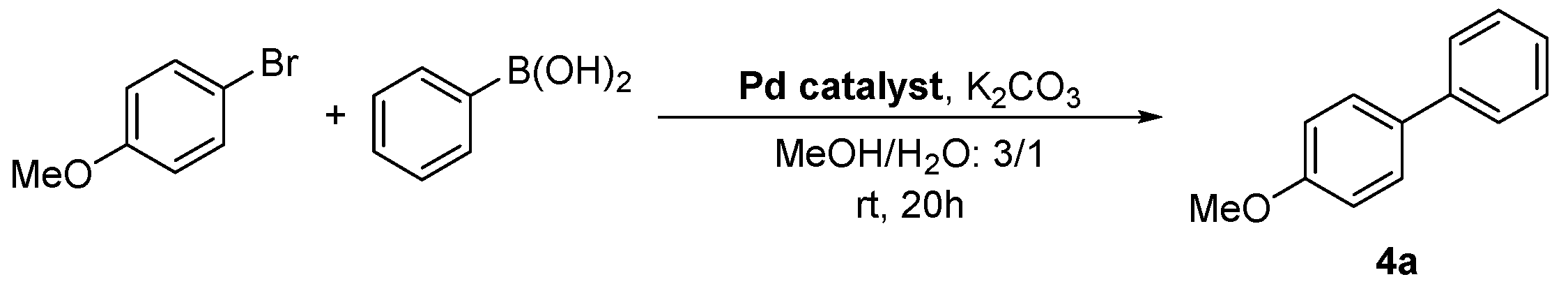
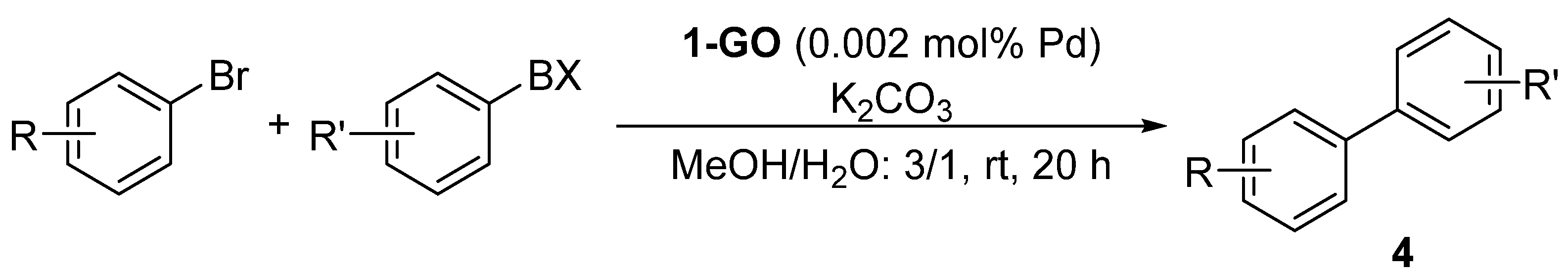Graphene Oxide-Supported Oxime Palladacycles as Efficient Catalysts for the Suzuki–Miyaura Cross-Coupling Reaction of Aryl Bromides at Room Temperature under Aqueous Conditions
Abstract
:1. Introduction
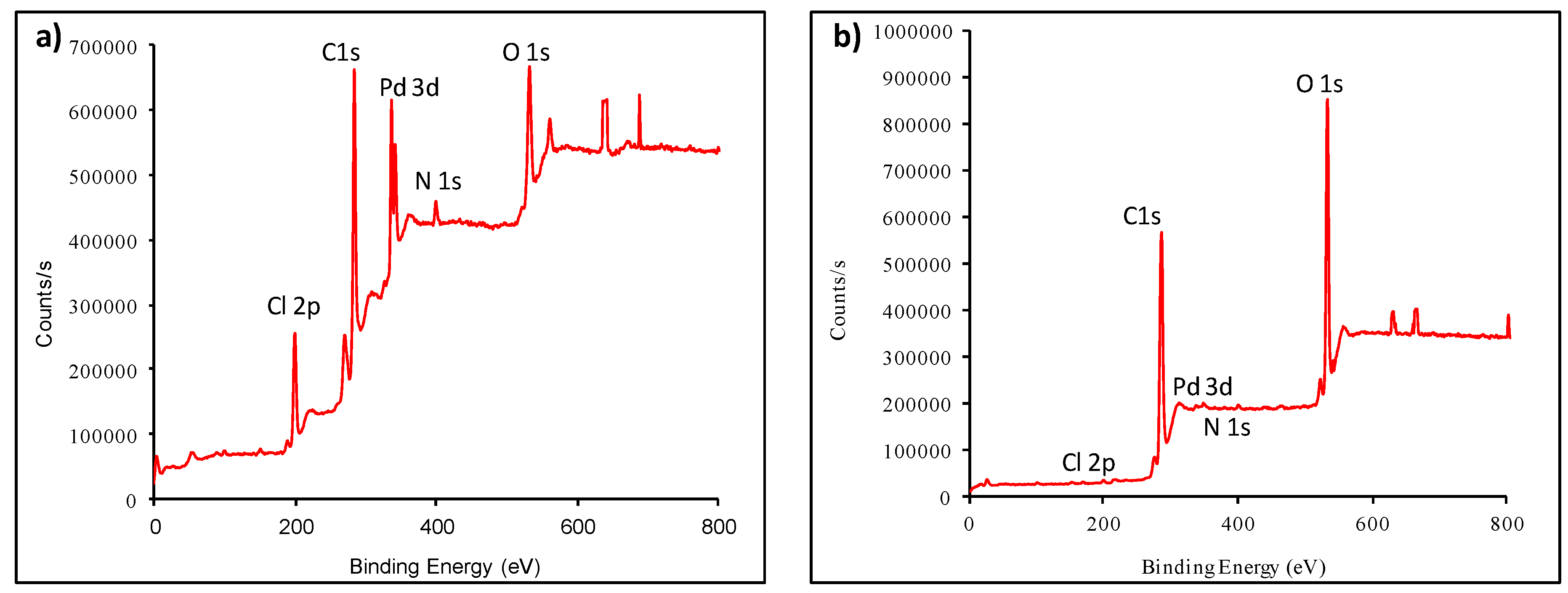
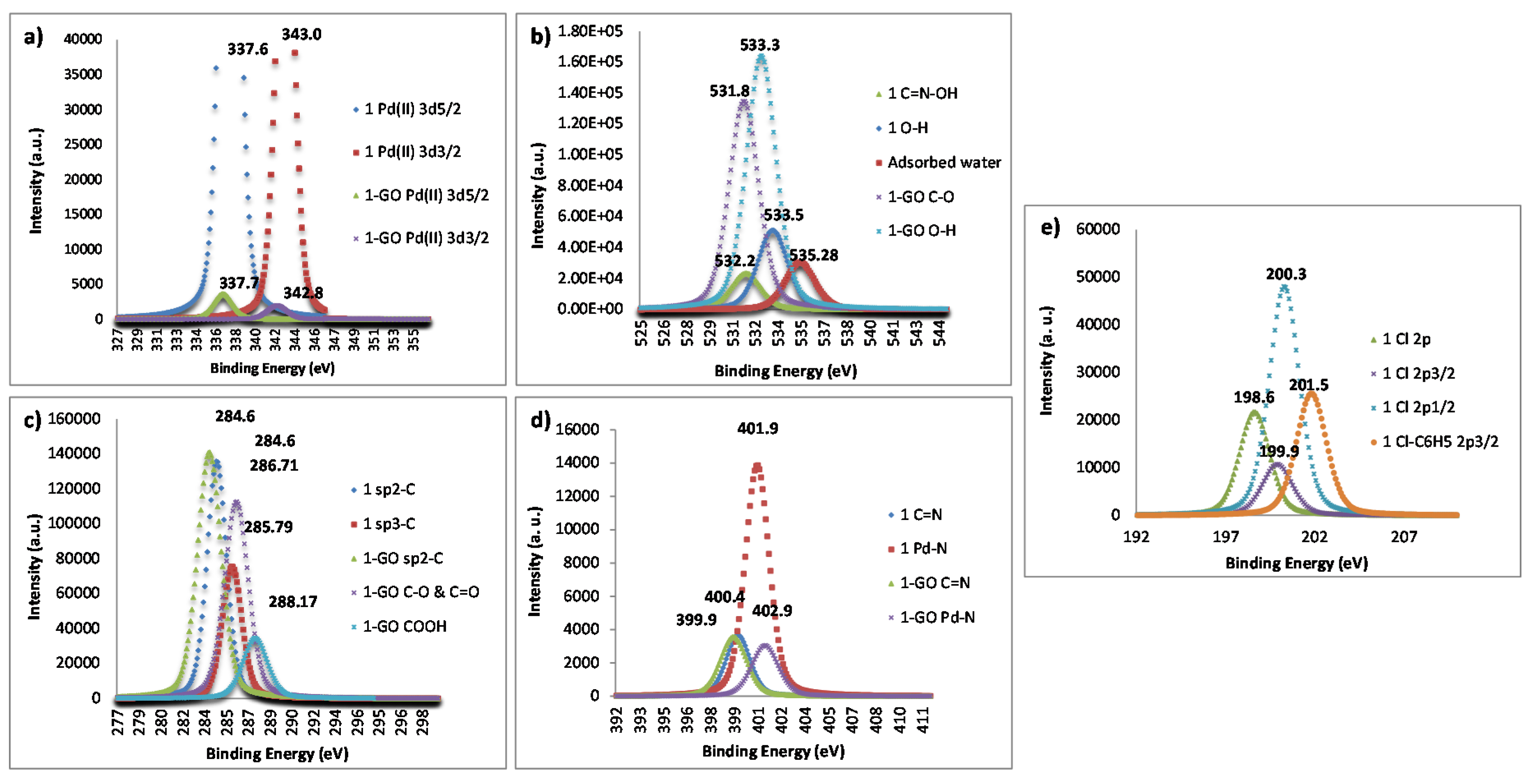
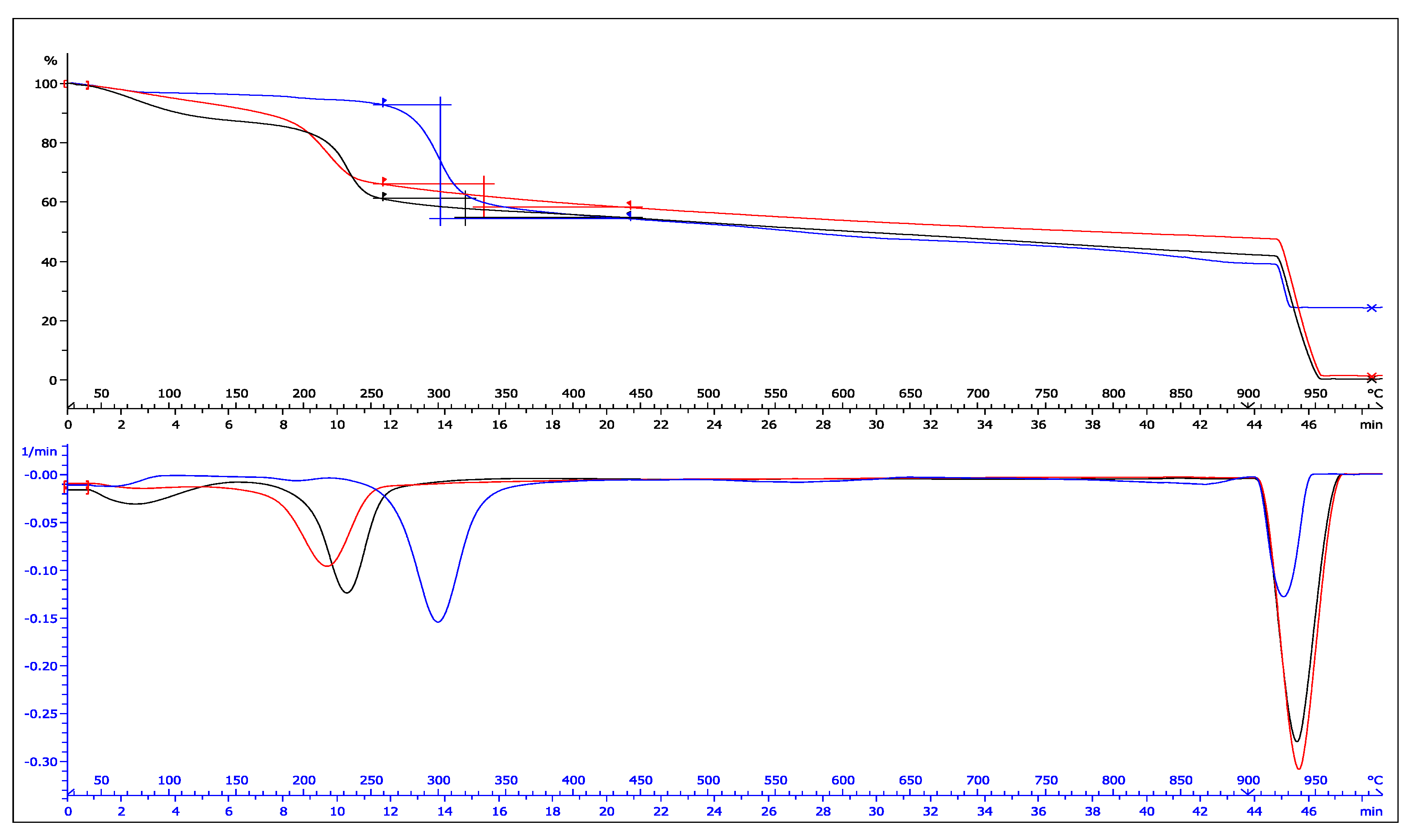
2. Results and Discussion
3. Materials and Methods
3.1. General
3.2. Synthesis of Oxime Palladacycle 1
3.3. Synthesis of 1-GO
3.4. Typical Procedure for the Suzuki–Miyaura Reaction
3.5. Typical Procedure for the Recovery of the Catalyst in the Suzuki–Miyaura Reaction
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References and Notes
- Diederich, F.; Stang, P.J. Metal-Catalyzed Cross-Coupling Reactions; Wiley-VCH: Weinheim, Germany, 1998. [Google Scholar]
- Beller, M.; Bolm, C. Transition Metals for Organic Synthesis. Building Blocks and Fine Chemicals; Wiley-VCH: Weinheim, Germany, 1998. [Google Scholar]
- Negishi, E. Handbook of Organopalladium Chemistry for Organic Synthesis; Wiley-Interscience: New York, NY, USA, 2002. [Google Scholar]
- Miyaura, N. Cross-Coupling Reactions. A Practical Guide; Springer: Berlin, Germany, 2002. [Google Scholar]
- Tsuji, J. Palladium Reagents and Catalysts. Innovations in Organic Synthesis; Wiley: Chichester, UK, 2004. [Google Scholar]
- Beller, M.; Bolm, C. Transition Metals for Organic Synthesis. Building Blocks and Fine Chemicals, 2nd ed.; Wiley-VCH: Weinheim, Germany, 2004. [Google Scholar]
- Diederich, F.; Stang, P.J. Metal-Catalyzed Cross-Coupling Reactions; Wiley-VCH: Weinheim, Germany, 2004. [Google Scholar]
- Farina, V.; Miyaura, N.; Buchwald, S.L. Special Issue on Cross-Coupling Chemistry. Adv. Synth. Catal. 2004, 346, 1505–1879. [Google Scholar]
- Negishi, E. Transition Metal-Catalyzed Organometallic Reactions that Have Revolutionized Organic Synthesis. Bull. Chem. Soc. Jpn. 2007, 80, 233–257. [Google Scholar] [CrossRef]
- Dupont, J.; Pfeffer, M. Palladacycles: Synthesis, Characterization and Applications; Wiley-VCH: Weinheim, Germany, 2008. [Google Scholar]
- Ratti, R. Palladacycles—Versatile Catalysts for Carbon-Carbon Coupling Reactions. Can. Chem. Trans. 2014, 2, 467–488. [Google Scholar]
- Das, P.; Linert, W. Schiff base-derived homogeneous and heterogeneous palladium catalysts for the Suzuki–Miyaura reaction. Coord. Chem. Rev. 2016, 311, 1–23. [Google Scholar] [CrossRef]
- Nájera, C. Oxime-Derived Palladacycles: Applications in Catalysis. ChemCatChem 2016, 8, 1865–1881. [Google Scholar] [CrossRef] [Green Version]
- Bedford, R.B.; Cazin, C.S.J.; Hursthouse, M.B.; Light, M.E.; Pike, K.J.; Wimperis, S. Silica-supported imine palladacycles—Recyclable catalysts for the Suzuki reaction? J. Organomet. Chem. 2001, 633, 173–181. [Google Scholar] [CrossRef]
- Baleizão, C.; Corma, A.; García, H.; Leyva, A. An oxime-carbapalladacycle complex covalently anchored to silica as an active and reusable heterogeneous catalyst for Suzuki cross-coupling in water. Chem. Commun. 2003, 606–607. [Google Scholar] [CrossRef]
- Yu, K.; Sommer, W.; Weck, M.; Jones, C.W. Silica and polymer-tethered Pd–SCS-pincer complexes: Evidence for precatalyst decomposition to form soluble catalytic species in Mizoroki–Heck chemistry. J. Catal. 2004, 226, 101–110. [Google Scholar] [CrossRef]
