Catalytic Transfer Hydrogenation of Biobased HMF to 2,5-Bis-(Hydroxymethyl)Furan over Ru/Co3O4
Abstract
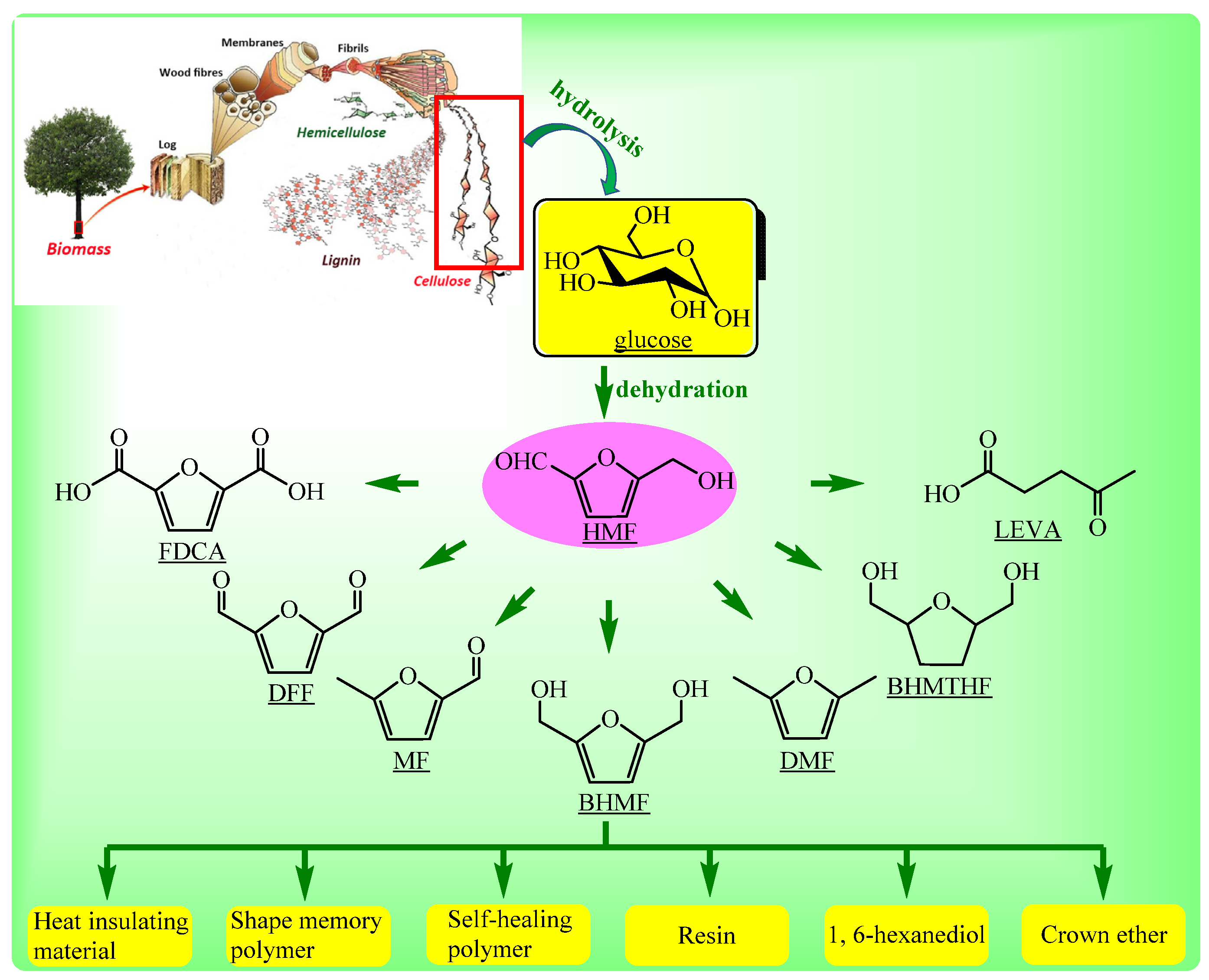
:1. Introduction
2. Results and Discussion
2.1. CTH of HMF to BHMF
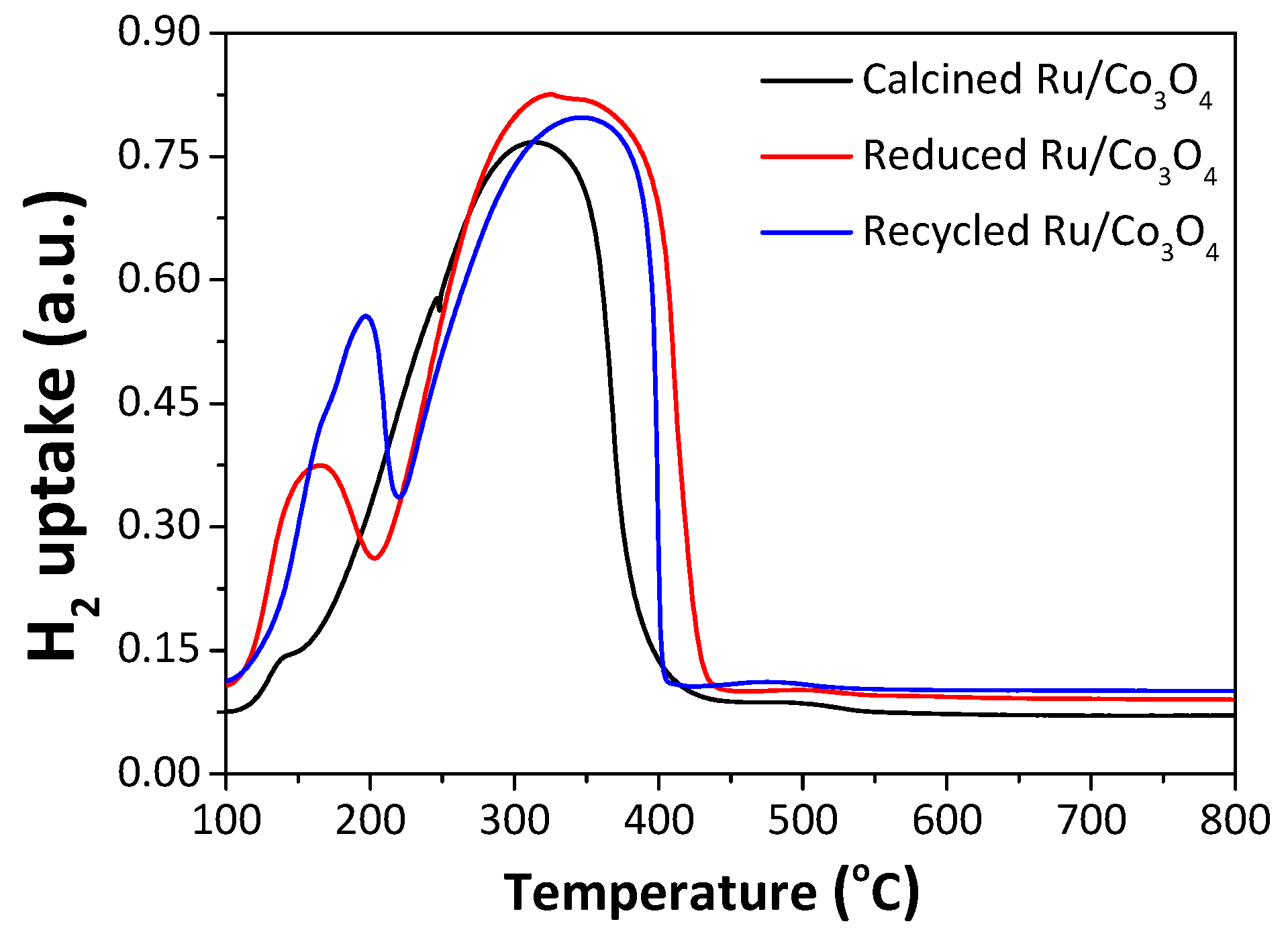
2.2. H2-TPR Profiles
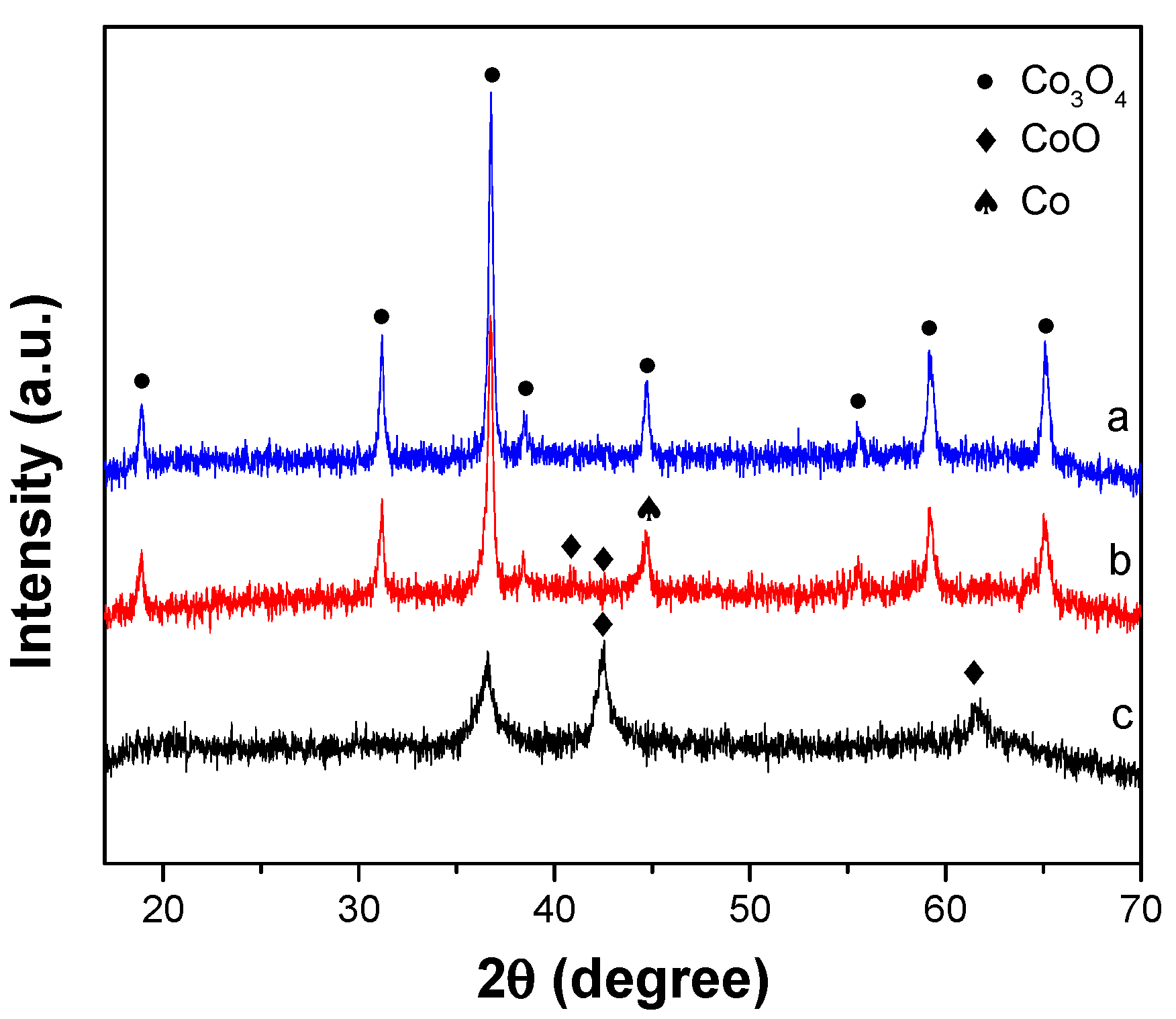
2.3. XRD Patterns
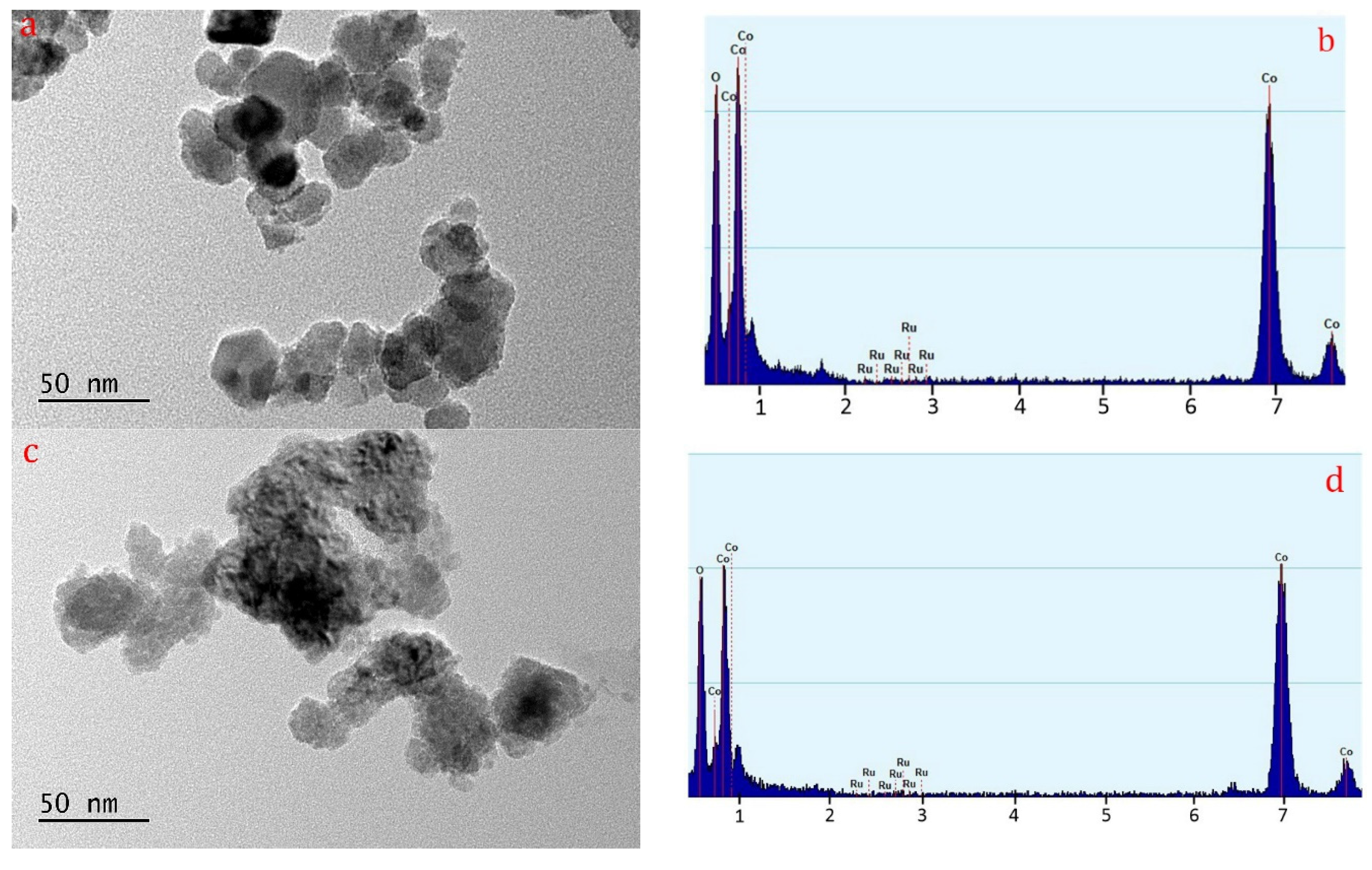
2.4. TEM Images
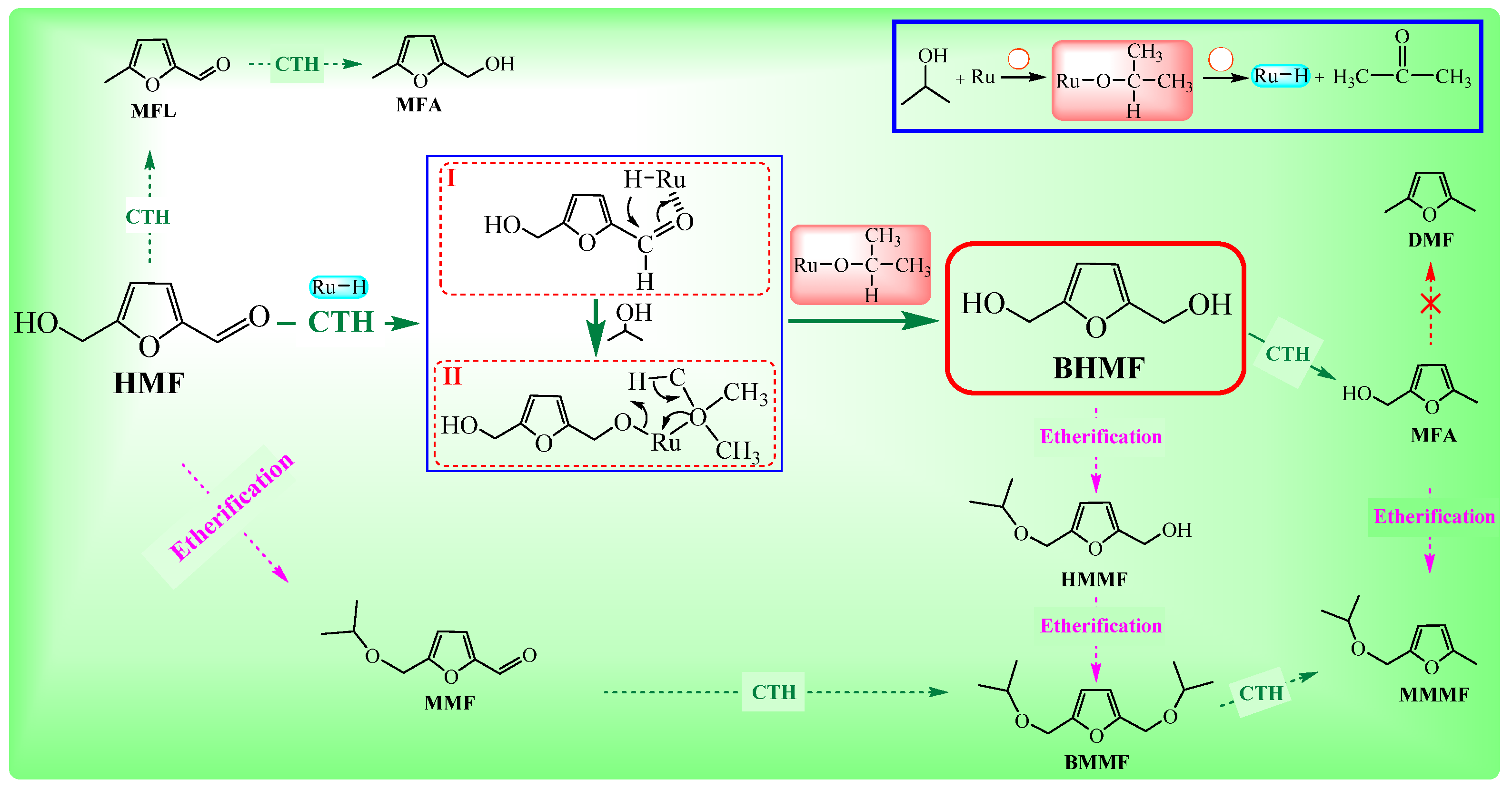
3. Reaction Processes for the CTH of HMF to BHMF
4. Materials and Methods
4.1. Chemicals and Materials
4.2. Catalyst Preparation
4.3. Catalytic Activity Tests
4.4. Catalyst Characterization
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Patel, M.; Kumar, A. Production of renewable diesel through the hydroprocessing of lignocellulosic biomass-derived bio-oil: A review. Renew. Sustain. Energy Rev. 2016, 58, 1293–1307. [Google Scholar] [CrossRef]
