Abstract
The oxidation of eight pharmaceutical micropollutants by chloroperoxidase derived from Caldaromyces fumago using hydrogen peroxide as an electron acceptor is reported. All the tested compounds, namely trazadone, sulfamethoxazole, naproxen, tetracycline, estradiol, ketoconazole, ketorolac, and diclofenac, were found to be substrates for oxidation by chloroperoxidase. The respective oxidation products were identified by electrospray ionization–mass spectrometry. All the products contain at least one chloride atom in their structure after the enzymatic oxidation. Degradability experiments indicated that most of the reaction products are more biodegradable than the corresponding unmodified compounds. The enzyme was found to be catalytically active in effluent from a water treatment facility, transforming the micropollutants with high reaction rates and conversions. The enzyme was immobilized in chitosan macrospheres, which allowed the catalyst to be recycled for up to three treatment cycles in simulated samples of treated residual water. The conversion was high in the first two cycles; however, in the third, a 50% reduction in the capacity of the enzyme to oxidize ketorolac was observed. Additionally, immobilization improved the performance of the enzyme over a wider pH range, achieving the conversion of ketorolac at pH 5, while the free enzyme was not active at this pH. Overall, the results of this study suggest that chloroperoxidase represents a powerful potential catalyst in terms of its catalytic activity for the transformation of pharmaceutical micropollutants.
1. Introduction
Pharmaceutical compounds (PhCs) are currently considered to be micropollutants due to their persistence in the environment at ultralow concentrations [1,2,3]. The environmental effects of most of these compounds are still under study, and regulations and standards are also limited or nonexistent for most [2,4,5]. Because conventional biological treatment processes are only partially successful in removing these compounds [6,7,8], the application of additional technologies is necessary to achieve their removal or transformation to innocuous compounds. Thus, several technologies have been proposed to achieve high removal rates [9,10,11]. Physicochemical and biological treatments are among the most promising technologies for the treatment of wastewater containing PhCs. These treatments represent green methods for the degradation of hazardous organic compounds into nontoxic by-products. For instance, advanced oxidation processes (AOPs) are a class of methodologies based on the generation of radicals (hydroxyl radicals) to react with organic compounds present in the medium and promote their degradation, and potentially, their mineralization. The advantage of hydroxyl radicals is that a sample containing several pollutants can be wholly treated without selectivity. The most common AOPs are photolysis, photocatalysis, Fenton, photo–Fenton, ozonation, sonolysis, radiolysis, and electrochemical methods [9,10,11]. The electrochemical techniques include anodic oxidation (AO), electro–Fenton (EF), photoelectro–Fenton (PEF), and sono-electrochemistry methods, which are based on the generation of reactive species. The advantages of AOPs include their easy operation, safety, low cost, environmental compatibility, versatility, short retention times compared to conventional methods, and potential to achieve a high degree of mineralization. AOPs offer a promising alternative for the treatment of recalcitrant organic pollutants present in air, soil, and water, including dyes, pesticides, herbicides, chelating agents, microcystin toxins, and others [12]. AOPs have been used for the oxidation of various pharmaceuticals, including antibiotics, analgesics, anti-inflammatories, and hormones, achieving high reaction rates and conversions, and even mineralization [9].
Another potentially attractive technology is biocatalysis [13], as the transformation of PhCs into less toxic and more biodegradable products using oxidative enzymes such as laccases and peroxidases has been reported [14,15,16]. Biocatalytic transformations are considered to be sustainable processes because they are carried out at mild reaction conditions, their energy demand is low, they do not generate sludge as a side product, and the reactions usually do not use toxic chemical agents. The use of enzymes as biocatalysts in PhC removal is convenient because of the high reaction rates, high conversions, and mild reaction conditions [17,18]. These oxidative enzymes have the capacity to recognize a wide variety of organic aromatic compounds as substrates, and subsequently oxidize these substrates in a selective and fast way. Thus, oxidative enzymes have potential as catalysts in chemical synthesis, with applications in diverse industrial fields. Furthermore, because of their wide substrate variability, these enzymes have also been proposed for the oxidative catalysis of aromatic pollutants such as organophosphorus pesticides, azo-like dyes, organochlorine and organophosphorus pesticides, polycyclic aromatic hydrocarbons, and, recently, pharmaceutical drugs.
The enzymatic oxidation of pollutants usually results in non-toxic products, or products with decreased toxicity and higher biodegradability [17,19]. For example, triclosan, an antibacterial and fungicidal agent widely used as a disinfectant in hospitals, homes, and industry, is enzymatically oxidized by the laccase of Ganoderma lucidum to produce triclosan dimers and trimers. Toxicity studies of these products revealed that this enzymatic oxidation effectively reduced the toxicity of triclosan, with a final conversion of 100% towards non-toxic or less toxic products [20]. Other pharmaceutical compounds, anti-inflammatory drugs (diclofenac and naproxen), and estrogen hormones (estrone, 17β-estradiol, and 17β-ethinylestradiol), have been transformed using laccases from other sources, such as Myceliophthora thermophile [21]. Peroxidase enzymes have also been reported to oxidize several drugs. Wen et al. (2009) reported the oxidation of two widely used antibiotics, tetracycline and oxytetracycline, by raw lignin peroxidase extracted from the fungus Phanerochaete chrysosporium, with a conversion of 95% in 5 min [22]. Manganese peroxidase from this same fungus can oxidize the steroid hormones 17β-estradiol and ethinylestradiol with a conversion of 95% and an 80% reduction of estrogenic activity. The enzymatic oxidation of several other drugs has also been reported recently [23].
The main limitation of environmental biocatalysis is the low operational stability of enzymes, which has delayed its application on an industrial scale. However, interesting studies into overcoming this limitation, mainly through the immobilization of the enzymes in organic, inorganic, and hybrid materials, have been reported. Through immobilization, the working pH range, thermostability, and working temperature range have been extended, and reuse of the enzyme has been achieved, indicating an improvement in operational stability [24,25,26].
Chloroperoxidase (CPO) from Caldaromyces fumago is a particularly interesting oxidative enzyme because of its potential applications in several areas, ranging from synthesis to remediation. This enzyme has the capability to transform compounds as different in chemical nature as pesticides [27], polycyclic aromatic hydrocarbons [28], azo dyes [29], and sulfur and nitrogen heterocyclic compounds [30,31]. The wide substrate variability of this enzyme makes it a good candidate for studies of the transformation of pharmaceutical micropollutants. However, its catalytic activity is dependent on the presence of activating salts such as chloride (or bromide) ions, which in certain cases can be counterproductive, especially if environmental remediation is envisaged. This information is important because the enzymes are biological catalysts, and these reactions could take place in nature, and a halogenating bioreaction would increase the environmental impact of the PhCs. Thus, in the present work, the biocatalytic transformations of several PhCs by free and immobilized CPO were studied. The kinetic constants were determined, the reaction products were identified, and the biodegradability indexes of the halogenation products were obtained. The catalytic capacities of the free and immobilized enzymes were assessed in simulated treated water samples from the secondary unit of a water treatment process to test the enzyme’s ability to operate in complex media.
