Synthesis of Biolubricant Basestocks from Epoxidized Soybean Oil
Abstract
:1. Introduction
2. Results and Discussion
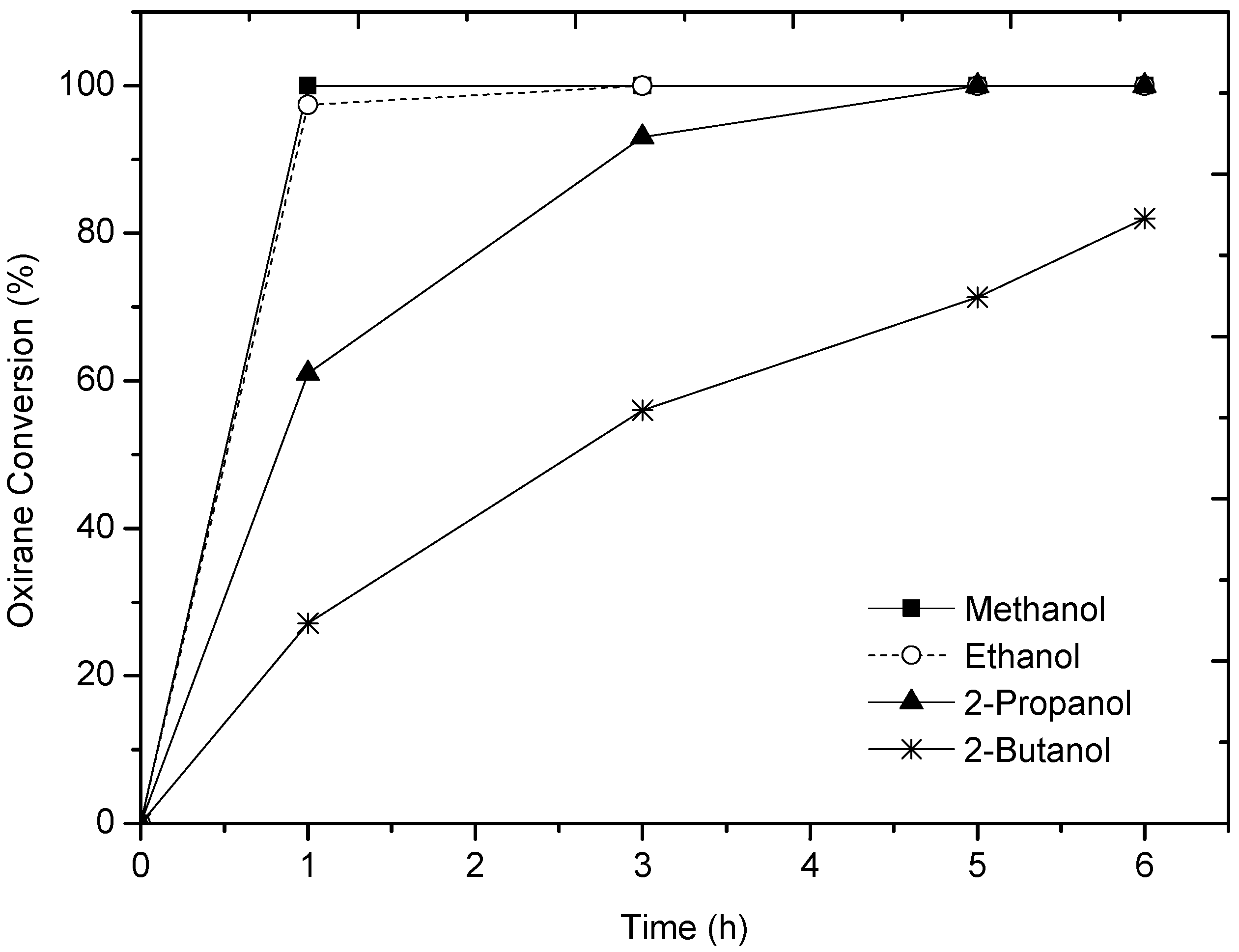
2.1. Alcohol Effect
