Catalytic Hydrogenation of CO2 to Methanol: Study of Synergistic Effect on Adsorption Properties of CO2 and H2 in CuO/ZnO/ZrO2 System
Abstract
:1. Introduction
2. Results and Discussion
2.1. Textural Properties of CZZ Catalysts
| Sample | Chemical Composition a (wt. %) | Cu:Zn (Atomic) | SBET (m2·g−1) | VP (cm3·g−1) | CuO b Dia. (nm) | ||
|---|---|---|---|---|---|---|---|
| CuO | ZnO | ZrO2 | |||||
| CZZ-271 | 21.5 | 67.0 | 11.5 | 0.3 | 81.8 | 0.31 | 10.3 |
| CZZ-361 | 31.0 | 57.7 | 11.3 | 0.5 | 70.1 | 0.22 | 12.6 |
| CZZ-451 | 41.2 | 48.6 | 11.7 | 0.8 | 61.6 | 0.24 | 13.4 |
| CZZ-541 | 52.0 | 37.7 | 10.3 | 1.3 | 48.3 | 0.27 | 14.0 |
| CZZ-631 | 60.8 | 28.2 | 11.0 | 2.0 | 44.6 | 0.26 | 14.7 |
| CZZ-721 | 70.4 | 18.8 | 10.8 | 3.6 | 41.2 | 0.16 | 16.2 |
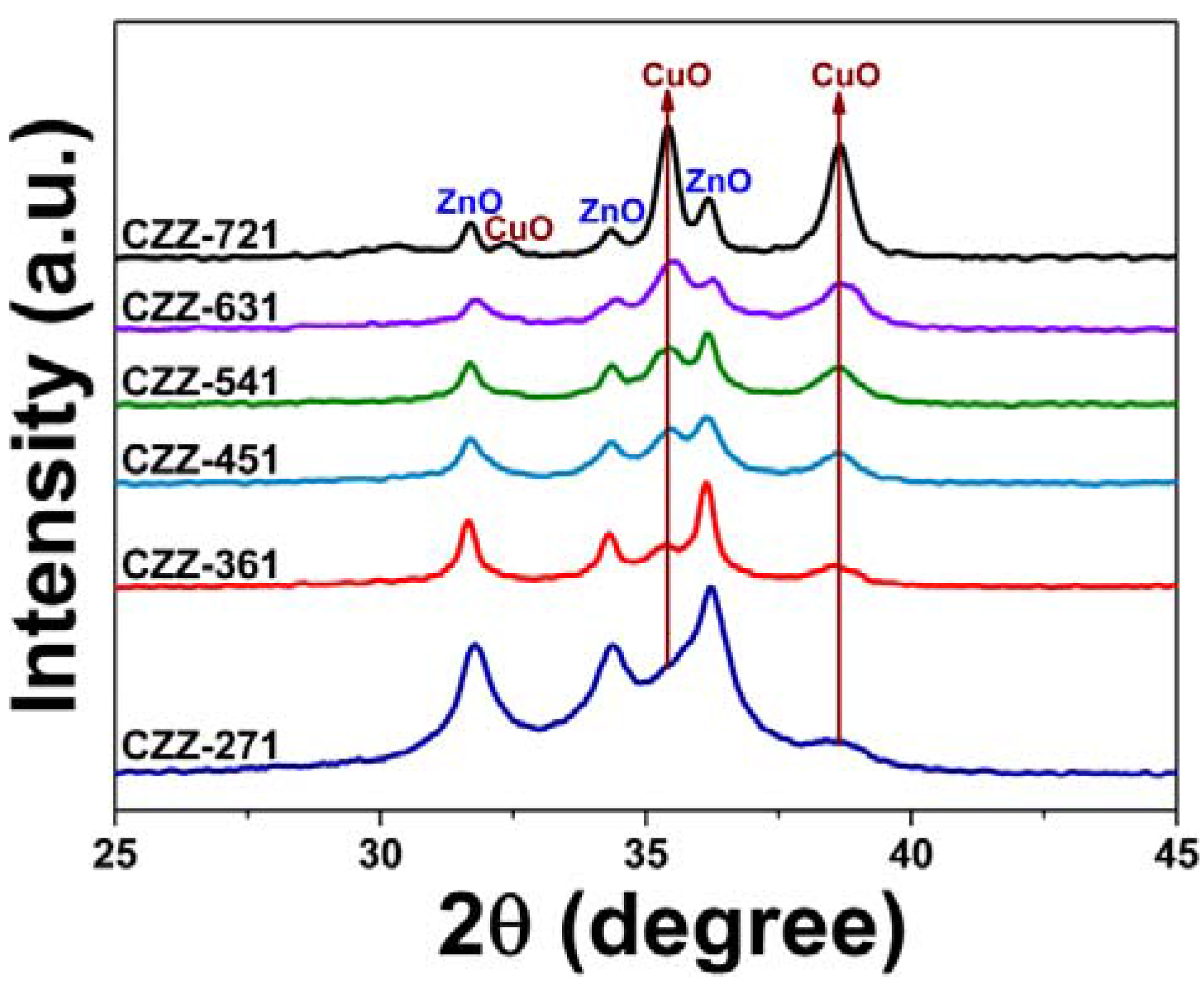
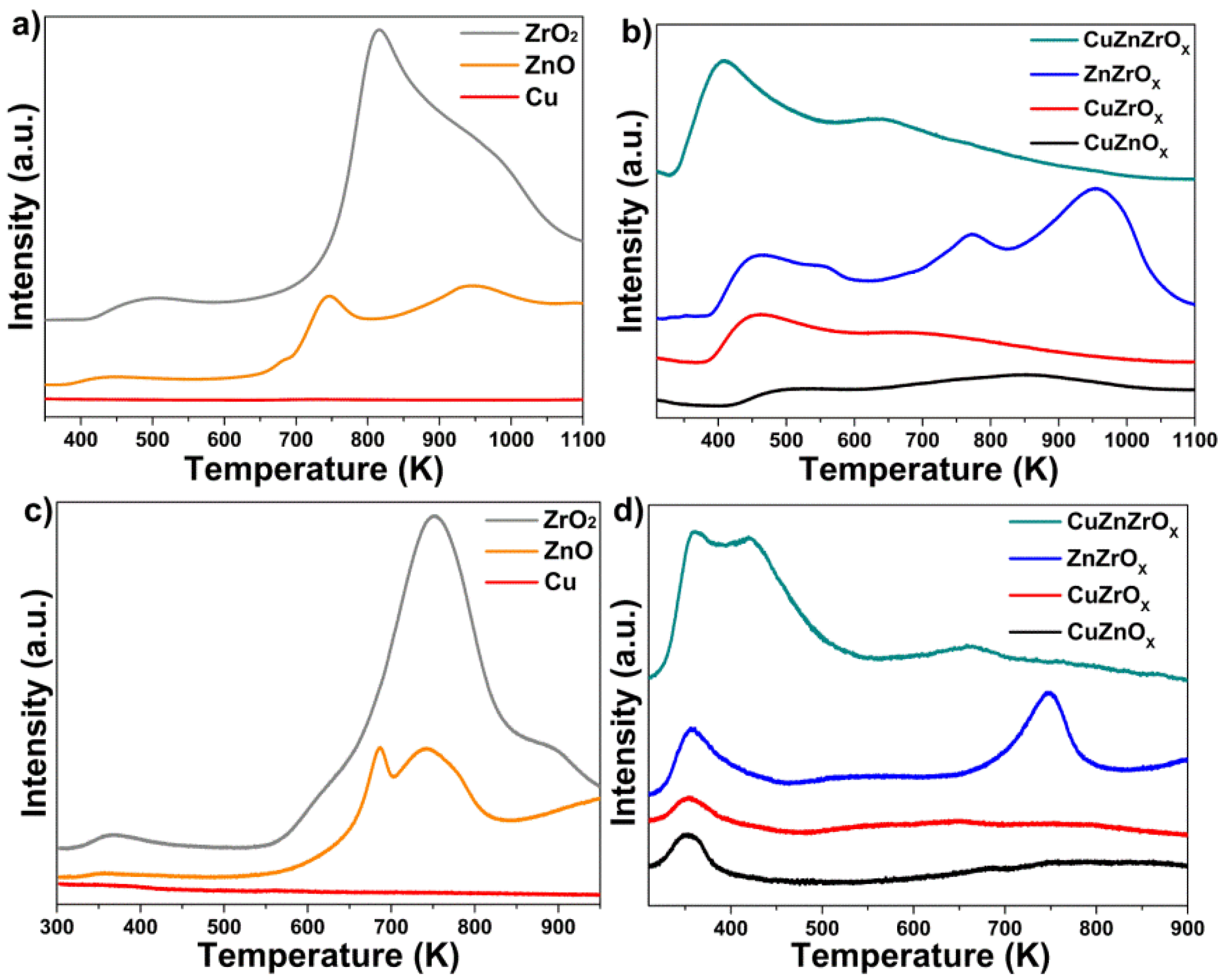
2.2. Reduction Behaviour of CZZ Catalysts

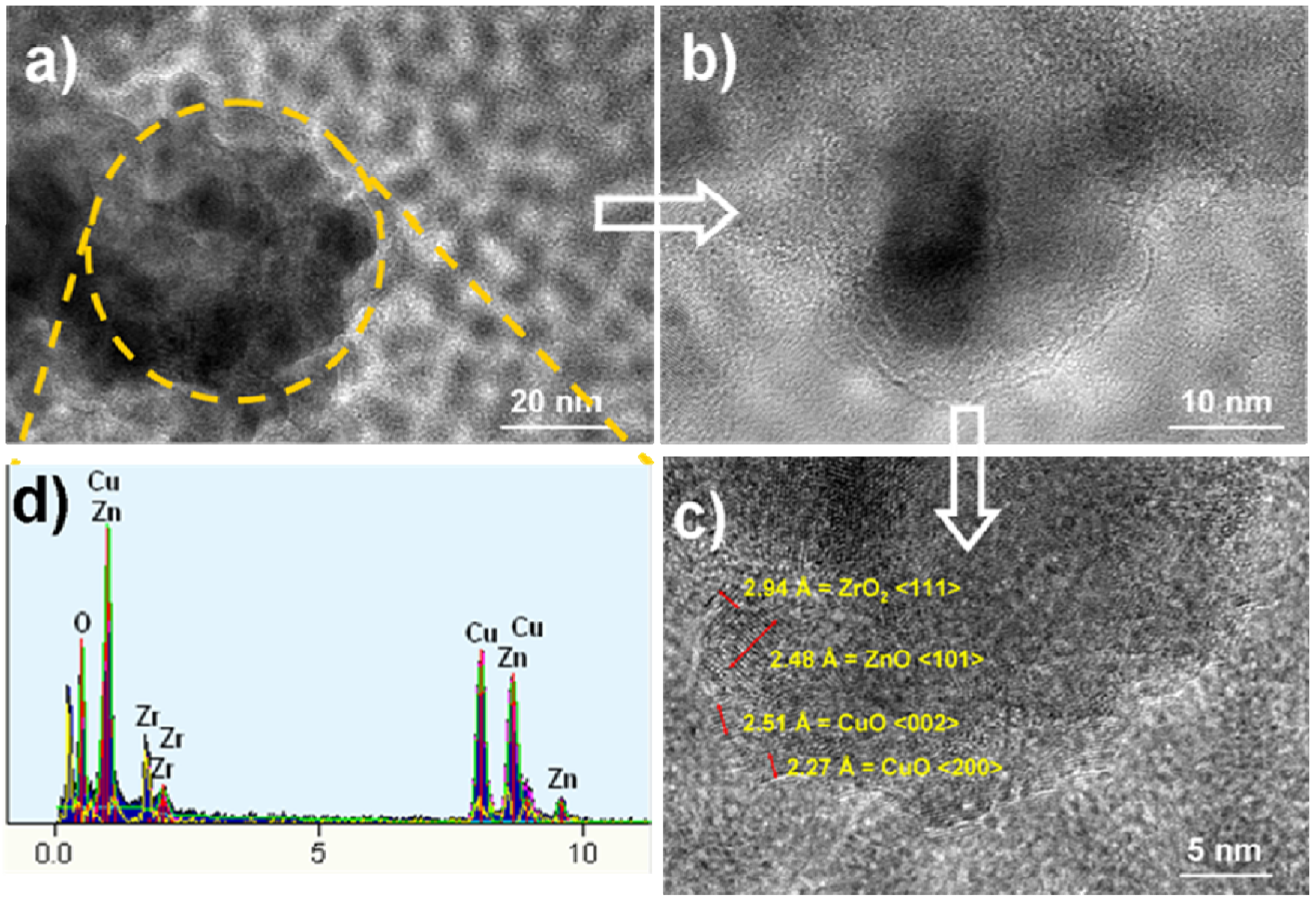
2.3. Morphologies of Catalysts

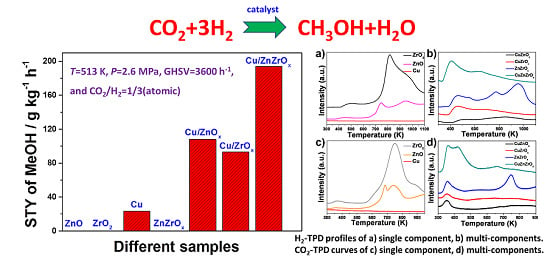
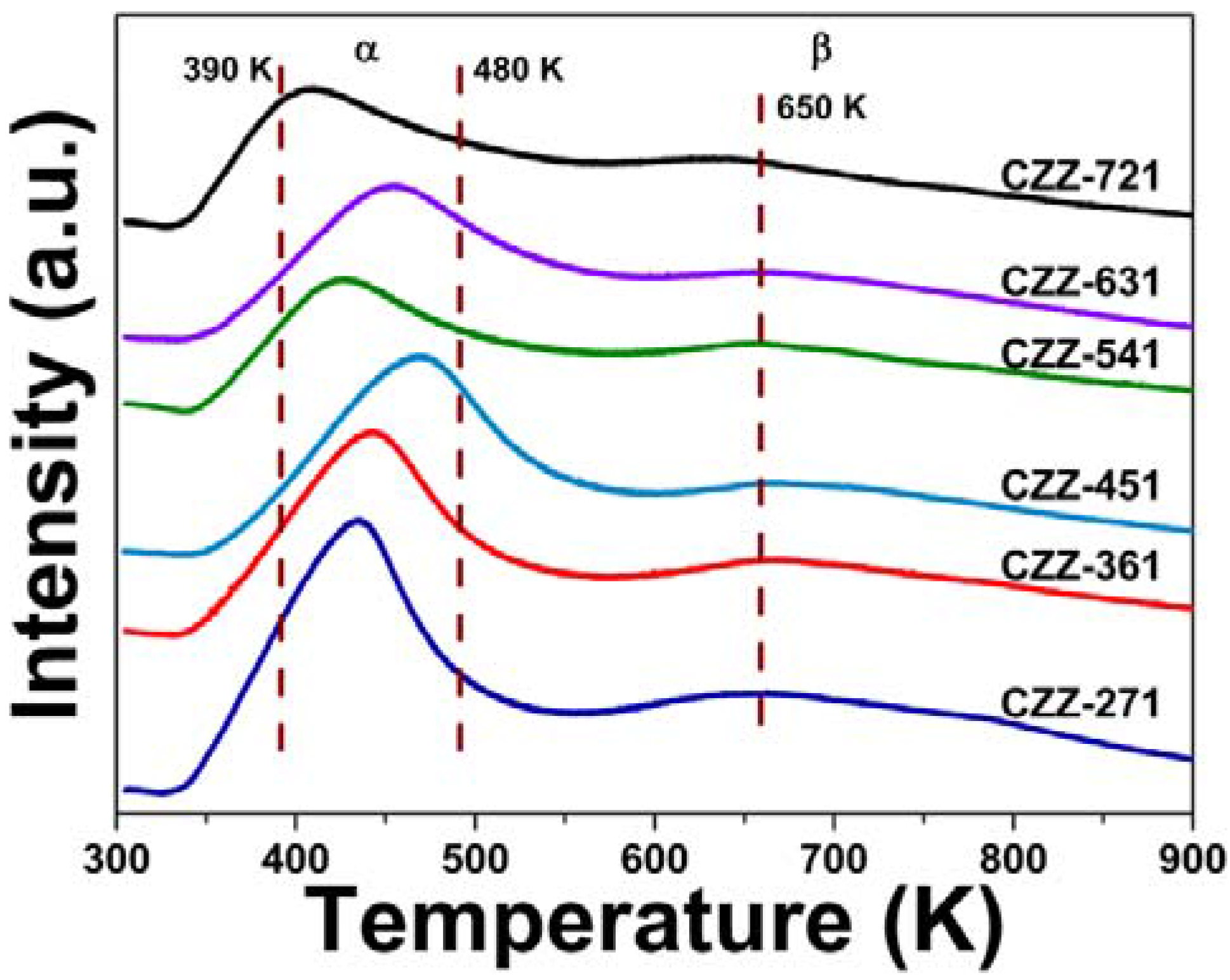
2.4. H2 Chemisorption
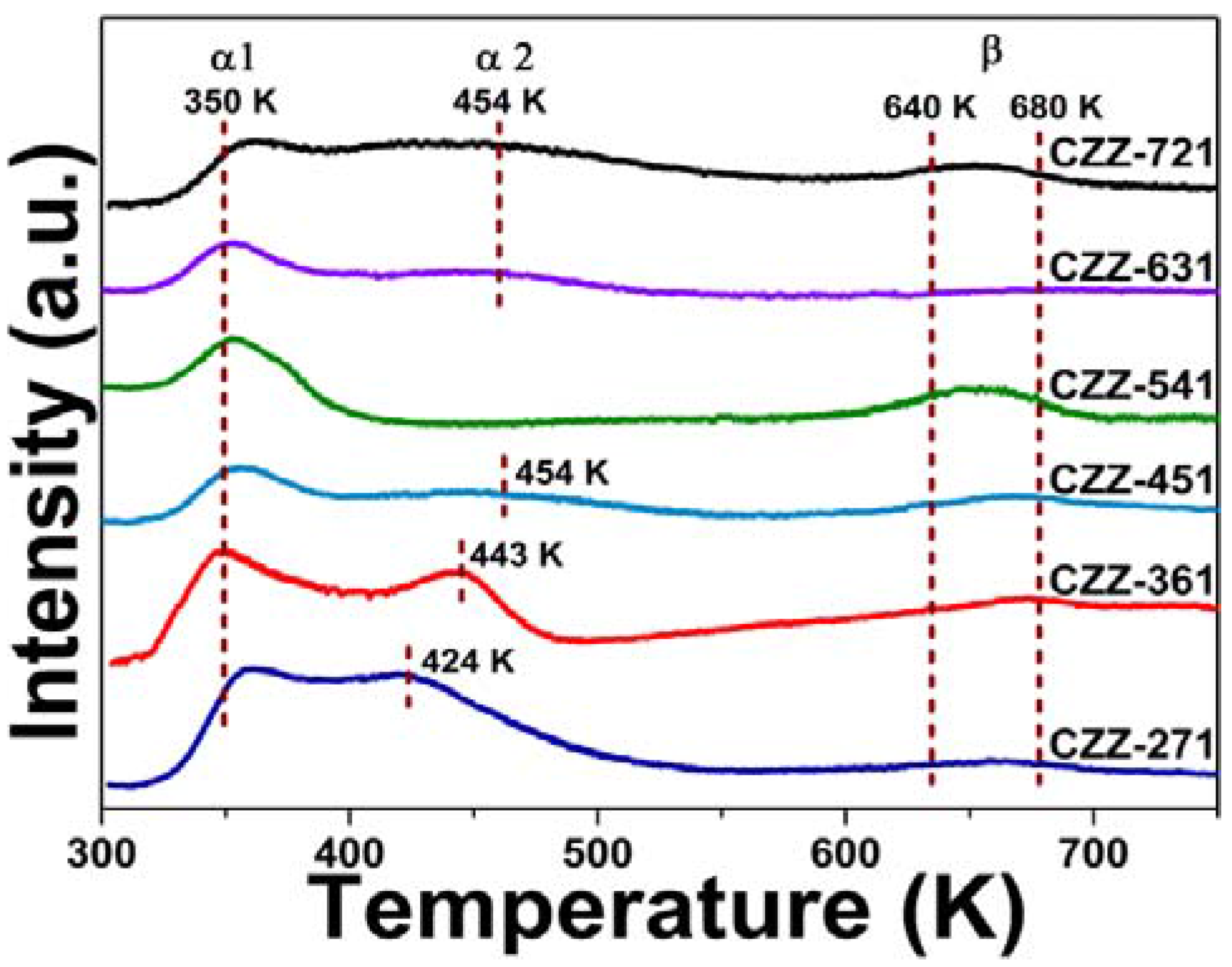
2.5. CO2 Chemisorption and XPS Analysis
| Element | Surface Atomic Concentrations of Calcined Catalysts (at.%) | |||||
|---|---|---|---|---|---|---|
| CZZ-271 | CZZ-361 | CZZ-451 | CZZ-541 | CZZ-631 | CZZ-721 | |
| Cu2+ | 4.2 | 5.7 | 13.0 | 13.2 | 14.9 | 19.6 |
| Zn2+ | 34.4 | 32.6 | 23.1 | 20.1 | 17.1 | 11.9 |
| Zr4+ | 5.6 | 5.8 | 3.9 | 3.6 | 6.3 | 7.0 |
2.6. Catalytic Performance of CO2 into Methanol
| Samples | CO2 Conversion (%) | Selectivity (C-mol %) | WTY of MeOH (g·kg−1·h−1) | |
|---|---|---|---|---|
| MeOH | CO | |||
| CZZ-271 | 32.9 | 45.8 | 54.2 | 193.9 |
| CZZ-361 | 31.9 | 39.3 | 60.7 | 161.1 |
| CZZ-451 | 30.7 | 33.2 | 66.8 | 138.2 |
| CZZ-541 | 28.5 | 29.7 | 70.3 | 108.7 |
