1. Introduction
Cellulases are classified as hydrolytic enzymes [
1] and include at least three types of biocatalysts: endo-(1,4)-
β-
d-glucanase (EC 3.2.1.4), exo-(1,4)-
β-
d-glucanase (EC 3.2.1.91), and
β-glucosidases (EC 3.2.1.21) [
2,
3,
4]. They are produced by bacteria and microbes; however, the main enzyme acquisition process is based on fungi, which provide the highest production index [
1,
5,
6]. The most frequently used for the production of cellulases are strains of cellulolytic fungi such as
Aspergillus,
Humicola,
Penicillium, and
Trichoderma [
7,
8]. Cellulases are responsible for the process of depolymerization of cellulose by delamination of the cell walls, which is a consequence of cellulose hydrolysis [
1,
6]. These biocatalysts have numerous practical applications in many fields of industry and agriculture. Commercial cellulases have been available for several decades, and have found applications in several important branches of industry, particularly in the wood and cellulose-paper industries, as well as in other branches including the conservation of thermoplastic polymers and plastics, bioconversion of cellulosic materials to organic solvents, fermentation processes, detergents, textiles, laundry, and the food and feed industries [
9]. In view of the broad application of these enzymes, it is necessary to develop novel and more stable materials to enable the aforementioned processes to be carried out more efficiently.
Immobilization is a technique of confining cells or enzymes on organic, inorganic, or hybrid carriers [
10]. Depending on the method of immobilization, the binding of the biocatalyst may occur more or less permanently. This technique is intended to increase the efficiency of catalytic processes, reduce the associated costs, and improve the properties of enzymes [
10], mainly its stability at harsh reaction conditions, as variable reaction parameters might affect enzymes’ properties. Among others, the most important properties are pH and temperature, as protein denaturation is caused mainly by abovementioned parameters [
11]. Immobilization of enzymes enables the retention of catalytic activity, which results in the possibility of reusing the biocatalytic system. Moreover, easier and faster separation of immobilized enzymes from the reaction mixture makes them more attractive for industrial applications [
11,
12]. The immobilization of biocatalysts also prevents protein from entering the process in a mobile phase reactor containing the reagents, resulting in cleaner products [
13].
As has already been noted, immobilization processes are of great importance and are used in many branches of industry, mainly in the pharmaceutical, food, chemical, and biological industries, but also in research and implementation of new technological solutions. The use of immobilized enzymes might result in greater process efficiency compared with the use of free biocatalysts, by better utilizing the potential of raw materials [
11,
12,
13,
14]. Various enzyme immobilization methods have enabled the development of industrial technology on a larger scale, reducing the costs of production. At present, there are several well-known techniques that rely on physical or chemical interactions [
11,
14]. Immobilized enzymes are a subject of great interest, which will certainly grow, as the properties of many of the biocatalysts are not yet fully known. Furthermore, the possibility of their immobilization may contribute to a steady progress in their development and use in bioprocesses and in the field of biosensing [
15,
16].
The continuous development of technology helps obtain more effective and stable materials with targeted industrial applications. Nowadays, novel hybrid or composite material with immobilized enzymes, achieved by creating an innovative biocatalytic system, are increasingly used in many fields of industry, like pharmaceutical, chemical, and food industries. A combination of inorganic and organic components can provide advanced features such as better thermal and chemical resistance or electrokinetic and biological stability. For instance, Tao et al., used magnetic Fe
3O
4–SiO
2 nanoparticles for the selective immobilization of cellulase [
17]. The novel hybrid material, due to the influence of the metallic oxides, exhibited enhanced adsorption parameters as well as biocompatibility and bioactivity. Similar findings are reported by Velmurugan et al., who confirmed that a newly developed MgO–Fe
3O
4 material was suitable for cellulose immobilization [
18]. Furthermore, Hong et al., obtained a polyhedral oligomeric silsesquioxane-silica-titania (POSS–SiO
2–TiO
2) hybrid material for cellulase immobilization for biocatalytic applications [
19]. In other studies laccase was successfully immobilized on TiO
2 nanoparticles and TiO
2 nanoparticle functionalized polyethersulfone (PES) membranes. The results revealed that both the immobilization procedures and the properties of the immobilization supports have significant impacts on the biocatalyst performance [
20]. In addition, Hou et al. showed in [
21] great potential for the application of the TiO
2 based biocatalytic nanoparticles and membranes for CO
2 conversion in a gas–liquid membrane contactor. These examples show that it is extremely important to continue research into the development of novel support materials for enzyme immobilization, which will lead to the creation of more effective and stable biocatalytic systems.
The main research goal of the present study was to evaluate a functional hybrid titanium dioxide–lignin hybrid material in terms of its suitability for the immobilization of cellulase, and to use the resulting biocatalytic system in the hydrolysis process of cellulose. The synthesized hybrid support was concisely characterized, immobilization of the enzyme was confirmed, and the effect of various operational conditions on the enzyme’s stability and activity, as well as its reusability and storage stability, were examined.
