Facile and Low-Cost Preparation of Nb/Al Oxide Catalyst with High Performance for the Conversion of Kiwifruit Waste Residue to Levulinic Acid
Abstract
:1. Introduction
2. Results and Discussion
2.1. Catalyst Characterization
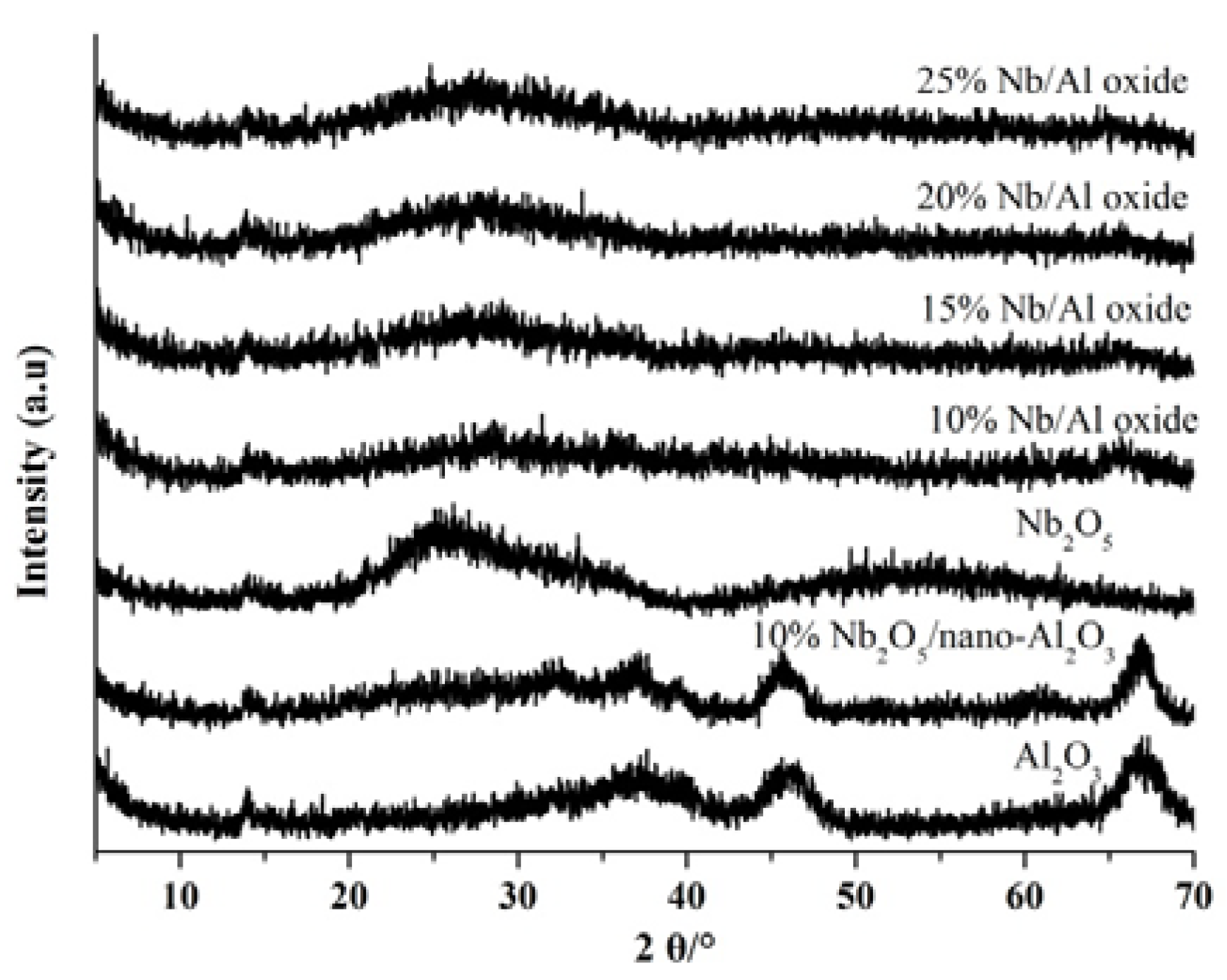
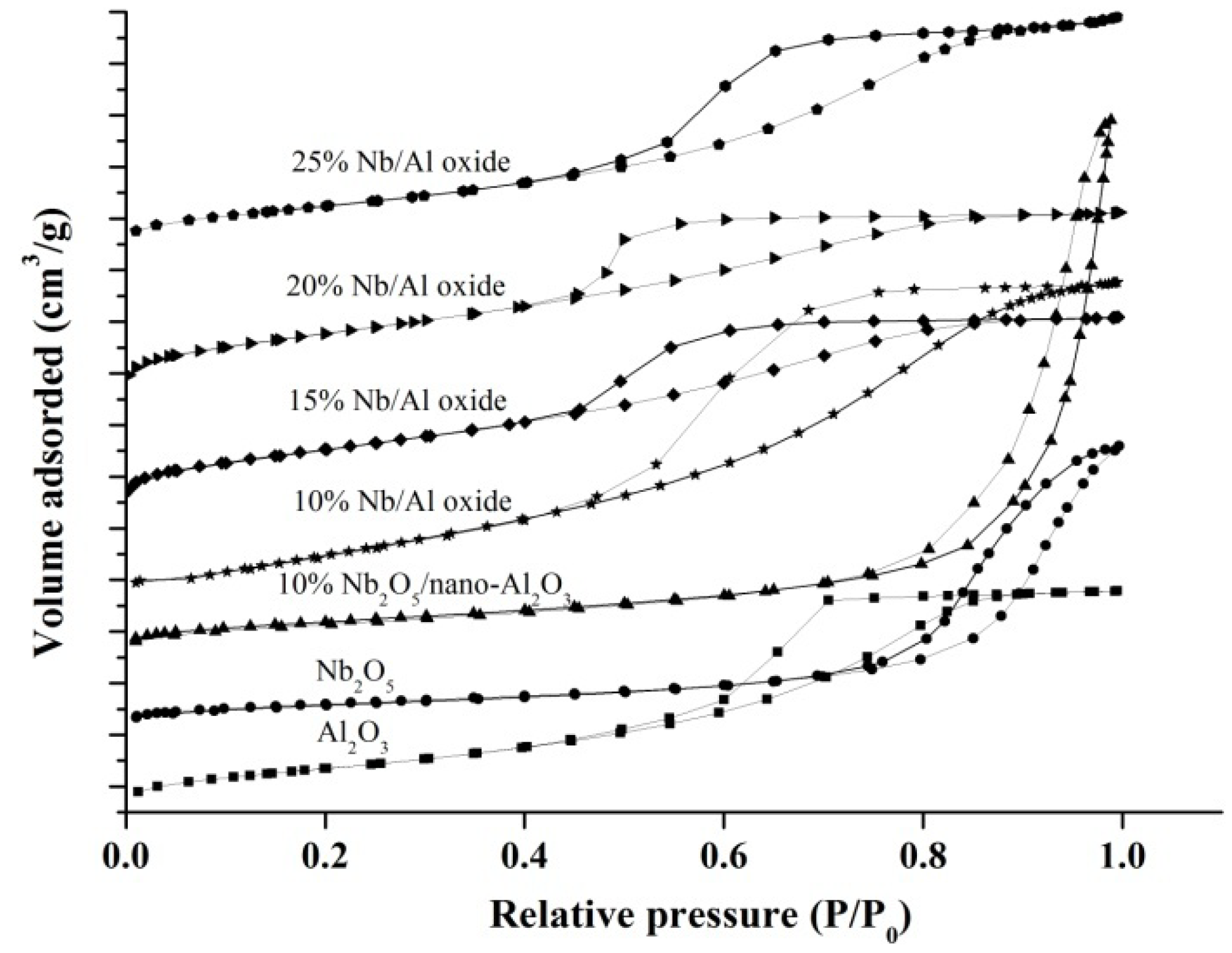

| Catalysts | SSA (m2/g) | AD(nm) | Surface Composition | Weak Acidity (%) | Moderate Acidity (%) | Strong Acidity (%) | Total Acidity (mmol/g) |
|---|---|---|---|---|---|---|---|
| 373–573 K | 573–773 K | >773 K | |||||
| 10% Nb/Al oxide | 296.55 | 3.97 | Nb0.036Al2.437O2.713 | 34.6 | 65.4 | 0 | 2.11 |
| 15% Nb/Al oxide | 288.98 | 4.20 | Nb0.045Al1.706O2.472 | 41.0 | 0 | 59.0 | 1.66 |
| 20% Nb/Al oxide | 281.47 | 4.49 | Nb0.077Al1.001O1.761 | 38.1 | 0 | 61.9 | 1.81 |
| 25% Nb/Al oxide | 264.84 | 5.45 | Nb0.081Al0.989O1.059 | 39.8 | 0 | 60.2 | 1.79 |
| 10%Nb2O5/nano-Al2O3 | 142.34 | 22.83 | - | 75 | 0 | 25 | 0.40 |
| Nb2O5 | 108.06 | 16.02 | - | 58.5 | 8.9 | 32.6 | 0.77 |
| Al2O3 | 207.77 | 5.69 | - | 98.8 | 0 | 1.2 | 0.83 |
2.2. Catalytic Reaction
2.2.1. Catalytic Conversion of Glucose and Fructose to LA with Different Metal Oxide Catalysts
2.2.2. Effect of Nb Loading of Nb/Al Oxides on Catalytic Conversion of Glucose and Fructose to LA
2.2.3. Effect of Reaction Temperature on Catalytic Conversion of Glucose and Fructose to LA with 20% Nb/Al Oxide
2.2.4. Effect of 20% Nb/Al Oxide Dosage on the Catalytic Conversion of Glucose and Fructose to LA
2.2.5. Effect of Reaction Time on Catalytic Conversion of Glucose and Fructose to LA with 20% Nb/Al Oxide
2.2.6. Catalytic Transformation of KWR into LA
| Reaction Condition | Glucose Conversion (%) | Fructose Conversion (%) | LA Concentration (mg/mL) |
|---|---|---|---|
| 10 wt. % of 20% Nb/Al oxide, 463 K, 10 min | 62.6 | 94.3 | 9.2 |
| 10 wt. % of 20% Nb/Al oxide, 463 K, 15 min | 67.3 | 97.3 | 9.8 |
| 10 wt. % of 20% Nb/Al oxide, 473 K, 10 min | 71.2 | 98.8 | 9.0 |
| 10 wt. % of 20% Nb/Al oxide, 473 K, 15 min | 76.2 | 99.3 | 8.5 |
3. Experimental Section
3.1. Materials
3.2. Glucose and Fructose Extraction from KWR
