Sustainable Carbon Dioxide Photoreduction by a Cooperative Effect of Reactor Design and Titania Metal Promotion
Abstract
1. Introduction
2. Results
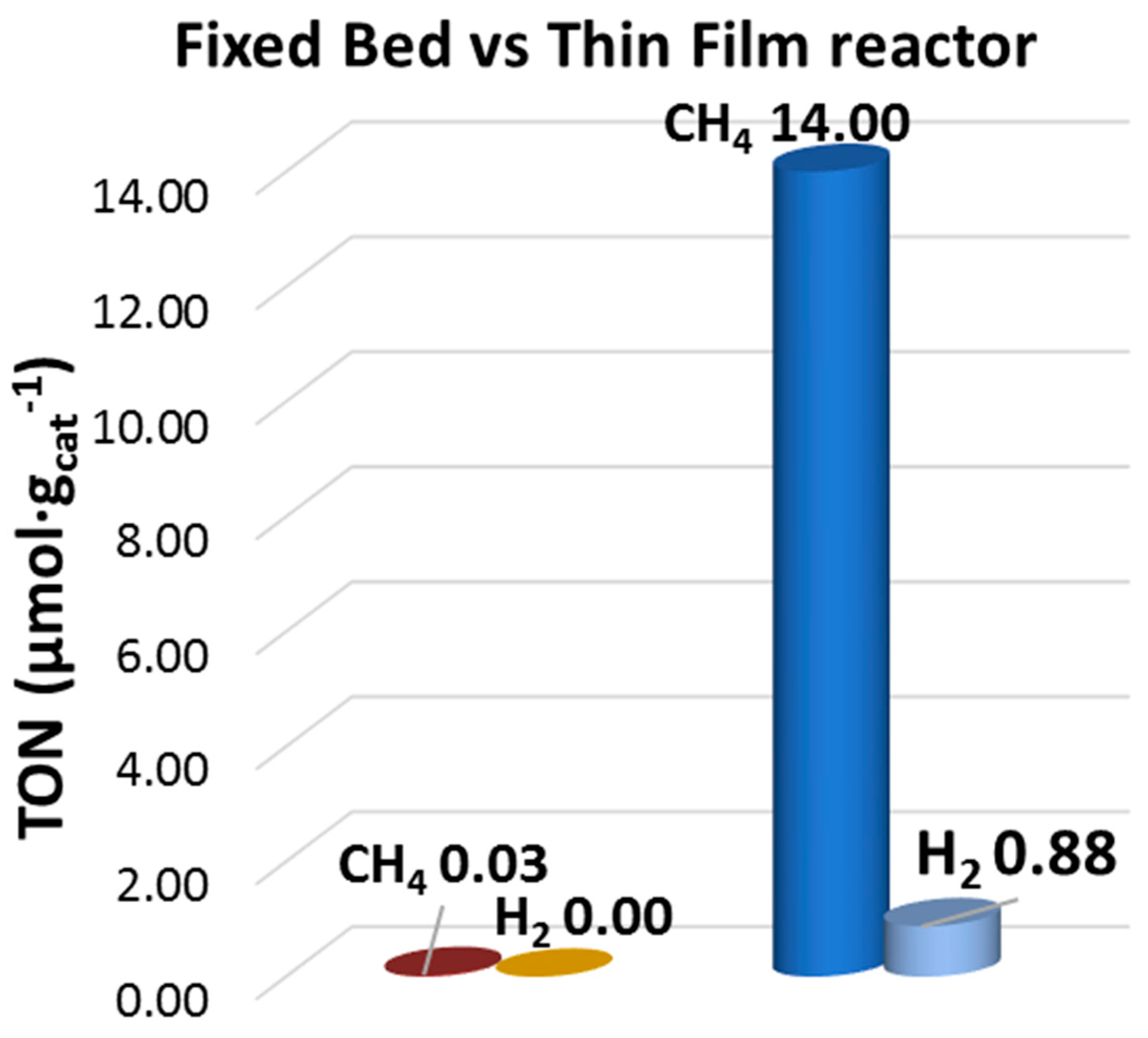
2.1. On the Reactor Design
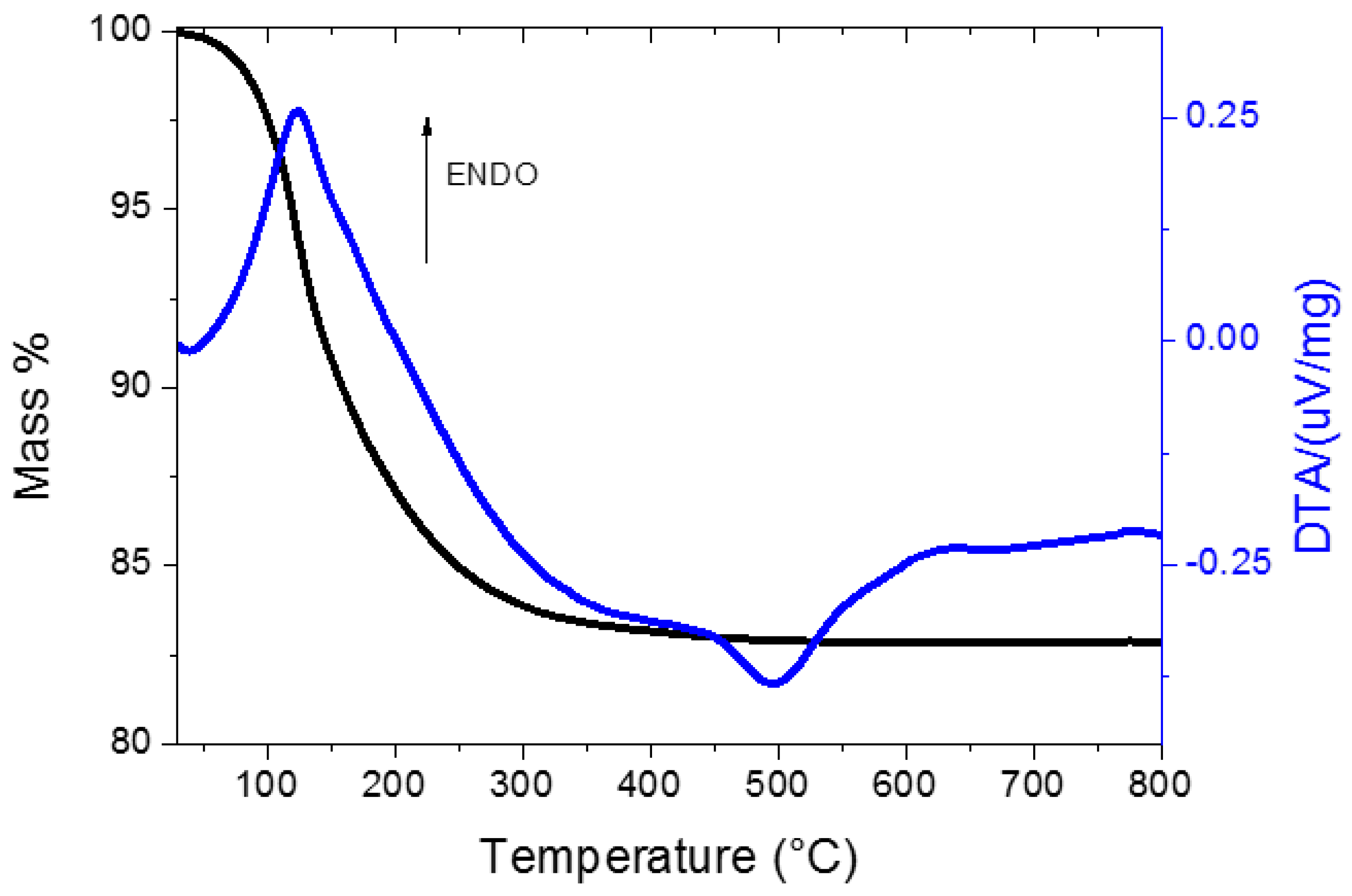
2.2. Characterisation of the Unpromoted TiO2 Photocatalyst
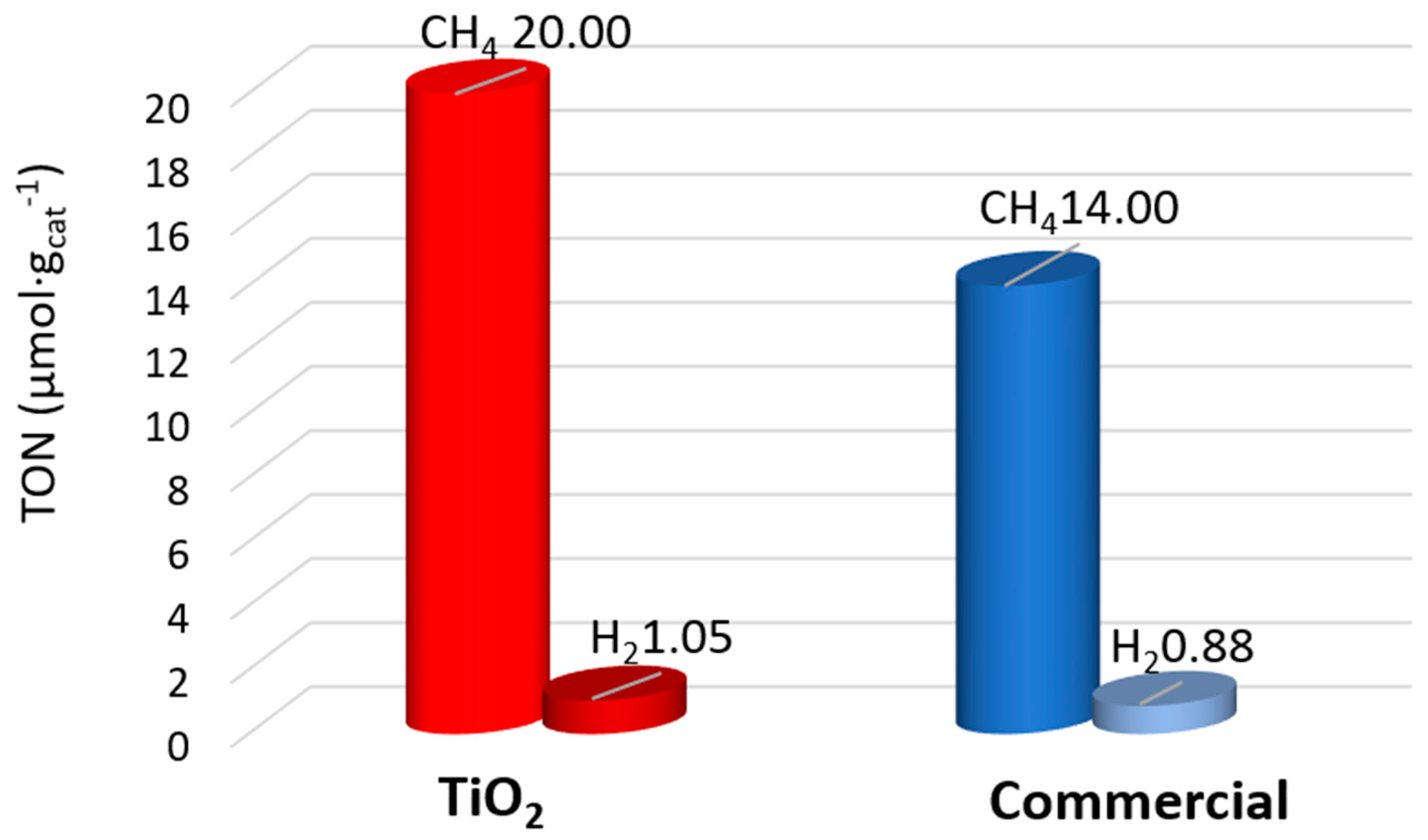
2.3. Effect of the Promoters on the Properties and Activity of the TiO2 Photocatalysts
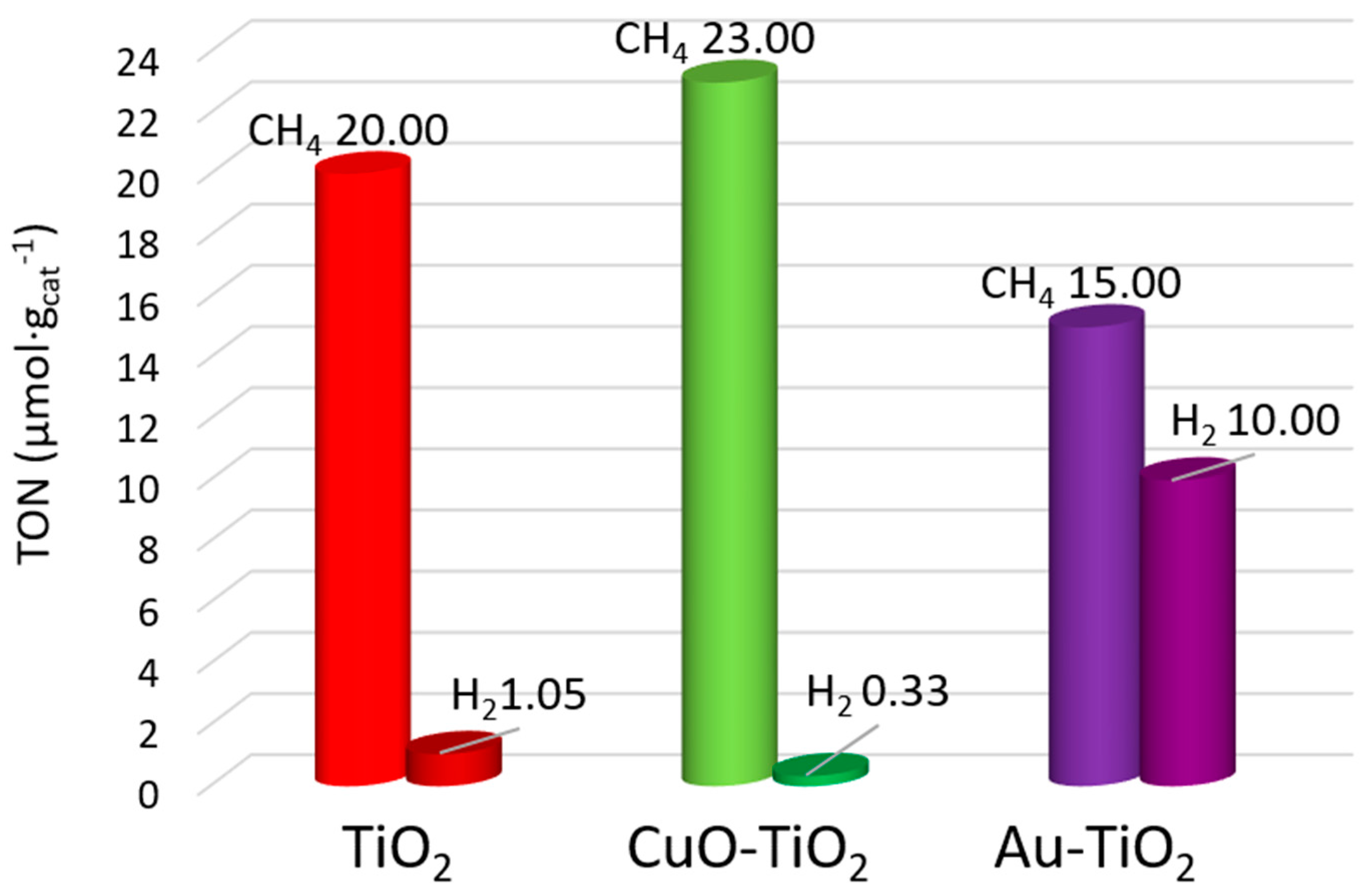
2.4. Influence of the Surface and Electronic Properties on the Photoactivity of CuO-TiO2 and Au-TiO2
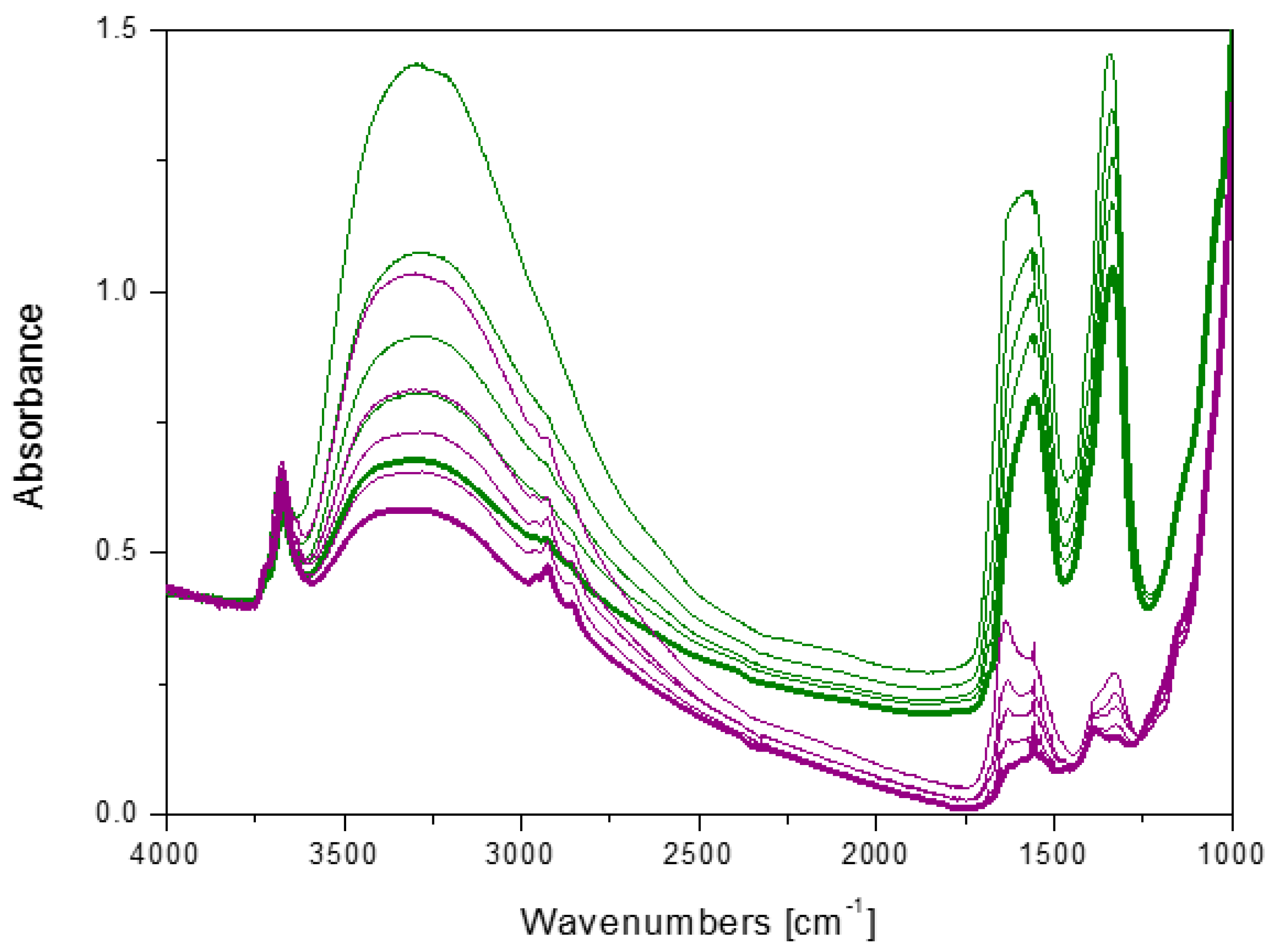
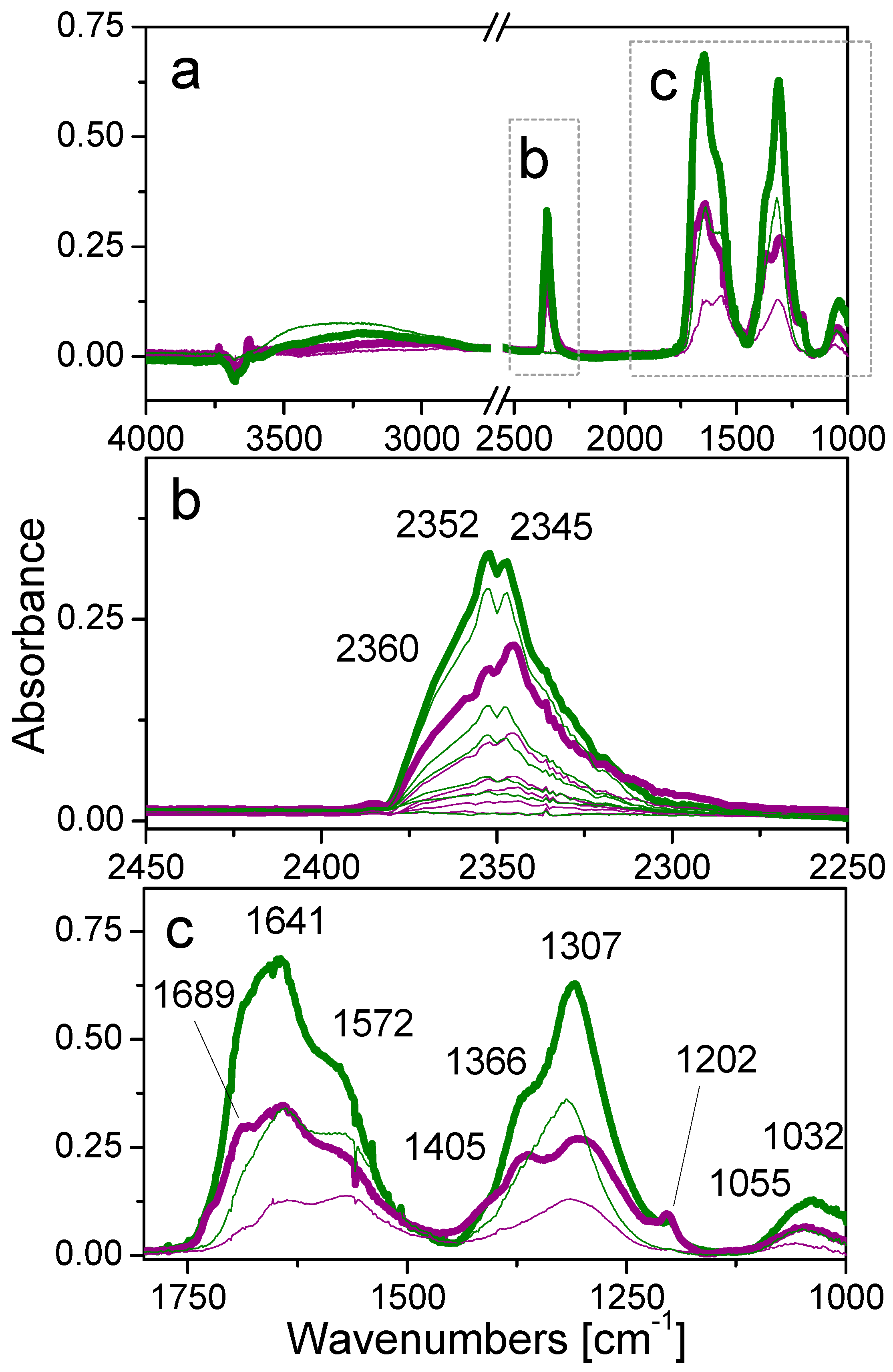
2.5. Interaction with CO2 at Room Temperature: Surface Reactivity
3. Materials and Methods
3.1. Materials
3.2. Synthesis of the Catalysts
3.2.1. Titania Synthesis
3.2.2. Copper Oxide Loading to Titania
3.2.3. Gold Introduction into Titania
3.3. Characterization of the Photocatalysts
3.4. Photoactivity Tests
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wang, Q.; Wu, S.D.; Zeng, Y.E.; Wu, B.W. Exploring the relationship between urbanization, energy consumption, and CO2 emissions in different provinces of China. Renew. Sustain. Energy Rev. 2016, 54, 1563–1579. [Google Scholar] [CrossRef]
- Höök, M.; Tang, M.X. Depletion of fossil fuels and anthropogenic climate change: A review. Energy Policy 2013, 52, 797–809. [Google Scholar] [CrossRef]
- Dai, H.; Xie, X.; Xie, Y.; Liu, J.; Masui, T. Green growth: The economic impacts of large-scale renewable energy development in China. Appl. Energy 2016, 162, 435–449. [Google Scholar] [CrossRef]
- Framework Convention on Climate Change (FCCC). Paris Agreement; Document FCCC/CP/2015/L.9/Rev.1; FCCC: Paris, France, 2015. [Google Scholar]
- Aresta, M.; Dibenedetto, A.; Angelini, A. The changing paradigm in CO2 utilization. J. CO2 Util. 2013, 3, 65–73. [Google Scholar] [CrossRef]
- Sakakura, T.; Choi, J.-C.; Yasuda, H. Transformation of Carbon Dioxide. Chem. Rev. 2007, 107, 2365–2387. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.; Varghese, O.; Paulose, M.; Grimes, C. Toward Solar Fuels: Photocatalytic Conversion of Carbon Dioxide to Hydrocarbons. ACS Nano 2010, 3, 1259–1278. [Google Scholar] [CrossRef] [PubMed]
- Ola, O.; Maroto-Valer, M. Review of material design and reactor engineering on TiO2 photocatalysis for CO2 reduction. J. Photochem. Photobiol. C 2015, 24, 16–42. [Google Scholar] [CrossRef]
- Tahir, M.; Amin, N. Recycling of carbon dioxide to renewable fuels by photocatalysis: Prospects and challenges. Renew. Sustain. Energy Rev. 2013, 25, 560–579. [Google Scholar] [CrossRef]
- Akhter, P.; Hussain, M.; Saracco, G.; Russo, N. Novel nanostructured-TiO2 materials for the photocatalytic reduction of CO2 greenhouse gas to hydrocarbons and syngas. Fuel 2015, 149, 55–65. [Google Scholar] [CrossRef]
- Neatu, S.; Maciá-Agulló, J.; Garcia, H. Solar Light Photocatalytic CO2 Reduction: General Considerations and Selected Bench-Mark Photocatalysts. Int. J. Mol. Sci. 2014, 15, 5246–5262. [Google Scholar] [CrossRef] [PubMed]