2. Results
2.1. Synthesis of Titania–Lignin Hybrid Material and Cellulase Immobilization
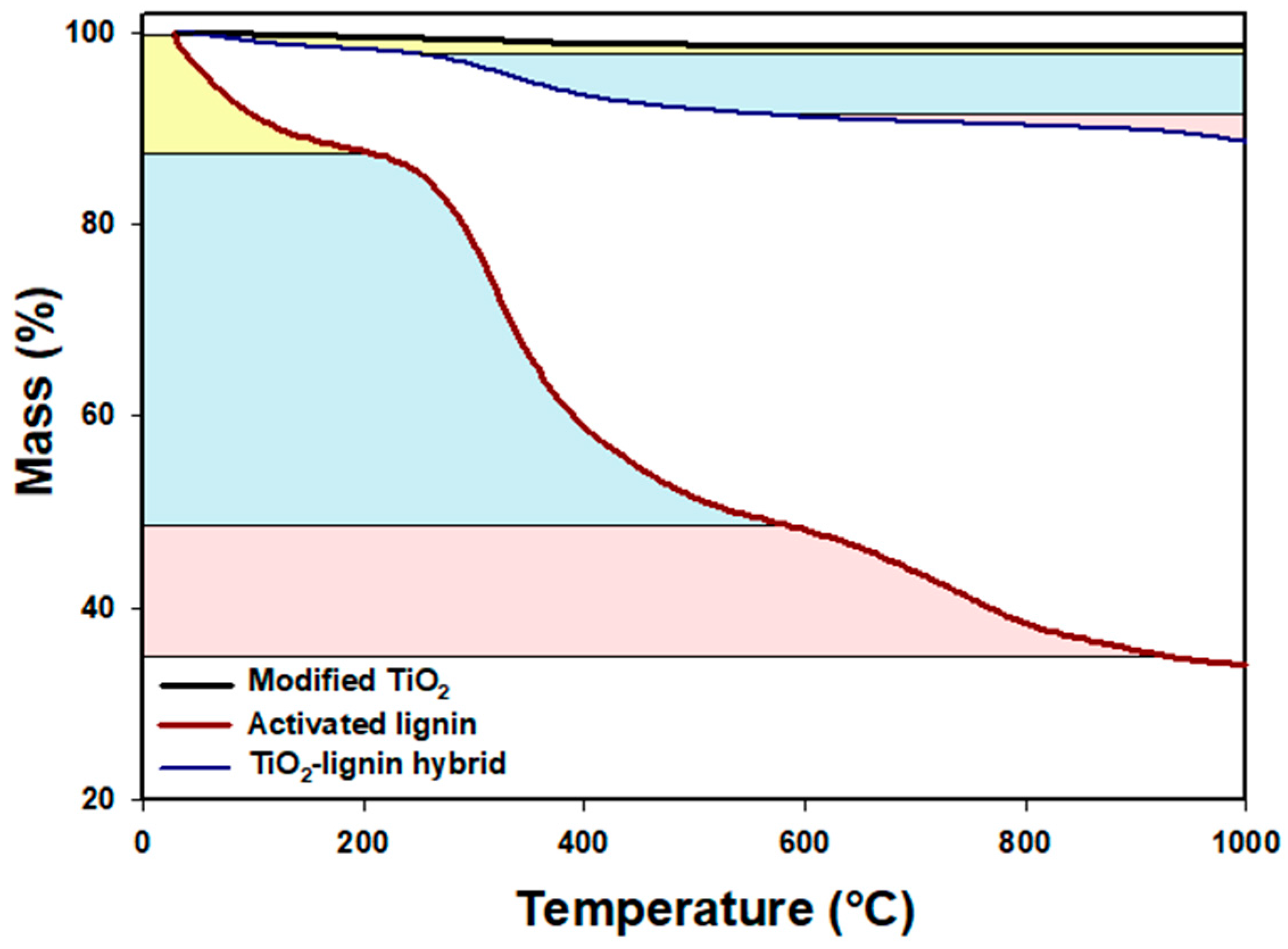
The thermal stability of the TiO
2 modified by poly-
l-lisine (PLL), lignin activated by sodium (meta)periodate and the synthesized TiO
2–lignin hybrid material was determined using thermogravimetric analysis (TGA) (
Figure 1). Commercial titanium dioxide after surface modification exhibited extremely high thermal stability, losing only about 2% of its mass in the analyzed temperature range, which might be explained by the evaporation of physically bound water. The thermogravimetric curve of the activated lignin showed a significant decrease in mass of the activated biopolymer consisted in three mass loss stages, at temperatures up to 200 °C (marked in yellow) and in the temperature ranges 200–600 °C (marked in blue) and 650–1000 °C (marked in red), linked respectively to the removal of physically bound water, decomposition of the lignin structure, and elimination of carbon and hydrogen atoms [
22]. The synthesized TiO
2–lignin hybrid showed relatively good thermal stability: up to a temperature of 1000 °C it lost about 10% of its mass. The high thermal stability (characteristic for TiO
2) and the shape of the TGA curve for the obtained material, similar to that recorded for lignin, also proved the effective connection of TiO
2 and lignin.
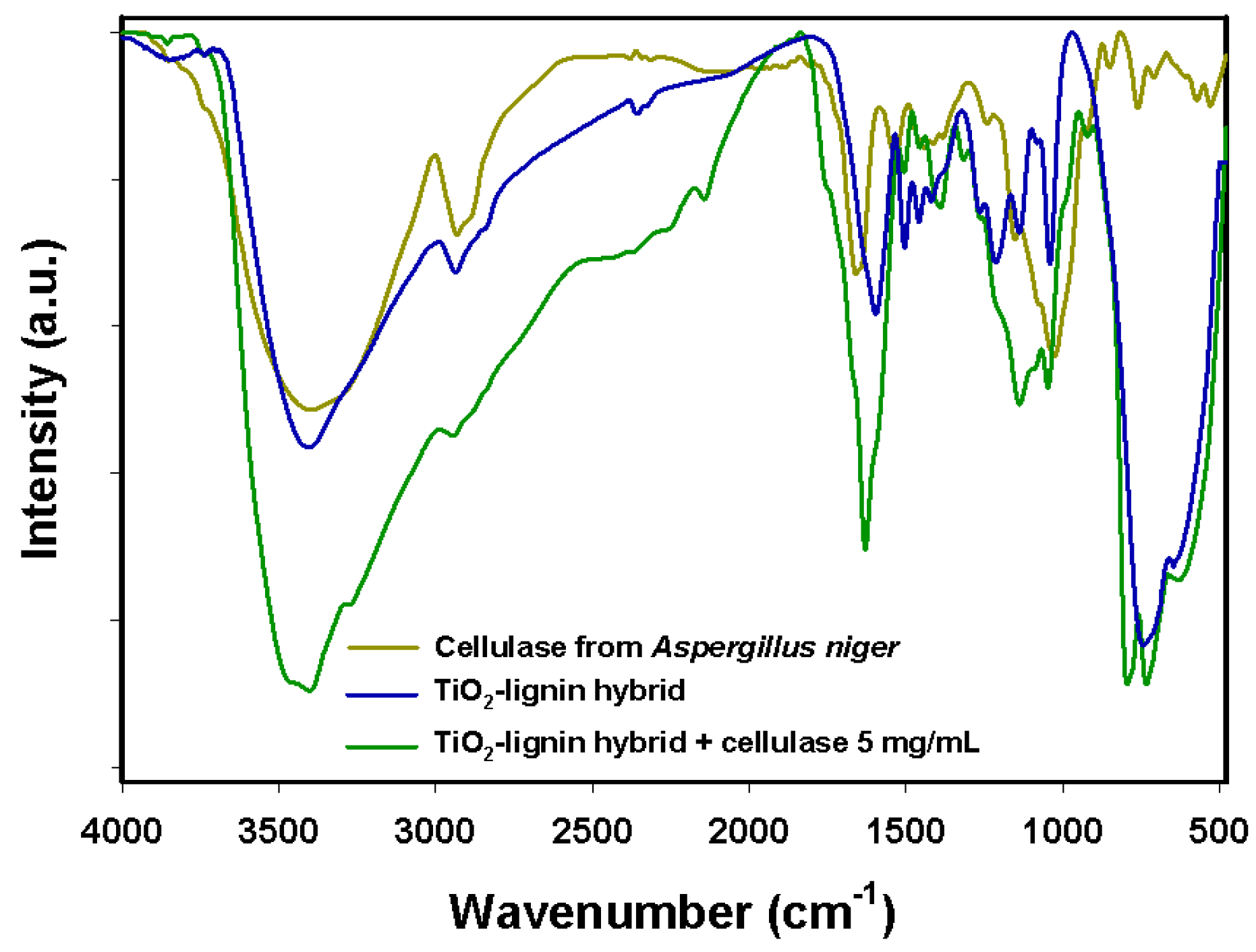
The Fourier-transform infrared (FTIR) spectrum of the TiO
2–lignin hybrid (
Figure 2) contained many signals characteristic for functional groups of both modified precursors, which indirectly confirmed the effective synthesis of the hybrid support. Among these signals, the most important are those at wavenumbers 3450 cm
−1, 2940 cm
−1, 1680 cm
−1, and around 1100 cm
−1, attributed respectively to stretching vibrations of –OH groups, C–H bonds, C=O groups, and C–O and C–O–C bonds in the lignin structure [
23]. Signals were also observed in the wavenumber range 1600–1450 cm
−1 and at 720 cm
−1, representing, respectively, stretching vibrations of C
Ar–C
Ar bonds in the structure of the biopolymer, and Ti–O–Ti bonds.