3.3. Catalyst Preparation
3.4. Catalyst Characterization
3.5. Catalyst Activity
3.6. Product Analysis
4. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bevilaqua, D.B.; Rambo, M.K.D.; Rizzetti, T.M.; Cardoso, A.L.; Martins, A.F. Cleaner production: Levulinic acid from rice husks. J. Clean Prod. 2013, 47, 96–101. [Google Scholar] [CrossRef]
- Uçkun Kıran, E.; Trzcinski, A.P.; Liu, Y. Platform chemical production from food wastes using a biorefinery concept. J. Chem. Technol. Biotechnol. 2015, 90, 1364–1379. [Google Scholar] [CrossRef]
- Jager, G.; Buchs, J. Biocatalytic conversion of lignocellulose to platform chemicals. Biotechnol. J. 2012, 7, 1122–1136. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Wu, L.P.; Wang, Y.; Song, Y.; Mourant, D.; Gunawan, R.; Gholizadeh, M.; Li, C.Z. Acid-catalyzed conversion of mono- and poly-sugars into platform chemicals: Effects of molecular structure of sugar substrate. Bioresour. Technol. 2013, 133, 469–474. [Google Scholar] [CrossRef] [PubMed]
- Yan, K.; Lafleura, T.; Wu, G.S.; Liao, J.Y.; Ceng, C.; Xie, X.M. Highly selective production of value-added γ-valerolactone frombiomass-derived levulinic acid using the robust Pd nanoparticles. Appl. Catal. A 2013, 468, 52–58. [Google Scholar]
- Yan, K.; Chen, A.C. Selective hydrogenation of furfural and levulinic acid to biofuelson the ecofriendly Cu-Fe catalyst. Fuel 2014, 115, 101–108. [Google Scholar] [CrossRef]
- Yan, K.; Lafleur, T.; Liao, J. Facile synthesis of palladium nanoparticles supportedon multi-walled carbon nanotube for efficient hydrogenationof biomass-derived levulinic acid. J. Nanopart. Res. 2013, 15, 1906–1913. [Google Scholar] [CrossRef]
- Pfaltzgraff, L.A.; de bruyn, M.; Cooper, E.C.; Budarin, V.; Clark, J.H. Food waste biomass: A resource for high-value chemicals. Green Chem. 2013, 15, 307–314. [Google Scholar]
- Lin, C.S.K.; Pfaltzgraff, L.A.; Herrero-Davila, L.; Mubofu, E.B.; Abderrahim, S.; Clark, J.H.; Koutinas, A.A.; Kopsahelis, N.; Stamatelatou, K.; Dickson, F. Food waste as a valuable resource for the production of chemicals, materials and fuels. Current situation and global perspective. Energy Environ. Sci. 2013, 6, 426–464. [Google Scholar] [CrossRef]