- Marszewski, M.; Cao, S.; Yu, J.; Jaronec, M. Semiconductor-based photocatalytic CO2 conversion. Mater. Horiz. 2015, 2, 261–278. [Google Scholar] [CrossRef]
- Centi, G.; Perathoner, S. Opportunities and prospects in the chemical recycling of carbon dioxide to fuels. Catal. Today 2009, 148, 191–205. [Google Scholar] [CrossRef]
- Izumi, Y. Recent advances in the photocatalytic conversion of carbon dioxide to fuels with water and/or hydrogen using solar energy and beyond. Coord. Chem. Rev. 2013, 257, 171–186. [Google Scholar] [CrossRef]
- Sastre, F.; Puga, A.; Liu, L.; Corma, A.; Garcia, H. Complete Photocatalytic Reduction of CO2 to Methane by H2 under Solar Light Irradiation. J. Am. Chem. Soc. 2014, 136, 6798–6801. [Google Scholar] [CrossRef] [PubMed]
- Olivo, A.; Trevisan, V.; Ghedini, E.; Pinna, F.; Bianchi, C.L.; Naldoni, A.; Cruciani, G.; Signoretto, M. CO2 photoreduction with water: Catalyst and process investigation. J. CO2 Util. 2015, 12, 86–94. [Google Scholar] [CrossRef]
- Peng, Y.; Yeh, Y.; Shah, S.; Huang, C.P. Concurrent photoelectrochemical reduction of CO2 and oxidation of methyl orange using nitrogen-doped TiO2. Appl. Catal. B 2012, 123, 414–423. [Google Scholar] [CrossRef]
- Lee, W.S.; Liao, C.H.; Tsai, M.F.; Huang, C.W.; Wu, J.C.S. A novel twin reactor for CO2 photoreduction to mimic artificial photosynthesis. Appl. Catal. B 2013, 132–133, 445–451. [Google Scholar] [CrossRef]
- De Richter, R.; Ming, T.; Caillol, S. Fighting global warming by photocatalytic reduction of CO2 using giant photocatalytic reactors. Renew. Sustain. Energy Rev. 2013, 19, 82–106. [Google Scholar] [CrossRef]
- Ni, M.; Leung, M.; Leung, D.; Sumathy, K.A. review and recent developments in photocatalytic water-splitting using TiO2 for hydrogen production. Renew. Sustain. Energy Rev. 2007, 11, 401–425. [Google Scholar] [CrossRef]
- Graves, C.; Ebbsen, S.; Morgensen, M.; Lackner, K.S. Sustainable hydrocarbon fuels by recycling CO2 and H2O with renewable or nuclear energy. Renew. Sustain. Energy Rev. 2011, 15, 1–23. [Google Scholar] [CrossRef]
- Arawaka, H.; Aresta, M.; Armor, J.N.; Barteau, M.A.; Beckman, E.J.; Bell, A.T.; Bercaw, J.E.; Creutz, C.; Dinjus, E.; Dixon, D.; et al. Catalysis Research of Relevance to Carbon Management: Progress, Challenges, and Opportunities. Chem. Rev. 2001, 101, 953–996. [Google Scholar] [CrossRef]
- Dhakshinamoorthy, A.; Navalon, S.; Corma, A.; Garcia, H. Photocatalytic CO2 reduction by TiO2 and related titanium containing solids. Energy Environ. Sci. 2011, 5, 9217–9233. [Google Scholar] [CrossRef]
- Liu, G.; Hoivik, N.; Wang, K.; Jakobsen, H. Engineering TiO2 nanomaterials for CO2 conversion/solar fuels. Sol. Energy Mater. Sol. Cells 2012, 105, 53–68. [Google Scholar] [CrossRef]
- Indrakanti, V.P.; Kubicki, J.D.; Schobert, H.H. Photoinduced activation of CO2 on Ti-based heterogeneous catalysts: Current state, chemical physics-based insights and outlook. Energy Environ. Sci. 2009, 2, 745–758. [Google Scholar] [CrossRef]
- Carp, O.; Hiusman, C.L.; Reller, A. Photoinduced reactivity of titanium dioxide. Prog. Solid State Chem. 2004, 32, 33–177. [Google Scholar] [CrossRef]
