Preparation and Performance of Modified Red Mud-Based Catalysts for Selective Catalytic Reduction of NOx with NH3
Abstract
:1. Introduction
2. Results
2.1. Effect of Dealkalization Method
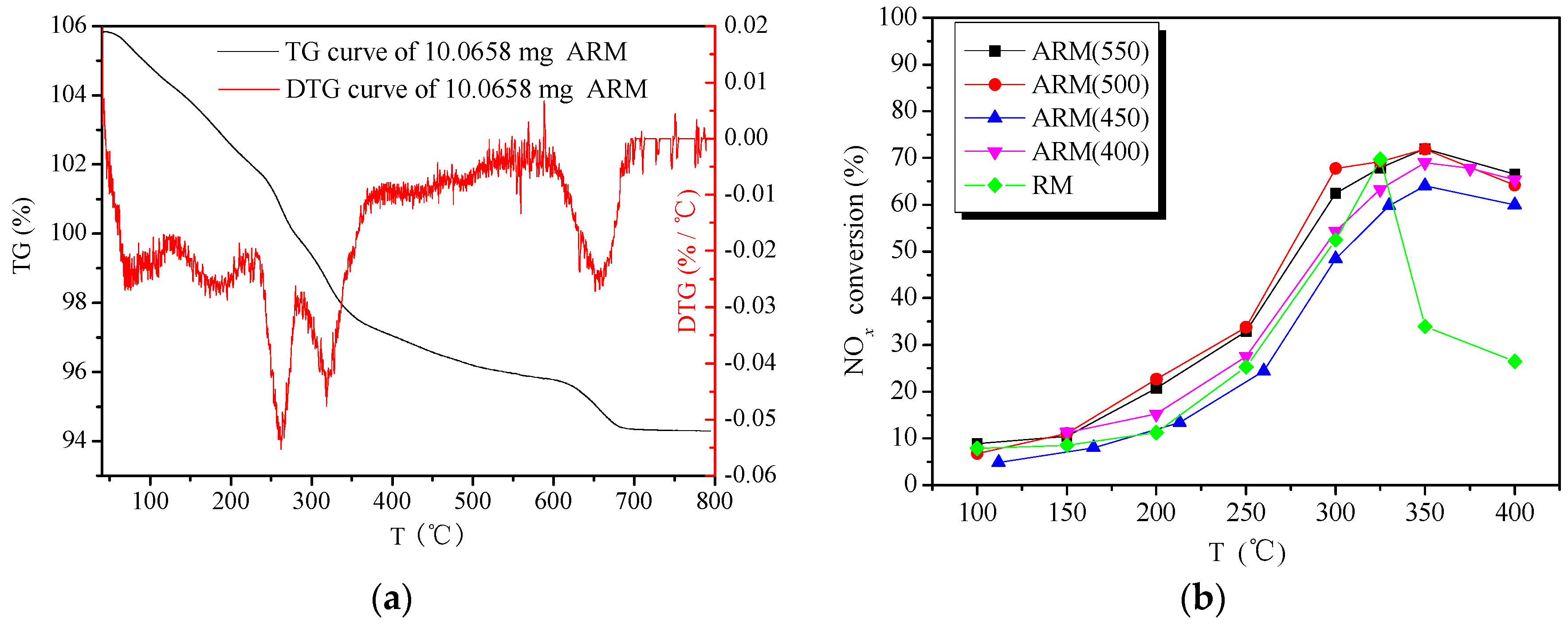
2.1.1. Components Analysis of RM and Alkali-Removed RM
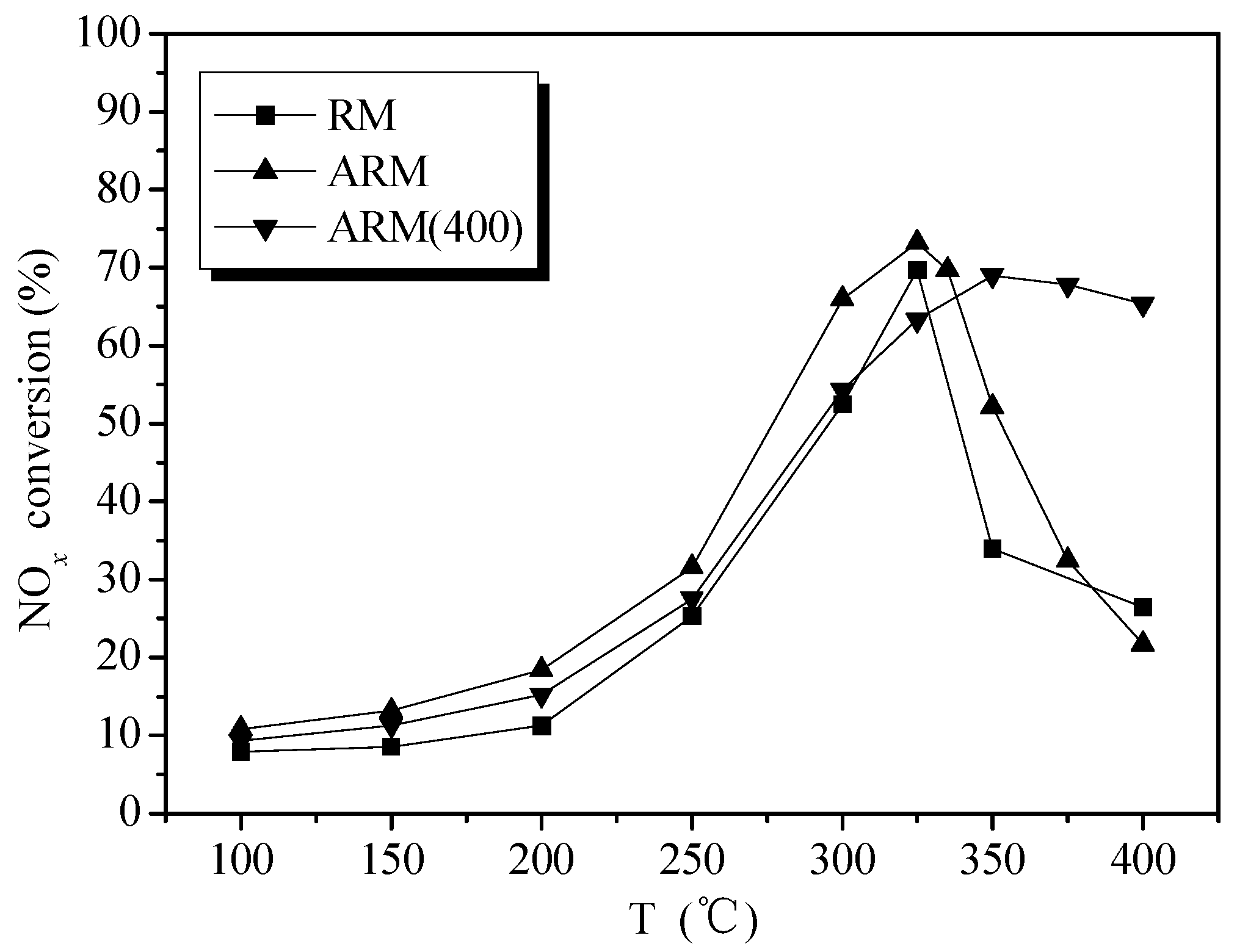
2.1.2. Catalytic Activity of RM and Alkali-Removed RM
2.1.3. Structure Characterization of RM and Alkali-Removed RM
2.1.4. Effect of Calcination Temperature on Catalytic Activity of ARM
2.2. Effect of Ce-Doping
2.2.1. Catalytic Activity of Ce/ARM
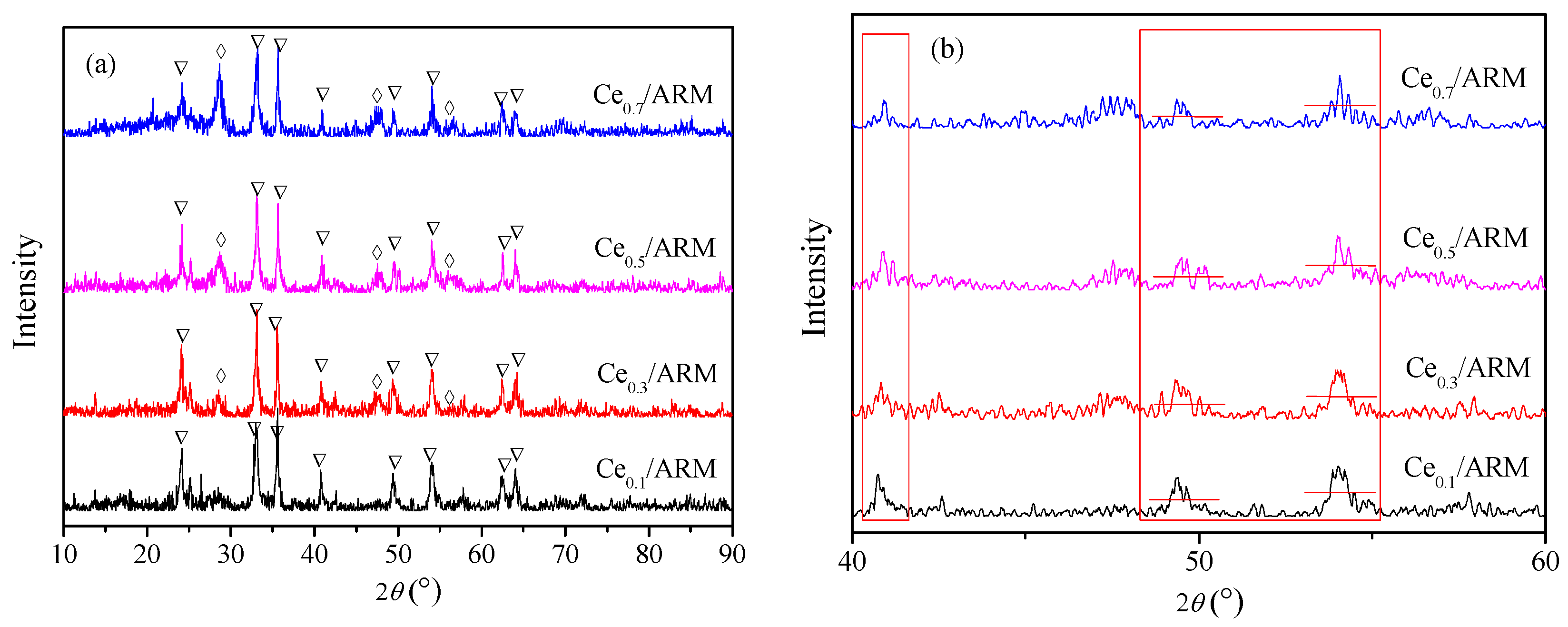
2.2.2. Structure Characterization of Ce/ARM
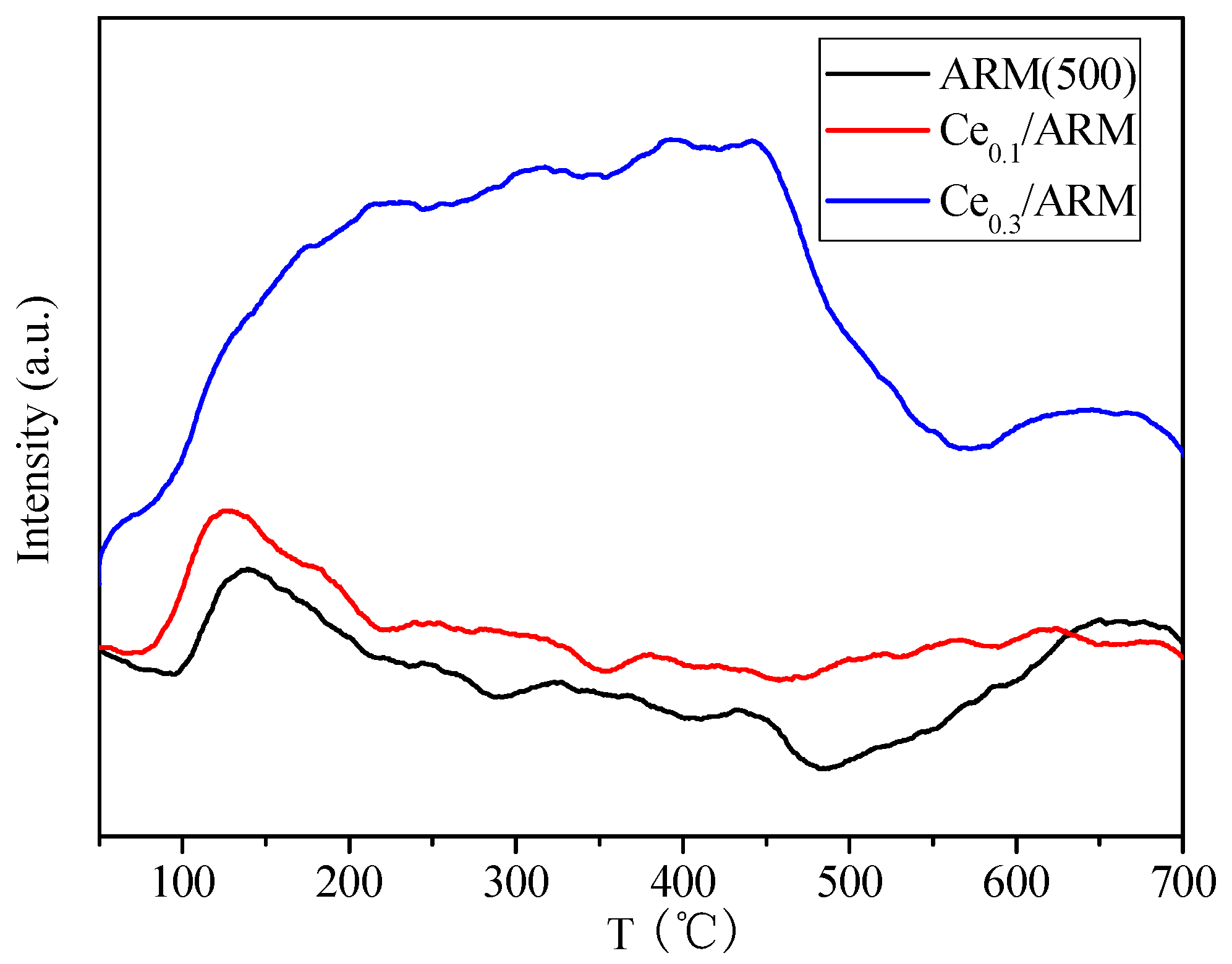
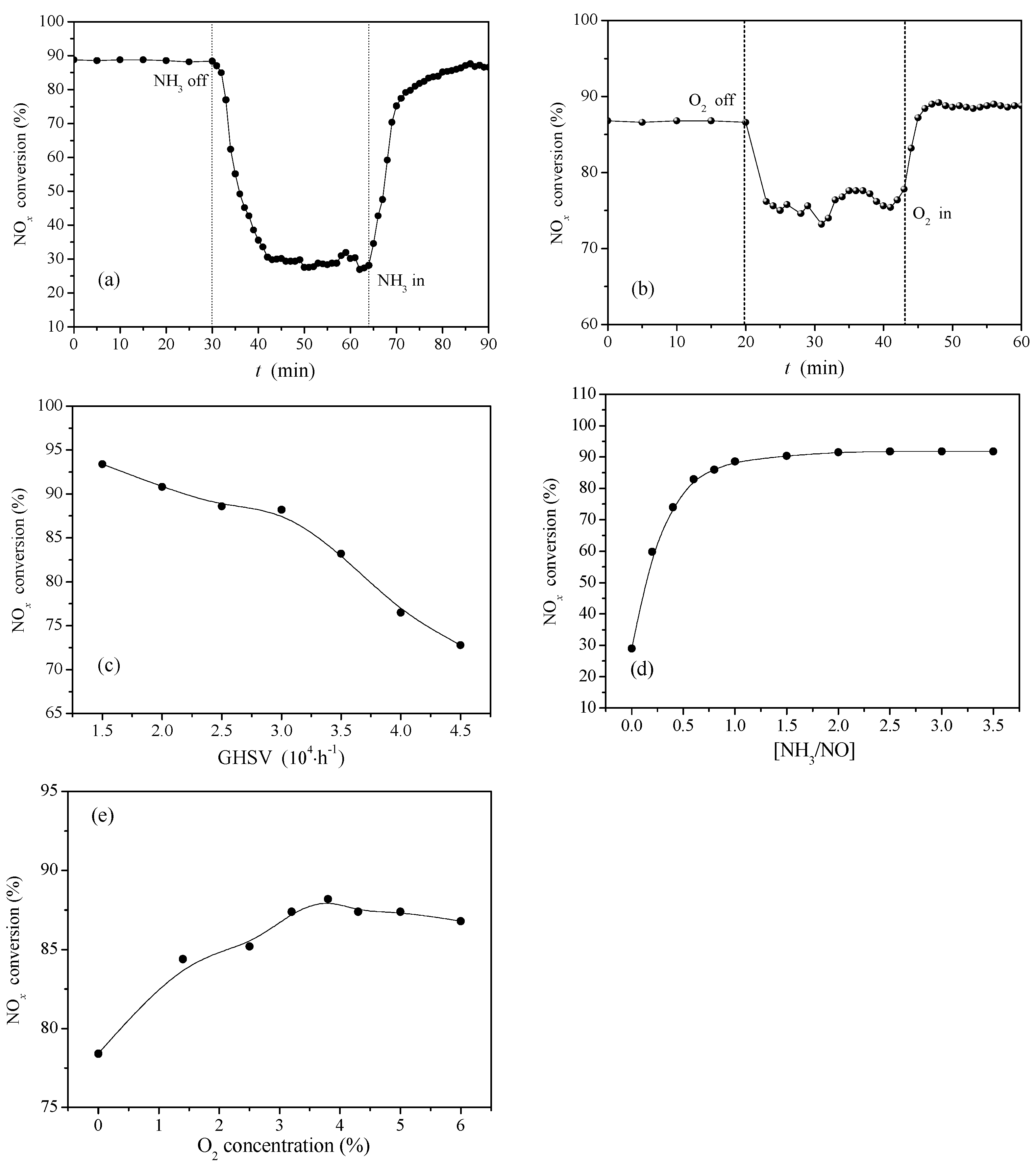
2.2.3. NH3 and O2 Transient Response of Ce0.3/ARM and the Effect of Major Operating Parameters
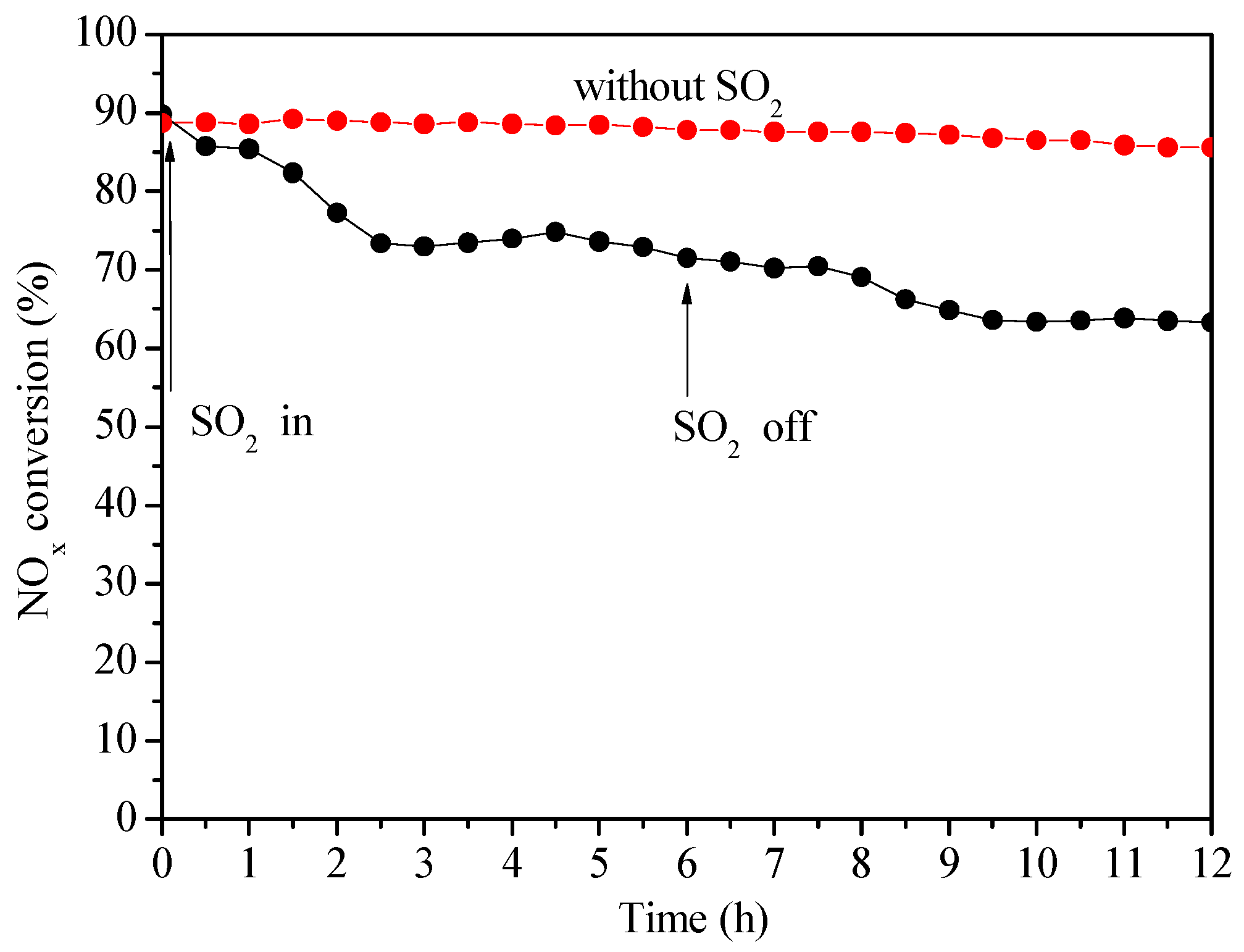
2.2.4. Effect of SO2 on Catalytic Activity of Ce0.3/ARM
3. Discussion
4. Materials and Methods
4.1. Preparation of RM and Alkali-Removed RM
4.2. Preparation of Ce-Doped Red Mud Catalyst
4.3. Catalytic deNOx Activity Measurements
4.4. Characterization of Catalysts
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hu, Z.P.; Zhao, H.; Gao, Z.M.; Yuan, Z.Y. High-surface-area activated red mud supported Co3O4 catalysts for efficient catalytic oxidation of CO. RSC Adv. 2016, 6, 94748–94755. [Google Scholar] [CrossRef]