In the FTIR spectrum of free cellulase from Aspergillus niger, the most important signal is that at 3430 cm−1, characteristic for amine groups, which are mainly responsible for attachment of the enzyme to the solid support, as well as peaks around 1665 cm−1 and 1530 cm−1, generated by stretching vibrations of amide I and amide II bands. The FTIR spectrum of the produced biocatalytic system contains signals characteristic for cellulase as well as for the hybrid matrix. This suggested that the biocatalyst was successfully deposited on the surface of the support material. This is proven by the presence of a peak at 1090 cm−1 (C–N stretching vibrations) and changes in the intensity of signals generated by amine groups (3430 cm−1), C–H bonds (2930 cm−1), and amide bands, in comparison with the FTIR spectrum of the TiO2–lignin hybrid before enzyme binding.
Based also on the results of the analysis of the porous structure parameters of the TiO2–lignin support material before and after immobilization, some additional conclusions can be drawn. The synthesized TiO2–lignin hybrid before enzyme attachment had a relatively low surface area of around 10 m2/g, while for the matrix after cellulase immobilization, this parameter decreased to about 8 m2/g. This might suggest that effective enzyme attachment was achieved. Moreover, a decrease was recorded in the pores diameter and pores volume in the samples after immobilization. The hybrid biocomposite had pores with a mean diameter of 3.3 nm and a volume of 0.005 cm3/g, while after cellulase immobilization these parameters fell to 1.7 nm and 0.003 cm3/g.
2.2. Cellulase Immobilization
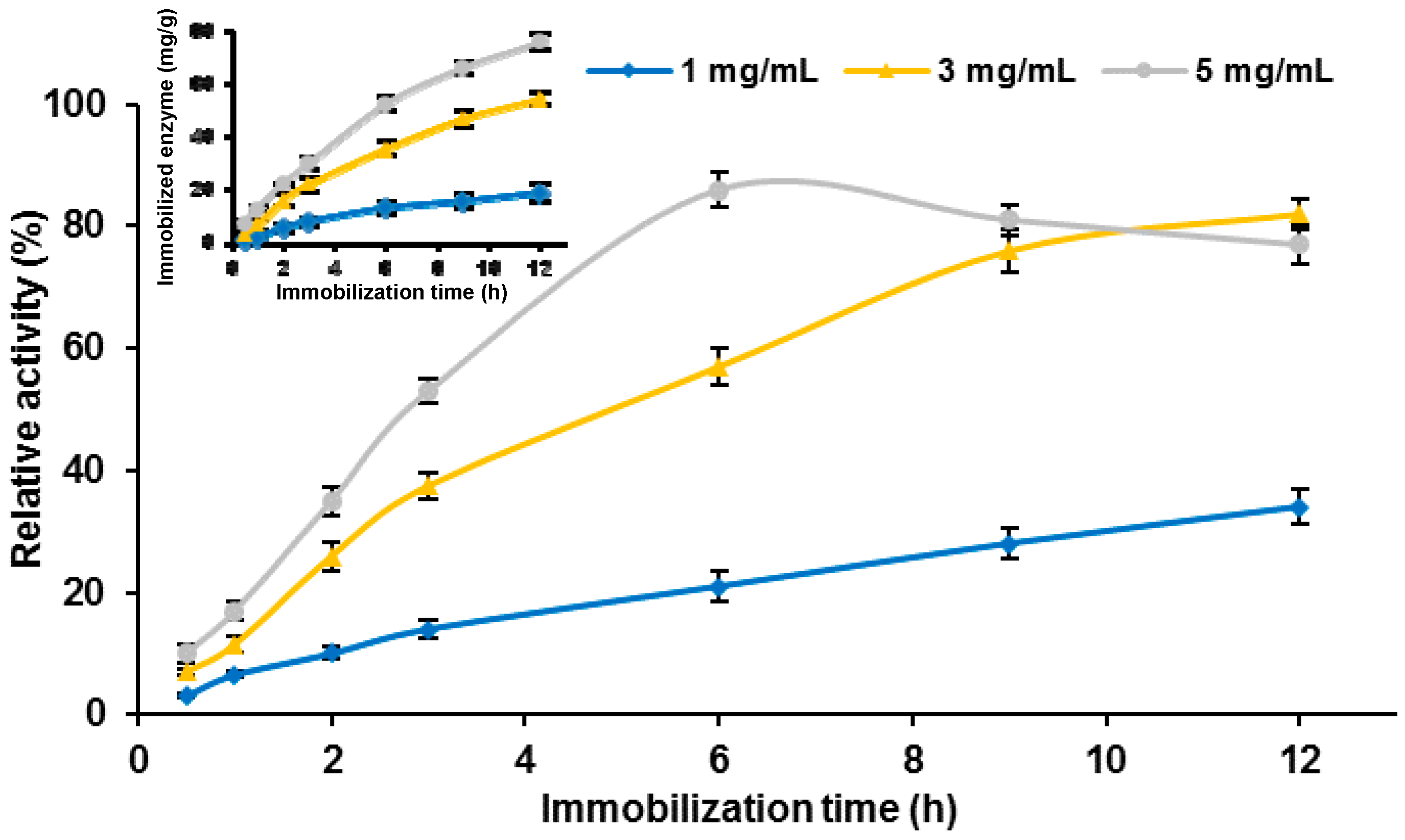
Besides confirmation of effective cellulase binding, the effect of initial immobilization parameters (process time and concentration of enzyme solution) on the quantity of immobilized enzyme and its relative activity was also evaluated (
Figure 3).
As
Figure 3 shows, the products after immobilization exhibited lower catalytic activity than free cellulase (relative activity up to 85%). When solution concentrations of 1 and 3 mg/mL were used, the highest relative activities, of around 30% and 80% respectively, were recorded after 12 h of the process. The amount of immobilized cellulase increased with process duration, irrespective of the concentration of the initial enzyme solution (
Figure 3 inset). After 12 h of immobilization, 19, 55, and 76 mg of the biocatalyst per 1 g of the support material was deposited from solutions of 1, 3, and 5 mg/mL respectively. The results indicated that with increasing process time, the relative activity of the immobilized enzyme increased along with the amount of bounded cellulase, except in the case of the 5 mg/mL solution. When cellulase solution at this concentration was employed, the highest relative activity, about 85%, was obtained after 6 h of immobilization. Further increase in the immobilization time caused the relative activity of the bound cellulase to decrease.
