High-Throughput Screening as a Supplemental Tool for the Development of Advanced Emission Control Catalysts: Methodological Approaches and Data Processing
Abstract
:1. Introduction
2. Results and Discussion
3. Experimental Section
3.1. Description of the HT Test Unit
- Forty eight reaction and 1 by-pass positions with liners made from stainless steel with an inner diameter of 7 mm for each individual reactor (cf. Figure 2).
- Liner filling up to 1 mL.
- T = 110–575 °C; atmospheric pressure; GHSV (gas hourly space velocity): 30,000–100,000 h−1.
- Dosing of gases (NO, NO2, NH3, N2O, HC, CO, CO2, O2, N2) and liquids (H2O, HC).
- Online gas analyzers for NO, NO2, N2O, NH3, HC, CO, CO2, O2 and H2, as well as a mass spectrometer for specific compounds (m/z 1–512).
- Changes in feed gas composition in the s range (e.g., lean/rich cycles).
- Flexible process control (“hteControl4”) for automated experiments with complex test protocols, as well as unattended and safe 24/7 operation.
- Easy change between preconfigured operation modes (diesel oxidation catalyst (DOC), TWC, selective catalytic reduction (SCR), lean NOx traps (LNT)).
- Automated data processing and data management system (“myhte”) allowing easy data export to sophisticated data analysis solutions.


3.2. S-Aging
- Temperatures: 300–450 °C.
- Feed gas: 10–100 ppm SO2, 10% H2O in air; optional: 100–200 ppm NO.
- Duration: 5–70 h.
- After exposure to SO2, H2O and NO (if used), the catalysts are cooled in air.
3.3. Test Procedure
3.3.1. Catalyst Amount and Shape
3.3.2. Hydrothermal Aging
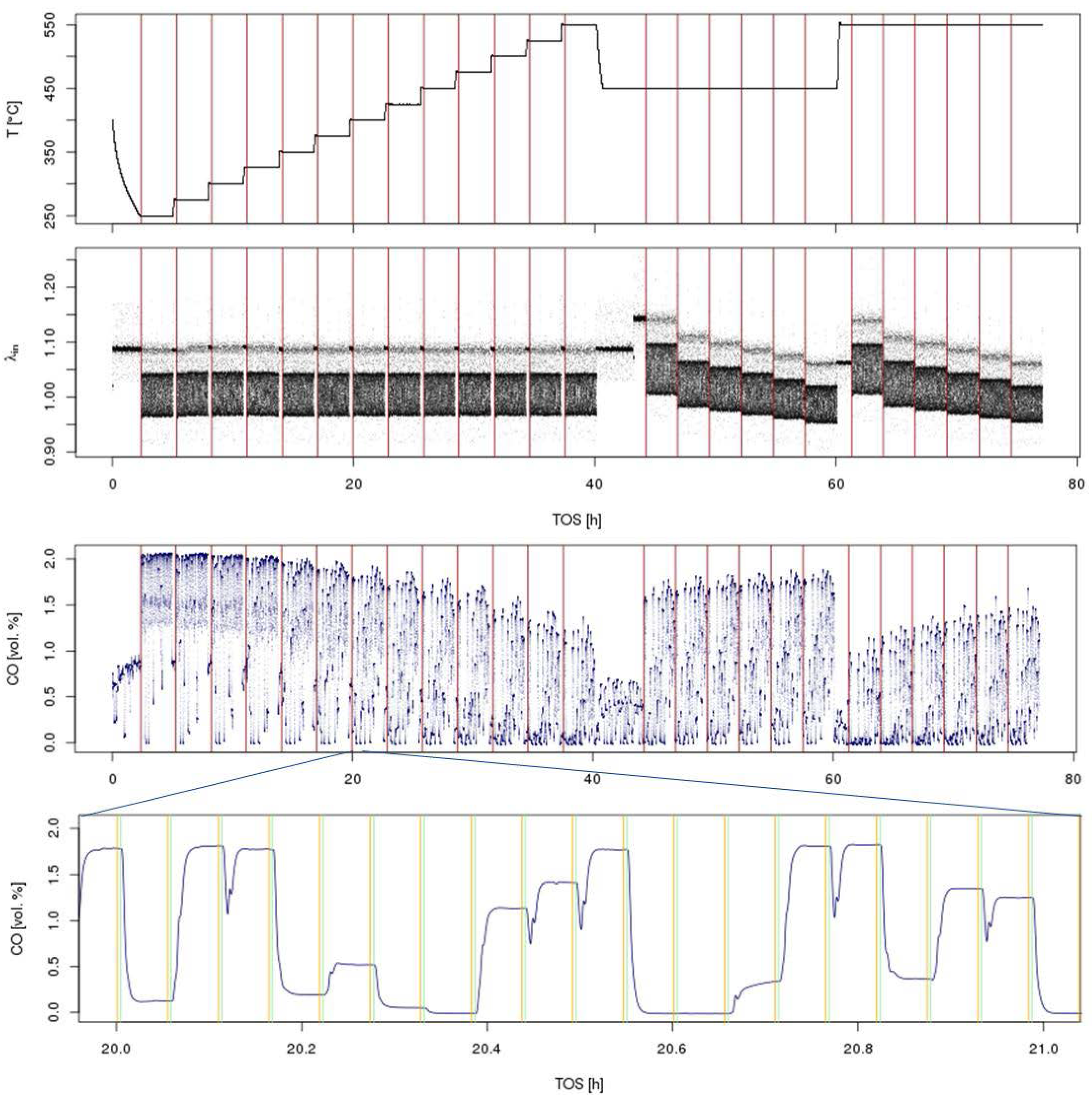
3.3.3. Screening Protocols
- Set first experimental condition (temperature, feed gas levels).