- Hu, Z.P.; Zhu, Y.P.; Gao, Z.M.; Wang, G.X.; Liu, Y.P.; Liu, X.Y.; Yuan, Z.Y. CuO catalysts supported on activated red mud for efficient catalytic carbon monoxide oxidation. Chem. Eng. J. 2016, 302, 23–32. [Google Scholar] [CrossRef]
- Paredes, J.R.; Ordonez, S.; Vega, A.; Diez, F.V. Catalytic combustion of methane over red mud-based catalysts. Appl. Catal. B 2004, 47, 37–45. [Google Scholar] [CrossRef]
- Bento, N.I.; Santos, P.S.C.; de Souza, T.E.; Oliveira, L.C.A.; Castro, C.S. Composites based on PET and red mud residues as catalyst for organic removal from water. J. Harzard. Mater. 2016, 314, 304–311. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.B.; Qi, F.; Sun, D.Z.; Chen, Z.L.; Robert, D. Cerium doped red mud catalytic ozonation for bezafibrate degradation in wastewater: Efficiency, intermediates, and toxicity. Chemosphere 2016, 146, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Koumanova, B.; Drame, M.; Popangelova, M. Phosphate removal from aqueous solutions using red mud wasted in bauxite Bayer’s process. Resour. Conserv. Recycl. 1997, 19, 11–20. [Google Scholar] [CrossRef]
- Ordonez, S.; Sastre, H.; Diez, F.V. Characterisation and deactivation studies of sulfided red mud used as catalyst for the hydrodechlorination of tetrachloroethylene. Appl. Catal. B 2001, 29, 263–273. [Google Scholar] [CrossRef]
- Li, C.M.; Zeng, H.; Liu, P.L.; Yu, J.; Guo, F.; Xu, G.W.; Zhang, Z.G. The recycle of red mud as excellent SCR catalyst for removal of NOx. RSC Adv. 2017, 7, 53622–53630. [Google Scholar] [CrossRef]