- Venkatesan, C.; Singh, A.P. Synthesis and characterization of carbometallated palladacycles over 3-hydroxypropyltriethoxysilyl-functionalized MCM-41. J. Catal. 2004, 227, 148–163. [Google Scholar] [CrossRef]
- Baleizão, C.; Corma, A.; García, H.; Leyva, A. Oxime Carbapalladacycle Covalently Anchored to High Surface Area Inorganic Supports or Polymers as Heterogeneous Green Catalysts for the Suzuki Reaction in Water. J. Org. Chem. 2004, 69, 439–446. [Google Scholar] [CrossRef] [PubMed]
- Corma, A.; Das, D.; García, H.; Leyva, A. A periodic mesoporous organosilica containing a carbapalladacycle complex as heterogeneous catalyst for Suzuki cross-coupling. J. Catal. 2005, 229, 322–331. [Google Scholar] [CrossRef]
- Mohamed, M.F.; Neverov, A.A.; Brown, R.S. An Immobilized Ortho-Palladated Dimethylbenzylamine Complex as an Efficient Catalyst for the Methanolysis of Phosphorothionate Pesticides. Inorg. Chem. 2009, 48, 1183–1191. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Yang, H.; Lia, W.; Zhang, G. Rationally designed palladium complexes on a bulky N-heterocyclic carbene-functionalized organosilica: An efficient solid catalyst for the Suzuki–Miyaura coupling of challenging aryl chlorides. Green Chem. 2011, 13, 2939–2947. [Google Scholar] [CrossRef]
- Farsadpour, S.; Ghoochany, L.T.; Shylesh, S.; Dörr, G.; Seifert, A.; Ernst, S.; Thiel, W.R. A Covalently Supported Pyrimidinylphosphane Palladacycle as a Heterogenized Catalyst for the Suzuki-Miyaura Cross Coupling. ChemCatChem 2012, 4, 401–407. [Google Scholar] [CrossRef]
- Lu, F-H.; Yue, P.; Wang, X.-R.; Lu, Z.-L. Synthesis and immobilization of oxime-derived palladacycles as effective and reusable catalysts for the degradation of phosphorothionate pesticides. Inorg. Chem. Commun. 2013, 34, 19–22. [Google Scholar] [CrossRef]
- Liu, H.; Li, T.; Xue, X.; Xua, W.; Wu, Y. The mechanism of a self-assembled Pd(ferrocenylimine)-Si compound-catalysed Suzuki coupling reaction. Catal. Sci. Technol. 2016, 6, 1667–1676. [Google Scholar] [CrossRef]
- Lin, C.-A.; Luo, F.-T. Polystyrene-supported recyclable palladacycle catalyst for Heck, Suzuki and Sonogashira reactions. Tetrahedron Lett. 2003, 44, 7565–7568. [Google Scholar] [CrossRef]
- Bedford, R.B.; Coles, S.J.; Hursthouse, M.B.; Scordia, V.J.M. Polystyrene-supported dicyclohexylphenylphosphine adducts of amine- and phosphite-based palladacycles in the Suzuki coupling of aryl chlorides. Dalton Trans. 2005, 991–995. [Google Scholar] [CrossRef] [PubMed]
- Luo, F.-T.; Xue, C.; Ko, S.-L.; Shao, Y.-D.; Wu, C.-J.; Kuo, Y.-M. Preparation of polystyrene-supported soluble palladacycle catalyst for Heck and Suzuki reactions. Tetrahedron 2005, 61, 6040–6045. [Google Scholar] [CrossRef]
- Corma, A.; Garcia, H.; Leyva, A. Comparison between polyethylenglycol and imidazolium ionic liquids as solvents for developing a homogeneous and reusable palladium catalytic system for the Suzuki and Sonogashira coupling. Tetrahedron 2005, 61, 9848–9854. [Google Scholar] [CrossRef]
- Corma, A.; Garcia, H.; Leyva, A. Polyethyleneglycol as scaffold and solvent for reusable C–C coupling homogeneous Pd catalysts. J. Catal. 2006, 240, 87–99. [Google Scholar] [CrossRef]
