environments and defect driven electrochemistry of Li- and Mn-rich Li-ion battery cathodes. J. Am. Chem. Soc. 2015, 6, 2328–2335. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.Q.; Xu, K.; Wu, C.Z.; Zhao, J.Y.; Xie, Y. Surface chemical-modification for engineering the intrinsic physical properties of inorganic two-dimensional nanomaterials. Chem. Soc. Rev. 2015, 44, 637–646. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, P.; Brezesinski, T.; Sann, J.; Lotnyk, A.; Eufinger, J.P.; Kienle, L.; Janek, J. Defect chemistry of oxide nanomaterials with high surface area: Ordered mesoporous thin films of the oxygen storage catalyst CeO2–ZrO2. ACS Nano 2013, 7, 2999–3013. [Google Scholar] [CrossRef] [PubMed]
- Dou, L.; Wong, A.B.; Yu, Y.; Lai, M.L.; Kornienko, N.; Eaton, S.W.; Fu, A.; Bischak, C.G.; Ma, J.; Ding, T.; et al. Atomically thin two-dimensional organic-inorganic hybrid perovskites. Science 2015, 349, 1518–1521. [Google Scholar] [CrossRef] [PubMed]
- Smith, R.J.; King, P.J.; Lotya, M.; Wirtz, C.; Khan, U.; De, S.; O’Neill, A.; Duesberg, G.C.; Grunlan, J.C.; Moriarty, G.; et al. Large–scale exfoliation of inorganic layered compounds in aqueous surfactant solutions. Adv. Mater. 2011, 23, 3944–3948. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Shao, M.F.; Zhang, C.; Zhao, J.W.; He, S.; Rao, D.M.; Wei, M.; Evans, D.G.; Duan, X. Hierarchical CoNi-sulfie nanosheet arrays derived from layered double hydroxides toward effiient hydrazine electrooxidation. Adv. Mater. 2017, 29, 1604080. [Google Scholar] [CrossRef] [PubMed]
- Subbaiah, Y.P.V.; Saji, K.J.; Tiwari, A. Atomically thin MoS2: A versatile nongraphene 2D material. Adv. Funct. Mater. 2016, 26, 2046–2069. [Google Scholar] [CrossRef]
- Zhang, R.K.; Shao, M.F.; Li, Z.H.; Ning, F.Y.; Wei, M.; Evans, D.G.; Duan, X. Photo electrochemical catalysis toward selective anaerobic oxidation of alcohols. Chem. Eur. J. 2017, 23, 8142–8147. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.F.; Zhang, X.; Jia, X.D.; Waterhouse, G.I.N.; Shi, R.; Zhang, X.R.; Zhan, F.; Tao, Y.; Wu, L.Z.; Tung, C.H.; et al. Sub-3 nm Ultrafine monolayer layered double hydroxide nanosheets for electrochemical water oxidation. Adv. Energy Mater. 2018. [Google Scholar] [CrossRef]
- Ma, W.L.; Wang, L.; Xue, J.Y.; Cui, H.T. Ultra-large scale synthesis of Co–Ni layered double hydroxides monolayer nanosheets by a solvent-free bottom-up strategy. J. Alloys Compd. 2016, 662, 315–319. [Google Scholar] [CrossRef]
- Hu, G.; Wang, N.; O’Hare, D.; Davis, J. One-step synthesis and AFM imaging of hydrophobic LDH monolayers. Chem. Commun. 2006, 0, 287–289. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.F.; Li, B.; Wang, Q.; Gao, W.; Wang, C.J.; Wei, M.; Evans, D.G.; Duan, X.; O’Hare, D. NiTi-Layered double hydroxides nanosheets as efficient photocatalysts for oxygen evolution from water using visible light. Chem. Sci. 2014, 5, 951–958. [Google Scholar] [CrossRef]