| CZZ-631 | 30.4 | 30.3 | 69.7 | 118.3 |
| CZZ-721 | 32.0 | 35.1 | 64.9 | 144.6 |
2.7. Discussion on the Synergetic Effect of CZZ Catalyst
| Sample | CO2 Conversion (%) | Selectivity (C-mol %) | WTY of MeOH (g·kg−1·h−1) | |
|---|---|---|---|---|
| MeOH | CO | |||
| ZnO | 1.5 | - | 100.0 | - |
| ZrO2 | 2.2 | - | 100.0 | - |
| Cu a | 6.3 | 28.8 | 71.2 | 23.4 |
| ZnO/ZrO2-071 | 3.3 | - | 100.0 | - |
| Cu a/ZnO-270 | 28.3 | 29.8 | 70.2 | 108.2 |
| Cu a/ZrO2-201 | 22.7 | 31.9 | 68.1 | 93.3 |
3. Experimental Section
3.1. Catalyst Preparation
3.2. Catalyst Characterizations
3.3. Catalysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Olah, G.A.; Prakash, G.K.S.; Goeppert, A. Anthropogenic chemical carbon cycle for a sustainable future. J. Am. Chem. Soc. 2011, 133, 12881–12898. [Google Scholar] [CrossRef] [PubMed]
- Zangeneh, F.T.; Sahebdelfar, S.; Ravanchi, M.T. Conversion of carbon dioxide to valuable petrochemicals: An approach to clean development mechanism. J. Nat. Gas Chem. 2011, 20, 219–231. [Google Scholar] [CrossRef]
- Olah, G. After oil and gas: Methanol economy. Catal. Lett. 2004, 93, 1–2. [Google Scholar] [CrossRef]
- Nakamura, J.; Uchijima, T.; Kanai, Y.; Fujitani, T. The role of ZnO in Cu/ZnO methanol synthesis catalysts. Catal. Today 1996, 28, 223–230. [Google Scholar] [CrossRef]
- Behrens, M. Heterogeneous catalysis of CO2 conversion to methanol on copper surfaces. Angew. Chem. Int. Ed. 2014, 53, 12022–12024. [Google Scholar] [CrossRef] [PubMed]
- Inui, T.; Hara, H.; Takeguchi, T.; Kim, J. Structure and function of Cu-based composite catalysts for highly effective synthesis of methanol by hydrogenation of CO2 and CO. Catal. Today 1997, 36, 25–32. [Google Scholar] [CrossRef]