- Solodenko, W.; Mennecke, K.; Vogt, C.; Gruhl, S.; Kirschning, A. Polyvinylpyridine, a Versatile Solid Phase for Coordinative Immobilisation of Palladium Precatalysts—Applications in Suzuki-Miyaura Reactions. Synthesis 2006, 1873–1881. [Google Scholar]
- Hershberger, J.C.; Zhang, L.; Lu, G.; Malinakova, H.C. Polymer-Supported Palladacycles: Efficient Reagents for Synthesis of Benzopyrans with Palladium Recovery. Relationship among Resin Loading, Pd:P Ratio, and Reactivity of Immobilized Palladacycles. J. Org. Chem. 2006, 71, 231–235. [Google Scholar] [CrossRef] [PubMed]
- Mennecke, K.; Solodenko, W.; Kirschning, A. Carbon-Carbon Cross-Coupling Reactions under Continuous Flow Conditions Using Poly(vinylpyridine) Doped with Palladium. Synthesis 2008, 1589–1599. [Google Scholar] [CrossRef]
- Alacid, E.; Najera, C. Kaiser oxime resin-derived palladacycle: A recoverable polymeric precatalyst in Suzuki–Miyaura reactions in aqueous media. J. Organomet. Chem. 2009, 694, 1658–1665. [Google Scholar] [CrossRef]
- Islam, M.; Mondal, P.; Roy, A.S.; Tuhina, K. Catalytic oxidation of organic substrates using a reusable polystyrene-anchored orthometallated palladium (II) complex. J. Appl. Polym. Sci. 2010, 118, 52–62. [Google Scholar] [CrossRef]
- Yang, Y.-C.; Toy, P.H. Self-Supported Ligands as a Platform for Catalysis: Use of a Polymeric Oxime in a Recyclable Palladacycle Precatalyst for Suzuki–Miyaura Reactions. Synlett 2014, 25, 1319–1324. [Google Scholar]
- Cho, H.-J.; Jung, S.; Kong, S.; Park, S.-J.; Lee, S.-M.; Lee, Y.-S. Polymer-Supported Electron-Rich Oxime Palladacycle as an Efficient Heterogeneous Catalyst for the Suzuki Coupling Reaction. Adv. Synth. Catal. 2014, 356, 1056–1064. [Google Scholar] [CrossRef]
- Sudheendran, M.; Eitel, S.H.; Naumann, S.; Buchmeiser, M.R.; Peters, R. Heterogenization of ferrocene palladacycle catalysts on ROMP-derived monolithic supports and application to a Michael addition. New J. Chem. 2014, 38, 5597–5607. [Google Scholar] [CrossRef]
- Karami, K.; Najvani, S.D.; Naeini, N.H.; Hervés, P. Palladium particles from oxime-derived palladacycle supported on Fe3O4/oleic acid as a catalyst for the copper-free Sonogashira cross-coupling reaction. Chin. J. Catal. 2015, 36, 1047–1053. [Google Scholar] [CrossRef]
- Gholinejad, M.; Razeghi, M.; Najera, C. Magnetic nanoparticles supported oxime palladacycle as a highly efficient and separable catalyst for room temperature Suzuki-Miyaura coupling reaction in aqueous media. RSC Adv. 2015, 5, 49568–49576. [Google Scholar] [CrossRef] [Green Version]
- Karami, K.; Haghighat Naeini, N. Palladium nanoparticles supported on cucurbit[6]uril: An efficient heterogeneous catalyst for the Suzuki reaction under mild conditions. Appl. Organomet. Chem. 2015, 29, 33–39. [Google Scholar] [CrossRef]
- Singh, V.; Ratti, R.; Kaur, S. Synthesis and characterization of recyclable and recoverable MMT-clay exchanged ammonium tagged carbapalladacycle catalyst for Mizoroki–Heck and Sonogashira reactions in ionic liquid media. J. Mol. Catal. A Chem. 2011, 334, 13–19. [Google Scholar] [CrossRef]
- Karami, K.; Hashemi, S.; Dinari, M. Investigation of catalytic properties of two new orthopalladated complexes supported on montmorillonite: Synthesis, characterization and application in aerobic oxidation of alcohols. Appl. Organomet. Chem. 2017, in press. [Google Scholar] [CrossRef]
