High Catalytic Activity of Heterometallic (Fe6Na7 and Fe6Na6) Cage Silsesquioxanes in Oxidations with Peroxides
Abstract
:1. Introduction
2. Results and Discussion
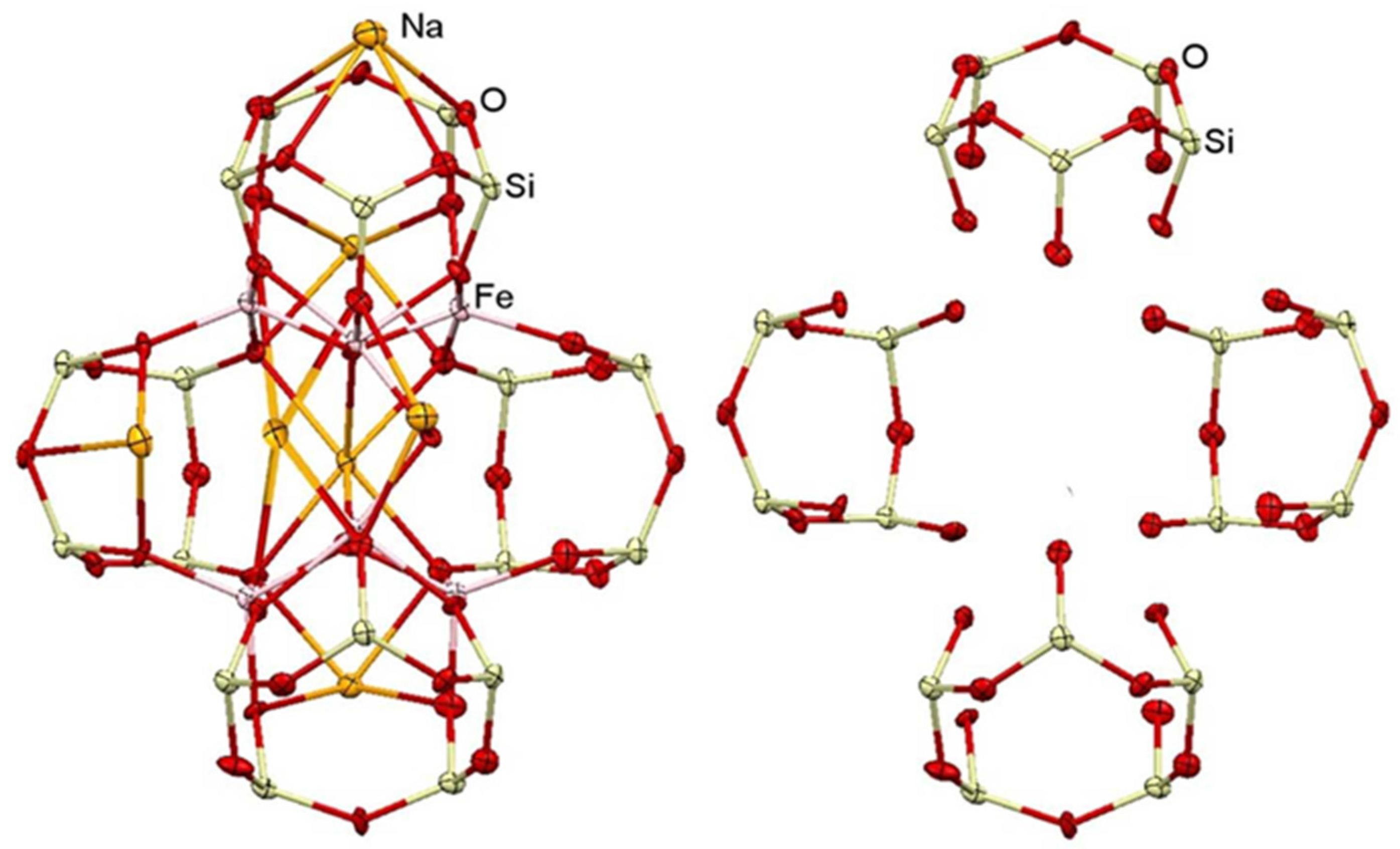
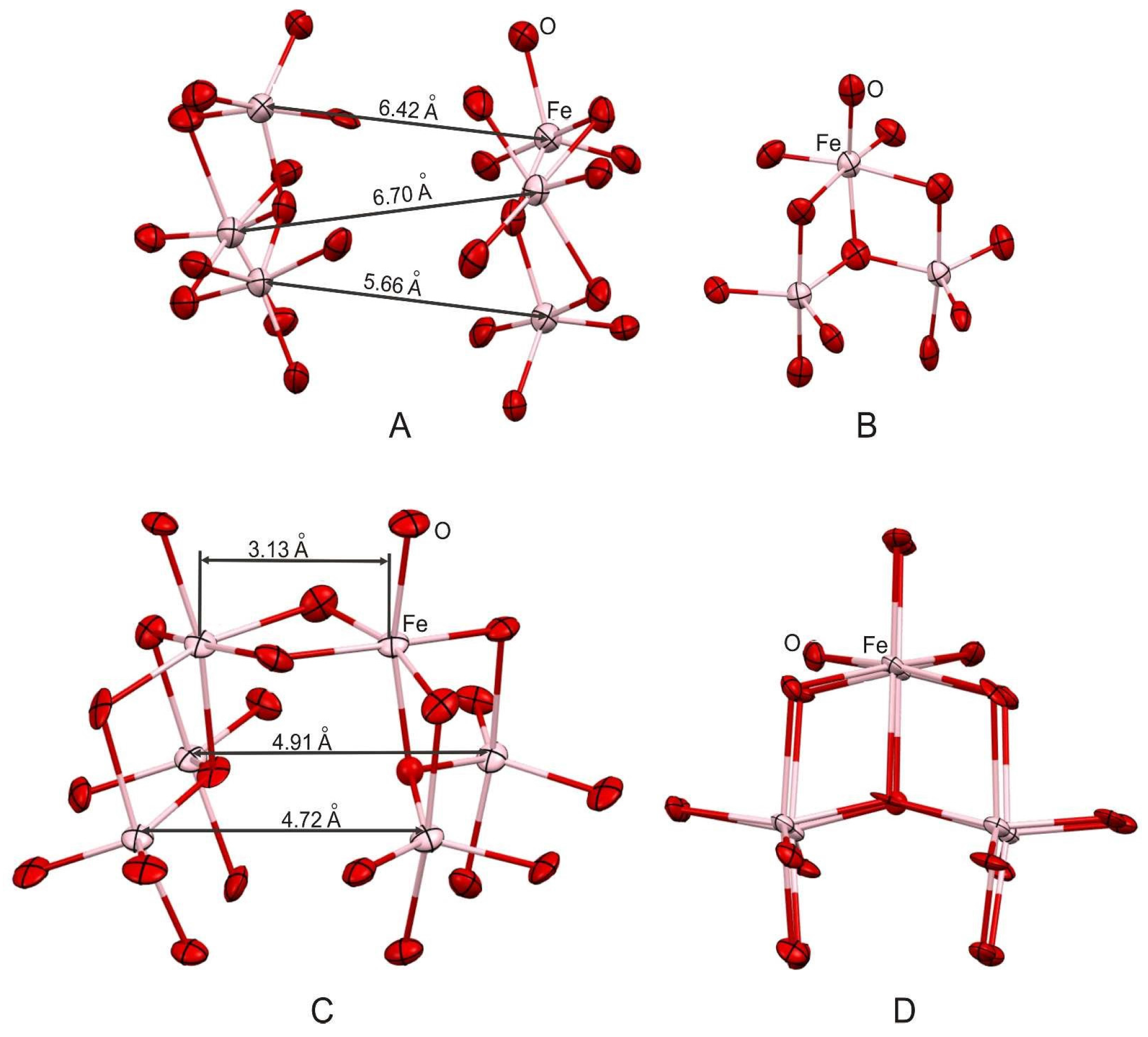
2.1. Syntheses and Structures of Catalysts
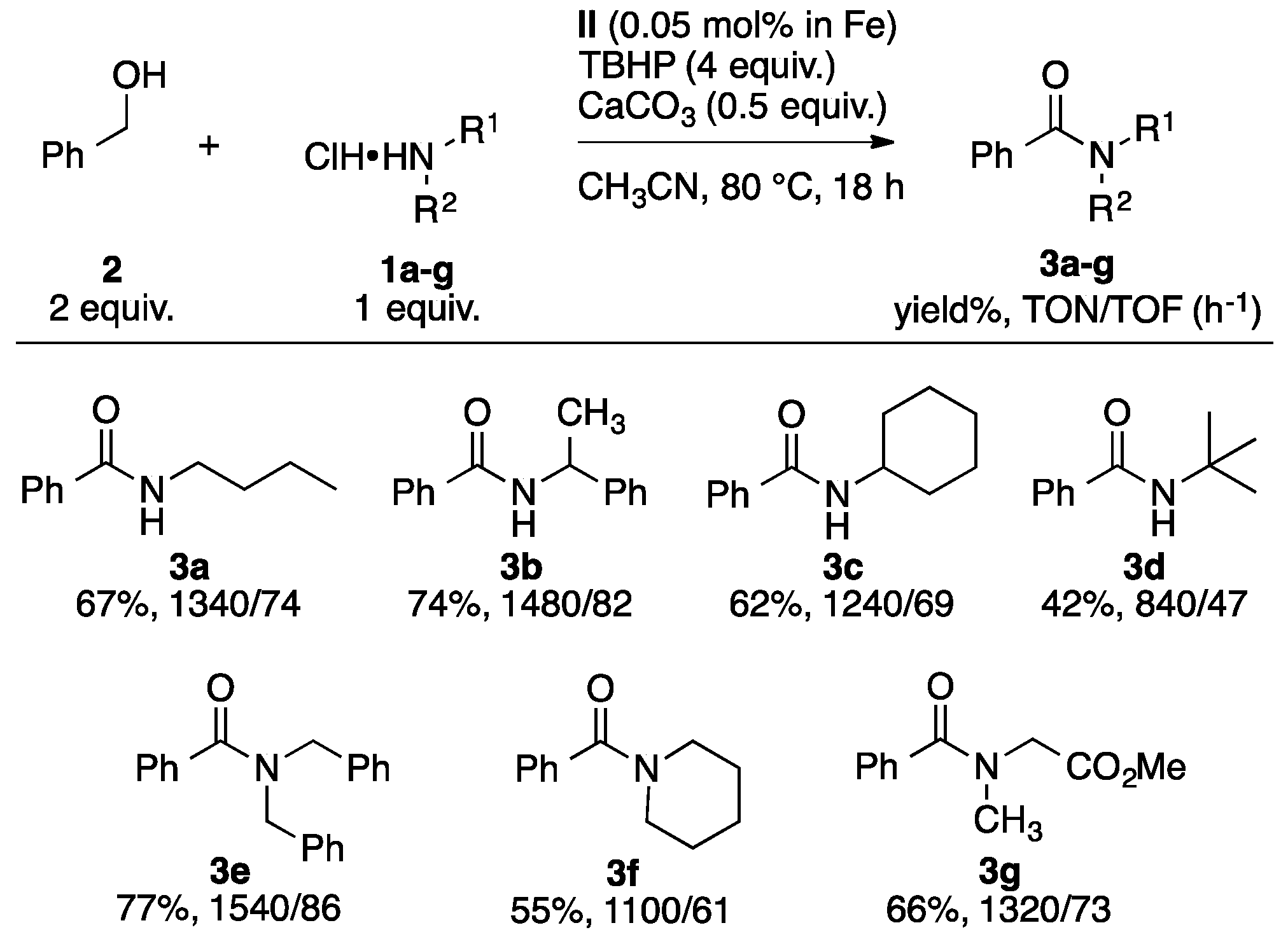
2.2. Catalytic Transformation of Alcohols and Amines into Amides
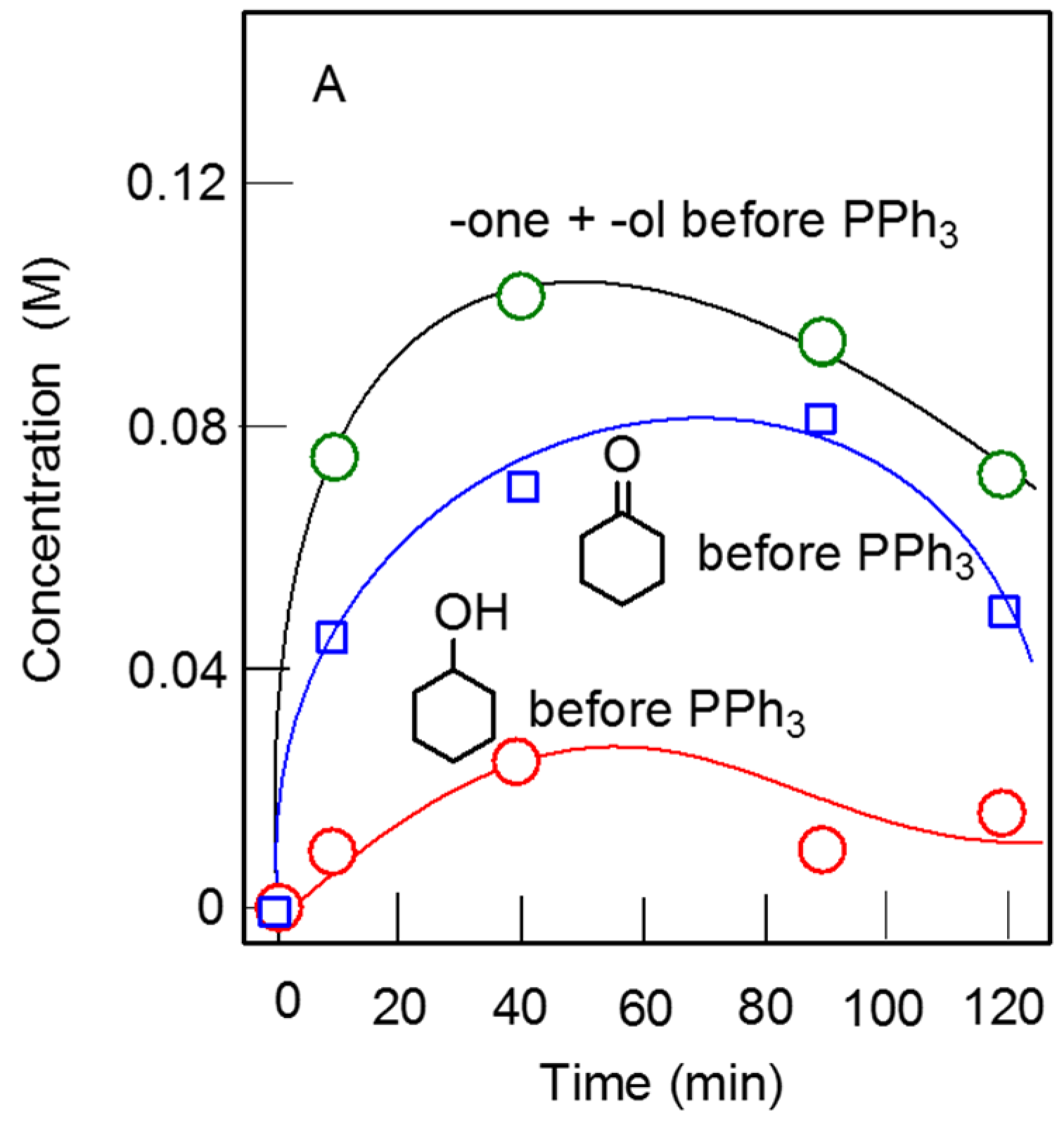
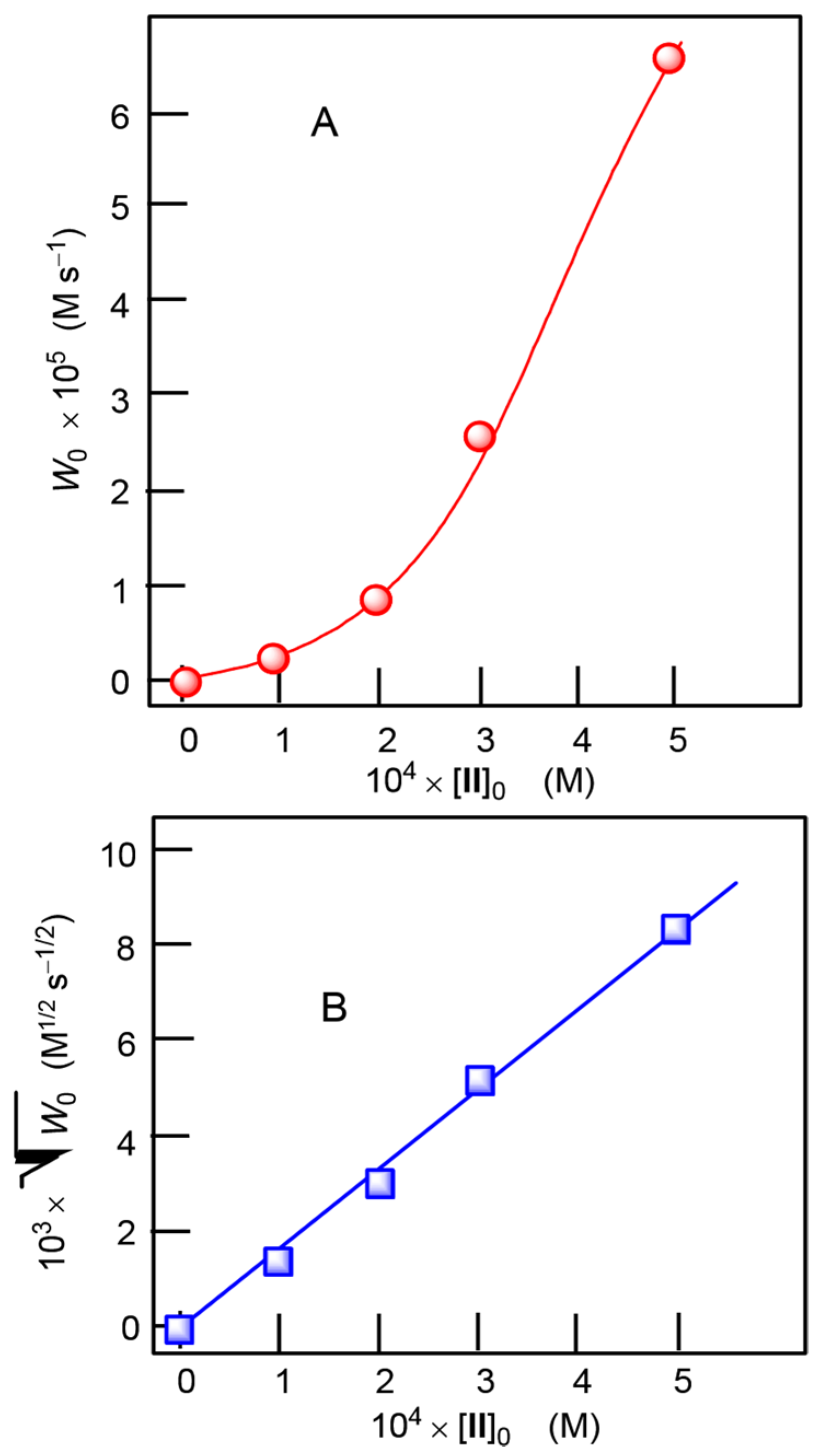
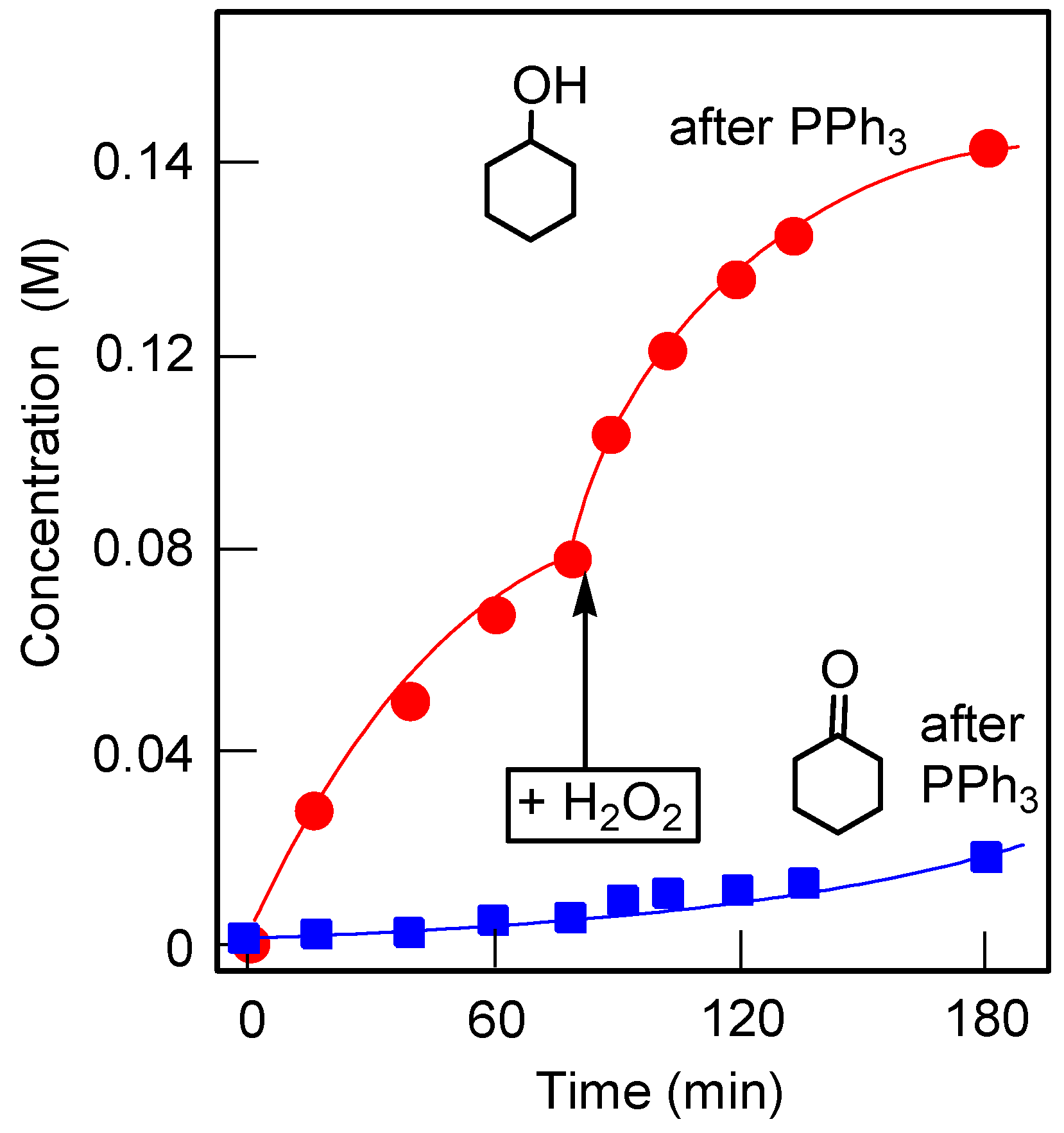
2.3. Catalytic Oxidation of Alcohols and Hydrocarbons
3. Materials and Methods
3.1. Synthesis of Compound I
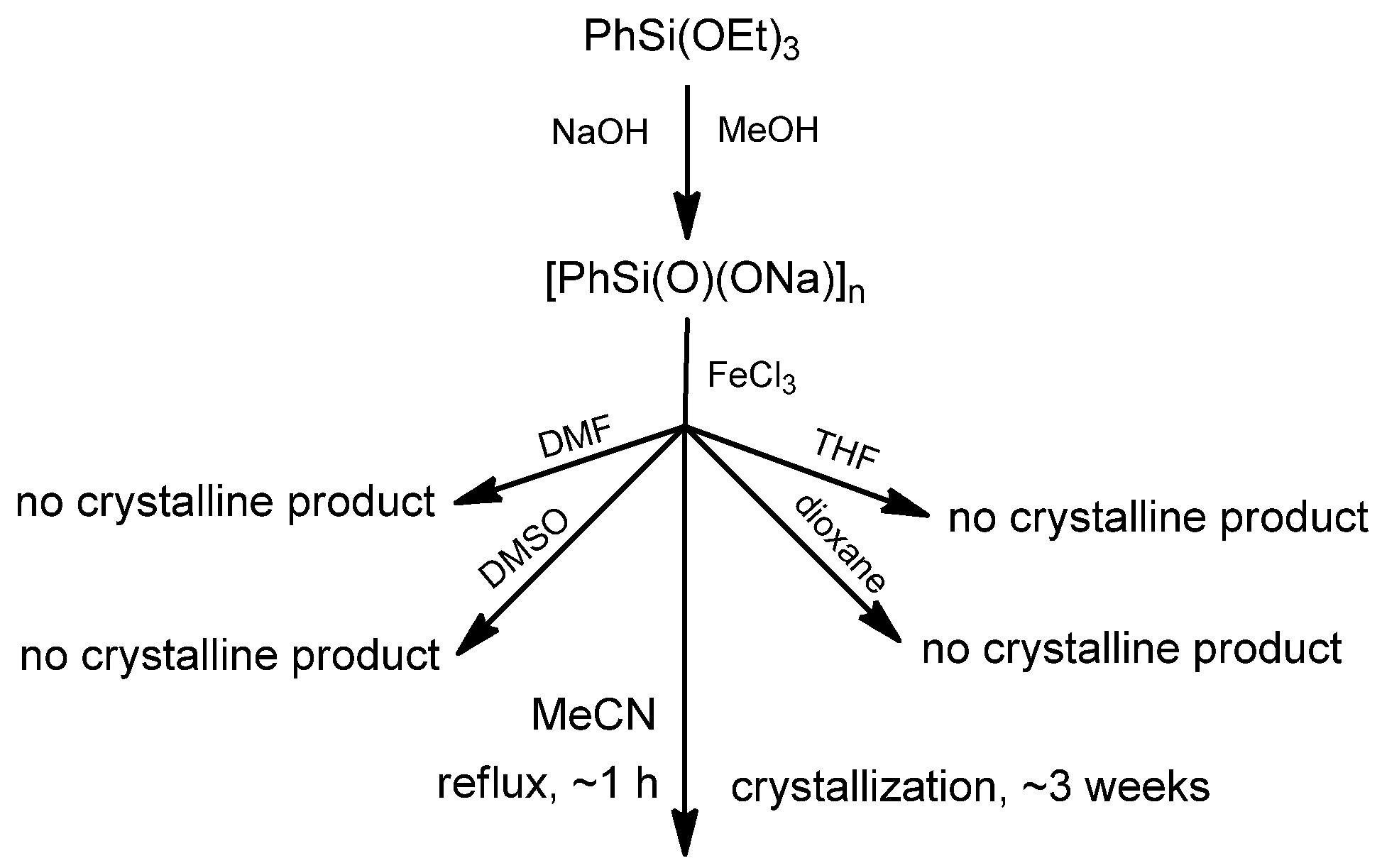
3.2. Synthesis of Compound II
3.3. X-ray Studies
3.4. Oxidation of Alcohols and Hydrocarbons with Peroxides
3.5. Selectivity in the Alkane Oxidations
3.6. General Procedure for Catalytic Amide Formation
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| TON | turnover number |
| TBHP | tert-butylhydroperoxide |
| GC | gas liquid chromatography |
| TFA | trifluoroacetic acid |
References
- Larsen, F.K.; McInnes, E.J.L.; Mkami, H.E.; Overgaard, J.; Piligkos, S.; Rajaraman, G.; Rentschler, E.; Smith, A.A.; Smith, G.M.; Boote, V.; et al. Synthesis and Characterization of Heterometallic {Cr7M} Wheels. Angew. Chem. Int. Ed. 2003, 42, 101–105. [Google Scholar] [CrossRef]
- Saalfrank, R.W.; Prakash, R.; Maid, H.; Hampel, F.; Heinemann, F.W.; Trautwein, A.X.; Bçttger, L.H. Synthesis and Characterization of Metal-Centered, Six-Membered, Mixed-Valent, Heterometallic Wheels of Iron, Manganese, and Indium. Chem. Eur. J. 2006, 12, 2428–2433. [Google Scholar] [CrossRef] [PubMed]
- Moushi, E.E.; Lampropoulos, C.; Wernsdorfer, W.; Nastopoulos, V.; Christou, G.; Tasiopoulos, A.J. A Large [Mn10Na]4 Loop of Four Linked Mn10 Loops. Inorg. Chem. 2007, 46, 3795–3797. [Google Scholar] [CrossRef] [PubMed]
- Milway, V.A.; Tuna, F.; Farrell, A.R.; Sharp, L.E.; Parsons, S.; Murrie, M. Directed Synthesis of {Mn18Cu6} Heterometallic Complexes. Angew. Chem. Int. Ed. 2013, 52, 1949–1952. [Google Scholar] [CrossRef] [PubMed]
- Ojea, M.J.H.; Wilson, C.; Coles, S.J.; Tunac, F.; Murrie, M. Directed synthesis of {CuII2ZnII2} and {CuII8ZnII8} heterometallic complexes. Dalton Trans. 2015, 44, 19275–19281. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Roesky, H.W. Robust and efficient molecular catalysts with a M–O–M′ framework. Dalton Trans. 2007, 1360–1370. [Google Scholar] [CrossRef] [PubMed]
- Mata, J.A.; Hahn, F.E.; Peris, E. Heterometallic complexes, tandem catalysis and catalytic cooperativity. Chem. Sci. 2014, 5, 1723–1732. [Google Scholar] [CrossRef]