- Gupta, S.M.; Tripathi, M. A review of TiO2 nanoparticles. Chin. Sci. Bull. 2011, 56, 1639–1657. [Google Scholar] [CrossRef]
- Inoue, T.; Fujishima, A.; Konishi, K.; Honda, K. Photoelectrocatalytic reduction of carbon dioxide in aqueous suspensions of semiconductor powders. Nature 1979, 277, 637–638. [Google Scholar] [CrossRef]
- Karamian, E.; Sharifnia, S. On the general mechanism of photocatalytic reduction of CO2. J. CO2 Util. 2016, 16, 194–203. [Google Scholar] [CrossRef]
- Do, J.Y.; Im, Y.; Kwak, B.S.; Kim, J.-Y.; Kang, M. Dramatic CO2 photoreduction with H2O vapors for CH4 production using the TiO2 (bottom)/Fe–TiO2 (top) double-layered films. Chem. Eng. J. 2015, 275, 288–297. [Google Scholar] [CrossRef]
- Nikokavoura, A.; Trapalis, C. Alternative photocatalysts to TiO2 for the photocatalytic reduction of CO2. Appl. Surf. Sci. 2017, 391, 149–174. [Google Scholar] [CrossRef]
- Vereb, G.; Manczinger, L.; Bozso, G.; Sienkiewicz, A.; Forro, L.; Mogyorosi, K.; Hernadi, K.; Dombi, A. Comparison of the photocatalytic efficiencies of bare and doped rutile and anatase TiO2 photocatalysts under visible light for phenol degradation and E. coli inactivation. Appl. Catal. B 2013, 129, 566–574. [Google Scholar] [CrossRef]
- Marschall, R.; Wang, L. Non-metal doping of transition metal oxides for visible-light photocatalysis. Catal. Today 2014, 225, 111–135. [Google Scholar] [CrossRef]
- Pelaez, M.; Nolan, N.T.; Pillai, S.C.; Seery, M.K.; Falaras, P.; Kontos, A.G.; Dunlop, P.S.M.; Hamilton, J.W.J.; Byrne, J.A.; O’Shea, K.; et al. A review on the visible light active titanium dioxide photocatalysts for environmental applications. Appl. Catal. B 2012, 125, 331–349. [Google Scholar] [CrossRef]
- Xu, H.; Ouyang, S.; Liu, L.; Umezawa, N.; Ye, J. Porous-structured Cu2O/TiO2 nanojunction material toward efficient CO2 photoreduction. J. Mater. Chem. A 2014, 2, 12462–12661. [Google Scholar] [CrossRef] [PubMed]
- Sun, T.; Liu, E.; Fan, J.; Hu, X.; Wu, F.; Hou, W. High photocatalytic activity of hydrogen production from water over Fe doped and Ag deposited anatase TiO2 catalyst synthesized by solvothermal method. Chem. Eng. J. 2013, 228, 896–906. [Google Scholar] [CrossRef]
- Linsebigler, A.; Lu, G.; Yates, J. Photocatalysis on TiO2 Surfaces: Principles, Mechanisms, and Selected Results. Chem. Rev. 1995, 95, 735–758. [Google Scholar] [CrossRef]
- Janus, M.; Inagaki, M.; Tryba, B.; Toyoda, M.; Morawski, A.W. New preparation of a carbon-TiO2 photocatalyst by carbonization of n-hexane deposited on TiO2. Appl. Catal. B 2006, 63, 272–276. [Google Scholar] [CrossRef]
- Kang, I.; Zhang, Q.; Yin, S.; Sato, T.; Saito, F. Preparation of a visible sensitive carbon doped TiO2 photo-catalyst by grinding TiO2 with ethanol and heating treatment. Appl. Catal. B 2008, 82, 81–87. [Google Scholar] [CrossRef]
- Das, S.; Wan Daud, W. Photocatalytic CO2 transformation into fuel: A review on advances in photocatalyst and photoreactor. Renew. Sustain. Energy Rev. 2014, 39, 765–805. [Google Scholar] [CrossRef]
- Diebold, U. The surface science of titanium dioxide. Surf. Sci. Rep. 2003, 48, 53–229. [Google Scholar] [CrossRef]
