Chemical Preparation of Supported Bimetallic Catalysts. Gold-Based Bimetallic, a Case Study
Abstract
:1. Introduction
- −
- The intrinsic properties of bimetallic particles may lead to catalytic properties different from those of the parent metals, e.g., to enhanced activity, selectivity, and/or stability;
- −
- The recent developments of characterization techniques, especially in electron microscopy allow now to characterize the bimetallic character of the particles much more accurately, e.g., to characterize the structure and composition of individual particles, and in addition, often in conditions in situ or operando.
- −
- The advances in catalyst preparation, especially the development of methods involving the use of colloids (Section 4) or of those based on surface reduction–oxidation reactions (Section 5.3).
- −
- The variety of possible metallic couples and their various extents of miscibility;
- −
- The variety of possible chemical compositions for a given bimetallic system and also the distribution in chemical compositions among the particles of a given sample;
- −
- The variety of particle structures that depend on the nature of the metals and on the synthesis methods, and their possible restructuration in reaction conditions.
2. Deposition-Reduction (Thermal Reduction)
2.1. Impregnation
2.2. Ion Adsorption
2.3. Deposition-Precipitation
2.4. Combination of Preparation Methods
3. One-Pot Deposition-Reduction (Reduction in Liquid Phase)
3.1. Chemical Reduction
3.2. Photo-Deposition
3.3. Reduction via the Support
4. Reduction-Deposition (Deposition of Preformed Metal Nanoparticles)
4.1. Colloidal Metal Particles
4.2. Metal Particles in Micelles
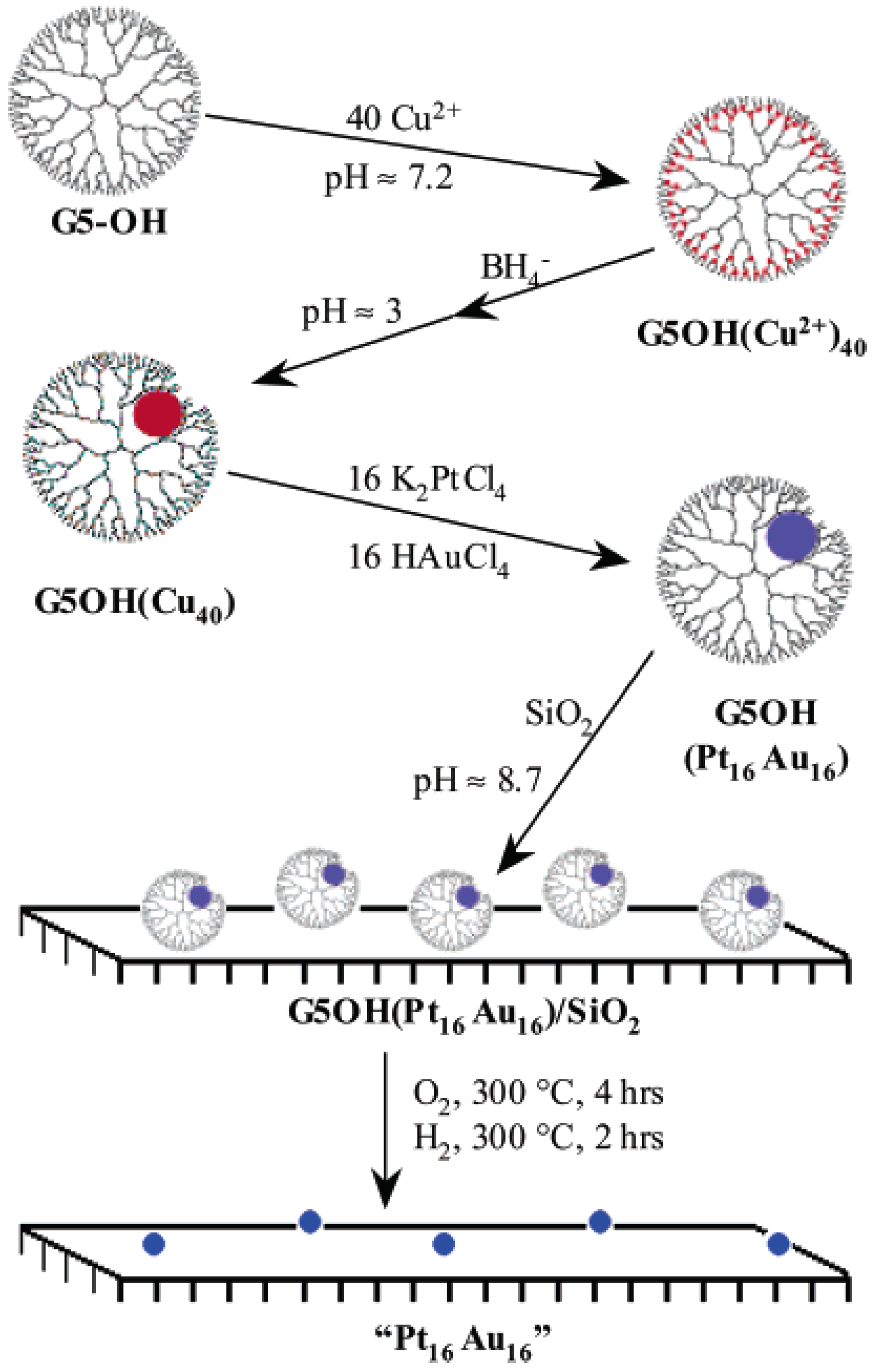
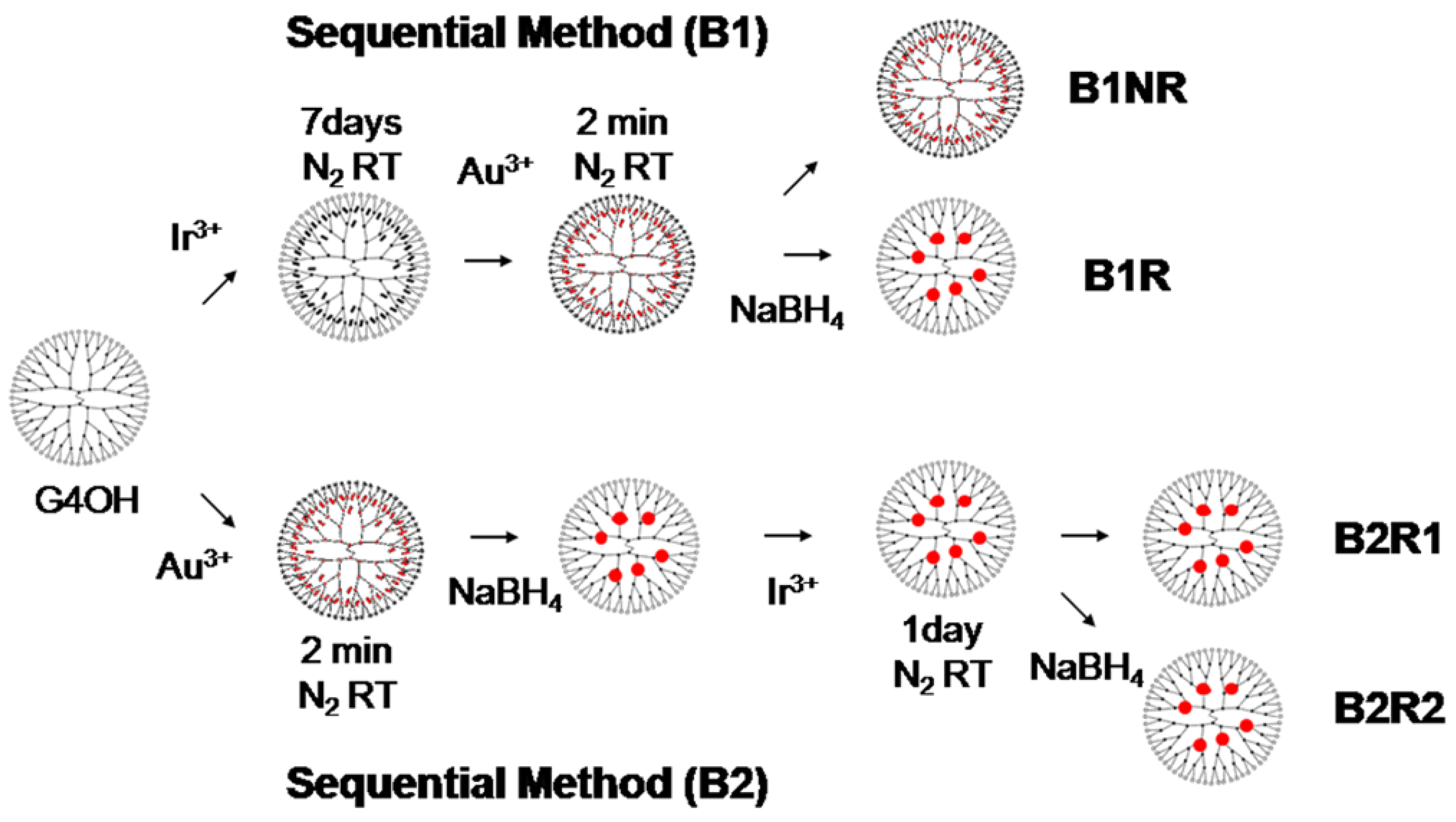
4.3. Metal Particles in Dendrimers
4.4. Microwave-Assisted Reduction
5. Specific Methods for the Preparation of Bimetallic Catalysts
5.1. Bimetallic Clusters
5.2. Radiolysis-Assisted Reduction
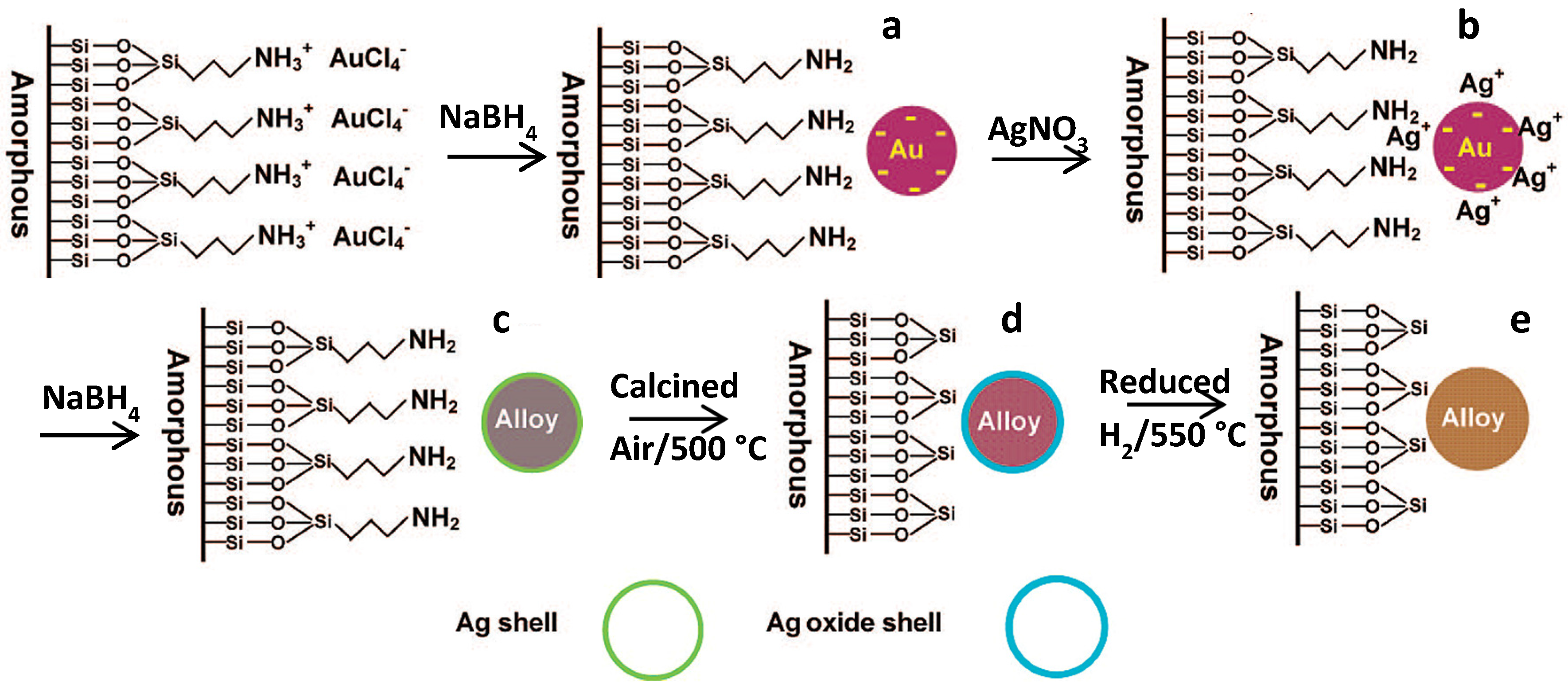
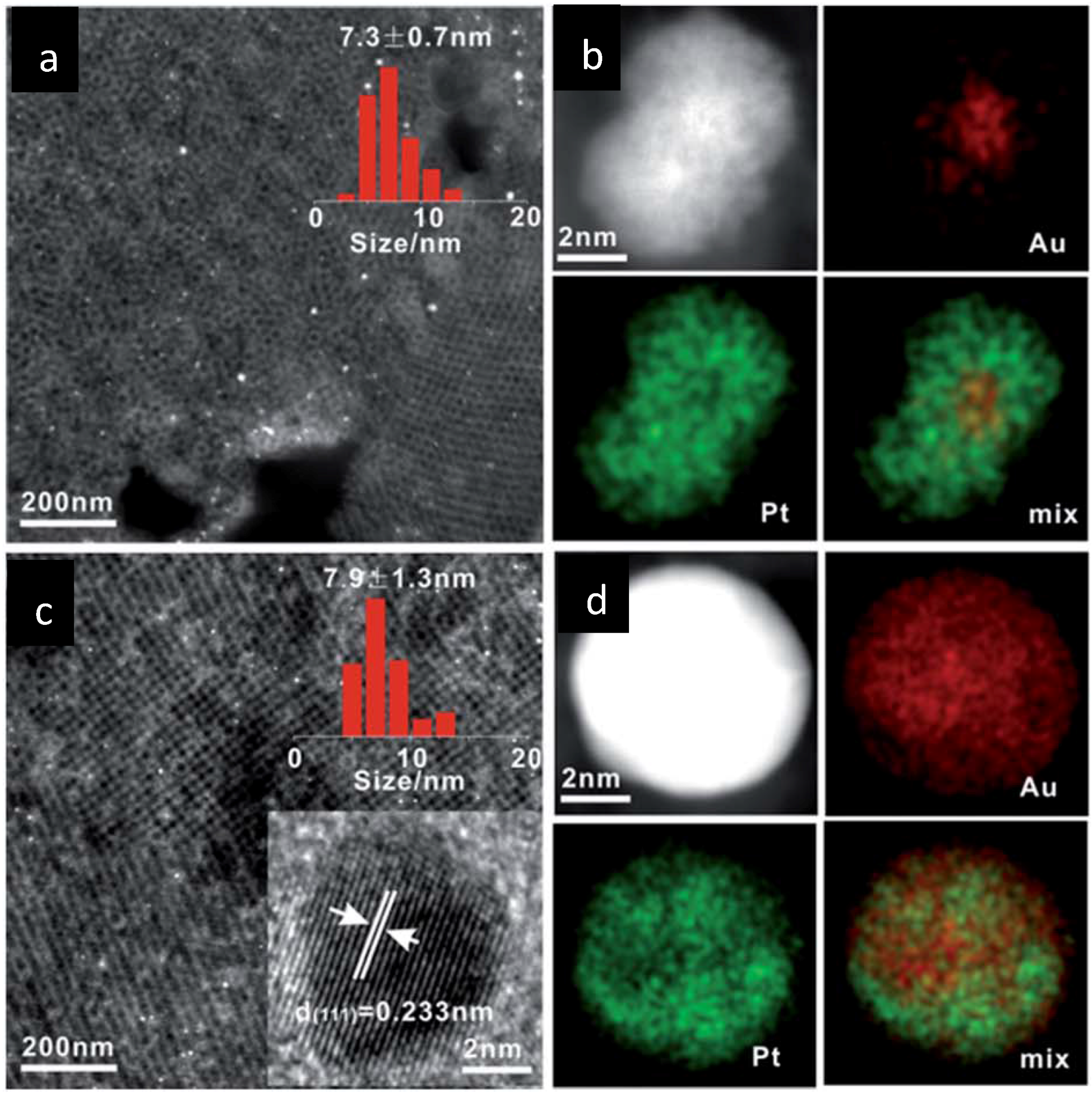
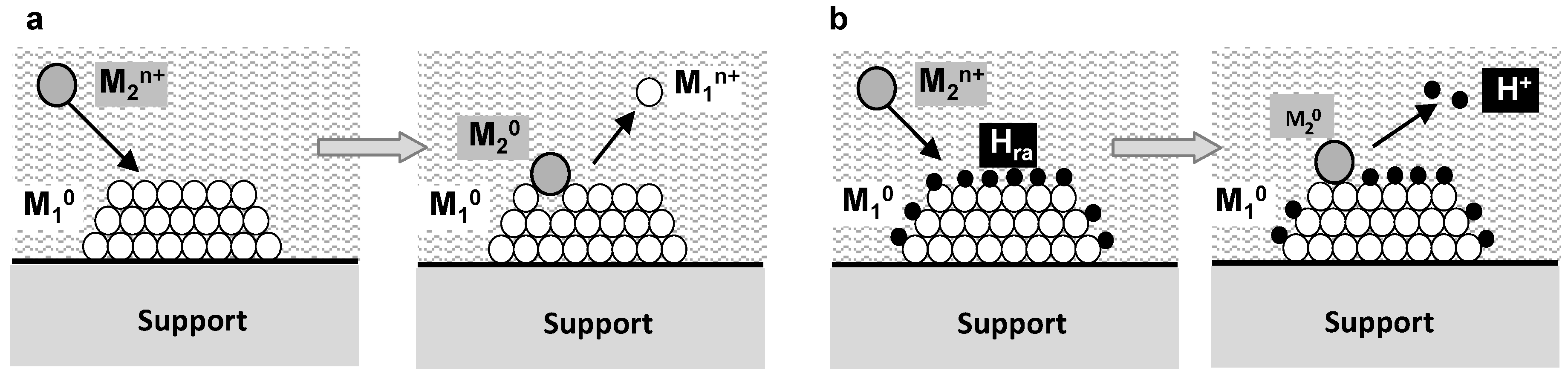
5.3. Surface Redox Methods
6. Gold-Based Bimetallic NPs Embedded in a Matrix
6.1. Embedding during Synthesis of Inorganic Matrix
6.2. Embedding after Matrix Synthesis
6.3. Embedding in Inorganic-Organic Matrix
7. Conclusions
Conflicts of Interest
References
- Haruta, M.; Kobayashi, T.; Sano, H.; Yamada, N. Novel gold catalysts for the oxidation of carbon monoxide at a temperature far below 0 °C. Chem. Lett. 1987, 2, 405–408. [Google Scholar] [CrossRef]
- Haruta, M.; Saika, K.; Kobayashi, T.; Tsubota, S.; Nakahara, Y. Preparation and catalytic properties of gold finely dispersed on beryllium oxide. Chem. Exp. 1988, 3, 159–162. [Google Scholar]
- Haruta, M. Size- and support-dependency in the catalysis of gold. Catal. Today 1997, 36, 153–166. [Google Scholar] [CrossRef]
- Wolf, A.; Schüth, F. A systematic study of the synthesis conditions for the preparation of highly active gold catalysts. Appl. Catal. A Gen. 2002, 226, 1–13. [Google Scholar] [CrossRef]
- Louis, C. Gold nanoparticles: Recent advances in CO oxidation. In Nanoparticles and Catalysis; Astruc, D., Ed.; Wiley-VCH: Weinheim, Germany, 2008. [Google Scholar]
- Widmann, D.; Behm, R.J. Activation of molecular oxygen and the nature of the active oxygen species for CO oxidation on oxide supported Au catalysts. Acc. Chem. Res. 2014, 47, 740–749. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.-H.; Wang, A.-Q.; Chi, Y.-S.; Lin, H.-P.; Mou, C.-Y. Synergistic effect in an Au-Ag alloy nanocatalyst: CO oxidation. J. Phys. Chem. B 2005, 109, 40–43. [Google Scholar] [CrossRef] [PubMed]
- Sandoval, A.; Aguilar, A.; Louis, C.; Traverse, A.; Zanella, R. Bimetallic Au-Ag/TiO2 catalyst prepared by deposition-precipitation. High activity and stability in CO oxidation. J. Catal. 2011, 281, 40–49. [Google Scholar] [CrossRef]
- Iizuka, Y.; Inoue, R.; Miura, T.; Morita, N.; Toshima, N.; Honma, T.; Oji, H. Chemical environment of Ag atoms contained in Au Ag bimetalliccatalysts and the generation of the catalytic activity for CO oxidation. Appl. Catal. A Gen. 2014, 483, 63–75. [Google Scholar] [CrossRef]
- McEwan, L.; Julius, M.; Roberts, S.; Fletcher, J.C.Q. A review of the use of gold catalysts in selective hydrogenation reactions. Gold Bull. 2010, 43, 298–306. [Google Scholar]