- Cao, J.L.; Wang, Y.; Li, G.J.; Li, K.; Wang, Y.; Ma, M. Mesoporous modified red mud supported CuO nanocatalysts for carbon monoxide oxidation. Curr. Nanosci. 2015, 11, 413–418. [Google Scholar] [CrossRef]
- Lamonier, J.F.; Leclerco, G.; Dufour, M.; Leclercq, L. Utilization of red mud. Catalytic properties in selective reduction of nitric oxide by ammonia. Récents Progrès en Génie des Procédés, Boues industrielles: Traitements. Recents Prog. Genie Procedes 1995, 43, 31–36. [Google Scholar]
- Bhattacharyya, A.; Rajanikanth, B.S. Discharge plasma combined with bauxite residue for biodiesel exhaust cleaning: A case study on NOx removal. IEEE Trans. Plasma Sci. 2015, 43, 1974–1982. [Google Scholar] [CrossRef]
- Bhattacharyya, A.; Rajanikanth, B.S. Biodiesel exhaust treatment with HFAC plasma supported by red mud: Study on DeNOx and power consumption. Energy Procedia 2015, 75, 2371–2378. [Google Scholar] [CrossRef]
- Liu, X.; Li, J.H.; Li, X.; Peng, Y.; Wang, H.; Jiang, X.M.; Wang, L.W. NH3 selective catalytic reduction of NO: A large surface TiO2 support and its promotion of V2O5 dispersion on the prepared catalyst. Chin. J. Catal. 2016, 37, 878–887. [Google Scholar] [CrossRef]
- Li, J.H.; Chang, H.Z.; Ma, L.; Hao, J.M.; Yang, R.T. Low-temperature selective catalytic reduction of NOx with NH3 over metal oxide and zeolite catalysts—A review. Catal. Today 2011, 175, 147–156. [Google Scholar] [CrossRef]