- Ma, R.Z.; Liu, R.P.; Li, L.; Iyia, N.; Sasak, T. Exfoliating layered double hydroxides in formamide: A method to obtain positively charged nanosheets. J. Mater. Chem. 2006, 16, 3809–3813. [Google Scholar] [CrossRef]
- Mao, N.; Zhou, C.H.; Tong, D.S.; Yu, W.H.; Lind, C.X.C. Exfoliation of layered double hydroxide solids into functional nanosheets. Appl. Clay Sci. 2017, 144, 60–78. [Google Scholar] [CrossRef]
- Yu, J.; Wang, Q.; O’Hare, D.; Sun, L.Y. Preparation of two dimensional layered double hydroxide nanosheets and their applications. Chem. Soc. Rev. 2017, 46, 5950–5974. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Cen, X.; Peng, H.D.; Guo, S.Z.; Wang, W.Z.; Liu, T.X. Heterogeneous ultrathin films of poly (vinyl alcohol)/layered double hydroxide and montmorillonite nanosheets via layer-by-layer assembly. J. Phys. Chem. B 2009, 46, 15225–15230. [Google Scholar] [CrossRef] [PubMed]
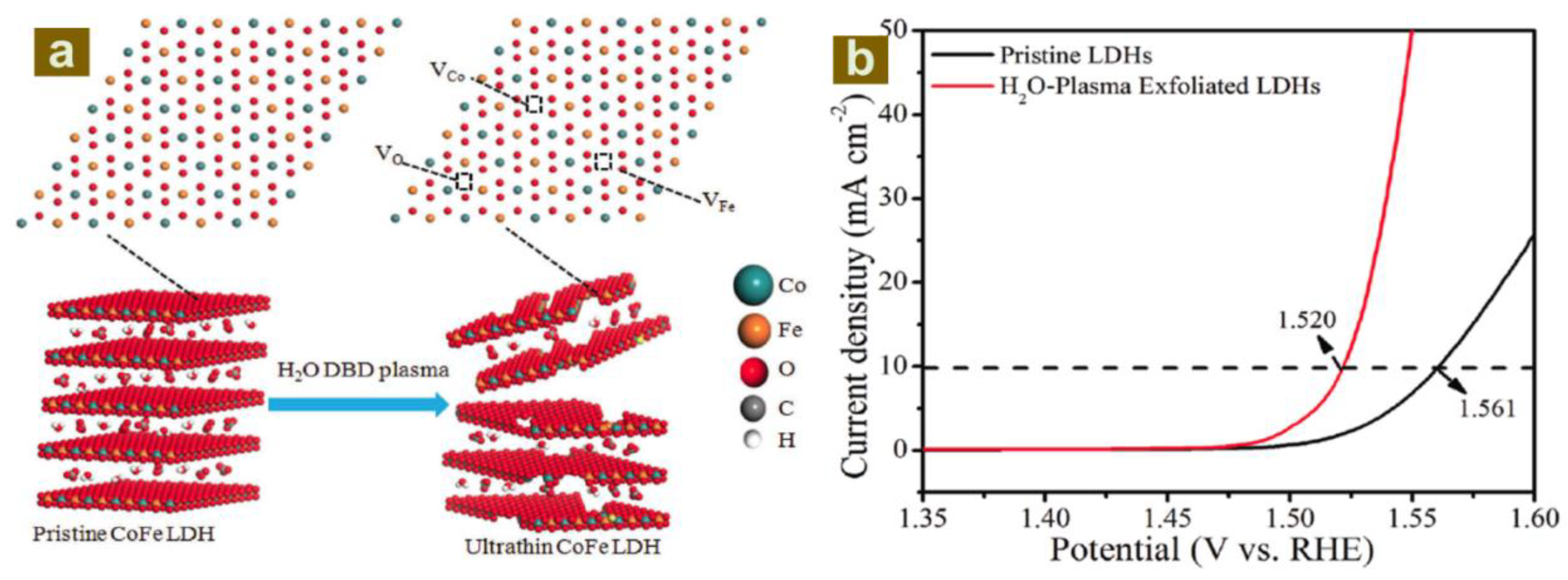
- Liu, R.; Wang, Y.Y.; Liu, D.D.; Zou, Y.Q.; Wang, S.Y. Water-plasma-enabled exfoliation of ultrathin layered double hydroxide nanosheets with multivacancies for water oxidation. Adv. Mater. 2017, 29, 1701546. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.K.; Shao, M.F.; Xu, S.M.; Ning, F.Y.; Zhou, L.; Wei, M. Photo-assisted synthesis of zinc-iron layered double hydroxides/TiO2 nanoarrays toward highly-efficient photoelectrochemical water splitting. Nano Energy 2017, 33, 21–28. [Google Scholar] [CrossRef]
- Zhou, L.; Jiang, S.; Liu, Y.K.; Shao, M.F.; Wei, M.; Duan, X. Ultrathin CoNiP@layered double hydroxides core–shell nanosheets arrays for largely enhanced overall water splitting. ACS Appl. Energy Mater. 2018, 1, 623–631. [Google Scholar] [CrossRef]
- Lin, J.H.; Jia, H.N.; Liang, H.Y.; Cheng, S.L.; Cai, Y.F.; Qi, J.L.; Qu, C.Q.; Cao, J.; Fei, W.D.; Feng, J.C. Hierarchical CuCo2S4@NiMn-layered double hydroxide core-shell hybrid arrays as electrodes for supercapacitors. Chem. Eng. J. 2018, 336, 562–569. [Google Scholar] [CrossRef]
- Wang, J.H.; Cui, W.; Liu, Q.; Xing, Z.C.; Asiri, A.M.; Sun, X.P. Recent progress in cobalt−based heterogeneous catalysts for electrochemical water splitting. Adv. Mater. 2016, 28, 215–230. [Google Scholar] [CrossRef] [PubMed]
- Chi, H.M.; Qin, B.; Fu, L.; Jia, J.; Yi, B.L.; Shao, Z.G. Vertically aligned FeOOH/NiFe layered double hydroxides electrode. ACS Appl. Mater. Interfaces 2017, 9, 464–471. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.; Jang, J.W.; Park, Y.B.; Kim, J.Y.; Magesh, G.; Kim, J.H.; Seol, M.; Yong, K.J.; Lee, K.H.; Lee, J.S. An exceptionally facile method to produce layered double hydroxides on a conducting substrate and their application for solar water splitting without an external bias. Energy Environ. Sci. 2014, 7, 2301–2307. [Google Scholar] [CrossRef]