- Hübner, S.; de Vries, J.G.; Farina, V. Why Does Industry Not Use Immobilized Transition Metal Complexes as Catalysts? Adv. Synth. Catal. 2016, 358, 3–25. [Google Scholar] [CrossRef]
- Scheuermann, G.M.; Rumi, L.; Steurer, P.; Bannwarth, W.; Mülhaupt, R. Palladium Nanoparticles on Graphite Oxide and Its Functionalized Graphene Derivatives as Highly Active Catalysts for the Suzuki-Miyaura Coupling Reaction. J. Am. Chem. Soc. 2009, 131, 8262–8270. [Google Scholar] [CrossRef] [PubMed]
- Siamaki, A.R.; Khder, A.E. R.S.; Abdelsayed, V.; El-Shall, M.S.; Gupton, B.F. Microwave-assisted synthesis of palladium nanoparticles supported on graphene: A highly active and recyclable catalyst for carbon–carbon cross-coupling reactions. J. Catal. 2011, 279, 1–11. [Google Scholar] [CrossRef]
- Rumi, L.; Scheuermann, G.M.; Mülhaupt, R.; Bannwarth, W. Palladium Nanoparticles on Graphite Oxide as Catalyst for Suzuki-Miyaura, Mizoroki-Heck and Sonogashira Reactions. Helv. Chim. Acta 2011, 94, 966–976. [Google Scholar] [CrossRef]
- Xiang, G.; He, J.; Li, T.; Zhuang, J.; Wang, X. Rapid preparation of noble metal nanocrystalsvia facile coreduction with graphene oxide and their enhanced catalytic properties. Nanoscale 2011, 3, 3737–3742. [Google Scholar] [CrossRef] [PubMed]
- Machado, B.F.; Serp, P. Graphene-based materials for catalysis. Catal. Sci. Technol. 2012, 2, 54–75. [Google Scholar] [CrossRef]
- Moussa, S.; Siamaki, A.R.; Gupton, B.F.; El-Shall, M.S. Pd-Partially Reduced Graphene Oxide Catalysts (Pd/PRGO): Laser Synthesis of Pd Nanoparticles Supported on PRGO Nanosheets for Carbon-Carbon Cross Coupling Reactions. ACS Catal. 2012, 2, 145–154. [Google Scholar] [CrossRef]
- Ioni, Y.V.; Lyubimov, S.E.; Korlyukov, A.A.; Antipin, M.Y.; Davankov, V.A.; Gubin, S.P. Activity of palladium nanoparticles on graphene oxide in the Suzuki-Miyaura reaction. Russ. Chem. Bull., Int. Ed. 2012, 61, 1825–1827. [Google Scholar] [CrossRef]
- Hu, J.; Wang, Y.; Han, M.; Zhou, Y.; Jiang, X.; Sun, P. A facile preparation of palladium nanoparticles supported on magnetite/s-graphene and their catalytic application in Suzuki-Miyaura reaction. Catal. Sci. Technol. 2012, 2, 2332–2340. [Google Scholar] [CrossRef]
- Qu, K.; Wu, L.; Ren, J.; Qu, X. Natural DNA-Modified Graphene/Pd Nanoparticles as Highly Active Catalyst for Formic Acid Electro-Oxidation and for the Suzuki Reaction. ACS Appl. Mater. Inter. 2012, 4, 5001–5009. [Google Scholar] [CrossRef] [PubMed]
- Shang, N.; Feng, C.; Zhang, H.; Gao, S.; Tang, R.; Wang, C.; Wang, Z. Suzuki-Miyaura reaction catalyzed by graphene oxide supported palladium nanoparticles. Catal. Commun. 2013, 40, 111–115. [Google Scholar] [CrossRef]
- Shang, N.; Gao, S.; Feng, C.; Zhang, H.; Wang, C.; Wang, Z. Graphene oxide supported N-heterocyclic carbene-palladium as a novel catalyst for the Suzuki-Miyaura reaction. RSC Adv. 2013, 3, 21863–21868. [Google Scholar] [CrossRef]
- Hoseini, S.J.; Dehghani, M.; Nasrabadi, H. Thin film formation of Pd/reduced-graphene oxide and Pd nanoparticles at oil-water interface, suitable as effective catalyst for Suzuki-Miyaura reaction in water. Catal. Sci. Technol. 2014, 4, 1078–1083. [Google Scholar] [CrossRef]
- Yamamoto, S.-I.; Kinoshita, H.; Hashimoto, H.; Nishina, Y. Facile preparation of Pd nanoparticles supported on single-layer graphene oxide and application for the Suzuki-Miyaura cross-coupling reaction. Nanoscale 2014, 6, 6501–6505. [Google Scholar] [CrossRef] [PubMed]