- Ma, Y.; Sun, Q.; Wu, D.; Fan, W.; Zhang, Y.; Deng, J. A practical approach for the preparation of high activity Cu/ZnO/ZrO2 catalyst for methanol synthesis from CO2 hydrogenation. Appl. Catal. A 1998, 171, 45–55. [Google Scholar] [CrossRef]
- Saito, M. R & D activities in Japan on methanol synthesis from CO2 and H2. Catal. Surv. Jpn. 1998, 2, 175–184. [Google Scholar]
- Saito, M.; Fujitani, T.; Takeuchi, M.; Watanabe, T. Development of copper/zinc oxide-based multicomponent catalysts for methanol synthesis from carbon dioxide and hydrogen. Appl. Catal. A 1996, 138, 311–318. [Google Scholar] [CrossRef]
- Melián-Cabrera, I.; López-Granados, M.; Fierro, J.L.G. Reverse Topotactic Transformation of a Cu-Zn-Al Catalyst during Wet Pd Impregnation: Relevance for the Performance in Methanol Synthesis from CO2/H2 Mixtures. J. Catal. 2002, 210, 273–284. [Google Scholar] [CrossRef]
- Waugh, K.C. Methanol Synthesis. Catal. Today 1992, 15, 51–75. [Google Scholar] [CrossRef]
- Sun, Q.; Liu, C.; Pan, W.; Zhu, Q.; Deng, J. In situ IR studies on the mechanism of methanol synthesis over an ultrafine Cu/ZnO/Al2O3 catalyst. Appl. Catal. A 1998, 171, 301–308. [Google Scholar] [CrossRef]
- Arena, F.; Mezzatesta, G.; Zafarana, G.; Trunfio, G.; Frusteri, F.; Spadaro, L. Effects of oxide carriers on surface functionality and process performance of the Cu-ZnO system in the synthesis of methanol via CO2 hydrogenation. J. Catal. 2013, 300, 141–151. [Google Scholar] [CrossRef]
- Sato, A.G.; Volanti, D.P.; Meira, D.M.; Damyanova, S.; Longo, E.; Bueno, J.M.C. Effect of the ZrO2 phase on the structure and behavior of supported Cu catalysts for ethanol conversion. J. Catal. 2013, 307, 1–17. [Google Scholar] [CrossRef]
- Raudaskoski, R.; Marita, V.N.; Keiski, R.L. The effect of ageing time on co-precipitated CuZnOZrO2 catalysts used in methanol synthesis from CO2 and H2. Top. Catal. 2007, 45, 57–60. [Google Scholar] [CrossRef]