- Wait until the whole reactor is equilibrated.
- Switch to Position 1.
- Equilibrate in stationary feed or run dynamic feed switching program.
- Repeat Steps 3 and 4 for all 48 reactor positions.
- Set next experimental condition (e.g., higher temperature).
- Continue with Steps 2–6 until all conditions are evaluated for all 48 reactors.
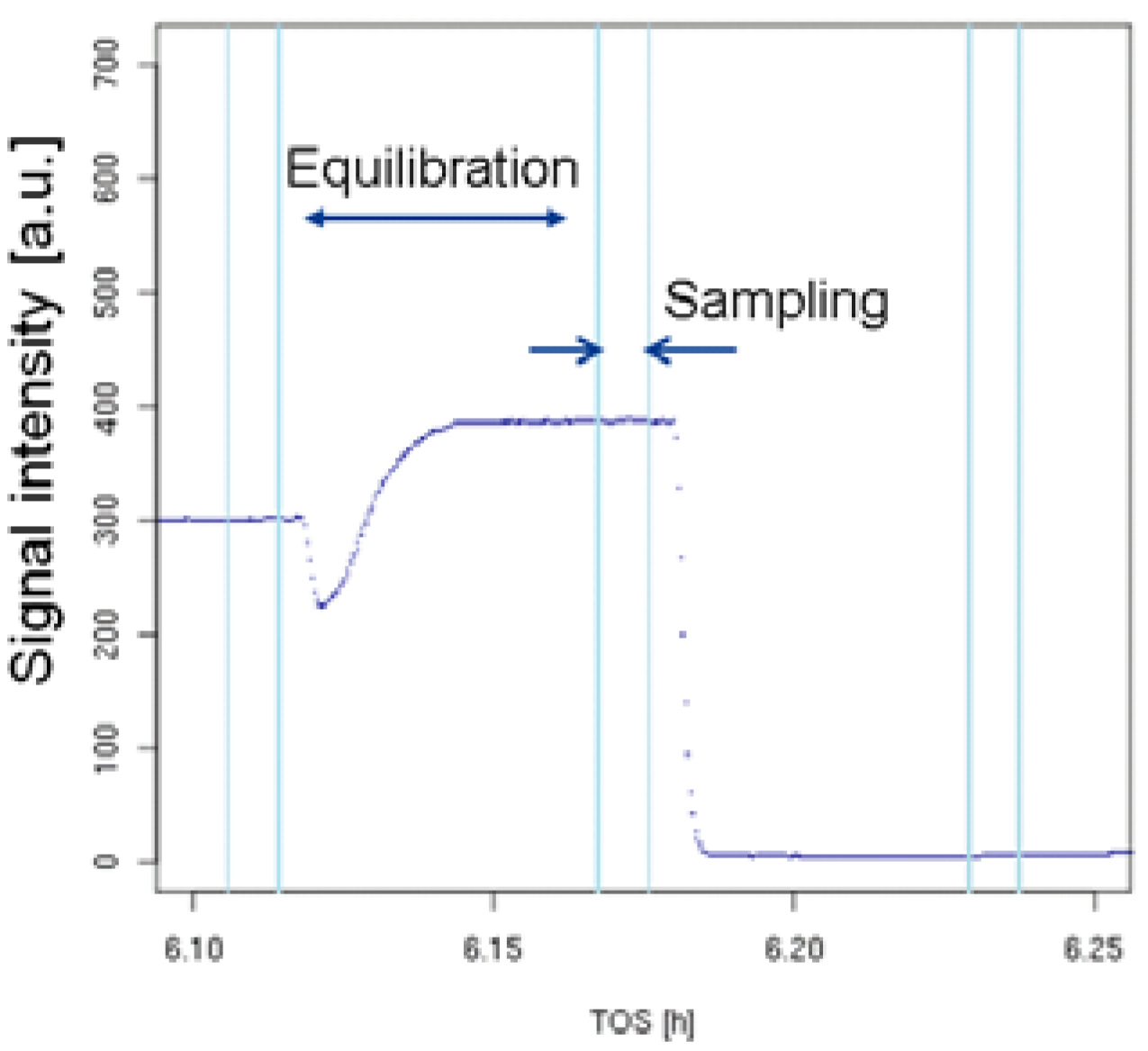
3.3.4. Data Processing
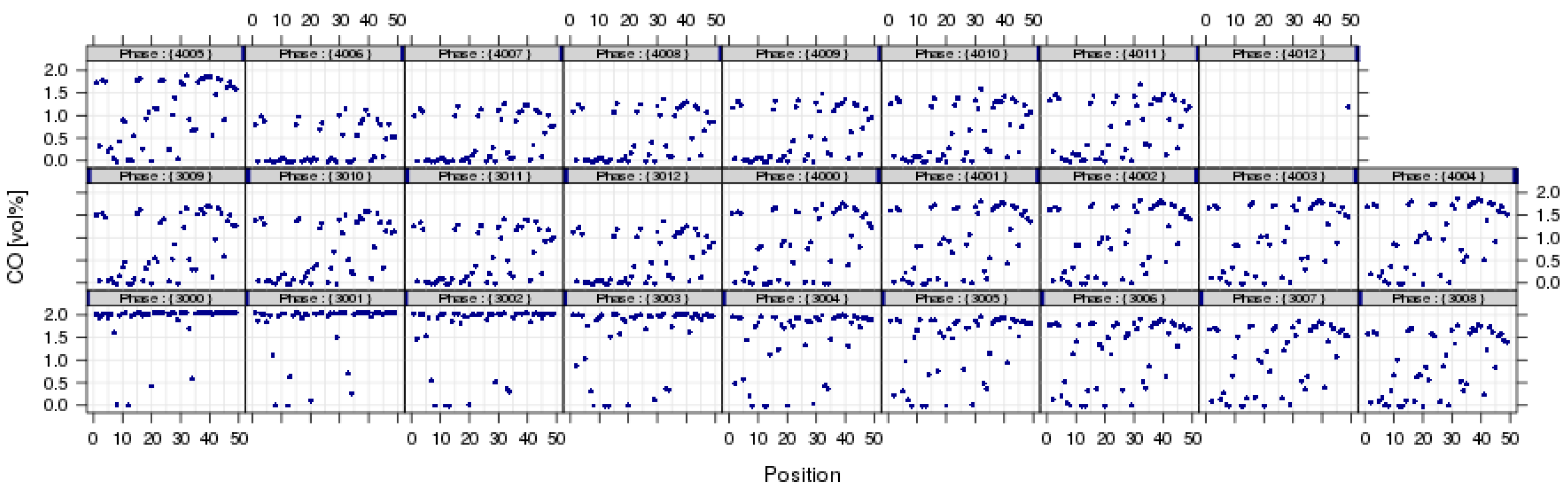
3.3.5. Quality Assurance
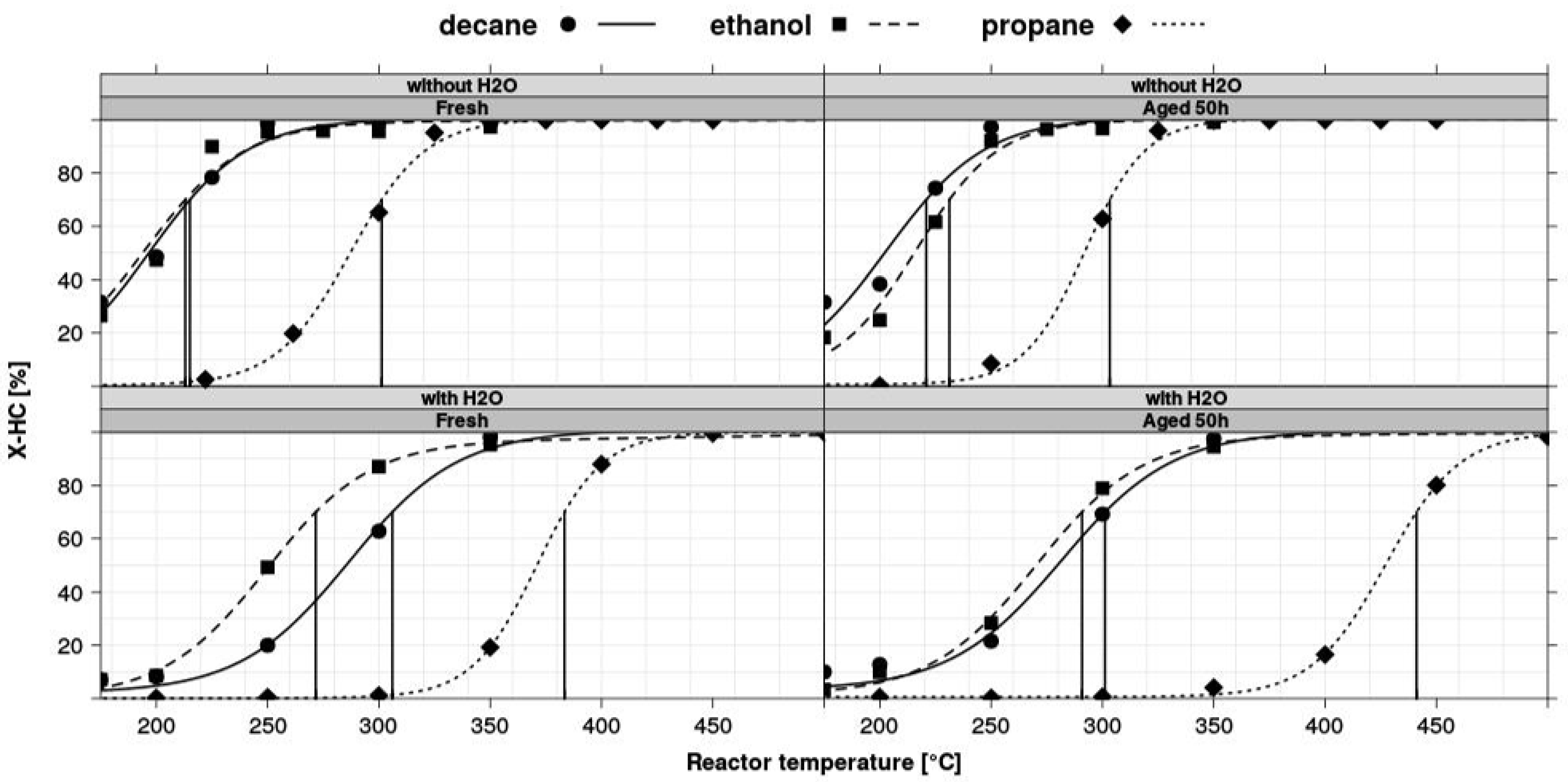
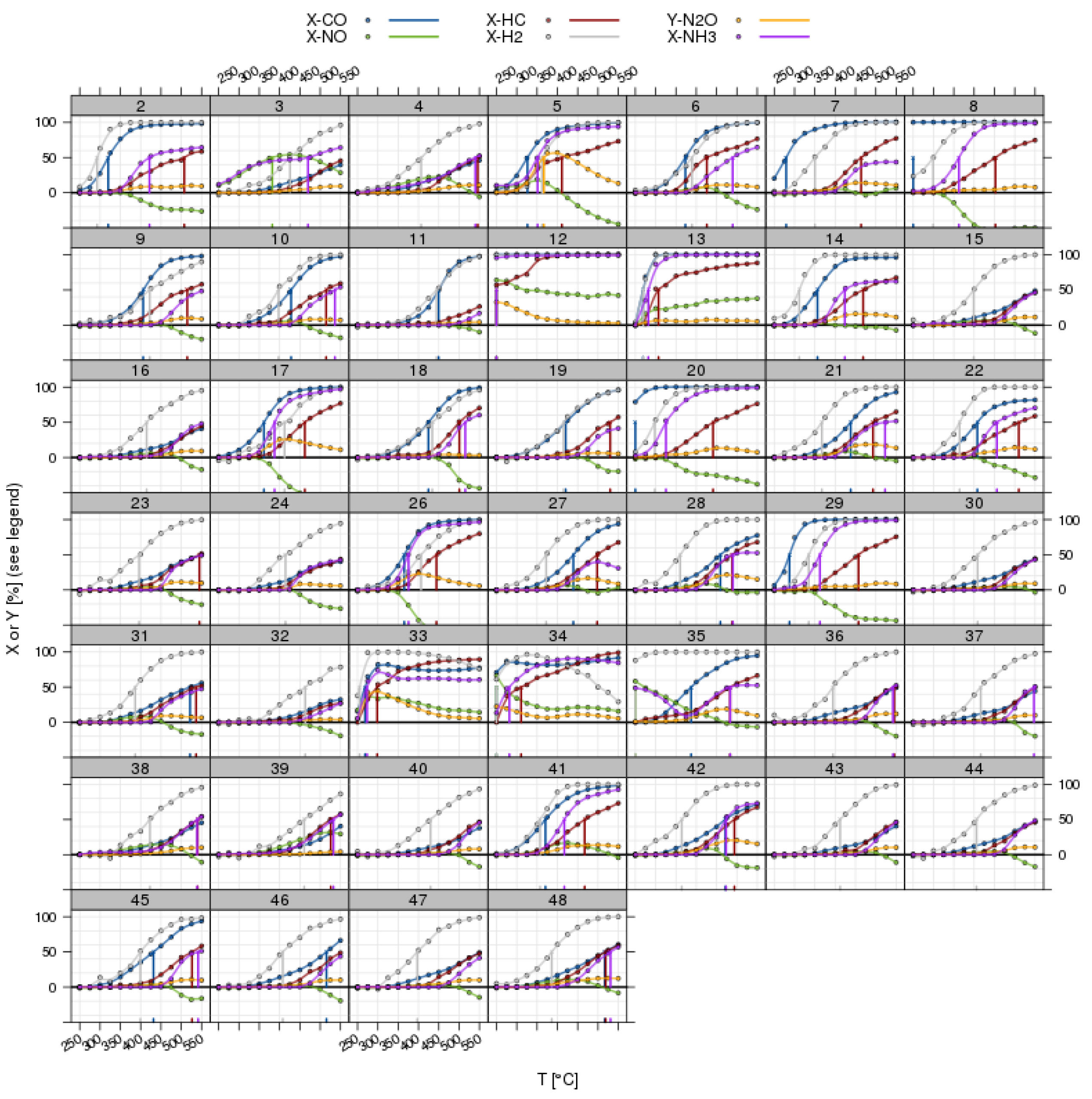
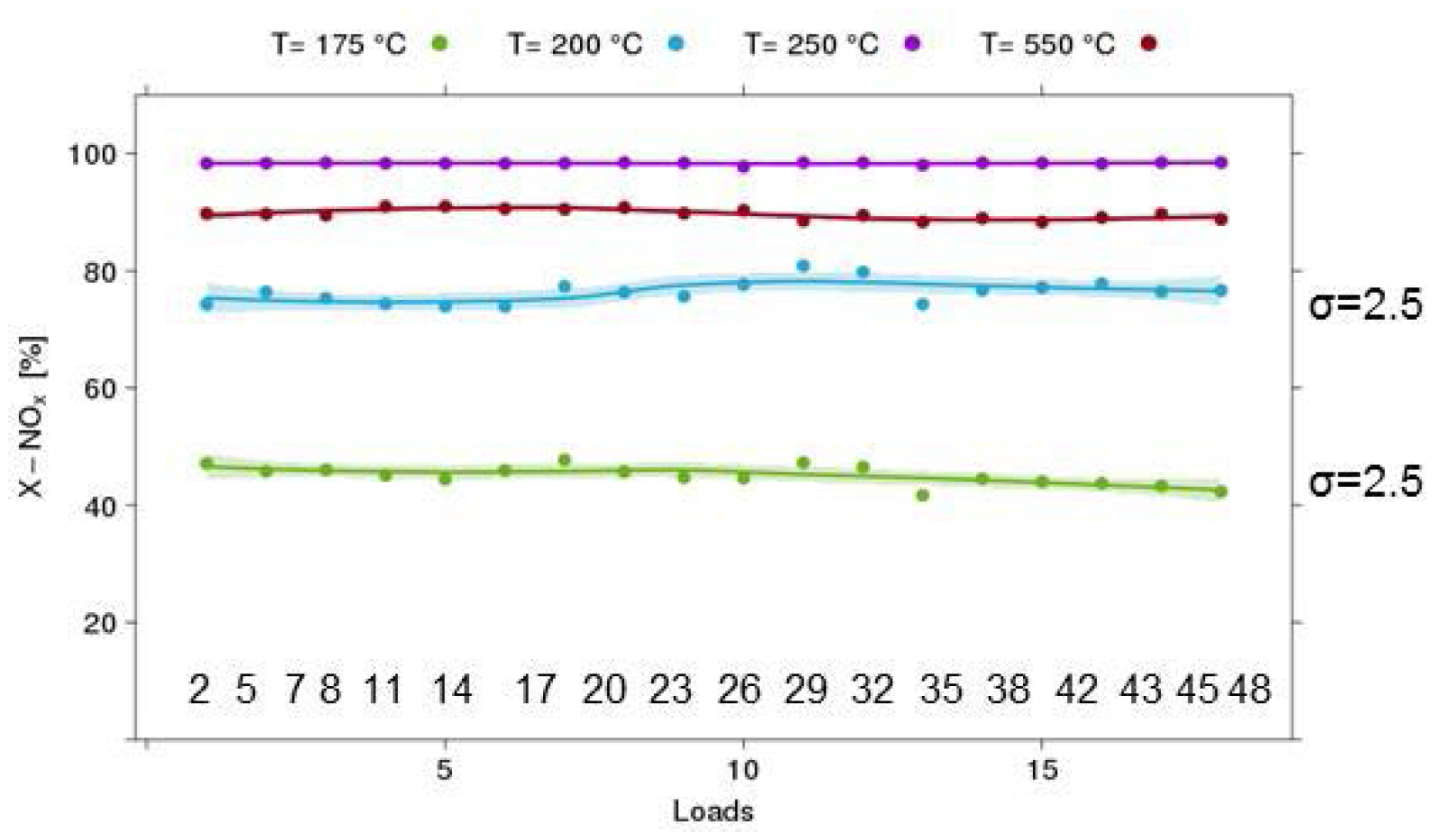
3.4. Test Protocols
3.4.1. Steady-State Tests
- Diesel oxidation catalysts (DOC): Although new diesel cars consist of rather complex system arrangements, including a particulate filter and components for the reduction of NOx emission, the catalytic functionality to oxidize CO and HC remains an important component of diesel