Techniques for Preparation of Cross-Linked Enzyme Aggregates and Their Applications in Bioconversions
Abstract
1. Introduction
2. Cross-Linking Enzyme Immobilization
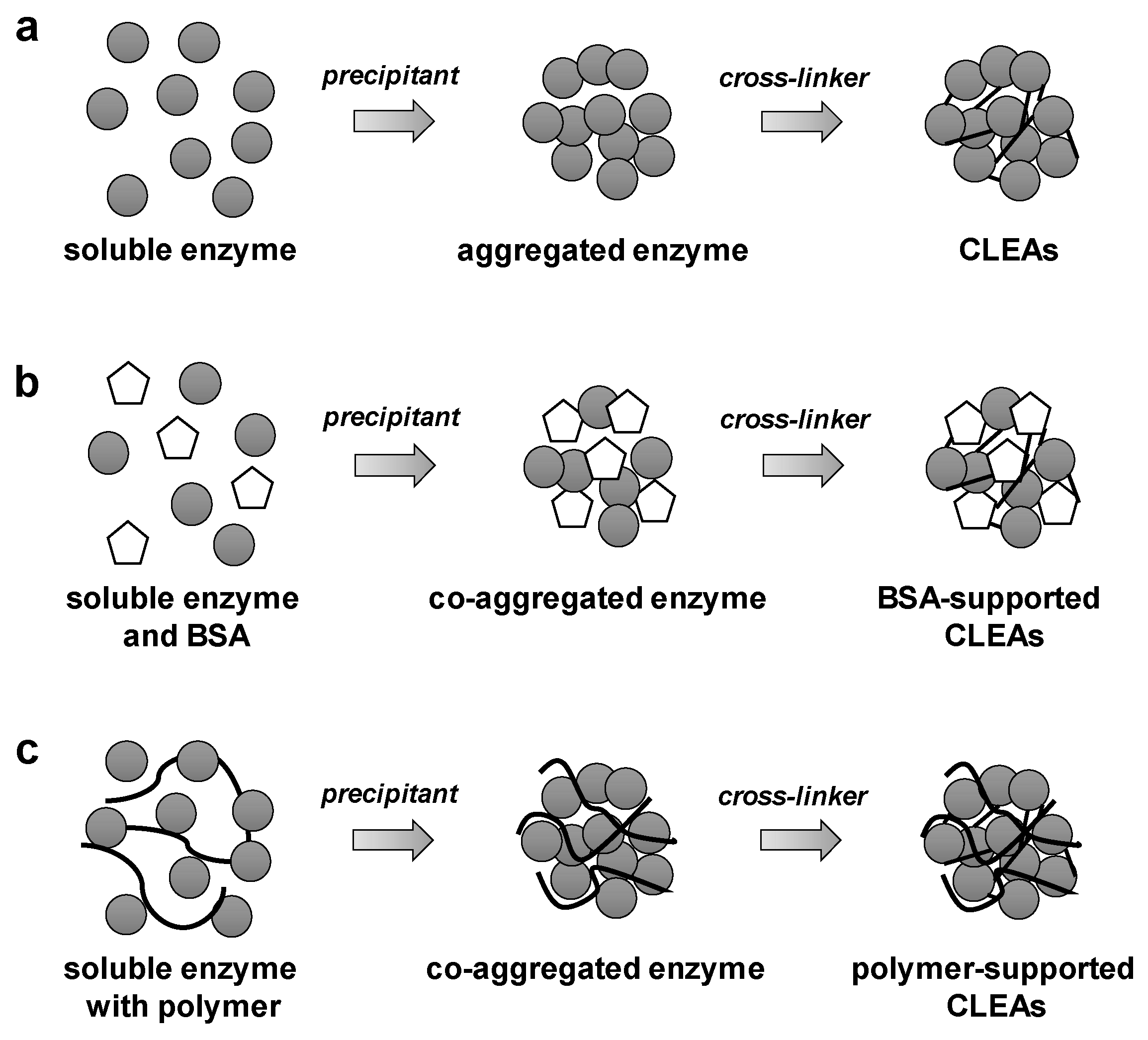
2.1. General Preparation of CLEAs
2.2. Optimization by Additives
2.2.1. Bovine Serum Albumin (BSA)
2.2.2. Ionic Polymer
2.2.3. Surfactants
2.2.4. Cations
2.3. Optimization by Precipitant Selection
2.4. Optimization by Cross-Linker
2.5. Novel Method for Preparation of CLEAs
2.5.1. CLEA Particle Size
2.5.2. Molecular-Imprinted CLEAs
2.6. Carrier-Bound CLEAs
2.6.1. CLEAs in Silica Material
2.6.2. Magnetic CLEAs
2.6.3. Entrapment CLEAs in Alginate Beads
3. Coimmobilization of Two or More Enzymes in CLEAs
3.1. Combi-CLEAs
3.2. Multi-CLEAs
4. Application for Processing by CLEAs
4.1. Chemical Synthesis
4.1.1. Pharmaceutical Chemicals
4.1.2. Polymers
4.1.3. Nanoparticles
4.2. Environmental Purposes
4.2.1. Decolorization and Detoxification of Dyes
4.2.2. Elimination of Endocrine-Disrupting Chemicals
4.2.3. Bioconversion of Agroindustrial Waste
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Madhavan, A.; Sindhu, R.; Binod, P.; Sukumaran, R.K.; Pandey, A. Strategies for design of improved biocatalysts for industrial applications. Bioresour. Technol. 2017, 245, 1304–1313. [Google Scholar] [CrossRef] [PubMed]
- Sheldon, R.A.; Woodley, J.M. Role of biocatalysis in sustainable chemistry. Chem. Rev. 2018, 118, 801–838. [Google Scholar] [CrossRef] [PubMed]
- Elleuche, S.; Schröder, C.; Sahm, K.; Antranikian, G. Extremozymes—Biocatalysts with unique properties from extremophilic microorganisms. Curr. Opin. Biotechnol. 2014, 29, 116–123. [Google Scholar] [CrossRef] [PubMed]
- Sheldon, R.A. Characteristic features and biotechnological applications of cross-linked enzyme aggregates (CLEAs). Appl. Microbiol. Biotechnol. 2011, 92, 467–477. [Google Scholar] [CrossRef] [PubMed]
- Franssen, M.C.; Steunenberg, P.; Scott, E.L.; Zuilhof, H.; Sanders, J.P. Immobilised enzymes in biorenewables production. Chem. Soc. Rev. 2013, 42, 6491–6533. [Google Scholar] [CrossRef] [PubMed]
- Bilal, M.; Iqbal, H.M.N.; Guo, S.; Hu, H.; Wang, W.; Zhang, X. State-of-the-art protein engineering approaches using biological macromolecules: A review from immobilization to implementation view point. Int. J. Biol. Macromol. 2018, 108, 893–901. [Google Scholar] [CrossRef] [PubMed]
- Miyazaki, M.; Portia Nagata, M.; Honda, T.; Yamaguchi, H. Bioorganic and biocatalytic reactions. In Microreactors in Organic Synthesis and Catalysis, 2nd ed.; Wiley-VCH: Hoboken, NJ, USA, 2013; Chapter 10. [Google Scholar]
- Honda, T.; Yamaguchi, H.; Miyazaki, M. Development of enzymatic reactions in miniaturized reactors. In Innovations and Future Directions Applied Bioengineering; Wiley-VCH: Hoboken, NJ, USA, 2017; pp. 99–166. [Google Scholar]
- Wang, L.S.; Khan, F.; Micklefield, J. Selective covalent protein immobilization: Strategies and applications. Chem. Rev. 2009, 109, 4025–4053. [Google Scholar] [CrossRef] [PubMed]