- Qin, Y.; Yu, L.; Wu, R.; Yang, D.; Qiu, X.; Zhu, J. Biorefinery Lignosulfonates from Sulfite-Pretreated Softwoods as Dispersant for Graphite. ACS Sustain. Chem. Eng. 2016, 4, 2200–2205. [Google Scholar] [CrossRef]
- Saha, B.; Abu-Omar, M.M. Advances in 5-hydroxymethylfurfural production from biomass in biphasic solvents. Green Chem. 2014, 16, 24–38. [Google Scholar] [CrossRef]
- Cai, H.; Li, C.; Wang, A.; Zhang, T. Biomass into chemicals: One-pot production of furan-based diols from carbohydrates via tandem reactions. Catal. Today 2014, 234, 59–65. [Google Scholar] [CrossRef]
- Timko, J.M.; Cram, D.J. Furanyl unit in host compounds. J. Am. Chem. Soc. 1974, 96, 7159–7160. [Google Scholar] [CrossRef]
- Zeng, C.; Seino, H.; Ren, J.; Hatanaka, K.; Yoshie, N. Bio-based furan polymers with self-healing ability. Macromolecules 2013, 46, 1794–1802. [Google Scholar] [CrossRef]
- Kang, E-S.; Chae, D.W.; Kim, B.; Kim, Y.G. Efficient preparation of DHMF and HMFA from biomass-derived HMF via a Cannizzaro reaction in ionic liquids. J. Ind. Eng. Chem. 2012, 18, 174–177. [Google Scholar]
- Subbiah, S.; Simeonov, S.P.; Esperanca, J.M.S.S.; Rebelo, L.P.N.; Afonso, C.A.M. Direct transformation of 5-hydroxymethylfurfural to the building blocks 2,5-dihydroxymethylfurfural (DHMF) and 5-hydroxymethyl furanoic acid (HMFA) via Cannizzaro reaction. Green Chem. 2013, 15, 2849–2853. [Google Scholar] [CrossRef]
- Zhu, Y.; Kong, X.; Zheng, H.; Ding, G.; Zhu, Y.; Li, Y.W. Efficient synthesis of 2,5-dihydroxymethylfuran and 2,5-dimethylfuran from 5-hydroxymethylfurfural using mineral-derived Cu catalysts as versatile catalysts. Catal. Sci. Technol. 2015, 5, 4208–4217. [Google Scholar] [CrossRef]
- Chatterjee, M.; Ishizaka, T.; Kawanami, H. Selective hydrogenation of 5-hydroxymethylfurfural to 2,5-bis-(hydroxymethyl)furan using Pt/MCM-41 in an aqueous medium: A simple approach. Green Chem. 2014, 16, 4734–4739. [Google Scholar] [CrossRef]