aftertreatment systems. Typical tests involve several stationary light-off experiments on fresh and oven-aged samples, including tests for performance after S-aging and thermal regeneration.
- Selective catalytic reduction (SCR): Experiments are performed with a simulated exhaust using NH3 as the reductant. Samples are tested under standard SCR conditions (i.e., without NO2 in the feed) and with varying levels of the NO2/NOx ratio. This test also involves a dynamic component to measure the NH3 storage capacity in a cycling feed with and without NH3.
- NH3 oxidation: An important component of an SCR aftertreatment system, the NH3 oxidation catalyst ensures removal of unconverted NH3 downstream of the SCR catalyst.
| Catalyst technology | Feed compositions | GHSV (kh−1) | Temperatures |
|---|---|---|---|
| Diesel oxidation catalyst (DOC) | 500–3000 ppm CO, 80–500 ppm-C1 HC (HC = methane, propene, octane, decane, toluene or mixtures thereof) 50–200 ppm NO 5%–10% O2, 5%–10% CO2, 5%–10% H2O | 45–80 | up to 12 temperature levels between 120 and 350 °C |
| Selective catalytic reduction (SRC) | 50–1000 ppm NO 50–300 ppm NO2 50–1000 ppm NH3 5%–10% O2, 5%–10% H2O Optional: 5%–10% CO2 50–500 ppm-C1 HC (see DOC) | 30–90 | 5–10 temperature levels between 150 and 575 °C |
| Ammonia oxidation | 50–1000 ppm NH3 5%–10% O2, 5%–10% H2O | 30–90 | 2 temperature levels between 200 and 575 °C |
3.4.2. Dynamic tests with feed switches (see Figure 9)
| Catalyst technology | Feed compositions | GHSV (kh−1) | Temperatures |
|---|---|---|---|
| Three-way catalysis | 1%–3% CO, 1000–4000 ppm-C1 HC (HC = methane, propene, propane, iso-butane, iso-octane, toluene or mixtures thereof) 1%–3% H2 500–2000 ppm NO 0.5%–3 % O2, 10%–15 % CO2, 10%–15 % H2O Oscillating lean/rich perturbations with defined average λ and amplitude (frequency 0.5–0.2 Hz) | 45–80 | up to 12 temperature levels between 200 and 550 °C |
| LNT (lean NOx trap) | 50–1000 ppm NO 50–300 ppm NO2 500–3000 ppm CO, 5%–10% O2, 5%–10% H2O Optional: 5%–10% CO2 50–500 ppm-C1 HC (see DOC) Rich spikes (3–10 s) with defined λ (by reducing O2 and increasing CO/H2) | 30–90 | 5–10 temperature levels between 150 and 575 °C |
| OSC (oxygen storage capacity) | Alternating gas atmosphere with 2% CO 1% O2 (variable duration 10/10 s, 30/30 s) | 60 | 2 temperature levels between 350 and 575 °C |
- TWC (three-way catalysis): Three-way catalysts operate best in an exhaust of an engine operated close to stoichiometry, an air:fuel ratio of 1. As the catalyst has to tolerate excursions from this optimal point, tests with λ perturbations are crucial. Several protocols are used to evaluate the different functionalities of fresh and aged catalysts.
- Light-off with average λ = 1 and high frequency (up to 0.5 Hz) λ perturbations.