- Liu, C.; Shi, J.W.; Gao, C.; Niu, C.M. Manganese oxide-based catalysts for low-temperature selective catalytic reduction of NOx with NH3: A review. Appl. Catal. A 2016, 522, 54–69. [Google Scholar] [CrossRef]
- Liang, H.; Gui, K.T.; Zha, X.B. DRIFTS study of γ-Fe2O3 nano-catalyst for low-temperature selective catalytic reduction of NOx with NH3. Can. J. Chem. Eng. 2016, 94, 1668–1675. [Google Scholar] [CrossRef]
- Sushil, S.; Batra, V.S. Modification of red mud by acid treatment and its application for CO removal. J. Harzard. Mater. 2012, 203–204, 264–273. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.B.; Ang, H.M.; Tade, M.O. Novel applications of red mud as coagulant, adsorbent and catalyst for environmentally benign processes. Chemosphere 2008, 72, 1621–1635. [Google Scholar] [CrossRef] [PubMed]
- Xuan, X.P.; Yue, C.T.; Li, S.Y.; Yao, Q. Selective catalytic reduction of NO by ammonia with fly ash catalyst. Fuel 2003, 82, 575–579. [Google Scholar] [CrossRef]
- Das, B.R.; Dash, B.; Tripathy, B.C.; Bhattacharya, I.N.; Das, S.C. Production of η-alumina from waste aluminium dross. Miner. Eng. 2007, 20, 252–258. [Google Scholar] [CrossRef]
- Zhao, H.; Zhang, D.X.; Wang, F.F.; Wu, T.T.; Gao, J.S. Modification of ferrite-manganese oxide sorbent by doping with cerium oxide. Process Saf. Environ. 2008, 86, 448–454. [Google Scholar] [CrossRef]