2. Results and Discussion
CPO was found to catalyze the oxidation of all eight PhCs assayed. Two of these PhCs are non-steroidal anti-inflammatories, two are antibiotics, one is a steroid hormone, one is an antifungal, one is analgesic, and one is an antidepressant. A conversion greater than 80% with a reaction time of 10 min was obtained for all substrates except trazadone, which was oxidized very slowly and for which only 4% conversion was observed. Figure 1 shows the time course curves for the oxidation of the eight PhCs by CPO using 0.1 mM H2O2. As can be seen, high conversions were reached during the first 5 min.
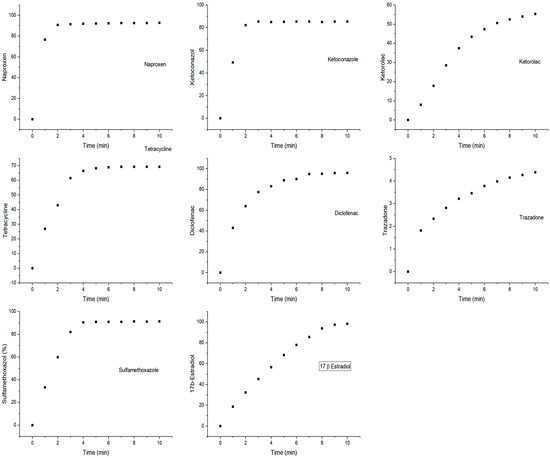
Figure 1.
Time course curves of the oxidation of the eight pharmaceutical compounds (PhCs) by chloroperoxidase (CPO).
The transformation of the antimicrobials sulfamethoxazole, tetracycline, and ketoconazole are of particular interest due to the current environmental problems related to antimicrobial resistance. The exposure of microorganisms to these compounds in the environment contributes to the appearance of resistant strains, which currently represents a serious health problem worldwide [32,33,34]. The transformation of endocrine disruptors is also relevant, given that water contamination by natural and synthetic hormones has frequently been reported, as have the toxic effects of hormone pollutants on various organisms [35]. Environmental contamination by anti-inflammatories has also recently been reported. Although their environmental impact has not been completely elucidated, some non-steroidal anti-inflammatory drugs have been reported to bioaccumulate in fish [36], to affect the balance of microbial populations [37], and to have toxic effects on agricultural plants [38]).
The catalytic parameters are given in Table 1. The parameter kcat is the first-order rate constant, which is often called the turnover number of the enzyme, because it represents the maximum number of substrate molecules that can be converted to products per active site per unit time. The kcat values of the tested PhC substrates were high, varying from 1141 min−1 for trazadone to 15,999 min−1 for naproxen. These values were higher than those reported for the oxidation of other contaminants by CPO under similar assay conditions [27,30,39]. Km is the apparent dissociation constant, which can be treated as the overall dissociation constant of all enzyme-bound species. The Km values for hydrogen peroxide were similar than those reported for other aromatic substrates [29,39] (Table 1). At high substrate concentrations, kcat is the parameter that best describes the suitability of a substrate for a particular enzyme; however, if the substrate concentration is lower that the Km value, the better substrate for the enzyme the one which has the lower Km value [40]). This contrariety can be resolved by using the parameter kcat/Km as a specificity constant [40]. Using this parameter, Table 1 shows that the best PhC substrate for CPO was naproxen, followed by diclofenac, and sulfamethoxazole. Again, some kcat/Km values were higher than those reported for other substrates of CPO under similar conditions. For example, the kcat/Km values for 2,3,5,6-tetrachlorophenol, pentachlorophenol, 2,3,5,6-tetrachloroaniline, and parathion are 2028, 470, 133, and 33 s−1·mM−1, respectively [27,39]. High conversions for the PhCs estradiol, sulfamethoxazole, diclofenac, and naproxen by CPO oxidation have also been previously reported [41,42]; however, no kinetic data were available for these compounds. Such data are useful to design the eventual catalytic process. Overall, PhCs seem to be readily oxidizable substrates for CPO.

Table 1.
Catalytic parameters of the oxidation of PhCs by CPO *.
2.1. Product Identification
To elucidate the degradation mechanism and evaluate the environmental safety of the enzymatic transformation, it is necessary to analyze the intermediate and final products generated in the degradation reaction. The versatile catalytic activity of CPO makes the production of a broad array of reaction products from PhCs possible. However, under the conditions employed in our study, only halogenation, hydroxylation, and decarboxylation activities were observed. Table S2 lists the molecular weights of the enzymatic oxidation products. According to these results, most of the PhCs were oxidized by the addition of one or even two chlorines to the molecule. No reaction was detected under peroxidase activity conditions (pH 5.0–6.0, and/or in the absence of chlorine ions) for any of the PhCs tested. Some of the reaction products have been previously reported [41,42,43]. However, two PhCs showed unique behavior under our tested conditions. Based on the ESI(+) mass spectrometry of the naproxen products, two main compounds were identified in the degradation process. In the first stage of the degradative CPO-catalyzed oxidation, naproxen was hydroxylated at the o- and m-positions. However, this product was not observed in the MS analysis. The observation of a molecular ion [M + H]+ at m/z 261.1231 suggested that this compound was further converted to the corresponding 1,4-naphthoquinone derivative (C14H13O5, m/ztheo 261.0763). Another degradation route could also be proposed based on the observation of another peak at m/z 217.0865. In this case, a decarboxylation step after hydroxylation in the CPO-catalyzed conversion could be achieved (Figure 2). In contrast, in the degradation of trazadone, the detected species suggested the sequential addition of chlorine at the o and m-positions. Two main ions with chlorine-specific isotopic characteristics were detected in the ESI(+) mass spectra (Figure 3). The first ion, detected at m/z 406.1082, was attributed to the addition of one chlorine at the o-position. The isotopic pattern was characteristic of the presence of two chlorine atoms, and a molecular formula of C19H21Cl2N5O (m/z 406.1196) can be corroborated. A second ion at m/z 440.0675 was assigned to the addition of two chlorine atoms. The isotopic pattern and the molecular weight confirmed the presence of a compound with a molecular formula of C19H20Cl3N5O (m/z 440.0806). No oxidation products were detected for diclofenac under these assay conditions during the ESI-MS analysis; oxidized products from the literature are described in Table S2 [41]. Estradiol was analyzed using ESI and GC-MS techniques. Halogenated products were detected only by GC-MS analysis following the procedure described in the Materials and Methods section [43]. Finally, the halogenated products of sulfamethoxazole were different from those previously reported. According to Zhang et al., sulfamethoxazole first undergoes halogenation, followed by ring opening [42]. In this work, only fragments were observed after the reaction: one product with a molecular formula of C10H11ClN3O3S (m/z 288.0254) and another one with a formula of C4H5NO3 (m/z 145.00), probably as a result of more extensive oxidation due to the concentration of activating salts.
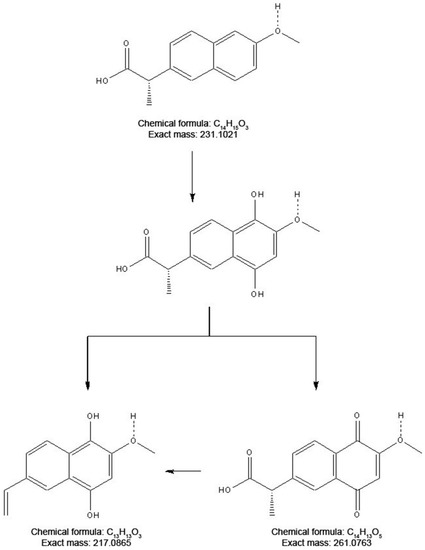
Figure 2.
