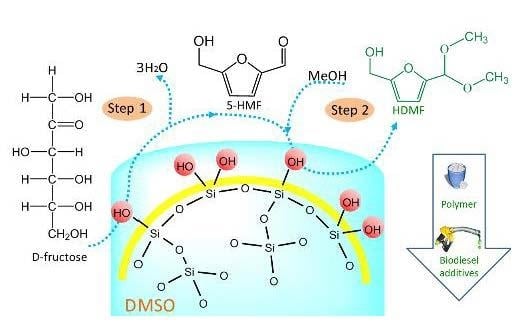
Facile, One-Pot, Two-Step, Strategy for the Production of Potential Bio-Diesel Candidates from Fructose
Abstract
:1. Introduction
2. Results and Discussion
2.1. Production of HDMF in Methanol Solvent
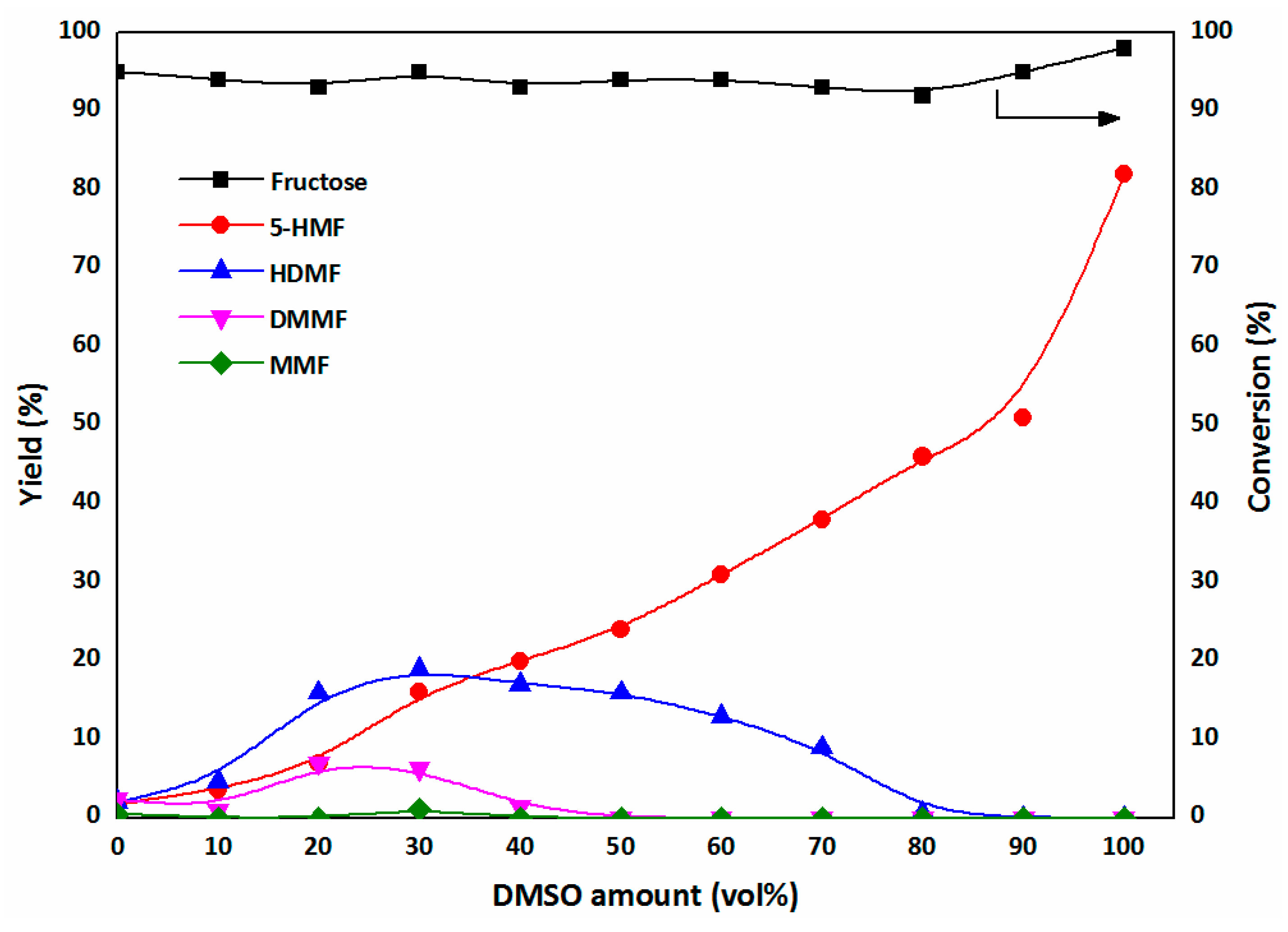
2.2. Production of HDMF in a DMSO/Methanol Biphasic System
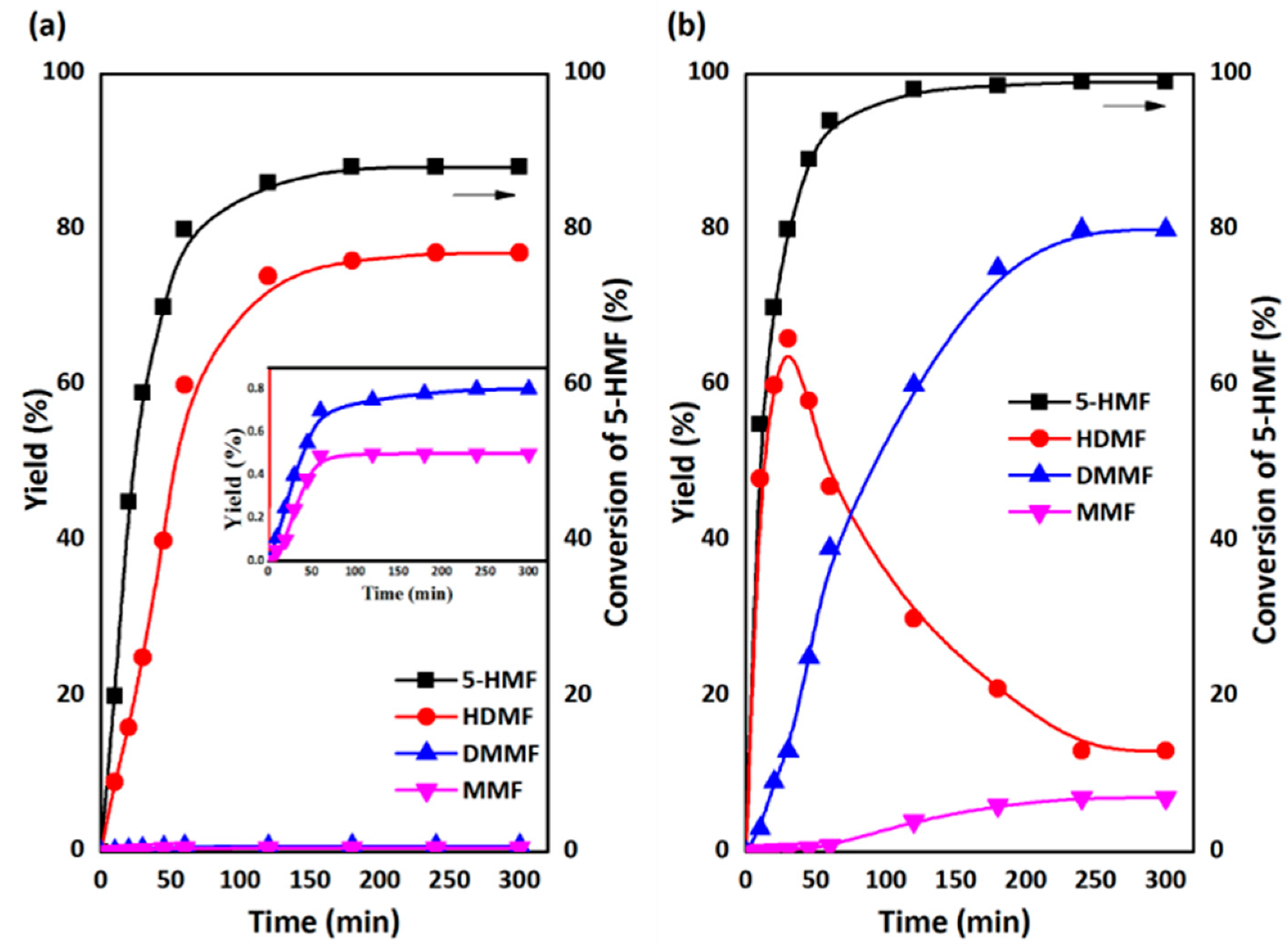
2.3. One-Pot, Two-Step Design for the Conversion of Fructose into HDMF
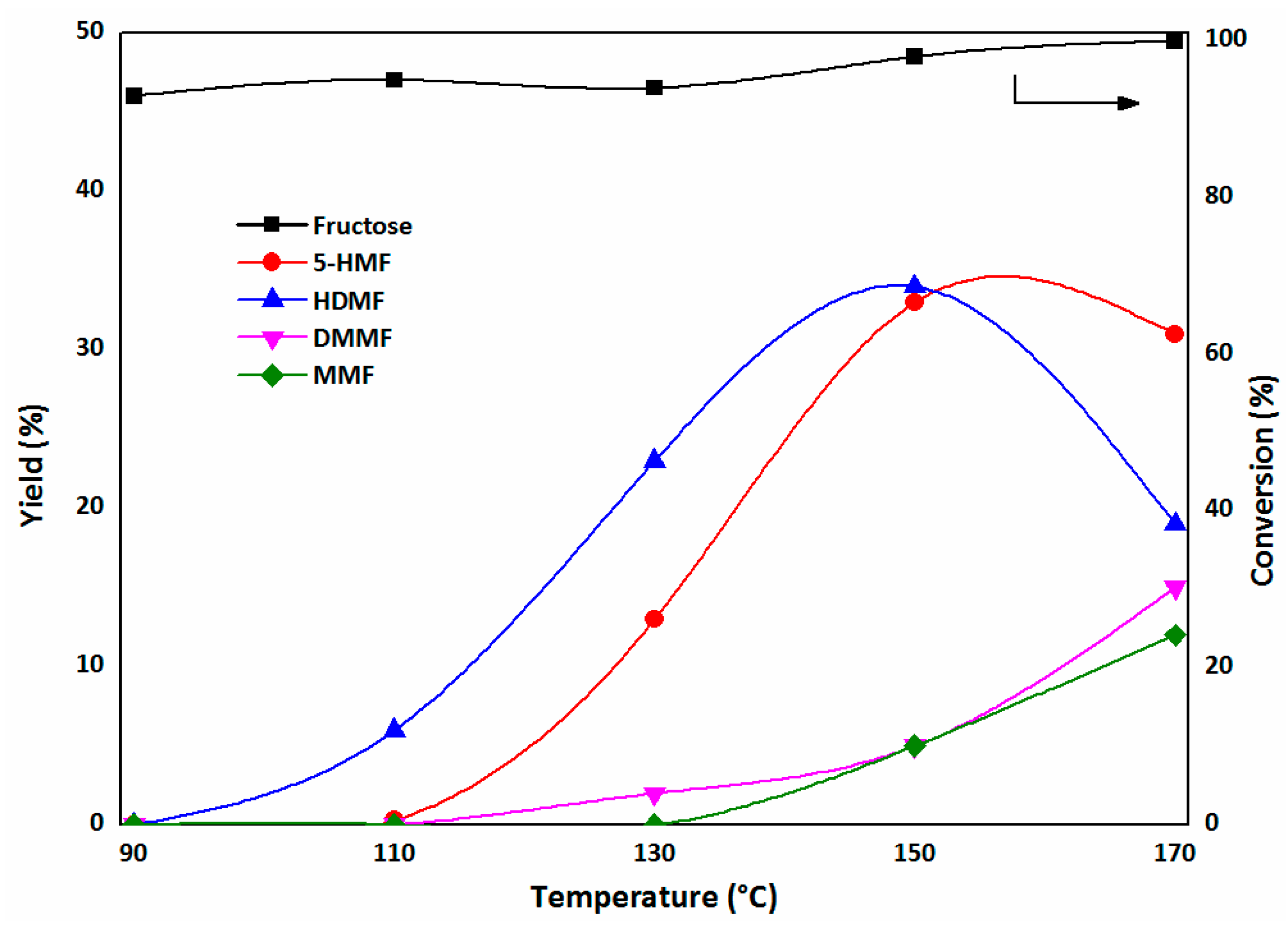
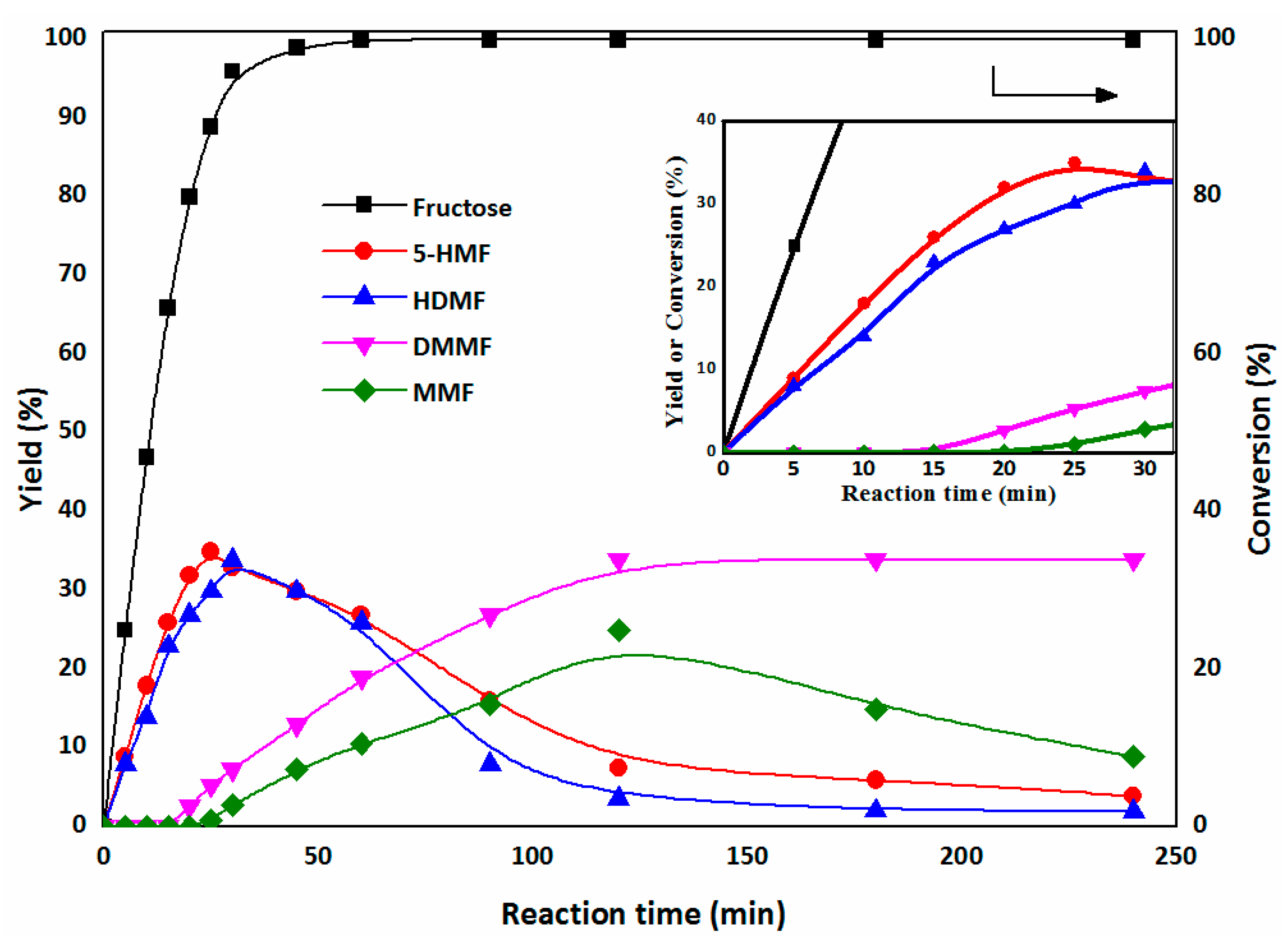
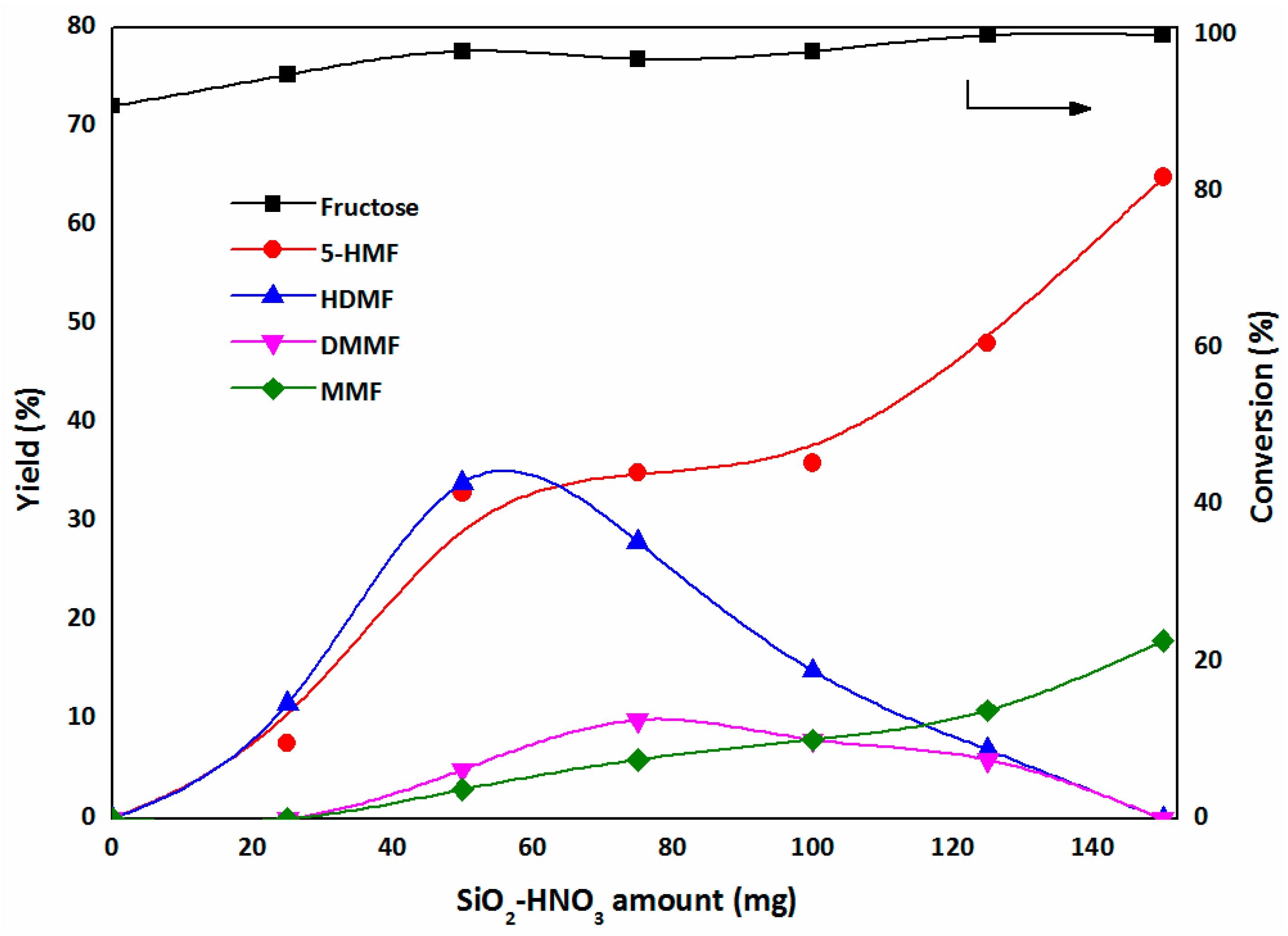
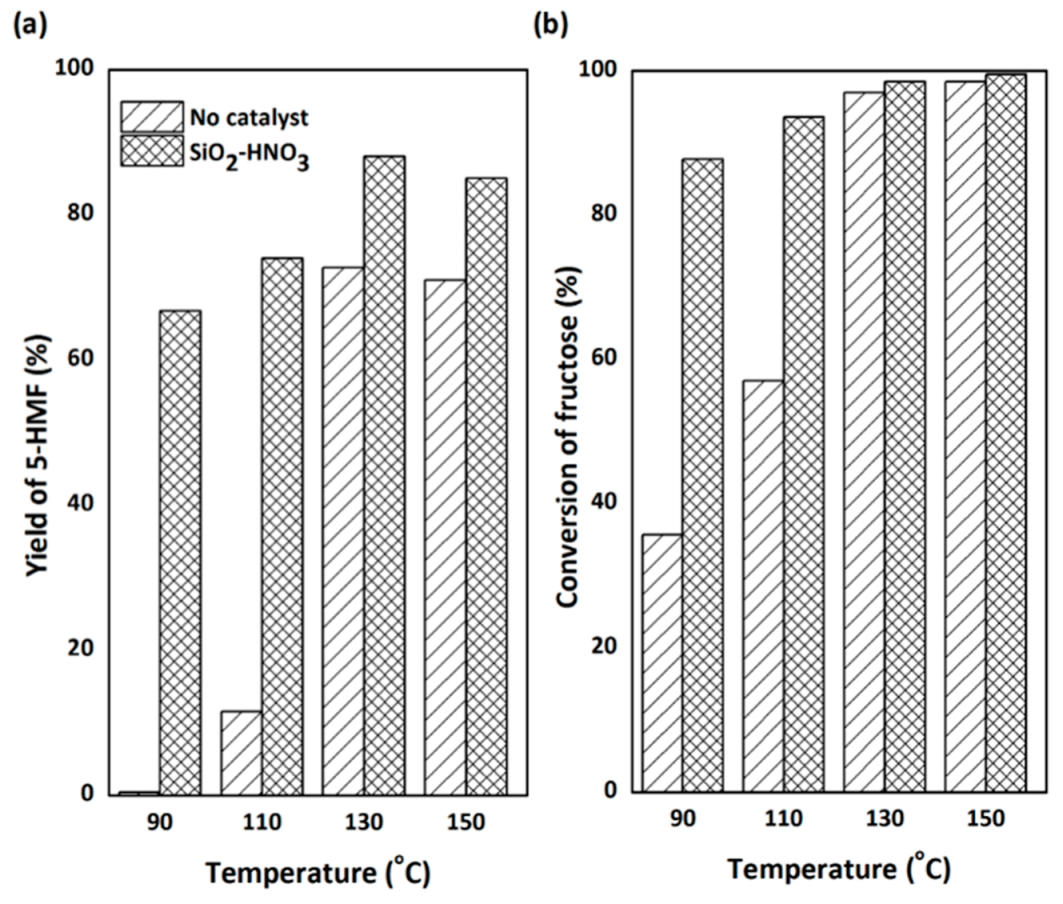
2.3.1. The First Step: Dehydration of Fructose to 5-HMF in DMSO
2.3.2. The Second Step: Acetalization of 5-HMF with the Addition of Methanol
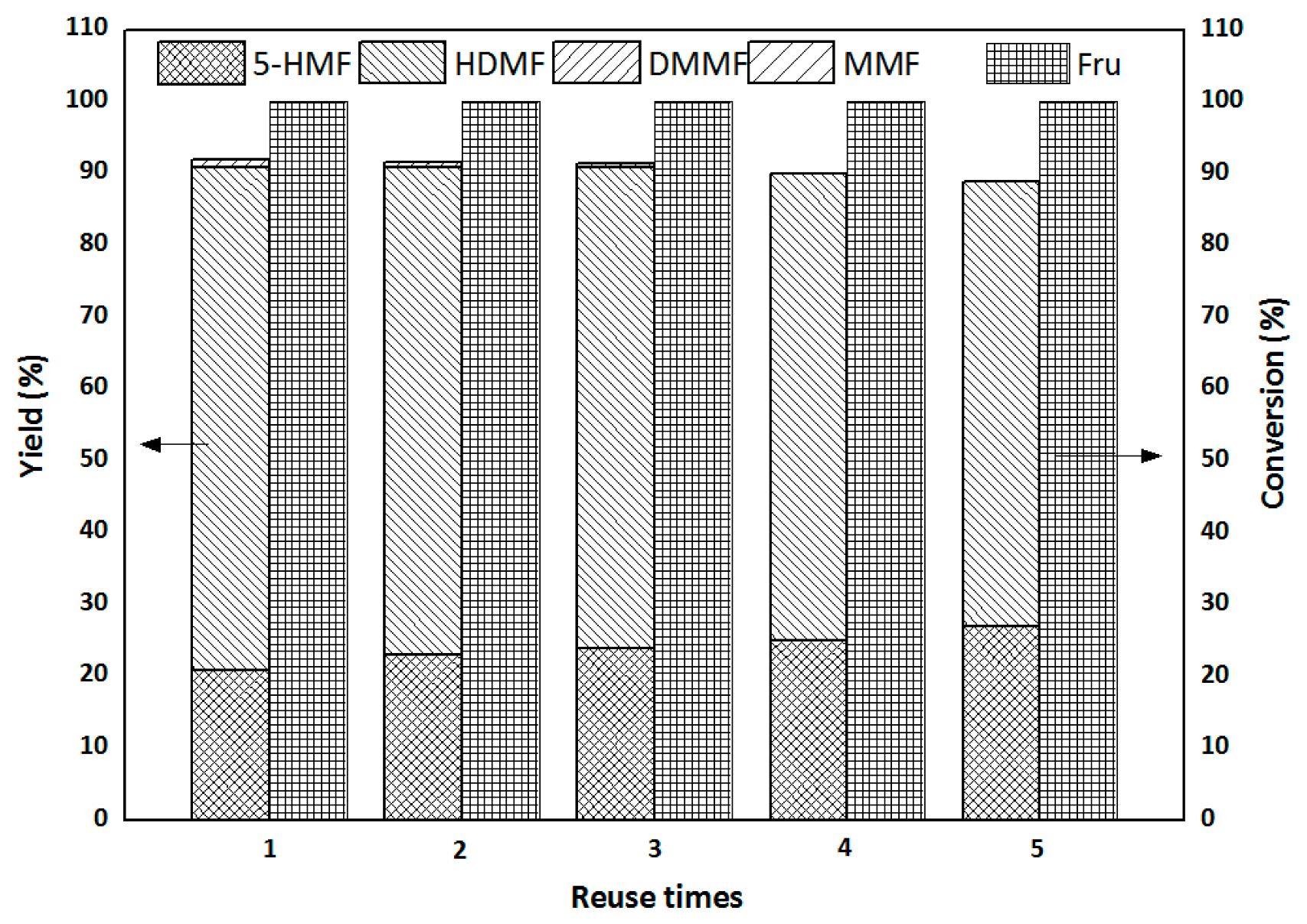
2.3.3. Catalyst Recycling
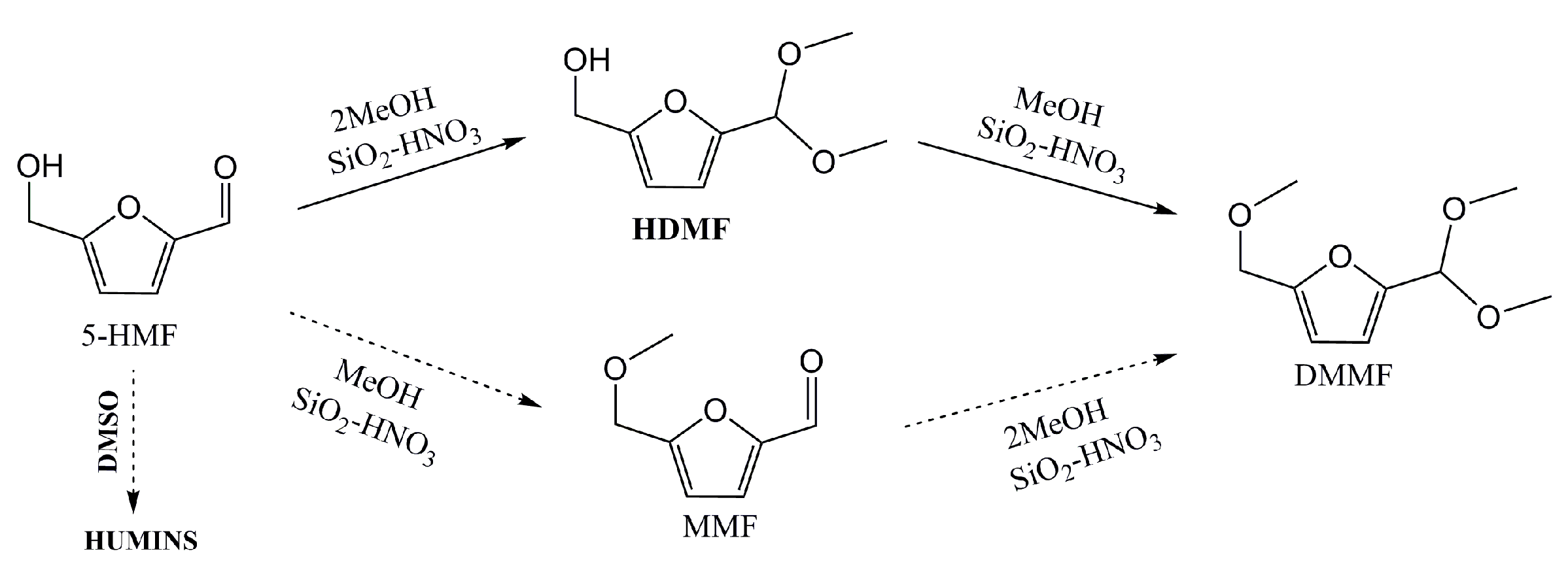
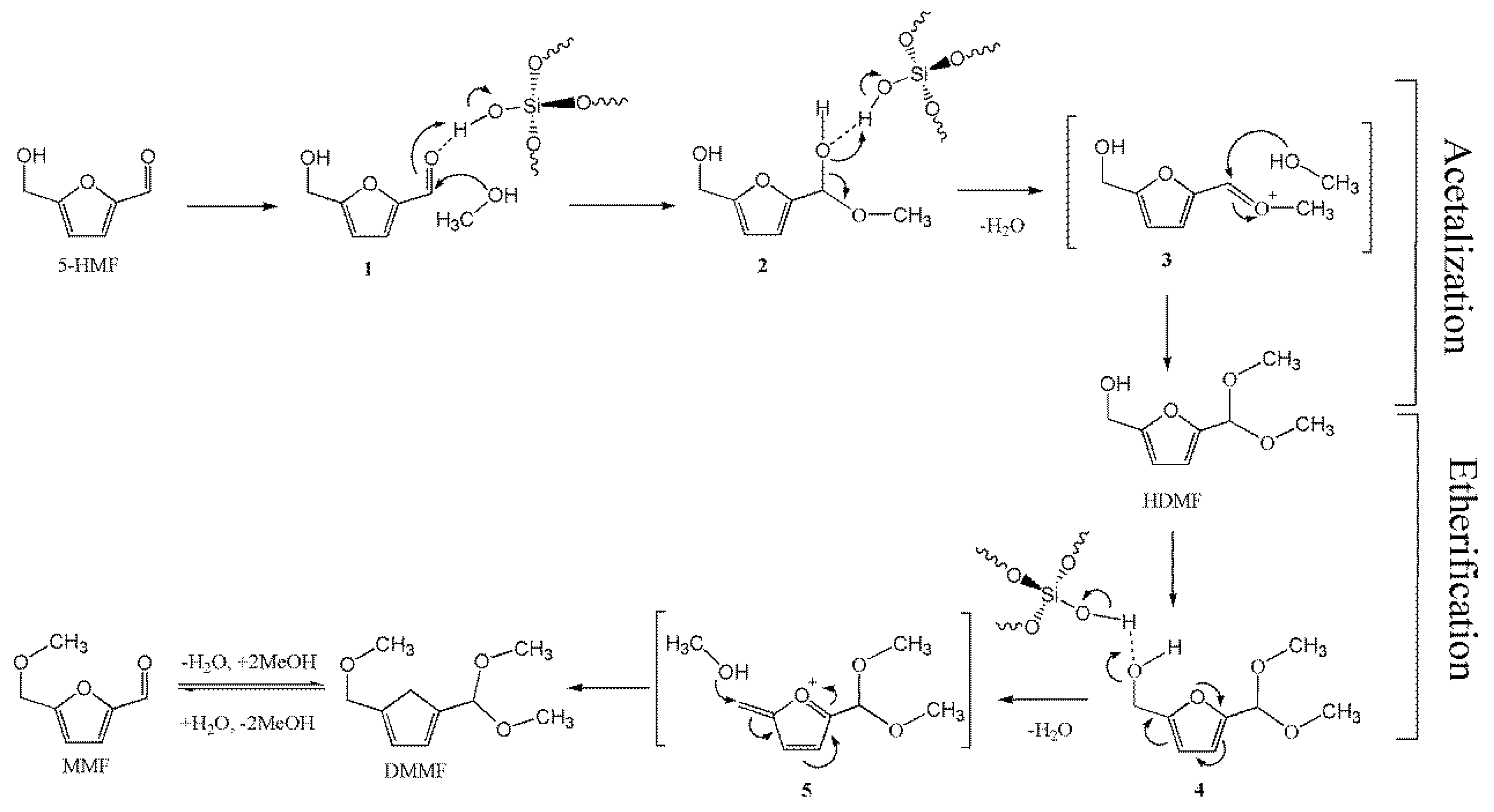
2.4. Proposed Mechanism for the Production of HDMF
3. Experimental Section
3.1. Materials
3.2. Preparation of Catalyst
3.3. General Reaction Procedure
3.4. Analysis Methods
3.5. Calculation of Yield and Conversion
- Conversion = (1-moles of fructose in product/initial moles of fructose) × 100%
- Yield = (moles of product produced/initial moles of fructose) × 100%