- Arena, F.; Katia, B.; Giuseppe, I.; Giuseppe, B.; Lorenzo, S.; Francesco, F. Synthesis, characterization and activity pattern of Cu-ZnO/ZrO2 catalysts in the hydrogenation of carbon dioxide to methanol. J. Catal. 2007, 249, 185–194. [Google Scholar] [CrossRef]
- Natesakhawat, S.; Lekse, J.W.; Baltrus, J.P.; Ohodnicki, P.R.; Howard, B.H.; Deng, X.; Matranga, C. Active Sites and Structure—Activity Relationships of Copper-Based Catalysts for Carbon Dioxide Hydrogenation to Methanol. ACS Catal. 2012, 2, 1667–1676. [Google Scholar] [CrossRef]
- Liao, F.; Zeng, Z.; Eley, C.; Lu, Q.; Hong, X.; Tsang, S.C.E. Electronic modulation of a copper/Zinc oxide catalyst by a heterojunction for selective hydrogenation of carbon dioxide to methanol. Angew. Chem. 2012, 124, 5934–5938. [Google Scholar] [CrossRef]
- Jia, L.; Gao, J.; Fang, W.; Li, Q. Influence of copper content on structural features and performance of pre-reduced LaMn1−xCuxO3 (0 ≤ x < 1) catalysts for methanol synthesis from CO2/H2. J. Rare Earth 2010, 28, 747–751. [Google Scholar]
- Słoczyński, J.; Grabowski, R.; Olszewski, P.; Kozłowska, A.; Stoch, J.; Lachowska, M.; Skrzypek, J. Effect of metal oxide additives on the activity and stability of Cu/ZnO/ZrO2 catalysts in the synthesis of methanol from CO2 and H2. Appl. Catal. A 2006, 310, 127–137. [Google Scholar] [CrossRef]
- Słoczyński, J.; Grabowski, R.; Kozłowska, A.; Olszewski, P.; Lachowska, M.; Skrzypek, J.; Stoch, J. Effect of Mg and Mn oxide additions on structural and adsorptive properties of Cu/ZnO/ZrO2 catalysts for the methanol synthesis from CO2. Appl. Catal. A 2003, 249, 129–138. [Google Scholar] [CrossRef]
