Study on the Relation between the Mn/Al Mixed Oxides Composition and Performance of FCC Sulfur Transfer Agent
Abstract
:1. Introduction
2. Results and Discussion
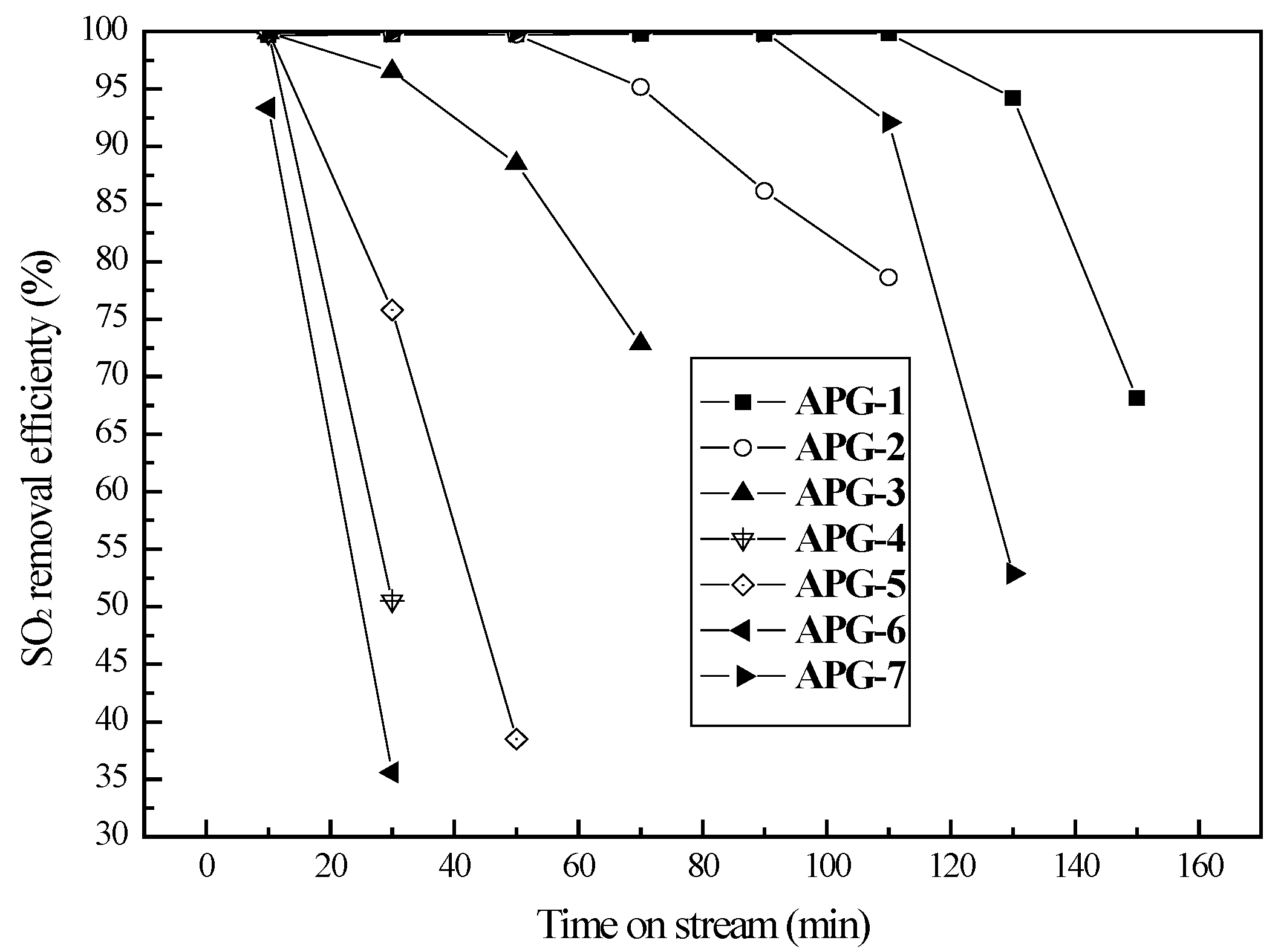
2.1. Evaluation of Activity of Sulfur Transfer Agent
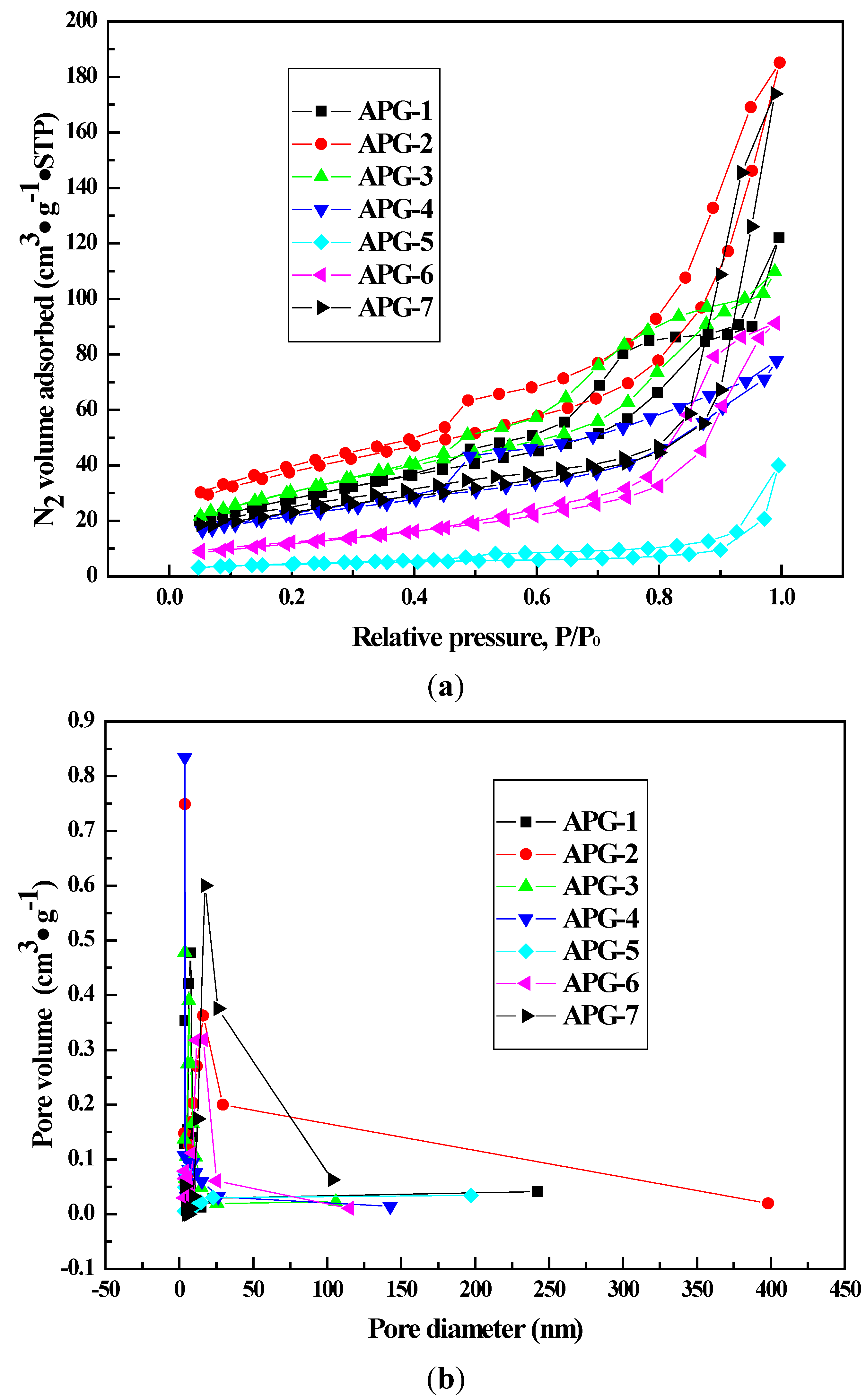
| Samples | Most Probable Pore Size/nm | SBET/m2·g−1 |
|---|---|---|
| APG-1 | 7.6 | 143.6 |
| APG-2 | 9.4 | 142.7 |
| APG-3 | 7.6 | 143 |
| APG-4 | 9.1 | 80.5 |
| APG-5 | 6.5 | 86.3 |
| APG-6 | 16.3 | 67.2 |
| APG-7 | 17.3 | 80.8 |
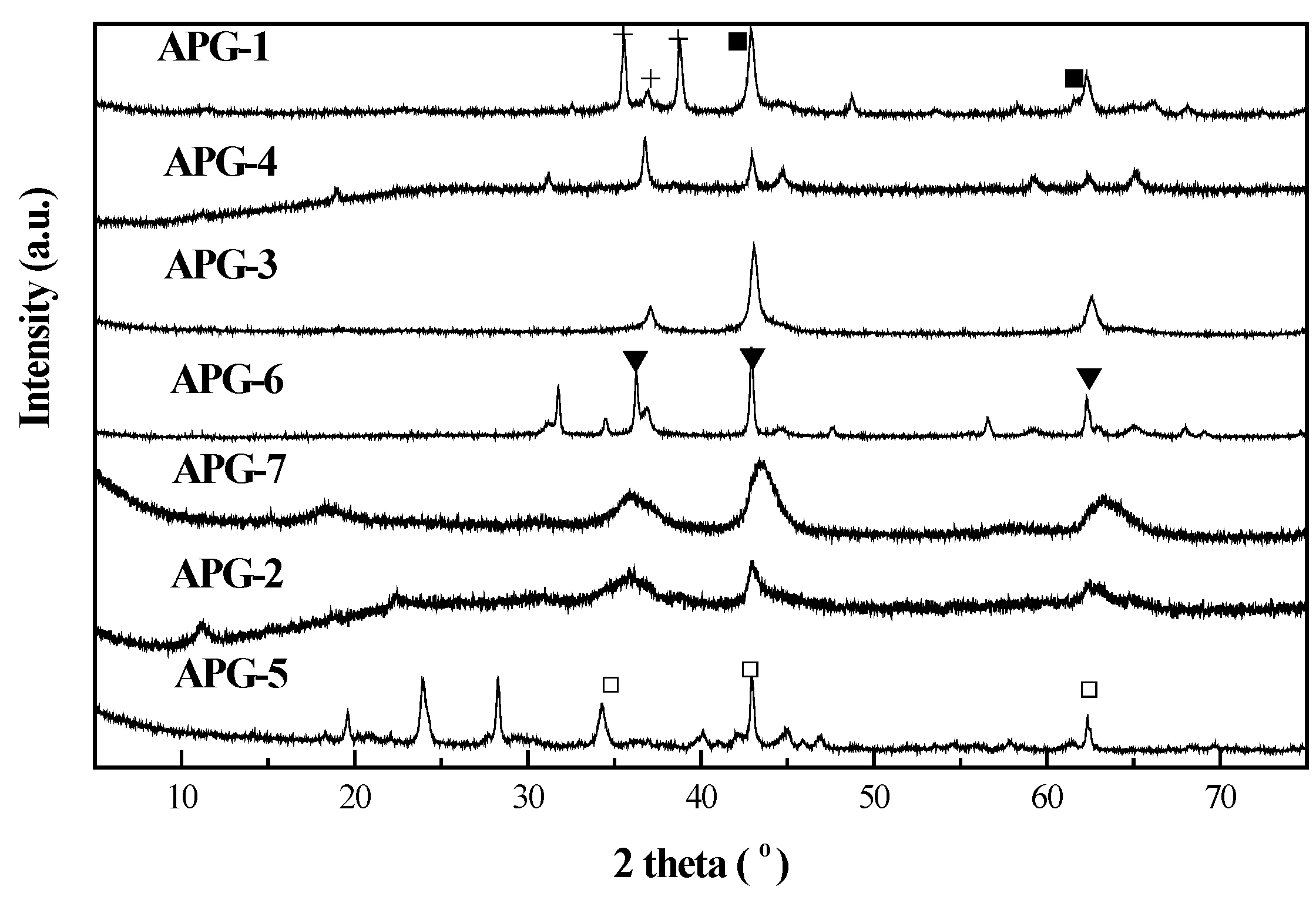
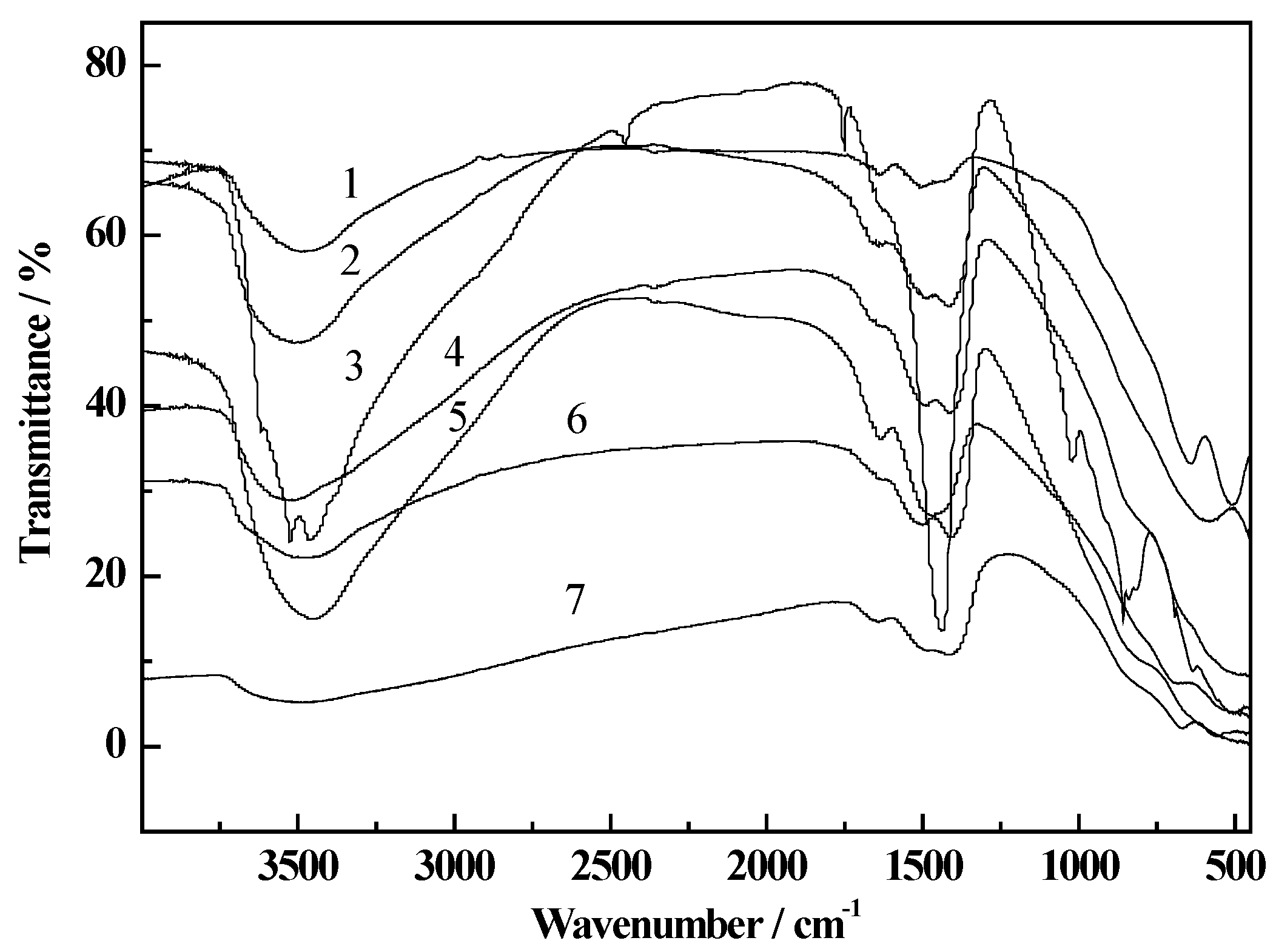
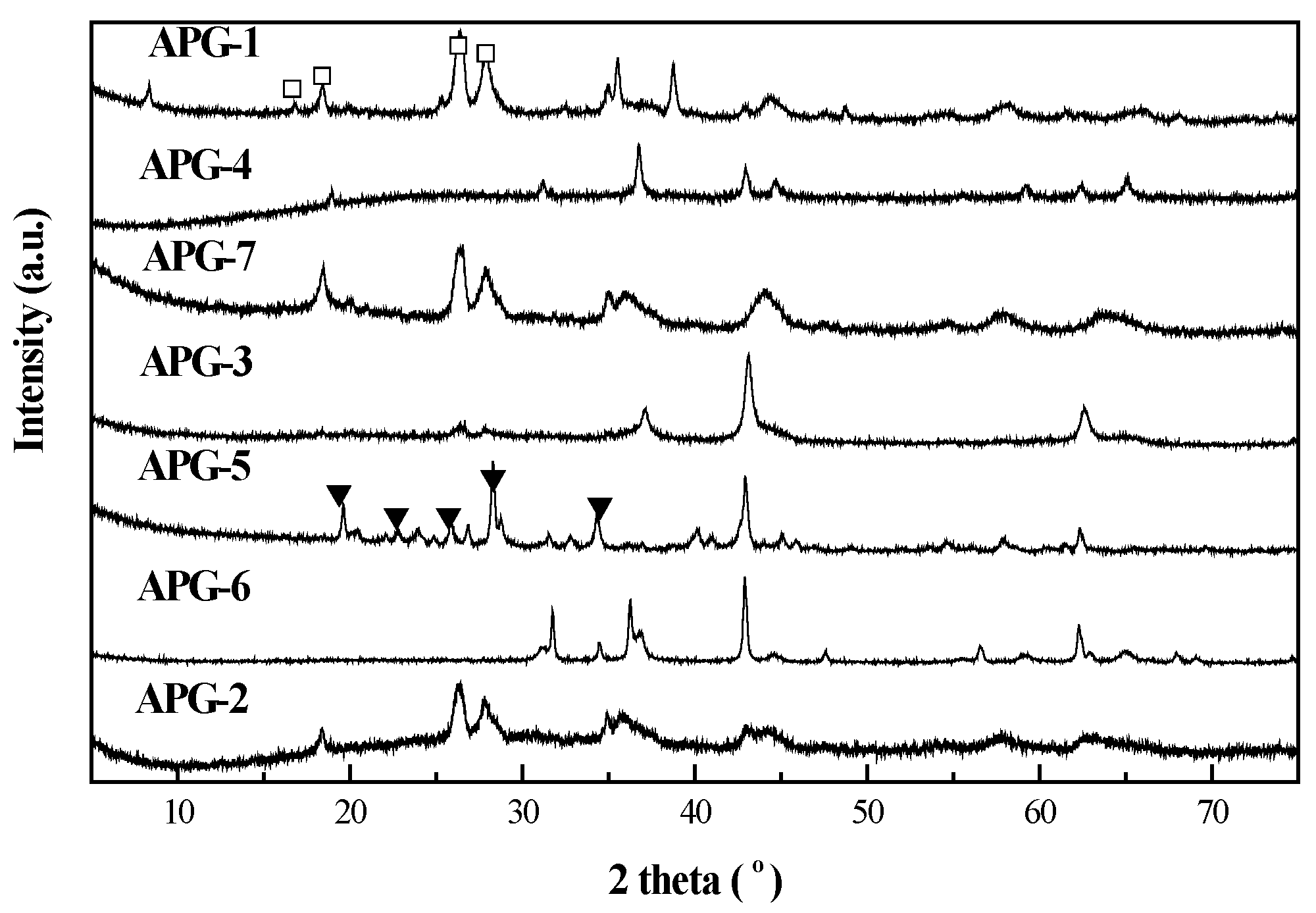
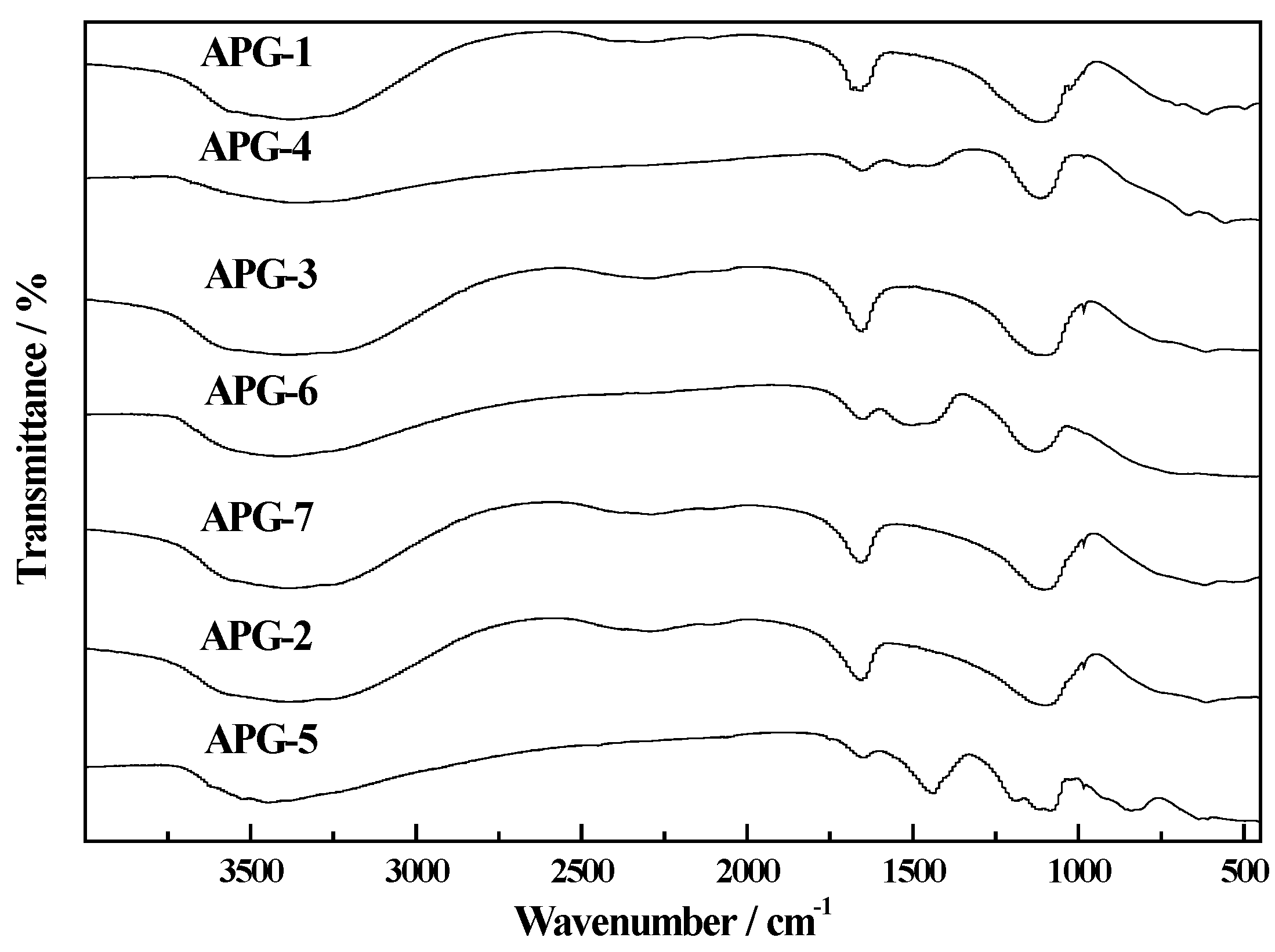
2.2. Thermogravimetry Reduction Analysis
| Sample | APG-1 | APG-2 | APG-3 | APG-4 | APG-5 | APG-6 | APG-7 | |
|---|---|---|---|---|---|---|---|---|
| Item | ||||||||
| Starting gravimetric temperature (°C) | 280 | 275 | 350 | 400 | 490 | 436 | 480 | |
| Final gravimetric temperature (°C) | 700 | 704 | 650 | 570 | 640 | 704 | 690 | |
| First weight loss ratio (%) | 21.26 | 33.99 | 21.26 | 11.44 | 7.42 | 12.77 | 27.76 | |