Proposed conversion products from the CPO-catalyzed oxidation of naproxen.
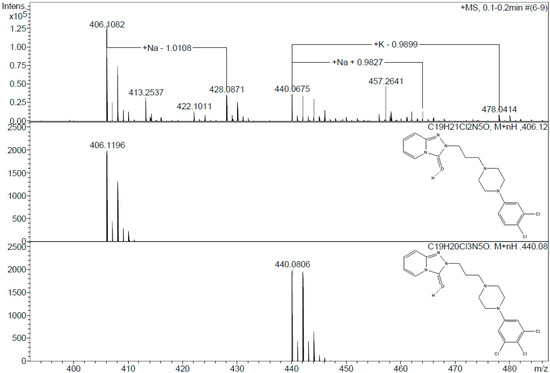
Figure 3.
ESI-mass spectra of the conversion products of the CPO-catalyzed oxidation of trazadone recorded in negative mode.
The chemical halogenation of PhCs has been detected in wastewater treatment plants. The application of chlorine (i.e., HOCl/OCl−) or chlorine dioxide (ClO2) during the final disinfection step can chlorinate micropollutants that are recalcitrant to biological treatment. For example, pharmaceuticals such as acetaminophen, sulfamethoxazole, diclofenac, metoprolol, gemfibrozil, and metoprolol are oxidized during chlorination [44,45]. The chlorination of some of these compounds gives rise to more toxic products; for example, several toxic products have been detected from the halogenation of paracetamol in simulated wastewater treatment plant effluents [46]. On the other hand, in some cases chlorination can produce less toxic or more biodegradable compounds. For instance, halogenated derivatives of β-estradiol have been reported to retain little estrogenic activity [43,47]. Additionally, in a previous work, the chlorination of sulfamethoxazole, diclofenac, and naproxen by CPO produced halogenated products with greater biodegradability and lower toxicity than the original drugs [41,42]. Because the authors of that study used an adapted inoculum from a biological reactor of a secondary treatment stage, an investigation of the biodegradability of the reaction products in natural water bodies, where non-adapted microorganisms could encounter these compounds, is still required.
2.2. Studies of the Biodegradability of the PhCs and the Enzymatic Reaction Products
The biochemical oxygen demand/theoretical oxygen demand (BOD28/ThOD) ratio was applied as an index of the biodegradability of a substance according to the OECD 301D specifications [48]. In this work, an inoculum of non-adapted microorganisms taken from the surface of a body of water was used. The biodegradability indexes of the oxidized products were higher than those of the corresponding unchlorinated substances, except in the case of estradiol (Figure 4). This last result may be explained by the fact that chlorinated products of estradiol are more toxic towards the inoculum than the original substance (Table S3); this suggests that the enzymatic products may have a different type of toxicity, not related to endocrine activity but another form of toxicity that inhibits the respiration of the microorganisms [41,42].
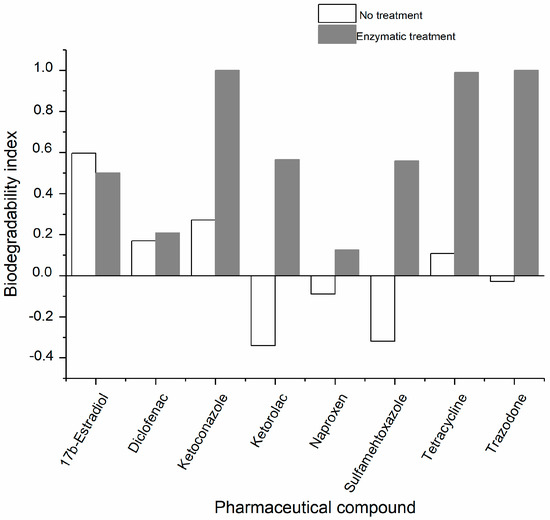
Figure 4.
Biodegradability indexes of the PhCs and their CPO oxidation products.
The enzymatic oxidation of trazadone, tetracycline, and ketoconazole produced readily biodegradable products, as reflected by their high biodegradability indexes.
For the calculation of the index, the highest theoretical oxygen demand (ThOD) was used in cases in which several oxidation products were identified. In the case of diclofenac, for which no products were detected, the data were calculated using the previously reported products [41]. Higher indexes indicate the higher biodegradability of the PhCs after being oxidized. Lower values indicate low biodegradability, and negative values may indicate some degree of toxicity of the PhCs toward the inoculum (less oxygen consumption was observed compared to the respiration of the blank assays) [48]. The toxicity testing recommended by OECD 301D was carried out for all the treated and untreated PhCs, in the presence of the reference compounds (dextrose and glutamic acid) (Table S3). Notably, ketorolac showed 100% inhibition of the respiration. Again, the oxidation products presented a lower inhibition than the non-oxidized substances, except in the case of estradiol and, to a lesser extent, sulfamethoxazole, suggesting that the enzymatic treatment could decrease the toxicity of most PhCs. However, additional toxicity tests are required to confirm the decrease in toxicity after the enzymatic oxidation.
2.3. Application of the Enzymatic Treatment in Simulated Treated Wastewater
Finally, the ability of CPO to chlorinate all the PhCs was determined in simulated samples of treated wastewater produced from a treatment plant in the state of Puebla, México (Table S1). This water did not contain measurable quantities of the tested PhCs. In addition, it should be noted that the wastewater did not contain peroxide or chlorides (the concentration of both was under the limit of detection), and that the addition of these compounds was necessary to affect the transformation of the PhCs. The pH of the samples also required adjustment to pH 3. CPO was found to be catalytically active for the oxidation of the tested compounds in the treated wastewater samples. The attained conversions were higher than 90% for five of the PhCs (diclofenac, sulfamethoxazole, naproxen, ketorolac, and tetracycline); estradiol and ketoconazole were oxidized with a conversion of approximately 60%; and trazadone was only oxidized to 12% conversion. Therefore, CPO was able to oxidize PhCs in complex treated wastewater media, suggesting that this method could potentially be applied as a polishing technology to oxidize compounds that are recalcitrant or toxic to conventional biological treatment. Such treatment would increase the biodegradability of the PhCs and facilitate their biological attenuation after the effluent is discharged into a natural water body.