- Li, K.; An, X.; Park, K.H.; Khraisheh, M.; Tang, J. A critical review of CO2 photoconversion: Catalysts and reactors. Catal. Today 2014, 224, 3–12. [Google Scholar] [CrossRef]
- Meng, X.; Ouyang, S.; Kako, T.; Li, P.; Yu, Q.; Wang, T.; Ye, J. Photocatalytic CO2 conversion over alkali modified TiO2 without loading noble metal cocatalyst. Chem. Commun. 2014, 50, 11517–11519. [Google Scholar] [CrossRef] [PubMed]
- Ohtani, B. Photocatalysis by inorganic solid materials. Inorg. Photochem. 2011, 63, 395–430. [Google Scholar] [CrossRef]
- Bai, S.; Yin, W.; Wang, L.; Li, Z.; Xiong, Y. Surface and interface design in cocatalysts for photocatalytic water splitting and CO2 reduction. RCS Adv. 2016, 6, 57446–57463. [Google Scholar] [CrossRef]
- Wang, Y.; Li, B.; Zhang, C.; Cui, L.; Kang, S.; Li, X.; Zhou, L. Ordered mesoporous CeO2-TiO2 composites: Highly efficient photocatalysts for the reduction of CO2 with H2O under simulated solar irradiation. Appl. Catal. B 2013, 130, 277–284. [Google Scholar] [CrossRef]
- Malato, S.; Fernandez-Ibanez, P.; Malodato, M.I.; Blanco, J.; Gerjakm, W. Decontamination and disinfection of water by solar photocatalysis: Recent overview and trends. Catal. Today 2009, 147, 1–59. [Google Scholar] [CrossRef]
- Yuan, L.; Xu, Y.J. Photocatalytic conversion of CO2 into value-added and renewable fuels. Appl. Surf. Sci. 2015, 342, 154–167. [Google Scholar] [CrossRef]
- Liu, L.; Gao, F.; Zhao, H.; Li, Y. Tailoring Cu valence and oxygen vacancy in Cu/TiO2 catalysts for enhanced CO2 photoreduction efficiency. Appl. Catal. B 2013, 134–135, 349–358. [Google Scholar] [CrossRef]
- Qin, S.; Xin, F.; Liu, Y.; Yin, X.; Ma, W. Photocatalytic reduction of CO2 in methanol to methyl formate over CuO–TiO2 composite catalysts. J. Colloid Interface Sci. 2011, 356, 257–261. [Google Scholar] [CrossRef] [PubMed]
- Isahak, W.N.R.W.; Ramli, Z.A.C.; Ismail, M.W.; Ismail, K.; Yosup, R.M.; Hisham, M.W.M.; Yarmo, M.A. Adsorption–desorption of CO2 on different type of copper oxides surfaces: Physical and chemical attractions studies. J. CO2 Util. 2013, 2, 8–15. [Google Scholar] [CrossRef]
- Ghosh, S.K.; Pal, T. Interparticle Coupling Effect on the Surface Plasmon Resonance of Gold Nanoparticles: From Theory to Applications. Chem. Rev. 2007, 107, 4797–4862. [Google Scholar] [CrossRef] [PubMed]
- Kaur, R.; Pal, B. Size and shape dependent attachments of Au nanostructures to TiO2 for optimum reactivity of Au–TiO2 photocatalysis. J. Mol. Catal. A 2012, 355, 39–43. [Google Scholar] [CrossRef]
- Tahir, B.; Tahir, M.; Amin, N. Photocatalytic CO2 conversion over Au/TiO2 nanostructures for dynamic production of clean fuels in a monolith photoreactor. Clean Technol. Environ. Policy 2016, 18, 2147–2160. [Google Scholar] [CrossRef]
- Silva, C.; Juarez, R.; Marino, T.; Molinari, R.; Garcia, H. Influence of excitation wavelength (UV or visible light) on the photocatalytic activity of titania containing gold nanoparticles for the generation of hydrogen or oxygen from water. J. Am. Chem. Soc. 2011, 133, 595–602. [Google Scholar] [CrossRef] [PubMed]