| Second weight loss ratio (%) | 32.55 | - | - | - | - | 12.99 | - | |
| Total weight loss ratio (%) | 53.81 | 33.99 | 21.26 | 11.44 | 7.42 | 25.76 | 27.76 | |
| Max reduction rate (%) | 0.55 | 0.56 | 0.39 | 0.32 | 0.23 | 0.38 | 1.02 | |
| Temperature in Max reduction rate (°C) | 407 | 416 | 499 | 490 | 579 | 577 | 627 | |
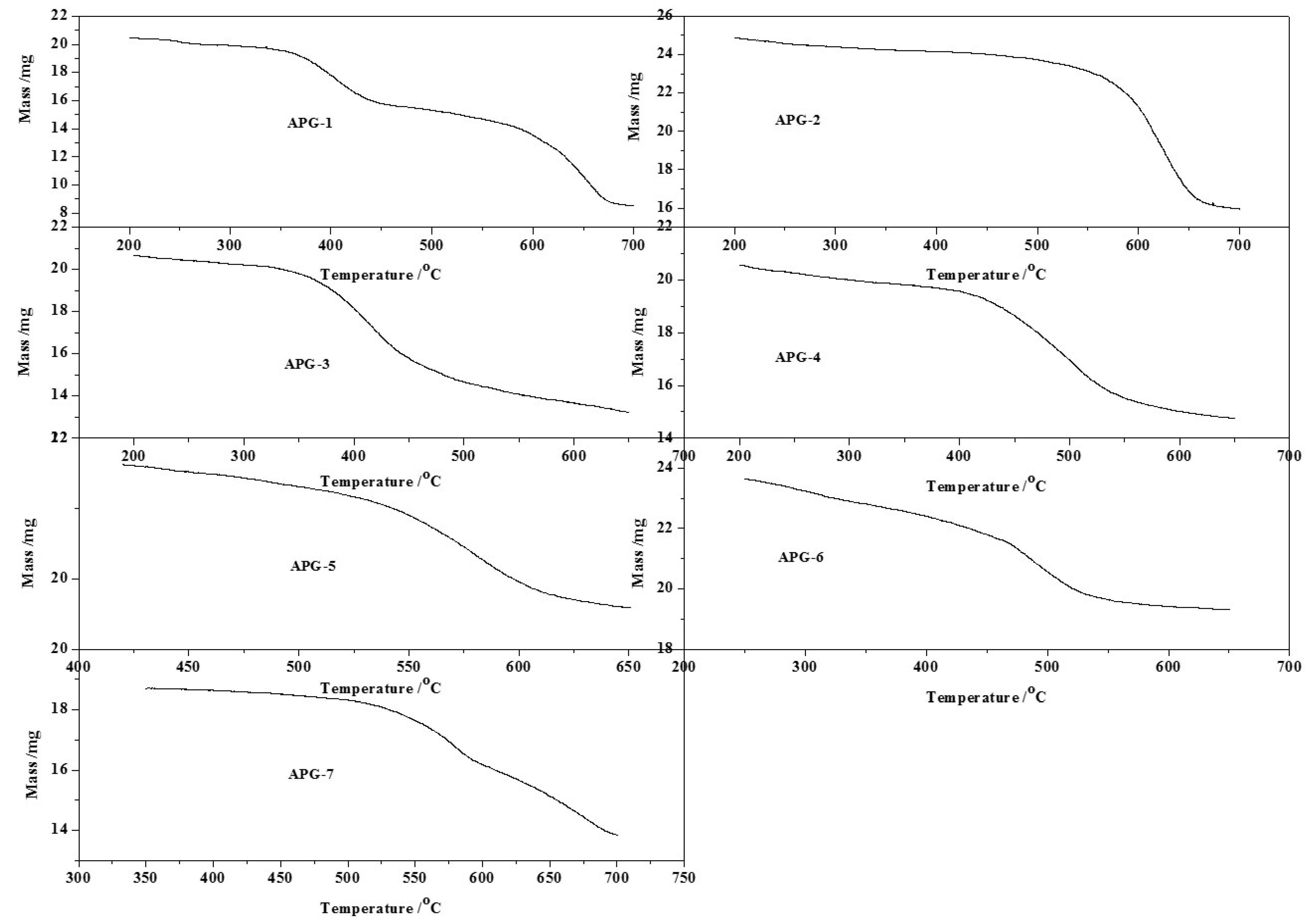
2.3. CO’s Influence on Oxidative Adsorption
| Samples | Gas | Cycles | ||||
|---|---|---|---|---|---|---|
| 0 | 1st, 20 min | 2nd, 20 min | 3rd, 20 min | 4th, 20 min | ||
| APG-1 | O2 | 7.6 | 5.4 | 5.5 | 7 | 7.8 |
| SO2 | 1900 | 6 | 6 | 215 | 941 | |
| CO2 | 10 | 11.8 | 11.7 | 10.6 | 9.9 | |
| CO | 8 | 6.2 | 6.3 | 7.4 | 7.8 | |
| APG-3 | O2 | 7.6 | 4.5 | 7.5 | 8 | - |
| SO2 | 1900 | 5 | 462 | 1082 | - | |
| CO2 | 10 | 12.4 | 10.2 | 9.7 | - | |
| CO | 8 | 5.6 | 7.8 | 7.6 | - | |
| APG-4 | O2 | 7.6 | 4.9 | 8 | - | - |
| SO2 | 1900 | 7 | 1113 | - | - | |
| CO2 | 10 | 12.1 | 9.8 | - | - | |
| CO | 8 | 5.9 | 7.9 | - | - | |
| APG-7 | O2 | 7.6 | 4.8 | 5.2 | 7.7 | 8 |