- Cárdenas-Lizana, F.; Keane, M.A. The development of gold catalysts for use in hydrogenation reactions. J. Mater. Sci. 2013, 48, 543–564. [Google Scholar] [CrossRef]
- Mitsudome, T.; Kaneda, K. Gold nanoparticle catalysts for selective hydrogenations. Green Chem. 2013, 15, 2636–2654. [Google Scholar] [CrossRef]
- Edwards, J.K.; Solsona, B.E.; Landon, P.; Carley, A.F.; Herzing, A.; Kiely, C.J.; Hutchings, G.J. Direct synthesis of hydrogen peroxide from H2 and O2 using TiO2-supported Au-Pd catalysts. J. Catal. 2005, 236, 69–79. [Google Scholar] [CrossRef]
- Sankar, M.; He, Q.; Morad, M.; Pritchard, J.; Freakley, S.J.; Edwards, J.K.; Taylor, S.H.; Morgan, D.J.; Carley, A.F.; Knight, D.W.; et al. Synthesis of stable ligand-free gold palladium nanoparticles using a simple excess anion method. ACS Nano 2012, 6, 6600–6613. [Google Scholar] [CrossRef] [PubMed]
- Hugon, A.; Delannoy, L.; Krafft, J.-M.; Louis, C. Supported gold-palladium catalysts for selective hydrogenation of 1,3 butadiene in an excess of propene. J. Phys. Chem. C 2010, 114, 10823–10835. [Google Scholar] [CrossRef]
- Yang, X.; Huang, C.; Fu, Z.; Song, H.; Liao, S.; Sud, Y.; Du, L.; Li, X. An effective Pd-promoted gold catalyst supported on mesoporous silica particles for the oxidation of benzyl alcohol. Appl. Catal. B Environ. 2013, 140–141, 419–425. [Google Scholar] [CrossRef]
- Edwards, J.K.; Pritchard, J.; Lu, L.; Piccinini, M.; Shaw, G.; Carley, A.F.; Morgan, D.J.; Kiely, C.J.; Hutchings, G.J. The direct synthesis of hydrogen peroxide using platinum-promoted gold-palladium catalysts. Angew. Chem. Int. Ed. 2014, 53, 2381–2384. [Google Scholar] [CrossRef] [PubMed]
- Gu, X.; Lu, Z.-H.; Jiang, H.-L.; Akita, T.; Xu, Q. Synergistic catalysis of metal organic framework-immobilized AuPd nanoparticles in dehydrogenation of formic acid for chemical hydrogen storage. J. Am. Chem. Soc. 2011, 133, 11822–11825. [Google Scholar] [CrossRef] [PubMed]
- Gudarzi, D.; Ratchananusorn, W.; Turunen, I.; Heinonen, M.; Salmi, T. Promotional effects of Au in Pd-Au bimetallic catalysts supported onactivated carbon cloth (ACC) for direct synthesis of H2O2 from H2 and O2. Catal. Today 2015, 248, 58–68. [Google Scholar] [CrossRef]
- Hao, Y.; Hao, G.-P.; Guo, D.-C.; Guo, C.-Z.; Li, W.-C.; Li, M.-R.; Lu, A.-H. Bimetallic Au-Pd nanoparticles confined in tubular mesoporous carbon as highly selective and reusable benzyl alcohol oxidation catalysts. ChemCatChem 2012, 4, 1595–1602. [Google Scholar] [CrossRef]
- Menegazzo, F.; Manzoli, M.; Signoretto, M.; Pinna, F.; Strukul, G. H2O2 direct synthesis under mild conditions on Pd-Au samples: Effectof the morphology and of the composition of the metallic phase. Catal. Today 2015, 248, 18–27. [Google Scholar] [CrossRef]
- Belin, S.; Bracey, C.L.; Briois, V.; Ellis, P.R.; Hutchings, G.J.; Hyde, T.I.; Sankar, G. CuAu/SiO2 catalysts for the selective oxidation of propene to acrolein: The impact of catalyst preparation variables on material structure and catalytic performance. Catal. Sci. Technol. 2013, 3, 2944–2957. [Google Scholar] [CrossRef]
- Chimentao, R.J.; Medina, F.; Fierro, J.L.G.; Llorca, J.; Sueiras, J.E.; Cesteros, Y.; Salagre, P. Propene epoxidation by nitrous oxide over Au-Cu/TiO2 alloy catalysts. J. Mol. Catal. A 2007, 274, 159–168. [Google Scholar] [CrossRef]
- Guan, Y.; Hensen, E.J.M. Selective oxidation of ethanol to acetaldehyde by AuIr catalysts. J. Catal. 2013, 305, 135–145. [Google Scholar] [CrossRef]
- Gallo, A.; Montini, T.; Marelli, M.; Minguzzi, A.; Gombac, V.; Psaro, R.; Fornasiero, P.; Santo, V.D. H2 production by renewables photoreforming on Pt-Au/TiO2 catalysts activated by reduction. ChemSusChem 2012, 5, 1800–1811. [Google Scholar] [CrossRef] [PubMed]
- Na, H.; Zhu, T.; Liu, Z. Effect of preparation method on the performance of Pt-Au/TiO2 catalysts for the catalytic co-oxidation of HCHO and CO. Catal. Sci. Technol. 2014, 4, 2051–2057. [Google Scholar] [CrossRef]
- Chen, C.; Yang, H.; Chen, J.; Zhang, R.; Guo, L.; Gan, H.; Song, B.; Zhu, W.; Hua, L.; Hou, Z. One-pot tandem catalytic synthesis of α,β-unsaturated nitriles from alcohol with nitriles in aqueous phase. Catal. Comm. 2014, 47, 49–53. [Google Scholar] [CrossRef]
- Haruta, M. Catalysis of gold nanoparticles deposited on metal oxides. Cattech 2002, 6, 102–115. [Google Scholar] [CrossRef]