- Asanomi, Y.; Yamaguchi, H.; Miyazaki, M.; Maeda, H. Enzyme-immobilized microfluidic process reactors. Molecules 2011, 16, 6041–6059. [Google Scholar] [CrossRef] [PubMed]
- Yiu, H.H.P.; Wright, P.A. Enzymes supported on ordered mesoporous solids: A special case of an inorganic–organic hybrid. J. Mater. Chem. 2005, 15, 3690–3700. [Google Scholar] [CrossRef]
- Cha, T.W.; Guo, A.; Zhu, X.Y. Enzymatic activity on a chip: The critical role of protein orientation. Proteomics 2005, 5, 416–419. [Google Scholar] [CrossRef] [PubMed]
- Zaborsky, O.R. Immobilized Enzymes; CRC Press: Cleveland, OH, USA, 1973. [Google Scholar]
- St. Clair, N.L.; Navia, M.A. Cross-linked enzyme crystals as robust biocatalysts. J. Am. Chem. Soc. 1992, 114, 7314–7316. [Google Scholar] [CrossRef]
- Roy, J.J.; Abraham, T.E. Strategies in making cross-linked enzyme crystals. Chem. Rev. 2004, 104, 3705–3722. [Google Scholar] [CrossRef]
- Cao, L.; van Langen, L.; Sheldon, R.A. Immobilised enzymes: Carrier-bound or carrier-free? Curr. Opin. Biotechnol. 2003, 14, 387–394. [Google Scholar] [CrossRef]
- Shah, S.; Sharma, A.; Gupta, M.N. Preparation of cross-linked enzyme aggregates by using bovine serum albumin as a proteic feeder. Anal. Biochem. 2006, 351, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Schoevaart, R.; Wolbers, M.W.; Golubovic, M.; Ottens, M.; Kieboom, A.P.; van Rantwijk, F.; van der Wielen, L.A.; Sheldon, R.A. Preparation, optimization, and structures of cross-linked enzyme aggregates (CLEAs). Biotechnol. Bioeng. 2004, 87, 754–762. [Google Scholar] [CrossRef] [PubMed]
- Majumder, A.B.; Gupta, M.N. Stabilization of Candida rugosa lipase during transacetylation with vinyl acetate. Bioresour. Technol. 2010, 101, 2877–2879. [Google Scholar] [CrossRef] [PubMed]
- Mateo, C.; Palomo, J.M.; van Langen, L.M.; van Rantwijk, F.; Sheldon, R.A. A new, mild cross-linking methodology to prepare cross-linked enzyme aggregates. Biotechnol. Bioeng. 2004, 86, 273–276. [Google Scholar] [CrossRef] [PubMed]
- López-Gallego, F.; Betancor, L.; Hidalgo, A.; Alonso, N.; Fernández-Lafuente, R.; Guisán, J.M. Coaggregation of enzymes and polyethyleneimine: A simple method to prepare stable and immobilized derivatives of glutaryl acylase. Biomacromolecules 2005, 6, 1839–1842. [Google Scholar] [CrossRef] [PubMed]
- Wilson, L.; Illanes, A.; Abián, O.; Pessela, B.C.; Fernández-Lafuente, R.; Guisán, J.M. Coaggregation of penicillin g acylase and polyionic polymers: An easy methodology to prepare enzyme biocatalysts stable in organic media. Biomacromolecules 2004, 5, 852–857. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Chen, Y.; Yang, L.; Li, M.; Zhang, J. Preparation of cross-linked enzyme aggregates of trehalose synthase via coaggregation with polyethyleneimine. Appl. Biochem. Biotechnol. 2014, 174, 2067–2078. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Ren, H.; Wang, Y.; Chen, K.; Fang, B.; Wang, S. Bioinspired immobilization of glycerol dehydrogenase by metal ion-chelated polyethyleneimines as artificial polypeptides. Sci. Rep. 2016, 6, 24163. [Google Scholar] [CrossRef] [PubMed]