| SO2 | 1900 | 12 | 7 | 258 | 1011 | |
| CO2 | 10 | 12.2 | 11.9 | 10 | 9.5 | |
| CO | 8 | 5.8 | 6.1 | 7.9 | 8 | |
2.4. Cycles Tests
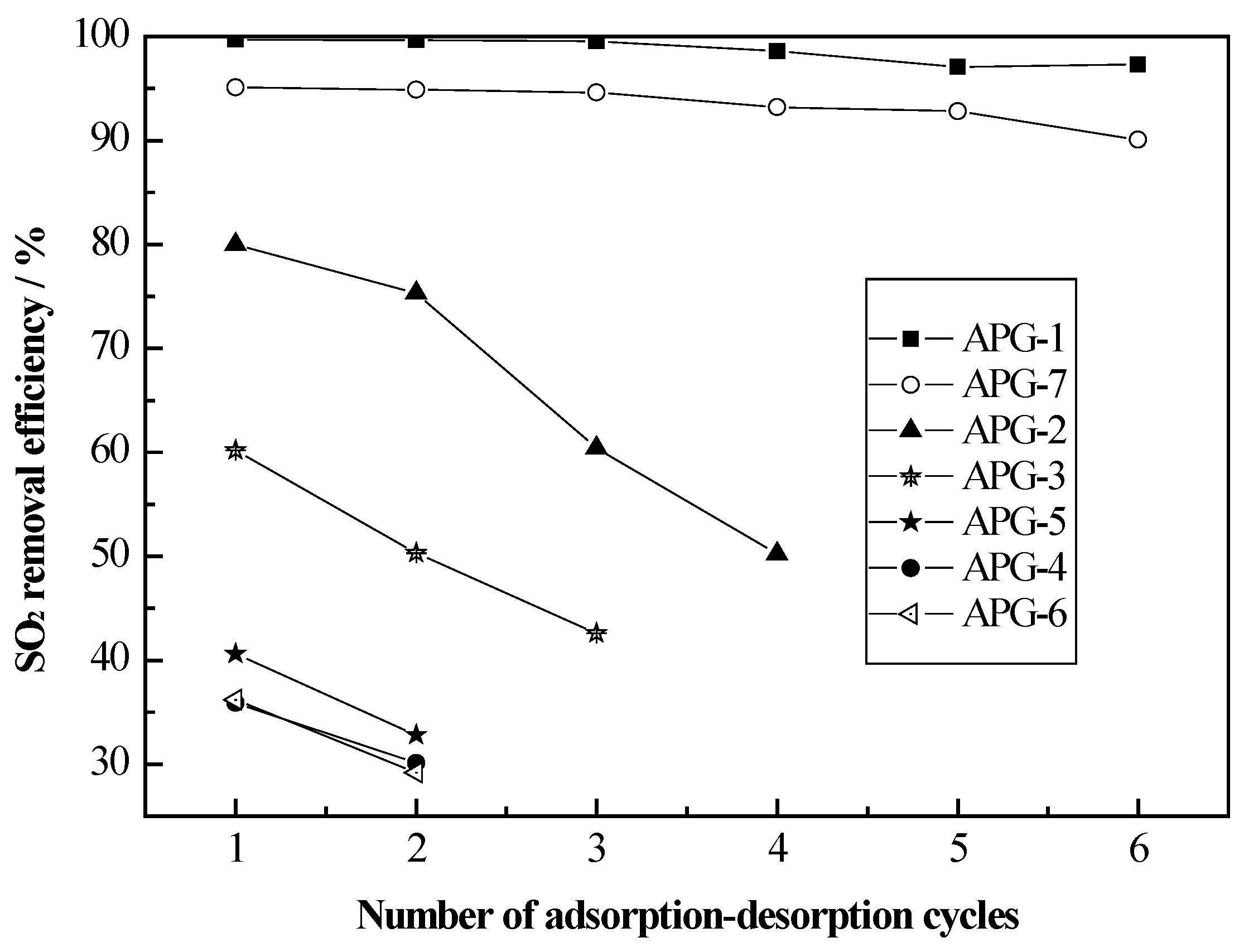
3. Experimental Section
3.1. Preparation of Sulfur Transfer Agents
| Samples | APG-1 | APG-2 | APG-3 | APG-4 | APG-5 | APG-6 | APG-7 |
|---|---|---|---|---|---|---|---|
| oxidative active components | Cu | Fe | Ni | Co | Ba | Zn | Cr |
3.2. Analysis and Characterization
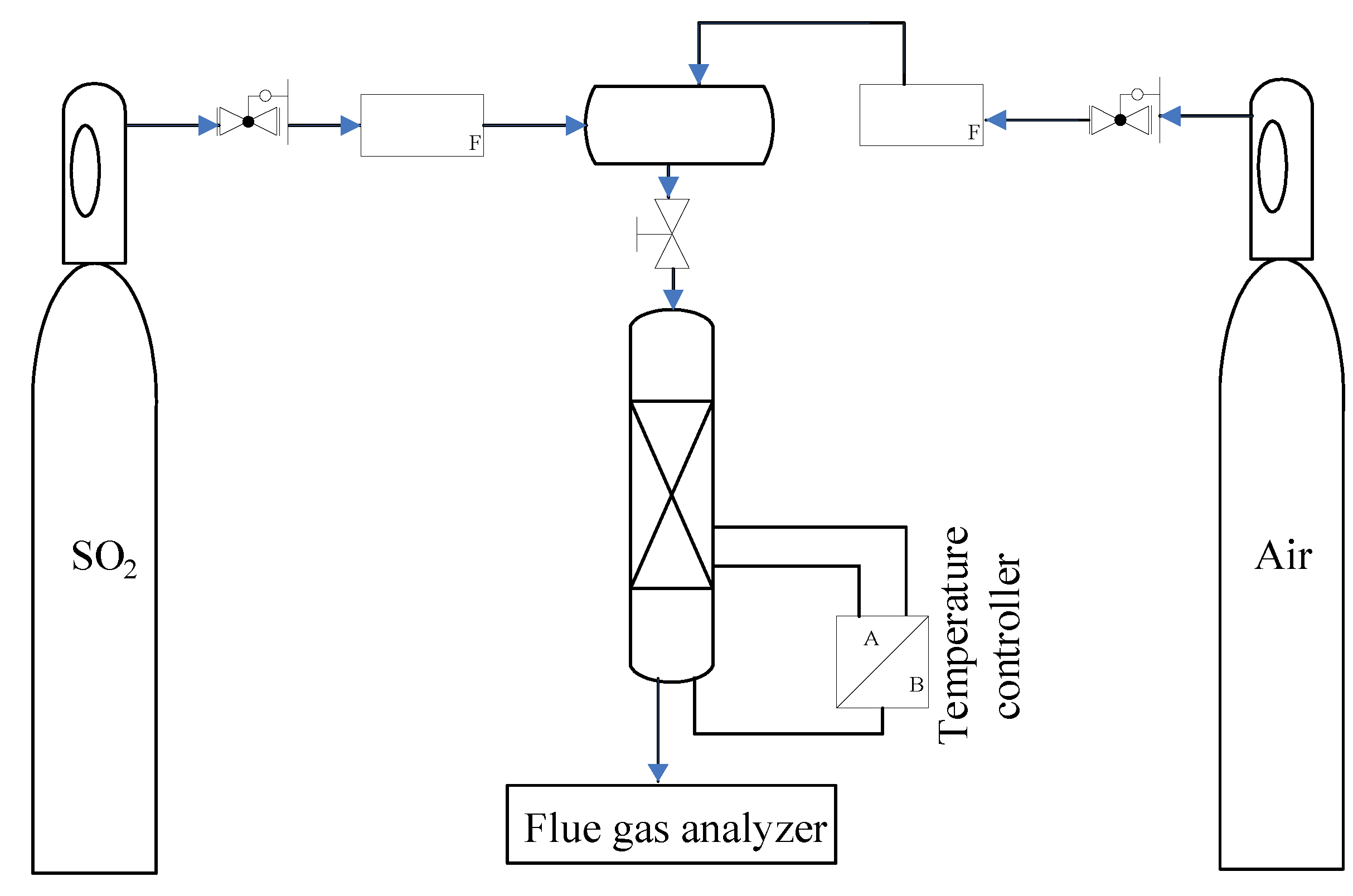
3.3. Data Processing
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lowell, P.S.; Schwitzgebel, K.; Parsons, T.B.; Sladek, K.J. Selection of metal oxides for removing SO2 from flue gas. Ind. Eng. Chem. Process Des. Dev. 1971, 10, 384–390. [Google Scholar] [CrossRef]
- Polato, C.M.S.; Henriques, C.A.; Neto, A.A.; Monteiro, J.L.F.J. Synthesis, characterization and evaluation of CeO2/Mg, Al-mixed oxides as catalysts for SOx removal. Mol. Catal. A 2005, 241, 184–193. [Google Scholar] [CrossRef]
- Cheng, W.; Yu, X.; Wang, W.; Liu, L.; Yang, J.; He, M. Synthesis, characterization and evaluation of Cu/MgAlFe as novel transfer catalyst for SOx removal. Catal. Commun. 2008, 9, 1505–1509. [Google Scholar] [CrossRef]