- Prati, L.; Martra, G. New gold catalysts for liquid phase oxidation. Gold Bull. 1999, 32. [Google Scholar] [CrossRef]
- Mimura, N.; Hiyoshi, N.; Date, M.; Fujitani, T.; Dumeignil, F. Microscope analysis of Au-Pd/TiO2 glycerol oxidation catalysts prepared by deposition—Precipitation method. Catal. Lett. 2014, 144, 2167–2175. [Google Scholar] [CrossRef]
- Sasirekha, N.; Sangeetha, P.; Chen, Y.-W. Bimetallic Au-Ag/CeO2 catalysts for preferential oxidation of CO in hydrogen-rich stream: Effect of calcination temperature. J. Phys. Chem. C 2014, 118, 15226–15233. [Google Scholar] [CrossRef]
- La Parola, V.; Testa, M.L.; Venezia, A.M. Pd and PdAu catalysts supported over 3-MPTES grafted HMS used in the HDS of thiophene. Appl. Catal. Environ. 2012, 119–120, 248–255. [Google Scholar] [CrossRef]
- Li, L.; Wang, C.; Ma, X.; Yang, Z.; Lu, X. An Au-Cu bimetal catalyst supported on mesoporous TiO2 with stable catalytic performance in CO oxidation. Chin. J. Catal. 2012, 33, 1778–1782. [Google Scholar] [CrossRef]
- Delannoy, L.; Thrimurthulu, G.; Reddy, P.S.; Méthivier, C.; Nelayah, J.; Reddy, B.M.; Ricolleau, C.; Louis, C. Selective hydrogenation of butadiene over TiO2 supported copper, gold and gold-copper catalysts prepared by deposition precipitation. Phys. Chem. Chem. Phys. 2014, 16, 26514–26527. [Google Scholar] [CrossRef] [PubMed]
- Aguilar-Tapia, A.; Zanella, R.; Calers, C.; Louis, C.; Delannoy, L. Synergistic effects of Ir-Au/TiO2 catalysts in the total oxidation of propene: Influence of the activation conditions. Phys. Chem. Chem. Phys. 2015, 17, 28022–28032. [Google Scholar] [CrossRef] [PubMed]
- Sandoval, A.; Delannoy, L.; Méthivier, C.; Louis, C.; Zanella, R. Synergetic effect in bimetallic Au-Ag/TiO2 catalysts for CO oxidation: Insights from in situ characterization. Phys. Chem. Chem. Phys. 2015, 17, 28022–28032. [Google Scholar] [CrossRef]
- Kittisakmontree, P.; Pongthawornsakun, B.; Yoshida, H.; Fujita, S.-I.; Arai, M.; Panpranot, J. The liquid-phase hydrogenation of 1-heptyne over Pd-Au/TiO2 catalysts prepared by the combination of incipient wetness impregnation and deposition—Precipitation. J. Catal. 2013, 297, 155–164. [Google Scholar] [CrossRef]
- Olmos, C.M.; Chinchilla, L.E.; Delgado, J.J.; Hungrıa, A.B.; Blanco, G.; Calvino, J.J.; Chen, X. CO oxidation over bimetallic Au-Pd supported on ceria-zirconia catalysts: Effects of oxidation temperature and Au:Pd molar ratio. Catal. Lett. 2016, 146, 144–156. [Google Scholar] [CrossRef]
- Chinchilla, L.E.; Olmos, C.M.; Villa, L.; Carlsson, A.; Prati, L.; Chen, X.; Blanco, G.; Calvino, J.J.; Hungría, A.B. Ru-modified Au catalysts supported on ceria-zirconia for the selectiveoxidation of glycerol. Catal. Today 2015, 253, 178–189. [Google Scholar] [CrossRef]
- Liao, X.; Chu, W.; Dai, X.; Pitchon, V. Bimetallic Au-Cu supported on ceria for PROX reaction: Effects of Cu/Au atomic ratios and thermal pretreatments. Appl. Catal. Environ. 2013, 142–143, 25–37. [Google Scholar] [CrossRef]
- Liu, X.; Wang, A.; Wang, X.; Mou, C.-Y.; Zhang, T. Au-Cu alloy nanoparticles confined in SBA-15 as a highly efficient catalyst for CO oxidation. Chem. Commun. 2008. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Wang, A.; Yang, X.; Zhang, T.; Mou, C.-Y.; Su, D.-S.; Li, J. Synthesis of thermally stable and highly active bimetallic Au-Ag nanoparticles on inert supports. Chem. Mater. 2009, 21, 410–418. [Google Scholar] [CrossRef]
- Liu, X.; Wang, A.; Zhang, T.; Su, D.-S.; Mou, C.-Y. Au-Cu alloy nanoparticles supported on silica gel as catalyst for CO oxidation: Effects of Au/Cu ratios. Catal. Today 2011, 160, 103–108. [Google Scholar] [CrossRef]
- Regan, M.R.; Banerjee, I.A. Preparation of Au-Pd bimetallic nanoparticles in porous germania nanospheres: A study of their morphology and catalytic activity. Scr. Mater. 2006, 54, 909–914. [Google Scholar] [CrossRef]
- Bauer, J.C.; Mullins, D.; Li, M.; Wu, Z.; Payzant, E.A.; Overbury, S.H.; Dai, S. Synthesis of silica supported AuCu nanoparticle catalysts and the effects of pretreatment conditions for the CO oxidation reaction. Phys. Chem. Chem. Phys. 2011, 13, 2571–2581. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Q.; Liu, Z.; Bo, A.; Zavahir, S.; Sarina, S.; Bottle, S.; Riches, J.D.; Zhu, H. Catalytic transformation of aliphatic alcohols to corresponding esters in O2 under neutral conditions using visible-light irradiation. J. Am. Chem. Soc. 2015, 137, 1956–1966. [Google Scholar] [CrossRef] [PubMed]