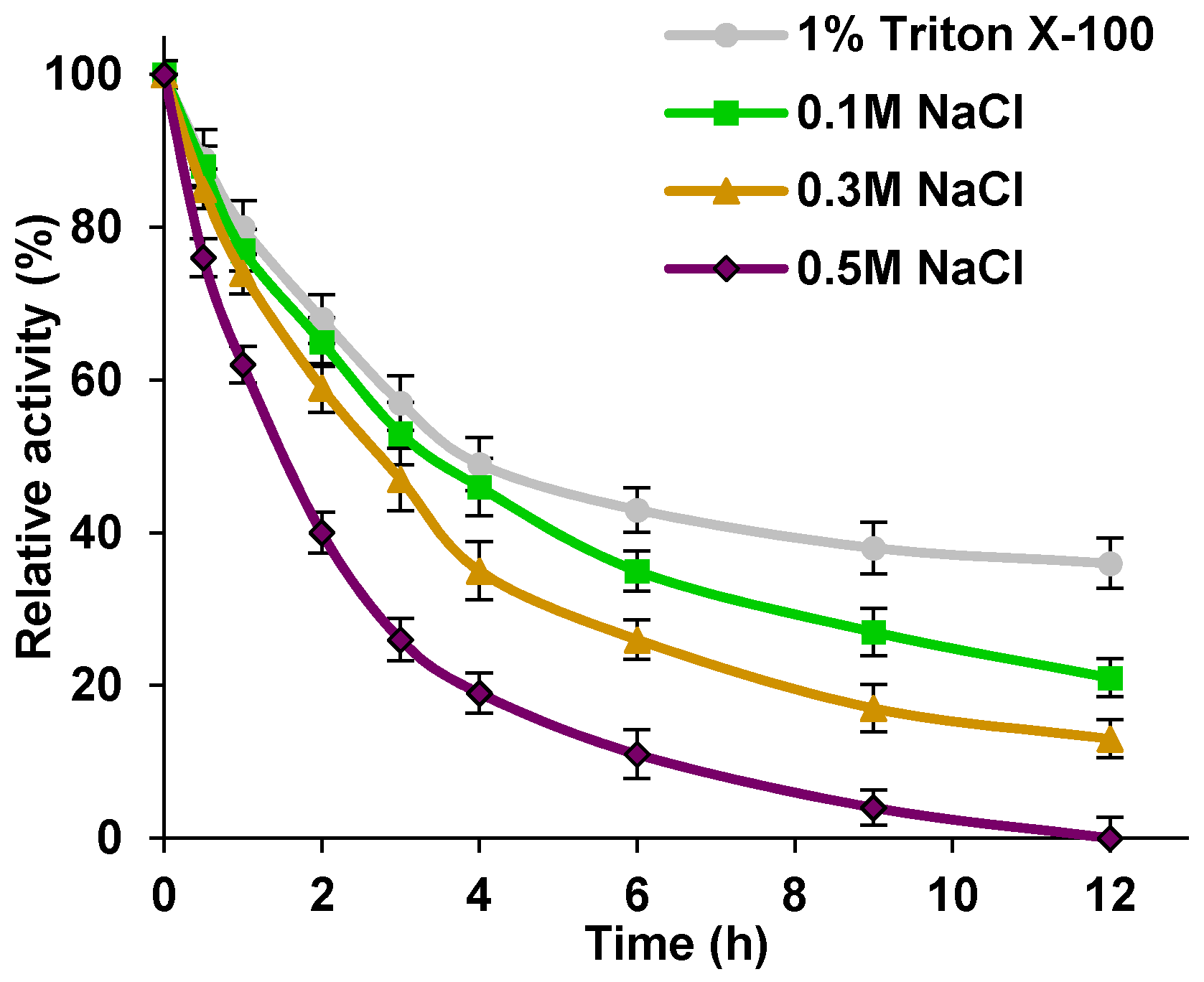
As has already been mentioned, the immobilization technique used led to the attachment of cellulase molecules mainly via physical and ionic interactions, but the formation of covalent bonds cannot be excluded. Electrokinetic measurements showed that the zeta potential of the titania–lignin hybrid matrix took negative values over the whole of the analyzed pH range, which indicates that the surface of the support material was negatively charged during immobilization. Under the immobilization conditions (acetate buffer at pH 4.8), the enzyme molecules were positively charged (the IEP of cellulase is around 5). These facts imply that effective biocatalyst immobilization occurred mainly via electrostatic and ionic interactions. To verify this statement, solutions of sodium chloride at various ionic strengths were applied in cellulase desorption tests, because this salt might elute the enzyme by way of ionic exchange (
Figure 4) [
24].
Figure 4 shows that the relative activity of the immobilized cellulase treated with 1% Triton X-100 and NaCl solutions at various molar concentrations declined gradually during the first 6 h of treatment, which suggests elution of the enzyme from the matrix. The further decrease in relative activity is insignificant (less than 10% in all cases), which indicates that desorption occurred in the initial stages of the test and is limited in its later stages. After treatment with 1% Triton X-100 for 12 h, the immobilized cellulase retained over 40% of its relative activity. When the immobilized biocatalyst was incubated in sodium chloride solutions at molarities of 0.1; 0.3, and 0.5 M, a more significant drop in the relative activity was observed. The produced systems finally exhibited 25%, 18%, and 0% of its relative activity, respectively. Additionally, to verify if the immobilized enzyme was eluted from the support material following the treatments or was deactivated by such treatments, amount of the immobilized enzyme retained on the matrix, after 6 and 12 h of desorption process was evaluated (
Table 1).
It can be seen from
Table 2, that amount of the immobilized enzyme that remained on the surface of the hybrid material after desorption, irrespectively of the type and ionic strength of the eluent, is about 10–20% higher than amount of the immobilized cellulase, which corresponds to the evaluated relative activity of the biocatalytic system after desorption. The biggest differences were noticed for 0.5 M NaCl solution. After 6 and 12 h of the process, the surface of the titania–lignin hybrid retained 9.7 and 5.1 mg of the enzyme per 1 g of the support. These values corresponded to the relative activity of 18.3% and 9.7%, meanwhile, the noticed values of relative activity were 11.2% and 0%.
2.3. Stability Study of Immobilized Cellulase
Various process parameters such as pH and temperature might affect the ability of the immobilized cellulase to degrade cellulosic material. Thus, in this study, the effect of the aforementioned parameters, as well as the chemical and thermal stability of the free and immobilized enzyme, were examined and compared. Moreover, the reusability and storage stability of the free and immobilized enzyme under different conditions were tested. Kinetic parameters for both forms of the enzyme were evaluated to verify how immobilization affected the affinity of the enzyme to the substrate molecules and its catalytic efficiency.
