Abstract
The bis(pyrazole)zinc(II) benzoate complexes bis(3,5-diphenylpyrazole)zinc(II) benzoate (1), bis(3,5-diphenylpyrazole)zinc(II) 3,5-dinitrobenzoate (2), bis(3,5-diphenylpyrazole)zinc(II) 4-hydroxybenzoate (3), and bis(3,5-di-tert-butylpyrazole)zinc(II) 2-chlorobenzoate (4) were synthesized from the reaction of 3,5-diphenylpyrazole (L1) or 3,5-di-tert-butylpyrazole (L2), zinc(II) acetate and the appropriate benzene carboxylic acid. The molecular structure of complex 2 confirmed that these zinc(II) benzoate complexes adopt a 4-coordinate tetrahedral geometry. All four complexes were screened as catalysts for the copolymerization of carbon dioxide (CO2) and cyclohexene oxide (CHO) and were found to be active for the formation of poly(cyclohexene carbonate) (PCHC) at CO2 pressures as low as 1.0 MPa under solvent-free conditions and without the use of a co-catalyst. At some reaction condition, most of the catalysts produced PCHC with high carbonate content of up to 98% and a good amount of cyclic cyclohexene carbonate (CCHC). The copolymers produced have low to moderate molecular weights (5200–12300 g/mol) and with polydispersity indices that vary from 1.19 to 2.50. Matrix Assisted Laser Desorption/Ionization-Time of Flight Mass Spectra (MALDI-TOF MS) of these copolymers showed they have benzoate and hydroxyl end groups.
1. Introduction
The fact that CO2 is readily available, inexpensive, and non-toxic renders it advantageous for utilization as a C1 synthetic feedstock, and thus its conversion to useful products has become highly desirable. The reaction of CO2 that has been studied extensively in the past five decades is the metal catalyzed alternating copolymerization of CO2 with epoxides to polycarbonates, some of which are biodegradable and are thus of great importance in the plastic industry. The alicyclic polycarbonate poly(cyclohexene carbonate) PCHC, resulting from the copolymerization of CO2 and CHO, possesses physical attributes very similar to polystyrene. For example, the glass transition temperature of PCHC (Tg = 115 °C) [1] is very close to that of polystyrene (Tg = 100 °C), hence the polymer has good dimensional stability at temperatures above 100 °C. Furthermore, the Tg of PCHC is slightly lower than that of the classical bisphenol-A polycarbonate (145 °C) [2]. This allows for the application of PCHC in a number of industrial processes [3,4]. In addition, PCHC displays excellent thermal performance with a one-step decomposition at ca. 310 °C [5], far higher than those of traditional CO2/epoxide copolymers like poly(propylene carbonate) (PPC); again allowing the use of PCHC as an environmentally friendly engineering thermoplastic [3]. As a new biodegradable polycarbonate suitable for high-temperature applications, PCHC may provide a solution to the poor dimensional stability of PPC.
The copolymerization of CO2 and epoxides is catalyzed by a number of metal complexes. Two reviews by Darensbourg [6] and Coates [1,7] and reports by Nozaki [8,9,10] and Lu [11,12] indicate that the best catalysts for this reaction are Schiff-base supported Cr, Co and Zn complexes. For homogeneous zinc-catalyzed copolymerization of CHO and CO2, three types of very active catalysts have been reported. These are: zinc carboxylates with fluorinated tails [13,14], bisphenoxides of zinc [15], and three-coordinate zinc complexes containing β-diiminate ligands [16]. Zn-based catalyst systems have, thus far, remained one of the most active catalysts for the CO2 and epoxide copolymerization reaction [17,18,19]. In general, zinc(II) complexes containing a Zn–O bond and supported by N-donor bidentate ligand systems have been reported to be highly active for the copolymerization reaction of CO2 and epoxides [1]. However, a significant drawback associated with using CO2 as a reagent in synthesis is the potential danger associated with operating at high temperatures and pressures. To overcome this drawback, it has been necessary to develop catalyst systems that can operate efficiently at low CO2 pressures. We herein report the syntheses of very simple bis(pyrazole)zinc(II) benzoates that catalyze the copolymerization of CO2 and CHO to both poly(cyclohexene carbonate) and cyclic cyclohexene carbonate), with some of the polymers having up to 98% carbonate content and at low CO2 pressures; in some cases as low as 1.0 MPa. Although the catalytic activity is not as high as have been reported for some zinc [1,7] and cobalt [8] catalysts, the current zinc catalyst system is very simple to prepare and thus provide readily accessible non-toxic catalysts for CO2/epoxide copolymerization.
2. Results and Discussion
2.1. Synthesis and Characterization of Zinc(II) Complexes
The pyrazole ligands, 3,5-diphenylpyrazole (L1) or 3,5-di-tert-butylpyrazole (L2), were reacted with zinc(II) acetate in a 2:1 ratio of ligand to metal precursor to give the zinc(II) benzoate complexes shown in Scheme 1. The products were characterized by a combination of 1H NMR spectroscopy, elemental analysis and, in the case of 2, by single crystal X-ray crystallography. The NMR data of these complexes have typical signals of the pyrazoles and the benzoates.
The crystal structure obtained for complex 2 (Figure 1) [20] shows that it is a monometallic zinc(II) tetrahedrally coordinated to two L1 ligands through the nitrogen atoms and to one oxygen atom of each of the two benzoate ligands. The crystallographic parameters for this complex are given in Table S1. Complex 2 was found to have Zn–O and Zn–N bond lengths of 1.94 and 2.00 Å, respectively. The angles around the zinc center varies from 100.70° for O(1)–Zn(1)–O(3) to 116.78° for O(3)–Zn(1)–N(3). These angles show the distorted tetrahedral geometry adopted by the complex (Figure 1). The structure of 2 can be compared to a similar complex of formula [Zn(L1)2(Bz)2] (where Bz = benzoate group) synthesized by Baruah [21], which has an average Zn–O bond length of 1.93 Å and is very close to the average Zn–O bond length in 2 (1.94 Å). The angles around the zinc center in [Zn(L1)2(Bz)2] are between 102.45° and 114.59° and for 2, these angles vary from 110.70° to 116.78°.