- Primo, A.; Corma, A.; Garcìa, H. Titania supported gold nanoparticles as photocatalyst. Phys. Chem. Chem. Phys. 2011, 13, 886–910. [Google Scholar] [CrossRef] [PubMed]
- Crisan, D.; Dragan, N.; Raileanu, M.; Crisan, M.; Ianculescu, A.; Luca, D.; Nastuta, A.; Mandare, D. Structural study of sol–gel Au/TiO2 films from nanopowders. Appl. Surf. Sci. 2011, 257, 4227–4231. [Google Scholar] [CrossRef]
- Korologos, C.A.; Nikolaki, M.D.; Zerva, C.N.; Philippopoulos, C.J.; Poupoulos, S.G. Photocatalytic oxidation of benzene, toluene, ethylbenzene and m-xylene in the gas-phase over TiO2-based catalysts. J. Photochem. Photobiol. A 2012, 244, 24–31. [Google Scholar] [CrossRef]
- Daous, M.; Iliev, V.; Petrov, L. Gold-modified N-doped TiO2 and N-doped WO3/TiO2 semiconductors as photocatalysts for UV-visible light destruction of aqueous 2,4,6-trinitrotoluene solution. J. Mol. Catal. A 2014, 392, 194–201. [Google Scholar] [CrossRef]
- Fujishima, A.; Zhang, X.; Tryk, D. Heterogeneous photocatalysis: From water photolysis to applications in environmental cleanup. Int. J. Hydrogen Energy 2007, 32, 2664–2672. [Google Scholar] [CrossRef]
- Camarillo, R.; Tostòn, S.; Martìnez, F.; Jimènez, C.; Rincòn, J. Enhancing the photocatalytic reduction of CO2 through engineering of catalysts with high pressure technology: Pd/TiO2 photocatalysts. J. Supercrit. Fluids 2017, 123, 18–27. [Google Scholar] [CrossRef]
- Corma, A.; Garcia, H. Photocatalytic reduction of CO2 for fuel production: Possibilities and challenges. J. Catal. 2013, 308, 168–175. [Google Scholar] [CrossRef]
- Tahir, M.; Tahir, B.; Amin, N.; Zakaria, Z.Y. Photo-induced reduction of CO2 to CO with hydrogen over plasmonic Ag-NPs/TiO2 NWs core/shell hetero-junction under UV and visible light. J. CO2 Util. 2017, 18, 250–260. [Google Scholar] [CrossRef]
- Naldoni, A.; D’Arienzo, M.; Altomare, M.; Marelli, M.; Scotti, R.; Morazzoni, F.; Selli, E.; Del Santo, V. Pt and Au/TiO2 photocatalysts for methanol reforming: Role of metal nanoparticles in tuning charge trapping properties and photoefficiency. Appl. Catal. B 2013, 130, 239–248. [Google Scholar] [CrossRef]
- Collado, L.; Reynal, A.; Coronado, J.M.; Serrano, D.P.; Durrant, J.R.; de la Pena O’Shea, V.A. Effect of Au surface plasmon nanoparticles on the selective CO2 photoreduction to CH4. Appl. Catal. B 2015, 178, 177–185. [Google Scholar] [CrossRef]
- Lo, C.C.; Hung, C.H.; Yuan, C.S.; Wu, J.F. Parameter Effects and Reaction Pathways of Photoreduction of CO2 over TiO2/SO42− Photocatalyst. Sol. Energy Mater. Sol. Cells 2007, 91, 1765–1774. [Google Scholar] [CrossRef]
- Wang, T.; Yang, L.; Du, X.; Yang, Y. Numerical investigation on CO2 photocatalytic reduction in optical fiber monolith reactor. Energy Convers. Manag. 2013, 65, 299–307. [Google Scholar] [CrossRef]
- Guan, G.; Kida, T.; Harada, T.; Iasayama, M.; Yoshida, A. Photoreduction of carbon dioxide with water over K2Ti6O13 photocatalyst combined with Cu/ZnO catalyst under concentrated sunlight. Appl. Catal. A 2003, 249, 11–18. [Google Scholar] [CrossRef]