2.3.1. Effect of pH
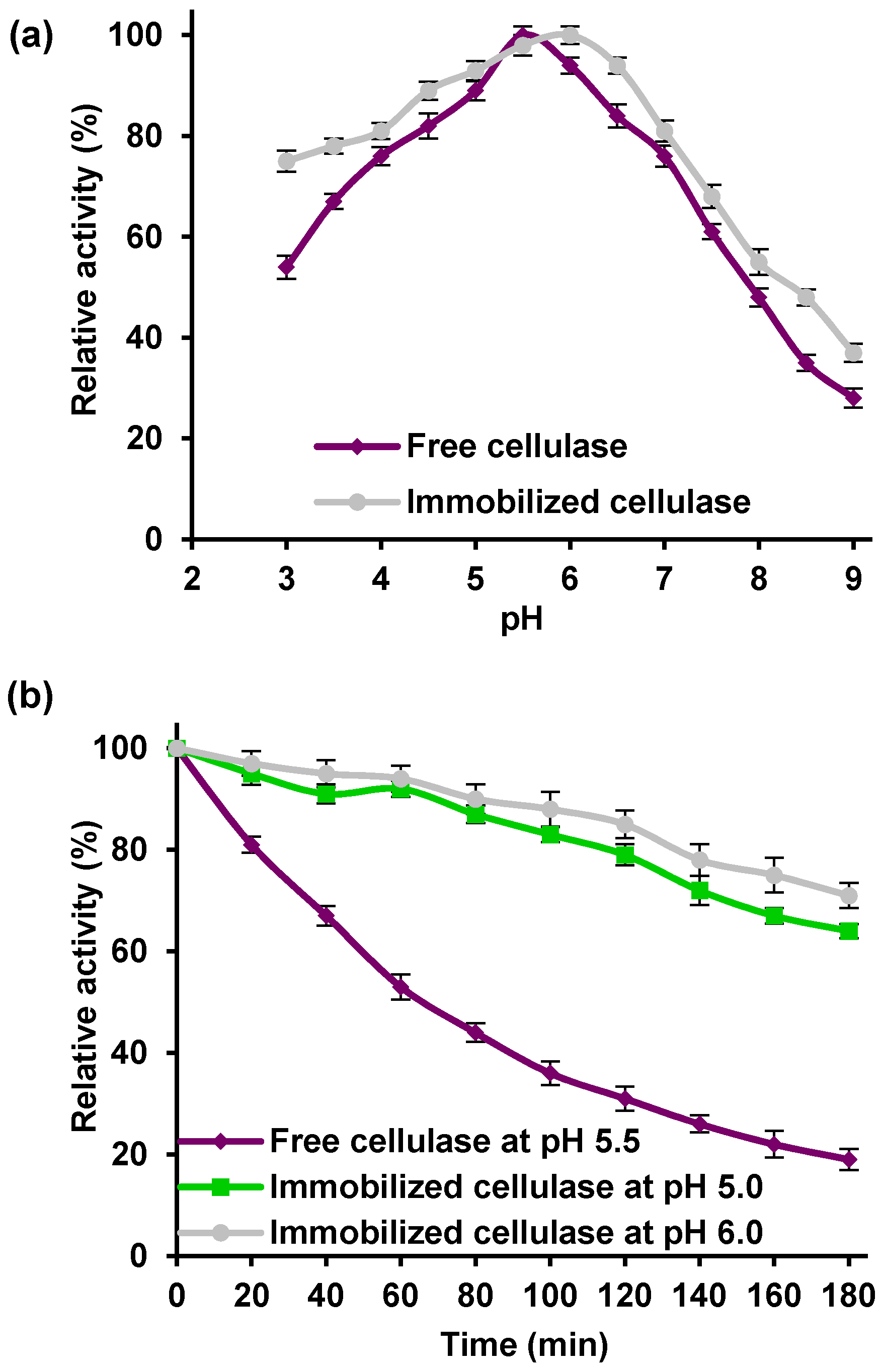
The effect of pH on the relative activity of free and immobilized cellulase was studied over a broad range of pH values, from 3.0 to 9.0 (
Figure 5a).
The maximum relative activity was recorded at pH 5.5 for the free enzyme, and at pH 6.0 for the immobilized enzyme, as expected, since acidic cellulase was used in this study. Although the graph showed similar trends for the free and immobilized biocatalyst over the whole of the analyzed pH range, the immobilized biomolecules have higher relative activity than the free ones in the same pH conditions. The immobilized cellulase exhibited over 80% of its relative activity at pH values from 4.5 to 6.0, as the free catalyst did so only at pH values from 5.0 to 6.0. The bound enzyme also exhibited about 20% higher relative activity (76%) than the free enzyme (54%) at pH 3.0. The results also indicated a significant decrease in cellulase activity when the pH is above or below its optimum value. This was especially visible in alkaline conditions: at pH 9.0, both biocatalysts retained less than 40% of their activity. To test the chemical stability of the free and immobilized cellulase, both enzymes were incubated for 3 h under their optimal reaction conditions (
Figure 5b). After 3 h, the free cellulase retained about 20% of its initial activity, as the immobilized enzymes incubated at pH 5.0 and 6.0 retained over 75% of their activity, which proved that cellulase attached to a TiO
2–lignin hybrid support has higher thermal stability than the free enzyme.
2.3.2. Effect of Temperature
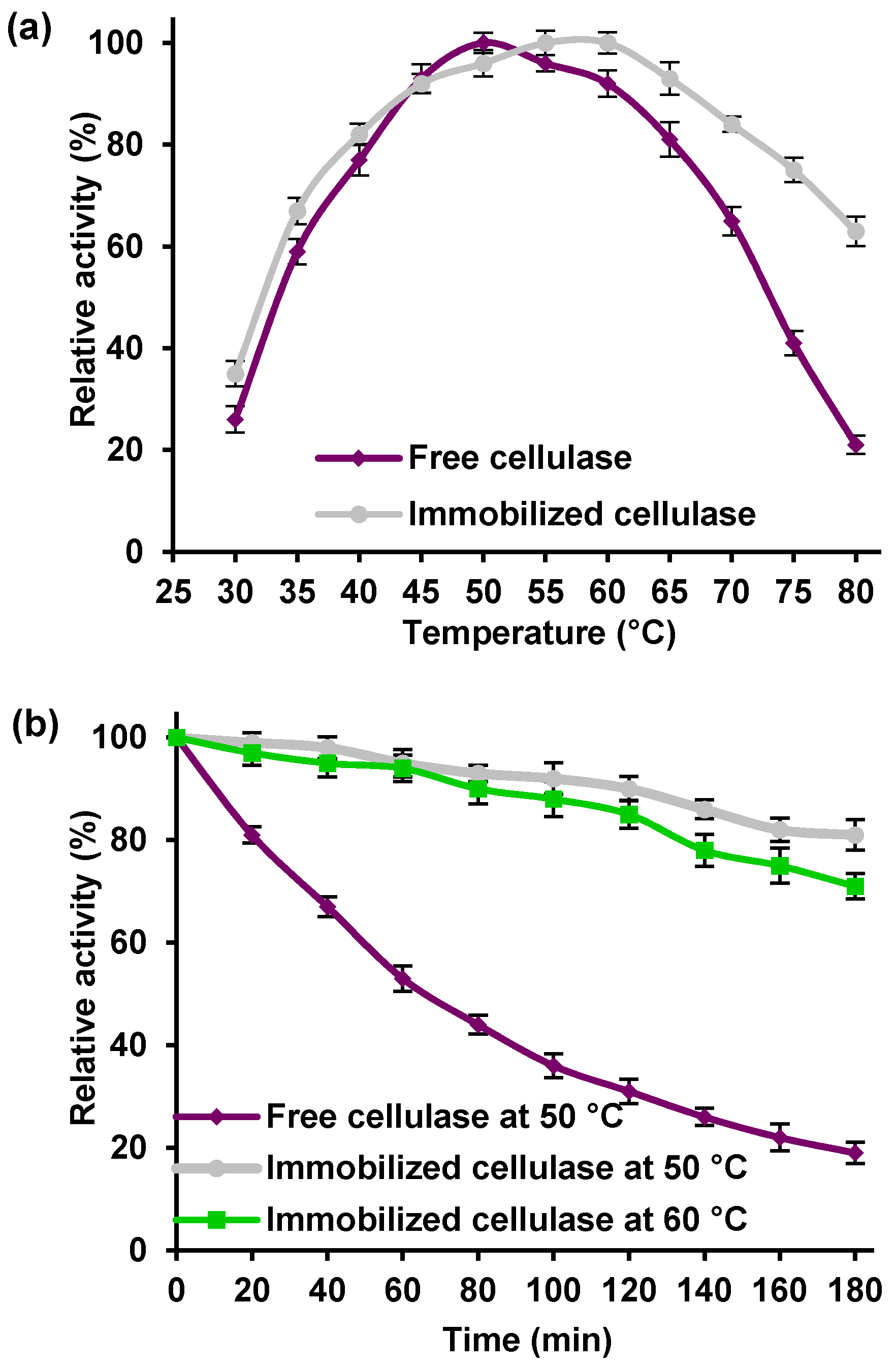
The effect of temperature on the relative activity of free and immobilized cellulase from
Aspergillus niger was studied between 30 and 80 °C, under optimal pH conditions for free and immobilized cellulase (
Figure 6a).