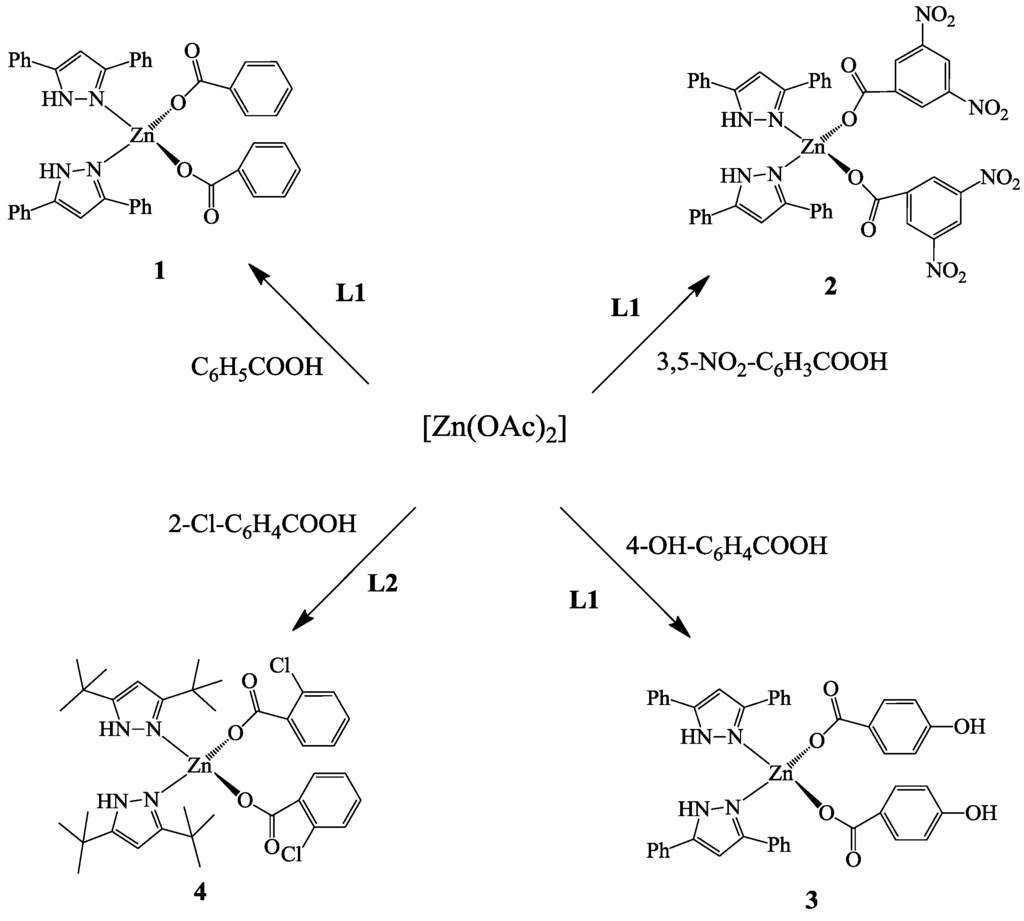
Scheme 1.
Synthesis of bis(pyrazole)zinc(II) benzoates pre-catalysts.
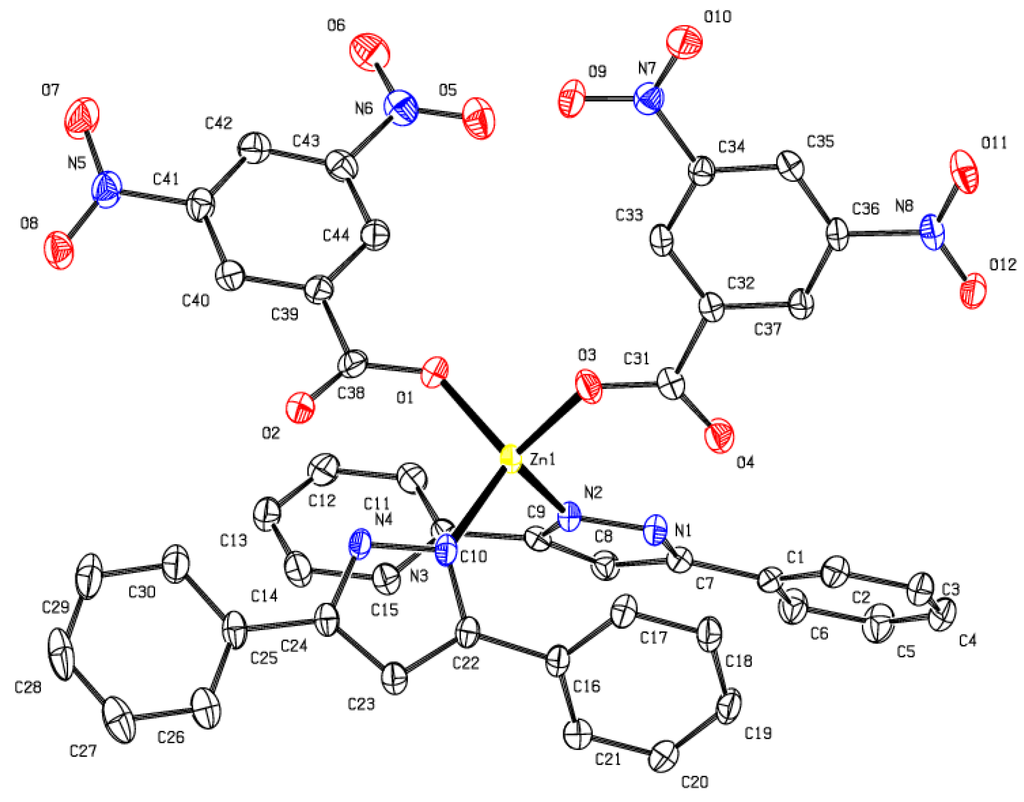
Figure 1.
ORTEP diagram of complex 2. All H atoms are omitted for clarity. Selected bond lengths (Å) and bond angles (°): O(1)–Zn(1), 1.9327(15); O(3)–Zn(1), 1.9509(15); N(3)–Zn(1), 1.9968(17); N(2)–Zn(1), 2.0016(18); O(1)–Zn(1)–O(3), 100.71(7); O(1)–Zn(1)–N(3), 111.63(6); O(3)–Zn(1)–N(3), 116.78(7); O(1)–Zn(1)–N(2), 107.04(7); O(3)–Zn(1)–N(2), 113.15(6); N(3)–Zn(1)–N(2), 107.11(7).
2.2. Ring-Opening Copolymerization of CO2 and CHO with Complexes 1–4
The four bis(pyrazole)zinc(II) benzoate complexes (1–4) were screened as catalysts for the copolymerization of CO2 and CHO. All four complexes (1–4) were found to be active for the copolymerization reaction at CO2 pressure of 1.0 MPa. A variation of monomer:catalyst ([M]:[C]) ratio showed that complexes 1 and 2 were the most active at [M]:[C] ratio of 500:1 (Table 1). When the amount of catalyst was increased (i.e., 250:1), there was negligible increase in the catalyst activities of 1 and 2. Complex 4 showed no catalytic activity when [M]:[C] ratios of 1000:1 and 500:1 were used. This observation is in line with suggestion by Klaus [22] that low concentration catalyst lowers activity even in bimetallic catalyst systems. Catalytic activity was only observed when higher catalyst loadings of [M]:[C] ratio of 250:1 were used. The requirement for high catalyst loading of these complexes shows that for copolymerization to occur, the active metal centers (Zn–O bonds) should be present in excess compared to the CHO comonomer. The decreased activity of these complexes can also be attributed to the bulky tertiary-butyl groups in the 3,5-position of the pyrazole moiety in 4; and for 3, the presence of electron-donating hydroxyl group on the benzoate group which makes its metal center less electrophilic and therefore less active towards the ring-opening copolymerization of CHO with CO2. For these reasons, further investigations involving catalysis with these complexes were carried out using [M]:[C] ratio of 500:1 for 1 and 2 and 250:1 for 3 and 4.