- Honda, T.; Miyazaki, M.; Nakamura, H.; Maeda, H. Immobilization of enzymes on a microchannel surface through cross-linking polymerization. Chem. Commun. 2005, 40, 5062–5064. [Google Scholar] [CrossRef] [PubMed]
- Honda, T.; Miyazaki, M.; Nakamura, H.; Maeda, H. Facile preparation of an enzyme-immobilized microreactor using a cross-linking enzyme membrane on a microchannel surface. Adv. Synth. Catal. 2006, 348, 2163–2171. [Google Scholar] [CrossRef]
- Honda, T.; Miyazaki, M.; Yamaguchi, Y.; Nakamura, H.; Maeda, H. Integrated microreaction system for optical resolution of racemic amino acids. Lab Chip 2007, 7, 366–372. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, H.; Miyazaki, M.; Maeda, H. Proteolysis approach without chemical modification for a simple and rapid analysis of disulfide bonds using thermostable protease-immobilized microreactors. Proteomics 2010, 10, 2942–2949. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, H.; Miyazaki, M.; Kawazumi, H.; Maeda, H. Multidigestion in continuous flow tandem protease-immobilized microreactors for proteomic analysis. Anal. Biochem. 2010, 407, 12–18. [Google Scholar] [CrossRef] [PubMed]
- Lloret, L.; Eibes, G.; Moreira, M.T.; Feijoo, G.; Lema, J.M.; Miyazaki, M. Improving the catalytic performance of laccase using a novel continuous-flow microreactor. Chem. Eng. J. 2013, 223, 497–506. [Google Scholar] [CrossRef]
- Yamaguchi, H.; Miyazaki, M.; Asanomi, Y.; Maeda, H. Poly-lysine supported cross-linked enzyme aggregates with efficient enzymatic activity and high operational stability. Catal. Sci. Technol. 2011, 1, 1256–1261. [Google Scholar] [CrossRef]
- Theil, F. Enhancement of selectivity and reactivity of lipases by additives. Tetrahedron 2000, 56, 2905–3076. [Google Scholar] [CrossRef]
- Gupta, P.; Dutt, K.; Misra, S.; Raghuwanshi, S.; Saxena, R.K. Characterization of cross-linked immobilized lipase from thermophilic mould Thermomyces lanuginosa using glutaraldehyde. Bioresour. Technol. 2009, 100, 4074–4076. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Lorente, G.; Palomo, J.M.; Mateo, C.; Munilla, R.; Ortiz, C.; Cabrera, Z.; Guisán, J.M.; Fernandez-Lafuente, R. Glutaraldehyde cross-linking of lipases adsorbed on aminated supports in the presence of detergents leads to improved performance. Biomacromolecules 2006, 7, 2610–2615. [Google Scholar] [CrossRef] [PubMed]
- De Rose, S.A.; Novak, H.; Dowd, A.; Singh, S.; Lang, D.A.; Littlechild, J. Stabilization of a lipolytic enzyme for commercial application. Catalysts 2017, 7, 91. [Google Scholar] [CrossRef]
- Burhan, A.; Nisa, U.; Gökhan, C.; Ömer, C.; Ashabil, A.; Osman, G. Enzymatic properties of a novel thermostable, thermophilic, alkaline and chelator resistant amylase from an alkaliphilic Bacillus sp. isolate ANT-6. Process Biochem. 2003, 38, 1397–1403. [Google Scholar] [CrossRef]
- Torabizadeh, H.; Tavakoli, M.; Safari, M. Immobilization of thermostable α-amylase from Bacillus licheniformis by cross-linked enzyme aggregates method using calcium and sodium ions as additives. J. Mol. Catal. B: Enzym. 2014, 108, 13–20. [Google Scholar] [CrossRef]