- Selectivity = (moles of product produced/moles of fructose converted) × 100%
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Van Putten, R.-J.; van der Waal, J.C.; de Jong, E.; Rasrendra, C.B.; Heeres, H.J.; de Vries, J.G. Hydroxymethylfurfural, a versatile platform chemical made from renewable resources. Chem. Rev. 2013, 113, 1499–1597. [Google Scholar] [CrossRef] [PubMed]
- Baliban, R.C.; Elia, J.A.; Floudas, C.A.; Xiao, X.; Zhang, Z.; Li, J.; Cao, H.; Ma, J.; Qiao, Y.; Hu, X. Thermochemical conversion of duckweed biomass to gasoline, diesel, and jet fuel: Process synthesis and global optimization. Ind. Eng. Chem. Res. 2013, 52, 11436–11450. [Google Scholar] [CrossRef]
- Ranoux, A.; Djanashvili, K.; Arends, I.W.C.E.; Hanefeld, U. 5-Hydroxymethylfurfural synthesis from hexoses is autocatalytic. ACS Catal. 2013, 3, 760–763. [Google Scholar] [CrossRef]
- Roylance, J.J.; Choi, K.S. Electrochemical reductive biomass conversion: Direct conversion of 5-hydroxymethylfurfural (HMF) to 2,5-hexanedione (HD) via reductive ring-opening. Green Chem. 2016, 18, 2956–2960. [Google Scholar] [CrossRef]