Table 1.
Effect of catalyst loading on catalytic activity a.
| Entry | Complex | [M]/[C] | Poly(cyclohexene carbonate) (PCHC) (%) b | Cyclic cyclohexene carbonate (CCHC) (%) b | Selectivity | TOF (h−1) c |
|---|---|---|---|---|---|---|
| 1 | 1 | 1000:1 | 4.4 | 2.1 | 68 | 4.1 |
| 2 | 1 | 500:1 | 18 | 5.6 | 76 | 7.7 |
| 3 | 1 | 250:1 | 19.3 | 6.1 | 76 | 7.7 |
| 4 | 2 | 1000:1 | 0 | 0 | - | - |
| 5 | 2 | 500:1 | 26.5 | 3.9 | 87 | 9.9 |
| 6 | 2 | 250:1 | 28 | 5.7 | 83 | 10 |
| 7 | 3 | 1000:1 | 0 | 0 | - | - |
| 8 | 3 | 500:1 | 8.2 | 2.6 | 76 | 3.5 |
| 9 | 3 | 250:1 | 11.2 | 4.5 | 71 | 2.5 |
| 10 | 4 | 1000:1 | 0 | 0 | - | - |
| 11 | 4 | 500:1 | 0 | 0 | - | - |
| 12 | 4 | 250:1 | 50.1 | 33.2 | 64 | 15 |
a Reactions were carried out in 1.0 mL CHO using 1.0 MPa CO2 pressure for 16 h at 120 °C; b % conversion, determined from 1H NMR and IR spectroscopy; c moles of CHO consumed per mole of catalyst per hour.
What is significant about using 1–4 as catalysts for copolymerization of CO2 and CHO is that they did not require the use of solvents or co-catalyst to catalyze the reaction but produced PCHC with turnover frequencies (TOFs) that are comparable to structurally-related monomeric zinc(II) carboxylate complexes reported in the literature [1,23]. Nevertheless, Coates’ zinc(II) carboxylate complexes still remain one of the most active catalysts for CO2 and epoxide copolymerization reaction, producing PCHC with carbonate content greater than 93% with narrow molecular weight distribution [24,25,26]. In addition to producing PCHC, the copolymerization reactions catalyzed by 1–4 also led to the production of cyclic cyclohexene carbonate (CCHC) as a byproduct. This is generally the result of back-biting of the growing copolymer chain [27].
The PCHC produced were of moderate to high number average molecular weight (Mn), with Mn in the range 5200–12300 g·mol−1. Molecular weight distribution in the copolymers, a measure of polydispersity index, was found to be moderate to wide in the range 1.19–2.50. The lower than expected Mn values for some of the PCHC produced in this work shows that the copolymerization process is not living but rather immortal. The immortal nature of the copolymerization could be due to the presence of chain transfer agents (CTAs). One possible CTA could be the presence of trace amounts of water from insufficiently dry CO2, glassware and possibly other equipment.
Variation of CO2 pressure while temperature (120 °C) and amount of CHO (9.88 mmol) were held constant resulted in significant increase in conversion of CHO to both PCHC and CCHC as CO2 pressure increased. Also significant was the favored production of PCHC over CCHC (Table 2). Although the increased formation of PCHC over CCHC with increasing CO2 pressure is consistent with literature reports [28], it is only complex 4 that had large amounts of CCHC formed. Complexes 1–3 had selectivity for the formation of PCHC ranging from 71%–89% as pressure increased; but complex 2 was the most active for PCHC formation with TOF of up to 32 h−1 and 89% selectivity for PCHC over CCHC at CO2 pressure of 4.0 MPa. The higher catalytic activity of 2 could be attributed to the electron-withdrawing nature of the nitro groups in the 3,5-position of the phenyl ring that facilitates the initial binding of the epoxide; thus making the zinc(II) center in 2 highly electrophilic and more active in binding the epoxide before its ring-opening. This observation is in accordance with the high activities reported in literature for zinc(II) complexes bearing electron-withdrawing groups [16,23,29,30]. In contrast, the complex bearing the OH group, 3, gave some of the lowest catalytic activities possibly because of a poorer electrophilic zinc center in this catalyst.
The kinetics of the copolymer catalyzed by 1 was studied using the in situ equipment ReactIR 45 that monitors stretching bands of IR active functional groups in the polymer. For PCHC and CCHC their carbonyl group stretching frequencies of 1750 cm−1 and 1825 cm−1, respectively, were followed as the reaction progressed (Figure S1). Figure S1 shows the asymmetric ν(C=O) stretch of PCHC as the stronger absorption peak at 1750 cm−1 and that of CCHC as the weaker absorption peak at 1825 cm−1 similar to observations made by Darensbourg and co-workers [31]. It is also clear from the figure that the initiation step in the polymerization, which involves the ring-opening of the three-membered cyclic ether in CHO, is rather slow with induction time of ~6 h. Because carboxylates are known to be weak nucleophiles, catalysts containing carboxylates are usually pre-activated with Lewis acid, which may be a cocatalyst [22]. Thus, in the absence of a cocatalyst, it is not surprising that reaction with complex 1 as catalysts had such long induction period. However, once the polymerization started the formation of PCHC was exponential with time, while the formation of CCHC increased linearly with time (Figure 2). The slower formation of CCHC than PCHC is attributed to the high activation energy barrier for CCHC formation, which is reported to be >80 kJ·mol−1 higher than that of PCHC production [29]. The 1H NMR data also indicate that the major linkage in the polymers were carbonate in most of the polymers (4.63 ppm); although in some of the polymers ether were substantial ether linkages (3.45 ppm).