- Liu, Y.; Sotelo-Boyás, R.; Murata, K.; Minowa, T.; Sakanishi, K. Production of Bio-Hydrogenated Diesel by Hydrotreatment of High-Acid-Value Waste Cooking Oil over Ruthenium Catalyst Supported on Al-Polyoxocation-Pillared Montmorillonite. Catalysts 2012, 2, 171–190. [Google Scholar] [CrossRef]
- Kraithong, W.; Viriya-empikul, N.; Laosiripojana, N. Catalytic Conversion of Wastewater from Starch Industry to Levulinic Acid. Innov. Sys. Des. Eng. 2014, 5, 1–5. [Google Scholar]
- Li, K.; Bai, L.; Amaniampong, P.N.; Jia, X.; Lee, J.M.; Yang, Y. One-Pot Transformation of Cellobiose to Formic Acid and Levulinic Acid over Ionic-Liquid-based Polyoxometalate Hybrids. ChemSusChem 2014, 7, 2670–2677. [Google Scholar] [PubMed]
- Morales, G.; Osatiashtiani, A.; Hernández, B.; Iglesias, J.; Melero, J.A.; Paniagua, M.; Brown, D.R.; Granollers, M.; Lee, A.F.; Wilson, K. Conformal sulfated zirconia monolayer catalysts for the one-pot synthesis of ethyl levulinate from glucose. Chem. Commun. 2014, 50, 11742–11745. [Google Scholar] [CrossRef] [PubMed]
- Ramli, N.A.S.; Amin, N.A.S. Catalytic hydrolysis of cellulose and oil palm biomass in ionic liquid to reducing sugar for levulinic acid production. Fuel Process. Technol. 2014, 128, 490–498. [Google Scholar] [CrossRef]
- Saravanamurugan, S.; Riisager, A. Zeolite catalyzed transformation of carbohydrates to alkyl levulinates. ChemCatChem 2013, 5, 1754–1757. [Google Scholar] [CrossRef]
- Upare, P.P.; Yoon, J.W.; Kim, M.Y.; Kang, H.Y.; Hwang, D.W.; Hwang, Y.K.; Kung, H.H.; Chang, J.S. Chemical conversion of biomass-derived hexose sugars to levulinic acid over sulfonic acid-functionalized graphene oxide catalysts. Green Chem. 2013, 15, 2935–2943. [Google Scholar] [CrossRef]
- Ramli, N.A.S.; Amin, N.A.S. Fe/HY zeolite as an effective catalyst for levulinic acid production from glucose: Characterization and catalytic performance. Appl. Catal. B 2015, 163, 487–498. [Google Scholar] [CrossRef]