- Agirre, I.; Güemez, M.B.; Ugarte, A.; Requies, J.; Barrio, V.L.; Cambra, J.F.; Arias, P.L. Glycerol acetals as diesel additives: Kinetic study of the reaction between glycerol and acetaldehyde. Fuel Process. Technol. 2013, 116, 182–188. [Google Scholar] [CrossRef]
- Cui, W.; Qi, M.; Li, X.; Huang, S.; Zhou, S.; Weng, J. Electrospun fibers of acid-labile biodegradable polymers with acetal groups as potential drug carriers. Int. J. Pharm. 2008, 361, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Balakrishnan, M.; Sacia, E.R.; Bell, A.T. Etherification and reductive etherification of 5-(hydroxymethyl) furfural: 5-(alkoxymethyl) furfurals and 2,5-bis (alkoxymethyl) furans as potential bio-diesel candidates. Green Chem. 2012, 14, 1626. [Google Scholar] [CrossRef]
- Lai, L.; Zhang, Y. The production of 5-hydroxymethylfurfural from fructose in isopropyl alcohol: A green and efficient system. ChemSusChem 2011, 4, 1745–1748. [Google Scholar] [CrossRef] [PubMed]
- Arias, K.S.; Climent, M.J.; Corma, A.; Iborra, S. Biomass-Derived Chemicals: Synthesis of Biodegradable Surfactant Ether Molecules from Hydroxymethylfurfural. ChemSusChem 2014, 7, 210–220. [Google Scholar] [CrossRef] [PubMed]