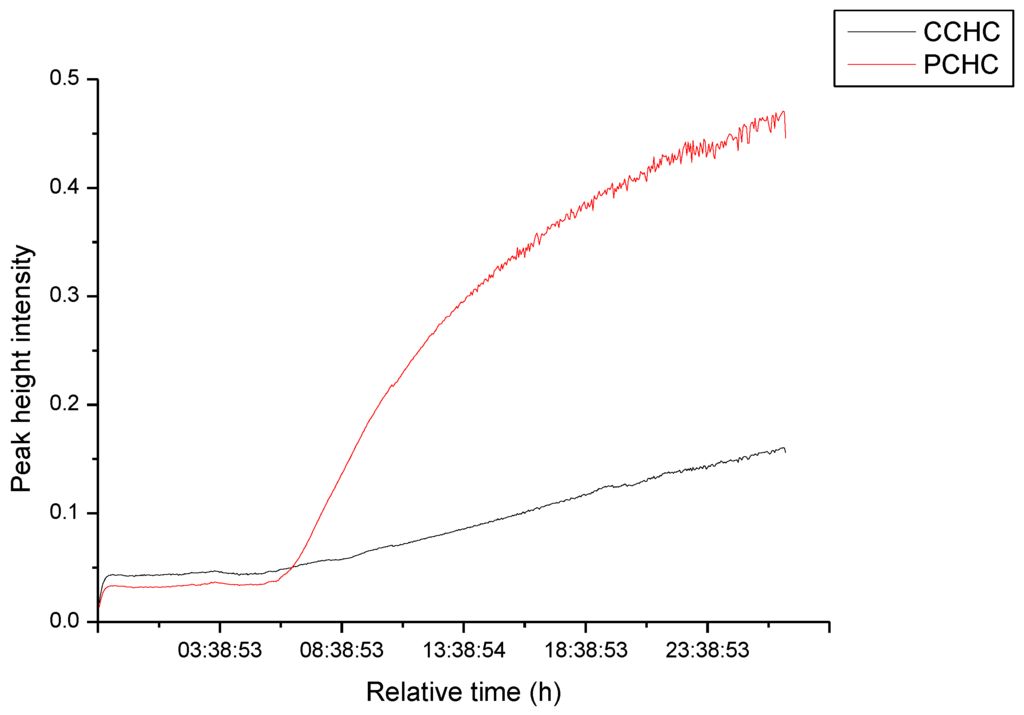
Figure 2.
Representative plot of poly(cyclohexene carbonate) (PCHC) and cyclic cyclohexene carbonate (CCHC) formation as a function of time using catalyst 1.

Table 2.
Effect of CO2 pressure on CHO and CO2 copolymerization a.
| Entry | Catalyst | CO2 Pressure (MPa) | Poly(cyclohexene carbonate) (PCHC) (%) b | Cyclic cyclohexene carbonate (CCHC) (%) b | Selectivity for PCHC (%) | Carbonate content in PCHC (%) c | TOF (h−1) d | Mn (g/mol) e | PDI (Mw/Mn) | Tg (°C) f |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 1 | 1 | 18.0 | 5.6 | 76 | 40 | 7.7 | 1600 | 1.64 | - |
| 2 | 2 | 1 | 26.5 | 3.9 | 87 | 15 | 9.9 | 5400 | 1.64 | - |
| 3 | 3 | 1 | 11.2 | 4.5 | 71 | 66 | 2.5 | - | - | - |
| 4 | 4 | 1 | 50.1 | 33.2 | 60 | 85 | 15 | 6300 | 1.61 | 109.4 |
| 5 | 1 | 2 | 25.0 | 5.8 | 81 | 81 | 10.1 | 9300 | 1.34 | - |
| 6 | 2 | 2 | 33.0 | 3.9 | 89 | 44 | 18 | 6500 | 2.05 | 116.7 |
| 7 | 3 | 2 | 11.6 | 4.3 | 73 | 76 | 2.5 | - | - | - |
| 8 | 4 | 2 | 55.6 | 41.5 | 57 | 89 | 17 | 6900 | 1.57 | 105.4 |
| 9 | 1 | 3 | 43.0 | 3.9 | 92 | 88 | 15.2 | 10,500 | 1.78 | 104.4 |
| 10 | 2 | 3 | 48.0 | 6.2 | 83 | 60 | 31 | 7700 | 2.19 | 114.4 |
| 11 | 3 | 3 | 31.0 | 7.0 | 82 | 89 | 6 | 6300 | 2.25 | - |
| 12 | 4 | 3 | 57.9 | 39.1 | 60 | 92 | 17 | 6300 | 1.44 | 107.8 |
| 13 | 1 | 4 | 58.0 | 10.9 | 84 | 98 | 22.3 | 12,300 | 1.19 | - |
| 14 | 2 | 4 | 55.0 | 6.5 | 89 | 77 | 32 | 5200 | 2.50 | 103.5 |
| 15 | 3 | 4 | 53.8 | 9.7 | 85 | 89 | 10 | - | - | - |
| 16 | 4 | 4 | 67.7 | 29.9 | 69 | 95 | 18 | 7600 | 1.52 | 112.9 |
a Reactions were carried out in 1.0 mL CHO at 120 °C for 16 h using [M]:[C] ratio of 500:1 for 1–2 and 250:1 for 3–4; b % conversion, determined from 1H NMR and IR spectroscopy; c determined from 1H NMR spectra by comparing integration of proton peaks at 4.63 ppm corresponding to carbonate linkage and 3.45 ppm for ether linkage; d moles of CHO consumed per mole of catalyst per hour; e determined by GPC; f from DSC measurements.
In several of the reactions, the PCHC produced had relatively high carbonate content (some as high as 98%) and generally found to be dependent on CO2 pressure; although there were a significant number of catalytic runs that produced polymers with significant ether content (Table 2, runs 1–3). A typical polymer showing high carbonate content is depicted by its 1H NMR spectrum (Figure S2). Higher CO2 pressures favored the formation of almost completely alternating copolymers with high carbonate content (Table 2, runs 10–16). In general, catalysts with less electrophilic metal centers gave PCHC with higher carbonate content. For example, copolymers with complex 4 possess higher carbonate content than those with complex 2 because the two chloro-substituted benzoates in 4 are less electron-withdrawing than the two nitro groups in 2. The relatively high carbonate content in PCHC obtained for catalysts with less electrophilic metal centers (1, 3 and 4) suggest that the insertion of CO2 into the growing chain is greater or equal to the rate of CHO ring-opening; hence the copolymerization is almost completely alternating. However, in the more electrophilic catalyst, 2, the metal center is more active in binding and ring-opening CHO for copolymerization. For this reason, catalyst 2 opens the epoxide ring in CHO more rapidly and this results in rapid insertion of CHO fragments into the growing chain and slow incorporation of CO2; hence the resulting copolymers have low carbonate content. The carbonate content of the copolymers was found to affect their glass transition temperature (Tg) (Table 2). Glass transition temperature in polycarbonates is affected by ether linkages in the polymer, so for our polymers with high carbonate content, it is not surprising that their Tg (103–112 °C) were lower than the average Tg in the literature (115 °C) [1].