The free enzyme exhibited its maximum activity at 50 °C and retained 80% of its activity in a temperature range from 45 to 60 °C. At temperatures below 50 and above 60 °C the catalytic activity of the free biocatalyst significantly decreased, suggesting that the free enzyme is unstable in these conditions due to denaturation of the peptide structure [
25]. By comparison, the immobilized cellulase exhibited its highest activity at temperatures of 55 and 60 °C, and retained over 80% of its maximum activity over a wide temperature range from 40 to 70 °C. In addition, the drop in relative activity above 70 °C is less significant that in the case of the free enzyme, while at 80 °C the bound cellulase exhibited over 60% of its relative activity. To determine the thermal stability of the immobilized cellulase, the free enzyme was incubated for 3 h at 50 °C and the immobilized cellulase at 50 and 60 °C (
Figure 6b). After 3 h, the relative activity of the immobilized cellulase remained at a high level (over 75% and 80%, at temperatures of 50 and 60 °C, respectively). Meanwhile, there was more significant decline in the catalytic properties of the free enzyme, which after 3 h of incubation had lost almost 80% of its activity.
2.3.3. Storage Stability and Reusability
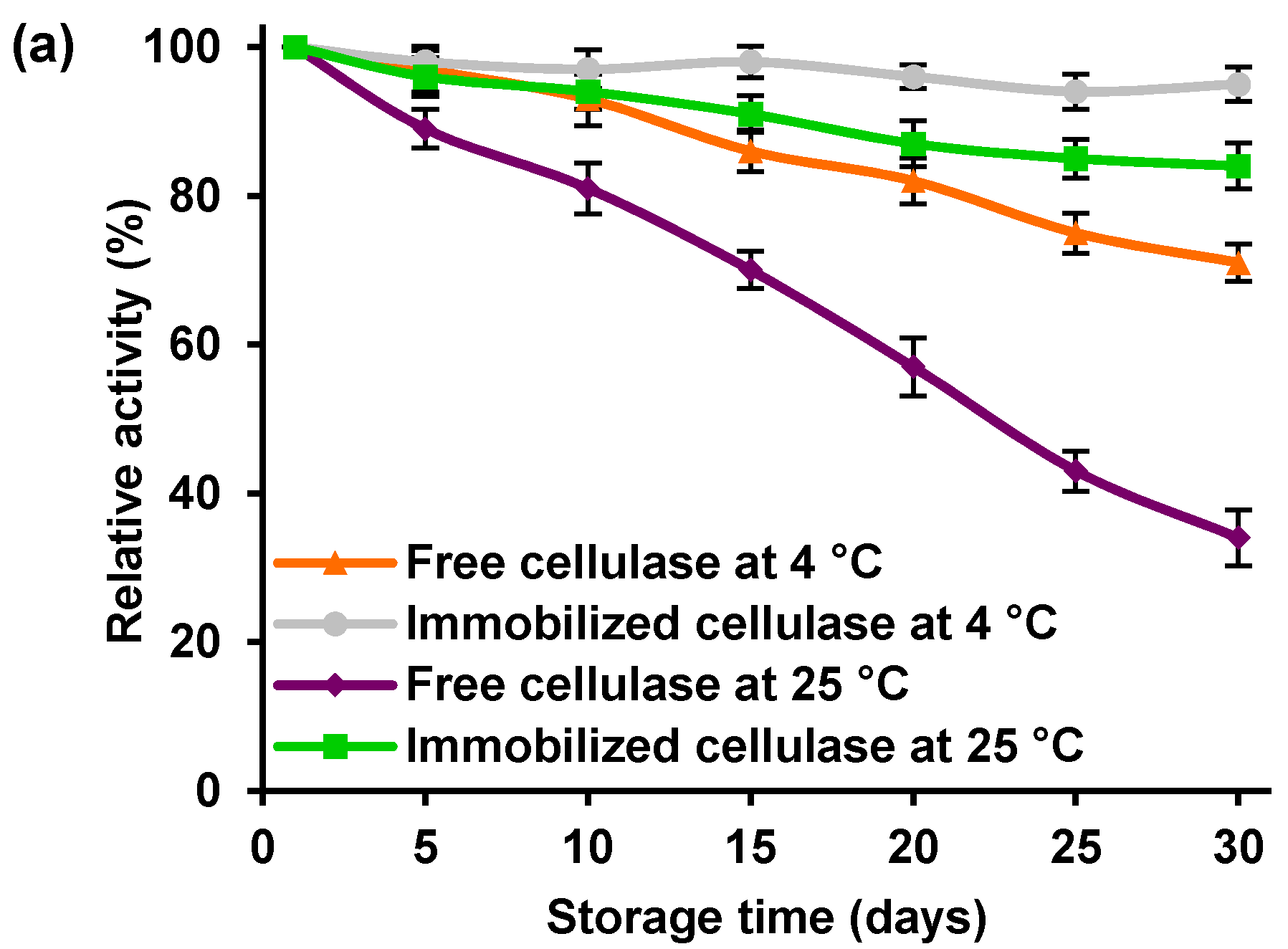
The storage stability of free and immobilized cellulase at 4 and 25 °C was evaluated by measuring the enzyme’s relative activity every 5 days for 30 days (
Figure 7a).