- Cannilla, C.; Bonura, G.; Rombi, E.; Arena, F.; Frusteri, F. Highly effective MnCeOx catalysts for biodiesel production by transesterification of vegetable oils with methanol. Appl. Catal. A 2010, 382, 158–166. [Google Scholar] [CrossRef]
- Pereira, H.B.; Polato, C.; Monteiro, J.L.F.; Henriques, C.A. Mn/Mg/Al-spinels as catalysts for SOx abatement: Influence of CeO2 incorporation and catalytic stability. Catal. Today 2010, 149, 309–315. [Google Scholar] [CrossRef]
- Jiang, R.; Shan, H.; Li, C.; Yang, C.J. Preparation and characterization of activated Mn/MgAlFe as transfer catalyst for SOx abatement. Nat. Gas Chem. 2011, 20, 191–197. [Google Scholar] [CrossRef]
- Jiang, R.; Shan, H.; Zhang, Q.; Li, C.; Yang, C. The influence of surface area of De-SOx catalyst on its performance. Sep. Purif. Technol. 2012, 95, 144–148. [Google Scholar] [CrossRef]
- Dimitriadis, D.V.; Vasalos, I.A. Evaluation and kinetics of commercially available additives for sulfur oxide (SOx) control in fluid catalytic cracking units. Ind. Eng. Chem. Res. 1992, 31, 2741–2748. [Google Scholar] [CrossRef]