The 13C{1H} NMR analyses (Figure S3) of all the PCHC produced indicate that the polymers are atactic and from MALDI-TOF mass spectrometry data, the polymer end-groups were either a benzoate ligand or a hydroxycyclohexyloxy group. Figure 3b is typical MALDI-TOF MS spectrum of our PCHCs produced with regular repeating mass units of 142.2. Figure 3a shows the benzoate group in our pre-catalyst 4 as the initiator of the copolymerization reaction. This particular polymer showed a significant ether linkages, depicted by peaks that conform to loss of CO2 (e.g., peak 6256 from 6299 minus 44). So from the MALDI-TOF mass spectral data we were able to establish that some of the polymers produced have both carbonate and ether linkages; although some polymers have very high carbonate content. Figure 3b shows that water was as a competing initiator in the copolymerization reaction. The mass values of the stronger signals in Figure 3a,b match (211.0 (benzoate) + 142.2n (repeating units) + 115.1 (hydroxyclohexyloxy group)) and (17.0 (hydroxyl) + 142.2n (repeating units) + 115.1 (hydroxyclohexyloxy group)), respectively. The mass spectrometry data therefore suggests that the copolymerization is terminated by protolysis to give either copolymer I or II. Copolymer I shows that the possible initiating mechanism for the copolymerization is the ring-opening of CHO by nucleophilic attack.
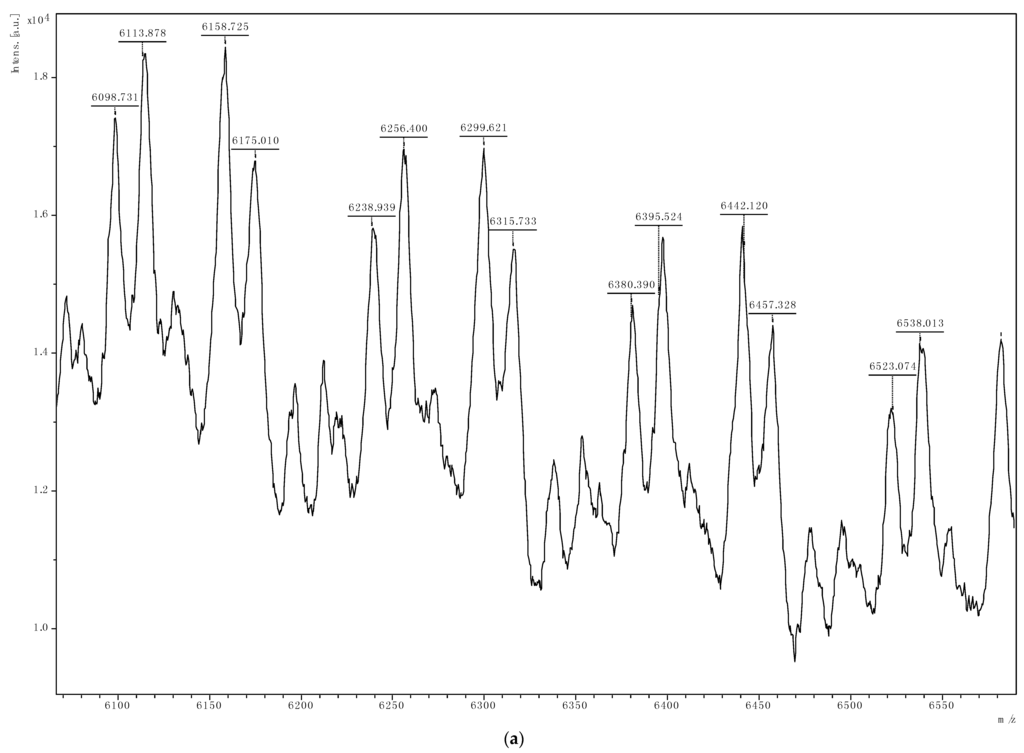
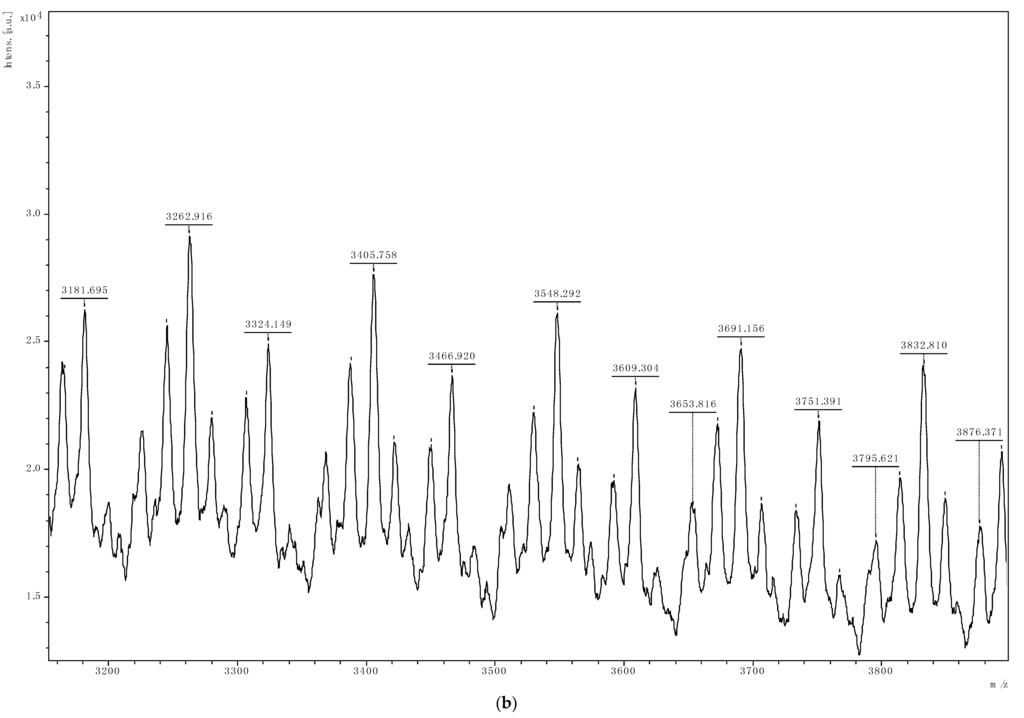
Figure 3.
Figure 3a,b are assigned to the mass spectra of the copolymers using complexes 2 and 1 respectively.
3. Experimental Section
3.1. Materials
Syntheses of all complexes were carried out under a dry nitrogen atmosphere using standard Schlenk techniques. Zinc(II) acetate ([Zn(OAc)2]), benzoic acid (C6H5COOH), 3,5-dinitrobenzoic acid (3,5-NO2-C6H3COOH), 4-hydroxybenzoic acid (4-OH-C6H4COOH), 2-chlorobenzoic acid (2-Cl-C6H4COOH) were used as received from Sigma Aldrich (Johannesburg, South Africa). All solvents were of analytical grade and were purchased from Sigma-Aldrich (Johannesburg, South Africa) and stored under inert conditions in a solvent drying system. 3,5-Diphenylpyrazole (L1) [31] and 3,5-di-tert-butylpyrazole (L2) [32] were synthesized according to literature procedures. Cyclohexene oxide was vacuum distilled over calcium hydride (Sigma Aldrich) before use. Carbon dioxide and phenanthrene were used as received.