- Buchwalter, P.; Rosé, J.; Braunstein, P. Multimetallic Catalysis Based on Heterometallic Complexes and Clusters. Chem. Rev. 2015, 115, 28–126. [Google Scholar] [CrossRef] [PubMed]
- Piquer, L.R.; Sañudo, E.C. Heterometallic 3d–4f single-molecule magnets. Dalton Trans. 2015, 44, 8771–8780. [Google Scholar] [CrossRef] [PubMed]
- Roques, N.; Mugnaini, V.; Veciana, J. Magnetic and porous molecule-based materials. Top. Curr. Chem. 2010, 293, 207–258. [Google Scholar] [PubMed]
- Costantino, F.; Ienco, A.; Taddei, M. Hybrid multifunctional materials based on phosphonates, phosphinates and auxiliary ligands. In Tailored Organic-Inorganic Materials; Brunet, E., Colón, J.L., Clearfield, A., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2015. [Google Scholar]
- Pardo, E.; Ruiz-Garcıa, R.; Cano, J.; Ottenwaelder, X.; Lescouezec, R.; Journaux, Y.; Lloret, F.; Julve, M. Ligand design for multidimensional magnetic materials: A metallosupramolecular perspective. Dalton Trans. 2008, 2780–2805. [Google Scholar] [CrossRef] [PubMed]
- Dul, M.-C.; Pardo, E.; Lescouëzec, R.; Journaux, Y.; Ferrando-Soriac, J.; Ruiz-García, R.; Cano, J.; Julve, M.; Lloret, F.; Cangussu, D.; et al. Supramolecular coordination chemistry of aromatic polyoxalamide ligands: A metallosupramolecular approach toward functional magnetic materials. Coord. Chem. Rev. 2010, 254, 2281–2296. [Google Scholar] [CrossRef]
- Murugavel, R.; Voigt, A.; Walawalkar, M.G.; Roesky, H.W. Hetero- and Metallasiloxanes Derived from Silanediols, Disilanols, Silanetriols, and Trisilanols. Chem. Rev. 1996, 96, 2205–2236. [Google Scholar] [CrossRef] [PubMed]
- Lorenz, V.; Fischer, A.; Gießmann, S.; Gilje, J.W.; Gun’ko, Y.; Jacob, K.; Edelmann, F.T. Disiloxanediolates and polyhedral metallasilsesquioxanes of the early transition metals and f-elements. Coord. Chem. Rev. 2000, 206–207, 321–368. [Google Scholar] [CrossRef]
- Ward, A.J.; Masters, A.F.; Maschmeyer, T. Metallasilsesquioxanes: Molecular Analogues of Heterogeneous Catalysts. Adv. Silicon Sci. 2011, 3, 135–166. [Google Scholar]
- Levitsky, M.M.; Bilyachenko, A.N. Modern Concepts and Methods in the Chemistry of Polyhedral Metallasiloxanes. Coord. Chem. Rev. 2016, 306, 235–269. [Google Scholar] [CrossRef]
- Pinkert, D.; Limberg, C. Iron Silicates, Iron-Modulated Zeolite Catalysts, and Molecular Models Thereof. Chem. Eur. J. 2014, 20, 9166–9175. [Google Scholar] [CrossRef] [PubMed]
- Shul’pin, G.B. New Trends in Oxidative Functionalization of Carbon–Hydrogen Bonds: A Review. Catalysts 2016, 6, 50. [Google Scholar] [CrossRef]
- Levitsky, M.M.; Yalymov, A.I.; Kulakova, A.N.; Petrov, А.А.; Bilyachenko, А.N. Cage-like metallasilsesquioxanes in catalysis: A review. J. Mol. Catal. A Chem. 2017, 426, 297–304. [Google Scholar] [CrossRef]