- Brown, N.J.; Weiner, J.; Hellgardt, K.; Shaffer, M.S.P.; Williams, C.K. Phosphinate stabilised ZnO and Cu colloidal nanocatalysts for CO2 hydrogenation to methanol. Chem. Commun. 2013, 49, 11074–11076. [Google Scholar] [CrossRef] [PubMed]
- Bonura, G.; Arena, F.; Mezzatesta, G.; Cannilla, C.; Spadaro, L.; Frusteri, F. Role of the ceria promoter and carrier on the functionality of Cu-based catalysts in the CO2-to-methanol hydrogenation reaction. Catal. Today 2011, 171, 251–256. [Google Scholar] [CrossRef]
- Wang, L.; Liu, Q.; Chen, M.; Liu, Y.; Cao, Y.; He, H.; Fan, K. Structural Evolution and Catalytic Properties of Nanostructured Cu/ZrO2 Catalysts Prepared by Oxalate Gel-Coprecipitation Technique. J. Phys. Chem. C 2007, 111, 16549–16557. [Google Scholar] [CrossRef]
- Ladera, R.; Pérez-Alonso, F.J.; González-Carballo, J.M.; Ojeda, M.; Rojas, S.; Fierro, J.L.G. Catalytic valorization of CO2 via methanol synthesis with Ga-promoted Cu-ZnO-ZrO2 catalysts. Appl. Catal. B 2013, 142–143, 241–248. [Google Scholar] [CrossRef]
- Liao, F.; Huang, Y.; Ge, J.; Zheng, W.; Tedsree, K.; Collier, P.; Hong, X.; Tsang, S.C. Morphology-dependent interactions of ZnO with Cu nanoparticles at the materials’ interface in selective hydrogenation of CO2 to CH3OH. Angew. Chem. Int. Ed. 2011, 50, 2162–2165. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Ma, Z.; Zhao, N.; Wei, W.; Hu, T.; Sun, Y. Methanol synthesis from CO2-rich syngas over a ZrO2 doped CuZnO catalyst. Catal. Today 2006, 115, 222–227. [Google Scholar] [CrossRef]
- Arena, F.; Italiano, G.; Barbera, K.; Bonura, G.; Spadaro, L.; Frusteri, F. Basic evidences for methanol-synthesis catalyst design. Catal. Today 2009, 143, 80–85. [Google Scholar] [CrossRef]