- Arias, K.S.; Al-Resayes, S.I.; Climent, M.J.; Corma, A.; Iborra, S. From Biomass to Chemicals: Synthesis of Precursors of Biodegradable Surfactants from 5-Hydroxymethylfurfural. ChemSusChem 2013, 6, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Gunawan, R.; Mourant, D.; Lievens, C.; Li, X.; Zhang, S.; Chaiwat, W.; Li, C.-Z. Acid-catalysed reactions between methanol and the bio-oil from the fast pyrolysis of mallee bark. Fuel 2012, 97, 512–522. [Google Scholar] [CrossRef]
- Hu, X.; Li, C.-Z. Levulinic esters from the acid-catalysed reactions of sugars and alcohols as part of a bio-refinery. Green Chem. 2011, 13, 1676. [Google Scholar] [CrossRef]
- Najafi, M.; Rostamian, R.; Rafati, A.A. Chemically modified silica gel with thiol group as an adsorbent for retention of some toxic soft metal ions from water and industrial effluent. Chem. Eng. J. 2011, 168, 426–432. [Google Scholar] [CrossRef]
- Kaur, M.; Sharma, S.; Bedi, P.M.S. Silica supported Brönsted acids as catalyst in organic transformations: A comprehensive review. Chin. J. Catal. 2015, 36, 520–549. [Google Scholar] [CrossRef]
- Xu, S.; Yan, X.; Bu, Q.; Xia, H. Highly efficient conversion of carbohydrates into 5-hydroxymethylfurfural using the bi-functional CrPO4 catalyst. RSC Adv. 2016, 6, 8048–8052. [Google Scholar] [CrossRef]
- Yu, Y.; Hu, C.; Abu-Omar, M.M. Conversion of glucose into furans in the presence of AlCl3 in an ethanol-water solvent system. Bioresour. Technol. 2012, 116, 190–194. [Google Scholar]