- Yan, S.; Gao, L.; Zhang, S.; Gao, L.; Zhang, W.; Li, Y. Investigation of AuNi/C anode catalyst for direct methanol fuel cells. Int. J. Hydr. Ener. 2013, 38, 12838–12846. [Google Scholar] [CrossRef]
- Qiao, P.; Zou, S.; Xu, S.; Liu, J.; Li, Y.; M, G.; Xiao, L.; Loua, H.; Fan, J. A general synthesis strategy of multi-metallic nanoparticles within mesoporous titania via in situ photo-deposition. J. Mater. Chem. A 2014, 2, 17321–17328. [Google Scholar] [CrossRef]
- Tanaka, A.; Hashimoto, K.; Kominami, H. Gold and copper nanoparticles supported on cerium(IV) oxide—A photocatalyst mineralizing organic acids under red light irradiation. ChemCatChem 2011, 3, 1619–1623. [Google Scholar] [CrossRef]
- Wu, T.; Ma, J.; Wang, X.; Liu, Y.; Xu, H.; Gao, J.; Wang, W.; Liu, Y.; Yan, J. Graphene oxide supported Au-Ag alloy nanoparticles with different shapes and their high catalytic activities. Nanotechnology 2013, 24, 125301. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Cai, Z.; Chen, X.; Oyama, M. AuPd bimetallic nanoparticles decorated on graphene nanosheets: Their green synthesis, growth mechanism and high catalytic ability in 4-nitrophenol reduction. J. Mater. Chem. A 2014, 2, 5668–5674. [Google Scholar] [CrossRef]
- Villa, A.; Wang, D.; Veith, G.M.; Vindigni, F.; Prati, L. Sol immobilization technique: A delicate balance between activity, selectivity and stability for gold catalyst. Catal. Sci. Technol. 2013, 3, 3036–3041. [Google Scholar] [CrossRef]
- Quintanilla, A.; Butselaar-Orthlieb, V.C.L.; Kwakernaak, C.; Sloof, W.G.; Kreutzer, M.T.; Kapteijn, F. Weakly bound capping agents on gold nanoparticles in catalysis: Surface poison? J. Catal. 2010, 271, 104–114. [Google Scholar] [CrossRef]
- Zhong, Z.; Lin, J.; Teh, S.-P.; Teo, J.; Dautzenberg, F.M. A rapid and efficient method to deposit gold particles on catalyst supports and its application for CO oxidation at low temperatures. Adv. Funct. Mater. 2007, 17, 1402–1408. [Google Scholar] [CrossRef]
- Menard, L.D.; Xu, F.; Nuzzo, R.G.; Yang, J.C. Preparation of TiO2-supported Au nanoparticle catalysts from a Au13 cluster precursor: Ligand removal using ozone exposure versus a rapid thermal treatment. J. Catal. 2006, 243, 64–73. [Google Scholar] [CrossRef]
- Llorca, J.; Casanovas, A.; Domınguez, M.; Casanova, I.; Angurell, I.; Seco, M.; Rossell, O. Plasma-activated core-shell gold nanoparticle films with enhanced catalytic properties. J. Nano. Res. 2008, 10, 537–542. [Google Scholar] [CrossRef]
- Zhong, R.-Y.; Yan, X.-H.; Gao, Z.-K.; Zhang, R.-J.; Xu, B.-Q. Stabilizer substitution and its effect on the hydrogenation catalysis by Au nanoparticles from colloidal synthesis. Catal. Sci. Technol. 2013, 3, 3013–3019. [Google Scholar] [CrossRef]
- Lopez-Sanchez, J.A.; Dimitratos, N.; Hammond, C.; Brett, G.L.; Kesavan, L.; White, S.; Miedziak, P.; Tiruvalam, R.; Jenkins, R.L.; Carley, A.F.; et al. Facile removal of stabilizer-ligands from supported gold nanoparticles. Nat. Chem. 2011, 3, 551–556. [Google Scholar] [CrossRef] [PubMed]
- Gandarias, I.; Miedziak, P.J.; Nowicka, E.; Douthwaite, M.; Morgan, D.J.; Hutchings, G.J.; Taylor, S.H. Selective oxidation of n-butanol using gold-palladium supported nanoparticles under base-free conditions. ChemSusChem 2015, 8, 473–480. [Google Scholar] [CrossRef] [PubMed]
- Luo, M.; Hong, Y.; Yao, W.; Huang, C.; Xu, Q.; Wu, Q. Facile removal of polyvinylpyrrolidone (PVP) adsorbates from Pt alloy nanoparticles. J. Mater. Chem. A 2015, 3, 2770–2775. [Google Scholar] [CrossRef]
- Naresh, N.; Wasim, F.G.S.; Ladewig, B.P.; Neergat, M. Removal of surfactant and capping agent from Pd nanocubes (Pd-NCs) using tert-butylamine: Its effect on electrochemical characteristics. J. Mater. Chem. A 2013, 1, 8553–8559. [Google Scholar] [CrossRef]
- Ansar, S.M.; Ameer, F.S.; Hu, W.; Zou, S.; Charles, U.; Pittman, J.; Zhang, D. Removal of molecular adsorbates on gold nanoparticles using sodium borohydride in water. Nano Lett. 2013, 13, 1226–1229. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Jia, L.; Medrano, J.A.; Ross, J.R.H.; Lefferts, L. Supported Pd catalysts prepared via colloidal method: The effect of acids. ACS Catal. 2013, 3, 2341–2352. [Google Scholar] [CrossRef]