- Yu, C.; Liu, Z.B.; Han, X.T.; Huang, H.W.; Zhao, C.T.; Yang, J.; Qiu, J. NiCo-layered double hydroxides vertically assembled on carbon fiber papers as binder-free high-active electrocatalysts for water oxidation. Carbon 2016, 110, 1–7. [Google Scholar] [CrossRef]
- Zhao, M.Q.; Zhang, Q.; Huang, J.Q.; Wei, F. Hierarchical nanocomposites derived from nanocarbons and layered double hydroxides−properties, synthesis, and applications. Adv. Funct. Mater. 2012, 22, 675–694. [Google Scholar] [CrossRef]
- Ma, Y.; Wang, Y.C.; Xie, D.H.; Gu, Y.; Zhang, H.M.; Wang, G.Z.; Zhang, Y.X.; Zhao, H.J.; Wong, P.K. NiFe-layered double hydroxide nanosheet arrays supported on carbon cloth for highly sensitive detection of nitrite. ACS Appl. Mater. Interfaces 2018, 10, 6541–6551. [Google Scholar] [CrossRef] [PubMed]
- Ning, F.Y.; Shao, M.F.; Zhang, C.L.; Xu, S.M.; Wei, M.; Duan, X. Co3O4@ layered double hydroxide core/shell hierarchical nanowire arrays for enhanced supercapacitance performance. Nano Energy 2014, 7, 134–142. [Google Scholar] [CrossRef]
- Li, X.Y.; Wang, X.; Yuan, X.T.; Dong, W.J.; Huang, F.Q. Rational composition and structural design of in situ grown nickel-based electrocatalysts for efficient water electrolysis. J. Mater. Chem. A 2016, 4, 167–172. [Google Scholar] [CrossRef]
- Lu, Z.Y.; Sun, M.; Xu, T.H.; Li, Y.J.; Xu, W.W.; Chang, Z.; Ding, Y.; Sun, X.M.; Jiang, L. Superaerophobic electrodes for direct hydrazine fuel cells. Adv. Mater. 2015, 27, 2361–2366. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Shao, M.F.; Zhou, L.; Li, Z.H.; Xiao, K.M.; Wei, M. Hierarchical NiFe layered double hydroxide hollow microspheres with highly-efficient behavior toward oxygen evolution reaction. ACS Appl. Mater. Interfaces 2016, 8, 33697–33703. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.P.; Li, Y.Y.; Fan, H.J.; Zhu, Z.H.; Jiang, J.; Ding, R.M.; Hu, Y.Y.; Huang, X.T. Iron oxide-based nanotube arrays derived from sacrificial template-accelerated hydrolysis: Large-area design and reversible lithium storage. Chem. Mater. 2010, 22, 212–217. [Google Scholar] [CrossRef]
- Tang, Y.Q.; Fang, X.Y.; Zhang, X.; Fernandes, G.; Yan, Y.; Yan, D.P.; Xiang, X.; He, J. Space–Confined earth–abundant bifunctional electrocatalyst for high-efficiency water splitting. ACS Appl. Mater. Interfaces 2017, 9, 36762–36771. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Li, T.; Lu, Z.Y.; Sun, X.M.; Liu, J.F. Hierarchical construction of an ultrathin layered double hydroxide nanoarray for highly-efficient oxygen evolution reaction. Nanoscale 2014, 6, 11789–11794. [Google Scholar] [CrossRef] [PubMed]
- Blanchard, P.; Huchet, L.; Lillain, E.; Roncali, J. ation template assisted electrosynthesis of a highly π-conjugated polythiophene containing oligooxyethylene segments. Electrochem. Commun. 2000, 2, 1–5. [Google Scholar] [CrossRef]