3.2. Instrumentation
1H and 13C{1H} NMR spectra were recorded in chloroform-d (CDCl3) and dimethylsulphoxide-d6 (DMSO) on a Bruker Ultrashield 400 instrument (Bruker, Karlsruhe, Germany) (400 MHz for 1H NMR and 100 MHz for 13C{1H} NMR) at room temperature. 1H and 13C{1H} NMR chemical shifts were referenced to the residual signals of the protons or carbons of the NMR solvents and are quoted in δ: CDCl3 at 7.26 and 77.16 ppm for 1H and 13C{1H} NMR spectra respectively; DMSO at 2.50 and 39.52 ppm for 1H and 13C{1H} NMR spectra respectively. Coupling constants are measured in Hertz (Hz). Infrared spectra were recorded on a Bruker FR-IR Tensor27 spectrometer fitted with an ATP-IR probe. Elemental analyses were performed on a Vario Elementar IIImicrocube CHNS analyzer at Rhodes University, South Africa. The crystal evaluation and data collection was performed on a Bruker Quazar SMART APEXII diffractometer using Mo radiation source.
For characterization of the copolymers produced, a JEOL JNM-ECP500 NMR spectrometer (1H 500 MHz, 13C{1H} 126 MHz) was used. Infrared spectra were recorded on a Shimadzu FTIR-8400 spectrometer. Gel permeation chromatography (GPC) analyses of the copolymers were carried out with two columns (Shodex KF-804L) using tetrahydrofuran as eluent at 40 °C at a flow rate of 1 mL/min. The molecular weights were calibrated against polystyrene standards. Differential scanning calorimetry (DSC) measurements were performed on a Seiko DSC 7020 analyzer at a heating and cooling rate of 10 °C/min; the reported glass transition temperature (Tg) values were determined from the second heating scan.
bis-(3,5-Diphenyl-pyrazolyl)zinc(II) benzoate (1)
A solution of [Zn(OAc)2] (0.18 g, 1.00 mmol) and C6H5COOH (0.24 g, 2.00 mmol) in 50 mL MeOH was stirred under reflux for 5 h. L1 (0.44 g, 2.00 mmol), dissolved in 2 mL MeOH, was added dropwise. The reaction was allowed to reflux for a further 16 h and the product isolated as a white solid after filtration and evaporation of filtrate. Yield: 0.54 g (72%). 1H NMR (DMSO): δ 7.18 (s, 2H, CH-Pz); 7.32 (t, 5JHH = 14.8 Hz, 6H, 4-Ph-Pz, 4-Bz); 7.46 (m, 10H, 3,5-Ph-Pz, 3,5-Bz); 7.83 (d, 3JHH = 7.6 Hz, 8H, 2,6-Ph-Pz); 7.95 (d, 3JHH = 7.6 Hz, 4H, 2,6-Bz). 13C{1H} NMR (DMSO): δ 99.7; 125.1; 127.8; 128.0; 128.9; 129.5; 131.3; 134.1; 171.0. Anal. Calcd for C44H34N4O4Zn.0.5CH3OH: C, 69.94; H, 4.75; N, 7.33%. Found: C, 69.62; H, 5.26; N, 7.22%.
bis-(3,5-Diphenyl-pyrazolyl)zinc(II) 3,5-dinitrobenzoate (2)
Reaction of [Zn(OAc)2] (0.18 g, 1.00 mmol), 3,5-(NO2)2-C6H3COOH (0.43 g, 2.00 mmol) and L1 (0.44 g, 2.00 mmol) in MeOH gave 2 as a white solid, obtained by evaporation of the solvent in vacuo. Single crystals suitable for X-ray were obtained from a solution of 2 in DMSO upon slow evaporation of the solvent. Yield: 0.61 g (66%). 1H NMR (CDCl3): δ 6.59 (s, 2H, CH-Pz); 7.10 (m, 8H, 3,5-Ph-Pz); 7.55 (m, 8H, 2,6-Ph-Pz); 7.72 (m, 4H, 4-Ph-Pz); 9.12 (s, 2H, 5-Bz); 9.24 (s, 4H, 2,6-Bz). 13C{1H} NMR (CDCl3): δ 99.6; 120.5; 125.1; 128.9; 138.8; 148.0; 166.6. Anal. Calcd for C44H30N8O12Zn: C, 56.94; H, 3.26; N, 12.07%. Found: C, 57.26; H, 3.29; N, 11.93%.
bis-(3,5-Diphenyl-pyrazolyl)zinc(II) 4-hydroxybenzoate (3)
Complex 3 was synthesized using [Zn(OAc)2] (0.18 g, 1.00 mmol), 4-OH-C6H4COOH (0.28 g, 2.00 mmol) and L1 (0.44 g, 2.00 mmol) and isolated as a white solid by filtration of the reaction mixture after evaporation of the solvent from the filtrate. Yield: 0.54 g (69%). 1H NMR (DMSO): δ 6.75 (d, 3JHH = 8.4 Hz, 4H, 3,5-Bz); 7.18 (s, 2H, CH-Pz); 7.32 (t, 5JHH = 14.4 Hz, 4H, 4-Ph-Pz); 7.44 (t, 5JHH = 15.2 Hz, 8H, 3,5-Ph-Pz); 7.78 (d, 3JHH = 8.0 Hz, 4H, 2,6-Bz); 7.83 (d, 3JHH = 7.6 Hz, 8H, 2,6-Ph-Pz). 13C{1H} NMR (DMSO): δ 99.7; 114.6; 125.1; 127.9; 128.9; 131.6; 160.4. Anal. Calcd for C44H34N4O6Zn.CH3OH: C, 66.54; H, 4.72; N, 6.90%. Found: C, 66.09; H, 4.65; N, 7.00%.
bis-(3,5-di-tert-Pyrazolyl)zinc(II) 2-chlorobenzoate (4)
Reaction of [Zn(OAc)2] (0.18 g, 1.00 mmol), 2-Cl-C6H4COOH (0.31 g, 2.00 mmol) and L2 (0.36 g, 2.00 mmol) gave 3 as a white solid, obtained by evaporation of the solvent in vacuo. Yield: 0.76 g (52%). 1H NMR (CDCl3): δ 1.27 (2s, 36H, CH3-Pz); 5.99 (s, 2H, CH-Pz); 7.20 (m, 4H, 3,5-Bz); 7.33 (d, 3JHH = 7.6 Hz, 2H, 4-Bz); 7.74 (s, 2H, 6-Bz); 15.45 (s, 2H, NH-Pz). 13C{1H} NMR (DMSO): δ 30.1; 31.6; 100.2; 126.2; 130.1; 130.4; 131.9; 173.0. Anal. Calcd for C36H48Cl2N4O4Zn: C, 58.66; H, 6.56; N, 7.60%. Found: C, 58.14; H, 6.65; N, 7.30%.