- Joshi, H.; Sharma, K.N.; Sharma, A.K.; Singh, A.K. Palladium-phosphorus/sulfur nanoparticles (NPs) decorated on graphene oxide: Synthesis using the same precursor for NPs and catalytic applications in Suzuki-Miyaura coupling. Nanoscale 2014, 6, 4588–4597. [Google Scholar] [CrossRef] [PubMed]
- Bai, C.; Zhao, Q.; Li, Y.; Zhang, G.; Zhang, F.; Fan, X. Palladium Complex Immobilized on Graphene Oxide as an Efficient and Recyclable Catalyst for Suzuki Coupling Reaction. Catal. Lett. 2014, 144, 1617–1623. [Google Scholar] [CrossRef]
- Park, J.H.; Raza, F.; Jeon, S.-J.; Kim, H.-I.; Kang, T.W.; Yim, S.; Kim, J.-H. Recyclable N-heterocyclic carbene/palladium catalyst on graphene oxide for the aqueous-phase Suzuki reaction. Tetrahedron Lett. 2014, 55, 3426–3430. [Google Scholar] [CrossRef]
- Movahed, S.K.; Esmatpoursalmani, R.; Bazgir, A. N-Heterocyclic carbene palladium complex supported on ionic liquid-modified graphene oxide as an efficient and recyclable catalyst for Suzuki reaction. RSC Adv. 2014, 4, 14586–14591. [Google Scholar] [CrossRef]
- Lin, J.; Mei, T.; Lv, M.; Zhang, C.; Zhao, Z.; Wang, X. Size-controlled PdO/graphene oxides and their reduction products with high catalytic activity. RSC Adv. 2014, 4, 29563–29570. [Google Scholar] [CrossRef]
- Pérez-Mayoral, E.; Calvino-Casilda, V.; Soriano, E. Metal-supported carbon-based materials: Opportunities and challenges in the synthesis of valuable products. Catal. Sci. Technol. 2016, 6, 1265. [Google Scholar] [CrossRef]
- Fath, R.H.; Hoseini, S.J. Covalently cyclopalladium(II) complex/reduced-graphene oxide as the effective catalyst for the Suzuki-Miyaura reaction at room temperature. J. Organomet. Chem. 2017, 828, 16–23. [Google Scholar] [CrossRef]
- Xue, Z.; Huang, P.; Li, T.; Qin, P.; Xiao, D.; Liu, M.; Chen, P.; Wu, Y. A novel “tunnel-like” cyclopalladated arylimine catalyst immobilized on graphene oxide nano-sheet. Nanoscale 2017, 9, 781–791. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Martínez, M.; Buxaderas, E.; Pastor, I.M.; Alonso, D.A. Palladium nanoparticles supported on graphene and reduced graphene oxide as efficient recyclable catalyst for the Suzuki-Miyaura reaction of potassium aryltrifluoroborates. J. Mol. Catal. A: Chem. 2015, 404, 1–7. [Google Scholar]
- Alonso, D.A.; Botella, L.; Nájera, C.; Pacheco, M.C. Synthetic Applications of Oxime-Derived Palladacycles as Versatile Catalysts in Cross-Coupling Reactions. Synthesis 2004, 1713–1718. [Google Scholar]
- Alacid, E.; Alonso, D.A.; Botella, L.; Nájera, C.; Pacheco, M.C. Oxime palladacycles revisited: Stone-stable complexes nonetheless very active catalysts. Chem. Rec. 2006, 6, 117–132. [Google Scholar] [CrossRef] [PubMed]
- Alonso, D.A.; Nájera, C. Oxime-derived palladacycles as source of palladium nanoparticles. Chem. Soc. Rev. 2010, 39, 2891–2902. [Google Scholar] [CrossRef] [PubMed]
- Alonso, D.A.; Nájera, C.; Pacheco, M.C. Oxime Palladacycles: Stable and Efficient Catalysts for Carbon−Carbon Coupling Reactions. Org. Lett. 2000, 2, 1823–1826. [Google Scholar] [CrossRef] [PubMed]
- GO Was Supplied by Nanoinnova Technologies S.L. Available online: http://www.nanoinnova.com/Product/Details/24 (accessed on 21 March 2017).