- Karinen, R.; Vilonen, K.; Niemelä, M. Biorefining: Heterogeneously catalyzed reactions of carbohydrates for the production of furfural and hydroxymethylfurfural. ChemSusChem 2011, 4, 1002–1016. [Google Scholar] [CrossRef] [PubMed]
- Kimura, H.; Nakahara, M.; Matubayasi, N. Solvent effect on pathways and mechanisms for D-fructose conversion to 5-hydroxymethyl-2-furaldehyde: In situ 13C NMR study. J. Phys. Chem. A 2013, 117, 2102–2113. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Tang, Y.; Wu, K.; Bi, C.; Cui, Q. Conversion of fructose into 5-hydroxymethylfurfural (HMF) and its derivatives promoted by inorganic salt in alcohol. Carbohydr. Res. 2012, 350, 20–24. [Google Scholar] [CrossRef] [PubMed]
- Bicker, M.; Kaiser, D.; Ott, L.; Vogel, H. Dehydration of D-fructose to hydroxymethylfurfural in sub-and supercritical fluids. J. Supercrit. Fluids 2005, 36, 118–126. [Google Scholar] [CrossRef]
- Shimizu, K.I.; Uozumi, R.; Satsuma, A. Enhanced production of hydroxymethylfurfural from fructose with solid acid catalysts by simple water removal methods. Catal. Commun. 2009, 10, 1849–1853. [Google Scholar] [CrossRef]