- Gao, X.; Jiang, Y.; Zhong, Y.; Luo, Z.Y.; Cen, K.F. The activity and characterization of CeO2-TiO2 catalysts prepared by the sol-gel method for selective catalytic reduction of NO with NH3. J. Harzard. Mater. 2010, 174, 734–739. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Li, J.H.; Li, X.; Peng, Y.; Wang, H.; Jiang, X.M.; Wang, L.W. Mechanism of selective catalytic reduction of NOx with NH3 over CeO2-WO3 catalysts. Chin. J. Catal. 2011, 32, 836–841. [Google Scholar]
- Wang, X.B.; Zhang, L.; Wu, S.G.; Zou, W.X.; Yu, S.H.; Shao, Y.; Dong, L. Promotional effect of Ce on iron-based catalysts for selective catalytic reduction of NO with NH3. Catalysts 2016, 6, 112. [Google Scholar] [CrossRef]
- Xu, C.H.F.; Chen, K.H.; Gu, Z.F.; Ma, D.D.; Rao, G.H. Effect of operation conditions on activity of de-NOx SCR catalysts before and after poisoned by alkali metal. Adv. Mater. Res. 2014, 955–959, 702–705. [Google Scholar]
- Seo, P.W.; Cho, S.P.; Hong, S.H.; Hong, S.C. The influence of lattice oxygen in titania on selective catalytic reduction in the low temperature region. Appl. Catal. A 2010, 380, 21–27. [Google Scholar] [CrossRef]
- Cao, S.T.; Ma, H.J.; Zhang, Y.; Chen, X.F.; Zhang, Y.F.; Zhang, Y. The phase transition in bayer red mud from China in high caustic sodium aluminate solutions. Hydrometallurgy 2013, 140, 111–119. [Google Scholar] [CrossRef]
- Cengeloglu, Y.; Tor, A.; Ersoz, M.; Arslan, G. Removal of nitrate from aqueous solution by using red mud. Sep. Purif. Technol. 2006, 51, 374–378. [Google Scholar] [CrossRef]