- Bilyachenko, A.N.; Yalymov, A.I.; Korlyukov, A.A.; Long, J.; Larionova, J.; Guari, Y.; Zubavichus, Y.V.; Trigub, A.L.; Shubina, E.S.; Eremenko, I.L.; et al. Heterometallic Na6Co3 Phenylsilsesquioxane Exhibiting Slow Dynamic Behavior in its Magnetization. Chem. Eur. J. 2015, 21, 18563–18565. [Google Scholar] [CrossRef] [PubMed]
- Bilyachenko, A.N.; Yalymov, A.I.; Korlyukov, A.A.; Long, J.; Larionova, J.; Guari, Y.; Vologzhanina, A.V.; Eskova, M.A.; Shubina, E.S.; Levitsky, M.M. Unusual penta- and hexanuclear Ni(II)-based silsesquioxane polynuclear complexes. Dalton Trans. 2016, 45, 7320–7327. [Google Scholar] [CrossRef] [PubMed]
- Bilyachenko, A.N.; Levitsky, M.M.; Yalymov, A.I.; Korlyukov, A.A.; Vologzhanina, A.V.; Kozlov, Y.N.; Shul’pina, L.S.; Nesterov, D.; Pombeiro, A.F.; Lamaty, F.; et al. Heterometallic (Fe6Na8) Cage-like Silsesquioxane: Synthesis, Structure, Spin Glass Behavior and High Catalytic Activity. RSC Adv. 2016, 6, 48165–48180. [Google Scholar] [CrossRef]
- Bilyachenko, A.N.; Yalymov, A.I.; Levitsky, M.M.; Korlyukov, A.A.; Eskova, M.A.; Long, J.; Larionova, J.; Guari, Y.; Shul’pina, L.S.; Ikonnikov, N.S.; et al. First Cage-Like Pentanuclear Co(II)-Silsesquioxane. Dalton Trans. 2016, 45, 13663–13666. [Google Scholar] [CrossRef] [PubMed]
- Roesky, H.W.; Anantharaman, G.; Chandrasekhar, V.; Jancik, V.; Singh, S. Control of Molecular Topology and Metal Nuclearity in Multimetallic Assemblies: Designer Metallosiloxanes Derived from Silanetriols. Chem. Eur. J. 2004, 10, 4106–4114. [Google Scholar] [CrossRef] [PubMed]
- Lorenz, V.; Edelmann, F.T. Metallasilsesquioxanes. Adv. Organomet. Chem. 2005, 53, 101–153. [Google Scholar]
- Jutzi, P.; Lindemann, H.M.; Nolte, J.-O.; Schneider, M. Synthesis, structure, and reactivity of novel oligomeric titanasiloxanes. In Silicon Chemistry: From the Atom to Extended Systems; Jutzi, P., Schubert, U., Eds.; Wiley: Hoboken, NJ, USA, 2007; pp. 372–382. [Google Scholar]
- Edelmann, F.T. Metallasilsesquioxanes. Synthetic and structural studies. In Silicon Chemistry: From the Atom to Extended Systems; Jutzi, P., Schubert, U., Eds.; Wiley: Hoboken, NJ, USA, 2007; pp. 383–394. [Google Scholar]
- Hirotsu, M.; Taruno, S.; Yoshimura, T.; Ueno, K.; Unno, M.; Matsumoto, H. Synthesis and Structures of the First Titanium(IV) Complexes with Cyclic Tetrasiloxide Ligands: Incomplete and Complete Cage Titanosiloxanes. Chem. Lett. 2005, 34, 1542–1543. [Google Scholar] [CrossRef]
- Anderson, T.M.; Zhang, X.; Hardcastle, K.I.; Hill, C.L. Reactions of Trivacant Wells-Dawson Heteropolytungstates. Ionic Strength and Jahn-Teller Effects on Formation in Multi-Iron Complexes. Inorg. Chem. 2002, 41, 2477–2488. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhao, J.-W.; Zhao, H.-Y.; Yang, B.-F.; He, H.; Yang, G.-Y. Syntheses, structures and properties of two multi-iron–samarium/multi-iron substituted germanotungstates. CrystEngComm 2014, 16, 252–259. [Google Scholar] [CrossRef]