In order to improve the biocatalytic performance of the CPO, it was immobilized in macrospheres of chitosan (CPO–chitosan macrospheres). The aim of the immobilization procedure was to enable the reuse of the enzyme and to increase its operational stability. The immobilization was carried out by physical adsorption, yielding synthesized macrospheres with an average diameter of 2 mm (Figure S1) that were brown in color due to the presence of the enzyme (Figure S1). This biocatalyst was analyzed using Raman spectrometry. The Raman spectra of the chitosan powder (Figure 5a), the chitosan macrospheres (Figure 5b), and the enzyme-chitosan powder (Figure 5c) are shown in Figure 5. A comparison of the spectra in Figure 5a,c, shows evident changes, which were attributed to the cross-linking reaction between chitosan and glutaraldehyde. In the double bond stretching region (1500 to 1700 cm−1) of the chitosan powder spectrum, the peak at 1586 cm−1 was assigned to the aromatic ring –C=C– stretching vibration and the band at 1618 cm−1 was associated with the double bond ring stretch modes (C=N) [49], providing evidence of crosslinked networks from the chitosan with glutaraldehyde. On the other hand, the intensity of the CH stretching vibration region between 2800 and 3000 cm−1 decreased considerably in the pellet spectrum of macrospheres which was related to the degree of cross-linking between the polymeric form of glutaraldehyde and chitosan. Finally, in the range from 250 to 1500 cm−1, multiple peaks associated with the chitosan powder spectrum decreased in intensity or were quenched, likely because these vibrations were associated with the non-conjugated form of chitosan, and were decreased by interactions with glutaraldehyde.
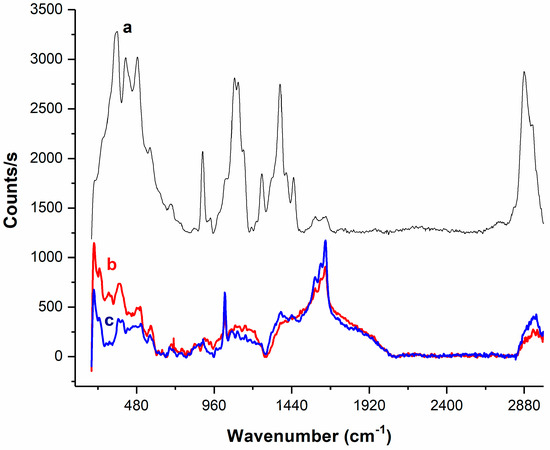
Figure 5.
Raman spectra of the chitosan powder (a); the chitosan macrospheres (b); and the enzyme-chitosan macrospheres (c).
The spectrum of the enzyme–chitosan macrospheres (Figure 5c) was very similar to that of the chitosan macrospheres (Figure 5b). The intensity of the CH stretching vibration region decreased when the protein was present. This observation was in agreement with the proposal that proteins may catalyze the aldol condensation/polymerization of glutaraldehyde, thus increasing the degree of cross-linking [50]. In the same spectrum, an increase in the intensity of the band at approximately 1030 cm−1, which is associated with the C-C stretching vibrations of the pyranose ring [51], was also observed. This behavior may be related to an increase in such functional groups due to the intricate chemical nature of the reaction of glutaraldehyde with the enzyme or to changes in the geometrical conformation of chitosan in the presence of the enzyme [52].
The CPO–chitosan macrospheres were then assayed for adsorption and oxidation of the PhCs, as shown in Table 2. For the heterogeneous catalysis process, the adsorption process was important for the subsequent oxidation, so it was desirable to measure and quantify the adsorption of the PhCs on the macrospheres. Up to 31% adsorption was observed for sulfamethoxazole, while practically no naproxen or ketorolac adsorption was observed. In general, little adsorption of the PhCs on the chitosan matrix was observed. The immobilized enzyme maintained the capacity to oxidize the PhCs with high conversion (up to 92% for ketorolac), except for trazadone (12%) (Table 2). The total removal of the PhCs, i.e., the sum of the adsorption and oxidation, reached values greater than 80% for all PhCs except trazadone.

Table 2.
Oxidation and adsorption of the PhCs by the CPO–chitosan macrospheres.
In addition, immobilization improved the activity profile with respect to pH, as shown in Figure 6. The free enzyme remained active until a pH of 5, at which its catalytic activity dropped to 62% of its maximum activity (pH 3), while the immobilized enzyme maintained 100% of its activity throughout the pH range of 3 to 5. At pH 6, both biocatalysts underwent a significant decrease in activity. The free enzyme achieved 0.13% conversion of ketorolac, while the immobilized enzyme degraded 4% of the ketorolac. It is known that immobilization with chitosan and other materials, such as silica derivatives, can improve the activity or stability of peroxidases by providing a protective microenvironment against fluctuations in pH [53,54]. The improvement in the stabilization or activity of enzymes by immobilization is often the result of a combination of physicochemical factors, such as enzyme rigidification, enzyme distortion, pH gradients, substrate or product gradients, partitioning (towards or away from the enzyme environment, for the substrate or the products), or the blocking of some enzyme areas (e.g., reduced inhibition), which may greatly improve enzyme performance [55].
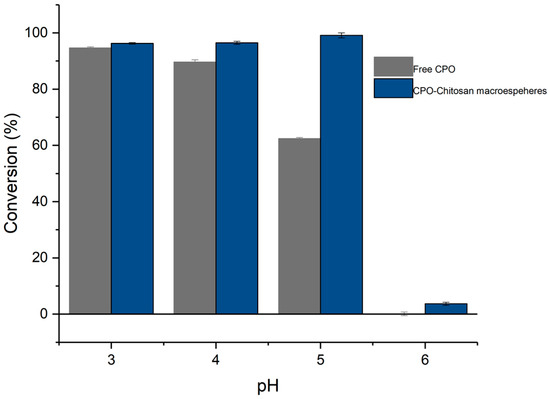
Figure 6.
Profile of activity versus pH for free CPO and the CPO–chitosan macrospheres during the oxidation of ketorolac.
The main advantage of the immobilized enzymes was their recyclability. As shown in Figure 7, the enzyme could be reused for up to three reaction cycles, although a pronounced loss of activity was observed in the third cycle (an approximately 65% decrease). In the fourth cycle, only a small amount of drug degradation was observed (less than 10%). Since hydrogen peroxide inactivation has been reported to be the main cause of peroxidase activity loss, the recyclability was determined at two different peroxide concentrations. However, the number of cycles in which the enzyme remained active did not change, and a small increase in the conversion of the third cycle was observed when the concentration of peroxide was decreased from 0.2 to 0.1 mM. These results suggested that another factor might be responsible for the inactivation of the immobilized enzymes.
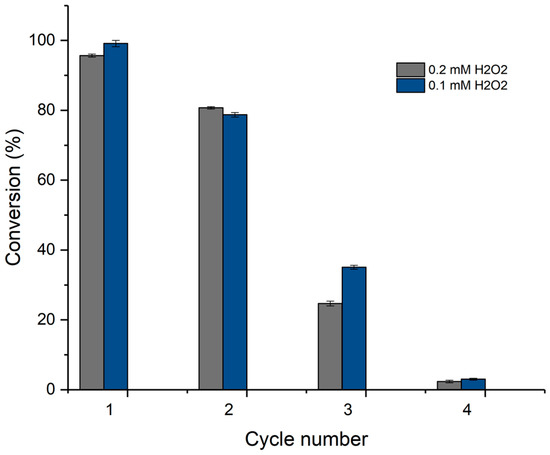
Figure 7.
Recyclability of the CPO–chitosan macrospheres for ketorolac oxidation at two hydrogen peroxide concentrations.