- Bazzo, A.; Urawaka, A. Origin of photocatalytic activity in continuous gas phase CO2 reduction over Pt/TiO2. ChemSusChem 2013, 6, 2095–2102. [Google Scholar] [CrossRef] [PubMed]
- Fang, B.; Xing, Y.; Bonakdapour, A.; Zhang, S.; Wilkinson, D.P. Hierarchical CuO–TiO2 Hollow Microspheres for Highly Efficient Photodriven Reduction of CO2 to CH4. ACS Sustain. Chem. Eng. 2015, 3, 2381–2388. [Google Scholar] [CrossRef]
- Zhao, C.; Krall, A.; Zhao, H.; Zhang, Q.; Li, Y. Ultrasonic spray pyrolysis synthesis of Ag/TiO2 nanocomposite photocatalysts for simultaneous H2 production and CO2 reduction. Int. J. Hydrogen Energy 2012, 37, 9967–9976. [Google Scholar] [CrossRef]
- Grigioni, I.; Dozzi, M.V.; Bernareggi, M.; Chiarello, G.L.; Selli, E. Photocatalytic CO2 reduction vs. H2 production: The effects of surface carbon-containing impurities on the performance of TiO2-based photocatalysts. Catal. Today 2017, 281, 214–220. [Google Scholar] [CrossRef]
- Camarillo, R.; Tostòn, S.; Martìnez, F.; Jimènez, C.; Rincòn, J. Preparation of TiO2-based catalysts with supercritical fluid technology: Characterization and photocatalytic activity in CO2 reduction. J. Chem. Technol. Biotechnol. 2017, 92, 1710–1720. [Google Scholar] [CrossRef]
- Camarillo, R.; Tostòn, S.; Martìnez, F.; Jimènez, C.; Rincòn, J. Supercritical synthesis of platinum-modified titanium dioxide for solar fuel production from carbon dioxide. Chin. J. Catal. 2017, 38, 636–650. [Google Scholar] [CrossRef]
- Tan, L.; Ong, W.; Chai, S.; Rahman, A. Photocatalytic reduction of CO2 with H2O over graphene oxide-supported oxygen-rich TiO2 hybrid photocatalyst under visible light irradiation: Process and kinetic studies. Chem. Eng. J. 2017, 308, 248–255. [Google Scholar] [CrossRef]
- Ola, O.; Maroto-Valer, M. Synthesis, characterization and visible light photocatalytic activity of metal based TiO2 monoliths for CO2 reduction. Chem. Eng. J. 2016, 283, 1244–1253. [Google Scholar] [CrossRef]
- Wu, J.C.S.; Huang, C.W. In situ DRIFTS study of photocatalytic CO2 reduction under UV irradiation. Front. Chem. Eng. China 2010, 4, 120–126. [Google Scholar] [CrossRef]
- Shibata, T.; Irie, H.; Ohmori, M.; Nakajima, A.; Watanabe, T.; Hashimoto, K. Comparison of photochemical properties of brookite and anatase TiO2 films. Phys. Chem. Chem. Phys. 2004, 6, 1359–1362. [Google Scholar] [CrossRef]
- Shelimov, B.N.; Tolkachev, N.N.; Tkachenko, O.P.; Baeve, G.N.; Klemen-tiev, K.V.; Stakheev, A.Y.; Kazansky, V.B. Enhancement effect of TiO2 dispersion over alumina on the photocatalytic removal of NOx admixtures from O2–N2 flow. J. Photochem. Photobiol. A 2008, 195, 81–88. [Google Scholar] [CrossRef]
- Olivo, A.; Ghedini, E.; Signoretto, M.; Compagnoni, M.; Rossetti, I. Liquid vs. Gas Phase CO2 Photoreduction Process: Which Is the Effect of the Reaction Medium? Energies 2017, 10, 1394. [Google Scholar] [CrossRef]
- Tahir, M.; Amin, N. Photocatalytic CO2 reduction with H2O vapors using montmorillonite/TiO2 supported microchannel monolith photoreactor. Chem. Eng. J. 2013, 230, 314–327. [Google Scholar] [CrossRef]
- Nguyen, T.; Wu, J.C.S. Photoreduction of CO2 in an optical-fiber photoreactor: Effects of metals addition and catalyst carrier. Appl. Catal. A 2008, 335, 112–120. [Google Scholar] [CrossRef]