- Zhao, J.; Han, Q.; Shi, D.; Chen, L.; Ma, P.; Wang, J.; Niu, J. Synthesis, structure and magnetism of a S-shaped multi-iron substituted arsenotungstate containing a trivacant Keggin [B-α-AsVW9O34]9− and a hexavacant Keggin [α-AsVW6O26]11− fragments. J. Solid State Chem. 2011, 184, 2756–2761. [Google Scholar] [CrossRef]
- Caneschi, A.; Cornia, A.; Fabretti, A.C.; Gatteschi, D.; Malavasi, W. Polyiron(III)-Alkoxo Clusters: A Novel Trinuclear Complex and Its Relevance to the Extended Lattices of Iron Oxides and Hydroxide. Inorg. Chem. 1995, 34, 4660–4668. [Google Scholar] [CrossRef]
- Li, B.; Zhao, J.-W.; Zheng, S.-T.; Yang, G.-Y. A banana-shaped iron(III)-substituted tungstogermanate containing two types of lacunary polyoxometalate units. Inorg. Chem. Comm. 2009, 12, 69–71. [Google Scholar] [CrossRef]
- Levitsky, M.M. Metallasiloxanes and lithosphere. Russ. Chem. J. 2002, XLVI, 51–63. [Google Scholar]
- Bilyachenko, A.N.; Zavin, B.G.; Shubina, E.S.; Filin, A.M.; Levitsky, M.M. Intermolecular rearrangement of cobalt phenylsiloxanes. Russ. Chem. Bull. 2004, 53, 1993–1995. [Google Scholar] [CrossRef]
- Levitsky, M.M.; Zavin, B.G.; Bilyachenko, A.N. Chemistry of metallasiloxanes. Current trends and new concepts. Russ. Chem. Rev. 2007, 76, 847–866. [Google Scholar] [CrossRef]
- Pietschnig, R.; Spirk, S. The chemistry of organo silanetriols. Coord. Chem. Rev. 2016, 323, 87–106. [Google Scholar] [CrossRef]
- Čas, D.; Hurkes, N.; Spirk, S.; Belaj, F.; Bruhn, C.; Rechberger, G.N.; Pietschnig, R. Dimer formation upon deprotonation: Synthesis and structure of a m-terphenyl substituted (R,S)-dilithium disiloxanolate disilanol. Dalton Trans. 2015, 44, 12818–12823. [Google Scholar] [CrossRef] [PubMed]
- Hurkes, N.; Bruhn, C.; Belaj, F.; Pietschnig, R. Silanetriols as Powerful Starting Materials for Selective Condensation to Bulky POSS Cages. Organometallics 2014, 33, 7299–7306. [Google Scholar] [CrossRef] [PubMed]
- Yalymov, A.I.; Dronova, M.S.; Filippov, O.A.; Bilyachenko, A.N.; Levitsky, M.M. Quantum chemical study of the template synthesis of cage-like metallasiloxanes. Russ. Chem. Bull. 2014, 63, 821–825. [Google Scholar] [CrossRef]
- Bilyachenko, A.N.; Dronova, M.S.; Yalymov, A.I.; Lamaty, F.; Bantreil, X.; Martinez, J.; Bizet, C.; Shul’pina, L.S.; Korlyukov, A.A.; Arkhipov, D.E.; et al. Cage-like Copper(II) Silsesquioxanes: Transmetalation Reactions and Structural, Quantum Chemical, and Catalytic Studies. Chem. Eur. J. 2015, 21, 8758–8770. [Google Scholar] [CrossRef] [PubMed]