- Kim, G.; Juskelis, M.V. Catalytic reduction of SO3 Stored in SOx transfer catalysts—A temperature-programmed reaction study. Stud. Surf. Sci. Catal. 1996, 101, 137–142. [Google Scholar]
- Centi, G.; Perathoner, S. Behaviour of SOx-traps derived from ternary Cu/Mg/Al hydrotalcite materials. Catal. Today 2007, 127, 219–229. [Google Scholar] [CrossRef]
- Bierla, A.; Fromentin, G.; Minfray, C.; Martin, J.M.; Mogne L., T.; Genet, N. Mechanical and physico-chemical study of sulfur additives effect in milling of high strength steel. Wear 2012, 286, 116–123. [Google Scholar] [CrossRef]
- Blanton, W.A., Jr.; Flanders, R.L. Process for Controlling Sulfur Oxides Using an Alumina-Impregnated Catalyst. U.S. Patent 4332672, 1 June 1982. [Google Scholar]
- Bhattacharyya, A.A.; Woltermann, G.M.; Yoo, J.S.; Karch, J.A.; Cormier, W.E. Catalytic SOx abatement: The role of magnesium aluminate spinel in the removal of SOx from fluid catalytic cracking flue gas. Ind. Eng. Chem. Res. 1988, 27, 1356–1360. [Google Scholar] [CrossRef]