- Xu, W.Q.; Yu, Y.B.; Zhang, C.B.; He, H. Selective catalytic reduction of NO by NH3 over a Ce/TiO2 catalyst. Catal. Commun. 2008, 9, 1453–1457. [Google Scholar] [CrossRef]
- Georgiadou, I.; Papadopoulou, C.; Matralis, H.K.; Voyiatzis, G.A.; Lycourghiotis, A.; Kordulis, C. Preparation, characterization, and catalytic properties for the SCR of NO by NH3 of V2O5/TiO2 catalysts prepared by equilibrium deposition filtration. J. Phys. Chem. B 1998, 102, 8459–8468. [Google Scholar] [CrossRef]
- Mamulova, K.K.; Tokarsky, J.; Kovar, P.; Vojteskova, S.; Kovarova, A.; Smetana, B.; Kukutschova, J.; Capkova, P.; Matejka, V. Preparation and characterization of photoactive composite kaolinite/TiO2. J. Harzard. Mater. 2011, 188, 212–220. [Google Scholar] [CrossRef] [PubMed]
- Iwasaki, M.; Shinjoh, H. A comparative study of “standard”, “fast” and “NO2” SCR reactions over Fe/zeolite catalyst. Appl. Catal. A 2010, 390, 71–77. [Google Scholar] [CrossRef]
- Jiang, B.Q.; Wu, Z.B.; Liu, Y.; Lee, S.C.; Ho, W.K. DRIFT study of the SO2 effect on low-temperature SCR reaction over Fe-Mn/TiO2. J. Phys. Chem. C 2010, 114, 4961–4965. [Google Scholar] [CrossRef]
- Wang, H.Q.; Gao, S.; Yu, F.X.; Liu, Y.; Weng, X.L.; Wu, Z.B. An effective way to control the performance of a ceria-based deNOx catalyst with improved alkali resistance: Acid-base adjusting. J. Phys. Chem. C 2015, 119, 15077–15084. [Google Scholar] [CrossRef]