- λ-sweep test at different temperatures. Performed by running light-off tests, but with different average lambda set points, and then plotting conversion as a function of lambda at constant temperature.
- OSC test (oxygen storage capacity): specific test for the oxygen-buffer functionality of the catalyst. Involves a feed cycling between CO and O2 in nitrogen and monitoring CO2 generation from stored oxygen. Results of this test have been previously reported [26].
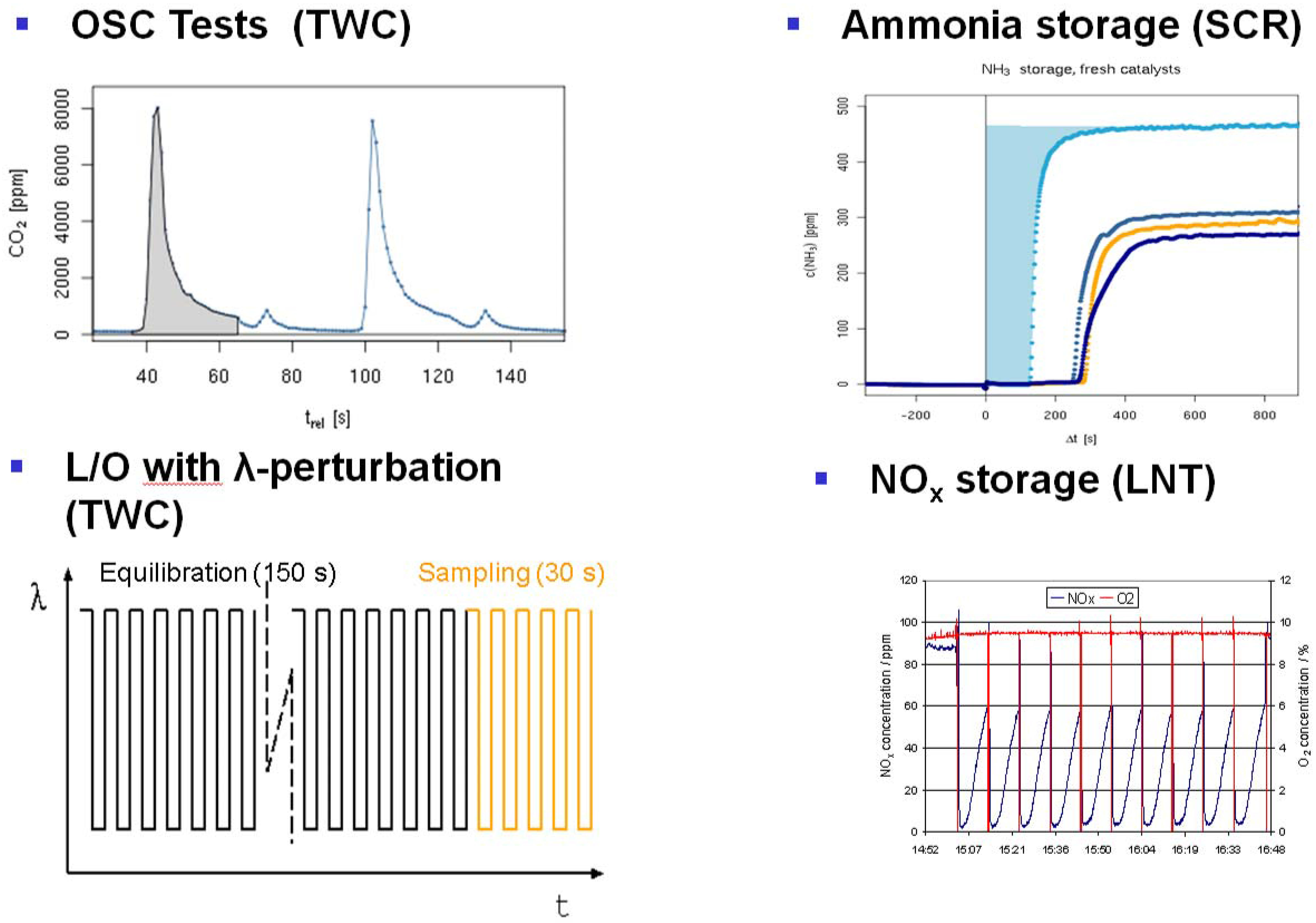
Figure 9. Examples for dynamic test protocols implemented on a 48-fold reactor system. Top left: OSC test, cycling O2 and CO containing feed and monitoring CO2 release in the absence of O2 in the gas phase, which is a measure for the oxygen storage capacity at a given temperature. Top right: NH3 storage test by cycling NH3 and monitoring the NH3 breakthrough under SCR reaction conditions (i.e., in the presence of NO, O2, H2O, optionally CO2 and/or HC). Bottom right: lean NOx traps (LNT) test with repeated rich regeneration cycles with defined λ. The NOx breakthrough is monitored and analyzed. Bottom left: TWC testing in a feed with rapid λ oscillations with defined amplitude. After an equilibration time of 150–180 s, the traces from relevant gas analyzers are averaged for 30 s.Figure 9. Examples for dynamic test protocols implemented on a 48-fold reactor system. Top left: OSC test, cycling O2 and CO containing feed and monitoring CO2 release in the absence of O2 in the gas phase, which is a measure for the oxygen storage capacity at a given temperature. Top right: NH3 storage test by cycling NH3 and monitoring the NH3 breakthrough under SCR reaction conditions (i.e., in the presence of NO, O2, H2O, optionally CO2 and/or HC). Bottom right: lean NOx traps (LNT) test with repeated rich regeneration cycles with defined λ. The NOx breakthrough is monitored and analyzed. Bottom left: TWC testing in a feed with rapid λ oscillations with defined amplitude. After an equilibration time of 150–180 s, the traces from relevant gas analyzers are averaged for 30 s. - LNT (lean NOx trap): Important for lab testing of NOx storage catalysts is the ability to generate reproducible rich pulses with defined rich λ and pulse width. Usually, 3–7 pulses are applied to each catalyst. After each rich pulse, the NOx breakthrough curve is recorded and integrated in several ways to calculate average NOx conversion or average NOx storage. Here, it is also important to compare storage in each individual rich/lean cycle and to monitor time on stream effects. The LNT test is usually run at several temperatures. A crucial component of LNT evaluation is activity after S-aging and lean/rich regeneration.