- Hu, X.; Wang, S.; Westerhof, R.J.; Wu, L.; Song, Y.; Dong, D.; Li, C.Z. Acid-catalyzed conversion of C6 sugar monomer/oligomers to levulinic acid in water, tetrahydrofuran and toluene: Importance of the solvent polarity. Fuel 2015, 141, 56–63. [Google Scholar] [CrossRef]
- Yang, F.; Liu, Q.; Bai, X.; Du, Y. Conversion of biomass into 5-hydroxymethylfurfural using solid acid catalyst. Bioresour. Technol. 2011, 3, 3424–3429. [Google Scholar] [CrossRef] [PubMed]
- Tanabe, K.; Okazaki, S. Various reactions catalyzed by niobium compounds and materials. Appl. Catal. A 1995, 133, 191–218. [Google Scholar] [CrossRef]
- Armaroli, T.; Busca, G.; Carlini, C.; Giuttari, M.; Raspolli Galletti, A.M.; Sbrana, G. Acid sites characterization of niobium phosphate catalysts and their activity in fructose dehydration to 5-hydroxymethyl-2-furaldehyde. J. Mol. Catal. A 2000, 151, 233–243. [Google Scholar] [CrossRef]
- Nakajima, K.; Fukui, T.; Kato, H.; Kitano, M.; Kondo, J.N.; Hayashi, S.; Hara, M. Structure and Acid Catalysis of Mesoporous Nb2O5·nH2O. Chem. Mater. 2010, 22, 3332–3339. [Google Scholar] [CrossRef]
- García-Sancho, C.; Rubio-Caballero, J.M.; Mérida-Robles, J.M.; Moreno-Tost, R.; Santamaría-González, J.; Maireles-Torres, P. Mesoporous Nb2O5 as solid acid catalyst for dehydration of dxylose into furfural. Catal. Today 2014, 234, 119–124. [Google Scholar] [CrossRef]
- García-Sancho, C.; Agirrezabal-Telleria, I.; Güemez, M.B.; Maireles-Torres, P. Dehydration of dxylose to furfural using different supported niobia catalysts. Appl. Catal. B 2014, 152–153, 1–10. [Google Scholar] [CrossRef]
- Esti, M.; Messia, M.C.; Bertocchi, P.; Sinesio, F.; Moneta, E.; Nicotra, A.; Fantechi, P.; Palleschi, G. Chemical compounds and sensory assessment of kiwifruit (Actinidia chinensis (Planch.) var. chinensis): Electrochemical and multivariate analyses. Food Chem. 1998, 61, 293–300. [Google Scholar] [CrossRef]
- Zhu, H.Y.; Riches, J.D.; Barry, J.C. γ-Alumina Nanofibers Prepared from Aluminum Hydrate with Poly(ethylene oxide) Surfactant. Chem. Mater. 2002, 14, 2086–2093. [Google Scholar] [CrossRef]