- Monzo, J.; Koper, M.T.M.; Rodriguez, P. Removing polyvinylpyrrolidone from catalytic Pt nanoparticles without modification of superficial order. ChemPhysChem 2012, 13, 709–715. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, C.L.; Canton, P.; Dimitratos, N.; Porta, F.; Prati, L. Selective oxidation of glycerol with oxygen using mono and bimetallic catalysts based on Au, Pd and Pt metals. Catal. Today 2005, 102–103, 203–212. [Google Scholar] [CrossRef]
- Wang, D.; Villa, A.; Porta, F.; Su, D.; Prati, L. Single-phase bimetallic system for the selective oxidation of glycerol to glycerate. Chem. Comm. 2006, 1956–1958. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Villa, A.; Prati, L.; Porta, F.; Su, D. Bimetallic gold/palladium catalysts: Correlation between nanostructure and synergistic effects. J. Phys. Chem. C 2008, 112, 8617–8622. [Google Scholar] [CrossRef]
- Villa, A.; Wang, D.; Su, D.; Veith, G.M.; Prati, L. Using supported Au nanoparticles as starting material for preparing uniform Au/Pd bimetallic catalysts. Phys. Chem. Chem. Phys. 2010, 12, 2183–2189. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Sanchez, J.A.; Dimitratos, N.; Miedziak, P.; Ntainjua, E.; Edwards, J.K.; Morgan, D.; Carley, A.F.; Tiruvalam, R.; Kiely, C.J.; Hutchings, G.J. Au-Pd supported nanocrystals prepared by a sol immobilisation technique as catalysts for selective chemical synthesis. Physi. Chem. Chem. Phys. 2008, 10, 1921–1930. [Google Scholar] [CrossRef] [PubMed]
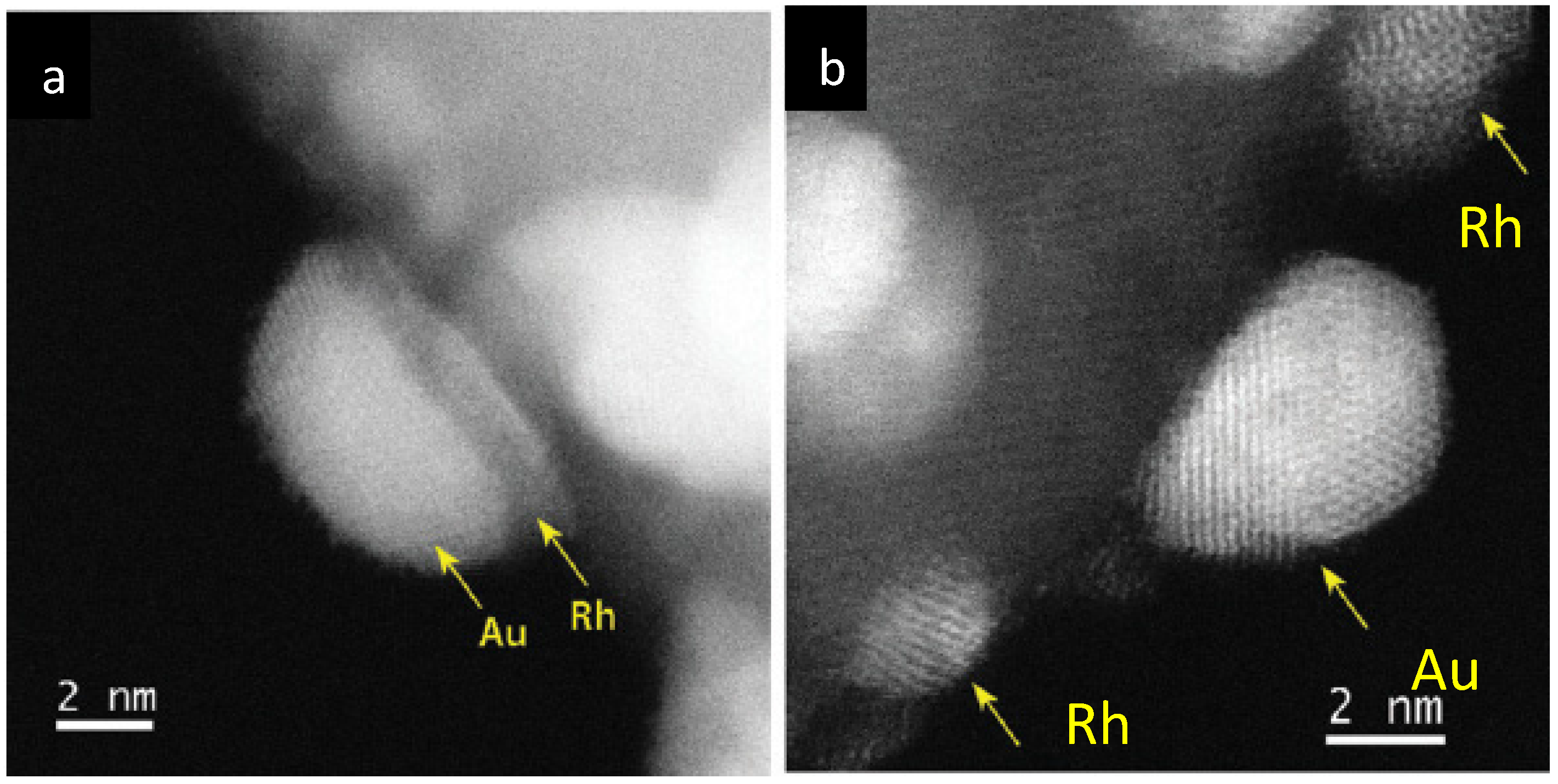
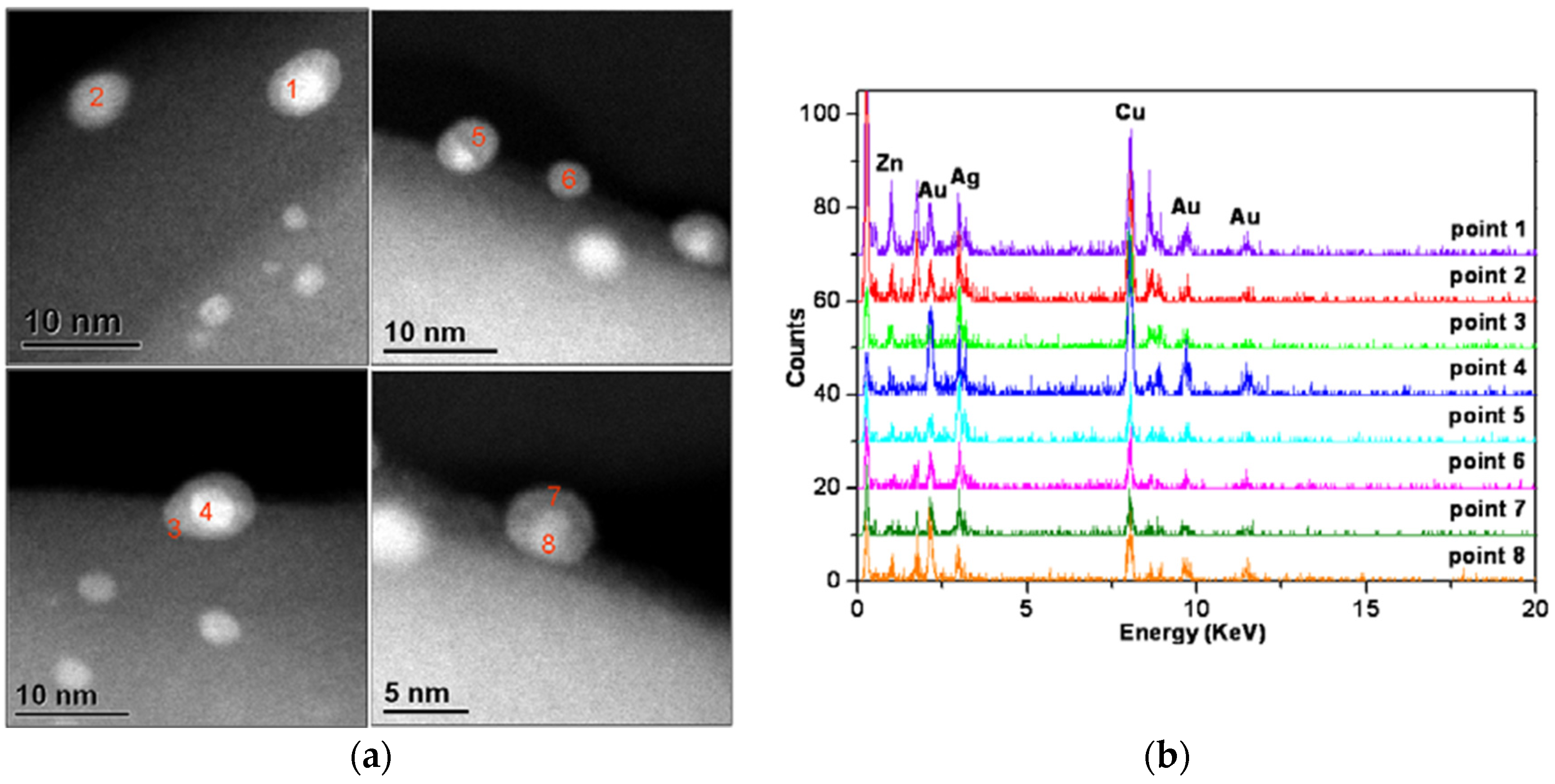
- Konuspayeva, Z.; Afanasiev, P.; Nguyen, T.-S.; Felice, L.D.; Morfin, F.; Nguyen, N.-T.; Nelayah, J.; Ricolleau, C.; Li, Z.Y.; Yuan, J.; et al. Au–Rh and Au–Pd nanocatalysts supported on rutile titania nanorods: Structure and chemical stability. Phys. Chem. Chem. Phys. 2015, 17, 28112–28120. [Google Scholar] [CrossRef] [PubMed]
- Benkó, T.; Beck, A.; Frey, K.; Srankó, D.F.; Geszti, O.; Sáfrán, G.; Maróti, B.; Schay, Z. Bimetallic Ag-Au/SiO2 catalysts: Formation, structure and synergisticactivity in glucose oxidation. Appl. Catal. A Gen. 2014, 479, 103–111. [Google Scholar] [CrossRef]
- Rodrigues, E.G.; Pereira, M.F.R.; Chen, X.; Delgado, J.J.; Órfão, J.J.M. Selective oxidation of glycerol over platinum-based catalysts supported on carbon nanotubes. Ind. Eng. Chem. Res. 2013, 52, 17390–17398. [Google Scholar] [CrossRef]
- Venezia, A.M.; Liotta, L.F.; Pantaleo, G.; Parola, V.L.; Deganello, G.; Beck, A.; Koppány, Z.; Frey, K.; Horváth, D.; Guczi, L. Activity of SiO2 supported gold-palladium catalysts in CO oxidation. Appl. Catal. A 2003, 251, 359–368. [Google Scholar] [CrossRef]
- Marx, S.; Krumeich, F.; Baiker, A. Surface properties of supported, colloid-derived gold/palladium mono- and bimetallic nanoparticles. J. Phys. Chem. C 2011, 115, 8195–8205. [Google Scholar] [CrossRef]
- Guczi, L.; Becka, A.; Horváth, A.; Koppány, Z.; Stefler, G.; Frey, K.; Sajó, I.; Geszti, O.; Bazin, D.; Lynch, J. AuPd bimetallic nanoparticles on TiO2: XRD, TEM, in situ EXAFS studies and catalytic activity in CO oxidation. J. Mol. Catal. A 2003, 204–205, 545–552. [Google Scholar] [CrossRef]
- Alshammari, A.; Kockritz, A.; Kalevaru, V.N.; Bagabas, A.; Martin, A. Potential of supported gold bimetallic catalysts for green synthesis of adipic acid from cyclohexane. Top. Catal. 2015, 58, 1069–1076. [Google Scholar] [CrossRef]
- Chen, H.; Li, Y.; Zhang, F.; Zhang, G.; Fan, X. Graphene supported Au-Pd bimetallic nanoparticles with core-shell structures and superior peroxidase-like activities. J. Mater. Chem. 2011, 21, 17658–17661. [Google Scholar] [CrossRef]
- Verbruggen, S.W.; Keulemans, M.; Filippousi, M.; Flahaut, D.; Tendeloo, G.V.; Lacombe, S.; Martens, J.A.; Lenaerts, S. Plasmonic gold-silver alloy on TiO2 photocatalysts with tunablevisible light activity. Appl. Catal. B Environ. 2014, 156–157, 116–121. [Google Scholar] [CrossRef]
- Llorca, J.; Domínguez, M.; Ledesma, C.; Chimentão, R.J.; Medina, F.; Sueiras, J.S.; Angurell, I.; Seco, M.; Rossell, O. Propene epoxidation over TiO2-supported Au-Cu alloy catalysts prepared from thiol-capped nanoparticles. J. Catal. 2008, 258, 187–198. [Google Scholar] [CrossRef]
- He, P.; Wang, Y.; Wang, X.; Pei, F.; Wang, H.; Liu, L.; Yi, L. Investigation of carbon supported Au-Ni bimetallic nanoparticles as electrocatalyst for direct borohydride fuel cell. J. Power Sour. 2011, 196, 1042–1047. [Google Scholar] [CrossRef]