- Khorshidi, K.J.; Lenjannezhadian, H.; Jamalan, M.; Zeinali, M. Preparation and characterization of nanomagnetic cross-linked cellulase aggregates for cellulose bioconversion. J. Chem. Technol. Biotechnol. 2016, 91, 539–546. [Google Scholar] [CrossRef]
- Dalal, S.; Sharma, A.; Gupta, M.N. A multipurpose immobilized biocatalyst with pectinase, xylanase and cellulase activities. Chem. Cent. J. 2007, 1, 16. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, L.T.; Yang, K.L. Uniform cross-linked cellulase aggregates prepared in millifluidic reactors. J. Colloid Interface Sci. 2014, 428, 146–151. [Google Scholar] [CrossRef] [PubMed]
- Perzon, A.; Dicko, C.; Çobanoğlu, Ö.; Yükselen, O.; Eryilmaz, J.; Dey, E.S. Cellulase cross-linked enzyme aggregates (CLEA) activities can be modulated and enhanced by precipitant selection. J. Chem. Technol. Biotechnol. 2017, 92, 1645–1649. [Google Scholar] [CrossRef]
- Devi, B.L.A.P.; Guo, Z.; Xu, X. Characterization of cross-linked lipase aggregates. J. Am. Oil Chem. Soc. 2009, 86, 637–642. [Google Scholar] [CrossRef]
- Rehman, S.; Bhatti, H.N.; Bilal, M.; Asgher, M. Cross-linked enzyme aggregates (CLEAs) of Pencilluim notatum lipase enzyme with improved activity, stability and reusability characteristics. Int. J. Biol. Macromol. 2016, 91, 1161–1169. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Xiao, C.; Chen, X.; Yang, L.; Qi, X.; Zheng, J.; Li, M.; Zhang, J. Preparation of cross-linked enzyme aggregates in water-in-oil emulsion: Application to trehalose synthase. J. Mol. Catal. B: Enzym. 2014, 100, 84–90. [Google Scholar] [CrossRef]
- Verheyen, E.; Schillemans, J.P.; van Wijk, M.; Demeniex, M.A.; Hennink, W.E.; van Nostrum, C.F. Challenges for the effective molecular imprinting of proteins. Biomaterials 2011, 32, 3008–3020. [Google Scholar] [CrossRef] [PubMed]
- Fishman, A.; Coganb, U. Bio-imprinting of lipases with fatty acids. J. Mol. Catal. B: Enzym. 2003, 22, 193–202. [Google Scholar] [CrossRef]
- Cabirol, F.L.; Tan, P.L.; Tay, B.; Cheng, S.; Hanefeld, U.; Sheldon, R.A. Linum usitatissimum Hydroxynitrile lyase cross-linked enzyme aggregates: A recyclable enantioselective catalyst. Adv. Synth. Catal. 2008, 350, 2329–2338. [Google Scholar] [CrossRef]
- De Winter, K.; Soetaert, W.; Desmet, T. An imprinted cross-linked enzyme aggregate (iCLEA) of sucrose phosphorylase: Combining improved stability with altered specificity. Int. J. Mol. Sci. 2012, 13, 11333–11342. [Google Scholar] [CrossRef] [PubMed]
- Sampath, C.; Belur, P.D.; Iyyasami, R. Enhancement of n-3 polyunsaturated fatty acid glycerides in Sardine oil by a bioimprinted cross-linked Candida rugosa lipase. Enzyme Microb. Technol. 2018, 110, 20–29. [Google Scholar] [CrossRef] [PubMed]