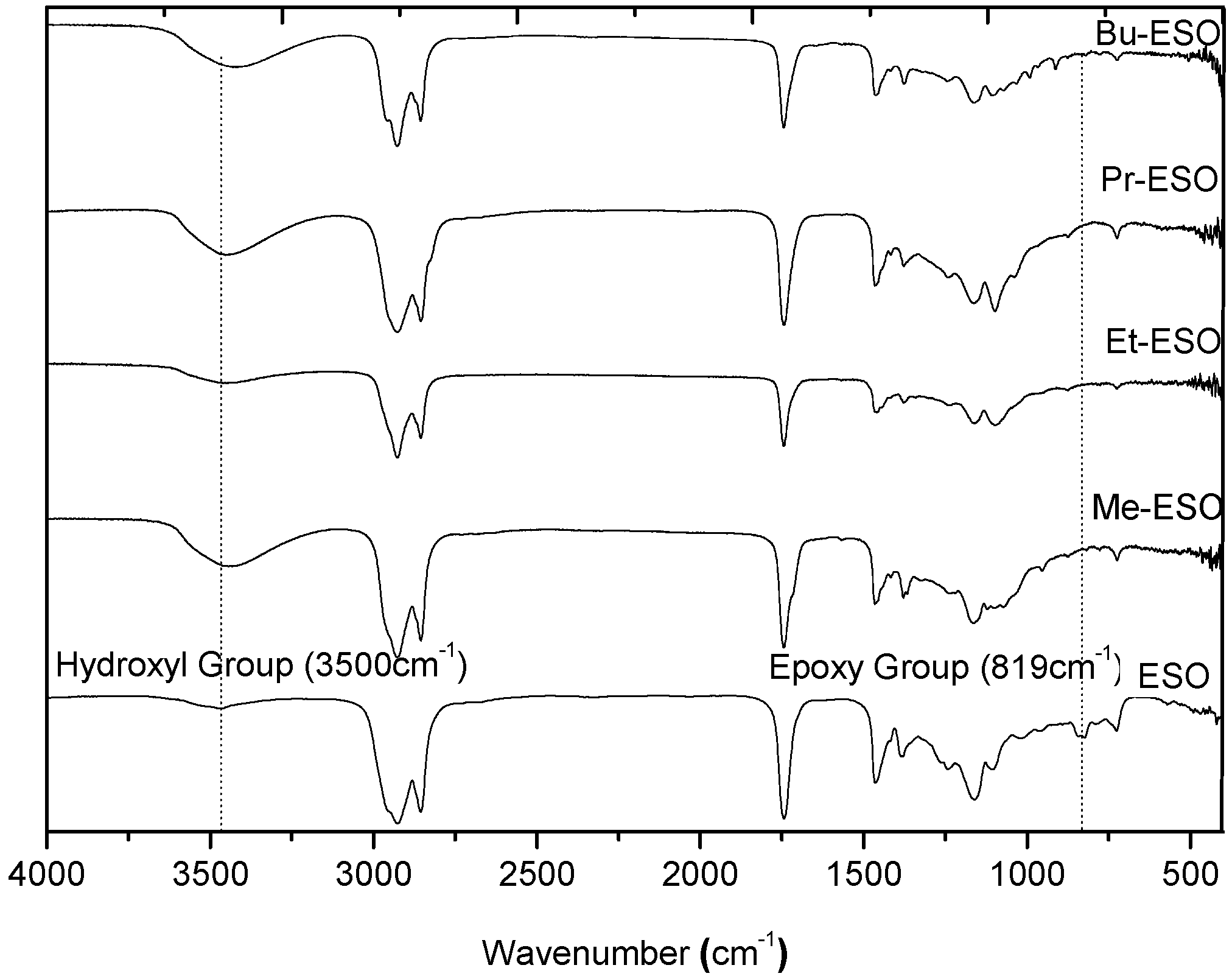
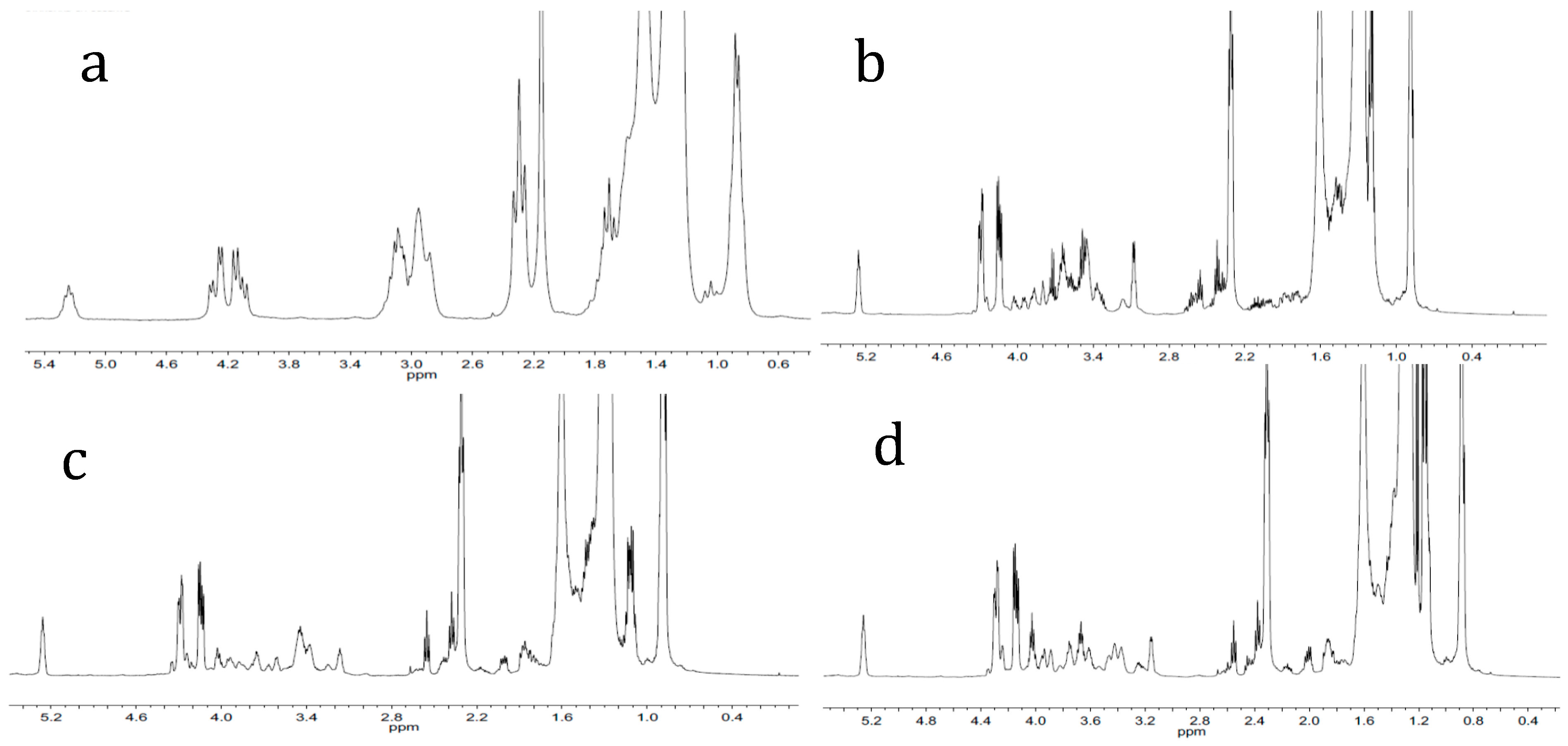
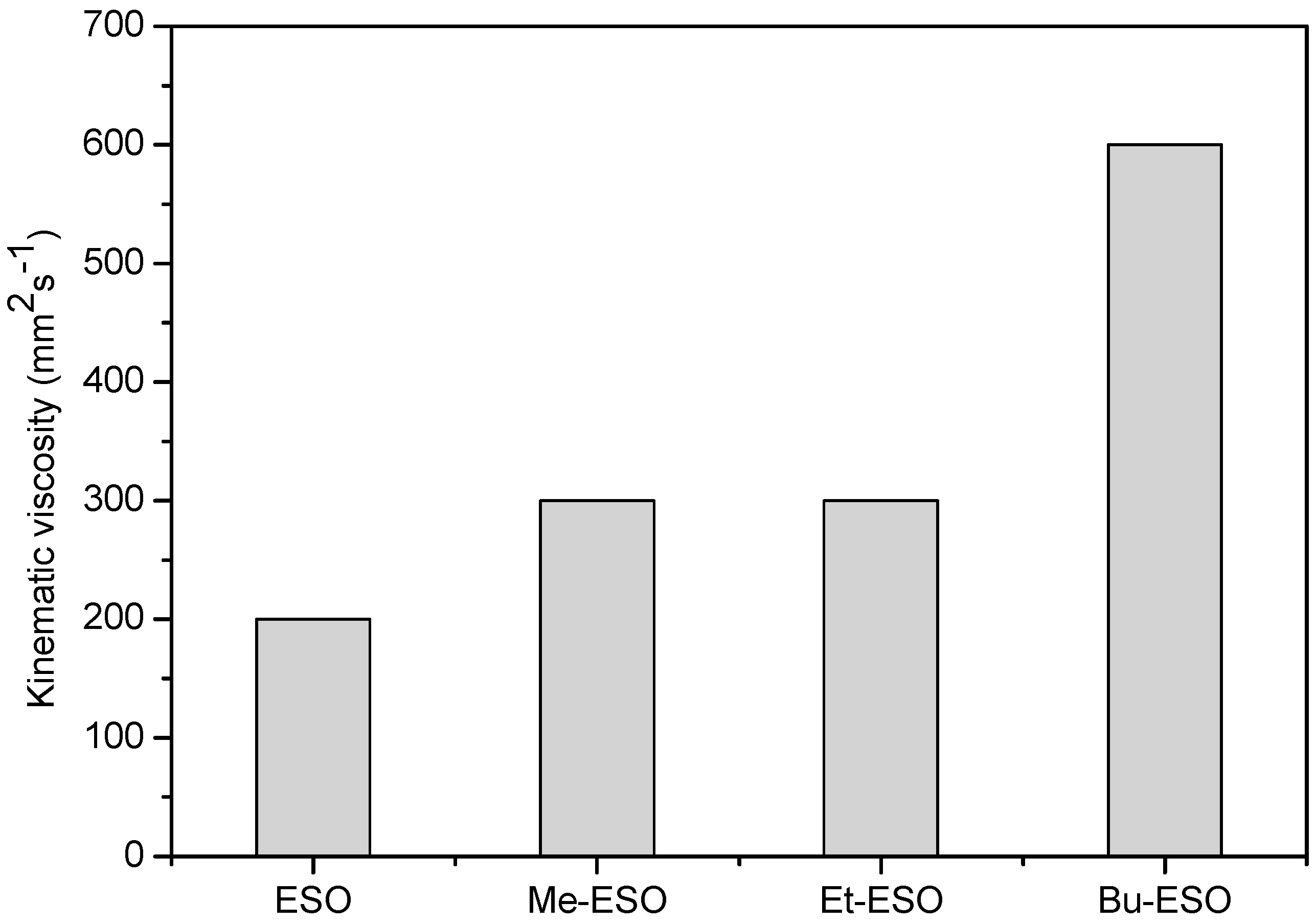
2.2. Characterization of Products
2.3. Kinetics
2.3.1. Diffusion Resistances