It can be seen that, irrespective of the storage temperature, the immobilized enzyme was characterized by higher activity than the free cellulase: after 30 days of storage at 4 and 25 °C, it retained over 95% and 85% of its initial activity, compared with less than 75% and less than 40% in the case of the free enzyme. These results indicated that the immobilization strategy used in this study improved the storage stability of the cellulase.
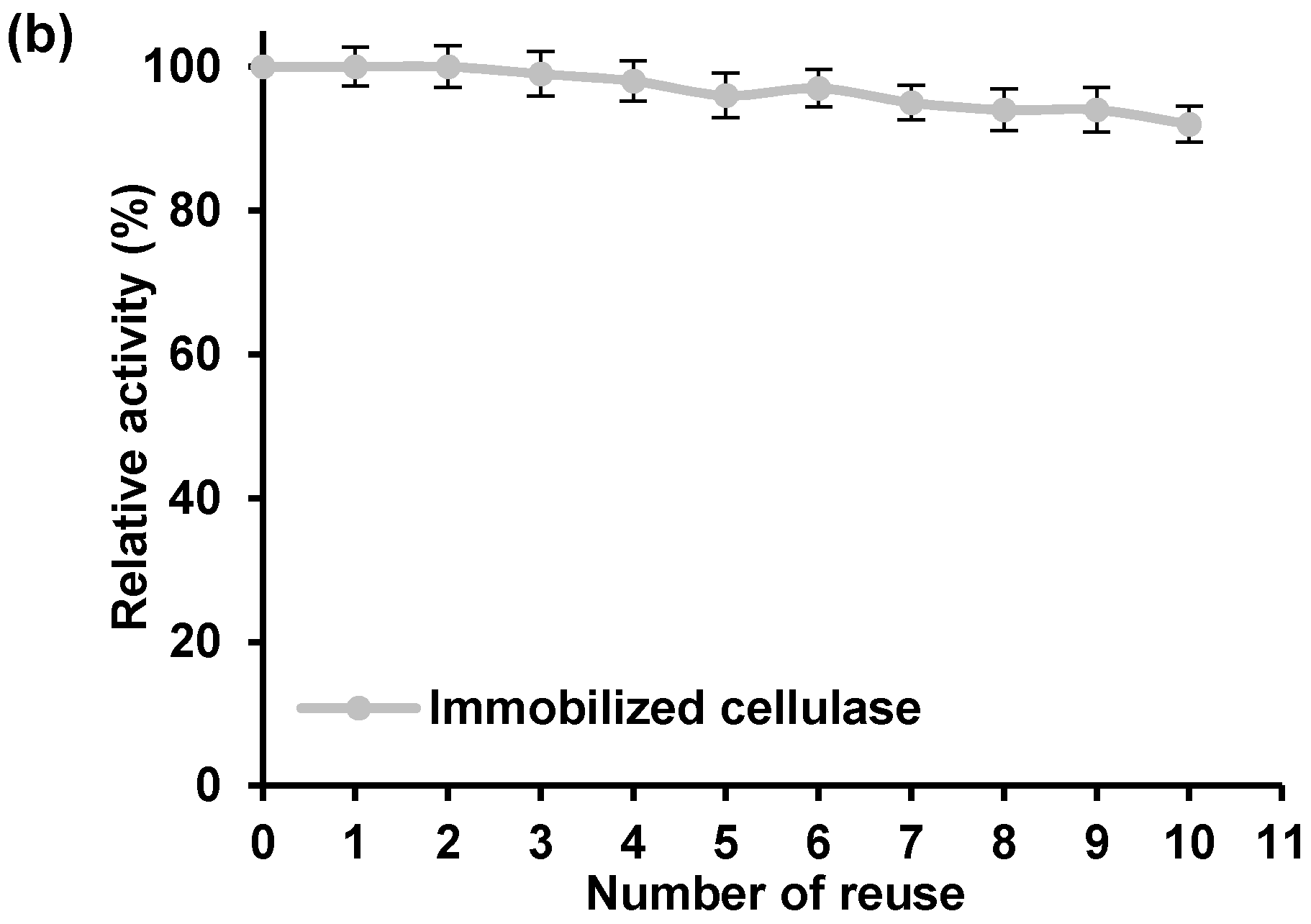
In this study, reusability of the bounded enzyme was evaluated over ten consecutive cycles of cellulose hydrolysis (
Figure 7b). As can be seen, the relative activity of the immobilized cellulase remained almost unaltered during repeated catalytic cycles, and after ten cycles it retained 93% of its initial activity.
2.4. Kinetic Parameters of Free and Immobilized Cellulase
It is clear that, with changes in the three-dimensional structure of the enzyme, and its pH and temperature profiles as a result of immobilization, its kinetic parameters might also be affected. Kinetic parameters, including
Km,
Vmax, and turnover number (
kcat), were determined for both free and immobilized cellulase by measuring the initial reaction rates during the hydrolysis of cellulose with various initial concentrations of substrate, under optimal process conditions (
Table 2).
It can be seen that for the immobilized cellulase the value of Km reached 2.63 mM, which is higher in comparison with free enzyme (2.06 mM), while the Vmax value recorded for the immobilized enzyme (159 U/mg) was lower than for free cellulase. Nevertheless, higher values of the Michaelis–Menten constant and lower values of Vmax after immobilization are typical for immobilized biocatalysts. A similar pattern was observed for turnover number (kcat), which after immobilization took a value of 114 s−1, and is lower than noticed for the free enzyme (145 s−1). In consequence, the value of catalytic efficiency (kcat/Km) recorded for the immobilized cellulase (43.6 s−1·mM−1) is also significantly lower than that of the free biocatalyst (70.5 s−1·mM−1). This can be explained by the decrease in the affinity of the immobilized biomolecules to the substrate molecules. Nevertheless, it should be emphasized that the half-life (t1/2) calculated for the immobilized cellulase (307 min) is almost five times as long as that of the free enzyme (63 min).
4. Materials and Methods
4.1. Materials
Cellulase from Aspergillus niger (EC 3.2.1.4, product number 22178), poly-l-lisine (PLL), kraft lignin, sodium (meta)periodate, cellulose, glucose, Whatman® qualitative filter paper (Grade 1 circles, diam. 15 mm), 3,5-dinitrosalicylic acid (3,5-DNS), sodium chloride, Triton X-100, phosphate buffer (PBS) at pH 7, acetate buffer at pH 4.8, Coomassie Brilliant Blue G-250 (CBB G-250), and bovine serum albumin (BSA) were delivered by Sigma-Aldrich (St. Louis, MO, USA). Commercially available titanium dioxide (product name Tytanpol® R-001) was supplied by Grupa Azoty SA (Tarnów, Poland). Sodium hydroxide, hydrochloric acid, dioxane, 96% ethyl alcohol, and 85% phosphoric acid were delivered by Chempur (Katowice, Poland).