- Cao, L. Immobilised enzymes: Science or art? Curr. Opin. Chem. Biol. 2005, 9, 217–226. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.I.; Kim, J.; Lee, J.; Jia, H.; Na, H.B.; Youn, J.K.; Kwak, J.H.; Dohnalkova, A.; Grate, J.W.; Wang, P.; et al. Crosslinked enzyme aggregates in hierarchically-ordered mesoporous silica: A simple and effective method for enzyme stabilization. Biotechnol. Bioeng. 2007, 96, 210–218. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Qi, W.; Yu, Q.; Su, R.; He, S. Cross-linking enzyme aggregates in the macropores of silica gel: A practical and efficient method for enzyme stabilization. Biochem. Eng. J. 2010, 52, 168–174. [Google Scholar] [CrossRef]
- Cui, J.D.; Li, L.L.; Bian, H.J. Immobilization of cross-linked phenylalanine ammonia lyase aggregates in microporous silica gel. PLoS ONE 2013, 8, e80581. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Shi, L.; Huang, Y.; Gao, J.; Zhang, X.; Zhou, L. Preparation of robust biocatalyst based on cross-linked enzyme aggregates entrapped in three-dimensionally ordered macroporous silica. ACS Appl. Mater. Interfaces 2014, 6, 2622–2628. [Google Scholar] [CrossRef] [PubMed]
- Talekar, S.; Ghodake, V.; Ghotage, T.; Rathod, P.; Deshmukh, P.; Nadar, S.; Mulla, M.; Ladole, M. Novel magnetic cross-linked enzyme aggregates (magnetic CLEAs) of alpha amylase. Bioresour. Technol. 2012, 123, 542–547. [Google Scholar] [CrossRef] [PubMed]
- Sekhon, S.S.; Ahn, J.-Y.; Shin, W.-R.; Ko, J.H.; Lee, L.; Dawes, M.; Tyler, J.; Han, J.; Kim, S.Y.; Min, J.; Kim, Y.-H. Immobilization of cross-linked cutinase aggregates onto magnetic beads for degradation of polycaprolactone. J. Nanosci. Nanotechnol. 2017, 17, 9306–9311. [Google Scholar] [CrossRef]
- Liu, Y.; Guo, C.; Liu, C.-Z. Novel magnetic cross-linked lipase aggregates for improving the resolution of (R, S)-2-octanol. Chirality 2015, 27, 199–204. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.-Y.; Yang, Z. Cross-linked tyrosinase aggregates for elimination of phenolic compounds from wastewater. Chemosphere 2013, 92, 391–398. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, L.T.; Lau, Y.S.; Yang, K.L. Entrapment of cross-linked cellulase colloids in alginate beads for hydrolysis of cellulose. Colloids Surf. B Biointerfaces. 2016, 145, 862–869. [Google Scholar] [CrossRef] [PubMed]
- Mateo, C.; Chmura, A.; Rustler, S.; van Rantwijk, F.; Stolz, A.; Sheldon, R.A. Synthesis of enantiomerically pure (S)-mandelic acid using an oxynitrilase-nitrilase bienzymatic cascade: A nitrilase surprisingly shows nitrile hydratase activity. Tetrahedron Asymm. 2006, 17, 320–323. [Google Scholar] [CrossRef]
- Talekar, S.; Pandharbale, A.; Ladole, M.; Nadar, S.; Mulla, M.; Japhalekar, K.; Pattankude, K.; Arage, D. Carrier free coimmobilization of alpha amylase, glucoamylase and pullulanase as combined cross-linked enzyme aggregates (combi-CLEAs): A tri-enzyme biocatalyst with one pot starch hydrolytic activity. Bioresour. Technol. 2013, 147, 269–275. [Google Scholar] [CrossRef] [PubMed]
- Periyasamy, K.; Santhalembi, L.; Mortha, G.; Aurousseau, M.; Subramanian, S. Carrier-free coimmobilization of xylanase, cellulase and β-1,3-glucanase as combined crosslinked enzyme aggregates (combi-CLEAs) for one-pot saccharification of sugarcane bagasse. RSC Adv. 2016, 6, 32849–32857. [Google Scholar] [CrossRef]