For the eventual practical application of a biocatalytic system based on CPO, some limitations remain to be overcome, such the need for pH adjustment and the addition of activating salts and hydrogen peroxide. Additionally, the catalytic activity of the enzyme at the low concentrations of contaminants present in natural systems must be determined. However, some surface water contains a sufficiently high peroxide concentration and chlorides to activate haloperoxidase enzymes [56]. With respect to the need for pH adjustment, some industrial effluents are acidic, such as mining or cleaning products; however, the pH of municipal effluents is usually nearly neutral. It is important to develop enzymatic preparations with catalytic activity in near real-world conditions, or alternatively, to have the infrastructure to lower the pH before treatment and neutralize it at the end of the treatment in controlled conditions, and to analyze the possible consequences of these treatments in advance. However, some potential applications for this enzyme are envisaged to treat effluents from small pharmaceutical industries or feed water for industries in which physicochemical conditions can be changed during treatment, and can be reconditioned before final discharge.
In conclusion, chloroperoxidase is an enzyme with the ability to oxidize several micropollutants to more biodegradable compounds quickly and efficiently (except for estradiol). As the enzyme maintains this capacity even in complex systems such as simulated residual treated water, it is conceived as a potential biocatalyst for eventual applications, once some limitations have been overcome, such as the working range of the pH and the reusability of the biocatalysts.
3. Materials and Methods
3.1. Chemicals
The pharmaceutical compounds diclofenac, 17β-estradiol, ketoconazole, ketorolac, tetracycline, sulfamethoxazole, naproxen, and trazadone were purchased from the company Sigma-Aldrich (St. Louis, MO, USA). Chloroperoxidase (CPO) from Caldaromyces fumago was purchased from Alltaenzymes (Edmonton, AB, Canada) with an RZ of 1.4 and a specific activity of 22,000 min−1 for the halogenation of monochlorodimedone. The buffer salts, 2-propanol (HPLC grade), dichloromethane (HPLC grade), sodium sulfate, hydrogen peroxide, and sodium chloride were purchased from J.T. Baker (Phillipsburg, NJ, USA).
3.2. Enzymatic Activity in Model Systems
The catalytic activities of CPO towards the PhCs was determined in a 1 mL reaction mixture containing 260 nM of the enzyme and 5 µM of the PhC in a medium containing a 10% isopropanol–60 mM phosphate buffer at pH 3 and 25 °C. The reactions were started by adding 0.1 mM of hydrogen peroxide. The control sample contained the same components, except for the enzyme. The reaction conversion was estimated by monitoring the change in the substrate peaks over 10 min using an HPLC system equipped with a UV-vis detector. The concentration of remaining substrate was measured every minute, by stopping the reaction by adding 1 mL of isopropanol. The peak areas were transformed using a previously prepared standard curve for each PhC. The conversion was calculated by the following formula:
where C0 and Ct are the initial concentration and the concentration after 10 min of reaction. The reported values are the mean of three replicates.
3.3. Kinetic Constants
The kinetic constants were calculated by determining the initial reaction rate of PhC transformation at different hydrogen peroxide concentrations while the PhC concentration was kept constant. A total of 5 µM of the PhC in a medium containing a 10% isopropanol–60 mM phosphate buffer at pH 3 and 25 °C was incubated with 260 nM of the enzyme, and the reaction was started by adding hydrogen peroxide (from 0.1 to 1 mM). After 20 s, the reaction was stopped by adding 1 mL of isopropanol. The change in the substrate concentration was divided by the protein concentration and reaction time. In this way, the initial reaction rates were calculated as moles of substrate converted per mole of enzyme per minute, or simply in min−1. The reported values are the mean of three replicates. The initial reaction rates were plotted versus the hydrogen peroxide concentration, and the data were fit to the Michaelis–Menten equation using an iteration procedure following the Marquardt–Levenberg nonlinear least-squares algorithm using Origin 9.0 software (Originlab Corporation, Northampton, MA, USA):
where vi is the specific initial reaction rate defined above, kcat is (Vmax/[E]), which is also the catalytic turnover (the catalytic constant for the conversion of substrate to product, calculated as the maximum velocity divided by the enzyme concentration), S is the hydrogen peroxide concentration and Km is the Michaelis constant (defined, operationally, as the substrate concentration at which the initial rate is one-half of the maximum velocity). The reported values are the mean of three replicates.
3.4. Enzyme Immobilization
Chitosan macrospheres were prepared using a previously reported precipitation method [57]). Chitosan (2 w/v %) was dissolved in an aqueous solution of acetic acid (2 w/v %). The solution was added drop-wise to a coagulation liquid (1 N sodium hydroxide and 26 v/v % ethanol) under stirring. The produced macrospheres were filtered and washed with distilled water until a neutral pH was reached. Cross-linking was carried out by shaking the chitosan macrospheres with a 2% glutaraldehyde solution for 4 h. Excess glutaraldehyde was removed by washing the macrospheres several times with distilled water. Immobilization of CPO on the chitosan macrospheres was carried out by incubating 1 g (wet weight) of the particles with 23 nmol of CPO for 24 h at 4 °C. Subsequently, the immobilized enzyme was centrifuged at 5000 rpm and washed with 60 mM potassium phosphate buffer (pH 4) until protein was no longer detected in the washing solution. The amount of protein immobilized was measured by difference, with protein concentrations determined at 398 nm (ε = 85,000 M−1·cm−1) before and after CPO adsorption (recovered enzyme in the supernatant after washing). The final enzyme preparation was kept in 1 mL of 60 mM phosphate buffer at pH 4.
The catalytic conversion of the PhCs by the CPO–chitosan macrospheres was determined in a volume of 1 mL containing 20 μM of the substrate, 20 mM of KCl, and 1 macrosphere in a 60 mM phosphate buffer at pH 3.0 under gentle agitation for 10 min. The conversion of the PhCs was monitored by HPLC. The removal was evaluated by both adsorption and conversion. For the first, the macrospheres were incubated in the absence of hydrogen peroxide. For the latter, the enzymatic conversion was measured by adding 0.2 mM of H2O2 (subtracting the percentage of adsorption). The conversion mediated by the CPO–chitosan macrospheres and the free enzyme was also determined in the pH range of 3–6 using ketorolac as the model substrate. Additionally, the recyclability of the immobilized enzyme was determined using ketorolac as the model substrate at pH 5. For this, the reaction was carried out for 10 min, after which the reaction mixture was centrifuged for 3 min at 3000 rpm to separate the macrospheres, which were then washed with phosphate buffer at pH 5, and added to a new reaction mixture for reuse. The ketorolac conversion was determined by HPLC as described above.
3.5. HPLC Analysis
The substrate concentrations were measured using a Perkin Elmer HPLC (Hopkinton, MA, USA) equipped with a reversed-phase C-18 column (Genesis, 5 µm), a binary pump system, and a 20 µL injection loop, and eluted using an pH 2 acetonitrile–phosphate buffer (70:30 v/v) solvent mixture at 0.7 mL/min. Substrate detection was carried out using UV-vis detection. The wavelengths used for detection are listed in Table 1.
3.6. Product Extraction
To obtain sufficient amounts of the oxidation products of the PhCs for their mass spectrometric identification, reactions were carried out at a 50 mL scale using the PhCs at a concentration of 20 µM. The products were extracted three times using methylene chloride (1:1 ratio), and the organic phase was collected and passed over 10 g of sodium sulfate to remove the excess water. Finally, the reaction products were obtained after the removal of methylene chloride on a rotary evaporator.