- Surendranath, Y.; Dincǎ, M.; Nocera, D.G. Electrolyte-dependent electrosynthesis and activity of cobalt-based water oxidation catalysts. J. Am. Chem. Soc. 2009, 131, 2615–2620. [Google Scholar] [CrossRef] [PubMed]
- Francke, R.; Little, R.D. Three-dimensional hierarchical metal oxide–carbon electrode materials for highly efficient microbial electrosynthesis. Chem. Soc. Rev. 2014, 43, 2492–2521. [Google Scholar] [CrossRef] [PubMed]
- Hamid, H.; Shiria, M.; Ehsani, S. Electrosynthesis of neodymium oxide nanorods and its nanocomposite with conjugated conductive polymer as a hybrid electrode material for highly capacitive pseudocapacitors. J. Colloid Interface Sci. 2017, 495, 102–110. [Google Scholar]
- Qiao, X.Y.; Wei, M.C.; Tian, D.; Xia, F.Q.; Chen, P.P.; Zhou, C.L. One-step electrosynthesis of cadmium/aluminum layered double hydroxides composite as electrochemical probe for voltammetric detection of anthracene. J. Electroanal. Chem. 2018, 808, 35–40. [Google Scholar] [CrossRef]
- Yamabe, K.; Goto, H. Electrosynthesis of conducting polymers in lecith in liquid crystal reaction field. Fibers Polym. 2018, 19, 248–253. [Google Scholar] [CrossRef]
- Shao, M.F.; Li, Z.H.; Zhang, R.K.; Ning, F.Y.; Wei, M.; Evans, D.G.; Duan, X. Hierarchical conducting polymer@clay core–shell arrays for flexible all-solid-state supercapacitor devices. Small 2015, 11, 3530–3538. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.X.; Hui, K.S.; Hui, K.N. One-dimensional core–shell architecture composed of silver nanowire@hierarchical nickel–aluminum layered double hydroxide nanosheet as advanced electrode materials for pseudocapacitor. J. Phys. Chem. C 2015, 119, 23358–23365. [Google Scholar] [CrossRef]
- Serrà, A.; Gómez, E.; Vallés, E. Novel electrodeposition media to synthesize CoNi-Pt core@shell stable mesoporous nanorods with very high active surface for methanol electro-oxidation. Electrochim. Acta 2015, 174, 630–639. [Google Scholar] [CrossRef]
- Yanilkina, V.V.; Nastapovaa, N.V.; Nasretdinovaa, G.R.; Fazleevaa, R.R.; Toropchinaa, А.V.; Osin, Y.N. Methylviologen mediated electrochemical reduction of AgCl—A new route to produce a silica core/Ag shell nanocomposite material in solution. Electrochem. Commun. 2015, 59, 60–63. [Google Scholar] [CrossRef]
- Li, Z.H.; Shao, M.F.; Zhou, L.; Zhang, R.K.; Zhang, C.; Wei, M.; Evans, D.G.; Duan, X. A flexible all-solid-state micro-supercapacitor based on hierarchical CuO@layered double hydroxide core–shell nanoarrays. Nano Energy 2016, 20, 294–304. [Google Scholar] [CrossRef]
- Dong, X.Y.; Wang, L.; Wang, D.; Li, C.; Jin, J. Layer-by-layer engineered Co–Al hydroxide nanosheets/graphene multilayer films as flexible electrode for supercapacitor. Langmuir 2012, 28, 293–298. [Google Scholar] [CrossRef] [PubMed]
- Han, J.B.; Lu, J.; Wei, M.; Wang, Z.L.; Duan, X. Heterogeneous ultrathin films fabricated by alternate assembly of exfoliated layered double hydroxides and polyanion. Chem. Commun. 2008, 5188–5190. [Google Scholar] [CrossRef] [PubMed]