- Guo, X.; Mao, D.; Lu, G.; Wang, S.; Wu, G. Glycine-nitrate combustion synthesis of CuO-ZnO-ZrO2 catalysts for methanol synthesis from CO2 hydrogenation. J. Catal. 2010, 271, 178–185. [Google Scholar] [CrossRef]
- Liu, X.; Lu, G.Q.; Yan, Z.; Beltramini, J. Recent Advances in Catalysts for Methanol Synthesis via Hydrogenation of CO and CO2. Ind. Eng. Chem. Res. 2003, 42, 6518–6530. [Google Scholar] [CrossRef]
- Zhang, Y.; Fei, J.; Yu, Y.; Zheng, X. Methanol synthesis from CO2 hydrogenation over Cu based catalyst supported on zirconia modified γ-Al2O3. Energy Convers. Manag. 2006, 47, 3360–3367. [Google Scholar] [CrossRef]
- Bonura, G.; Cordaro, M.; Cannilla, C.; Arena, F.; Frusteri, F. The changing nature of the active site of Cu-Zn-Zr catalysts for the CO2 hydrogenation reaction to methanol. Appl. Catal. B 2014, 152–153, 152–161. [Google Scholar] [CrossRef]
- Guo, X.; Mao, D.; Wang, S.; Wu, G.; Lu, G. Combustion synthesis of CuO-ZnO-ZrO2 catalysts for the hydrogenation of carbon dioxide to methanol. Catal. Commun. 2009, 10, 1661–1664. [Google Scholar] [CrossRef]
- Karelovic, A.; Bargibant, A.; Fernández, C.; Ruiz, P. Effect of the structural and morphological properties of Cu/ZnO catalysts prepared by citrate method on their activity toward methanol synthesis from CO2 and H2 under mild reaction conditions. Catal. Today 2012, 197, 109–118. [Google Scholar] [CrossRef]
- Słoczyński, J.; Grabowski, R.; Kozłowska, A.; Olszewski, P.; Stoch, J.; Skrzypek, J.; Lachowska, M. Catalytic activity of the M/(3ZnO·ZrO2) system (M = Cu, Ag, Au) in the hydrogenation of CO2 to methanol. Appl. Catal. A 2004, 278, 11–23. [Google Scholar] [CrossRef]
- Kanai, Y.; Watanabe, T.; Fujitani, T.; Uchijima, T.; Nakamura, J. The synergy between Cu and ZnO in methanol synthesis catalysts. Catal. Lett. 1996, 38, 157–163. [Google Scholar] [CrossRef]