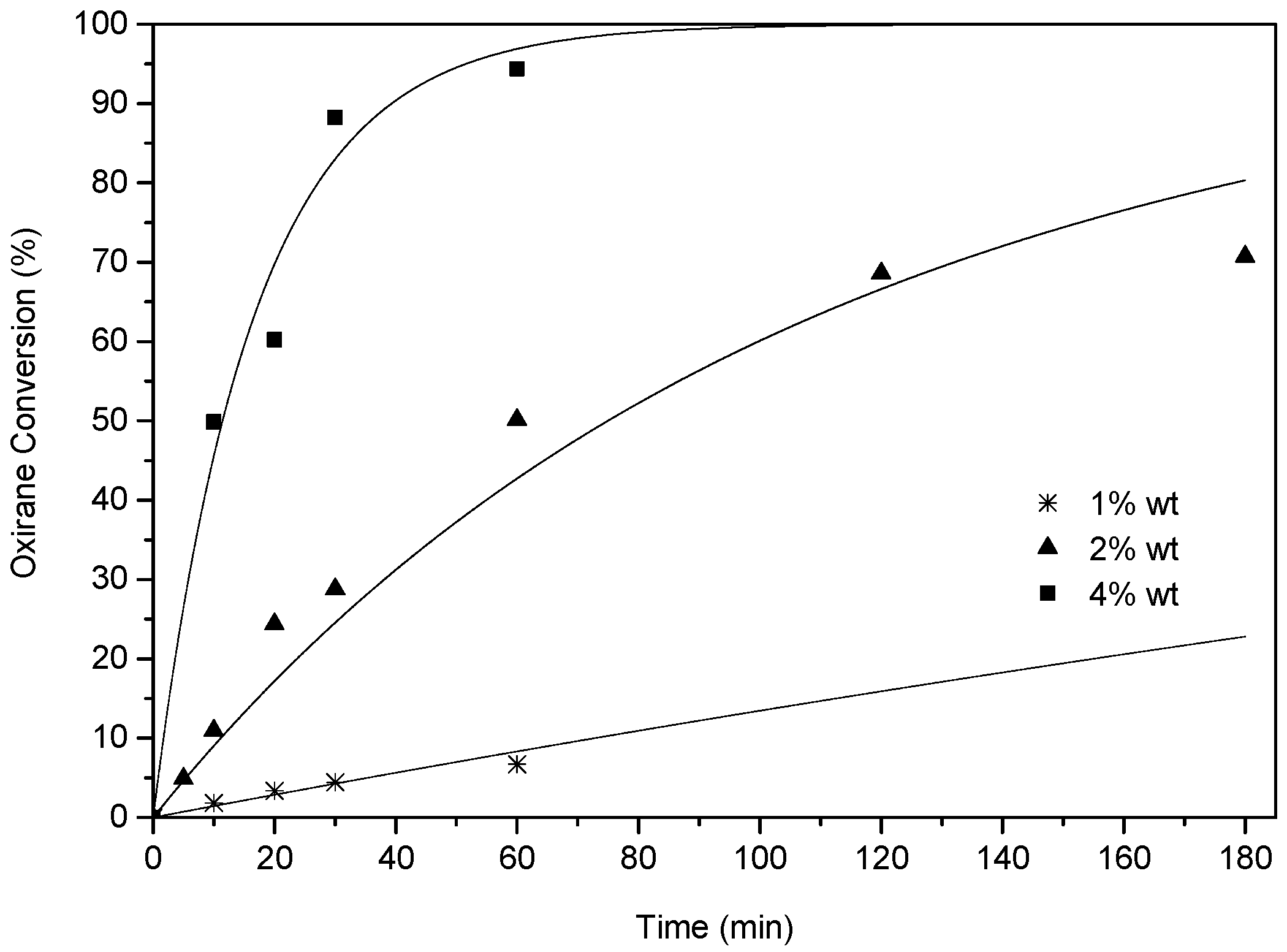
2.3.2. Effect of Catalyst Loading and Temperature
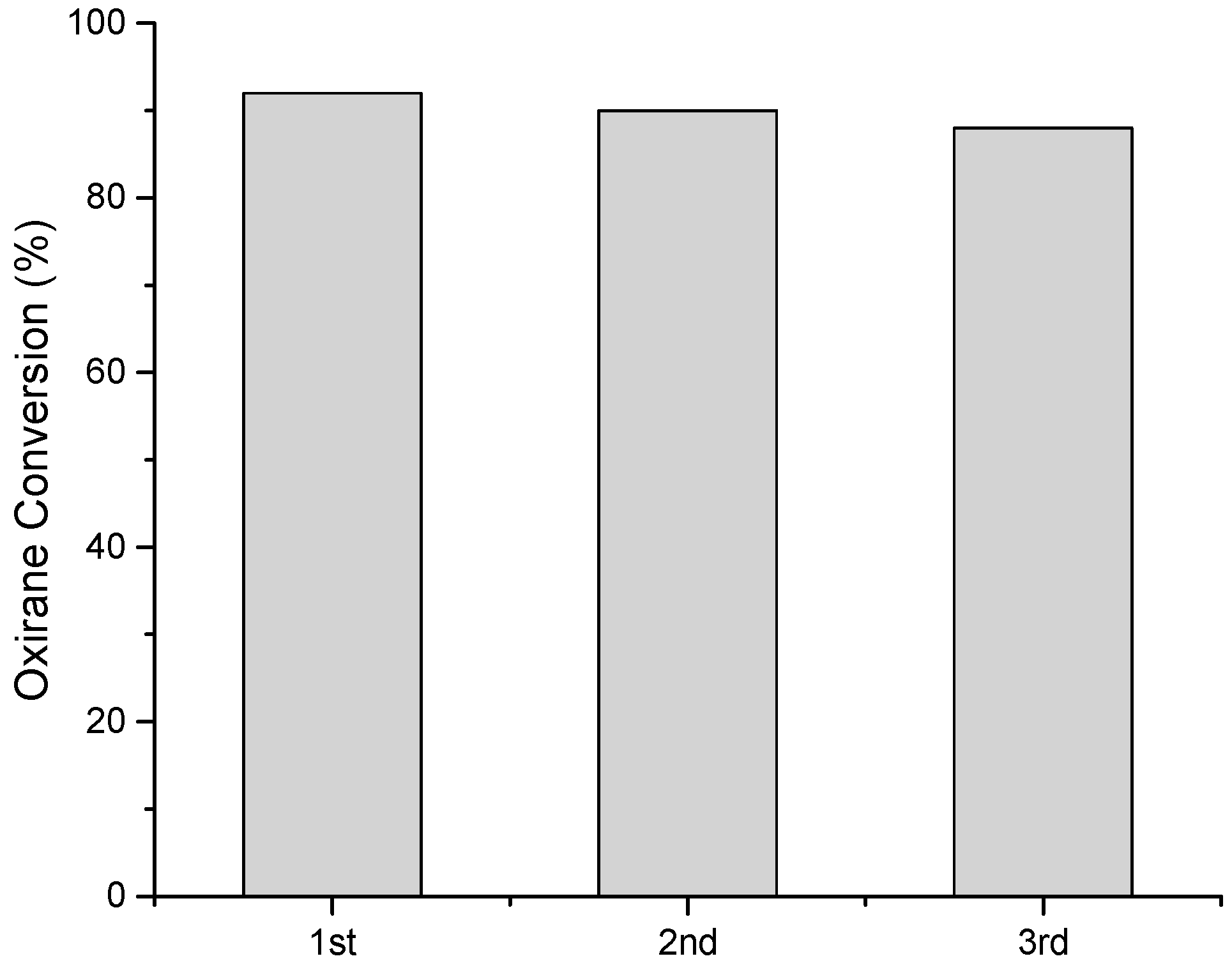
2.3.3. Reuses Tests
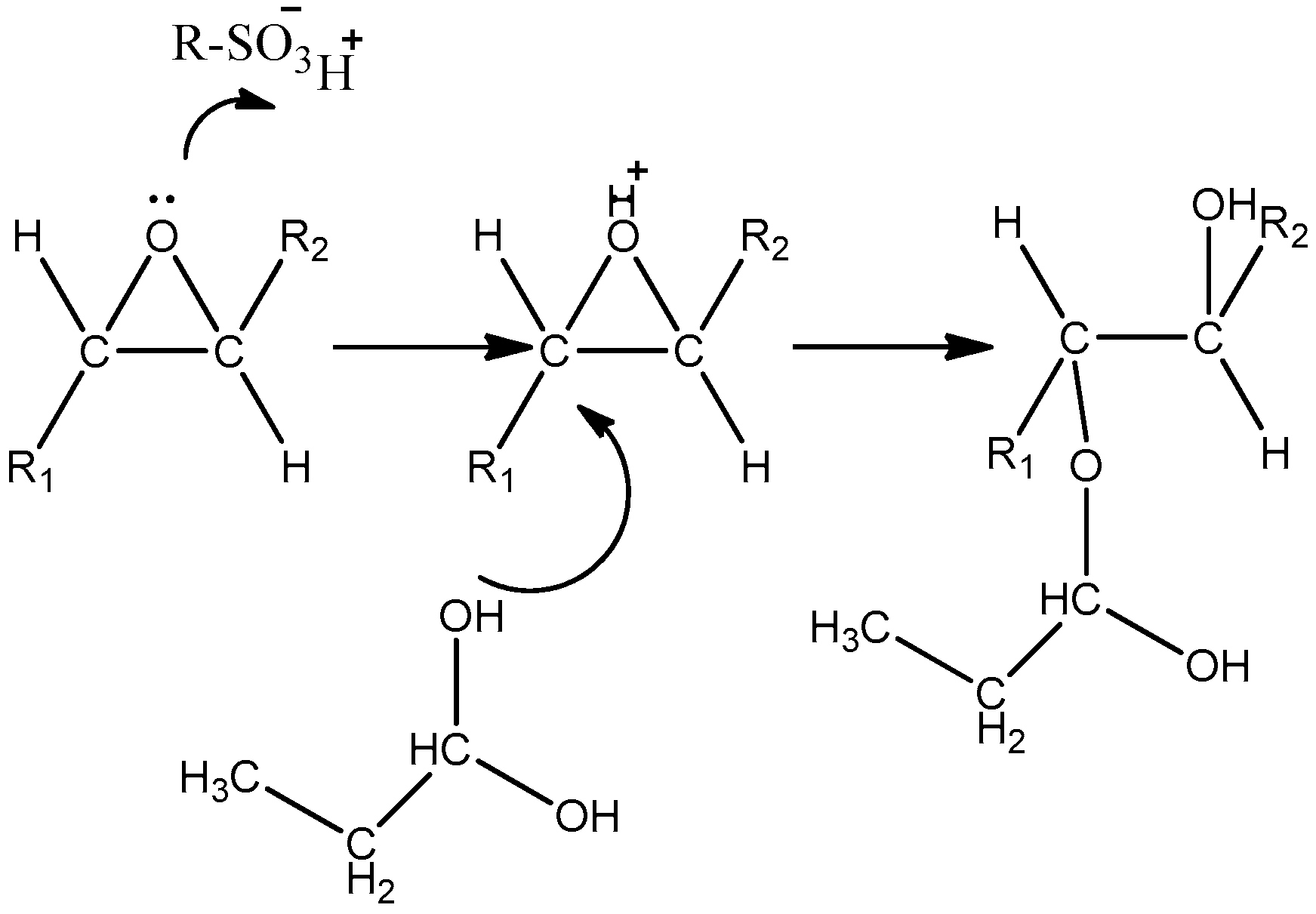
2.3.4. Development of a Kinetic Model and Reaction Mechanism for the Ring-Opening Reaction of Epoxidized Soybean Oil to Hydroxyl-Ether Products
- the evolution of the concentration of the oxirane ring and 2-butanol was followed along with time, being the time-derivative of the above concentrations, through the solution of an ordinary differential 2-equation system (ode45 MATLAB algorithm);
- , and were obtained by parameter estimation activity, implanting the lsqnonlin function present in the MATLAB library. 95% confidence intervals were calculated by implementing the nlparci MATLAB function.