- Yi, G.S.; Zhang, Y.G. One-Pot Selective Conversion of Hemicellulose (Xylan) to Xylitol under Mild Conditions. ChemSusChem 2012, 5, 1383–1387. [Google Scholar] [CrossRef] [PubMed]
- Hansen, T.S.; Barta, K.; Anastas, P.T.; Ford, P.C.; Riisager, A. One-pot reduction of 5-hydroxymethylfurfural via hydrogen transfer from supercritical methanol. Green Chem. 2012, 14, 2457–2461. [Google Scholar] [CrossRef]
- Jae, J.; Zheng, W.Q.; Lobo, R.F.; Vlachos, D.G. Production of Dimethylfuran from Hydroxymethylfurfural through Catalytic Transfer Hydrogenation with Ruthenium Supported on Carbon. ChemSusChem 2013, 6, 1158–1162. [Google Scholar] [CrossRef] [PubMed]
- Thananatthanachon, T.; Rauchfuss, T.B. Efficient Production of the Liquid Fuel 2,5-Dimethylfuran from Fructose Using Formic Acid as a Reagent. Angew. Chem. Int. Ed. 2010, 49, 6616–6618. [Google Scholar] [CrossRef] [PubMed]
- Zu, Y.; Yang, P.; Wang, J.; Liu, X.; Ren, J.; Lu, G.; Wang, Y. Efficient production of the liquid fuel 2,5-dimethylfuran from 5-hydroxymethylfurfural over Ru/Co3O4 catalyst. Appl. Catal. B Environ. 2014, 146, 244–248. [Google Scholar] [CrossRef]
- Yu, J.; Zhao, D.; Xu, X.; Wang, X.; Zhang, N. Study on RuO2/SnO2: Novel and active catalysts for CO and CH4 oxidation. ChemCatChem 2012, 4, 1122–1132. [Google Scholar] [CrossRef]
- Harrison, P.G.; Ball, I.K.; Daniell, W.; Lukinskas, P.; Céspedes, M.; Miró, E.E.; Ulla, M. Cobalt catalysts for the oxidation of diesel soot particulate. Chem. Eng. J. 2003, 95, 47–55. [Google Scholar] [CrossRef]
- Guiral, V.; Delbecq, F.; Sautet, P. Origin of the Enantioselectivity in the Hydrogen Transfer Reduction of Carbonyls by a Rhodium(I) Complex: A Theoretical Study. J. Am. Chem. Soc. 2001, 20, 2207–2214. [Google Scholar] [CrossRef]