- Polato, C.M.S.; Henriques, C.A.; Rodrigues, A.C.C.; Monteiro, J.L.F. De-SOx additives based on mixed oxides derived from Mg, Al-hydrotalcite-like compounds containing Fe, Cu, Co or Cr. Catal. Today 2008, 133, 534–540. [Google Scholar] [CrossRef]
- Wang, J.; Chen, L.; Li, C.J. Roles of cerium oxide and the reducibility and recoverability of the surface oxygen species in the CeO2/MgAl2O4 catalysts. Mol. Catal. A 1999, 139, 315–323. [Google Scholar] [CrossRef]
- Jiang, R.Y.; Jing, W.J.; Shan, H.H.; Li, C.Y.; Yang, C.H. Studies on regeneration mechanism of sulfur transfer additives of FCC flue gas by H2 reduction. Catal. Commun. 2011, 13, 97–100. [Google Scholar] [CrossRef]
- Wang, J.; Li, C. SO2 adsorption and thermal stability and reducibility of sulfates formed on the magnesium-aluminate spinel sulfur-transfer catalyst. Appl. Surf. Sci. 2000, 161, 406–416. [Google Scholar] [CrossRef]
- Wang, J.; Chen, L.; Limas-Ballesteros, R.; Montoya, A.; Dominguez, J.J. Evaluation of crystalline structure and SO2 storage capacity of a series of composition-sensitive De-SO2 catalysts. Mol. Catal. A 2003, 194, 181–193. [Google Scholar] [CrossRef]