3.7. Mass Spectrometry Analysis
The reaction products were resuspended in 1 mL of methanol and filtered through a 0.25 μm polytetrafluoroethylene (PTF) filter to remove any impurities. After this, the samples were acidified with 5 μL of 0.01% trifluoroacetic acid and analyzed in a micrOTOF-QII spectrometer (Bruker Daltonics, Billerica, MA, USA) using DIESI-MS in positive or negative mode. A constant volumetric flow rate (8 μL·min−1) was achieved using a 74900-00-05 Cole Palmer syringe pump (Billerica, MA, USA) loaded with 100 μL of the sample. The capillary voltage was set to 2700 V, and nitrogen was used as the drying and nebulizing gas, with a flow rate of 4 L min−1 (0.4 bar) and a gas temperature of 180 °C. Continuous spectra were collected in the m/z range of 50−1500, with a total run duration of 1 min, a scan time of 10 s, and an interscan time of 0.1 s, producing six spectra per sample.
The mass spectrometer was operated at a resolution of 11,000 (FWHM) at mass 301.9981 m/z in positive ion mode at a capillary voltage of 2700 V (positive). The spectrometer was calibrated with an ESI-TOF tuning mix calibrant (Sigma-Aldrich, Toluca, Mexico).
Finally, precursor ion scans (MS/MS) were performed using positive electrospray ionization (ESI+) with an appropriate set mass. Based on the obtained pattern, suitable fragments were analyzed using Bruker Compass DataAnalysis 4.0 (Bruker Daltonics), which provided a list of possible elemental formulas using Generate Molecular Formula Editor, as well as a sophisticated comparison of the theoretical and measured isotope pattern (σ value) for increased confidence in the suggested molecular formula (Bruker Daltonics Technical Note 008, 2004). The widely accepted accuracy threshold for the confirmation of elemental compositions was set as 5 ppm.
3.8. Enzymatic Oxidation of PhCs in Simulated Treated Water
The oxidative capability of CPO was determined for all the PhCs in a simulated wastewater effluent, produced from the secondary treatment stage of the municipal wastewater treatment plant of Puebla City, Mexico. The samples taken from this effluent were filtered to remove particulate matter and suspended solids, and then stored at 4 °C until their use. The wastewater sample was analyzed according to standard methods, and its main characteristics are summarized in Table S1 of the supplementary material. The water samples were spiked with 20 µM of each PhC, and the transformation was assayed as described for the model systems. Three replicate experiments were performed for all samples.
3.9. Biodegradability Determination
The ratio BOD28/ThOD was adopted as the biodegradability index. The evaluation of BOD values at 28 days were performed in BOD bottles with glass stoppers, following standard procedures for Winkler titration [48]. The inoculum was derived from the surface water of the Nexapa River in the state of Puebla, Mexico.
3.10. Raman Measurements
For the Raman analysis of the unmodified and enzyme-modified chitosan pellets, a Micro-Raman system (Xplora Plus microscope from Horiba, Kyoto, Japan) equipped with a confocal microscope (Olympus BX51, Rochester, NY, USA) and a laser excitation at 785 nm were used. The spectra were obtained using a 1200 gr·mm−1 grating with 10 s of acquisition time, and an accumulation of two spectra. The laser power was 15 W·mm−2.
Supplementary Materials
The following are available online at www.mdpi.com/2073-4344/8/1/32/s1, Figure S1: CPO-chitosan macrospheres prepared by precipitation and cross-linking method (scale bar 1 mm), Table S1: Partial physicochemical characterization of treated water from municipal treatment plant from Puebla State, Table S2: Oxidation products of the CPO enzymatic oxidation, Table S3: Toxicity test, containing of both the PhC (before and after enzymatic transformation).
Acknowledgments
This work was supported by PRODEP (Grant DSA/103.5/14/10819).
Author Contributions
José Luis García-Zamora and Karina León-Aguirre carried out the experiments about enzyme kinetics; René Quiroz-Morales and Georgette Rebollar-Pérez performed the biodegradability experiments, and also Rebollar-Pérez wrote the manuscript. Mayra Beatriz Gómez-Patiño and Daniel Arrieta-Baez performed the MS experiments and analyzed the data for product identification. Roberto Parra an Eduardo Torres conceived the experiments, analyzed all the data, and revised the manuscript.
Conflicts of Interest
The authors declare no conflict of interest. The founding sponsors had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, and in the decision to publish the results.
References
- Tijani, J.O.; Fatoba, O.O.; Babajide, O.O.; Petrik, L.F. Pharmaceuticals, endocrine disruptors, personal care products, nanomaterials and perfluorinated pollutants: A review. Environ. Chem. Lett. 2016, 14, 27–49. [Google Scholar] [CrossRef]
- Pal, A.; Gin, K.Y.; Lin, A.Y.; Reinhard, M. Impacts of emerging organic contaminants on freshwater resources: Review of recent occurrences, sources, fate and effects. Sci. Total Environ. 2010, 408, 6062–6069. [Google Scholar] [CrossRef] [PubMed]
- Fatta-Kassinos, D.; Meric, S.; Nikolaou, A. Pharmaceutical residues in environmental waters and wastewater: Current state of knowledge and future research. Anal. Bioanal. Chem. 2011, 399, 251–275. [Google Scholar] [CrossRef] [PubMed]
- Escher, B.I.; Baumgartner, R.; Koller, M.; Treyer, K.; Lienert, J.; McArdell, C.S. Environmental toxicology and risk assessment of pharmaceuticals from hospital wastewater. Water Res. 2011, 45, 75–92. [Google Scholar] [CrossRef] [PubMed]
- Fent, K.; Weston, A.A.; Caminada, D. Ecotoxicology of human pharmaceuticals. Aquat. Toxicol. 2006, 76, 122–159. [Google Scholar] [CrossRef] [PubMed]
- Batt, A.L.; Kim, S.; Aga, D.S. Comparison of the occurrence of antibiotics in four full-scale wastewater treatment plants with varying designs and operations. Chemosphere 2007, 68, 428–435. [Google Scholar] [CrossRef] [PubMed]
- Kümmerer, K. Antibiotics in the aquatic environment—A review—Part I. Chemosphere 2009, 75, 417–434. [Google Scholar] [CrossRef] [PubMed]