- Tan, G.; Yang, Y.; Chu, C.; Zhu, H.; Roesky, H.W. Cu24O24Si8R8: Organic Soluble 56-Membered Copper(I) Siloxane Cage and Its Use in Homogeneous Catalysis. J. Am. Chem. Soc. 2010, 132, 12231–12233. [Google Scholar] [CrossRef] [PubMed]
- Bantreil, X.; Kanfar, N.; Gehin, N.; Golliard, E.; Ohlmann, P.; Martinez, J.; Lamaty, F. Iron-catalyzed benzamide formation. Application to the synthesis of moclobemide. Tetrahedron 2014, 70, 5093–5099. [Google Scholar] [CrossRef]
- Bantreil, X.; Navals, P.; Martinez, J.; Lamaty, F. Iron/Caffeine as a Catalytic System for Microwave-Promoted Benzamide Formation. Eur. J. Org. Chem. 2015, 417–422. [Google Scholar] [CrossRef]
- De Araújo, M.L.; Mandelli, D.; Kozlov, Y.N.; Carvalho, W.A.; Shul’pin, G.B. Oxidation of hydroxyacetone (acetol) with hydrogen peroxide in acetonitrile solution catalyzed by iron(III) chloride. J. Mol. Catal. A Chem. 2016, 422, 103–114. [Google Scholar] [CrossRef]
- Shilov, A.E.; Shul’pin, G.B. Activation of C–H Bonds by Metal Complexes. Chem. Rev. 1997, 97, 2879–2932. [Google Scholar] [CrossRef] [PubMed]
- Shilov, A.E.; Shul’pin, G.B. Activation and Catalytic Reactions of Saturated Hydrocarbons in the Presence of Metal Complexes; Kluwer Academic Publishers: Dordrecht, The Netherlands; Boston, MA, USA; London, UK, 2000. [Google Scholar]
- Schröder, K.; Junge, K.; Bitterlich, B.; Beller, M. Fe-catalyzed Oxidation Reactions of Olefins, Alkanes and Alcohols: Involvement of Oxo- and Peroxo Complexes. Top. Organomet. Chem. 2011, 33, 83–109. [Google Scholar]
- Kirillov, A.M.; Shul’pin, G.B. Pyrazinecarboxylic acid and analogs: Highly efficient co-catalysts in the metal-complex-catalyzed oxidation of organic compounds. Coord. Chem. Rev. 2013, 257, 732–754. [Google Scholar] [CrossRef]
- Sorokin, A.B. Phthalocyanine Metal Complexes in Catalysis. Chem. Rev. 2013, 113, 8152–8191. [Google Scholar] [CrossRef] [PubMed]
- Martins, L.M.D.R.S.; Pombeiro, A.J.L. Tris(pyrazol-1-yl)methane metal complexes for catalytic mild oxidative functionalizations of alkanes, alkenes and ketones. Coord. Chem. Rev. 2014, 265, 74–88. [Google Scholar] [CrossRef]
- Afanasiev, P.; Sorokin, A.B. μ-Nitrido Diiron Macrocyclic Platform: Particular Structure for Particular Catalysis. Acc. Chem. Res. 2016, 49, 583–593. [Google Scholar] [CrossRef] [PubMed]
- Blomberg, M.R.A.; Borowski, T.; Himo, F.; Liao, R.-Z.; Siegbahn, P.E.M. Quantum Chemical Studies of Mechanisms for Metalloenzymes. Chem. Rev. 2014, 114, 3601–3658. [Google Scholar] [CrossRef] [PubMed]
- Poulos, T.L. Heme Enzyme Structure and Function. Chem. Rev. 2014, 114, 3919–3962. [Google Scholar] [CrossRef] [PubMed]