- Emeis, C.A. Determination of Integrated Molar Extinction Coefficients for Infrared Absorption Bands of Pyridine Adsorbed on Solid Acid Catalysts. J. Catal. 1993, 141, 347–354. [Google Scholar] [CrossRef]
- Ya’aini, N.; Amin, N.A.S.; Asmadi, M. Optimization of levulinic acid from lignocellulosic biomass using a new hybrid catalyst. Bioresour. Technol. 2012, 116, 58–65. [Google Scholar] [CrossRef] [PubMed]
- Qi, L.; Mui, Y.F.; Lo, S.W.; Lui, M.Y.; Akien, G.R.; Horváth, I.T. Catalytic Conversion of Fructose, Glucose, and Sucrose to 5-(Hydroxymethyl)furfural and Levulinic and Formic Acids in γ-Valerolactone as a Green Solvent. ACS Catal. 2014, 4, 1470–1477. [Google Scholar] [CrossRef]
- Shen, Y.; Xu, Y.; Sun, J.; Wang, B.; Xu, F.; Sun, R. Efficient conversion of monosaccharides into 5-hydroxymethylfurfural and levulinic acid in InCl3-H2O medium. Catal. Commun. 2014, 50, 17–20. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, R.; Xie, X.; Liu, Y.; Liu, Z.; Xie, G.; Ji, N.; Ma, L.; Tang, M. Facile and Low-Cost Preparation of Nb/Al Oxide Catalyst with High Performance for the Conversion of Kiwifruit Waste Residue to Levulinic Acid. Catalysts 2015, 5, 1636-1648. https://doi.org/10.3390/catal5041636
Wang R, Xie X, Liu Y, Liu Z, Xie G, Ji N, Ma L, Tang M. Facile and Low-Cost Preparation of Nb/Al Oxide Catalyst with High Performance for the Conversion of Kiwifruit Waste Residue to Levulinic Acid. Catalysts. 2015; 5(4):1636-1648. https://doi.org/10.3390/catal5041636
Chicago/Turabian StyleWang, Rui, Xiaolin Xie, Yanxiu Liu, Zhigang Liu, Guofang Xie, Ning Ji, Lizhi Ma, and Mingquan Tang. 2015. "Facile and Low-Cost Preparation of Nb/Al Oxide Catalyst with High Performance for the Conversion of Kiwifruit Waste Residue to Levulinic Acid" Catalysts 5, no. 4: 1636-1648. https://doi.org/10.3390/catal5041636
APA StyleWang, R., Xie, X., Liu, Y., Liu, Z., Xie, G., Ji, N., Ma, L., & Tang, M. (2015). Facile and Low-Cost Preparation of Nb/Al Oxide Catalyst with High Performance for the Conversion of Kiwifruit Waste Residue to Levulinic Acid. Catalysts, 5(4), 1636-1648. https://doi.org/10.3390/catal5041636