- Wang, H.; Deng, T.; Wang, Y.; Qi, Y.; Hou, X.; Zhu, Y. Efficient catalytic system for the conversion of fructose into 5-ethoxymethylfurfural. Bioresour. Technol. 2013, 136, 394–400. [Google Scholar] [CrossRef] [PubMed]
- Qu, Y.; Huang, C.; Song, Y.; Zhang, J.; Chen, B. Efficient dehydration of glucose to 5-hydroxymethylfurfural catalyzed by the ionic liquid, 1-hydroxyethyl-3-methylimidazolium tetrafluoroborate. Bioresour. Technol. 2012, 121, 462–466. [Google Scholar] [CrossRef] [PubMed]
- Rosatella, A.A.; Simeonov, S.P.; Frade, R.F.M.; Afonso, C.A.M. 5-Hydroxymethylfurfural (HMF) as a building block platform: Biological properties, synthesis and synthetic applications. Green Chem. 2011, 13, 754–793. [Google Scholar] [CrossRef]
- Laugel, C.; Estrine, B.; Le Bras, J.; Hoffmann, N.; Marinkovic, S.; Muzart, J. NaBr/DMSO-Induced Synthesis of 2,5-Diformylfuran from Fructose or 5-(Hydroxymethyl) furfural. ChemCatChem 2014, 6, 1195–1198. [Google Scholar] [CrossRef]
- Despax, S.; Maurer, C.; Estrine, B.; Bras, J.L.; Hoffmann, N.; Marinkovic, S.; Muzart, J. Fast and efficient DMSO-mediated dehydration of carbohydrates into 5-hydroxymethylfurfural. Catal. Commun. 2014, 51, 5–9. [Google Scholar] [CrossRef]