- He, P.; Wang, X.; Liu, Y.; Liu, X.; Yi, L. Comparison of electrocatalytic activity of carbon-supported Au-M (M [ Fe, Co, Ni, Cu and Zn) bimetallic nanoparticles for direct borohydride fuel cells. Int. J. Hydr. Ener. 2012, 37, 11984–11993. [Google Scholar] [CrossRef]
- Szumełda, T.; Drelinkiewicz, A.; Kosydar, R.; Gurgul, J. Hydrogenation of cinnamaldehyde in the presence of PdAu/C catalystsprepared by the reverse “water-in-oil” microemulsion method. Appl. Catal. A Gen. 2014, 487, 1–15. [Google Scholar] [CrossRef]
- Menezes, W.G.; Zielasek, V.; Thiel, K.; Hartwig, A.; Bäumer, M. Effects of particle size, composition, and support on catalytic activity of AuAg nanoparticles prepared in reverse block copolymer micelles as nanoreactors. J. Catal. 2013, 299, 222–231. [Google Scholar] [CrossRef]
- Lee, L.-C.; Xiao, C.; Huang, W.; Zhao, Y. Palladium-gold bimetallic nanoparticle catalysts prepared by “controlled release” from metalloaded interfacially cross-linked reverse micelles. New J. Chem. 2015, 39, 2459–2466. [Google Scholar] [CrossRef]
- Scott, R.W.J.; Wilson, O.M.; Crooks, R.M. Synthesis, characterization, and applications of dendrimer-encapsulated nanoparticles. J. Phys. Chem. B 2005, 109, 692–704. [Google Scholar] [CrossRef] [PubMed]
- Chandler, B.D.; Gilbertson, J.D. PAMAM dendrimer templated nanoparticle catalysts. In Nanoparticles and Catalysis; Astruc, D., Ed.; Wiley-VCH: Weinheim, Germany, 2007. [Google Scholar]
- Lang, H.; Maldonado, S.; Stevenson, K.J.; Chandler, B.D. Synthesis and characterization of dendrimer templated supported bimetallic Pt-Au nanoparticles. J. Am. Chem. Soc. 2004, 126, 12949–12956. [Google Scholar] [CrossRef] [PubMed]
- Auten, B.J.; Lang, H.; Chandler, B.D. Dendrimer templates for heterogeneous catalysts: Bimetallic Pt-Au nanoparticles on oxide supports. Appl. Catal. B 2008, 81, 225–235. [Google Scholar] [CrossRef]
- Chandler, B.D.; Long, C.G.; Gilbertson, J.D.; Pursell, C.J.; Vijayaraghavan, G.; Stevenson, K.J. Enhanced oxygen activation over supported bimetallic Au-Ni catalysts. J. Phys. Chem. C 2010, 114, 11498–11508. [Google Scholar] [CrossRef]
- Song, Y.-J.; Jesús, Y.M.L.-D.; Fanson, P.T.; Williams, C.T. Preparation and characterization of dendrimer-derived bimetallic Ir-Au/Al2O3 catalysts for CO oxidation. J. Phys. Chem. C 2013, 117, 10999–11007. [Google Scholar] [CrossRef]
- Zheng, Z.; Li, H.; Liu, T.; Cao, R. Monodisperse noble metal nanoparticles stabilized in SBA-15: Synthesis, characterization and application in microwave-assisted Suzuki-Miyaura coupling reaction. J. Catal. 2010, 270, 268–274. [Google Scholar] [CrossRef]
- Pasini, T.; Piccinini, M.; Blosi, M.; Bonelli, R.; Albonetti, S.; Dimitratos, N.; Lopez-Sanchez, J.A.; Sankar, M.; He, Q.; Kiely, C.J.; et al. Selective oxidation of 5-hydroxymethyl-2-furfural using supported gold-copper nanoparticles. Green Chem. 2011, 13, 2091–2099. [Google Scholar] [CrossRef]
- Yin, M.; Huang, Y.; Lv, Q.; Liang, L.; Liao, J.; Liu, C.; Xing, W. Improved direct electrooxidation of formic acid by increasing Au fraction on the surface of PtAu alloy catalyst with heat treatment. Electrochim. Acta 2011, 58, 6–11. [Google Scholar] [CrossRef]
- Tang, S.; Vongehr, S.; Meng, X. Controllable incorporation of Ag and Ag-Au nanoparticles in carbon spheres for tunable optical and catalytic properties. J. Mater. Chem. 2010, 20, 5436–5445. [Google Scholar] [CrossRef]
- Tang, S.; Vongehr, S.; He, G.; Chen, L.; Meng, X. Highly catalytic spherical carbon nanocomposites allowing tunable activity via controllable Au-Pd doping. J. Coll. Interf. Sci. 2012, 375, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Chandler, B.D.; Schabel, A.B.; Blanford, C.F.; Pignolet, L.H. Preparation and characterization of supported bimetallic Pt-Au particle catalysts from molecular cluster and chloride salt precursors. J. Catal. 1999, 187, 367–384. [Google Scholar] [CrossRef]
- Albonetti, S.; Bonelli, R.; Delaigle, R.; Femoni, C.; Gaigneaux, E.M.; Morandi, V.; Ortolani, L.; Tiozzo, C.; Zacchini, S.; Trifiro, F. Catalytic combustion of toluene over cluster-derived gold/iron catalysts. Appl. Catal. A Gen. 2010, 372, 138–146. [Google Scholar] [CrossRef]
- Redjala, T.; Remita, H.; Apostolescu, G.; Mostafavi, M.; Thomazeau, C.; Uzio, D. Bimetallic Au-Pd and Ag-Pd clusters synthesised by γ or electron beam radiolysis and study of the reactivity/structure relationships in the selective hydrogenation of buta-1,3-diene. Oil Gas Sci. Tech. Rev. IFP 2006, 61, 789–797. [Google Scholar] [CrossRef]