- Ning, C.; Su, E.; Tian, Y.; Wei, D. Combined cross-linked enzyme aggregates (combi-CLEAs) for efficient integration of a ketoreductase and a cofactor regeneration system. J. Biotechnol. 2014, 184, 7–10. [Google Scholar] [CrossRef] [PubMed]
- Su, E.; Meng, Y.; Ning, C.; Ma, X.; Deng, S. Magnetic combined cross-linked enzyme aggregates (combi-CLEAs) for cofactor regeneration in the synthesis of chiral alcohol. J. Biotechnol. 2018, 271, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, L.T.; Yang, K.L. Combined cross-linked enzyme aggregates of horseradish peroxidase and glucose oxidase for catalyzing cascade chemical reactions. Enzyme Microb. Technol. 2017, 100, 52–59. [Google Scholar] [CrossRef] [PubMed]
- Jung, D.-H.; Jung, J.-H.; Seo, D.-H.; Ha, S.-J.; Kweon, D.-K.; Park, C.-S. One-pot bioconversion of sucrose to trehalose using enzymatic sequential reactions in combined cross-linked enzyme aggregates. Bioresour. Technol. 2013, 130, 801–804. [Google Scholar] [CrossRef] [PubMed]
- Scism, R.A.; Bachmann, B.O. Five-component cascade synthesis of nucleotide analogues in an engineered self-immobilized enzyme aggregate. ChemBioChem 2010, 11, 67–70. [Google Scholar] [CrossRef] [PubMed]
- Mahmod, S.S.; Yusof, F.; Jami, M.S.; Khanahmadi, S.; Shah, H. Development of an immobilized biocatalyst with lipase and protease activities as a multipurpose cross-linked enzyme aggregate (multi-CLEA). Process Biochem. 2015, 50, 2144–2157. [Google Scholar] [CrossRef]
- Chen, Z.; Wang, Y.; Liu, W.; Wang, J.; Chen, H. A novel cross-linked enzyme aggregates (CLEAs) of papain and neutrase-production, partial characterization and application. Int. J. Biol. Macromol. 2017, 95, 650–657. [Google Scholar] [CrossRef] [PubMed]
- Hickey, A.M.; Ngamsom, B.; Wiles, C.; Greenway, G.M.; Watts, P.; Littlechild, J.A. A microreactor for the study of biotransformations by a cross-linked γ-lactamase enzyme. Biotechnol. J. 2009, 4, 510–516. [Google Scholar] [CrossRef] [PubMed]
- Lanfranchi, E.; Grill, B.; Raghoebar, Z.; Van Pelt, S.; Sheldon, R.A.; Steiner, K.; Glieder, A.; Winkler, M. Production of hydroxynitrile lyase from D. tyermannii (DtHNL) in Komagataella phaffii and its immobilization as a CLEA to generate a robust biocatalyst. ChemBioChem 2018, 19, 312–316. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.-Y.; Chen, J.-Y.; Yang, Z. Use of cross-linked tyrosinase aggregates as catalyst for synthesis of L-DOPA. Biochem. Eng. J. 2012, 63, 88–94. [Google Scholar] [CrossRef]
- Nuijens, T.; Cusan, C.; Schepers, A.C.H.M.; Kruijtzer, J.A.W.; Quaedflieg, P.J.L.M. Enzymatic synthesis of activated esters and their subsequent use in enzyme-based peptide synthesis. J. Mol. Catal. B: Enzym. 2011, 71, 79–84. [Google Scholar] [CrossRef]
- Stavila, E.; Alberda van Ekenstein, G.R.O.; Loos, K. Enzyme-catalyzed synthesis of aliphatic–aromatic oligoamides. Biomacromolecules 2013, 14, 1600–1606. [Google Scholar] [CrossRef] [PubMed]
- Talekar, S.; Joshi, A.; Chougle, R.; Nakhe, A.; Bhojwani, R. Immobilized enzyme mediated synthesis of silver nanoparticles using cross-linked enzyme aggregates (CLEAs) of NADH-dependent nitrate reductase. Nano-Struct. Nano-Objects. 2016, 6, 23–33. [Google Scholar] [CrossRef]