- Trehoux, A.; Mahy, J.-P.; Avenier, F. A growing family of O2 activating dinuclear iron enzymes with key catalytic diiron(III)-peroxo intermediates: Biological systems and chemical models. Coord. Chem. Rev. 2016, 322, 142–158. [Google Scholar] [CrossRef]
- Shul’pin, G.B. Selectivity in C–H Functionalizations. In Comprehensive Inorganic Chemistry II, 2nd ed.; Reedijk, J., Poeppelmeier, K., Casella, L., Eds.; Elsevier: Amsterdam, The Netherlands, 2013; Volume 6, pp. 79–104. [Google Scholar]
- Bilyachenko, A.N.; Levitsky, M.M.; Yalymov, A.I.; Korlyukov, A.A.; Khrustalev, V.N.; Vologzhanina, A.V.; Shul’pina, L.S.; Ikonnikov, N.S.; Trigub, A.E.; Dorovatovskii, P.V.; et al. Cage-like Fe,Na-Germsesquioxanes: Structure, Magnetism, and Catalytic Activity. Angew. Chem. Int. Ed. 2016, 55, 15360–15363. [Google Scholar] [CrossRef] [PubMed]
- Shul’pin, G.B. C–H functionalization: Thoroughly tuning ligands at a metal ion, a chemist can greatly enhance catalyst’s activity and selectivity, Perspective. Dalton Trans. 2013, 42, 12794–12818. [Google Scholar] [CrossRef]
- Shul’pin, G.B. Selectivity enhancement in functionalization of C–H bonds: A review. Org. Biomol. Chem. 2010, 8, 4217–4228. [Google Scholar] [CrossRef] [PubMed]
- Shul’pin, G.B.; Druzhinina, A.N. Hydroperoxidation of alkanes by atmospheric oxygen in the presence of hydroquinone or quinone catalyzed by copper(II) acetate under visible light irradiation. React. Kinet. Catal. Lett. 1992, 47, 207–211. [Google Scholar] [CrossRef]
- Shul’pin, G.B. Metal-catalyzed hydrocarbon oxygenations in solutions: The dramatic role of additives: A review. J. Mol. Catal. A Chem. 2002, 189, 39–66. [Google Scholar] [CrossRef]
- Shul’pin, G.B. Metal-catalysed hydrocarbon oxidations. C. R. Chim. 2003, 6, 163–178. [Google Scholar] [CrossRef]
- Shul’pin, G.B. Hydrocarbon Oxygenations with Peroxides Catalyzed by Metal Compounds. Mini Rev. Org. Chem. 2009, 6, 95–104. [Google Scholar] [CrossRef]
- Shul’pin, G.B.; Kozlov, Y.N.; Shul’pina, L.S.; Kudinov, A.R.; Mandelli, D. Extremely Efficient Alkane Oxidation by a New Catalytic Reagent H2O2/Os3(CO)12/Pyridine. Inorg. Chem. 2009, 48, 10480–10482. [Google Scholar] [CrossRef] [PubMed]
- Shul’pin, G.B.; Kozlov, Y.N.; Shul’pina, L.S.; Petrovskiy, P.V. Oxidation of alkanes and alcohols with hydrogen peroxide catalyzed by complex Os3(CO)10(mu-H)2. Appl. Organomet. Chem. 2010, 24, 464–472. [Google Scholar]
- Olivo, G.; Lanzalunga, O.; Di Stefano, S. Non-Heme Imine-Based Iron Complexes as Catalysts for Oxidative Processes. Adv. Synth. Catal. 2016, 358, 843–863. [Google Scholar] [CrossRef]
- Garcia-Bosch, I.; Siegel, M.A. Copper-Catalyzed Oxidation of Alkanes with H2O2 under a Fenton-like Regime. Angew. Chem. Int. Ed. 2016, 128, 13065–13068. [Google Scholar] [CrossRef]