- Wang, Y.; Saad, A.M.; Saur, O.; Lavalley, J.; Morrow, B. FTIR study of adsorption and reaction of SO2 and H2S on Na/SiO2. Appl. Catal. B 1998, 16, 279–290. [Google Scholar] [CrossRef]
- Centi, G.; Passarini, N.; Perathoner, S.; Riva, A. Combined DeSOx/DeNOx reactions on a copper on alumina sorbent-catalyst. 1. Mechanism of sulfur dioxide oxidation-adsorption. Ind. Eng. Chem. Res. 1992, 31, 1947–1955. [Google Scholar] [CrossRef]
- Rodriguez, J.A. Electronic and chemical properties of mixed-metal oxides: Basic principles for the design of DeNOx and DeSOx catalysts. Catal. Today 2003, 85, 177–1992. [Google Scholar] [CrossRef]
- Palomares, A.; López-Nieto, J.; Lázaro, F.; López, A.; Corma, A. Reactivity in the removal of SO2 and NOx on Co/Mg/Al mixed oxides derived from hydrotalcites. Appl. Catal. B 1999, 20, 257–266. [Google Scholar] [CrossRef]
- Polato, C.M.S.; Rodrigues, A.C.C.; Monteiro, J.L.F.; Henrique, C.A. High Surface Area Mn, Mg, Al-Spinels as Catalyst Additives for SOx Abatement in Fluid Catalytic Cracking Units. Ind. Eng. Chem. Res. 2009, 49, 1252–1258. [Google Scholar] [CrossRef]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiang, R.; Yu, S.; Zhou, Y.; Zhu, T. Study on the Relation between the Mn/Al Mixed Oxides Composition and Performance of FCC Sulfur Transfer Agent. Catalysts 2016, 6, 20. https://doi.org/10.3390/catal6020020
Jiang R, Yu S, Zhou Y, Zhu T. Study on the Relation between the Mn/Al Mixed Oxides Composition and Performance of FCC Sulfur Transfer Agent. Catalysts. 2016; 6(2):20. https://doi.org/10.3390/catal6020020
Chicago/Turabian StyleJiang, Ruiyu, Shuanglin Yu, Yudong Zhou, and Tao Zhu. 2016. "Study on the Relation between the Mn/Al Mixed Oxides Composition and Performance of FCC Sulfur Transfer Agent" Catalysts 6, no. 2: 20. https://doi.org/10.3390/catal6020020
APA StyleJiang, R., Yu, S., Zhou, Y., & Zhu, T. (2016). Study on the Relation between the Mn/Al Mixed Oxides Composition and Performance of FCC Sulfur Transfer Agent. Catalysts, 6(2), 20. https://doi.org/10.3390/catal6020020