- Xue, L.; Zhang, C.; He, H.; Teraoka, Y. Catalytic decomposition of N2O over CeO2 promoted Co3O4 spinel catalyst. Appl. Catal. B Environ. 2007, 75, 167–174. [Google Scholar] [CrossRef]
- Liu, H.; Xu, J.; Zheng, Z.; Li, S.; Yao, M. Effects of fuel properties on combustion and emissions under both conventional and low temperature combustion mode fueling 2,5-dimethylfuran/diesel blends. Energy 2013, 62, 215–223. [Google Scholar] [CrossRef]
| Entry | T (°C) | t (h) | Conv. (%) | Yield % | |
|---|---|---|---|---|---|
| BHMF | MFA | ||||
| 1 | 130 | 10 | 37.1 | 24.4 | - |
| 2 | 150 | 10 | 66.7 | 51.7 | - |
| 3 | 170 | 10 | 94.4 | 80.3 | - |
| 4 | 190 | 10 | 100 | 79.3 | 9.0 |
| 5 | 210 | 10 | 100 | 67.7 | 17.2 |
| 6 | 190 | 6 | 100 | 82.8 | - |
| 7 | 190 | 8 | 100 | 79.9 | 7.7 |
| 8 * | 170 | 10 | 95.3 | 75.2 | - |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, T.; Zhang, J.; Xie, W.; Tang, Y.; Guo, D.; Ni, Y. Catalytic Transfer Hydrogenation of Biobased HMF to 2,5-Bis-(Hydroxymethyl)Furan over Ru/Co3O4. Catalysts 2017, 7, 92. https://doi.org/10.3390/catal7030092
Wang T, Zhang J, Xie W, Tang Y, Guo D, Ni Y. Catalytic Transfer Hydrogenation of Biobased HMF to 2,5-Bis-(Hydroxymethyl)Furan over Ru/Co3O4. Catalysts. 2017; 7(3):92. https://doi.org/10.3390/catal7030092
Chicago/Turabian StyleWang, Ting, Junhua Zhang, Wenxing Xie, Yanjun Tang, Daliang Guo, and Yonghao Ni. 2017. "Catalytic Transfer Hydrogenation of Biobased HMF to 2,5-Bis-(Hydroxymethyl)Furan over Ru/Co3O4" Catalysts 7, no. 3: 92. https://doi.org/10.3390/catal7030092
APA StyleWang, T., Zhang, J., Xie, W., Tang, Y., Guo, D., & Ni, Y. (2017). Catalytic Transfer Hydrogenation of Biobased HMF to 2,5-Bis-(Hydroxymethyl)Furan over Ru/Co3O4. Catalysts, 7(3), 92. https://doi.org/10.3390/catal7030092