- Shul’pin, G.B.; Kozlov, Y.N.; Nizova, G.V.; Süss-Fink, G.; Stanislas, S.; Kitaygorodskiy, A.; Kulikova, V.S. Oxidations by the reagent “O2–H2O2–vanadium derivative–pyrazine-2-carboxylic acid”. Part 12. Main features, kinetics and mechanism of alkane hydroperoxidation. J. Chem. Soc. Perkin Trans. 2001, 2, 1351–1371. [Google Scholar] [CrossRef]
- Kozlov, Y.N.; Romakh, V.B.; Kitaygorodskiy, A.; Buglyó, P.; Süss-Fink, G.; Shul’pin, G.B. Oxidation of 2-Propanol and Cyclohexane by the Reagent “Hydrogen Peroxide-Vanadate Anion-Pyrazine-2-carboxylic Acid”: Kinetics and Mechanism. J. Phys. Chem. A 2007, 111, 7736–7752. [Google Scholar] [CrossRef] [PubMed]
- Shul’pin, G.B.; Kozlov, Y.N.; Shul’pina, L.S.; Carvalho, W.; Mandelli, D. Oxidation reactions catalyzed by osmium compounds. Part 4. Highly efficient oxidation of hydrocarbons and alcohols including glycerol by the H2O2/Os3(CO)12/pyridine reagent. RSC Adv. 2013, 3, 15065–15074. [Google Scholar] [CrossRef]
- Vinogradov, M.M.; Shul’pina, L.S.; Kozlov, Y.N.; Kudinov, A.R.; Ikonnikov, N.S.; Shul’pin, G.B. Oxidation of hydrocarbons and alcohols with peroxides catalyzed by new pi-cymene osmium complexes. J. Organometal. Chem. 2015, 784, 52–61. [Google Scholar] [CrossRef]
- Shul’pina, L.S.; Kudinov, A.R.; Mandelli, D.; Carvalho, W.A.; Kozlov, Y.N.; Vinogradov, M.M.; Ikonnikov, N.S.; Shul’pin, G.B. Oxidation of alkanes and benzene with hydrogen peroxide catalyzed by ferrocene in the presence of acids. J. Organometal. Chem. 2015, 793, 217–231. [Google Scholar] [CrossRef]
- Gryca, I.; Machura, B.; Małecki, J.G.; Kusz, J.; Shul’pina, L.S.; Ikonnikov, N.S.; Shul’pin, G.B. p-Tolylimido rhenium(V) complexes with phenolate-based ligands: Synthesis, X-ray studies and catalytic activity in oxidation with tert-butylhydroperoxide. Dalton Trans. 2016, 45, 334–351. [Google Scholar] [CrossRef] [PubMed]
- Gryca, I.; Machura, B.; Shul’pina, L.S.; Shul’pin, G.B. Synthesis, structures and catalytic activity of p-tolylimido rhenium(V) complexes incorporating quinoline-derived ligands. Inorg. Chim. Acta 2017, 455, 683–695. [Google Scholar] [CrossRef]
- Bilyachenko, A.N.; Kulakova, A.N.; Levitsky, M.M.; Petrov, A.A.; Korlyukov, A.A.; Shul’pina, L.S.; Khrustalev, V.N.; Dorovatovskii, P.V.; Vologzhanina, A.V.; Tsareva, U.S.; et al. Unusual Tri-, Hexa-, and Nonanuclear Cu(II) Cage Methylsilsesquioxanes: Synthesis, Structures, and Catalytic Activity in Oxidations with Peroxides. Inorg. Chem. 2017, 56. [Google Scholar]
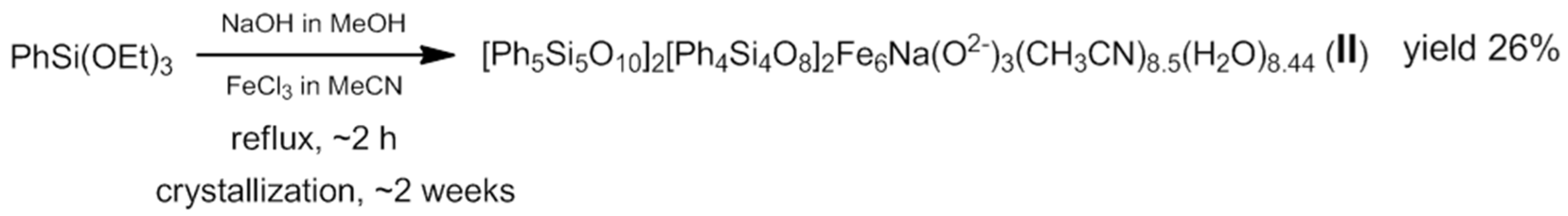




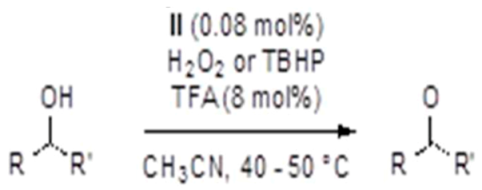
| Entry | Alcohol | Oxidant | Time (h) | Ketone Yield (%) |
|---|---|---|---|---|