3.3. Representative Procedure for CO2 and CHO Copolymerization Reaction
In a typical reaction, to a clean autoclave (dried at 80 °C overnight in dynamic vacuum) containing the appropriate amount of the catalyst, was added 2.0 mL CHO under inert conditions. The reaction was initiated by pressurizing the autoclave with CO2 and left to stir at the required temperature for the appropriate time. The autoclave was then vented, allowed to cool to room temperature and a small amount of phenanthrene (internal standard) added. A small aliquot was removed for 1H NMR and IR analysis at the end of each reaction and the remaining mixture was quenched with five drops of a 5% HCl solution in methanol.
The copolymerization reaction that was monitored by IR spectroscopy was performed in a 150 mL Parr autoclave fitted with a ReactIR probe. The Parr autoclave, containing the appropriate amount of catalyst, was charged with toluene (7.0 mL) and CHO (1.0 mL) via the injection pot. A single 120 scan background spectrum was collected. The autoclave was pressurized to 3.0 MPa of CO2 and heated at 120 °C as the IR probe began collecting scans. The probe was set up to collect one spectrum every minute for a period of 26 h 56 min.
4. Conclusions
We have prepared the first pyrazole zinc(II) benzoate complexes that are active and highly selective for the formation of PCHC from CO2 and CHO copolymerization reaction. All the PCHC copolymers produced have glass transition temperatures that are indicative of almost completely alternating copolymers (high carbonate content). Our catalyst systems achieved TOFs that are higher than those reported by Darensbourg using monomeric zinc(II) carboxylate complexes containing electron-withdrawing substituents. The two most active catalysts are the ones with the electron-deficient 3,5-dinitrobenzoate and 2-chlorobenzoate groups (2 and 4), indicating that the higher electrophilicity of the zinc centers in these two catalysts is responsible for the higher activity. The results obtained also show that catalysts 1–3 are more selective for PCHC formation than catalyst 4, which only shows less than 70% selectivity for PCHC. We can thus conclude that catalysts 1–3 are best for the selective formation of PCHC while catalyst 4 would be best for CCHC formation; but, in general, we see these pyrazole supported zinc(II) benzoates as new catalysts systems in the copolymerization of epoxide and carbon dioxide that can be tuned to make either predominantly polycarbonates or cyclic carbonates and are currently working on various modifications of metal complexes using pyrazolyl ligands for this and other ring-opening polymerization reactions.
Supplementary Materials
Electronic supplementary information (ESI) available. CCDC 1000656 contains the supplementary crystallographic data for this paper. This data can be obtained free from the Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/data_request/cif.
Acknowledgments
We are grateful to the National Research Foundation (NRF) for the support received under a South Africa-Japan Joint Science project and the University of Johannesburg for funding this project under the Synthesis and Catalysis Research Centre.
Author Contributions
Divambal Appavoo synthesized the initial zinc complex; Bernard O. Owaga determined the crystal structure of complex 2; Mapudumo L. Lephoto prepared the zinc complexes and performed the catalysis and wrote the initial draft of the manuscript; Koji Nakano worked with MLL on the catalysis as MML’s immediate supervisor and also corrected the initial draft of the manuscript; Kyoko Nozaki and James Darkwa conceived this work as a collaborative project. All authors were given the opportunity to read the manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References and Notes
- Coates, G.W.; Moore, D.R. Discrete metal-based catalysts for the copolymerization of CO2 and epoxides: Discovery, reactivity, optimization, and mechanism. Angew. Chem. Int. Ed. 2004, 43, 6618–6639. [Google Scholar] [CrossRef]
- Koning, C.; Wildeson, J.; Parton, R.; Plum, B.; Steeman, P.; Darensbourg, D. Synthesis and characterization of poly(cyclohexane carbonate), synthesis from CO2 and cyclohexene oxide. J. Polym. 2001, 42, 3995–4004. [Google Scholar] [CrossRef]
- Harnett, C.K.; Coates, G.W.; Craighead, H.G. Heat-depolymerizable polycarbonates as electron beam patternable sacrificial layers for nanofluidics. J. Vac. Sci. Technol. 2001, B19, 2842–2845. [Google Scholar] [CrossRef]
- Harnett, C.K.; Satyalakshmi, K.M.; Coates, G.W.; Craighead, H.G. Direct electron-beam patterning of surface coating and sacrificial layers for micro-total analysis systems. J. Photopolym. Sci. Technol. 2002, 15, 493–496. [Google Scholar] [CrossRef]
- Wu, G.-P.; Jiang, S.-D.; Lu, X.-B.; Ren, W.-M.; Yan, S.-K. Stereoregular poly(cyclohexene carbonate)s: Unique crystallization behaviour. Chin. J. Polym. Sci. 2012, 30, 487–492. [Google Scholar] [CrossRef]
- Darensbourg, D.J. Making plastics from carbon dioxide: salen metal complexes for the production of polycarbonates from expoxides and CO2. Chem. Rev. 2007, 107, 2388–2410. [Google Scholar] [CrossRef]
- Coates, G.W.; Jeske, R.C. Handbook of Green Chemistry; Crabtree, R.H., Ed.; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2009; p. 343. [Google Scholar]
- Nakano, K.; Hashimoto, S.; Nozaki, K. Bimetallic mechanism operating in the copolymerization of proplyene oxide and carbon dioxide catalyzed by cobalt-salen complexes. Chem. Sci. 2010, 1, 369–373. [Google Scholar] [CrossRef]
- Nakano, K.; Nakamura, M.; Nozaki, K. Alternating copolymerization of cyclohexene oxide and carbon dioxide catalyzed by (salalen)CrCl complexes. Macromolecules 2009, 42, 6972–6980. [Google Scholar] [CrossRef]