- Mushrif, S.H.; Caratzoulas, S.; Vlachos, D.G. Understanding solvent effects in the selective conversion of fructose to 5-hydroxymethyl-furfural: A molecular dynamics investigation. Phys. Chem. Chem. Phys. 2012, 14, 2637–2644. [Google Scholar] [CrossRef] [PubMed]
- Antal, M.J., Jr.; Mok, W.S.L.; Richards, G.N. Mechanism of formation of 5-(hydroxymethyl)-2-furaldehyde from D-fructose and sucrose. Carbohydr. Res. 1990, 199, 91–109. [Google Scholar] [CrossRef]
- Morales, G.; Melero, J.A.; Paniagua, M.; Iglesias, J.; Hernández, B.; Sanz, M. Sulfonic acid heterogeneous catalysts for dehydration of C 6-monosaccharides to 5-hydroxymethylfurfural in dimethyl sulfoxide. Chin. J. Catal. 2014, 35, 644–655. [Google Scholar] [CrossRef]
- Hu, X.; Lievens, C.; Larcher, A.; Li, C.Z. Reaction pathways of glucose during esterification: Effects of reaction parameters on the formation of humin type polymers. Bioresour. Technol. 2011, 102, 10104–10113. [Google Scholar] [CrossRef] [PubMed]
- Cottier, L.; Descotes, G.; Soro, Y. Synthesis of Acetylated Ranunculin Diastereoisomers and δ-Glucosyloxy-γ-Oxo Esters from α or β Glucosylmethylfurfural. J. Carbohydr. Chem. 2005, 24, 55–71. [Google Scholar] [CrossRef]

| Temp. (°C) | Time (h) | Fructose Conversion (%) | 5-HMF Yield (%) | HDMF Yield (%) | DMMF Yield (%) | MMF Yield (%) |
|---|---|---|---|---|---|---|