| 1 | 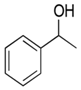 | H2O2 | 3 | 30 |
| 2 | 5 | 50 | ||
| 3 | TBHP | 4 | 80 | |
| 4 | 5 | 92 | ||
| 5 | 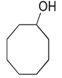 | TBHP | 3 | 71 |
| 6 | 4 | 80 | ||
| 7 | 5 | 85 |
| Compound | I | II |
|---|---|---|
| Brutto formula | C133.50H128.25Fe6N5.75Na7O46Si20 | C125H117.96Fe6N8.5Na6O44.36Si18 |
| Formula weight | 3606.99 | 3427.79 |
| Wavelength, Å | 0.96600 | 1.5418 |
| T, K | 100 | 120 |
| Space group | P21/n | Pn |
| Z | 4 | 2 |
| a, Å | 27.130(5) | 17.7227(10) |
| b, Å | 18.150(4) | 15.9140(9) |
| c, Å | 32.830(7) | 28.4984(17) |
| β, ° | 91.75(3) | 100.136(3) |
| V, Å3 | 16158(6) | 7912.2(8) |
| ρcalc, g·cm−3 | 1.483 | 1.439 |
| μ, cm−1 | 17.86 | 64.35 |
| F(000) | 7402 | 3513 |
| 2θmax, ° | 70.84 | 135.72 |
| Reflections collected | 234,993 | 20,450 |
| Independent reflections | 22,897 | 20,450 |
| Independent reflections with I > 2σ(I) | 18,123 | 17,065 |
| Parameters | 1681 | 1825 |
| R1 [I > 2σ(I)] | 0.1274 | 0.0925 |
| wR2 (all reflections) | 0.2763 | 0.2347 |
| GOF | 1.065 | 0.973 |
| Residual electron density, e·Å3 (ρmin/ρmax) | 1.72/−1.10 | 2.08/−1.15 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yalymov, A.I.; Bilyachenko, A.N.; Levitsky, M.M.; Korlyukov, A.A.; Khrustalev, V.N.; Shul’pina, L.S.; Dorovatovskii, P.V.; Es’kova, M.A.; Lamaty, F.; Bantreil, X.; et al. High Catalytic Activity of Heterometallic (Fe6Na7 and Fe6Na6) Cage Silsesquioxanes in Oxidations with Peroxides. Catalysts 2017, 7, 101. https://doi.org/10.3390/catal7040101
Yalymov AI, Bilyachenko AN, Levitsky MM, Korlyukov AA, Khrustalev VN, Shul’pina LS, Dorovatovskii PV, Es’kova MA, Lamaty F, Bantreil X, et al. High Catalytic Activity of Heterometallic (Fe6Na7 and Fe6Na6) Cage Silsesquioxanes in Oxidations with Peroxides. Catalysts. 2017; 7(4):101. https://doi.org/10.3390/catal7040101
Chicago/Turabian StyleYalymov, Alexey I., Alexey N. Bilyachenko, Mikhail M. Levitsky, Alexander A. Korlyukov, Victor N. Khrustalev, Lidia S. Shul’pina, Pavel V. Dorovatovskii, Marina A. Es’kova, Frédéric Lamaty, Xavier Bantreil, and et al. 2017. "High Catalytic Activity of Heterometallic (Fe6Na7 and Fe6Na6) Cage Silsesquioxanes in Oxidations with Peroxides" Catalysts 7, no. 4: 101. https://doi.org/10.3390/catal7040101
APA StyleYalymov, A. I., Bilyachenko, A. N., Levitsky, M. M., Korlyukov, A. A., Khrustalev, V. N., Shul’pina, L. S., Dorovatovskii, P. V., Es’kova, M. A., Lamaty, F., Bantreil, X., Villemejeanne, B., Martinez, J., Shubina, E. S., Kozlov, Y. N., & Shul’pin, G. B. (2017). High Catalytic Activity of Heterometallic (Fe6Na7 and Fe6Na6) Cage Silsesquioxanes in Oxidations with Peroxides. Catalysts, 7(4), 101. https://doi.org/10.3390/catal7040101