- Doherty, R.P.; Krafft, J.-M.; Méthivier, C.; Casale, S.; Remita, H.; Louis, C.; Thomas, C. On the promoting effect of Au on CO oxidation kinetics of Au-Pt bimetallic nanoparticles supported on SiO2: An electronic effect? J. Catal. 2012, 287, 102–113. [Google Scholar] [CrossRef]
- Yamamoto, T.A.; Nakagawa, T.; Seino, S.; Nitani, H. Bimetallic nanoparticles of PtM (M = Au, Cu, Ni) supported on iron oxide: Radiolytic synthesis and CO oxidation catalysis. Appl. Catal. A 2010, 387, 195–202. [Google Scholar] [CrossRef]
- Barbier, J. Handbook of Heterogeneous Catalysis; Ertl, G., Knözinger, H., Weitkamp, J., Eds.; Wiley VCH: Weinheim, Germany, 1997; p. 257. [Google Scholar]
- Epron, F.; Especel, C.; Lafaye, G.; Marécot, P. Multimetallic nanoparticles prepared by redox processes applied in catalysis. In Nanoparticles and Catalysis; Astruc, D., Ed.; Wiley VCH: Weinheim, Germany, 2007; pp. 281–304. [Google Scholar]
- Especel, C.; Duprez, D.; Epron, F. Bimetallic catalysts for hydrogenation in liquid phase. C. R. Chim. 2014, 17, 790–800. [Google Scholar] [CrossRef]
- Beard, K.D.; Schaal, M.T.; Zee, J.W.V.; Monnier, J.R. Preparation of highly dispersed PEM fuel cell catalysts using electroless deposition methods. Appl. Catal. B Environ. 2007, 72, 262–271. [Google Scholar] [CrossRef]
- Rebelli, J.; Rodriguez, A.A.; Ma, S.; Williams, C.T.; Monnier, J.R. Preparation and characterization of silica-supported, group IB-Pd bimetallic catalysts prepared by electroless deposition methods. Catal. Today 2011, 160, 170–178. [Google Scholar] [CrossRef]
- An, Q.; Yu, M.; Zhang, Y.; Ma, W.; Guo, J.; Wang, C. Fe3O4 carbon microsphere supported Ag-Au bimetallic nanocrystals with the enhanced catalytic activity and selectivity for the reduction of nitroaromatic compounds. J. Phys. Chem. C 2012, 116, 22432–22440. [Google Scholar] [CrossRef]
- Guan, Y.; Zhao, N.; Tang, B.; Jia, Q.; Xu, X.; Liu, H.; Boughton, R.I. A stable bimetallic Au-Ag/TiO2 nanopaper for aerobic oxidation of benzyl alcohol. Chem. Comm. 2013, 49, 11524–11526. [Google Scholar] [CrossRef] [PubMed]
- Jia, Q.; Zhao, D.; Tang, B.; Zhao, N.; Li, H.; Sang, Y.; Bao, N.; Zhang, X.; Xu, X.; Liu, H. Synergistic catalysis of Au-Cu/TiO2-NB nanopaper in aerobic oxidation of benzyl alcohol. J. Mater. Chem. A 2014, 2, 16292–16298. [Google Scholar] [CrossRef]
- Sarkany, A.; Horvath, À.; Beck, A. Hydrogenation of acetylene over low loaded Pd and Pd-Au/SiO2 catalysts. Appl. Catal. A Gen. 2002, 229, 117–125. [Google Scholar] [CrossRef]
- Choudhary, T.V.; Sivadinarayana, C.; Datye, A.K.; Kumar, D.; Goodman, D.W. Acetylene hydrogenation on Au-based catalysts. Catal. Lett. 2003, 86, 1–8. [Google Scholar] [CrossRef]
- Espinosa, G.; Angel, G.D.; Barbier, J.; Marécot, P.; Schifter, I. Preparation of alumina-supported palladium platinum catalysts by surface redox reactions. Activity for complete hydrocarbon oxidation. Stud. Surf. Sci. Catal. 1997, 111, 421–426. [Google Scholar]
- Maris, E.P.; Ketchie, W.C.; Murayama, M.; Davis, R.J. Glycerol hydrogenolysis on carbon-supported PtRu and AuRu bimetallic catalysts. J. Catal. 2007, 251, 281–294. [Google Scholar] [CrossRef]
- Rebelli, J.; Detwiler, M.; Ma, S.; Williams, C.T.; Monnier, J.R. Synthesis and characterization of Au-Pd/SiO2 bimetallic catalysts prepared by electroless deposition. J. Catal. 2010, 270, 224–233. [Google Scholar] [CrossRef]
- Alba-Rubio, A.C.; Plauck, A.; Stangland, E.E.; Mavrikakis, M.; Dumesic, J.A. Direct synthesis of hydrogen peroxide over Au-Pd catalysts prepared by electroless deposition. Catal. Lett. 2015, 145, 2057–2065. [Google Scholar] [CrossRef]
- Griffin, M.B.; Rodriguez, A.A.; Montemore, M.M.; Monnier, J.R.; Williams, C.T.; Medlin, J.W. The selective oxidation of ethylene glycol and 1,2-propanediol on Au, Pd, and Au-Pd bimetallic catalysts. J. Catal. 2013, 307, 111–120. [Google Scholar] [CrossRef]
- Monyanon, S.; Pongstabodee, S.; Luengnaruemitchai, A. Preferential oxidation of carbon monoxide over Pt, Au monometallic catalyst, and Pt-Au bimetallic catalyst supported on ceria in hydrogen-rich reformate. J. Chin. Inst. Chem. Eng. 2007, 38, 435–441. [Google Scholar] [CrossRef]