- Nakano, K.; Kamada, T.; Nozaki, K. Selective formation of polycarbonate over cyclic carbonate: copolymerization of epoxides and carbon dioxide catalyzed by a cobalt(III) with piperidinium end-capping arm. Angew. Chem. Int. Ed. 2006, 45, 7274–7277. [Google Scholar] [CrossRef]
- Ren, W.-M.; Zhang, X.; Liu, Y.; Li, J.-F.; Wang, H.; Lu, X.-B. Highly active, bifunctional Co(III)salen catalyst for alternating copolymerization of CO2 with cyclohexene oxide and terpolymerization with aliphatic epoxides. Macromolecules 2010, 43, 1396–1402. [Google Scholar] [CrossRef]
- Liu, Y.; Ren, W.-M.; Liu, J.; Lu, X.-B. Asymmetric copolymerization of CO2 with meso-epoxides mediated by dinuclear cobalt(III) complexes: unprecedented enantioselectivity and activity. Angew. Chem. Int. Ed. 2013, 52, 11594–11598. [Google Scholar] [CrossRef]
- Super, M.; Berluche, E.; Costello, C.; Beckman, E. Copolymerization of 1,2-epoxicyclohexane and carbon dioxide using carbon dioxide as both reactant and solvent. Macromolecules 1997, 30, 368–372. [Google Scholar]
- Super, M.; Beckman, E. Co polymerization of CO2 and cyclohexene oxide. J. Macromol. Symp. 1998, 127, 89–108. [Google Scholar]
- Darensbourg, D.J.; Holtcamp, M. Catalytic activity of zinc(II) phenoxides which possess readily accessible coordination sites. Copolymerization and terpolymerization of epoxides and carbon dioxide. Macromolecules 1995, 28, 7577–7579. [Google Scholar]
- Cheng, M.; Lobkovsky, E.B.; Coates, G.W. Catalytic reactions involving C1 feedstocks: new high-activity zinc(II)-based catalysts for the alternating copolymerization of carbon dioxide and epoxides. J. Am. Chem. Soc. 1998, 120, 11018–11019. [Google Scholar]
- Chapman, A.M.; Keyworth, C.; Kembert, M.R.; Lennox, A.J.J.; Williams, C.K. Adding value to power station captured CO2: tolerant Zn and Mg homogeneous catalysts for polycarbonate polyol production. ACS Catal. 2015, 5, 1581–1588. [Google Scholar] [CrossRef]
- Kembert, M.R.; Knight, P.D.; Reung, P.T.; Williams, C.K. Highly active dizinc catalyst for the copolymerization of carbon dioxide and cyclohexene oxide at one atmosphere pressure. Angew. Chem. Int. Ed. 2009, 48, 931–933. [Google Scholar] [CrossRef]
- Elmas, S.; Subhani, M.A.; Leitner, W.; Muller, T.E. Anion effect controlling the selectivity in the zinc catalysed copolymerisation of CO2 and cyclohexene oxide. Beilstein J. Org. Chem. 2015, 11, 42–49. [Google Scholar] [CrossRef]
- CCDC 1000656 Contains the Supplementary Crystallographic Data for this Paper. This Data Can Be Obtained Free from the Cambridge Crystallographic Data Centre. Available online www.ccdc.cam.ac.uk/data_request/cif (accessed on 18 January 2016).
- Sarma, R.; Kalita, D.; Baruah, B. Solvent induced reactivity of 3,5-dimethylpyrazole towards zinc(II) carboxylates. Dalton Trans. 2009, 7428–7436. [Google Scholar] [CrossRef]
- Klaus, S. Development of efficient catalysts for the CO2/epoxide copolymerization reaction. Ph.D. Thesis, Technical University of Munich, Munich, Germany, 2011. [Google Scholar]
- Darensbourg, D.J.; Wildeson, J.R.; Yarbrough, J.C. Coplymerization and terpolymerization of CO2 and epoxides using soluble zinc crotonate catalyst precursor. Inorg. Chem. 2002, 41, 973–980. [Google Scholar] [CrossRef]
- Cheng, M.; Moore, D.R.; Reczek, J.J.; Chamberlain, B.M.; Lobkovsky, E.B.; Coates, G.W. Single-site β-diimine zinc catalysts for the alternating copolymerization of CO2 and expoxides: catalyst synthesis and unprecedented activity. J. Am. Chem. Soc. 2001, 123, 8738–8749. [Google Scholar] [CrossRef]
- Moore, D.R.; Cheng, M.; Lobkovsky, E.B.; Coates, G.W. Electronic and steric effects on CO2/epoxide polymerization: subtle modification resulting in superior activities. Angew. Chem. Int. Ed. 2002, 41, 2599–2602. [Google Scholar] [CrossRef]
- Cheng, M.; Moore, D.R.; Lobkovsky, E.B.; Coates, G.W. Mechanism for the polymerization of epoxides and CO2 using β-diiminate zinc catalysts: evidence for bimetallic epoxide enchainment. J. Am. Chem. Soc. 2003, 125, 11911–11924. [Google Scholar]
- Darensbourg, D.J.; Mackiewicz, R.M.; Phelps, A.L.; Billodeaux, D.R. Copolymerization of epoxides catalyzed by metal salen complexes. Acc. Chem. Res. 2004, 37, 836–844. [Google Scholar] [CrossRef]
- Darensbourg, D.J.; Yarbrough, J.C.; Ortiz, C.; Fang, C.C. Comparative kinetics studies of the copolymerization of cyclohexene oxide and propylene oxide with carbon dioxide in the presence of chromium salen derivatives. In situ FTIR measurements of copolymers vs cyclic carbonate production. J. Am. Chem. Soc. 2003, 125, 7586–7591. [Google Scholar] [CrossRef]
- Darensbourg, D.J.; Yarbrough, J.C.; Wildeson, J.R.; Reibenspiea, J.H. Bis 2,6-difluorophenoxide dimeric complexes of zinc and cadmium and their phosphine adducts: lessons learnt relative to carbon dioxide/cyclohexene oxide alternation copolymerization processes catalyzed by zinc phenoxides. J. Am. Chem. Soc. 2000, 122, 12487–12496. [Google Scholar] [CrossRef]
- Darensbourg, D.J.; Mackiewicz, R.M.; Phelps, A.L.; Rodgers, J.L. The coupling of epoxides and carbon dioxide in the presence of homogeneous transition-metal catalysts. Production of polycarbonates vs cyclic carbonates. Prepr. Pap-Am. Chem. Soc. Div. Fuel Chem. 2004, 49, 5–6. [Google Scholar]
- Kitajima, N.; Fujisawa, K.; Fujimoto, C.; Morooka, Y.; Hashimoto, S.; Kitagawa, T.; Toriumi, K.; Tatsumi, K.; Nakamura, A. A new model for dioxygen binding in hemocyanin. Synthesis, characterization and molecular structure of the.mu.-.eta.2:.eta.2 peroxo dinuclear copper(II) complexes, [Cu(HB(3,5-R2pz)3]2(O2) (R = isopropyl and Ph). J. Am. Chem. Soc. 1992, 114, 1277–1291. [Google Scholar] [CrossRef]
- Appavoo, D. Pyrazole and pyrazolyl copper and zinc complexes in ring opening polymerization of ε-caprolactone and d,L-lactide. Master’s Thesis, University of Johannesburg, Johannesburg, South Africa, September 2011. [Google Scholar]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).