- Singh, G.; Botcha, V.C.; Sutar, D.S.; Talwar, S.S.; Srinivasa, R.S.; Major, S.S. Graphite mediated reduction of graphene oxide monolayer sheets. Carbon 2015, 95, 843–851. [Google Scholar] [CrossRef]
- Yu, B.; Wang, X.; Qian, X.; Xing, W.; Yang, H.; Ma, L.; Lin, Y.; Jiang, S.; Song, L.; Hu, Y.; et al. Functionalized graphene oxide/phosphoramide oligomer hybrids flame retardant prepared via in situ polymerization for improving the fire safety of polypropylene. RSC Adv. 2014, 4, 31782–31794. [Google Scholar] [CrossRef]
- Lin, Y.; Pan, X.; Qi, W.; Zhang, B.; Su, D.S.J. Nitrogen-doped onion-like carbon: A novel and efficient metal-free catalyst for epoxidation reaction. Mater. Chem. A 2014, 2, 12475–12483. [Google Scholar] [CrossRef]
- Paredes, J.I.; Villar-Rodil, S.; Martínez-Alonso, A.; Tascón, J.M.D. Au/graphene hydrogel: Synthesis, characterization and its use for catalytic reduction of 4-nitrophenol. Langmuir 2008, 24, 10560–10564. [Google Scholar] [CrossRef] [PubMed]
- Alonso, D.A.; Nájera, C.; Pacheco, M.C. Highly Active Oxime-Derived Palladacycle Complexes for Suzuki−Miyaura and Ullmann-Type Coupling Reactions. J. Org. Chem. 2002, 67, 5588–5594. [Google Scholar] [CrossRef] [PubMed]
- Botella, L.; Nájera, C. A Convenient Oxime-Carbapalladacycle-Catalyzed Suzuki Cross-Coupling of Aryl Chlorides in Water. Angew. Chem. Int. Ed. 2002, 41, 179–181. [Google Scholar] [CrossRef]
- Kim, J.; Cote, L.J.; Kim, F.; Yuan, W.; Shull, K.R.; Huang, J. Graphene Oxide Sheets at Interfaces. J. Am. Chem. Soc. 2010, 132, 8180–8186. [Google Scholar] [CrossRef] [PubMed]
- Bedford, R.B.; Cazin, C.S.J.; Coles, S.J.; Gelbrich, T.; Horton, P.N.; Hursthouse, M.B.; Light, M.E. High-Activity Catalysts for Suzuki Coupling and Amination Reactions with Deactivated Aryl Chloride Substrates: Importance of the Palladium Source. Organometallics 2003, 22, 987–999. [Google Scholar] [CrossRef]