- Verlicchi, P.; Al Aukidy, M.; Zambello, E. Occurrence of pharmaceutical compounds in urban wastewater: Removal, mass load and environmental risk after a secondary treatment—A review. Sci. Total Environ. 2012, 429, 123–155. [Google Scholar] [CrossRef] [PubMed]
- Méndez, E.; González-Fuentes, M.A.; Rebollar-Perez, G.; Méndez-Albores, A.; Torres, E. Emerging pollutant treatments in wastewater: Cases of antibiotics and hormones. J. Environ. Sci. Health Part A 2017, 52, 235–253. [Google Scholar] [CrossRef] [PubMed]
- Rivera-Utrilla, J.; Sánchez-Polo, M.; Ferro-García, M.Á.; Prados-Joya, G.; Ocampo-Pérez, R. Pharmaceuticals as emerging contaminants and their removal from water. A review. Chemosphere 2013, 93, 1268–1287. [Google Scholar] [CrossRef] [PubMed]
- Ncibi, M.C.; Mahjoub, B.; Mahjoub, O.; Sillanpää, M. Remediation of Emerging Pollutants in Contaminated Wastewater and Aquatic Environments: Biomass-Based Technologies. CLEAN—Soil Air Water 2017, 45. [Google Scholar] [CrossRef]
- Wu, W.; Huang, Z.H.; Lim, T.T. Recent development of mixed metal oxide anodes for electrochemical oxidation of organic pollutants in water. Appl. Catal. A Gen. 2014, 480, 58–78. [Google Scholar] [CrossRef]
- Eibes, G.; Debernardi, G.; Feijoo, G.; Moreira, M.T.; Lema, J.M. Oxidation of pharmaceutically active compounds by a ligninolytic fungal peroxidase. Biodegradation 2011, 22, 539–550. [Google Scholar] [CrossRef] [PubMed]
- Majeau, J.A.; Brar, S.K.; Tyagi, R.D. Laccases for removal of recalcitrant and emerging pollutants. Bioresour. Technol. 2010, 101, 2331–2350. [Google Scholar] [CrossRef] [PubMed]
- Taboada-Puig, R.; Eibes, G.; Lloret, L.; Lú-Chau, T.A.; Feijoo, G.; Moreira, M.T.; Lema, J.M. Fostering the action of versatile peroxidase as a highly efficient biocatalyst for the removal of endocrine disrupting compounds. New Biotechnol. 2016, 33, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Torres-Duarte, C.; Viana, M.T.; Vazquez-Duhalt, R. Laccase-Mediated Transformations of Endocrine Disrupting Chemicals Abolish Binding Affinities to Estrogen Receptors and Their Estrogenic Activity in Zebrafish. Appl. Biochem. Biotechnol. 2012, 168, 864–876. [Google Scholar] [CrossRef] [PubMed]
- Torres-Duarte, C.; Vazquez-Duhalt, R. Applications and Prospective of Peroxidase Biocatalysis in the Environmental Field. In Biocatalysis Based on Heme Peroxidases; Torres, E., Ayala, M., Eds.; Springer: Berlin, Germany, 2010. [Google Scholar]
- Garcia-Morales, R.; Rodríguez-Delgado, M.; Gomez-Mariscal, K.; Orona-Navar, C.; Hernandez-Luna, C.; Torres, E.; Parra, R.; Cárdenas-Chávez, D.; Mahlknecht, J.; Ornelas-Soto, N. Biotransformation of Endocrine-Disrupting Compounds in Groundwater: Bisphenol A, Nonylphenol, Ethynylestradiol and Triclosan by a Laccase Cocktail from Pycnoporus sanguineus CS43. Water Air Soil Pollut. 2015, 226, 251. [Google Scholar] [CrossRef] [PubMed]
- Torres, E.; Bustos-Jaimes, I.; le Borgne, S. Potential use of oxidative enzymes for the detoxification of organic pollutants. Appl. Catal. B Environ. 2003, 46, 1–15. [Google Scholar] [CrossRef]
- Murugesan, K.; Chang, Y.Y.; Kim, Y.M.; Jeon, J.R.; Kim, E.J.; Chang, Y.S. Enhanced transformation of triclosan by laccase in the presence of redox mediators. Water Res. 2010, 44, 298–308. [Google Scholar] [CrossRef] [PubMed]
- Lloret, L.; Eibes, G.; Lú-Chau, T.A.; Moreira, M.T.; Feijoo, G.; Lema, J.M. Laccase-catalyzed degradation of anti-inflammatories and estrogens. Biochem. Eng. J. 2010, 51, 124–131. [Google Scholar] [CrossRef]
- Wen, X.; Jia, Y.; Li, J. Degradation of tetracycline and oxytetracycline by crude lignin peroxidase prepared from Phanerochaete chrysosporium—A white rot fungus. Chemosphere 2009, 75, 1003–1007. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, K.; Hirai, H.; Murata, H.; Nishida, T. Removal of estrogenic activities of 17β-estradiol and ethinylestradiol by ligninolytic enzymes from white rot fungi. Water Res. 2003, 37, 1972–1975. [Google Scholar] [CrossRef]
- Montiel, C.; Terrés, E.; Domínguez, J.M.; Aburto, J. Immobilization of chloroperoxidase on silica-based materials for 4,6-dimethyl dibenzothiophene oxidation. J. Mol. Catal. B Enzym. 2007, 48, 90–98. [Google Scholar] [CrossRef]
- Takahashi, H.; Li, B.; Sasaki, T.; Miyazaki, C.; Kajino, T.; Inagaki, S. Catalytic activity in organic solvents and stability of immobilized enzymes depend on the pore size and surface characteristics of mesoporous silica. Chem. Mater. 2000, 12, 3301–3305. [Google Scholar] [CrossRef]
- Pešić, M.; López, C.; Álvaro, G.; López-Santín, J. A novel immobilized chloroperoxidase biocatalyst with improved stability for the oxidation of amino alcohols to amino aldehydes. J. Mol. Catal. B Enzym. 2012, 84, 144–151. [Google Scholar] [CrossRef]
- Hernandez, J.; Robledo, N.R.; Velasco, L.; Quintero, R.; Pickard, M.A.; Vazquez-Duhalt, R. Chloroperoxidase-Mediated Oxidation of Organophosphorus Pesticides. Pestic. Biochem. Physiol. 1998, 61, 87–94. [Google Scholar] [CrossRef]
- Vázquez-Duhalt, R.; Ayala, M.; Márquez-Rocha, F.J. Biocatalytic chlorination of aromatic hydrocarbons by chloroperoxidase of Caldariomyces fumago. Phytochemistry 2001, 58, 929–933. [Google Scholar] [CrossRef]
- Guerrero, E.; Aburto, P.; Terrés, E.; Villegas, O.; González, E.; Zayas, T.; Hernández, F.; Torres, E. Improvement of catalytic efficiency of chloroperoxidase by its covalent immobilization on SBA-15 for azo dye oxidation. J. Porous Mater. 2013, 20, 387–396. [Google Scholar] [CrossRef]
- Ayala, M.; Robledo, N.R.; Lopez-Munguia, A.; Vazquez-Duhalt, R. Substrate Specificity and Ionization Potential in Chloroperoxidase-Catalyzed Oxidation of Diesel Fuel. Environ. Sci. Technol. 2000, 34, 2804–2809. [Google Scholar] [CrossRef]
- Correa-Basurto, J.; Aburto, J.; Trujillo-Ferrara, J.; Torres, E. Ligand recognition by chloroperoxidase using molecular interaction fields and quantum chemistry calculations. Mol. Simul. 2007, 33, 649–654. [Google Scholar] [CrossRef]