| 80 | 4 | 98 | 0 | 1.1 | 0 | 0 |
| 8 | 98 | 0 | 3 | 1 | 0 | |
| 24 | 100 | 2 | 1 | 5 | 1 | |
| 100 | 4 | 99 | 1 | 2 | 4 | 0 |
| 8 | 99 | 2 | 3 | 4 | 0 | |
| 24 | 100 | 3 | 1 | 6 | 6 | |
| 120 | 4 | 99 | 2 | 1.3 | 3 | 4 |
| 8 | 100 | 4 | 1 | 8 | 6 | |
| 24 | 100 | 6 | 2 | 6 | 8 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qiu, G.; Wang, X.; Huang, C.; Li, Y.; Chen, B. Facile, One-Pot, Two-Step, Strategy for the Production of Potential Bio-Diesel Candidates from Fructose. Catalysts 2017, 7, 237. https://doi.org/10.3390/catal7080237
Qiu G, Wang X, Huang C, Li Y, Chen B. Facile, One-Pot, Two-Step, Strategy for the Production of Potential Bio-Diesel Candidates from Fructose. Catalysts. 2017; 7(8):237. https://doi.org/10.3390/catal7080237
Chicago/Turabian StyleQiu, Guo, Xincheng Wang, Chongpin Huang, Yingxia Li, and Biaohua Chen. 2017. "Facile, One-Pot, Two-Step, Strategy for the Production of Potential Bio-Diesel Candidates from Fructose" Catalysts 7, no. 8: 237. https://doi.org/10.3390/catal7080237
APA StyleQiu, G., Wang, X., Huang, C., Li, Y., & Chen, B. (2017). Facile, One-Pot, Two-Step, Strategy for the Production of Potential Bio-Diesel Candidates from Fructose. Catalysts, 7(8), 237. https://doi.org/10.3390/catal7080237