- Yan, D.P.; Lu, J.; Wei, M.; Han, J.B.; Li, F.; Evans, D.G.; Duan, X. Ordered poly(p-phenylene)/layered double hydroxide ultrathin films with blue luminescence by layer-by-layer assembly. Angew. Chem. 2009, 121, 3119–3122. [Google Scholar] [CrossRef]
- Han, J.B.; Xu, X.Y.; Rao, X.Y.; Wei, M.; Evans, D.G.; Duan, X. Layer-by-layer assembly of layered double hydroxide/cobalt phthalocyanine ultrathin film and its application for sensors. J. Mater. Chem. 2011, 21, 2126–2130. [Google Scholar] [CrossRef]
- Liang, R.Z.; Tian, R.; Shi, W.Y.; Liu, Z.H.; Yan, D.P.; Wei, M.; Evans, D.G.; Duan, X. Temperature sensor based on CdTe quantum dots–layered double hydroxide ultrathin films via layer-by-layer assembly. Chem. Commun. 2013, 49, 969–971. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Zhao, J.W.; Zhou, L.; Li, Z.H.; Shao, M.F.; Wei, M. Layer-by-layer assembly of exfoliated layered double hydroxide nanosheets for enhanced electrochemical oxidation of water. J. Mater. Chem. A 2016, 4, 11516–11523. [Google Scholar] [CrossRef]
- Hunter, B.M.; Hieringer, W.; Winkler, J.R.; Gray, H.B.; Muller, A.M. Effect of interlayer anions on [NiFe]-LDH nanosheet water oxidation activity. Energy Environ. Sci. 2016, 9, 1734–1743. [Google Scholar] [CrossRef]
- Zhang, J.F.; Liu, J.Y.; Xi, L.F.; Yu, Y.F.; Chen, N.; Sun, S.H.; Wang, W.C.; Lange, K.M.; Zhang, B. Single-atom Au/NiFe layered double hydroxide electrocatalyst: Probing the origin of activity for oxygen evolution reaction. J. Am. Chem. Soc. 2018, 140, 3876–3879. [Google Scholar] [CrossRef] [PubMed]
- Ahn, H.S.; Bard, A.J. Surface interrogation scanning electrochemical microscopy of Ni1−xFexOOH (0 < x < 0.27) oxygen evolving catalyst: Kinetics of the “fast” iron sites. J. Am. Chem. Soc. 2016, 138, 313–318. [Google Scholar] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, J.; Jiang, S.; Shao, M.; Wei, M. Host-Guest Engineering of Layered Double Hydroxides towards Efficient Oxygen Evolution Reaction: Recent Advances and Perspectives. Catalysts 2018, 8, 214. https://doi.org/10.3390/catal8050214
Li J, Jiang S, Shao M, Wei M. Host-Guest Engineering of Layered Double Hydroxides towards Efficient Oxygen Evolution Reaction: Recent Advances and Perspectives. Catalysts. 2018; 8(5):214. https://doi.org/10.3390/catal8050214
Chicago/Turabian StyleLi, Jianming, Shan Jiang, Mingfei Shao, and Min Wei. 2018. "Host-Guest Engineering of Layered Double Hydroxides towards Efficient Oxygen Evolution Reaction: Recent Advances and Perspectives" Catalysts 8, no. 5: 214. https://doi.org/10.3390/catal8050214
APA StyleLi, J., Jiang, S., Shao, M., & Wei, M. (2018). Host-Guest Engineering of Layered Double Hydroxides towards Efficient Oxygen Evolution Reaction: Recent Advances and Perspectives. Catalysts, 8(5), 214. https://doi.org/10.3390/catal8050214