- Askgaard, T.S.; Norskov, J.K.; Ovesen, C.V.; Stoltze, P. A Kinetic Model of Methanol Synthesis. J. Catal. 1995, 156, 229–242. [Google Scholar] [CrossRef]
- Morikawa, M.; Ahmed, N.; Yoshida, Y.; Izumi, Y. Photoconversion of carbon dioxide in zinc-copper-gallium layered double hydroxides: The kinetics to hydrogen carbonate and further to CO/methanol. Appl. Catal. B 2014, 144, 561–569. [Google Scholar] [CrossRef]
- Dong, X.; Zhang, H.; Lin, G.; Yuan, Y.; Tsai, K.R. Highly Active CNT-Promoted Cu-ZnO-Al2O3 Catalyst for Methanol Synthesis from H2/CO/CO2. Catal. Lett. 2003, 85, 237–246. [Google Scholar] [CrossRef]
- Genger, T.; Hinrichsen, O.; Muhler, M. The temperature-programmed desorption of hydrogen from copper surfaces. Catal. Lett. 1999, 59, 137–141. [Google Scholar] [CrossRef]
- Ahlers, J.; Grasser, J.; Loveless, B.; Muggli, D. Room-temperature oxidation of reduced Cu/ZnO surfaces by lattice oxygen diffusion. Catal. Lett. 2007, 114, 185–191. [Google Scholar] [CrossRef]
- Wilmer, H.; Genger, T.; Hinrichsen, O. The interaction of hydrogen with alumina-supported copper catalysts: A temperature-programmed adsorption/temperature-programmed desorption/isotopic exchange reaction study. J. Catal. 2003, 215, 188–198. [Google Scholar] [CrossRef]
- Tao, J.; Zhang, W.; Chen, M.; Au, C.T. TPD and TPSR studies of CO2 adsorption on Cu-baseed catalysts. Nat. Gas Chem. Ind. 1998, 23, 14–17. [Google Scholar]
- Gao, P.; Li, F.; Zhao, N.; Xiao, F.; Wei, W.; Zhong, L.; Sun, Y. Influence of modifier (Mn, La, Ce, Zr and Y) on the performance of Cu/Zn/Al catalysts via hydrotalcite-like precursors for CO2 hydrogenation to methanol. Appl. Catal. A 2013, 468, 442–452. [Google Scholar] [CrossRef]
- Sun, Q.; Xie, J.; Zhang, T. Dissociated adsorption of H2 on surfaces of Ni Pd and Cu. Acta Phys. Sin. 1995, 44, 1805–1813. [Google Scholar]