- Menegazzo, F.; Signoretto, M.; Marchese, D.; Pinna, F.; Manzoli, M. Structure—Activity relationships of Au/ZrO2 catalysts for 5-hydroxymethylfurfural oxidative esterification: Effects of zirconia sulphation on gold dispersion, position and shape. J. Catal. 2015, 326, 1–8. [Google Scholar] [CrossRef]
- Manzoli, M.; Menegazzo, F.; Signoretto, M.; Cruciani, G.; Pinna, F. Effects of synthetic parameters on the catalytic performance of Au/CeO2 for furfural oxidative esterification. J. Catal. 2015, 330, 465–473. [Google Scholar] [CrossRef]
- Yashnik, S.; Ismagilov, Z.; Anufrienko, V. Catalytic properties and electronic structure of copper ions in Cu-ZSM-5. Catal. Today 2005, 110, 310–322. [Google Scholar] [CrossRef]
- Bravo-Suárez, J.J.; Subramaniam, B.; Chaudhari, R.V. Ultraviolet–Visible Spectroscopy and Temperature-Programmed Techniques as Tools for Structural Characterization of Cu in CuMgAlOx Mixed Metal Oxides. J. Phys. Chem. C 2012, 116, 18207–18221. [Google Scholar] [CrossRef]
- Anpo, M.; Yamashita, H.; Ichihashi, Y.; Ehara, S. Photocatalytic reduction of CO2 with H2O on various titanium oxide catalysts. J. Electroanal. Chem. 1995, 396, 21–26. [Google Scholar] [CrossRef]
- Carneiro, J.T.; Yang, C.C.; Moma, J.A.; Moulijin, J.A.; Mul, G. How gold deposition affects anatase performance in the photo-catalytic oxidation of cyclohexane. Catal. Lett. 2009, 129, 12–19. [Google Scholar] [CrossRef]
- Martra, G. Lewis acid and base sites at the surface of microcrystalline TiO2 anatase: Relationships between surface morphology and chemical behaviour. Appl. Catal. A 2000, 200, 275–285. [Google Scholar] [CrossRef]
- Morterra, C. An infrared spectroscopic study of anatase properties. Part 6—Surface hydration and strong Lewis acidity of pure and sulphate-doped preparations. J. Chem. Soc. Faraday Trans. 1 1988, 84, 1617–1637. [Google Scholar] [CrossRef]
- Cerrato, G.; Marchese, L.; Morterra, C. Structural and morphological modifications of sintering microcrystalline TiO2: An XRD, HRTEM and FTIR study. Appl. Surf. Sci. 1993, 70–71, 200–205. [Google Scholar] [CrossRef]
- Morterra, C.; Cerrato, G.; Emanuel, C. End-on surface coordinated (adsorbed) CO2: A specific ligand for surface Lewis acidic centres. Mater. Chem. Phys. 1991, 29, 447–456. [Google Scholar] [CrossRef]
- Busca, G.; Lorenzelli, V. Infrared spectroscopic identification of species arising from reactive adsorption of carbon oxides on metal oxide surfaces. Mater. Chem. 1982, 7, 89–126. [Google Scholar] [CrossRef]
- Baltrusaitis, J.; Schuttlefield, J.; Zeitler, E.; Grassian, V.H. Carbon dioxide adsorption on oxide nanoparticle surfaces. Chem. Eng. J. 2011, 170, 471–481. [Google Scholar] [CrossRef]
- Ramis, G.; Busca, G.; Lorenzelli, V. Low-temperature CO2 adsorption on metal oxides: Spectroscopic characterization of some weakly adsorbed species. Mater. Chem. Phys. 1991, 29, 425–435. [Google Scholar] [CrossRef]
- Tabatabai, M.A. A Rapid Method for Determination of Sulfate in Water Samples. Environ. Lett. 1974, 7, 237–243. [Google Scholar] [CrossRef]