- Ammonia storage for SCR to measure the NH3 storage capacity in a cycling feed with and w/o NH3.
| Topic | Performance characteristics/Protocols | Typical throughput (samples/week) | Data output (data points/week) |
|---|---|---|---|
| DOC | Light-off performance (CO/HC/NO) and sulfur resistance 2–5 light-offruns per sample | 45–135 | 4500–11,250 |
| SCR | Low and high temperature SCR performance 4 protocols (standard. and fast SCR, ammonia oxidation and storage) | 30–45 (all protocols) 45–135 (w/o NH3 storage) | 2800–4200 |
| TWC | Oxygen storage capacity (OSC) Catalytic performance: light-off, λ-sweep for CO/HC/NO | 225 (OSC) 45–90 (L/O and λ-sweep) | 900 (OSC) 6000–12,000- (L/O and λ-sweep) |
| LNT | NOx efficiencies (lean/rich and lean) and NOx storage (lean) at 3 temperatures, 5 cycles per position, two sample loads | 45 | 1800–2700 |
4. Summary
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Heck, R.M.; Farrauto, R.J.; Gulati, S.T. Catalytic Air Pollution Control, Commercial Technology, 3rd ed.; John Wiley & Sons: Hoboken, New Jersey, 2009. [Google Scholar]
- Deutschmann, O.; Grunwaldt, J.-D. Abgasnachbehandlung in mobilen Systemen: Stand der Technik, Herausforderungen und Perspektiven. Chem. Ing. Tech. 2013, 85, 595–617. [Google Scholar] [CrossRef]
- Rodemerck, U.; Wolf, D.; Buyevskaya, O.V.; Claus, P.; Senkan, S.; Baerns, M. High-throughput synthesis and screening of catalytic materials: Case study on the search for a low-temperature catalyst for the oxidation of low-concentration propane. Chem. Eng. J. 2001, 82, 3–11. [Google Scholar] [CrossRef]
- Hendershot, R.J.; Lasko, S.S.; Fellmann, M.F.; Oskarsdottir, G.; Delgass, W.N.; Snively, C.M.; Lauterbach, J. A novel reactor system for high throughput catalyst testing under realistic conditions. Appl. Catal. A 2003, 254, 107–120. [Google Scholar] [CrossRef]
- Lucas, M.; Claus, P. High throughput screening in monolith reactors for total oxidation reactions. Appl. Catal. A 2003, 254, 35–43. [Google Scholar] [CrossRef]
- Mills, P.L.; Nicole, J.F. A novel reactor for high-throughput screening of gas-solid catalyzed reactions. Chem. Eng. Sci. 2004, 59, 5345–5354. [Google Scholar] [CrossRef]
- Paul, J.S.; Jacobs, P.A.; Weiss, P.-A.W.; Maier, W.F. Combinatorial discovery of new catalysts for the selective oxidation of isobutene. Appl. Catal. A 2004, 265, 185–193. [Google Scholar]
- Zech, T.; Bohner, G.; Klein, J. High-throughput screening of supported catalysts in massively parallel single-bead microreactors: Workflow aspects related to reactor bonding and catalyst preparation. Catal. Today 2005, 110, 58–67. [Google Scholar] [CrossRef]
- Brooks, C.; Cypes, S.; Grasselli, R.K.; Hagemeyer, A.; Hogan, Z.; Lesik, A.; Streukens, G.; Volpe, A.F., Jr.; Turner, H.W.; Weinberg, W.H.; et al. High throughput discovery of CO oxidation/VOC combustion and water-gas shift catalysts for industrial multi-component streams. Top. Catal. 2006, 38, 195–209. [Google Scholar] [CrossRef]
- Farrusseng, D. High-throughput heterogeneous catalysis. Surf. Sci. Rep. 2008, 63, 487–513. [Google Scholar] [CrossRef]
- Moehmel, S.; Steinfeldt, N.; Engelschalt, S.; Holena, M.; Kolf, S.; Baerns, M.; Dingerdissen, U.; Wolf, D.; Weber, R.; Bewersdorf, M. New catalytic materials for the high-temperature synthesis of hydrocyanic acid from methane and ammonia by high-throughput approach. Appl. Catal. A 2008, 334, 73–83. [Google Scholar] [CrossRef]
- Turner, H.W.; Volpe, A.F., Jr.; Weinberg, W.H. High-throughput heterogeneous catalyst research. Surf. Sci. 2009, 603, 10–12. [Google Scholar] [CrossRef]
- Gaudillere, C.; Vernoux, P.; Mirodatos, C.; Caboche, G.; Farrusseng, D. Screening of ceria-based catalysts for internal methane reforming in low temperature SOFC. Catal. Today 2010, 157, 263–269. [Google Scholar] [CrossRef]
- Valtchev, M.; Hammes, M.; Richter, R.; Holtzen, H.; Stowe, K.; Maier, W.F. Corrosion-Resistant Parallel Fixed-Bed Reactors for High-Throughput Screening of New Deacon Reaction Catalysts. Chem. Eng. Technol. 2014, 37, 1251–1260. [Google Scholar] [CrossRef]
- Zhu, H.B.; Laveille, P.; Rosenfeld, D.C.; Hedhili, M.N.; Basset, J.M. A high-throughput reactor system for optimization of Mo-V-Nb mixed oxide catalyst composition in ethane ODH. Catal. Sci. Technol. 2015, 5, 4164–4173. [Google Scholar] [CrossRef]