3. Experimental
3.1. Materials
3.2. Analytical Methods
3.3. Ring-Opening Reaction
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
List of Symbols
| 2-butanol concentration, [mol L−1] | |
| Catalyst concentration, [g L−1] | |
| Oxirane ring concentration, [mol L−1] | |
| Activation energy, [kJ mol−1] | |
| Kinetic constant, [Ln+1g−n mol−1 min−1] | |
| Kinetic constant at 353 K, [Ln+1g−nmol−1min−1] | |
| Catalyst reaction order, [-] | |
| Oxirane number, [mol oxirane ring/100 g substrate] | |
| Reaction rate, [mol L−1 min−1] | |
| Ideal gas constant, [kcal K−1 mol−1] | |
| Temperature, [K] | |
| Reference temperature, [K] |
References
- Salimon, J.; Smith, N.; Yousilf, E. Bio-lubricants: Raw materials, chemical modifications and environmental benefits. Eur. J. Lipid Sci. Technol. 2010, 112, 519–530. [Google Scholar]
- Mobarak, H.M.; Niza Mohamad, E.; Masjuki, H.H.; Kalam, M.A.; Mahmud, A.; Habibullah, M.; Ashraful, A.M. The prospect of biolubricants as alternatives in automative applications. Renew. Sustain. Energy Rev. 2014, 33, 34–43. [Google Scholar] [CrossRef]
- MacNutt, J.; He, Q.S. Development of biolubricants from vegetable oils chemical modification. J. Ind. Eng. Chem. 2016, 36, 1–12. [Google Scholar] [CrossRef]
- Madankar, C.; Dalai, A.; Naik, S. Green synthesis of bio-lubricant basestock from canola oil. Ind. Crops Prod. 2013, 44, 139–144. [Google Scholar] [CrossRef]
- Di Serio, M.; Russo, V.; Santacesaria, E.; Tesser, R.; Turco, R.; Vitiello, R. Liquid–Liquid–Solid Model for the Epoxidation of Soybean Oil Catalyzed by Amberlyst-16. Ind. Eng. Chem. Res. 2017. [Google Scholar] [CrossRef]
- Turco, R.; Vitiello, R.; Russo, V.; Tesser, R.; Santacesaria, E.; Di Serio, M. Selective epoxidation of soybean oil with performic acid catalyzed by acidic ionic exchange resins. Green Process. Synth. 2013, 2, 427–432. [Google Scholar] [CrossRef]
- Turco, R.; Pischetola, C.; Di Serio, M.; Vitiello, R.; Tesser, R.; Santacesaria, E. Selective Epoxidation of Soybean Oil in the Presence of H–Y Zeolite. Ind. Eng. Chem. Res. 2017, 56, 7930–7936. [Google Scholar] [CrossRef]
- Adhvaryu, A.; Erhan, S.Z. Epoxidized soybean oil as a potential source of high-temperature lubricants. Ind. Crop Prod. 2002, 15, 247–254. [Google Scholar] [CrossRef]
- Sharma, B.; Advariuy, A.Z.; Erhan, S. Chemical modification of vegetable oils for lubricant applications. J. Am. Oil Chem. Soc. 2006, 8, 129–136. [Google Scholar] [CrossRef]
- Sharma, R.V.; Dalai, A. Synthesis of bio-lubricant from epoxy canola oil using sulfated Ti-SBA-15 catalyst. Appl. Catal. B Environ. 2013, 142–143, 604–614. [Google Scholar] [CrossRef]
- Adhvaryu, A.; Liu, Z.; Erhan, S.Z. Synthesis of novel alkoxylatetriacylglycerols and their lubricant base oil properties. Ind. Crops Prod. 2005, 21, 113–119. [Google Scholar] [CrossRef]
- Campanella, A.; Rustoy, E.; Baldessari, A.; Baltanas, A. Lubricants from chemically modified vegetable oils. Bioresour. Technol. 2010, 101, 245–254. [Google Scholar] [CrossRef] [PubMed]
- Aejung, K.; Seyed Mahdi, R.; Shahrouz, A.; Mohammadreza, S. Palladium Nanocatalysts Confined in Mesoporous Silica for Heterogeneous Reduction of Nitroaromatics. Energy Environ. Focus. 2015, 4, 18–23. [Google Scholar]