- Zanella, R.; Giorgio, S.; Henry, C.R.; Louis, C. Alternative Methods for the Preparation of Gold Nanoparticles Supported on TiO2. J. Phys. Chem. B 2002, 106, 7634–7642. [Google Scholar] [CrossRef]
- Menegazzo, F.; Signoretto, M.; Pinna, F.; Manzoli, M.; Aina, V.; Cerrato, G.; Boccuzzi, F. Oxidative esterification of renewable furfural on gold-based catalysts: Which is the best support? J. Catal. 2014, 309, 241–247. [Google Scholar] [CrossRef]
- Brunauer, S.; Emmett, P.H.; Teller, E. Adsorption of Gases in Multimolecular Layers. J. Am. Chem. Soc. 1938, 60, 309–319. [Google Scholar] [CrossRef]
- Barrett, E.P.; Joyner, L.S.; Halenda, P.P. The Determination of Pore Volume and Area Distributions in Porous Substances. I. Computations from Nitrogen Isotherms. J. Am. Chem. Soc. 1951, 73, 373–380. [Google Scholar] [CrossRef]
- Kubelka, P.; Munk, F. Ein Beitrag Zur Optik Der Farbanstriche. Zeitschrift Technische Physik 1931, 12, 593–601. [Google Scholar]
- Liu, D.; Fernandez, Y.; Ola, O.; Maroto-Valer, M.; Parlett, C.M.A.; Lee, A.F.; Wu, J.C.S. On the impact of Cu dispersion on CO2 photoreduction over Cu/TiO2. Catal. Commun. 2012, 25, 78–82. [Google Scholar] [CrossRef]
- Tahir, M.; Amin, N. Photocatalytic reduction of carbon dioxide with water vapors over montmorillonite modified TiO2 nanocomposites. Appl. Catal. B 2013, 142–143, 512–522. [Google Scholar] [CrossRef]
- Braslavsky, S.E.; Braun, A.M.; Cassano, A.E.; Emeline, A.V.; Litter, M.I.; Palmisano, L.; Parmon, V.N.; Serpone, N. Glossary of terms used in photocatalysis and radiation catalysis (IUPAC Recommendations 2011). Pure Appl. Chem. 2011, 83, 931–1014. [Google Scholar] [CrossRef]
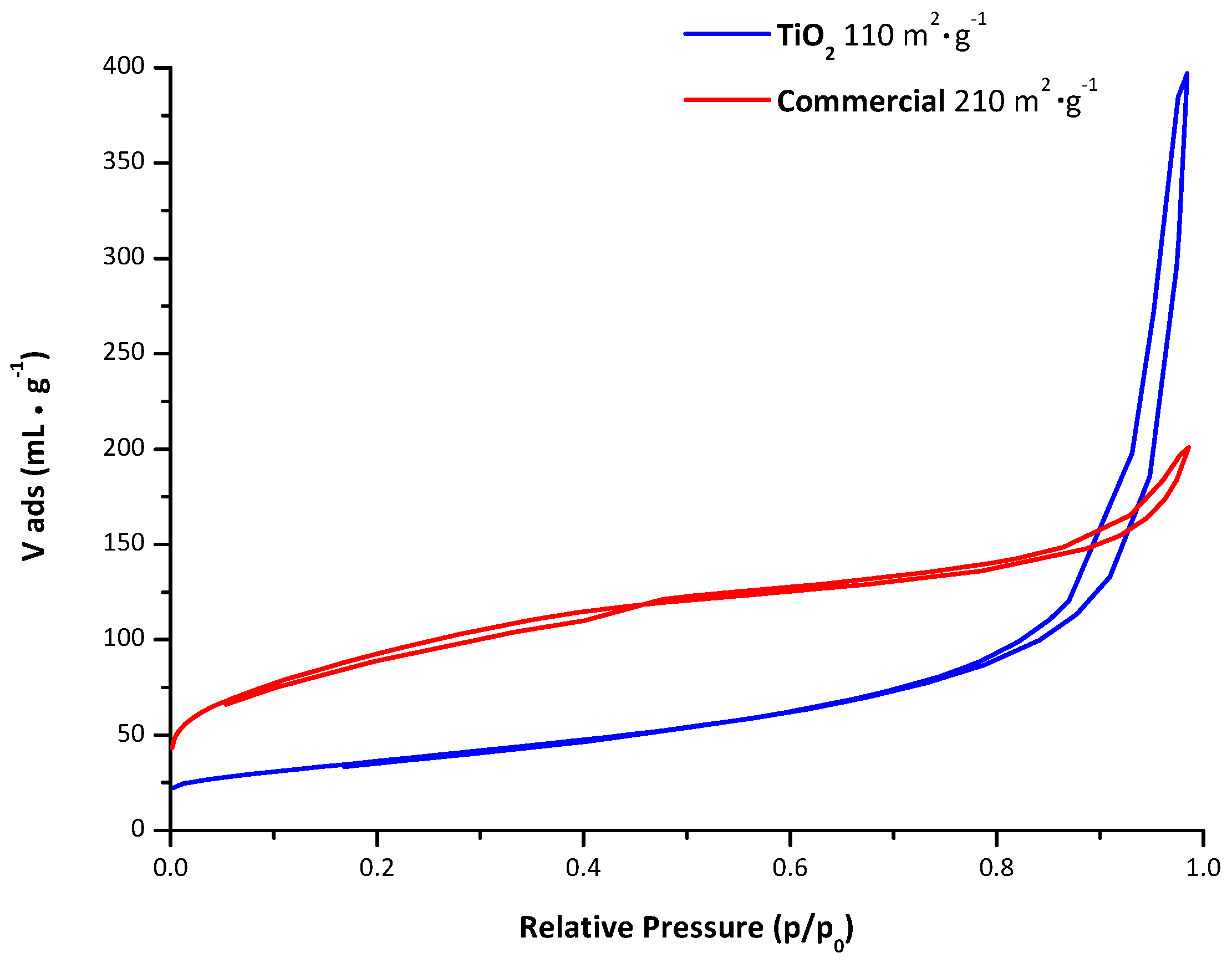
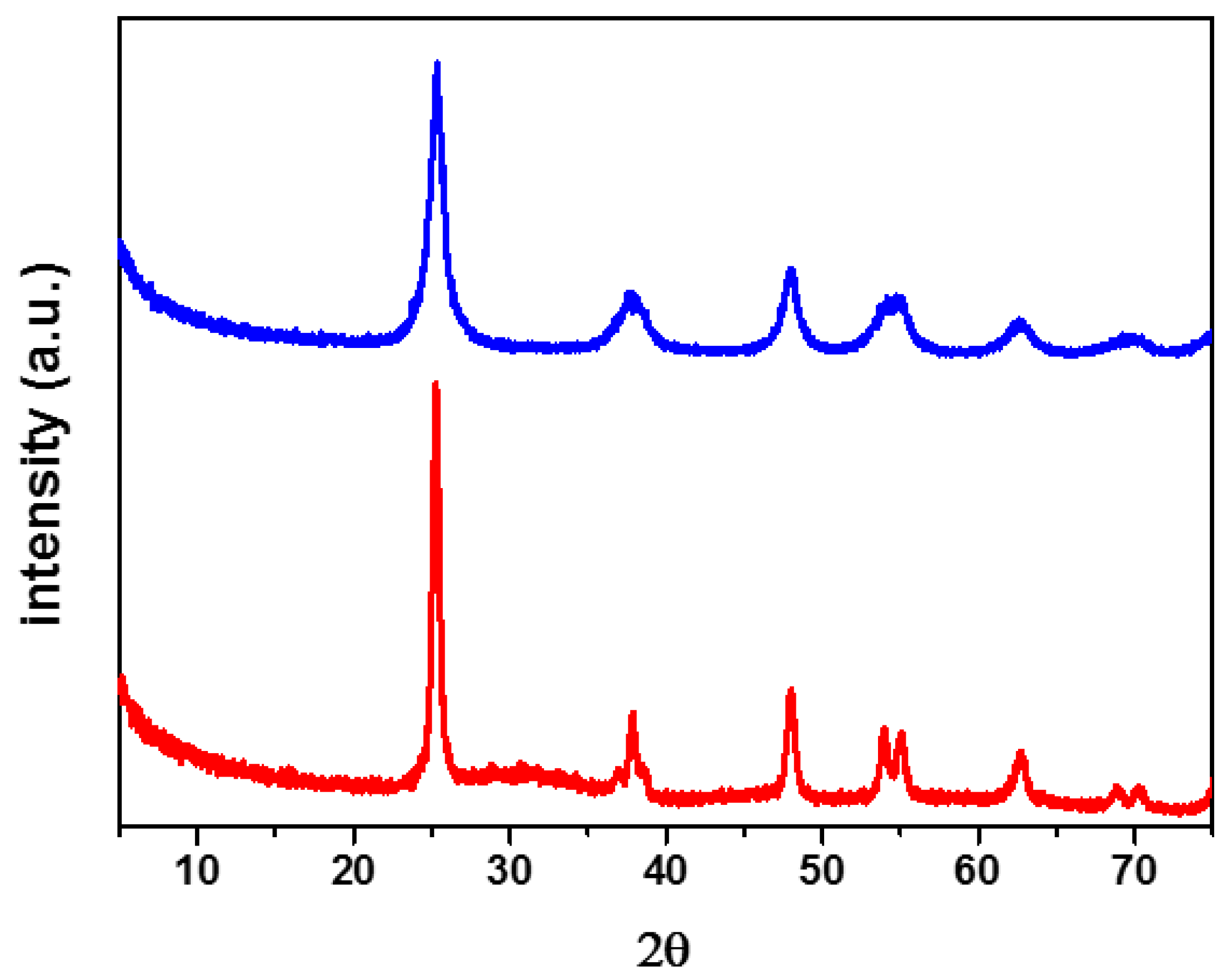
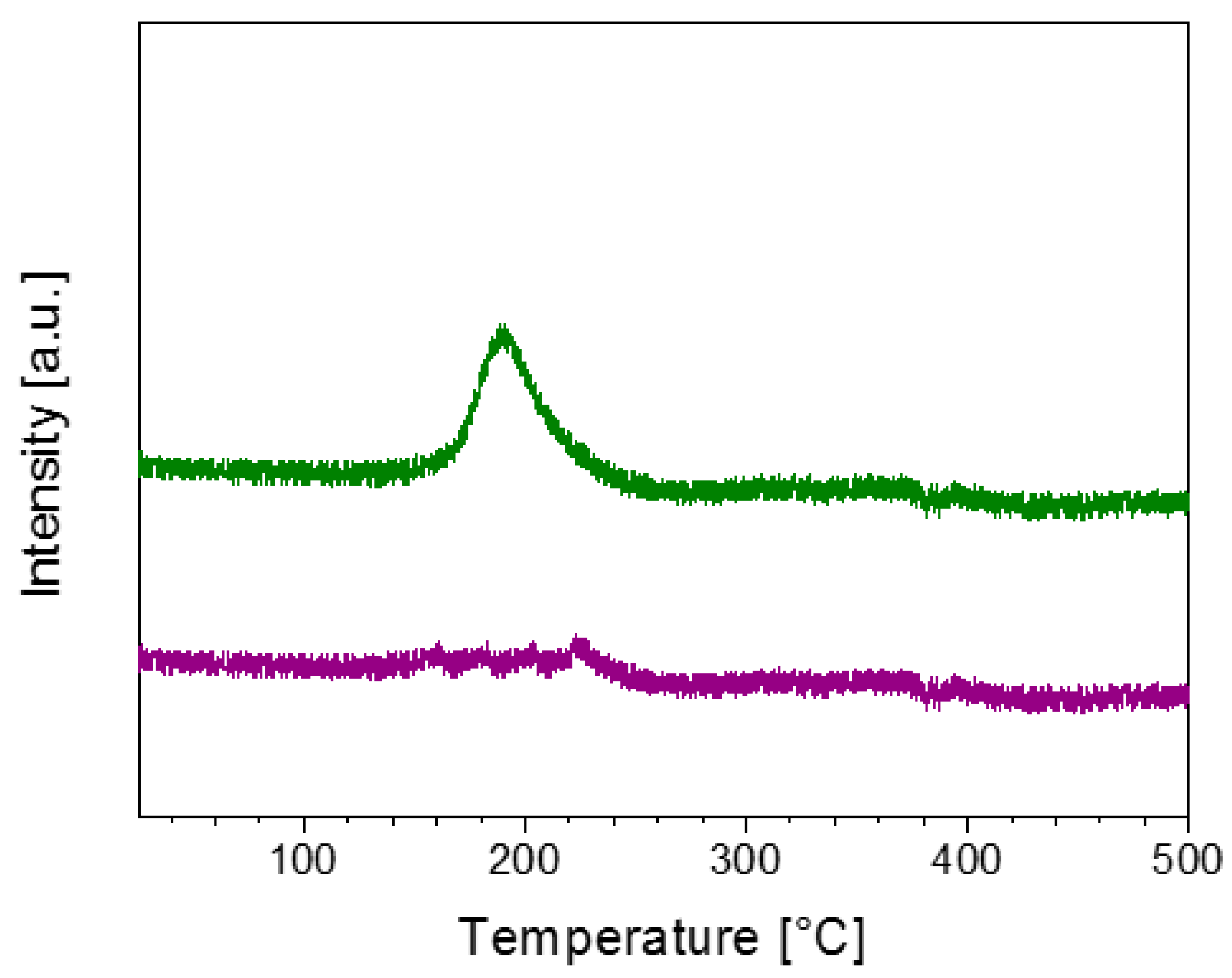
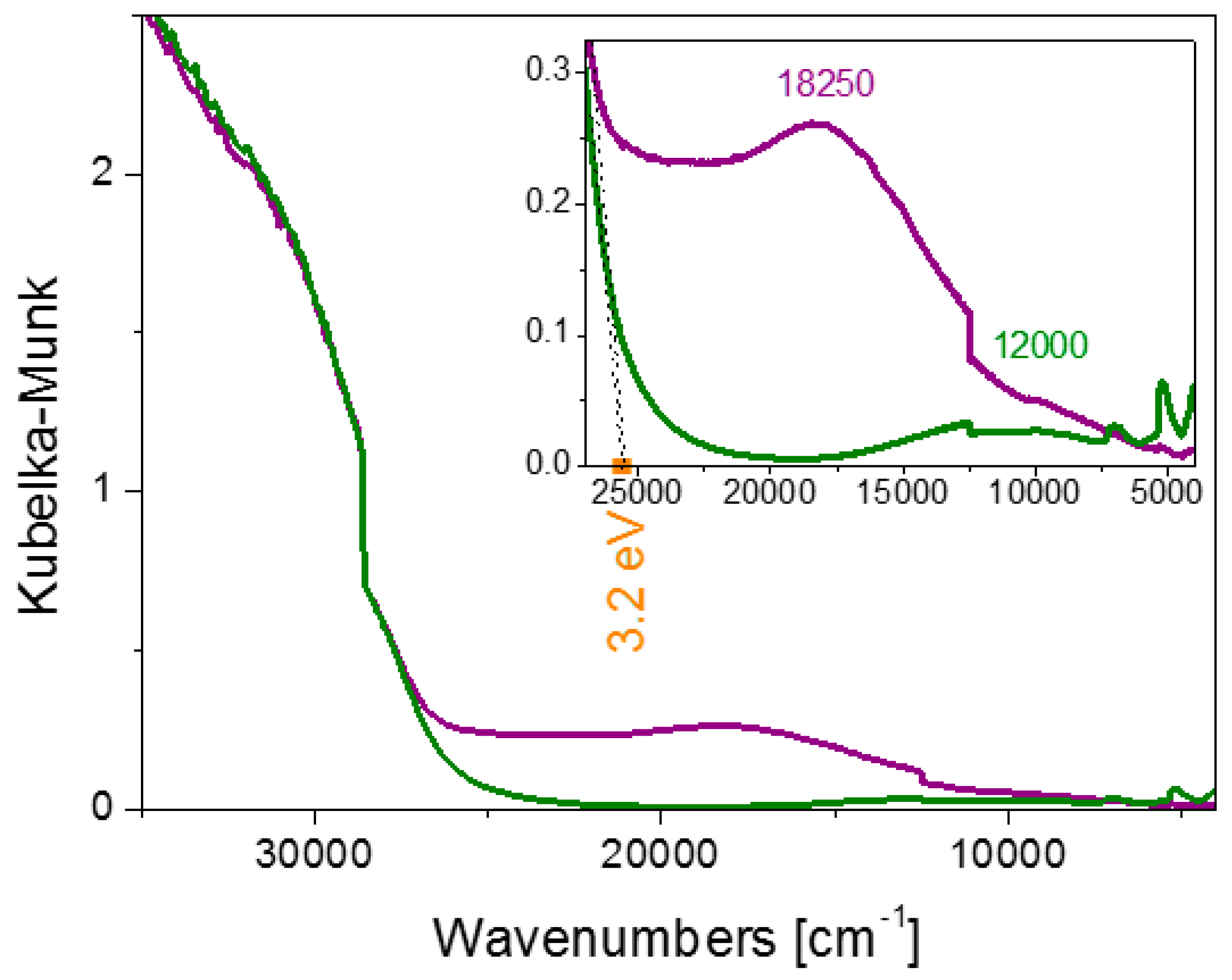

| Photocatalyst | BET Specific Surface Area (m2/g) |
|---|---|
| MIRKAT 211 | 217 |
| TiO2 | 110 |
| CuO-TiO2 | 100 |
| Au-TiO2 | 100 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olivo, A.; Ghedini, E.; Pascalicchio, P.; Manzoli, M.; Cruciani, G.; Signoretto, M. Sustainable Carbon Dioxide Photoreduction by a Cooperative Effect of Reactor Design and Titania Metal Promotion. Catalysts 2018, 8, 41. https://doi.org/10.3390/catal8010041
Olivo A, Ghedini E, Pascalicchio P, Manzoli M, Cruciani G, Signoretto M. Sustainable Carbon Dioxide Photoreduction by a Cooperative Effect of Reactor Design and Titania Metal Promotion. Catalysts. 2018; 8(1):41. https://doi.org/10.3390/catal8010041
Chicago/Turabian StyleOlivo, Alberto, Elena Ghedini, Paolina Pascalicchio, Maela Manzoli, Giuseppe Cruciani, and Michela Signoretto. 2018. "Sustainable Carbon Dioxide Photoreduction by a Cooperative Effect of Reactor Design and Titania Metal Promotion" Catalysts 8, no. 1: 41. https://doi.org/10.3390/catal8010041
APA StyleOlivo, A., Ghedini, E., Pascalicchio, P., Manzoli, M., Cruciani, G., & Signoretto, M. (2018). Sustainable Carbon Dioxide Photoreduction by a Cooperative Effect of Reactor Design and Titania Metal Promotion. Catalysts, 8(1), 41. https://doi.org/10.3390/catal8010041