- Forootanfar, H.; Faramarzi, M.A. Insights into laccase producing organisms, fermentation states, purification strategies, and biotechnological applications. Biotechnol. Prog. 2015, 31, 1443–1463. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.V.; Sivanesan, S.; Cabana, H. Magnetic cross-linked laccase aggregates—Bioremediation tool for decolorization of distinct classes of recalcitrant dyes. Sci. Total Environ. 2014, 487, 830–839. [Google Scholar] [CrossRef] [PubMed]
- Bilal, M.; Iqbal, H.M.N.; Hu, H.; Wang, W.; Zhang, X. Development of horseradish peroxidase-based cross-linked enzyme aggregates and their environmental exploitation for bioremediation purposes. J. Environ. Manag. 2017, 188, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, L.T.; Seow, N.; Yang, K.-L. Hollow cross-linked enzyme aggregates (h-CLEA) of laccase with high uniformity and activity. Colloids Surf. B Biointerfaces. 2017, 151, 88–94. [Google Scholar] [CrossRef] [PubMed]
- Special Report on Environmental Endocrine Disruption: An Effects Assessment and Analysis. 1997. Available online: https://archive.epa.gov/raf/web/html/special-rpt-endocrine-disruption.html (accessed on 8 February 2018).
- Taboada-Puig, R.; Junghanns, C.; Demarche, P.; Moreira, M.T.; Feijoo, G.; Lema, J.M.; Agathos, S.N. Combined cross-linked enzyme aggregates from versatile peroxidase and glucose oxidase: Production, partial characterization and application for the elimination of endocrine disruptors. Bioresour. Technol. 2011, 102, 6593–6599. [Google Scholar] [CrossRef] [PubMed]
- Hero, J.S.; Romero, C.M.; Pisa, J.H.; Perotti, N.I.; Olivaro, C.; Martinez, M.A. Designing cross-linked xylanase aggregates for bioconversion of agroindustrial waste biomass towards potential production of nutraceuticals. Int. J. Biol. Macromol. 2018, 111, 229–236. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, A.; Pletschke, B.I. Magnetic cross-linked enzyme aggregates (CLEAs): A novel concepttowards carrier free immobilization of lignocellulolytic enzymes. Enzyme Microb. Technol. 2014, 61–62, 17–27. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yamaguchi, H.; Kiyota, Y.; Miyazaki, M. Techniques for Preparation of Cross-Linked Enzyme Aggregates and Their Applications in Bioconversions. Catalysts 2018, 8, 174. https://doi.org/10.3390/catal8050174
Yamaguchi H, Kiyota Y, Miyazaki M. Techniques for Preparation of Cross-Linked Enzyme Aggregates and Their Applications in Bioconversions. Catalysts. 2018; 8(5):174. https://doi.org/10.3390/catal8050174
Chicago/Turabian StyleYamaguchi, Hiroshi, Yuhei Kiyota, and Masaya Miyazaki. 2018. "Techniques for Preparation of Cross-Linked Enzyme Aggregates and Their Applications in Bioconversions" Catalysts 8, no. 5: 174. https://doi.org/10.3390/catal8050174
APA StyleYamaguchi, H., Kiyota, Y., & Miyazaki, M. (2018). Techniques for Preparation of Cross-Linked Enzyme Aggregates and Their Applications in Bioconversions. Catalysts, 8(5), 174. https://doi.org/10.3390/catal8050174