- Mohammadreza, S. Magnetically Separable and Sustainable Nanostructured Catalysts for Heterogeneous Reduction of Nitroaromatics. Catalysts 2015, 5, 534–560. [Google Scholar]
- Seyed Mahdi, R.; Aejung, K.; Mohammadreza, S. Gadolinium Triflate Immobilized on Magnetic Nanocomposites as Recyclable Lewis Acid Catalyst for Acetylation of Phenols. Nanosci. Nanotechnol. Lett. 2014, 6, 309–313. [Google Scholar]
- Vlcček, T.; Petrović, Z.S. Optimization of the chemoenzymatic epoxidation of soybean oil. J. Am. Oil Chem. Soc. 2006, 83, 247–252. [Google Scholar] [CrossRef]
- Rudnick, L.R. Synthetics, Mineral Oils, and Bio-Based Lubricants; Taylor & Francis: London, UK, 2006. [Google Scholar]
- Ahn, J.K.; Ihm, S.K.; Park, K.S. The effect of the local concentration and distribution of sulfonic acid groups on 1-butene isomerization catalyzed by macroporous ion-exchange resin catalysts. J. Catal. 1988, 13, 434–443. [Google Scholar] [CrossRef]
- Al-Jarallah, A.M.; Siddiqui, M.A.; Lee, A.K.K. Kinetics of methyl tertiary butyl ether synthesis catalyzed by ion exchange resin. Can. J. Chem. Eng. 1988, 66, 802–807. [Google Scholar] [CrossRef]
- Ancillotti, F.; Mauri, M.; Pescarollo, E. Ion Exchange resin catalyzed addition of alcohols to olefins. J. Catal. 1977, 46, 49–57. [Google Scholar] [CrossRef]
- Paquot, C.H. A Commission on Oils Fats and Derivatives: Standard Methods for the Analysis of Oils, Fats and Derivatives; Blackwell Scientific Publications: London, UK, 1987. [Google Scholar]

| RUN # | Alcohol | Alcohol/ESO (mol/mol) | Catalyst | Catalyst/ESO (wt %) | (°C) |
|---|---|---|---|---|---|
| 1 | methanol | 10 | SAC-13 a | 10 | 65 |
| 2 | ethanol | 10 | SAC-13 a | 10 | 78 |
| 3 | 2-propanol | 10 | SAC-13 a | 10 | 80 |
| 4 | 2-butanol | 10 | SAC-13 a | 10 | 80 |
| 5 | 2-butanol | 10 | SAC-13 | 10 | 80 |
| 6 | 2-butanol | 10 | No catalyst | - | 80 |
| 7 | 2-butanol | 10 | SAC-13 | 2 | 80 |
| 8 | 2-butanol | 10 | SAC-13 | 4 | 80 |
| 9 | 2-butanol | 10 | SAC-13 | 1 | 80 |
| 10 | 2-butanol | 10 | SAC-13 | 4 | 60 |
| 11 | 2-butanol | 10 | SAC-13 | 4 | 70 |
| 12 | 2-butanol | 10 | SAC-13 | 2 | 80 |
| 13 | 2-butanol | 10 | SAC-13 | 2 | 80 |
| Parameter | Value | Unit | |
| kref | 2.84 × 10−5 ± 1.32 × 10−6 | Ln+1 g−n mol−1 min−1 | |
| Ea | 154.47 ± 10.50 | kJ mol−1 | |
| n | 2.7 ± 0.1 | - | |
| Correlation Matrix | |||
| Ea | kref | n | |
| Ea | 1 | ||
| kref | −0.5 | 1 | |
| n | 0.6 | 0.7 | 1 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Turco, R.; Tesser, R.; Vitiello, R.; Russo, V.; Andini, S.; Serio, M.D. Synthesis of Biolubricant Basestocks from Epoxidized Soybean Oil. Catalysts 2017, 7, 309. https://doi.org/10.3390/catal7100309
Turco R, Tesser R, Vitiello R, Russo V, Andini S, Serio MD. Synthesis of Biolubricant Basestocks from Epoxidized Soybean Oil. Catalysts. 2017; 7(10):309. https://doi.org/10.3390/catal7100309
Chicago/Turabian StyleTurco, Rosa, Riccardo Tesser, Rosa Vitiello, Vincenzo Russo, Salvatore Andini, and Martino Di Serio. 2017. "Synthesis of Biolubricant Basestocks from Epoxidized Soybean Oil" Catalysts 7, no. 10: 309. https://doi.org/10.3390/catal7100309
APA StyleTurco, R., Tesser, R., Vitiello, R., Russo, V., Andini, S., & Serio, M. D. (2017). Synthesis of Biolubricant Basestocks from Epoxidized Soybean Oil. Catalysts, 7(10), 309. https://doi.org/10.3390/catal7100309