- Xiong, Z.B.; Lu, C.M.; Guo, D.X.; Zhang, X.L.; Han, K.H. Selective catalytic reduction of NOx with NH3 over iron-cerium mixed oxide catalyst: Catalytic performance and characterization. J. Chem. Technol. Biotechnol. 2013, 88, 1258–1265. [Google Scholar]



| Samples | LOSS | SiO2 | Al2O3 | Fe2O3 | CaO | MgO | SO3 | TiO2 | K2O | Na2O | Others |
|---|---|---|---|---|---|---|---|---|---|---|---|
| RM | 13.02 | 20.74 | 24.26 | 23.89 | 2.77 | 0.56 | 0.93 | 2.95 | 0.57 | 10.13 | 0.18 |
| WRM1 | 13.75 | 19.75 | 27.81 | 22.61 | 5.52 | 0.37 | 0.85 | 1.55 | 0.07 | 7.43 | 0.29 |
| WRM2 | 14.14 | 19.26 | 27.40 | 23.23 | 5.42 | 0.36 | 1.01 | 1.43 | 0.13 | 7.35 | 0.27 |
| ARM | 13.06 | 18.19 | 35.34 | 23.12 | 3.28 | 0.24 | 0.25 | 2.17 | - | 4.23 | 0.12 |
| Samples | BET Specific Surface Area (m2·g−1) | Pore Volume (cm3·g−1) | Average Pore Diameter (nm) |
|---|---|---|---|
| RM | 42.7 | 0.1919 | 109.2 |
| ARM | 50.5 | 0.1048 | 82.9 |
| ARM(400) | 57.2 | 0.1041 | 72.8 |
| Samples | BET Specific Surface Area (m2·g−1) | Pore Volume (cm3·g−1) | Average Pore Diameter (nm) |
|---|---|---|---|
| Ce0.1/ARM | 46.5 | 0.0922 | 87.9 |
| Ce0.3/ARM | 54.4 | 0.1002 | 81.6 |
| Ce0.5/ARM | 45.1 | 0.1071 | 98.0 |
| Ce0.7/ARM | 46.1 | 0.1181 | 103.1 |
| Options | Reference [8] | This Study |
|---|---|---|
| Synthesis method | acid-base neutralization | acid washing; impregnation |
| Activation treatment | washing, drying and calcination; SO2-activated | washing, drying and calcination |