- Rosner, T.; Le Bars, J.; Pfaltz, A.; Blackmond, D.G. Kinetic Studies of Heck Coupling Reactions Using Palladacycle Catalysts: Experimental and Kinetic Modeling of the Role of Dimer Species. J. Am. Chem. Soc. 2001, 123, 1848–1855. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.J.; Zhang, J.; Zhao, X.; Xu, L.L.; Lyu, Z.Y.; Laia, M.; Chen, W. Palladium nanoparticle functionalized graphene nanosheets for Li–O2 batteries: Enhanced performance by tailoring the morphology of the discharge product. RSC Adv. 2015, 5, 73451–73456. [Google Scholar] [CrossRef]
- Chua, C.K.; Pumera, M. Chemical reduction of graphene oxide: A synthetic chemistry viewpoint. Chem. Soc. Rev. 2014, 43, 291–312. [Google Scholar] [CrossRef] [PubMed]
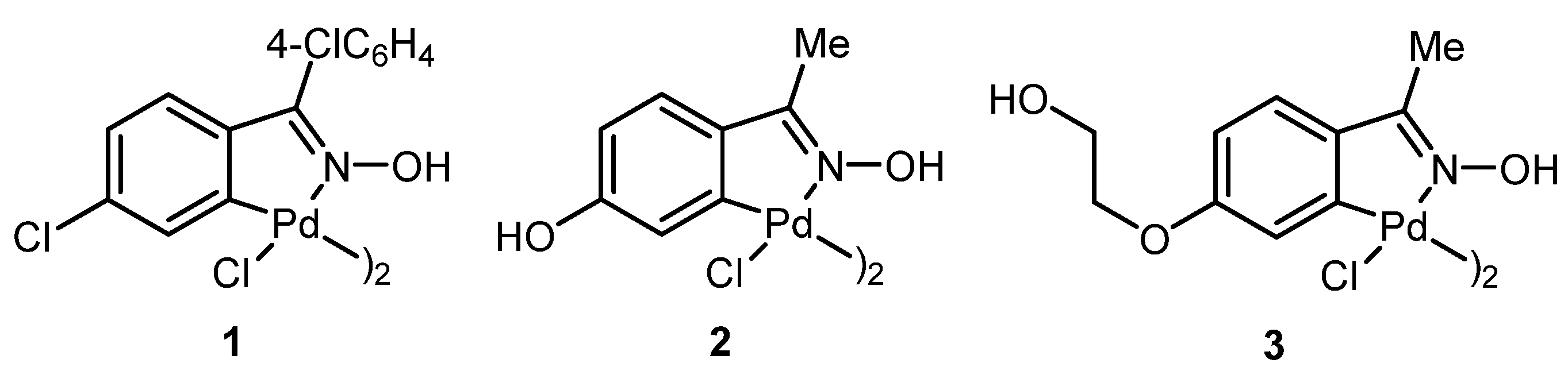
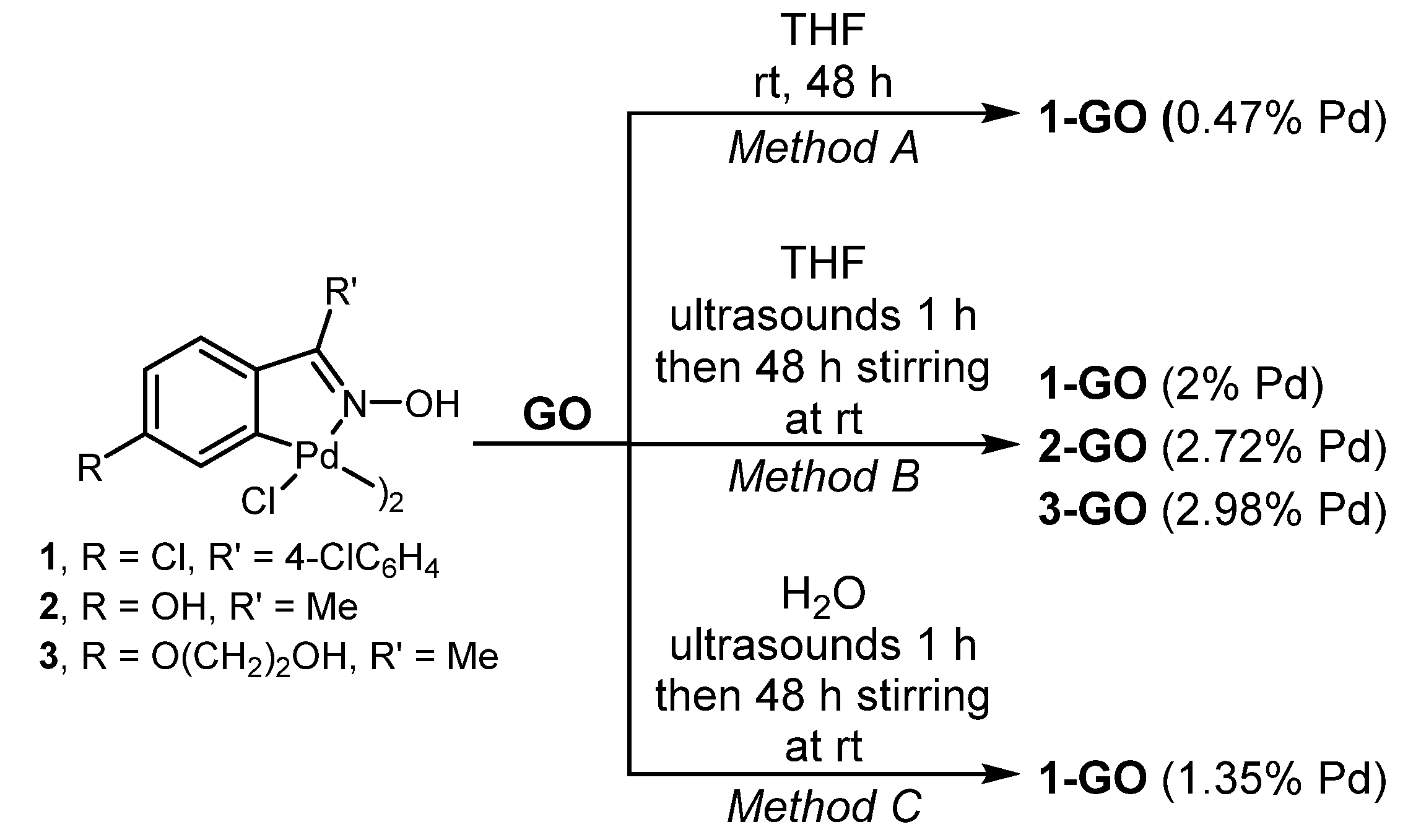

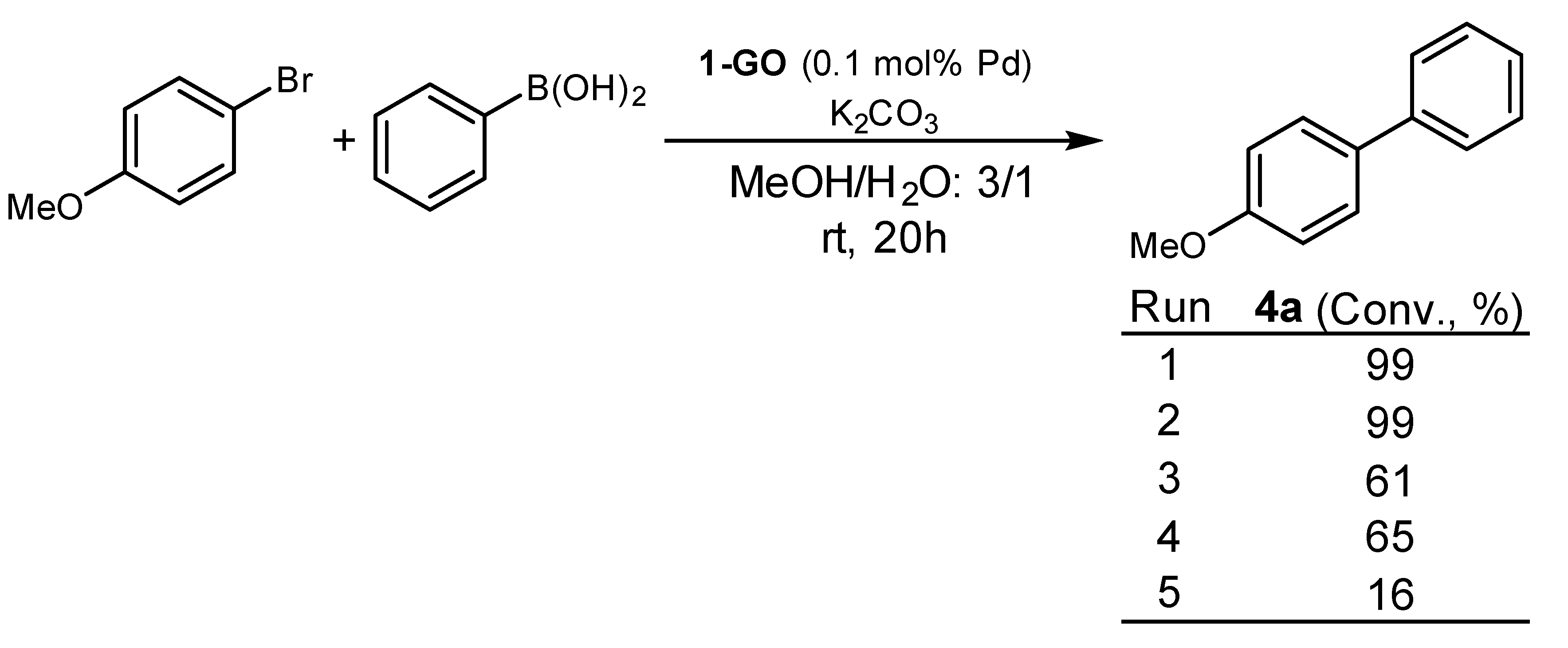
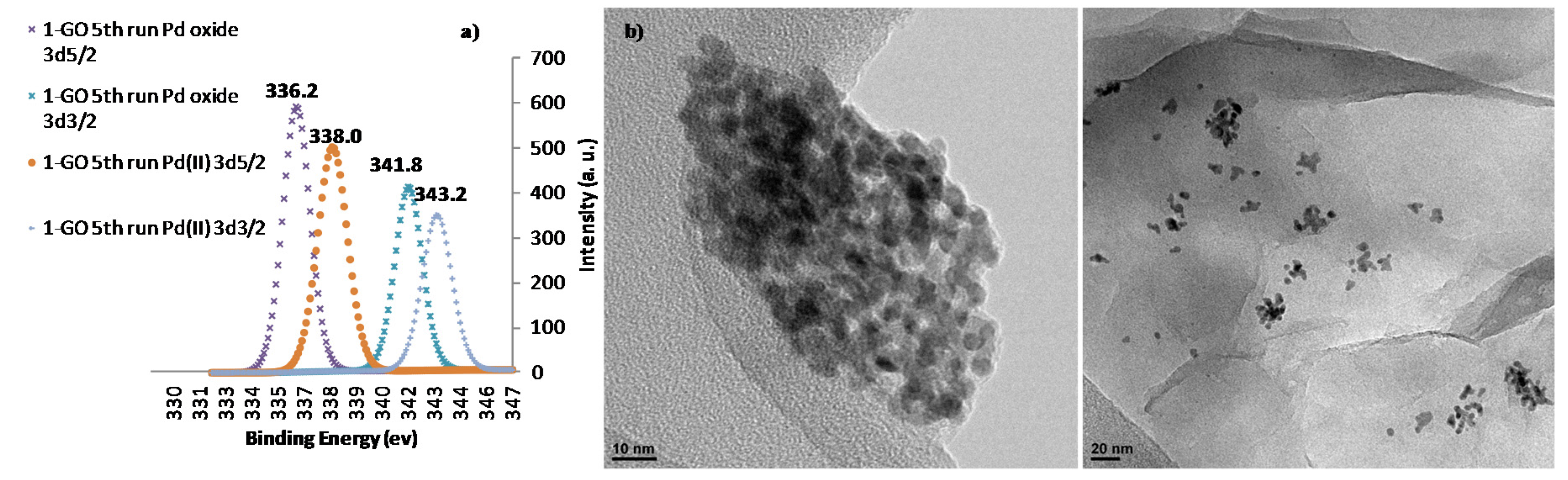

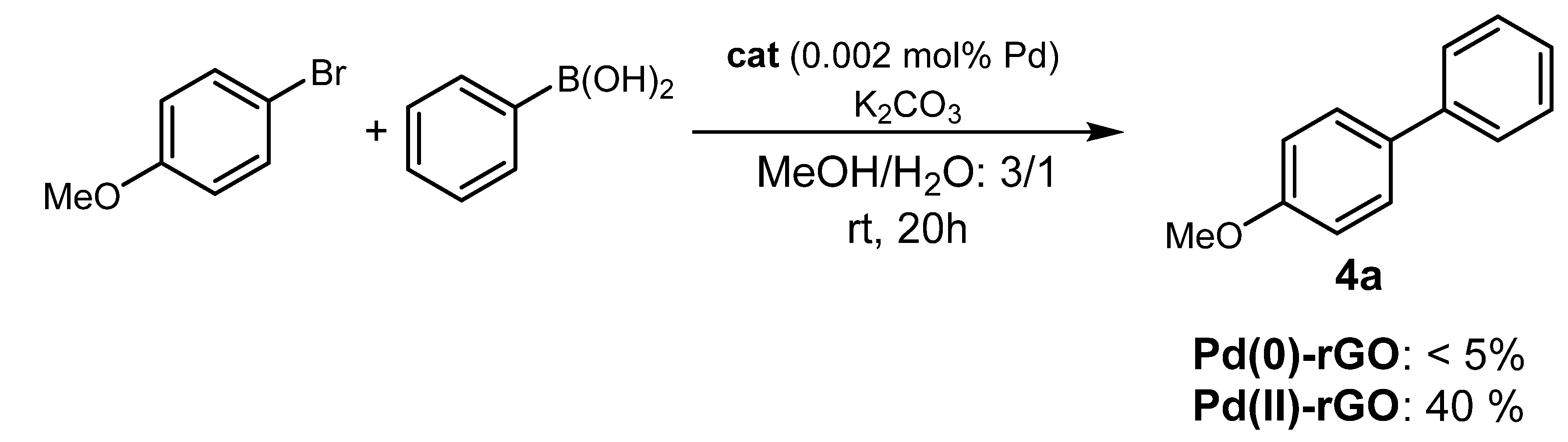
| Entry | Pd Catalyst | Mol % Pd | 4a (Conv., %) 1 |
|---|---|---|---|
| 1 | GO | - | 0 |
| 2 | 1 | 0.02 | 83 |
| 3 | 2 | 0.02 | 80 |
| 4 | 3 | 0.02 | 99 |
| 5 | 1-GO | 0.02 | 85 |
| 6 | 2-GO | 0.02 | 69 |
| 7 | 3-GO | 0.02 | 86 |
| 8 | 5 | 0.02 | 80 |
| 9 | 5-GO | 0.02 | 81 |
| 10 | PdNPs-G | 0.02 | 30 |
| 11 | PdNPs-rGO | 0.02 | 40 |
| 12 | 1-GO | 0.002 | 99 (73) |
| 13 | 2-GO | 0.002 | 45 |
| 14 | 3-GO | 0.002 | 33 |
| 15 | 5-GO | 0.002 | 15 |
| Entry | R | R’ | BX | Product | Yield (%) 2 | TON |
|---|---|---|---|---|---|---|
| 1 | 4-OMe | H | B(OH)2 | 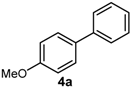 | 73 | 36,500 |
| 2 | 4-OMe | H | BF3K | 33 | 16,500 | |
| 3 | 4-OMe | H | B(OCMe2)2 | 27 | 13,500 | |
| 4 | 4-OMe | H | B(OCOCH2)2NMe | 5 | - | |