- Singer, A.C.; Shaw, H.; Rhodes, V.; Hart, A. Review of Antimicrobial Resistance in the Environment and Its Relevance to Environmental Regulators. Front. Microbiol. 2016, 7, 1728. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, E.M.; Hickey, R.; Hsu, T.; Betancourt Román, C.M.; Chen, J.; Schwager, R.; Kline, J.; Brown, G.Z.; Halden, R.U.; Huttenhower, C.; et al. Antimicrobial Chemicals Are Associated with Elevated Antibiotic Resistance Genes in the Indoor Dust Microbiome. Environ. Sci. Technol. 2016, 50, 9807–9815. [Google Scholar] [CrossRef] [PubMed]
- Van Breda, L.K.; Ward, M.P. Evidence of antimicrobial and disinfectant resistance in a remote, isolated wild pig population. Prev. Vet. Med. 2017, 147, 209–212. [Google Scholar] [CrossRef] [PubMed]
- Adeel, M.; Song, X.; Wang, Y.; Francis, D.; Yang, Y. Environmental impact of estrogens on human, animal and plant life: A critical review. Environ. Int. 2017, 99, 107–119. [Google Scholar] [CrossRef] [PubMed]
- Schwaiger, J.; Ferling, H.; Mallow, U.; Wintermayr, H.; Negele, R.D. Toxic effects of the non-steroidal anti-inflammatory drug diclofenac: Part I: Histopathological alterations and bioaccumulation in rainbow trout. Aquat. Toxicol. 2004, 68, 141–150. [Google Scholar] [CrossRef] [PubMed]
- Bácsi, I.; Viktória, B.; Kókai, Z.; Gonda, S.; Novák, Z.; Nagy, S.A.; Vasas, G. Effects of non-steroidal anti-inflammatory drugs on cyanobacteria and algae in laboratory strains and in natural algal assemblages. Environ. Pollut. 2016, 212, 508–518. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, W.; Redshaw, C.H. Evaluation of biological endpoints in crop plants after exposure to non-steroidal anti-inflammatory drugs (NSAIDs): Implications for phytotoxicological assessment of novel contaminants. Ecotoxicol. Environ. Saf. 2015, 112, 212–222. [Google Scholar] [CrossRef] [PubMed]
- Longoria, A.; Tinoco, R.; Vázquez-Duhalt, R. Chloroperoxidase-mediated transformation of highly halogenated monoaromatic compounds. Chemosphere 2008, 72, 485–490. [Google Scholar] [CrossRef] [PubMed]
- Eisenthal, R.; Danson, M.J.; Hough, D.W. Catalytic efficiency and kcat/KM: A useful comparator? Trends Biotechnol. 2007, 25, 247–249. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; He, Q.; Li, H.; Gao, X.; Hu, M.; Li, S.; Zhai, Q.; Jiang, Y.; Wang, X. Bioconversion of non-steroidal anti-inflammatory drugs diclofenac and naproxen by chloroperoxidase. Biochem. Eng. J. 2017, 120, 7–16. [Google Scholar] [CrossRef]
- Zhang, X.; Li, X.; Jiang, Y.; Hu, M.; Li, S.; Zhai, Q. Combination of enzymatic degradation by chloroperoxidase with activated sludge treatment to remove sulfamethoxazole: Performance, and eco-toxicity assessment. J. Chem. Technol. Biotechnol. 2016, 91, 2802–2809. [Google Scholar] [CrossRef]
- Salcedo, K.; Torres-Ramírez, E.; Haces, I.; Ayala, M. Halogenation of β-estradiol by a rationally designed mesoporous biocatalyst based on chloroperoxidase. Biocatalysis 2015, 1, 33–43. [Google Scholar] [CrossRef]
- Glassmeyer, S.T.; Shoemaker, J.A. Effects of Chlorination on the Persistence of Pharmaceuticals in the Environment. Bull. Environ. Contam. Toxicol. 2005, 74, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Dodd, M.C.; Huang, C.H. Transformation of the Antibacterial Agent Sulfamethoxazole in Reactions with Chlorine: Kinetics, Mechanisms, and Pathways. Environ. Sci. Technol. 2004, 38, 5607–5615. [Google Scholar] [CrossRef] [PubMed]
- Bedner, M.; MacCrehan, W.A. Transformation of Acetaminophen by Chlorination Produces the Toxicants 1,4-Benzoquinone and N-Acetyl-p-benzoquinone Imine. Environ. Sci. Technol. 2005, 40, 516–522. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, F.; Liu, H.; Burdette, J.E.; Li, Y.; Overk, C.R.; Pisha, E.; Yao, J.; van Breemen, R.B.; Swanson, S.M.; et al. Effect of Halogenated Substituents on the Metabolism and Estrogenic Effects of the Equine Estrogen, Equilenin. Chem. Res. Toxicol. 2003, 16, 741–749. [Google Scholar] [CrossRef] [PubMed]
- The Organisation for Economic Co-Operation and Development (OECD). Test No. 301: Ready Biodegradability; OECD Publishing: Paris, France, 1992. [Google Scholar]
- Liu, C.; Thormann, E.; Claesson, P.M.; Tyrode, E. Surface Grafted Chitosan Gels. Part II. Gel Formation and Characterization. Langmuir 2014, 30, 8878–8888. [Google Scholar] [CrossRef] [PubMed]
- Migneault, I.; Dartiguenave, C.; Bertrand, M.J.; Waldron, K.C. Glutaraldehyde: Behavior in aqueous solution, reaction with proteins, and application to enzyme crosslinking. Biotechniques 2004, 37, 790–806. [Google Scholar] [PubMed]
- Praveena, S.D.; Ravindrachary, V.; Bhajantri, R.F.; Ismayil. Free volume-related microstructural properties of lithium perchlorate/sodium alginate polymer composites. Polym. Compos. 2014, 35, 1267–1274. [Google Scholar] [CrossRef]
- Boujday, S.; Chapelle, M.L.; Srajer, J.; Knoll, W. Enhanced Vibrational Spectroscopies as Tools for Small Molecule Biosensing. Sensors 2015, 15, 21239–21264. [Google Scholar] [CrossRef] [PubMed]
- Vazquez-Duhalt, R.; Tinoco, R.; D’Antonio, P.; Topoleski, L.T.; Payne, G.F. Enzyme Conjugation to the Polysaccharide Chitosan: Smart Biocatalysts and Biocatalytic Hydrogels. Bioconjug. Chem. 2001, 12, 301–306. [Google Scholar] [CrossRef] [PubMed]
- Aburto, J.; Ayala, M.; Bustos-Jaimes, I.; Montiel, C.; Terrés, E.; Dominguez, J.M.; Torres, E. Stability and catalytic properties of chloroperoxidase immobilized on SBA-16 mesoporous materials. Microporous Mesoporous Mater. 2005, 83, 193–200. [Google Scholar] [CrossRef]
- Rodrigues, R.C.; Ortiz, C.; Berenguer-Murcia, Á.; Torres, R.; Fernández-Lafuente, R. Modifying enzyme activity and selectivity by immobilization. Chem. Soc. Rev. 2013, 42, 6290–6307. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.; Shiller, A.M. Distribution of hydrogen peroxide in the northwest Pacific Ocean. Geochem. Geophys. Geosyst. 2005, 6. [Google Scholar] [CrossRef]
- Chiu, S.H.; Chung, T.W.; Giridhar, R.; Wu, W.T. Immobilization of β-cyclodextrin in chitosan beads for separation of cholesterol from egg yolk. Food Res. Int. 2004, 37, 217–223. [Google Scholar] [CrossRef]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).