| Catalyst composition | Fe2O3-TiO2/(SiO2-Al2O3) (RM-based catalyst) | Ce-doped mixed oxides with inevitable residual alkali (Ce/ARM) |
| Major active component | α-Fe2O3, Fe2(SO4)3 | α-Fe2O3, CeO2 and others |
| Active temperature window | 350–450 °C | 275–375 °C |
| Operating conditions | GHSV of 60,000 h−1 | GHSV of 30,000 h−1 |
| Samples | Ce Content (wt%) |
|---|---|
| Ce0.1/ARM | 1.40 |
| Ce0.3/ARM | 4.20 |
| Ce0.5/ARM | 7.01 |
| Ce0.7/ARM | 9.81 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, J.; Gong, Z.; Lu, C.; Niu, S.; Ding, K.; Xu, L.; Zhang, K. Preparation and Performance of Modified Red Mud-Based Catalysts for Selective Catalytic Reduction of NOx with NH3. Catalysts 2018, 8, 35. https://doi.org/10.3390/catal8010035
Wu J, Gong Z, Lu C, Niu S, Ding K, Xu L, Zhang K. Preparation and Performance of Modified Red Mud-Based Catalysts for Selective Catalytic Reduction of NOx with NH3. Catalysts. 2018; 8(1):35. https://doi.org/10.3390/catal8010035
Chicago/Turabian StyleWu, Jingkun, Zhiqiang Gong, Chunmei Lu, Shengli Niu, Kai Ding, Liting Xu, and Kang Zhang. 2018. "Preparation and Performance of Modified Red Mud-Based Catalysts for Selective Catalytic Reduction of NOx with NH3" Catalysts 8, no. 1: 35. https://doi.org/10.3390/catal8010035
APA StyleWu, J., Gong, Z., Lu, C., Niu, S., Ding, K., Xu, L., & Zhang, K. (2018). Preparation and Performance of Modified Red Mud-Based Catalysts for Selective Catalytic Reduction of NOx with NH3. Catalysts, 8(1), 35. https://doi.org/10.3390/catal8010035