| 5 | 4-OH | H | B(OH)2 |  | 61 | 30,500 |
| 6 | 4-Ac | H | B(OH)2 |  | 66 | 33,000 |
| 7 | H | 4-Me | B(OH)2 | 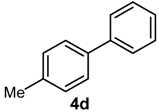 | 94 | 47,000 |
| 8 | 2-Me | H | B(OH)2 | 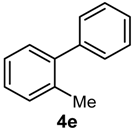 | 42 | 21,000 |
| 9 | 4-OMe | 2,6-(Me)2 | B(OH)2 |  | 5 | - |
| 10 | 4-OMe | 4-Me | B(OH)2 | 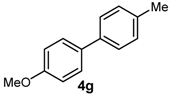 | 68 | 34,000 |
| 11 | 4-OMe | 4-CF3 | B(OH)2 |  | 12 | 6000 |
| 12 | 4-OMe | -3 | B(OH)2 | 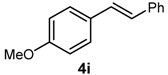 | 35 | 17,500 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gómez-Martínez, M.; Baeza, A.; Alonso, D.A. Graphene Oxide-Supported Oxime Palladacycles as Efficient Catalysts for the Suzuki–Miyaura Cross-Coupling Reaction of Aryl Bromides at Room Temperature under Aqueous Conditions. Catalysts 2017, 7, 94. https://doi.org/10.3390/catal7030094
Gómez-Martínez M, Baeza A, Alonso DA. Graphene Oxide-Supported Oxime Palladacycles as Efficient Catalysts for the Suzuki–Miyaura Cross-Coupling Reaction of Aryl Bromides at Room Temperature under Aqueous Conditions. Catalysts. 2017; 7(3):94. https://doi.org/10.3390/catal7030094
Chicago/Turabian StyleGómez-Martínez, Melania, Alejandro Baeza, and Diego A. Alonso. 2017. "Graphene Oxide-Supported Oxime Palladacycles as Efficient Catalysts for the Suzuki–Miyaura Cross-Coupling Reaction of Aryl Bromides at Room Temperature under Aqueous Conditions" Catalysts 7, no. 3: 94. https://doi.org/10.3390/catal7030094
APA StyleGómez-Martínez, M., Baeza, A., & Alonso, D. A. (2017). Graphene Oxide-Supported Oxime Palladacycles as Efficient Catalysts for the Suzuki–Miyaura Cross-Coupling Reaction of Aryl Bromides at Room Temperature under Aqueous Conditions. Catalysts, 7(3), 94. https://doi.org/10.3390/catal7030094