- Serna, P.; Baumes, L.A.; Moliner, M.; Corma, A. Combining high-throughput experimentation, advanced data modeling and fundamental knowledge to develop catalysts for the epoxidation of large olefins and fatty esters. J. Catal. 2008, 258, 25–34. [Google Scholar] [CrossRef]
- Brenner, A.; Schüth, F.; Schunk, S.A.; Stichert, W. Anordnung zum Testen der katalytischen Aktivität von einem Reaktionsgas ausgesetzten Feststoffen. DE 198 61 316 B4, 6 October 2005. [Google Scholar]
- Haas, A.; Strehlau, W.; Brenner, A.; Koechel, O.; Friess, M.; Zech, T. Device and method for pressure and flow control in parallel reactors. WO 2005063372 A2, 17 July 2005. [Google Scholar]
- Strehlau, W.; Gerlach, O.; Maier, J.; Gabriel, T. Catalysts for the simultaneous removal of carbon monoxide and hydrocarbons from oxygen-rich exhaust gases and processes for the manufacture thereof. WO 2005102513 A1, 3 November 2005. [Google Scholar]
- Strehlau, W.; Gerlach, O.; Maier, J.; Gabriel, T. Catalyst for the treatment of exhaust gases and processes for producing the same. WO 2006120013 A1, 16 November 2006. [Google Scholar]
- Hendershot, R.J.; Rogers, W.B.; Snively, C.A.; Ogunnaike, B.A.; Lauterbach, J. Development and optimization of NOx storage and reduction catalysts using statistically guided high-throughput experimentation. Catal. Today 2004, 98, 375–385. [Google Scholar] [CrossRef]
- Hendershot, R.J.; Vijay, R.; Snively, C.M.; Lauterbach, J. High-throughput study of the performance of NOx storage and reduction catalysts as a function of cycling conditions and catalyst composition. Chem. Eng. Sci. 2006, 61, 3907–3916. [Google Scholar] [CrossRef]
- Iojoiu, E.E.; Bassou, B.; Guilhaume, N.; Farrusseng, D.; Desmartin-Chomel, A.; Lombaert, K.; Bianchi, D.; Mirodatos, C. High-throughput approach to the catalytic combustion of diesel soot. Catal. Today 2008, 137, 103–109. [Google Scholar] [CrossRef]
- Kern, P.; Klimczak, M.; Heinzelmann, T.; Lucas, M.; Claus, P. High-throughput study of the effects of inorganic additives and poisons on NH3-SCR catalysts. Part II: Fe-zeolite catalysts. Appl. Catal. B 2010, 95, 48–56. [Google Scholar] [CrossRef]
- Gärtner, A.; Lenk, T.; Kiemel, R.; Casu, S.; Breuer, C.; Stöwe, K. High-throughput screening approach to identify new catalysts for total oxidation of methane from gas fueled lean burn engines. In Proceedings of the Preprints of the tenth international congress on Catalysis and automotive pollution control, Brussels, Belgium, 28–30 October 2015.
- Sundermann, A.; Gerlach, O. High-Throughput Screening Technology for Automotive Applications. Chem. Ing. Tech. 2014, 86, 1941–1947. [Google Scholar] [CrossRef]
- Montgomery, D.C. Design and Analysis of Experiments, 8th ed.; John Wiley & Sons: Hoboken, New Jersey, 2012. [Google Scholar]
- Box, G.E.P.; Hunter, J.S.; Hunter, W.G. Statistics for Experimenters: Design, Innovation, and Discovery, 2nd ed.; Wiley: New York, NY, USA, 2005. [Google Scholar]
- Team, R.C. R Foundation for Statistical Computing, Vienna, Austria. Available online: http://www.R-project.org/ (accessed on 25 January 2016).
- Ricketts, J.H.; Head, G.A. A five-parameter logistic equation for investigating asymmetry of curvature in baroreflex studies. Am. J. Phys.-Regul. Integr. Comp. Phys. 1999, 277, R441–R454. [Google Scholar]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sundermann, A.; Gerlach, O. High-Throughput Screening as a Supplemental Tool for the Development of Advanced Emission Control Catalysts: Methodological Approaches and Data Processing. Catalysts 2016, 6, 23. https://doi.org/10.3390/catal6020023
Sundermann A, Gerlach O. High-Throughput Screening as a Supplemental Tool for the Development of Advanced Emission Control Catalysts: Methodological Approaches and Data Processing. Catalysts. 2016; 6(2):23. https://doi.org/10.3390/catal6020023
Chicago/Turabian StyleSundermann, Andreas, and Olga Gerlach. 2016. "High-Throughput Screening as a Supplemental Tool for the Development of Advanced Emission Control Catalysts: Methodological Approaches and Data Processing" Catalysts 6, no. 2: 23. https://doi.org/10.3390/catal6020023
APA StyleSundermann, A., & Gerlach, O. (2016). High-Throughput Screening as a Supplemental Tool for the Development of Advanced Emission Control Catalysts: Methodological Approaches and Data Processing. Catalysts, 6(2), 23. https://doi.org/10.3390/catal6020023