- Chinchen, G.C.; Spencer, M.S.; Waugh, K.C.; Whan, D.A. Promotion of methanol synthesis and the water-gas shift reactions by adsorbed oxygen on supported copper catalysts. Faraday Trans. 1 1987, 83, 2193–2212. [Google Scholar] [CrossRef]
- Hadden, R.A.; Vandervell, H.D.; Waugh, K.C.; Webb, G. The adsorption and decomposition of carbon dioxide on polycrystalline copper. Catal. Lett. 1988, 1, 27–33. [Google Scholar] [CrossRef]
- Bonura, G.; Cordaro, M.; Spadaro, L.; Cannilla, C.; Arena, F.; Frusteri, F. Hybrid Cu-ZnO-ZrO2/H-ZSM5 system for the direct synthesis of DME by CO2 hydrogenation. Appl. Catal. B 2013, 140–141, 16–24. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, C.; Chen, S.; Fei, X.; Liu, D.; Zhang, Y. Catalytic Hydrogenation of CO2 to Methanol: Study of Synergistic Effect on Adsorption Properties of CO2 and H2 in CuO/ZnO/ZrO2 System. Catalysts 2015, 5, 1846-1861. https://doi.org/10.3390/catal5041846
Huang C, Chen S, Fei X, Liu D, Zhang Y. Catalytic Hydrogenation of CO2 to Methanol: Study of Synergistic Effect on Adsorption Properties of CO2 and H2 in CuO/ZnO/ZrO2 System. Catalysts. 2015; 5(4):1846-1861. https://doi.org/10.3390/catal5041846
Chicago/Turabian StyleHuang, Chunjie, Shaoyun Chen, Xiaoyao Fei, Dai Liu, and Yongchun Zhang. 2015. "Catalytic Hydrogenation of CO2 to Methanol: Study of Synergistic Effect on Adsorption Properties of CO2 and H2 in CuO/ZnO/ZrO2 System" Catalysts 5, no. 4: 1846-1861. https://doi.org/10.3390/catal5041846
APA StyleHuang, C., Chen, S., Fei, X., Liu, D., & Zhang, Y. (2015). Catalytic Hydrogenation of CO2 to Methanol: Study of Synergistic Effect on Adsorption Properties of CO2 and H2 in CuO/ZnO/ZrO2 System. Catalysts, 5(4), 1846-1861. https://doi.org/10.3390/catal5041846