- Pramanik, S.; Mishra, M.K.; De, G. Crystal structure tailoring of Au-Cu alloy NPs using the embedding film matrix as template. Cryst. Eng. Comm. 2014, 16, 56–63. [Google Scholar] [CrossRef]
- Bönnemann, H.; Endruschat, U.; Tesche, B.; Rufınska, A.; Lehmann, C.W.; Wagner, F.E.; Filoti, G.; Parvulescu, V.; Parvulescu, V.I. An SiO2-embedded nanoscopic Pd/Au alloy colloid. Eur. J. Inorg. Chem. 2000, 12, 819–822. [Google Scholar] [CrossRef]
- Dash, P.; Bond, T.; Fowler, C.; Hou, W.; Coombs, N.; Scott, R.W.J. Rational design of supported PdAu nanoparticle catalysts from structured nanoparticle precursors. J. Phys. Chem. C 2009, 113, 12719–12730. [Google Scholar] [CrossRef]
- Jiang, H.-L.; Umegaki, T.; Akita, T.; Zhang, X.-B.; Haruta, M.; Xu, Q. Bimetallic Au-Ni nanoparticles embedded in SiO2 Nanospheres: Synergetic catalysis in hydrolytic dehydrogenation of ammonia borane. Chem. Eur. J. 2010, 16, 3132–3137. [Google Scholar] [CrossRef] [PubMed]
- Scott, R.W.J.; Sivadinarayana, C.; Wilson, O.M.; Yan, Z.; Goodman, D.W.; Crooks, R.M. Titania-supported PdAu bimetallic catalysts prepared from dendrimer-encapsulated nanoparticle precursors. J. Am. Chem. Soc. 2005, 127, 1380–1381. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Lim, H.; Tang, Q.; Gao, Y.; Sun, T.; Yan, Q.; Yang, Y. Solvent-free aerobic oxidation of benzyl alcohol over Pd monometallic and Au-Pd bimetallic catalysts supported on SBA-16 mesoporous molecular sieves. Appl. Catal. A 2010, 380, 55–65. [Google Scholar] [CrossRef]
- Zheng, J.; Lin, H.; Wang, Y.; Zheng, X.; Duan, X.; Yuan, Y. Efficient low-temperature selective hydrogenation of esters on bimetallic Au-Ag/SBA-15 catalyst. J. Catal. 2013, 297, 110–118. [Google Scholar] [CrossRef]
- Lin, H.; Zheng, J.; Zheng, X.; Gu, Z.; Yuan, Y.; Yang, Y. Improved chemoselective hydrogenation of crotonaldehyde over bimetallic AuAg/SBA-15 catalyst. J. Catal. 2015, 330, 135–144. [Google Scholar] [CrossRef]
- Qu, Z.; Ke, G.; Wang, Y.; Liu, M.; Jiang, T.; Gao, J. Investigation of factors influencing the catalytic performance of CO oxidation over Au-Ag/SBA-15 catalyst. Appl. Surf. Sci. 2013, 277, 293–301. [Google Scholar] [CrossRef]
- Sobczak, I.; Dembowiak, E. The effect of AuAg-MCF and AuAg-NbMCF catalysts pretreatment onthe gold-silver alloy formation and the catalytic behavior in selectivemethanol oxidation with oxygen. J. Mol. Catal. A 2015, 409, 137–148. [Google Scholar] [CrossRef]
- Jiang, H.-L.; Akita, T.; Ishida, T.; Haruta, M.; Xu, Q. Synergistic catalysis of Au@Ag core-shell nanoparticles stabilized on metal-organic framework. J. Am. Chem. Soc. 2011, 133, 1304–1306. [Google Scholar] [CrossRef] [PubMed]
- Long, J.; Liu, H.; Wu, S.; Liao, S.; Li, Y. Selective oxidation of saturated hydrocarbons using Au-Pd alloy nanoparticles supported on metal-organic frameworks. ACS Catal. 2013, 3, 667–654. [Google Scholar] [CrossRef]
- Zhu, Q.-L.; Li, J.; Xu, Q. Immobilizing metal nanoparticles to metal-organic frameworks with size and location control for optimizing catalytic performance. J. Am. Chem. Soc. 2013, 135, 10210–10213. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhua, Q.-L.; Xu, Q. Highly active AuCo alloy nanoparticles encapsulated in the pores of metal–organic frameworks for hydrolytic dehydrogenation of ammonia borane. Chem. Comm. 2014, 50, 5899–5901. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Jia, S.-Y.; Wu, S.-H.; Li, P.-L.; Liu, C.-J.; Xu, Y.-M.; Qin, F.-X. Synthesis of highly dispersed metallic nanoparticles inside the pores of MIL-101(Cr) via the new double solvent method. Catal. Commun. 2015, 70, 44–48. [Google Scholar]


© 2016 by the author; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Louis, C. Chemical Preparation of Supported Bimetallic Catalysts. Gold-Based Bimetallic, a Case Study. Catalysts 2016, 6, 110. https://doi.org/10.3390/catal6080110
Louis C. Chemical Preparation of Supported Bimetallic Catalysts. Gold-Based Bimetallic, a Case Study. Catalysts. 2016; 6(8):110. https://doi.org/10.3390/catal6080110
Chicago/Turabian StyleLouis, Catherine. 2016. "Chemical Preparation of Supported Bimetallic Catalysts. Gold-Based Bimetallic, a Case Study" Catalysts 6, no. 8: 110. https://doi.org/10.3390/catal6080110
APA StyleLouis, C. (2016). Chemical Preparation of Supported Bimetallic Catalysts. Gold-Based Bimetallic, a Case Study. Catalysts, 6(8), 110. https://doi.org/10.3390/catal6080110