4.2. Synthesis of TiO2–Lignin Hybrid Support Material
The synthesis of a titania–lignin hybrid support was carried out in three steps, using the method described in our previous work [
45,
46] with some modifications. In this study, the titanium dioxide was modified with poly-
l-lysine (PLL), in view of its peptide nature and to increase the amount of reactive chemical groups for the effective binding of lignin. For surface functionalization of TiO
2, it was suspended in PBS at pH 7, then a 10% (w/w) solution of PLL was added. The mixture was shaken for 12 h (KS260 Basic, IKA Werke GmbH, Staufen im Breisgau, Germany) at 4 °C and centrifuged (Eppendorf Centrifuge 5810 R, Hamburg, Germany), and washed with deionized water to remove unbound PLL and PBS. In the next step, kraft lignin was activated by sodium (meta)periodate. Finally, the activated lignin and modified titanium dioxide were linked at a mass ratio of 1:1.
4.3. Immobilization of Cellulase from Aspergillus niger
Immobilization was carried out using 0.25 g of the previously obtained TiO2–lignin hybrid support, to which 10 mL of a solution of cellulase from Aspergillus niger, at concentrations of 1.0, 3.0, and 5.0 mg/mL in acetate buffer at pH 4.8, was added. The mixture was shaken for a specified period of time (1, 2, 3, 6, 9, or 12 h) in a KS 4000i Control incubator (IKA Werke GmbH, Staufen im Breisgau, Germany) at a temperature of 4 °C. Finally, the products were centrifuged (Eppendorf Centrifuge 5810 R, Hamburg, Germany) and washed several times with the acetate buffer to remove unbound enzyme.
4.4. Characterization of the Hybrid Support and Product after Immobilization
Thermogravimetric curves for the modified TiO2, activated lignin, and synthesized TiO2–lignin hybrid material (sample weight approximately 10 mg) were obtained using a Jupiter STA449F3 apparatus (Netzsch, Selb, Germany). Measurements were made at a heating rate of 10 °C/min over the temperature range 25–1000 °C under nitrogen flow (10 mL/min).
Porous structure parameters (BET surface area, pores diameter and pores volume) were determined using an ASAP 2020 instrument (Micromeritics Instrument Co., Norcross, GA, USA). The surface area was evaluated based on the multipoint BET (Brunauer–Emmett–Teller) method using data for nitrogen adsorption under relative pressure (p/p0). The BJH (Barrett–Joyner–Halenda) algorithm was applied to examine the mean size and total volume of pores.
Zeta potential measurements were made on a Zetasizer Nano ZS instrument (Malvern Instruments Ltd., Malvern, UK) equipped with an autotitrator. The measurements were performed in a 0.001 M NaCl solution over the pH range 2.0–11.0. The zeta potential was computed using Henry’s equation.
Fourier transform infrared (FTIR) spectra were obtained using a Vertex 70 spectrophotometer (Bruker, Billerica, MA, USA), analyzing samples in the form of KBr pellets at a resolution of 0.1 cm−1 over a wavenumber range of 4000–400 cm−1. Pellets were made by mixing 200 mg of anhydrous potassium bromide and 2 mg of the sample.
The amount of cellulase immobilized on the hybrid support was evaluated based on the Bradford method [
47]. Briefly, amount of the cellulase was measured before and after immobilization at wavelength 595 nm, using a calibration curve based on BSA solutions at known concentrations to calculate the quantity of immobilized enzyme present, in mg of cellulase per gram of support.
4.5. Activity and Stability of Free and Immobilized Cellulase
The catalytic activity of free and immobilized cellulase was evaluated by measuring the quantity of reducing sugars (glucose) during hydrolysis of the cellulose substrate. The concentration of glucose was quantified using the previously described DNS method [
48]. The activity measurements were carried out as follows: 50 mg of cellulosic substrate (Whatman
® paper) was placed in acetate buffer at pH 4.8, and 10 mg of free or immobilized cellulase was added to the reactor. The mixture was incubated for 60 min at 50 °C. The reaction was terminated by the addition of 2 mL of 3,5-DNS. The resulting solution was then incubated at 100 °C for 5 min in an oil bath, and after that time was immediately cooled in ice. Samples were then diluted with distilled water and centrifuged (Eppendorf Centrifuge 5810 R, Hamburg, Germany) to remove solid particles, and were subjected to spectrophotometric measurements at 540 nm using a Jasco V-750 UV–Vis spectrophotometer (Jasco, Tokyo, Japan). The calibration curve of glucose was used to determine the relation between absorbance and the quantity of reducing sugars. All measurements were made in triplicate. One enzyme activity unit (U) of free and immobilized cellulase was defined as the amount of enzyme that produced 1 μmol of glucose per minute.
The relative activity (
AR) (Equation (1)) was defined as the percentage ratio of the activity of cellulase at a specific value (
Ai) to the enzyme’s maximum activity (
Amax).
Amax is the highest activity among all values of enzymatic activity recorded in this study.
Also based on the above-mentioned reaction, the effect of pH and temperature on the activity of immobilized cellulase, as well as its thermal and chemical stability, storage stability, and reusability, were evaluated. The effect of pH was studied by incubating the reaction mixture with free or immobilized cellulase at pH values ranging from 3 to 9 (pH was adjusted by the addition of 0.1 M HCl or NaOH). The effect of temperature was evaluated over the temperature range 30–80 °C by incubating the reaction mixture under the desired temperature conditions. pH and thermal inactivation curves for free cellulase were examined after incubation of the free enzyme at pH 5.5 and a temperature of 50 °C for 3 h. Thermal inactivation curves of immobilized cellulase were evaluated after 3 h of incubation at pH 6 at temperatures of 50 and 60 °C, while pH inactivation curves were examined after 3 h of incubation at 60 °C, at pH 5 and 6. The reusability of the immobilized cellulase was examined by measuring enzymatic activity in ten consecutive reaction cycles. After each hydrolysis cycle, the immobilized cellulase was separated from the reaction mixture by centrifugation and washed with buffer solution before the next cycle. The enzymatic activity in the first cycle was defined as 100%, and relative activity was calculated for the following cycles. Storage stability was evaluated every 5 days under optimum reaction conditions for free and immobilized cellulase stored at 4 and 25 °C, in phosphate buffer at pH 7. The initial activity was defined as 100%.
The effect of 1% Triton X-100 and sodium chloride solutions at various concentrations (0.1–0.5 M) on the relative activity of immobilized cellulase from Aspergillus niger was evaluated over a time of 12 h. For this purpose, the immobilized enzyme was dispersed in sodium chloride or Triton X-100 solution. After the specified period of time, the relative activity of the immobilized enzyme was evaluated based on the hydrolysis reaction of the cellulosic substrate.
4.6. Kinetic Parameters of Free and Immobilized Enzyme
The Lineweaver–Burk plots were used to evaluate the kinetic parameters: the Michaelis–Menten constant (Km), maximum reaction rate (Vmax), specificity constant (kcat/Km), and turnover number (kcat) of free and immobilized cellulase. These parameters were evaluated based on the hydrolysis reaction of cellulose substrate at different concentrations. Initial reaction rates were evaluated under optimum reaction conditions.